Clinacanthusnutans (CN) plants have been received much interest from phytochemical researchers because of their plentiful bioactive compounds of total phenolic compound (TPC) and total flavonoid compound (TFC). Many of report study exhibit wide varieties of biological activities. In this study, herbal ethyl acetate extracts was isolated from C. nutans (CN) leaf, identified by Thin layer Chromatography. Phytochemical screening illustrated to the tannins compound. Crude extracted was freshly prepared by solubilizing in DMSO and immediately diluted in Müller-Hinton broth for antibacterial testing. The data showed that Staphylococcus aureus and Methicillin resistant Staphylococcus aureus (MRSA) of three strains such as ATCC 29213, MRSA 4738 and MRSA 20649 were susceptible to crude extracts in disc diffusion method.
## I. INTRODUCTION
Currently, the numbers of nosocomial infections have increased continuously. Staphylococcus aureus is a major problem in nosocomial infections disease such as pneumonia, operative wound infections and sepsis [1]. S. aureus was caused infections include skin lesions such as boils, furuncles and more serious infections, for example, phlebitis, meningitis, endocarditis and urinary tract infections. The mortality rate for nosocomial endocarditis is found higher than that for urinary tract infection when the pathogen is S. aureus [2]. The lesion of staphylococcal infection is the abscess, which consists of a fibrin wall surrounded by inflamed tissues enclosing a central core of pus containing organisms and leukocytes. The organisms may be disseminated hematogenously, even from the smallest abscess. S. aureus has a tendency to spread to particular sites, including the bones, joints, kidneys, and lungs. This may result in virulent sepsis. The presentation of staphylococcal sepsis is similar to that of gram-negative sepsis, with fever, hypotension, tachycardia, and tachypnea. Severe cases progress to many organs dysfunction, lactic acidosis and death.
Many strains of S. aureus are developing resistance to available antibacterial agents, creating a serious problem in public health such as methicillin-resistant S.aureus (MRSA). The organism may acquire genes encoding enzymes, for example $\beta$ -lactamase that destroys the antibacterial agent before it can have an effect. For these problems, searching and development of novel antibacterial compounds are urgently required.
Clinacanthus nutans are well known and interesting sources for new antibacterial agents. It is usually slithered along other trees, about 1-3 meters tall, The trunk is round, the skin is smooth and green. Propagate with the method of cutting or separating the rhizomes to plant. Grows well in all kinds of soils. There are distribution zones in China, Vietnam, Indonesia, Malaysia and Thailand. In Thailand, it is commonly found in mixed forests in all parts of the country, or grown in common houses. The appearance of the leaves is spear-shaped, oval, narrow, parallel edges. The leaves are dark green and smooth, the weavers bloom at the ends of the branches, each inflorescences have 3-6 flowers, the petals are red-orange, the petals are welded together into tubes. The tip is divided into 2 mouths, the lower and upper mouths have 5 petals, the petals are cylindrical, and the petals are green, equally long, with sticky glands around them. The flowers contain 2 male pollen, while the female pollen is hairless, flowering from around October to January (but rarely flowering). They are ubiquitous in antibacterial compounds and are commonly found in traditional medicine. These compounds have been used in traditional herbal medicine as the principal physiologically active constituents to treat human diseases for centuries. In addition, this class of natural products is becoming the subject of antimicrobial research. Many groups of pure compound possessing antiviral, antifungal or antibacterial activities have been isolated and identified for the structure [3].
Clinacanthus plants have been received much interest from phytochemical researchers because of their plentiful bioactive compounds of flavonoids, betulin, lupeol, n-butanol and glycoglycerolipids. Many of pure compounds exhibit wide varieties of biological activities. For example, botulin and lupeol have been reported to possess anti-action and anti-inflammatory activity [4]. There are several lines of evidence demonstrating its antimicrobial potential, including antiviral, antibacterial and antimycobacterial activities. Several studies have demonstrated the mechanisms of action underlying antimicrobial effects of flavonoids extracted from medicinal plants. Because of a variety of the structures in this phytochemical class, the mechanism of action previously established by researchers varies dramatically. Antimicrobial activities of the plant has been demonstrated, however its mechanism of action has never been documented. Therefore, in the present study, bioactive compounds will be purified from C. nutans leaves. Then screening test for antibacterial activities of crude extract against MRSA will be performed. Bacteria which are susceptible to crude extract will be used to investigate its mechanism of action. From the preliminary data, C.
nutans crude extract showed best activity against the Gram-positive bacteria S. aureus. Moreover, the pilot study also revealed that the extract caused damage of bacterial cell wall and/or cell membrane. The data obtained from this study will provide scientific evidence to support the use or development of this compound as antimicrobial agent.
## II. SUBJECTS AND METHODS
### a) Chemicals
Hexane, ethanol, methanol, dichrolomethane, ethyl acetate, NaCl, Agar, Mueller-Hinton, 0.5 McFarland, $\mathrm{CDCl}_3$, glutaraldehyde, acetone and dimethyl sulfoxide were purchased from Carlo Erba (Italy). Vancomycin was purchased from Sigma-Aldrich (USA).
### b) Plant Material and Herbal Crude Extraction
Clinacanthus nutans was collected from Phetchabun province, Thailand. Botanical identification was performed by Dr. Surangrut Punsang, Program of Biology, Phetchabun Rajabhat university (PCRU). A voucher specimen (PCRU-CN-001) was deposited at Program of Public Health, PCRU. The leaves was air dried at room temperature. The leaves was stored at room temperature until used for extraction.
100 grams of dried leaves was extracted with $500\mathrm{ml}$ ethyl acetate, using maceration for 7 days. The total extracts were filtered with Whatman No.1. Then the extracts were rotary evaporated and freeze dry. Thin layer chromatography (TLC) was used to confirm purity of the extract. Crude extract obtained was dissolved in ethanol and $5\mu l$ of the solution was submitted to TLC on silica gel $\mathsf{G}_{60}\mathsf{F}_{254}$ aluminium plates. Dichrolomethane: Methanol (95:5) was used as the eluent. Spot was detected by UV light at 254 to confirm the purification.
### c) Determination of Constituents of Crude Extract by Phytochemical Screening
Phytochemical screening was carried out to identify constituents of C. nutans extract. The extract was screened for terpenoids, flavonoids, saponins, tannins and cardiac glycosides. Phytochemical screening was performed based on previously reported methods [5].
### d) Antibacterial Assays
## i. Disc Diffusion
Bacteria used in this study was obtained from Department of Medical Sciences Thailand (DMST). The antibacterial activities of ethyl acetate crude extract from C. nutans was evaluated against Staphylococcus aureus ATTC 29213, Staphylococcus aureus (MRSA) DMST 20649, Staphylococcus aureus (MRSA) DMST 4738. The screening of the antibacterial activity was done using disc diffusion method. Bacterial suspensions was prepared by inoculating one loopful of a pure colony into Mueller-Hinton Broth (MHB), incubated overnight and diluted in $0.85\%$ NaCl. Cell suspensions, of which adjusted turbidity equivalent to that of a 0.5 McFarland standard, contains about $10^{8}$ cfu/ml. These was used to inoculate on Mueller-Hinton Agar (MHA) plates by swabbing over the entire agar surface. The CN crude extract (25, 50, 75 $\mu$ g/disc) was impregnated to filter paper discs (Whatman No.1, 6 mm diameter) and then placed on the previously inoculated agar plate. After 24 h of incubation at $37^{\circ}\mathrm{C}$, the antibacterial activity was determined by measuring the diameter of the inhibition zones formed around the disc. Vancomycin and DMSO was used as a positive and vehicle controls, respectively[6].
## ii. Determination of Minimum Inhibitory Concentration (MIC) and Minimum Bactericidal Concentration (MBC)
A modified broth microdilution method according to Clinical and Laboratory Standard Institute Guidelines [7] was used to determine MIC and MBC of the extract. It was dissolved in DMSO and two-fold serial dilutions will be made in Mueller-Hinton broth (MHB) using 96-well flat bottom microtiter plate (Corning Life Sciences, USA). Suspension of bacteria in MHB was prepared from the overnight broth culture. The final bacterial cell concentration was adjusted to $5 \times 10^{5}$ cfu/ml. The final concentration of the CN extract was ranged from $0.25 - 512 \mu \mathrm{g} / \mathrm{ml}$. Vancomycin and DMSO was used as positive and negative controls, respectively. The MIC was considered as the lowest concentration of the agents showing no visible growth of microorganism after incubation at $37^{\circ} \mathrm{C}$ for $24 \mathrm{~h}$. The MBC determination was carried out by subculturing 20
$\mu l$ from the broth with no growth onto Mueller-Hinton Agar (MHA) plates followed by incubation for $24h$ at $37^{\circ}C$. The lowest concentration with no visible growth was taken as the MBC. All tests was performed in triplicate independent experiments.
### e) Transmission Electron Microscopy (TEM)
Transmission electron microscopy (TEM) was used to visualize the change in morphology at the membrane and cell wall ultrastructure of Methicillin resistant S. aureus after treatment with the CN extract. TEM preparations were made in accordance with the previously reported method with slight modifications [8].
The bacterial samples were prepared similar to the SEM method. After the extract treatment for $12\mathrm{h}$, cells were gently washed with $0.1\mathrm{M}$ PBS $(\mathsf{pH}7.2)$, fixed with $2.5\%$ glutaraldehyde in PBS and rinsed with PBS. Post-fixation was then carried out with $1\%$ osmium tetroxide (Electron Microscopy Sciences: Hatfield, PA, USA) in $0.1\mathrm{M}$ PBS for $2\mathrm{h}$ at room temperature. After washing in the buffer, the samples were dehydrated using sequential exposure for acetone concentrations ranging from 20 to $100\%$. Subsequently, infiltration and embedding were performed using Spurr's resin (EMS). Finally, the samples were sectioned using an ultramicrotome with a diamond knife and were mounted on copper grids. They were stained with $2\%$ uranyl acetate and lead citrate. The samples were viewed with a JEM-1230 electron microscope (Tokyo, Japan). The morphology of bacterial cells was observed and compared to vancomycin-treated cells as positive control.
## III. RESULTS
### a) Phytochemical Screening
The result showed that the CN extract exposed tannins compound (Table 1).
Table 1: Phytochemicals screening of ethyl acetate extract from C. nutans leaves
<table><tr><td>Compounds</td><td>CN extract</td></tr><tr><td>Flavonoid</td><td>-</td></tr><tr><td>Saponins</td><td>-</td></tr><tr><td>Cardiac glycoside</td><td>-</td></tr><tr><td>Tannins</td><td>+</td></tr><tr><td>Terpenoids</td><td>-</td></tr></table>
### b) Thin Layer Chromatography (TLC)
The results of the active compound extraction of C. nutans leaves are shown a dark green viscous after maceration. Thin layer chromatography (TLC) used to confirm purity of the extract. Dark green viscous obtained dissolved in ethanol and submitted to TLC on silica gel $\mathrm{G}_{60} \mathrm{~F}_{254}$ aluminium plates that shown in Figure 1. Dichrolomethane:Methanol (95:5) used as the eluent.
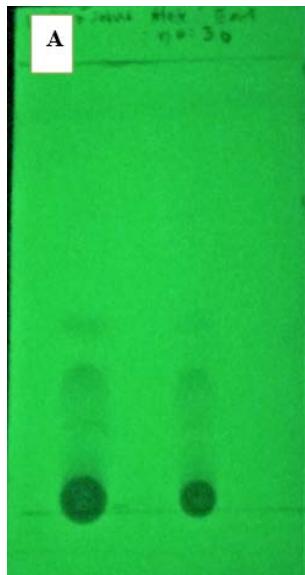 Figure 1: TLC of the CN extract was detected by UV light at $254~\mathrm{nm}$
### c) Antibacterial Activity
By disc diffusion assay, it was found that the CN crude extract can inhibit growth of Staphylococcus aureus ATTC 29213, Staphylococcus aureus (MRSA)
DMST 20649, Staphylococcus aureus (MRSA) DMST 4738 at 25, 50 and $75\mu \mathrm{g}$, respectively (Table 2). Whereas, Vancomycin was used as a positive control.
Table 2: Antibacterial activity of the CN crude extract using Disc diffusion
<table><tr><td rowspan="3">Microorganism</td><td colspan="3">Diameter of inhibitions zone (mm)</td><td rowspan="2">Vancomycin (μg)</td></tr><tr><td colspan="3">CN (μg/disc)</td></tr><tr><td>25</td><td>50</td><td>75</td><td>30</td></tr><tr><td>Staphylococcus aureus 29213</td><td>12 ± 0.2</td><td>20 ± 0.6</td><td>25 ± 0.2</td><td>37 ± 0.5</td></tr><tr><td>Staphylococcus aureus (MRSA) 4738</td><td>10± 0.5</td><td>23 ± 0.6</td><td>25 ± 0.3</td><td>24 ± 0.7</td></tr><tr><td>Staphylococcus aureus (MRSA) 20649</td><td>10 ± 0.1</td><td>23 ± 0.2</td><td>26 ± 0.5</td><td>28 ± 0.7</td></tr><tr><td>Data are means ± SD (n=3)</td><td></td><td></td><td></td><td></td></tr></table>
The MIC and MBC values of the CN crude extract obtained using the microdilution method as shown in Table 3. The active compound of C. nutans leaves gave MIC and MBC values at 8 and $16\mu \mathrm{g / ml}$ against Staphylococcus aureus 29213, respectively. In addition, the antibacterial activity showed MIC and MBC values at 3.125 and $6.25\mu \mathrm{g / ml}$ against Staphylococcus aureus (MRSA) 4738 and Staphylococcus aureus (MRSA) 20649 when compared with control cell after 24 h of incubation at $37^{\circ}C.$ The assay were carried out in triplicate.
Table 3: The minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) of the CN crude extract against MRSA compared with vancomycin
<table><tr><td rowspan="2">Microorganisms</td><td colspan="2">CN</td><td colspan="2">Vancomycin</td></tr><tr><td>MIC (mg /ml)</td><td>MBC (mg /ml)</td><td>MIC (μg/ml)</td><td>MBC (μg/ml)</td></tr><tr><td>Staphylococcus aureus 29213</td><td>8</td><td>16</td><td>4</td><td>8</td></tr><tr><td>Staphylococcus aureus (MRSA) 4738</td><td>3.125</td><td>6.25</td><td>4</td><td>8</td></tr><tr><td>Staphylococcus aureus (MRSA) 20649</td><td>3.125</td><td>6.25</td><td>4</td><td>8</td></tr></table>
### d) Transmission Electron Microscopy (TEM)
TEM analysis was conducted and the data showed that after incubation with MIC for $12\mathrm{hr}$, the CN crude extract, as well as vancomycin, obviously ruptured bacterial cell membrane and/or cell wall (Figure 2). Cell death and irregular shape of bacterial cells were seen in the treated groups, the CN extract and vancomycin. Damage of cell wall and cell membrane of dividing cells were observed after $12\mathrm{h}$ of incubation with the CN extract and vancomycin, compared with control.
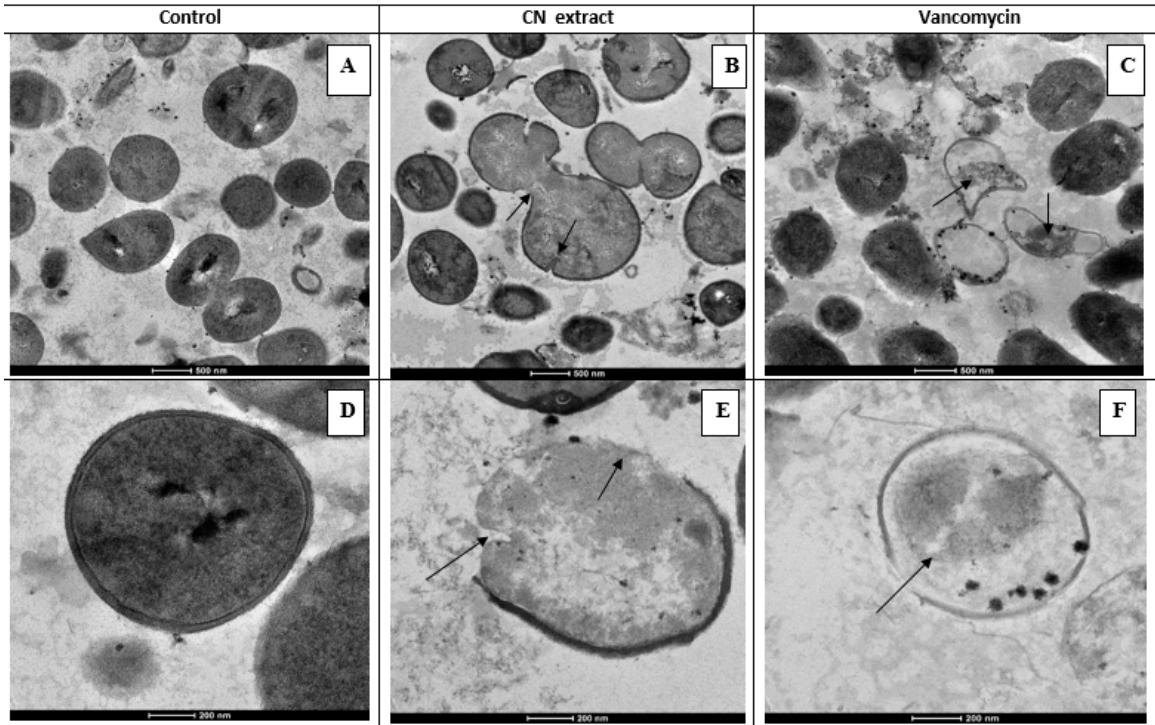 Figure 2: Transmission electron micrograph of MRSA treated with CN extract. A, B and C are overview of control and cells treated with CN extract and vancomycin respectively. Cell death and irregular shape of bacterial cells were seen in the treated groups, CN extract (B) and vancomycin (C). Damage of cell wall and cell membrane of dividing cells (indicated by arrows) were observed after $12\mathrm{h}$ of incubation with CN extract (E) and vancomycin (F), compared with control (D). Enlargement: bar = 200 nm, and 500 nm
## IV. DISCUSSION
According to this research, when extracting substances from the leaves of C. nutans, it was found that the extract was coarse. It has a sticky, dark green, pungent and sour odor, which can be an important characteristic of various substances, including flavonoids, which have anti-inflammatory effects, monoglycosyl diglycerides such as 1,2-O-dilinolenoyl-3-O-b-d-glucopyranosyl-sn-glycerol, and glycoglycerolipids, which inhibit the herpes simplex virus [9].
In this study, methicillin resistant Staphylococcus aureus (MRSA) was tested for antimethicillin resistant Staphylococcus aureus (MRSA) with Minimal inhibitory concentration (MIC) and Minimal bactericidal concentration (MBC). The results showed that coarse extracts can inhibit bacterial growth by causing inhibition zones on MHB compared to vancomycin, with statistically significant differences $(p < 0.05)$ as shown in Table 2. Antibacterial were identified by using MIC and MBC. The CN extract showed MIC of $3.125\mu \mathrm{g / ml}$ and MBC of $6.25\mu \mathrm{g / ml}$ against MRSA (Table 3). In addition, the researchers investigated whether all three strains were MRSA based on MIC values of ampicillin found to be greater than 512 $\mu$ g/ml (data not shown). To investigate the mechanism of action, Transmission electron microscope (TEM), a powerful technique was applied to determine the physiological changes in bacterial cells. TEM is an technique with the change in morphology of extracellular and intracellular bacteria as shown in Figure 2. Moreover, the extract caused membrane bleb, ruffling or detachment, hypodense cytoplasmic release and cell vacuolization compared to control. After damaging the cell membrane, which acts as a barrier for most molecules, bacteria degrade the cell's permeability control, resulting in an increase in intracellular pressure and subsequently destruction of the cell wall [10,16].
It is widely known that total phenolic compounds in plants comprise several groups of phytochemicals such as tannins, terpenoids and flavonoids. Over the years, C. nutans have been reported to produce numerous betulin, lupeol, $\beta$ -sitosterol, flavonoids and glycoglycerolipids compounds[11]. It have been found to possess significant antimicrobial, antiviral, and anti-inflammatory in mouse. The leaves of C. nutans are rich in natural antibacterial total phenolic compound such as tannins. In the present study, phytochemical screening was identified to flavonoids, saponins, cardiacglycosides, tannins and triterpenoids. It was found that the CN crude extracts exhibited tannins compound (Table 1). In general, tannins are found naturally in various parts of plants. The main chemical properties are antibacterial, antioxidant, anticancer and anti-inflammatory activity [12].
Tannin, sometimes called tannic acid, is a compound derived from phenolic acids and belongs to a group of compounds called polyphenols. Other compounds in the polyphenols group also have the ability to adhere to other molecules such as proteins, cellulose, starch and minerals easily[13], making tannins more insoluble and more resistant to decomposition than other compounds in the same group. As a matter of time, tannins can be found naturally. In different parts of the plant, both edible and inedible parts, such as root trees and bark of plants produce tannins to help protect yourself from natural pests. Foods containing tannins are most commonly found in tea, coffee, chocolate and wine. Tannins are substances that belong to the group of polyphenol compounds. Chemicals with the main properties are antioxidants[14,15]. It helps prevent inflammation and may help against cancer. Eating foods high in tannins can help fight free radicals, one of the major problems that contribute to cell degeneration in the body and can lead to cancer. Antioxidants like tannins can also help prevent and reduce inflammation. This makes the wound regenerate more quickly. In conclusion, one recent study found that C.nutans have antimicrobial properties, these medicinal plants can be a source for isolation of anti-MRSA compounds.
### ACKNOWLEDGEMENT
This research project was funded by the National Research Council of Thailand (Grant No: PCRU-2562-KN031).
Generating HTML Viewer...
References
16 Cites in Article
Fd Lowy (1998). Staphylococcus aureus infections.
C Ml Fernandez-Guerrero,J Verdejo,M Azofra,Gorgolas De (1995). Hospital-acquired infectious endocarditis not associated with cardiac surgery: an emerging problem.
Willow Liu (2011). Introduction to Traditional Herbal Medicines and their Study.
Santi Sakdarat,Aussavashai Shuyprom,Chamsai Pientong,Tipaya Ekalaksananan,Sasithorn Thongchai (2009). Bioactive constituents from the leaves of Clinacanthus nutans Lindau.
Pakarang Kumkrai,Sirilak Kamonwannasit,Nuannoi Chudapongse (2014). Cytoprotective and anti-diabetic effects of Derris reticulata aqueous extract.
Kamol Yusook,Pettaya Panvongsa (2017). Antibacterial Activity of Lupinifolin from Derris reticulata and Its Effect on Cytoplasmic Membrane of Methicillin Resistant.
(2012). An Overview of the Clinical and Laboratory Standards Institute (CLSI) and Its Impact on Antimicrobial Susceptibility Tests.
Sudipta Ghosh,Kiran Indukuri,Somasekhar Bondalapati,Anil Saikia,Latha Rangan (2013). Unveiling the mode of action of antibacterial labdane diterpenes from Alpinia nigra (Gaertn.) B. L. Burtt seeds.
Herbs used in basic public health.
K Yusook,O Weeranantanapan,Y Hua,P Kumkrai,N Chudapongse (2017). Lupinifolin from Derris reticulata possesses bactericidal activity on Staphylococcus aureus by disrupting bacterial cell membrane.
T Cushnie,Andrew Lamb (2005). Errata for “Antimicrobial activity of flavonoids” [Int. J. Antimicrob. Agents 26 (2005) 343–356].
Chulabhorn Mahidol,Hunsa Prawat,Vilailak Prachyawarakorn,Somsak Ruchirawat (2002). Investigation of some bioactive Thai medicinal plants.
Nantiya Joycharat,Sonesay Thammavong,Surasak Limsuwan,Sirilux Homlaead,Supayang Voravuthikunchai,Boon-Ek Yingyongnarongkul,Sukanya Dej-Adisai,Sanan Subhadhirasakul (2013). Antibacterial substances from Albizia myriophylla wood against cariogenic Streptococcus mutans.
Nisa Humeera,Azra Kamili,Suhaib Bandh,Shajr-Ul- Amin,Bashir Lone,Nisa Gousia (2013). Antimicrobial and antioxidant activities of alcoholic extracts of Rumex dentatus L..
H Mahidol,V Prawat,S Prachyawarakorn,Ruchirawat (2002). Investigation of some bioactive Thai medicinal plants.
Poonam Tyagi,Madhuri Singh,Himani Kumari,Anita Kumari,Kasturi Mukhopadhyay (2015). Bactericidal Activity of Curcumin I Is Associated with Damaging of Bacterial Membrane.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Kamol Yusook. 2026. \u201cThe Antibacterial Activity against Methicillin-Resistant Staphylococcus Aureus from the Ethyl Acetate Extract in Clinacanthusnutans\u201d. Global Journal of Science Frontier Research - B: Chemistry GJSFR-B Volume 22 (GJSFR Volume 22 Issue B1).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.