Saliva has been studied extensively as a potential diagnostic tool over the last decade due to its ease and non-invasive accessibility along with its abundance of biomarkers, such as genetic material and proteins. The activity of protein in saliva increased during ovulation. When we monitored salivary protein activity in 40 different women volunteers during various stages of reproduction like (prepubertal, parous, non-parous, menopausal and in the metabolic disorder state diabetic condition it has been observed that highly significant (p
## I. INTRODUCTION
As a diagnostic fluids Body fluids like blood, saliva, tears, sweat and urine are a source of pathological biochemical markers in which saliva is an excellent biological fluid that is useful for noninvasive exploration of the human diseases and physiological conditions (Villiger et al., 2018). Salivary protein concentration is dependent on gland production at time of day, diet, age, gender and presence of disease (Ferreiro et al., 2002). Saliva consists mainly of water, enzymes, ions and amino acids and performs several important functions in oral health. It contains various biomolecules such as proteins, enzymes and hormones. Saliva contains a large number of proteins that participate in the protection of the oral tissues, for instance lysozyme, lactoferrin, lactoperoxidase, immunoglobulins, ag-glutinin and mucins. Nieuw Amerongen and Veer-man 2002, Denny et al., 2008 and Yan et al., 2008 reported about two thousand proteins in human saliva using mass spectrometry. Sanjay et al., 2008 reported significant high total salivary protein in the saliva of cancer patient. In the beginning of menstruation and during ovulation, the protein content of saliva increases considerably, which turns out to be a rich source of nutrient to bacteria, the count of which may increase during menstruation and ovulation. Protein in the serum is made up of albumin and globulin. Albumin is made mainly in the liver which helps tissue growth and healing. Globulins is made up of different proteins called alpha, beta and gamma. Some globulins are made by liver while others are made by immune systems. Salivary proteins, such as mucins, $\alpha$ -amylase, lysozyme, and peroxidase are synthesized and packed into secretory granules in acinar cells. Salivary protein secretion is strongly enhanced by the sympathetic nerve stimulation. Perinpanayagam et al., 1995 reported that small peptide in saliva showed proteolytic activity. Chicharro et al., 1998 indicated that the salivary level of total protein increases also through $\beta$ -sympathetic activity in salivary glandssince saliva secretion is mainly evoked by the action of androgenic mediators. Hu et al., 2004 and Huang, 2004 studied the structure and function of a large number of proteins in human saliva with traditional biochemical techniques including chromatography, gel electrophoresis, mass spectrometry, immunoassay. Tabak, 2001 and Simpson et al., 2005 also detected various enzymes, enzyme inhibitors, hormones (growth factor) and cytokines (Interlukin-8) in the saliva. Van Nieum Amerongen & Veerman 2002 studied the functional aspect of important proteins in saliva which act as mineralization of the enamel, taste, perception, digestion, inhibition, cell proliferation, chemotaxis, and cell motility. Zachariasen, 1992 also indicated that changes in ovarian hormone levels during puberty, parous pregnancy menstrual cycle and after oral contraceptive use appear to co-relate with decrease in various proteins. Ben-Aryeh et al., 1986 and Arranz et al., 1992 also reported increase in total protein and reduction in amylase activity with age in parotid saliva. Lakshmi et al., 2015 showed a higher level of salivary total proteins and $\alpha$ -amylase in patients with DM. Panchbhai et al., 2010 studied on salivary total proteins, and $\alpha$ -amylase of well-controlled and poorly-controlled DM patients compared with healthy individuals and showed a significantly lower level of salivary $\alpha$ -amylase in patients with well-controlled DM compared to healthy subjects.
## II. MATERIALS AND METHODS
The studies were performed in 63 different human female volunteers of age group (7 to 45 yrs.) categorized as prepubertal, parous, non-parous, menopausal and diabetogenic. The human female unstimulated whole saliva was collected during various periods of parous and non-parous volunteers, viz. preovulatory, ovulatory and post ovulatory phases and also from pre-pubertal, menopausal and diabetogenic stages and were stored at $-70^{\circ}\mathrm{C}$ for further use (Navazesh and Christensen, 1982). Subjects were asked not to swallow any saliva during the collection period. Saliva volume was measured with the tube sealed and then frozen in dry ice until taken back to the laboratory for processing. All the saliva samples of different category of female subjects were placed into salivate tube using a natural cotton swab insert and centrifuged at $400\mathrm{g}$ for 10 minutes at $4^{\circ}\mathrm{C}$. The Total protein was measured as per method of Lowry et al., 1951 by Folin-ciocalteau phenol reagent.
## III. RESULTS & DISCUSSION
Table 1: Level of Total Protein in saliva of different conditions in human female subjects
<table><tr><td>SI No.</td><td>Name of different conditions with symbols</td><td>Level of Total Protein(mg/dl), Mean and SE of 6 samples</td><td>P-Value</td></tr><tr><td>1</td><td>Pre-pubertal-(a)</td><td>9.51 ± 0.153</td><td></td></tr><tr><td rowspan="4">2</td><td>Parous</td><td></td><td>a to c - (p<0.001) HS</td></tr><tr><td>Pre-ovulatory-(b)</td><td>9.40 ± 0.184</td><td>a to e - (p<0.001) HS</td></tr><tr><td>Ovulatory - (c)</td><td>16.50 ± 0.149</td><td>a to f - (p<0.001) HS</td></tr><tr><td>Post ovulatory-(d)</td><td>11.28 ± 0.181</td><td>a to g - (p<0.01) HS</td></tr><tr><td rowspan="4">3</td><td>Non-Parous</td><td></td><td></td></tr><tr><td>Pre-ovulatory-(e)</td><td>7.47 ± 0.264</td><td>b to e - (p<0.001) HS</td></tr><tr><td>Ovulatory - (f)</td><td>11.58 ± 0.147</td><td>c to f - (p<0.001) HS</td></tr><tr><td>Post ovulatory-(g)</td><td>10.38 ± 0.153</td><td>d to g - (p<0.01) S</td></tr><tr><td rowspan="6">4</td><td rowspan="6">Menopausal-(h)</td><td rowspan="6">7.61 ± 0.150</td><td>a to h - (p<0.001) HS</td></tr><tr><td>b to h - (p<0.001) HS</td></tr><tr><td>c to h - (p<0.001) HS</td></tr><tr><td>d to h - (p<0.001) HS</td></tr><tr><td>f to h - (p<0.001) HS</td></tr><tr><td>g to h - (p<0.001) HS</td></tr><tr><td rowspan="8">5</td><td rowspan="8">Diabetogenic-(i)</td><td rowspan="8">6.58± 0.164</td><td>a to i - (p<0.001) HS</td></tr><tr><td>b to i - (p<0.001) HS</td></tr><tr><td>c to i - (p<0.001) HS</td></tr><tr><td>d to i - (p<0.001) HS</td></tr><tr><td>e to i - (p<0.02) S</td></tr><tr><td>f to i - (p<0.001) HS</td></tr><tr><td>g to i - (p<0.001) HS</td></tr><tr><td>h to i - (p<0.01) HS</td></tr></table>
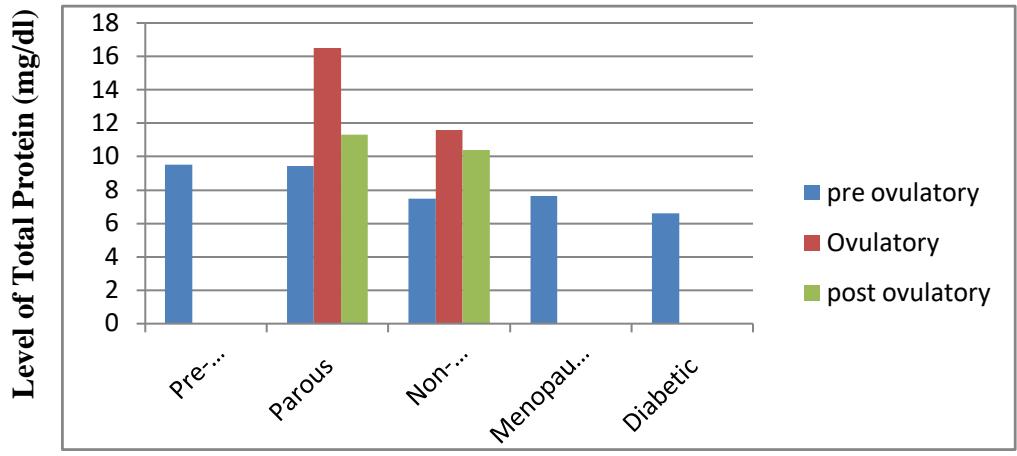 Name of Different condition Figure As per our findings a highly significant increased salivary total protein in parous and nonparous ovulatory and post ovulatory phase of menstrual cycle in comparison to prepubertal salivary total protein might be due to the diminished level of ovarian steroid estrogen & progesterone hormone in pubertal stage of female subject and active secretion and synthesis of steroid hormone in parous & non- parous human female subjects. A highly significant decreased level of salivary protein in non-parous preovulatory, ovulatory, post ovulatory phases of menstrual cycle than parous women phases might be an indication of disturbed hormone level in non- parous women. Earlier report of Alagendran et al., 2013 indicated that protein range was highest at ovulatory phase than pre and post ovulatory phases due to estrogen peak. Thus, increased salivary protein level may be estrogen dependent. He further observed that total protein level in saliva were maximum during ovulation and minimum during post ovulatory phase. But in menopausal women's saliva the total protein showed as decreased level due to physiological endocrinelogical changes in menopausal women. As earlier report of Ambatipudi et al., 2009 indicated that age related changes in protein abundance were observed. But in diabetogenic women a significant & highly significant increased level of salivary protein in comparison to prepubertal, parous and non-parous reproductive cycle phases salivary protein and from menopausal women might be due to disturbed metabolic activity in diabetic condition of women might be due to reduced salivary fluid secretion and various range of underlying pathogenic factors. Earlier report of Antonio et al., 2004 support our findings that diabetic patients were attributed to reduced salivary fluid secretion. Our findings are consistent with the findings of Dodds and Dodds 1997 that increased salivary protein were observed in diabetic patients. Twetman et al., 2002 also observed increased salivary total protein level in type -1 diabetic human female subjects than type-2.
### ACKNOWLEDGEMENTS
Generating HTML Viewer...
References
24 Cites in Article
S Alagendran,S Saibaba,R Muthukumar,Rajkumar,G Guzman,Archunan (2013). Characterization of salivary protein during ovulatory phase of menstrual cycle through MALDI-TOF/MS.
Kiran Ambatipudi,Bingwen Lu,Fred Hagen,James Melvin,John Yates (2009). Quantitative Analysis of Age Specific Variation in the Abundance of Human Female Parotid Salivary Proteins.
Antonio Mata,Duarte Marques,Sara Rocha,Helena Francisco,Carolina Santos,Maria Mesquita,Jaipaul Singh (2004). Effects of diabetes mellitus on salivary secretion and its composition in the human.
E Arranz,S O'mahony,J Barton,A Ferguson (1992). Immunosenescence and mucosal immunity: significant effects of old age on secretory IgA concentrations and intraepithelial lymphocyte counts..
H Ben-Aryeh,A Shalev,R Szargel,A Laor,D Laufer,D Gutman (1986). The salivary flow rate and composition of whole and parotid resting and stimulated saliva in young and old healthy subjects.
José Chicharro,Alejandro Lucía,Margarita Pérez,Almudena Vaquero,Rosario Ureña (1998). Saliva Composition and Exercise.
Chun-Ming Huang (2004). Comparative proteomic analysis of human whole saliva.
Paul Denny (2008). The proteomes of human parotid and submandibular/sublingual gland saliva collected as the ductal secretions.
Anne Dodds,Dodds (1997). Effects of glycemic control on saliva flow rates and protein composition in noninsulin-dependent diabetes mellitus.
C Ferreiro,M,M Hermida,S Prieto,R Rodriguez,A Vazquez,Castro,P Iglesias,Diz Dios (2002). Whole stimulated salivary flow in patients with chronic hepatitis C virus Journal of oral pathology & medicine.
Lawrence Tabak (2001). A Revolution in Biomedical Assessment: The Development of Salivary Diagnostics.
P Lakshmi,E Sridevi,A Sankar,M Kumar,M Sridhar,B Sujatha (2015). Diagnostic perspective of saliva in insulin dependent diabetes mellitus children: an in vivo study.
N J O H Lowry,A L Rosebrough,R J Farr,Randall (1951). Protein measurement with the Folin phenol reagent.
M Navazesh,C Christensen (1982). A Comparison of Whole Mouth Resting and Stimulated Salivary Measurement Procedures.
A Nieuw Amerongen,Eci Veerman (2002). Saliva -the defender of the oral cavity.
Arati Panchbhai,Shirish Degwekar,Rahul Bhowte (2010). Estimation of salivary glucose, salivary amylase, salivary total protein and salivary flow rate in diabetics in India.
H Perinpanayagam,B Vanwuyckhuyse,Z Ji,L Tabak (1995). Characterization of Low-molecular-weight Peptides in Human Parotid Saliva.
P Sanjay,K Hallikeri,A Shivashankara (2008). Evaluation of salivary sialic acid, total protein, and total sugar in oral cancer: A preliminary report.
Shen Hu,Patricia Denny,Paul Denny,Yongming Xie,Joseph Loo,Lawrence Wolinsky,Yang Li,Jim Mcbride,Rachel Ogorzalek Loo,Mavash Navazesh,David Wong (2004). Differentially expressed protein markers in human submandibular and sublingual secretions.
J Simpson,L Wood,P Gibson (2005). Inflammatory mediators in exhaled breath, induced sputum and saliva.
S Twetman,I Johansson,D Birkhed,T Nederfors (2002). Caries Incidence in Young Type 1 Diabetes mellitus Patients in Relation to Metabolic Control and Caries-Associated Risk Factors.
M Villiger,R Stoop,T Vetsch,E Hohenauer,M Pini,P Clarys,F Pereira,R Clijsen (2018). Evaluation and review of body fluids saliva, sweat and tear compared to biochemical hydration assessment markers within blood and urine.
Weihong Yan (2009). Systematic comparison of the human saliva and plasma proteomes.
R D Zachariasen (1993). The Effect of Elevated Ovarian Hormones on Periodontal Health.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Shweta. 2026. \u201cChanges in Total Protein Level in Human Female Saliva during Ovulatory Functions and Metabolic Disorders\u201d. Global Journal of Science Frontier Research - C: Biological Science GJSFR-C Volume 22 (GJSFR Volume 22 Issue C3).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.