Detailed male meiosis has been made in one species of genus Withania collected from Ujjain region of Madhya Pradesh, India. The study revealed 2n=48 for the species. In addition to this, some cells showed the occurrence of various meiotic irregularities for example, lagging chromosomes (1.76%), chromatin bridges (1.76%) and cytomixis (0.87%). The fertility of pollen grains was also determined which was observed 97.52%. The species are widely used for various medicinal purposes by local/tribal people of the state.
## I. INTRODUCTION
The genus Withania belongs to the family Solanaceae. The family comprises of 100 genera and about 2500 species (Hunziker 2001, Olmstead et al. 2008). The genus is distributed throughout the tropical and sub-tropical regions of the world with 26 species (Ahmad 2014). W. somnifera is well distributed in India with and is growing well in dry parts of tropical and subtropical regions extending to the elevation of $1500\mathrm{m}$. The plant possesses antitumor, antimicrobial and anti-inflammatory properties. Since the plant is toxic in nature so it should be used with caution (Purohit and Yyas 2004). Flowers of its species are used to treat nervous exhaustion, insomnia and impotence. The meiotic course of the species reveals the presence of $2n = 48$ from West Pakistan (Baquar 1967) as well as outside India. W. somnifera commonly known as "Ashwangandha" or "Indian ginseng" is extensively studied from India by Bir et al. (1978) Bir and Sidhu (1979 and 1980) from Punjab plains, Koul et al. (1976) from Jammu and Kashmir, Madhavadian (1968) from Tamil Nadu, Bhaduri (1933), Datta et al. (2005), Iqbal and Datta (2007) from West Bengal. It is also extensively worked out from Pakistan by Baquar (1967) and Khatton and Ali (1982) and from Saudi Arabia by Al-Turki et al. (2000). Earlier meiotic studies reveals the presence of intraspecific diploid cytotype $(2n = 24)$, tetraploid cytotype $(2n = 48)$ and hexaploid cytotypes $(2n = 72)$. The karyotype analysis of the species shows seven groups of the chromosomes with occurrence of metacentric and sub-metacentric types (Samaddar et al. 2012). The species also shows polysomatomy $(2n = 12,18,24,36,48,72)$ with predominance of $2n = 48$.
Present research work is undertaken by keeping in view the existence of cytological diversity.
## II. MATERIALS AND METHODS
### a) Collection of plants materials
Species was collected from Ujjain city especially in the premises of Vikram University Ujjain. The photographs of the collected plants species were preferably taken from their natural habitats. The photography was done during the complete flowering seasons. For identification of plants species, different floras were consulted during the present study such as Flora of Madhya Pradesh Western Part by Singh (2012), various internet sources. The plant is an important medicinal plant of the Indian subcontinent. In additions to this, the plant specimens were taken to some taxonomists for further documentation. The identified plants specimens have been reposted in School of Studies in Botany, Vikram University, Ujjain (India).
### b) Fixation and preservation of plant materials
Floral buds were fixed in freshly prepared Carnoy's fixative (6 parts of absolute alcohol: 3 parts of chloroform: 1 part of glacial acetic acid) for 24-48 hours. Afterwards, these were transferred to $70\%$ ethyl alcohol and stored in refrigerator at $4^{\circ}\mathrm{C}$ until use. For chromosomal preparations, anthers were crushed and tapped to prepare a smear of pollen mother cells (PMCs) in $1\%$ acetocarmine (Belling 1921). A number of PMCs were observed and chromosome counts were confirmed. In case of species with meiotic abnormalities, large numbers of PMCs are observed to confirm frequency of various abnormalities. Pollen fertility was observed by mounting the pollen grains in $50\%$ glycerol-aceto carmine (1:1) solution (Marks 1954). Pollen grains with stained nuclei were taken as fertile and viable, whereas, unstained pollen grains marked as sterile ones.
### c) Photomicrography
Photomicrographs were taken from freshly prepared temporary slides. The photographs were taken with the aid of the microscope Camera (Digital Eyepiece) on a field of $40\%$ objective lens and 10X eyepiece of Olympus microscope.
## III. RESULTS
### a) Taxonomical and morphological studies
Kingdom: Plantae (Plants);
Sub-kingdom: Tracheobionta (Vascular plants);
Super division: Spermatophyta (Seeds plants);
Division: Angiosperma
Class: Dicotyledons
Order: Tubiflorae
Family: Solanaceae
Genus: Withania
Species: Somnifera Dunal
The plant is usually erect, green, branched/unbranched herb, up to $1.25 \mathrm{~m}$ in height. The aerial part, especially the stem, leaves and calyx are sparsely covered with fine hairy tomentum. Branches are round; leaves simple, petiolated, ovate, entire, shiny, smooth and opposite; flowers inconspicuous, greenish or yellow, in axillary umbellate cymes, bisexual; fruit is berry in a persistent calyx; seeds are small, flat, yellow, reniform, very light. In this species, the period of flowering and fruiting occurred during the months from August to October. The plant has tap root system having $15 - 25 \mathrm{~cm}$ in length and light yellow in colour. The branches are erect and are of about 60-120 cm in length. It requires dry weather conditions for development of better root quality and alkaloid content.
Male meiotic studies In this plant species, meiotic characterization of anthers revealed regular 24 bivalents $(2n = 48)$ in cells at diakinesis as well as metaphase I. At anaphase I, the chromosomes successively demonstrated normal segregation of 24:24 chromosomes towards opposite poles (Figs. A-I). In addition to this, some cells showed the occurrence of various meiotic irregularities (Figs. A-I; Table-1-3), for example, lagging chromosomes $(1.76\%)$, chromatin bridges $(1.76\%)$ and cytomixis $(0.87\%)$. The incidence of chromosome association in the form of ring and rod bivalents per PMC ranged from 13 to 22 (mean $= 17.2$ and 2 to 11 (mean $= 6.8$ ), respectively. The frequency of chiasma per cells, on an average, was observed 45.8 $(SD = \pm 3.25)$ in which 41.20 chiasma $(SD = \pm 3.12)$ were found terminalized and 4.60 $(SD = \pm 0.80)$ were identified as untereminalized. As a result, the terminalization coefficient was calculated as 0.90 (Table-8). The fertility of pollen grains was also determined which was observed $97.52\%$.
## IV. DISCUSSION AND CONCLUSIONS
Meiosis is most sensitive stage in the life cycle for all sexual species and has direct relevance to natural selection; it leads to the formation of gametes, contributes to genome stability and generates genetic diversity. The process of meiosis depends upon interrelated events of homologous chromosome recognition, intimate association, synapsis and recombination (Hamant et al. 2006, de Muyt et al. 2009). In plants, it is affected by various genetic and environmental factors (Ahmad et al. 1984, Viccini and Carvalho, 2002, Sun et al. 2004, Bajpai and Singh 2006, Rezaei et al. 2010). There are various meiotic abnormalities which hinder the path of normal meiosis and are the cause of changes in the morphology and genetic constitutions of the plant. The evolution of vascular plants is dependent upon the variation in chromosome numbers which may be caused due to genomic mutations especially polyploidy (auto or allopolyploidy) (Soltis et al. 2009, Bedini et al. 2012). There are number of research papers on the phenomena of polyploidy, emphasizing its origin, impact and role in speciation (Stebbins 1985, Ramsey and Schemske 1998, Otto and Whitton 2000, Cifuentes et al. 2010, Jiao et al. 2011). The autotetraploids are generally characterized by the presence of quadrivalents due to homology of 4 sets of chromosomes, whereas, in allopolyploids there is normal pairing because of existence of two separate sets of chromosomes. On the other hand in segmental allotetraploids due to the partial homology of two genomes there is low frequency of quadrivalent formation. In the present study W. somnifera shows normal bivalent formation in all the PMCs, without any quadrivalent formation which indicates its allotetraploid behavior. However, the absence of quadrivalents does not confirm that it is an allotetraploid because there are many artificially produced autotetraploids where there is only bivalent formation because the formation of quadrivalents depends upon many other factors such as localization of chiasmata, small size of chromosomes, and presence of some suppressor genes etc., which does not allow the pairing between the homologous chromosomes (Morrison and Rajhathy 1960, Gottschalk 1978). On the other hand in W. somnifera the meiosis is abnormal with the presence of spindle abnormalities which indicates the absence of multivalents and also indicates that it might be hybrid or more probably due the presence of specific genes which interfere in the pairing and functioning of spindle (Baum et al.1992, Risser-Pascotto et al. 2003; Kumar and Singhal 2008; Singhal and Kaur 2009). The basic function of the spindle is to attach at kinetochore and separate the chromosome or chromatids at anaphases (Wadsworth et al. 2011), these attach to the centromeres (Qu and Vorsa 1999) and rearrange the chromosomes on the equatorial plate and bring them together at metaphase-I (Qu and Vorsa 1999). But, if due to some factors (genetic or environmental) the spindle activity fails then chromosomes are unable to line up in the equator and then separate at Anaphases of the meiosis, which leads to abnormal meiotic course. Earlier, a number of plants have been reported with abnormalities like irregular spindle activity, cytomixis and chromatin stickiness
leading to abnormal microsporogenesis (Baum et al. 1992, Caetano-Pereira and Pagliarinini 2001, Kumar and Singhal 2008, Rai and Kumar 2010, Singhal and Kaur 2009). Abnormalities like lagging chromosomes, chromatin bridges and cytomixis which ultimately lead to abnormal microsporogenesis with the production of dyads, triads, polyads, tetrads with micronuclei, and sterile and fertile pollen grains.
## ACKNOWLEDGMENTS
Authors are thankful to U.G.C., New Delhi for providing financial assistance to research work. The authors are also thankful to Head, Department of Botany, Vikram University, Ujjain, for all the necessary laboratory facilities.

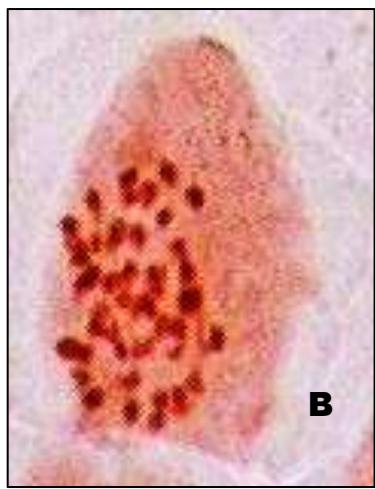
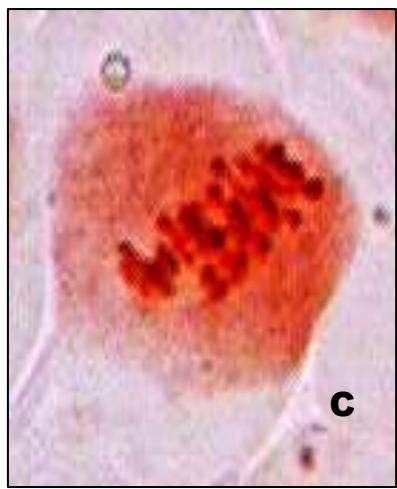
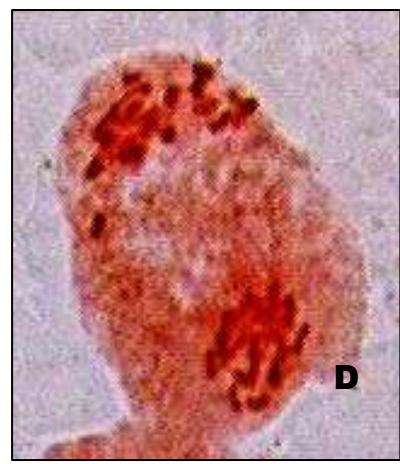
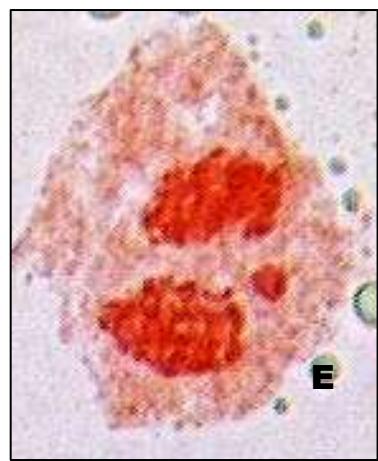
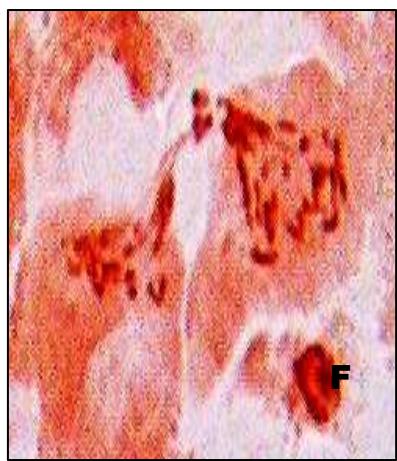
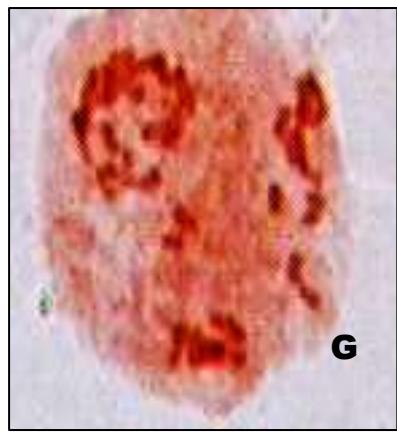
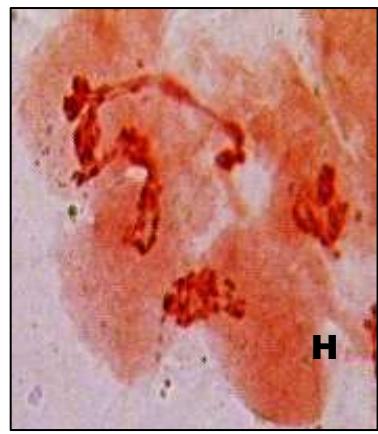
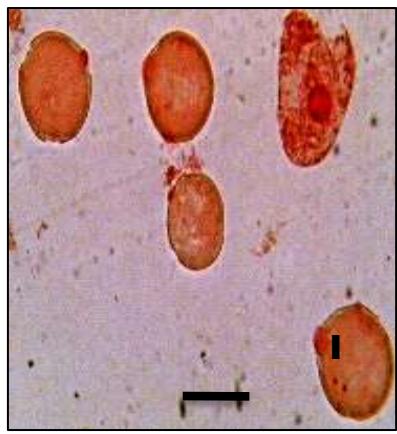 Figure (A-I): Microsporocytes showing different meiotic stages in Withania somnifera Dunal. A) Morphology of Withania somnifera Dunal. B) Diakinesis showing 24 bivalents $(2n = 48)$ C) Metaphase I D) Anaphase I showing 24:24 chromosome segregation E) Laggard at telophase I F) Cytomixis G) Tripolar movement with laggards H) Bridge at telophase II I) Pollens (Scale bar $= 10\mu m$ ).
Table 1: Place of collection, meiotic chromosome count, ploidy level and pollen fertility in Withania somnifera L.
<table><tr><td>Name of taxon</td><td>Place of collection</td><td>Meiotic chromosome Number (n)</td><td>Ploidy level</td><td>Pollen fertility</td></tr><tr><td>Withania somniferaL.</td><td>VU Campus, Ujjain</td><td>n = 24</td><td>4x</td><td>97.52</td></tr></table>
Table 2: Chromosome association (Mean and range) and distribution of chromosomes at anaphase I in Withania somnifera L.
<table><tr><td></td><td rowspan="2">No. of Cells analyzed</td><td colspan="2">Ring bivalents</td><td colspan="2">Rod bivalents</td><td rowspan="2">Anaphase separation</td></tr><tr><td>Species</td><td>Mean</td><td>Range</td><td>Mean</td><td>Range</td></tr><tr><td>Withania somnifera</td><td>25</td><td>17.2</td><td>13-22</td><td>6.8</td><td>2-11</td><td>24:24</td></tr></table>
Table 3: Average chiasma frequency per PMC, terminalized, untereminalized and terminalization coefficient of Withania somnifera L.
<table><tr><td>Taxon</td><td>No. of cells analyzed</td><td>Chiasma/cell Mean ± SD</td><td>Terminalized</td><td>Untereminalized</td><td>Terminalization Coefficient</td></tr><tr><td>Withania somnifera</td><td>25</td><td>45.8 ± 3.25</td><td>41.20 ± 3.12</td><td>4.60 ± 0.80</td><td>0.90</td></tr></table>
Generating HTML Viewer...
References
24 Cites in Article
Q Ahmad,E Britten,D Byth (1984). Effect of interacting genetic factors and temperature on meiosis and fertility in soybean×Glycine soja hybrids.
A Bajpai,A Singh (2006). Meiotic behavior of Carica papaya L. spontaneous chromosome instability and elimination in important cvs in North Indian conditions.
M Baum,E Lagudah,R Appels (1992). Wide Crosses in Cereals.
Gianni Bedini,Fabio Garbari,Lorenzo Peruzzi (2012). Does chromosome number count? Mapping karyological knowledge on Italian flora in a phylogenetic framework.
Creucí Caetano-Pereira,Maria Pagliarini (2001). A new meiotic abnormality in<i>Zea mays</i>: multiple spindles associated with abnormal cytokinesis in both divisions.
Marta Cifuentes,Laurie Grandont,Graham Moore,Anne Chèvre,Eric Jenczewski (2010). Genetic regulation of meiosis in polyploid species: new insights into an old question.
A De Muyt,R Mercier,C Mezard,M Grelon (2009). Meiotic recombination and crossovers in plants.
W Gottschalk (1978). Open problems in polyploidy research.
Olivier Hamant,Hong Ma,W Cande (2006). GENETICS OF MEIOTIC PROPHASE I IN PLANTS.
P Kumar,V Singhal (2008). Cytology of Caltha palustris L. (Ranunculaceae) from cold regions of Western Himalayas.
J Morrison,T Rajhathy (1960). Frequency of Quadrivalents in Autotetraploid Plants.
Sarah Otto,Jeannette Whitton (2000). POLYPLOID INCIDENCE AND EVOLUTION.
L Qu,N Vorsa (1999). Desynapsis and spindle abnormalities leading to 2n pollen formation in Vaccinium darrowi.
P Rai,G Kumar (2010). The genotoxic potential of two heavy metals in inbred lines of maize (Zea mays L.).
Justin Ramsey,Douglas Schemske (1998). PATHWAYS, MECHANISMS, AND RATES OF POLYPLOID FORMATION IN FLOWERING PLANTS.
Claudicéia Risso-Pascotto,Maria Pagliarini,Cacilda Valle (2003). A mutation in the spindle checkpoint arresting meiosis II in<i>Brachiaria ruziziensis</i>.
Masoumeh Rezaei,Ahmad Arzani,Badraldin Sayed-Tabatabaei (2010). Meiotic behaviour of tetraploid wheats (Triticum turgidum L.) and their synthetic hexaploid wheat derivates influenced by meiotic restitution and heat stress.
G Stebbins (1985). Polyploidy, Hybridization, and the Invasion of New Habitats.
V Singhal,D Kaur (2009). Spontaneous occurrence of meiotic spindle abnormalities in 'Long Headed Poppy' (Papaver dubium L.) from Indian cold regions.
Kelian Sun,Kimberly Hunt,Bernard Hauser (2004). Ovule Abortion in Arabidopsis Triggered by Stress.
Douglas Soltis,Victor Albert,Jim Leebens‐mack,Charles Bell,Andrew Paterson,Chunfang Zheng,David Sankoff,Claude De Pamphilis,P Wall,Pamela Soltis (2009). Polyploidy and angiosperm diversification.
L Viccini,C Carvalho (2002). Meiotic chromosomal variation resulting from irradiation of pollen in maize.
Patricia Wadsworth,Wei-Lih Lee,Takashi Murata,Tobias Baskin (2011). Variations on theme: spindle assembly in diverse cells.
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.