A Comparative Study on the Larvicidal Effect of Ethanol Leaf Extracts of Cymbopogon Citratus (Lemongrass) and Ximenia Americana (Sea Lemon) on Anopheles and Culex Larva
## I. INTRODUCTION
Mosquitoes are the most important single group of insects in terms of public health, which transmit a number of diseases such as malaria, filariasis, dengue and zika virus, causing millions of deaths every year [28, 4]. Malaria is transmitted by female anopheles mosquito from person to person. Various species have been found to be the vectors in different parts of the world. Anopheles gambiae complex is the principal vector in Africa [7]. Vector control strategies have traditionally focused on killing mosquitoes using a variety of insecticides. Environmental management (through reduction or removal of mosquito breeding sites) has often been used alongside chemicals or microbiological ovidcides, larvicides and pupicides [31] in areas where endemics mosquito-borne diseases occur. The use of synthetic insecticides has to be regulated given that the development of insecticide resistance is widespread and that there is concern regarding the damage of the environment, effect on human health and non-target organisms [21]. Hence there is a need to develop and incorporate new alternative insecticidal agent.
In recent years, the emphasis on control of the mosquito population has shifted steadily from the use of conventional chemicals towards more specific and environmentally friendly materials, which are generally of botanical origin. Plant products have been used traditionally by human communities in many part of the world against the vector and pest species of insects [25]. The plant derived natural products as larvicides have the advantage of being harmless to beneficial nontarget organisms and environment when compared to synthetic insecticides [27]. The demand for plant-based insecticides is that they are non-toxic, easily available at affordable prices, biodegradable and show broad spectrum, target specific activities against various species of mosquitoes. A lot of phytochemicals extracted from various plant species have been tested for their larvicidal actions against mosquitoes [25]. The use of active toxic agent from plant extract as an alternative mosquito control strategy was dated back to ancient time as such many studies on plant extract and their active constituent compounds against mosquito larvae have been carried out in different parts of the world [16, 18]. Although, there are several reports on the antibacterial effect of Ximenia Americana, there is dearth of information on the larvicidal effect of the plant, thus this work is aimed atcomparing the larvicidal effect of ethanol leaf extracts of Cymbopogon ctratus and Ximenia americana.
## II. MATERIALS AND METHODS
### a) Plant identification and harvesting
The plant samples of C.citratus and X. americana leave were collected from the botanical garden behind Aminu Kano Teaching Hospital (AKTH) and Jibga Town in Bebeji Local Government Area (LGA) Kano respectively. Each plant specimen was submitted to the Herbarium division of Plant Science Department, Bayero University Kano (BUK) and then identification was confirmed in the laboratory according to Delziel [9].
### b) Processing of samples to remove pesticides residues
### c) Extraction of leaves component
The plant powder was extracted according to the method of Anees [2], using soxhlet extraction device. Fifty grams each of the plant fine powder of Cymbopogon citratus and Ximenia americana were extracted using the soxhlet extraction with ethanol as a solvent. The solvent ethanol was then evaporated to obtain ethanolic extract of the leaves using rotary evaporator.
### d) Phytochemical Screening
The plant extracts were analyzed for the presence of Alkaloids, flavonoids, saponins, phytosteroids and tannins as described by Ngbede et al. [22] and Amzad et al. [1].
## i. Test for alkaloids
About 1g powder sample was mixed with 3ml of ammonia solution in a conical flask. It was then allowed to stand for 3 minutes to evaluate for free alkaloids. Chloroform (10ml) was added to the conical flask, shaken and then filtered. The chloroform was evaporated from the crude extract by water bath and Mayer's reagent (3ml) was added. Observation for cream precipitate was done and result was recorded.
## ii. Test for flavonoids
About 1ml of stock solution was taken in a test tube, four drop of dilute NaOH solution was added. An intense yellow colour appeared in the test tube. It became colorless upon addition of few drop of dilute acid which indicate the presence of flavonoids.
## iii. Test for saponins
About 1ml of stock solution was taken in a test tube and diluted with 20ml of distilled water. It was shaken by hand for 15minutes. Observation for foam layer was done and the result recorded.
## iv. Test for phytosteroids
About $1\mathrm{ml}$ of the crude plant extract was taken and mixed with chloroform (10ml) and then equal volume of concentrated sulphuric acid was added to the mixture. Observation for colour change was done and result recorded.
## v. Test for tannins
About 3ml of the crude extract mixed with chloroform and 1ml of acetic anhydride was added. Finally, 1ml of sulphuric acid was added carefully by the side of test tube to the solution. A green color shows the presence of tannins.
## vi. Test for resins
About $5 \mathrm{ml}$ of petroleum ether was added to $1 \mathrm{~g}$ of the powdered extract. Equal volume of copper acetate solution was added and shaken vigorously, then allowed to separate. A green color indicates the presence of resins.
### e) Mosquito larval collection and identification
Anopheles larvae were collected from rice field, and small temporary rain pools in Doko town Garki L.G.A, Jigawa State, Northern Nigeria while Culex larvae were collected from drainage behind provost office of college of health sciences, Aminu Kano Teaching Hospital, Kano. Larvae were collected by dipping method using entomological larval spoons, plastic cups and suitable containers. The larvae were processed at site of collection, worms and other insects were removed according to the method described by Cheah et al., [6].
Morphological features such as presence or absence of siphons, resting position to water surface contained in taxonomic keys were used to identify the Anopheles and Culex larvae as described by Gillies and Coetzee [13].
### f) Transportation of mosquito larvae
All specimen collected from a particular breeding place was kept in a plastic bucket and labeled. (Date and site of collection). The buckets were not filled to the brim in other to allow air space for the larvae to breath. The buckets were well covered before they were transported to the laboratory [29].
### g) Rearing of mosquito larvae
The collected larvae were kept inside in a rearing bowl, at 270C. The larvae were fed daily with yeast according to WHO [34].
### h) Larvicidal effect of the plant extracts
The efficacy of the plants extracts as larvicides against the Anopheles and culex mosquito larvae were evaluated in accordance with guidelines of World Health Organization (WHO) standard method [34]. Ten milliliter $(10\mathrm{ml})$ of the solution for each concentration $30\mathrm{mg / ml}$, $20\mathrm{mg / ml}$, $10\mathrm{mg / ml}$ and $5\mathrm{mg / ml}$ was placed in a small plastic container, mixed thoroughly with $90\mathrm{ml}$ of distilled water. Batches of 25 matured larvae in four replicates were exposed to each of test concentrations. During the period of exposure larvae were fed with yeast [34]. The control contains $100\mathrm{ml}$ of distilled water with $0.2\mathrm{ml}$ of ethanol.
### i) Determination of mortality
The effect of each plant extract was carefully monitored. Larvae were considered dead if they were unable to move after touching with a needle. Moribund larvae were unable to rise to the surface when the water was disturbed. The mortality was observed counted and recorded after 24 and 48 hours of exposure and the percentage mortality was computed using the expression below according to WHO [34].
$$
\text{PercentageMotality} = \frac{\text{Numberofdeadlarvae}}{\text{Numberoflarvaeintroduced}} x 1 0 0
$$
### j) Statistical analysis
The statistical tools used in this study include; Arithmetic mean to get the average number of dead larvae and percentage mortality as well Probit Analysis using SPSS 20 software to calculate $\mathrm{LC}_{50}$ and $\mathrm{LC}_{90}$ values to determine lethal concentrations of the plant extracts on mosquito larvae at 24 and 48 hours of treatment. Analysis of variance (ANOVA) and a two tailed type one t-test was used to determine whether there exists a significant difference in the mean mortality over the period of observation with the $5\%$ confidence interval.
## III. RESULTS
### a) Phytochemicals constituents of the plants extract
The result shows the phytochemical constituents of the C. citratus and X. americana leave extracts. As shown on table 3.1, C. citratus contains alkaloids, saponins, flavanoids, phytosteroids, tannins and resins while X. americana contains saponnins, flavanoids, tannins and resins only, it does not contain alkaloids and phytosteroids.
Table 3.1: Phytochemical constituents of the plants extracts
<table><tr><td>Plants</td><td>Alkaloids</td><td>Saponins</td><td>Flavonoids</td><td>Steroids</td><td>Tannins</td><td>Resins</td></tr><tr><td>C. citratus</td><td>+</td><td>+</td><td>+</td><td>+</td><td>+</td><td>+</td></tr><tr><td>X.americana</td><td>-</td><td>+</td><td>+</td><td>-</td><td>+</td><td>+</td></tr></table>
### b) Larvicidal activities of plants extracts against Anopheles larvae
The result shows the larvicidal effects of C. citratus and X. americana against Anopheles larvae. At concentration of $5\mathrm{mg / ml}$ C. citratus shows $45\%$ mortality after 24 hours and $65\%$ mortality after 48 hours of exposure. At concentration $10\mathrm{mg / ml}$ it shows $73\%$ mortality after 24 hours and $87\%$ after 48 hours. At concentration of $20\mathrm{mg / ml}$ it shows $90\%$ mortality after 24 hours and $100\%$ after 48 hours. At concentration of $30\mathrm{mg / ml}$ it shows $99\%$ mortality after 24 hours and $100\%$ after 48 hours while X. americana on the other hand shows $38\%$ mortality after 24 hours and $53\%$ after 48 hours at concentration of $5\mathrm{mg / ml}$. At concentration of $10\mathrm{mg / ml}$ it shows $54\%$ mortality after 24 hours and $69\%$ after 48 hours. At concentration of $20\mathrm{mg / ml}$ it shows $72\%$ mortality after 24 hours and $92\%$ after 48 hours. At concentration of $30\mathrm{mg / ml}$ it shows $85\%$ mortality after 24 hours and $100\%$ after 48 hours. There is significant association between Anopheles larvae mortality and the concentration of the extracts at $P < 0.05$.
Table 3.2: Larvicidal effects of plants extracts of against Anopheles larvae
<table><tr><td>Plant type</td><td>Concentration (mg/ml)</td><td>n</td><td>Percentage Mean 24 hours</td><td>Mortality 48 hours</td></tr><tr><td>Control</td><td></td><td>25</td><td>0</td><td>0</td></tr><tr><td rowspan="4">C. citratus</td><td>5</td><td>25</td><td>45.00±2.00</td><td>65.00±3.83</td></tr><tr><td>10</td><td>25</td><td>73.00±2.00</td><td>87.00±2.00</td></tr><tr><td>20</td><td>25</td><td>90.00±2.31</td><td>100.00±0.00</td></tr><tr><td>30</td><td>25</td><td>99.00±2.00</td><td>100.00±0.00</td></tr><tr><td>P-value</td><td></td><td></td><td>0.0000</td><td>0.0000</td></tr><tr><td rowspan="4">X. americana</td><td>5</td><td>25</td><td>38.00±2.31</td><td>53.00±2.00</td></tr><tr><td>10</td><td>25</td><td>54.00±2.31</td><td>69.00±3.83</td></tr><tr><td>20</td><td>25</td><td>72.00±3.27</td><td>92.00±0.00</td></tr><tr><td>30</td><td>25</td><td>85.00±2.00</td><td>100.00±0.00</td></tr><tr><td>P-value</td><td></td><td></td><td>0.0000</td><td>0.0000</td></tr></table>
### c) Larvicidal activities of plants extracts against Culex larvae
The result shows the larvicidal effects of C. citratus and X. americana against Culex larvae. At concentration of $5\mathrm{mg / ml}$ C. citratus shows $37\%$ mortality after 24 hours and $67\%$ mortality after 48 hours of exposure. At concentration $10\mathrm{mg / ml}$ it shows $68\%$ mortality after 24 hours and $90\%$ after 48 hours. At concentration of $20\mathrm{mg / ml}$ it shows $83\%$ mortality after 24 hours and $100\%$ after 48 hours. At concentration of $30\mathrm{mg / ml}$ it shows $92\%$ mortality after 24 hours and
100% after 48 hours while X. americana on the other hand shows $28\%$ mortality after 24 hours and $40\%$ after 48 hours at concentration of $5\mathrm{mg / ml}$. At concentration of $10\mathrm{mg / ml}$ it shows $47\%$ mortality after 24 hours and $67\%$ after 48 hours. At concentration of $20\mathrm{mg / ml}$ it shows $67\%$ mortality after 24 hours and $84\%$ after 48 hours. At concentration of $30\mathrm{mg / ml}$ it shows $81\%$ mortality after 24 hours and $100\%$ after 48 hours. There is significant association between Anopheles larvae mortality and the concentration of the extracts at $\mathsf{P} < 0.05$.
Table 3.3: Larvicidal effects of plants extracts of against Culex larvae
<table><tr><td>Plant type</td><td>Concentration (mg/ml)</td><td>n</td><td>Percentage Mean 24 hours</td><td>Mortality 48 hours</td></tr><tr><td>Control</td><td></td><td>25</td><td>0</td><td>0</td></tr><tr><td rowspan="4">C. citratus</td><td>5</td><td>25</td><td>37.00±2.00</td><td>67.00±2.00</td></tr><tr><td>10</td><td>25</td><td>68.00±4.62</td><td>90.00±2.31</td></tr><tr><td>20</td><td>25</td><td>83.00±2.00</td><td>100.00±0.00</td></tr><tr><td>30</td><td>25</td><td>92.00±0.00</td><td>100.00±0.00</td></tr><tr><td>P-value</td><td></td><td></td><td>0.0000</td><td>0.0000</td></tr><tr><td rowspan="4">X. americana</td><td>5</td><td>25</td><td>28.00±0.00</td><td>40.00±0.00</td></tr><tr><td>10</td><td>25</td><td>47.00±2.00</td><td>67.00±2.00</td></tr><tr><td>20</td><td>25</td><td>67.00±2.00</td><td>84.00±0.00</td></tr><tr><td>30</td><td>25</td><td>81.00±2.00</td><td>100.00±0.00</td></tr><tr><td>P-value</td><td></td><td></td><td>0.0000</td><td>0.0000</td></tr></table>
### d) Comparative larvicidal effects of plants extracts against Anopheles larvae
At concentration of $5\mathrm{mg / ml}$ C. citratus shows $45\%$ mortality while X. americana shows $38\%$ with a p-value of 0.8184 after 24 hours of exposure and after 48 hours C. citratus shows $65\%$ while X. americana shows $53\%$ with a p-value of 0.0000. At concentration of $10\mathrm{mg / ml}$ C. citratus shows $73\%$ mortality while X. americana shows $54\%$ with a p-value of 0.8184 after 24 hours of exposure and after 48 hours C. citratus shows $87\%$ while X. americana shows $69\%$ with a p-value of
0.8184. At concentration of $20\mathrm{mg / ml}$ C. citratus shows $90\%$ mortality while X. americana shows $72\%$ with a p-value of 0.5825 after 24 hours of exposure and after 48 hours C. citratus shows $100\%$ while X. americana shows $92\%$ with an undefined p-value. At concentration of $30\mathrm{mg / ml}$ C. citratus shows $99\%$ mortality while X. americana shows $85\%$ with a p-value of 1.0000 after 24 hours of exposure and after 48 hours C. citratus shows $100\%$ while X. americana shows $100\%$ with an undefined p-value.
Table 3.4: Comparative larvicidal effects of plants extracts against Anopheles larvae Concentration of plant extract (mg/ml)
<table><tr><td colspan="6">Concentration of plant extract (mg/ml)</td></tr><tr><td>Plant type</td><td>Time (hrs)</td><td>5</td><td>10</td><td>20</td><td>30</td></tr><tr><td>C. citratus</td><td>24</td><td>45.00±2.00</td><td>73.00±2.00</td><td>90.00±2.31</td><td>99.00±2.00</td></tr><tr><td>X. americana</td><td></td><td>38.00±2.31</td><td>54.00±2.31</td><td>72.00±3.27</td><td>85.00±2.00</td></tr><tr><td>P-value</td><td></td><td>0.8184</td><td>0.8184</td><td>0.5825</td><td>1.0000</td></tr><tr><td>C. citratus</td><td>48</td><td>65.00±3.83</td><td>87.00±2.00</td><td>100.00±0.00</td><td>100.00±0.00</td></tr><tr><td>X. americana</td><td></td><td>53.00±2.00</td><td>69.00±3.83</td><td>92.00±0.00</td><td>100.00±0.00</td></tr><tr><td>P- value</td><td></td><td>0.0000</td><td>0.8184</td><td>-</td><td>-</td></tr></table>
### e) Comparative larvicidal effects of plants extracts on Culex larvae
The result shows the comparative larvicidal effects of C. citratus and X. americana against Culex larvae. At concentration of $5\mathrm{mg / ml}$ C. citratus shows $37\%$ mortality while X. americana shows $28\%$ with a p-value of 0.0000 after 24 hours of exposure and after 48 hours C. citratus shows $67\%$ while X. americana shows $40\%$ with a p-value of 0.0000. At concentration of $10\mathrm{mg / ml}$ C. citratus shows $68\%$ mortality while X. americana shows $47\%$ with a p-value of 0.2025 after 24 hours of exposure and after 48 hours C. citratus shows
90% while X. americana shows $67\%$ with a p-value of 0.8184. At concentration of $20\mathrm{mg / ml}$ C. citratus shows $83\%$ mortality while X. americana shows $67\%$ with a p-value of 1.0000 after 24 hours of exposure and after 48 hours C. citratus shows $100\%$ while X. americana shows $84\%$ with an undefined p-value. At concentration of $30\mathrm{mg / ml}$ C. citratus shows $92\%$ mortality while X. americana shows $81\%$ with a p-value of 0.0000 after 24 hours of exposure and after 48 hours C. citratus shows $100\%$ while X. americana shows $100\%$ with an undefined p-value.
Table 3.5: Comparative larvicidal effects of plant extract against Culex larvae
<table><tr><td colspan="7">Concentration of plants extract (mg/ml)</td></tr><tr><td>Plant type</td><td>Time (hrs)</td><td>5</td><td>10</td><td>20</td><td>30</td><td></td></tr><tr><td>C. citratus</td><td>24</td><td>37.00±2.00</td><td>68.00±4.62</td><td>83.00±2.00</td><td>92.00±0.00</td><td rowspan="6">X. americana</td></tr><tr><td>28.00±0.00</td><td></td><td>47.00±2.00</td><td>67.00±2.00</td><td>81.00±2.00</td><td></td></tr><tr><td>P-value</td><td></td><td>0.0000</td><td>0.2025</td><td>1.0000</td><td>0.0000</td></tr><tr><td>C. citratus</td><td></td><td>4867.00±2.00</td><td>90.00±2.31</td><td>100.00±0.00</td><td>100.00±0.00</td></tr><tr><td>X. americana</td><td></td><td>40.00±0.00</td><td>67.00±2.00</td><td>84.00±0.00</td><td>100.00±0.00</td></tr><tr><td>P- value</td><td></td><td>0.0000</td><td>0.8184</td><td>-</td><td>-</td></tr></table>
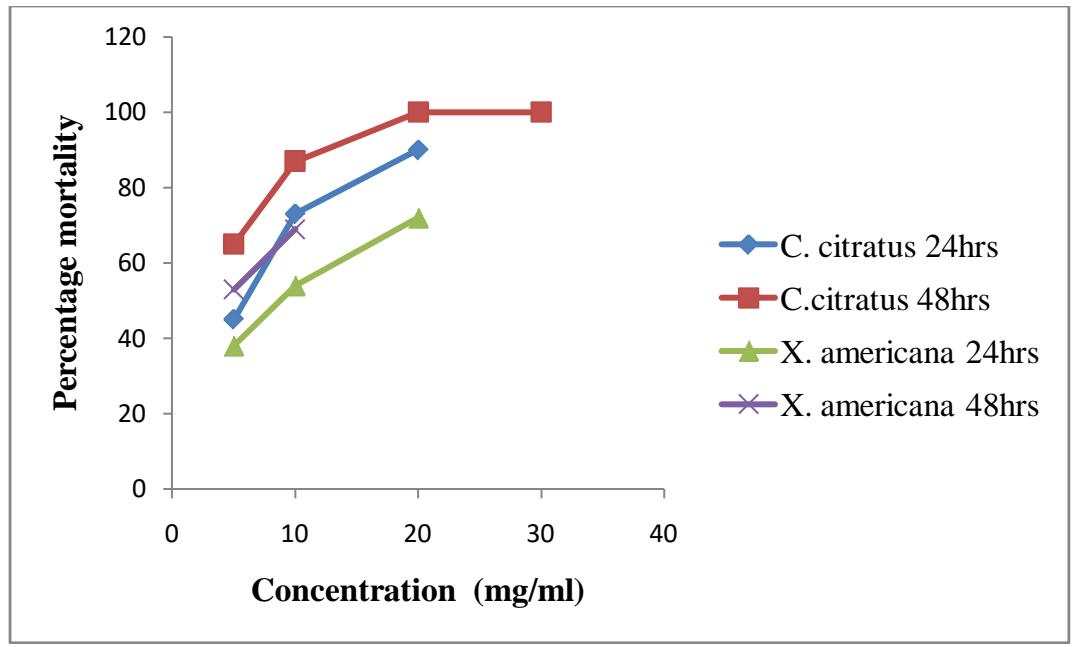 Figure 3.1: Percentage mortality of anopheles in plants extracts
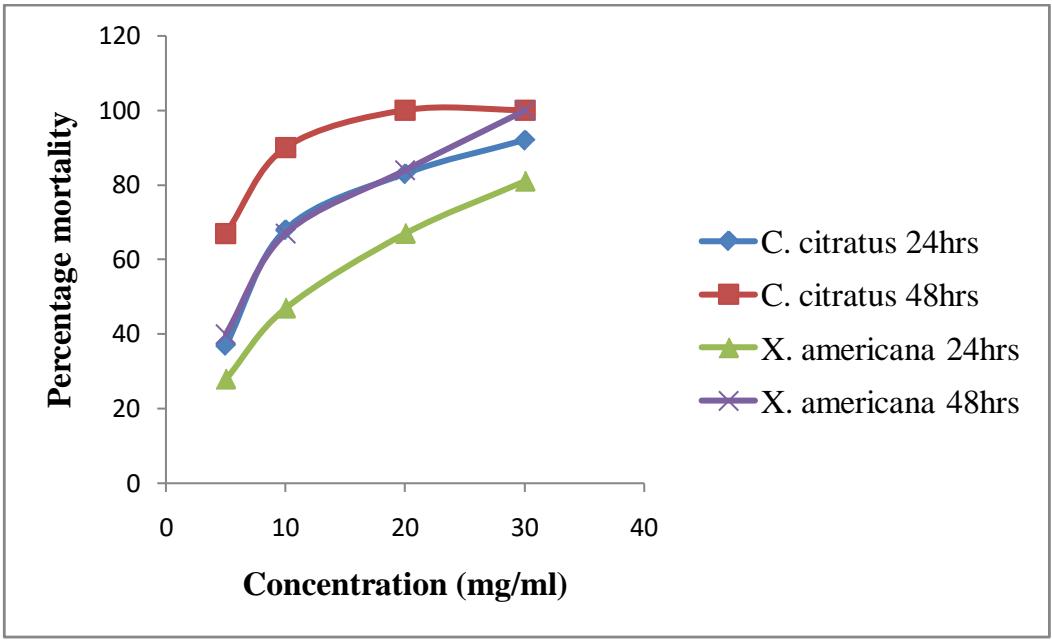 Figure 3.2: Percentage mortality of culex in plants extracts
$f)$ $LC_{50}$ and $LC_{90}$ of plants extracts on Anopheles larvae
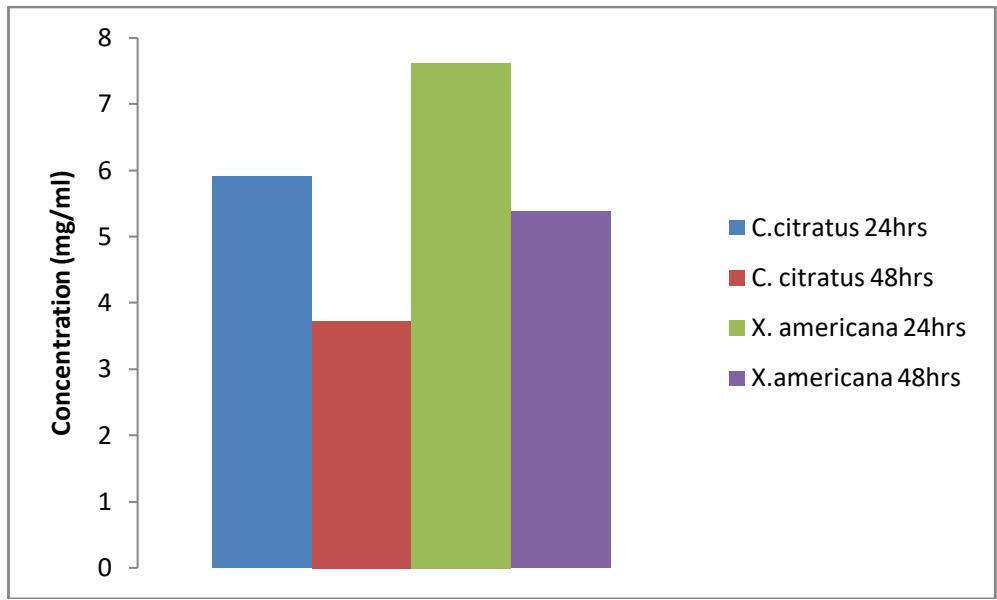 Figure 3.3: $\mathrm{LC}_{50}$ of extract of the plants against anopheles larvae
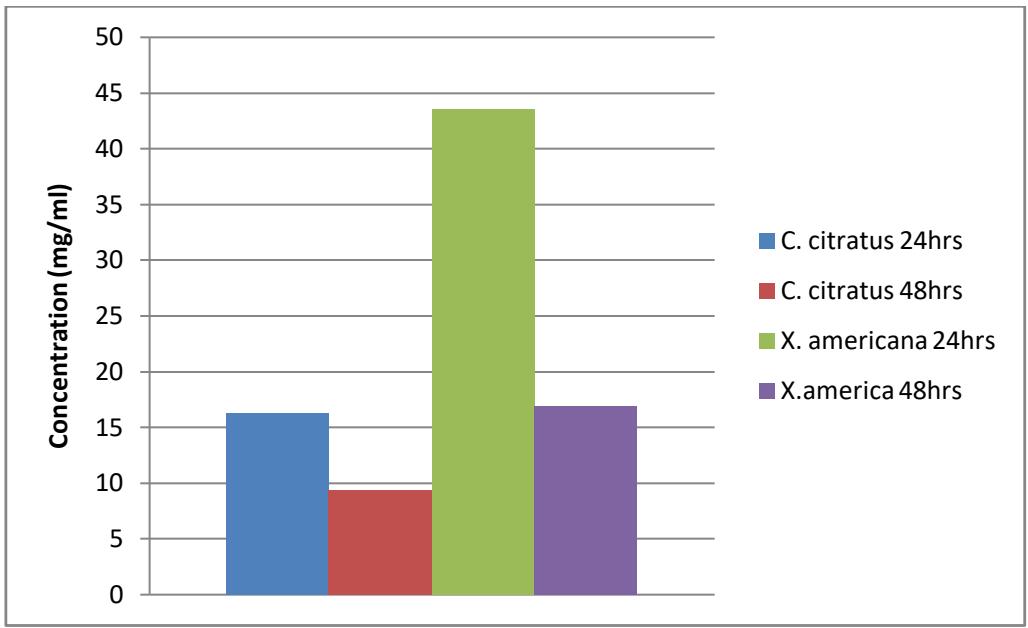 Figure 3.4: $\mathsf{LC}_{90}$ of extract of the plants against anopheles larvae
### g) $LC_{50}$ and $LC_{90}$ of plants extracts on Culex larvae
The result of probit analysis on Anopheles larvae to different concentration of C. citratus after the period of exposure showed that the lethal concentration capable of killing $50\%$ of the larvae $\mathrm{LC}_{50}$ after 24 and 48 hours were 6.851 and $3.7741\mathrm{mg / ml}$ and $\mathrm{LC}_{90}$ were 25.678 and $8.584\mathrm{mg / ml}$ respectively. While for X. americana $\mathrm{LC}_{50}$ were 10.626 and $6.534\mathrm{mg / ml}$ and $\mathrm{LC}_{90}$ were 54.434 and $20.130\mathrm{mg / ml}$ after 24 and 48 hours respectively.
Table 3.8: ${\mathrm{{LC}}}_{50}$ and ${\mathrm{{LC}}}_{90}$ of plants extracts on Culex larvae
<table><tr><td>Plants</td><td>Times (hrs)</td><td>LC50(mg/ml)</td><td>LC90(mg/ml)</td></tr><tr><td rowspan="2">C. citrates</td><td>24</td><td>6.851</td><td>25.678</td></tr><tr><td>48</td><td>3.774</td><td>8.584</td></tr><tr><td rowspan="2">X. Americana</td><td>24</td><td>10.626</td><td>54.434</td></tr><tr><td>48</td><td>6.534</td><td>20.130</td></tr></table>
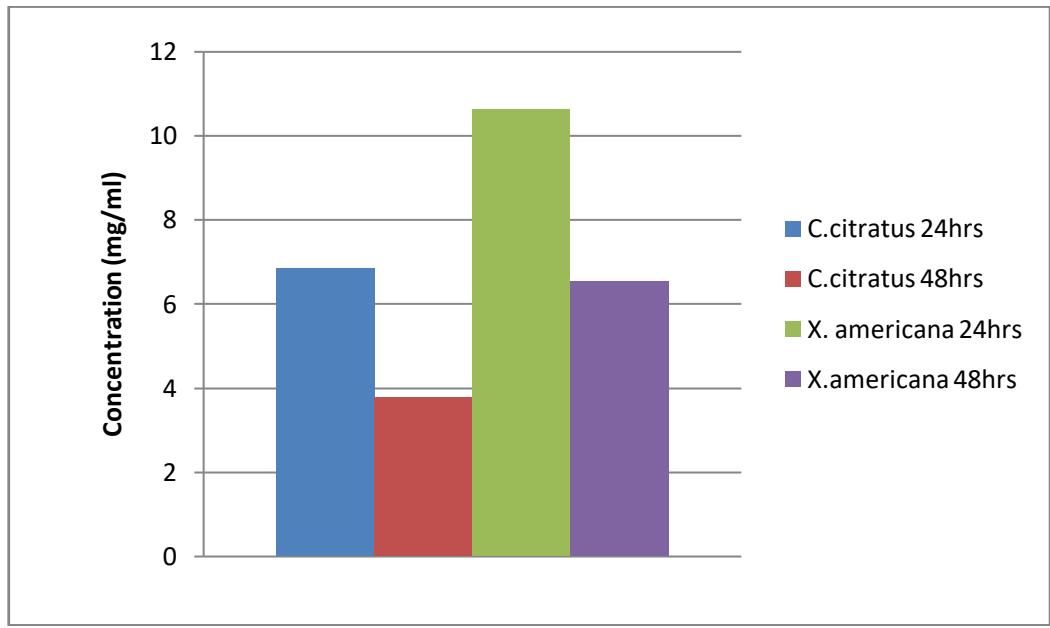
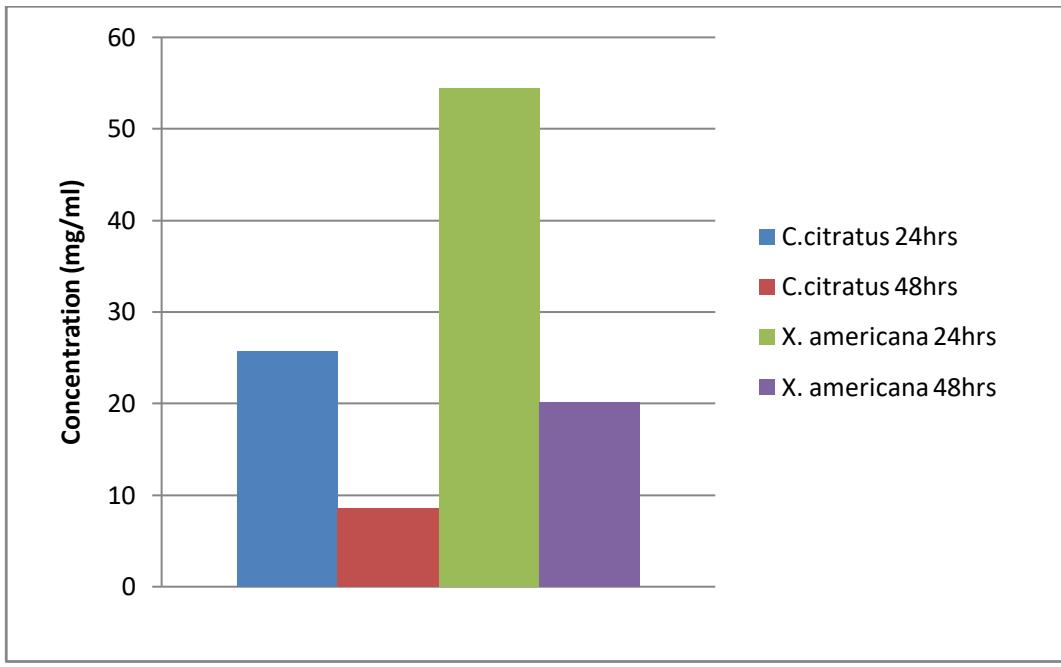 Figure 3.5: $\mathrm{LC}_{50}$ of extract of the plants against culex larvae Figure 3.6: $\mathsf{LC}_{90}$ of extract of the plants against culex larvae
## IV. DISCUSSION
Result of the phytochemical constituents C. citratus show that it contains alkaloids, saponins, flavanoids, phytosteroids, tannins and resins.
Researchers such as Hasim et al.[14], identified the same phytochemicals in C. citratus. Egunyomi et al. [11] worked on the phytochemical component of Nigerian medicinal plants and found the above mentioned active components in Citrus sinensis and C. citratus. Otabor et al.[24]also reported the presence of the above components in the methanolic extract of C. citratus.
However, X. americana contains saponnins, flavanoids, tannins and resins only, it does not contain alkaloids and phytosteroids which is in line with the findings of Shagal et al. [32], Ogunleye and Ibiotoye [23] also reported the absence of alkaloid in X. americana. This variation may be due to geographical location and soil type. More so, Phytochemicals are biologically active, naturally occurring chemical compounds found in plants, they protect plants from diseases, damage and also contribute to the plants colour, aroma and flavor [30]. Insecticidal effects of plants extracts vary not only according to plant species, mosquito species, geographical variation and plant part used, but also due to method of extraction followed and the polarity of the solvent used during extraction, Phytochemicals are extracted either from the whole body of little herbs or from various parts like fruits, leaves, stem bark and roots etc of larger plants or trees. In all cases where the most toxic substances are concentrated are usually extracted for mosquito control [3].
From the mortality bioassay of the ethanolic leaves extract of C. citratus and X. americana shows a proven larvicidal effect against both the mosquito larvae tested (Anopheles and Culex) at different concentrations. The highest concentration of $30\mathrm{mg / ml}$ from C. citratus shows $99\%$ mortality within 24 hours of exposure against Anopheles larvae while against Culex larvae the same extract at same concentration shows $92\%$ mortality. Similarly, other concentrations, $20\mathrm{mg / ml}$ and $10\mathrm{mg / ml}$ shows almost $100\%$ mortality on both Anopheles and Culex mosquito larvae after 48 hours of exposure with the same plant extract. The $5\mathrm{mg / ml}$ concentration shows $65\%$ mortality against Anopheles larvae and $67\%$ mortality against Culex larvae after 48 hours of exposure with the same plant extract.
In contrast, the highest concentration (30mg/ml) of X. americana leave extract shows $85\%$ mortality within 24 hours of exposure against Anopheles larvae and $81\%$ against Culex larvae. Other concentrations, 20mg/ml and 10mg/ml shows more than $50\%$ mortality against both Anopheles and Culex mosquito larvae after 48 hours of exposure with the same plant extract.
The leaves extract of C. citratus and X. americana were found to be effective in killing the mosquito larvae, but C. citratus extract have more effect on the larvae compared to X. americana extract. The results of larvicidal activity showed that the percentage mortality of larvae increases with the increase in concentration of the extract with prolong exposure to treatment. However, in all the experiments no mortality was recorded in the control group which shows that the mortality observed in treating the larvae with extract is due to the presence of some active ingredients that are proven to have some larvicidal properties against mosquito larvae as described by Anupam et al., [3].
Higher percentage mortality was observed on anopheles larvae than on culex larvae which could be as a result of their breeding habitat, Anopheles larvae breed in a fresh water that are free of pollutant, therefore find the plant extracts more toxic than Culex larvae which breed in a stagnant water containing pollutants and other toxic substances, therefore they are more resistant to the plant extracts. The comparison of percentage mean mortality of larvae in extract of C. citratus and that of X. americana showed significant difference at various concentrations and hours of exposure while some showed no significant difference at all concentration and hours of exposure. This could be due to the possession of various phytochemicals constituents that are responsible for the larval mortality, some of these constituents are common within the plants while some are not, therefore the variation in the percentage mortality of the larvae could be as a result of the phytochemical constituents present in the plants. The result of this research shows that leave extracts of X. americana has larvicidal effect on mosquito larvae and it could be due to the presence of secondary metabolites present in the plant such as flavonoids, saponins, tannins, and resins. Flavonoids have been reported to has insecticidal properties acting as a mitochondrial poison, which blocks the electron transport chain and prevent energy production (Musau et al., 2016). Flavonoids have also been reported to inhibit Acetylcholinesterase, act as an insect growth regulator and antifeedant (Jagruti et al., 2014). Saponins on the other hand are freely soluble and can be extracted in both aqueous and organic solvents and perform their action by attacking the cuticle membrane of the larvae, eventually disturbing the membrane which is the main cause for larval death [15].
However, in this study probit analysis shows that C. citratus has the highest effects against Anopheles and Culex larvae with $\mathrm{LC}_{50}$ 5.905mg/ml and $\mathrm{LC}_{90}$ 16.241mg/ml while against Culex larvae with $\mathrm{LC}_{50}$ 6.851mg/ml and $\mathrm{LC}_{90}$ 25.678mg/ml. The result shows that C. citratus has the least concentration required to kill $50\%$ and $90\%$ of the larvae within 24 hours of exposure, compared to X. americana which has the least effect by having the highest concentration required to kill $50\%$ and $90\%$ of the larvae within the time of exposure with $\mathrm{LC}_{50}$ 7.617mg/ml and $\mathrm{LC}_{90}$ 43.471mg/ml against Anopheles larvae while $\mathrm{LC}_{50}$ 10.626mg/ml and $\mathrm{LC}_{90}$ 16.241mg/ml against Culex larvae. Ebe et al. [10] in their findings also revealed that C. citratus have some larvicidal effects when tested against Anopheles gambiae, Aedes eagypti and Culex quinquefaciatus. Musa et al.[19] also revealed that C.citratus have some larvicidal and insecticidal effect when tested against Anopheles mosquitoes. Several researchers studied the components of essentials part of plants such as bark [15] oil from the leaves [19] etc against different species of insects which were proven to have some insecticidal and larvicidal properties.
However, the nature of the bioactive components of plants activities depend on the nature of the solvent used during the extraction. In this study ethanol was used which is among the good solvent for extraction of polar organic compounds. Different compounds have been found to be present in the plants used such as flavonoid, alkaloid, saponin, tannin, resin and phytosteroid which have some insecticidal properties. Furthermore, Eliman et al.,[12] suggest that the use of plants available stand a better option when compared to chemicals for the control of mosquito larvae as the affect non-target organisms and environmental hazards.
## V. CONCLUSION
Plants derived natural products possess a number of phytochemicals that have been proven for larvicidal effect against mosquitoes and that they are non-toxic, easily available at affordable prices, biodegradable and show broad spectrum, targets specific activities. The result for phytochemical screening indicated that alkaloid, flavonoid, saponin, tannin, steroid and resin were present in C.citratus while alkanoid and steroid were absent in X. americana. The leaves extract of C. citrates kills more mosquito larvae than that of X. americana. Therefore, in this study it was concluded that the leaves extract of C. citrates was found to be more effective against both species of mosquitoes exposed (Anopheles and Culex) compared to the leaves extract of X. americana. The higher mortality of the mosquito larvae recorded with C. citrates may be due to the presence of more bioactive compounds found in it compared to X. americana.
This study shows that treating the larvae with plant extracts prevent them from molting to pupae while in the control group the larvae successfully molted and emerged into larvae.
### Competing Interest
The authors of this article declare no conflict of interest through the processes of this work.
#### ACKNOWLEDGEMENT
Our profound gratitude goes to the entire members of staff of the department of Microbiology, Bayero University Kano Nigeria for their academic and technical support for the success of this work. A special thanks also goes to the staff members of pharmaceutical laboratory section of Aminu Kano Teaching Hospital, Kanofor their unflinching support during the data collections phase of this work.
Generating HTML Viewer...
References
34 Cites in Article
M Amzad,K Hossain,Z Salim,Al-Mijizy Am,Q Weli (2013). Study of total phenol, flavonoids contents and phytochemical screening of various leaves crude extracts of locally grown Thymus vulgaris.
A Anees,F Abbas,H Sufia,W Khoo (2010). Optimization of soxhlet extraction of herbaleonuri using factorial design of experiment.
G Anupam,C Nandita,C Goutam (2012). Plant extract as potential mosquito larvicide.
Neeta Sharma (2010). Indigenous Plants and their Larvicidal Potential against Indian Mosquito Vectors: A Review.
S Cheah (2014). Toxicity and sublethal effects of Artemisia annua Linnaeus on Aedes aegypti, Aedes albopictus, Anopheles sinensis and Culex quinquefasciatus.
Shao-Xiong Cheah,Jia-Wei Tay,Lai-Keng Chan,Zairi Jaal (2013). Larvicidal, oviposition, and ovicidal effects of Artemisia annua (Asterales: Asteraceae) against Aedes aegypti, Anopheles sinensis, and Culex quinquefasciatus (Diptera: Culicidae).
Maureen Coetzee (2010). DISTRIBUTION OF THE AFRICAN MALARIA VECTORS OF THE ANOPHELES GAMBIAE COMPLEX.
C Connelly,Elizabeth Bolles,D Culbert,James Devalerio,Michael Donahoe,K Gabel,R Jordi,J Mclaughlin,Anita Neal,Sally Scalera,E Toro,Joseph Walter (2014). Integrated Pest Management for Mosquito Reduction around Homes and Neighborhoods.
J Dalziel (1937). The useful plants of tropical west Africa. Crown agents for overseas and Government and adains.
E Ebe,M Ifeyinwa,Roselyn Njoku,T Chinedu,I Emanuel,E (2015). Larvicidal effect of cymbopogom citratus root and leaf on first instar larval stage of Anophelesgambie, culexquinquefasciatus and Aedeseagypti.
A Egunyomi,I Gbadamosi,K Osinama (2010). Comparative effectiveness of ethanol botanical mosquito repellents.
Abdalla Elimam,Khitma Elmalik,Faysal Ali (2009). Efficacy of leaves extract of Calotropis procera Ait. (Asclepiadaceae) in controlling Anopheles arabiensis and Culex quinquefasciatus mosquitoes.
M Gillies,M Coetzee (1987). A supplement to the anophelinae of Africa south of the sahara (Afrotropical Region). South African Institude for Medical Research.
S Hasim,R Falah,Ayunda Dn (2015). Antioxidant and Antifungal Potential of Murraya Koenigii Leaves Extracts (Crude) and Essential Oil.
M Jagruti,J Kevalia,P Patel (2014). Mode of action of phytochemicals in target insect body.
C Kamaraj,A Bagavan,A Rahuman,A Zahir,G Elango,G Pandiyan (2010). Larvicidal potential of medicinal plant extracts against Anopheles subpictus Grassi and Culex tritaeniorhynchus Giles (Diptera: Culicidae).
Rajan Maheswaran,Savarimuthu Ignacimuthu (2012). A novel herbal formulation against dengue vector mosquitoes Aedes aegypti and Aedes albopictus.
R Maheswaran,S Ignacimuthu,S Sathish (2008). Larvicidal activity of Leucasaspera against the larvae of Culexquinquefasciatus and Aedesaegypti.
A Musa,B Aleiro,A Aleiro,I Tafinta (2015). Larvicidal and insecticidal effect of Cymbopogoncitratus (Lemongrass) on Anopheles mosquitoes in Sokoto State, Nigeria.
J Musau,J Mbaria,J Nguta,M Mathiu (2016). Phytochemical composition and larvicidal properties of plants used for mosquito control in Kwale County, Kenya.
Muhammad Naqqash,Ayhan Gökçe,Allah Bakhsh,Muhammad Salim (2016). Insecticide resistance and its molecular basis in urban insect pests.
J Ngbede,R Yakubu,D Nyam (2008). Phytochemical screening for active compound in Canarium schweinfurthii (Atile) leaves from Jos North, Plateau State, Nigeria.
D Ogunleye,S Ibitoye (2003). Short Communication: Studies of antimicrobial activity and chemical constituents of <i>Ximenia americana</i>.
J Otabor,J Rotimi,L Opoggen,I Egbon,O Uyi (2019). Phytochemical constituents and larvicidal efficacy of methanolic extracts of Cymbopogon citratus, ocimumgratissimum and Vernoniaamygdalina against culex quinqufasciatus larvae.
R Pavela (2007). Possibilities of botanical insecticides exploitation in plant protection.
R Pavela (2008). Larvicidal effects of various Euro-Asiatic plants against Culex quinquefasciatus larvae (Diptera: Culicidae).
B Pitasawat,D Champakaew,W Choochote,A Jitpakdi,U Chaithong,D Kanjanapothi,E Rattanachanpichai,P Tippawangkosol,D Riyong,B Tuetun,D Chaiyasit (2007). Aromatic plant-derived essential oil: An alternative larvicide for mosquito control.
A Rahuman,A Bagavan,C Kamaraj,E Saravanan,A Zahir,G Elango (2009). Efficacy of larvicidal botanical extracts against Culex quinquefasciatus Say (Diptera: Culicidae).
Paulo Rufalco-Moutinho,Nicolás Schweigmann,Denise Bergamaschi,Maria Mureb Sallum (2016). Larval habitats of Anopheles species in a rural settlement on the malaria frontier of southwest Amazon, Brazil.
M Saxena,J Saxena,N Rajeev,D Signh,G Abhishek (2013). Phytochemistry of medicinal plants.
Margit Semmler,Fathy Abdel-Ghaffar,Khaled Al-Rasheid,Heinz Mehlhorn (2009). Nature helps: from research to products against blood-sucking arthropods.
M Shagal,D Kubmarawa,S Bamina (2013). Evaluation of Antimicrobial property of Ximenia americana.
Saghir Sheikh,Shafi Nizamani,Asif Jamali,Aasia Panhwar,Mahvish Channa,Beenish Mirani (2012). Removal of Pesticide Residues from Okra Vegetable through Traditional Processing.
(2005). World Health Organization Communicable disease control, prevention and eradication.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Abdullahi Hasan Amoto. 2026. \u201cA Comparative Study on the Larvicidal Effect of Ethanol Leaf Extracts of Cymbopogon Citratus (Lemongrass) and Ximenia Americana (Sea Lemon) on Anopheles and Culex Larva\u201d. Global Journal of Science Frontier Research - C: Biological Science GJSFR-C Volume 22 (GJSFR Volume 22 Issue C2).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.
A Comparative Study on the Larvicidal Effect of Ethanol Leaf Extracts of Cymbopogon Citratus (Lemongrass) and Ximenia Americana (Sea Lemon) on Anopheles and Culex Larva
Abdullahi Hasan Amoto<p>Bayero University Kano</p>