A study on electrochemical deposition in a triple-component Co-Ni-Fe system from a chloride electrolyte solution with equal concentrations of Co, Ni, and Fe was performed. The concentrations used were 0.48; 0.083; 0.00625 mol/l, and the temperature was 70°C. The relative content of the components in the film approached the composition of the electrolyte, when the concentration of each component was slightly decreased. The dependence of the composition of films on the current density is explained by concentration polarization.
## I. INTRODUCTION
Thin films of triple alloys consisting of the ferromagnetic metals Fe, Co and Ni have a high magnetization saturation, high magnetic permeability and low coercivity, and they are used in many areas, such as computers, read/write heads and microelectromechanical systems (MEMS). Electrochemical deposition is one of the most preferred methods for producing thin layers of alloy due to its simplicity, cost-effectiveness, versatility and relatively rapid deposition.
A characteristic feature of the electrochemical deposition of Co-Ni-Fe films is the discrepancy between the fraction of elements in the electrolyte and in the film, which makes it difficult to obtain a film with the desired electrophysical properties. This paper presents the results of a study on electrochemical deposition of a triple Co-Ni-Fe system from a chloride electrolyte solution with equivalent concentrations of Co, Ni, and Fe at 0.48, 0.083, and $0.00625\mathrm{mol} / \mathrm{l}$ and a temperature of $70^{\circ}\mathrm{C}$. The study of electrochemical deposition of Co-Ni-Fe alloys provides new insight into the mechanisms that determine the composition of the precipitated layers and their dependence on the composition of the electrolyte.
Finding the composition of the electrolyte to obtain the desired composition of the precipitated film is a very time-consuming task. The decision on the composition of the electrolyte depends on the introduction of additives into the electrolyte that improves the mechanical properties, adhesion and morphology of the films. The conducted studies of congruent electrochemical deposition of the triple Co-Ni-Fe system with equal concentrations of components in the electrolyte allow us to approach finding a solution to the electrolyte composition problem that determines the composition of precipitated films of complex composition.
## II. ELECTROCHEMICAL DEPOSITION OF CO-NI-FE FILMS
For the deposition of Co-Ni-Fe films, a chloride electrolyte solution containing three components, $\mathrm{CoCl}_2\cdot 6\mathrm{H}_2\mathrm{O}$, $\mathrm{FeCl}_2\cdot 4\mathrm{H}_2\mathrm{O}$, and $\mathrm{NiCl}_2\cdot 6\mathrm{H}_2\mathrm{O}$, was used, with the components in a 1:1:1 molar ratio; three concentrations were tested: 0.48, 0.083, and 0.00625 mol/l [1-3]. Various additives were added to the electrolyte solution in the following concentrations: $\mathrm{H}_3\mathrm{BO}_3 - 20\mathrm{g} / \mathrm{l}$, $\mathrm{C}_7\mathrm{H}_4\mathrm{NaNO}_3\mathrm{S}\cdot 2\mathrm{H}_2\mathrm{O} - 1.5\mathrm{g} / \mathrm{l}$, $\mathrm{HCl} - 3\mathrm{ml} / \mathrm{l}$. The film from the specified electrolyte was deposited in a galvanic bath with a volume of 2 liters and a graphite anode. A vertically-oriented metallized silicon wafer was used as the cathode. The distance between the anode and the cathode was $8\mathrm{cm}$.
Insoluble hydroxides were removed by filtration. An alloy film with a diameter of $8\mathrm{cm}$ was obtained on the metallized Ni surface of a $100\mathrm{mm}$ silicon wafer. The electrolyte was heated by a submersible heater to $70^{\circ}\mathrm{C}$ and mixed with a magnetic stirrer. A constant current density of 3 to $40\mathrm{mA/cm}^2$ was maintained in the deposition area on the silicon wafer.
As the concentration of $\mathrm{CoCl}_2$, $\mathrm{NiCl}_2$, and $\mathrm{FeCl}_2$ salts in the electrolyte solution increased, the resistance of the solution decreased, and this resistance determined the amount of current passing between the anode and the cathode and the voltage that changed linearly with increasing current. At a current density of 16 mA/cm2, the voltage between the anode and the cathode was 2 V, 3.5 V, and 5.5 V in the different electrolyte solutions.
With a difference in electrolyte concentrations of 77 times, the growth rate of Co-Ni-Fe films practically does not depend on the concentration of the electrolyte. The growth rate of Co-Ni-Fe films increases with increasing current density and half the values calculated according to Faraday's law, i.e. the cathode current output is 0.5.
1. Electrolytes with Co, Ni, and Fe concentrations of $0.48\mathrm{mol} / \mathrm{l}$ [2] precipitated at a current density of $3.6\mathrm{mA} / \mathrm{cm}^2$. The composition of the film was $\mathrm{Co}_{32}\mathrm{Ni}_{61.5}\mathrm{Fe}_{6.5}$. At a low current density, the composition of the film was characterized by a high nickel content and low iron and content. When the current density of the Co-Ni-Fe film increased to $15\mathrm{mA} / \mathrm{cm}^2$, the iron content rose to $41.3\%$, and at a current density of $20\mathrm{mA} / \mathrm{cm}^2$, it remained approximately the same at $42.35\%$. When the current density of the Co-Ni-Fe film increased to $15\mathrm{mA} / \mathrm{cm}^2$, the nickel content decreased to $7.9\%$, and at a current density of $20\mathrm{mA} / \mathrm{cm}^2$, it remained at the same level of $7.85\%$. The actual change in the iron and nickel content at a current density of $10\mathrm{mA} / \mathrm{cm}^2$ changed the relative cobalt content to $59\%$. At current densities of 15 and $20\mathrm{mA} / \mathrm{cm}^2$, the cobalt content had similar values of 49 and $48\%$.
Fig. 1: Dependence of the composition of films obtained from a three-component solution on the electric current density of $3.6 \div 20 \mathrm{~mA} / \mathrm{cm}^2$. The solution contained $\mathrm{CoCl}_2$, $\mathrm{NiCl}_2$, and $\mathrm{FeCl}_2$ with each component at a concentration of $0.48 \mathrm{~mol} / \mathrm{l}$, and it also contained the following additives: $\mathrm{H}_3\mathrm{BO}_3 - 20 \mathrm{~g} / \mathrm{l}$, $\mathrm{C}_7\mathrm{H}_4\mathrm{NaNO}_3\mathrm{S} \cdot 2\mathrm{H}_2\mathrm{O} - 1.5 \mathrm{~g} / \mathrm{l}$, and $\mathrm{HCl} - 3 \mathrm{ml} / \mathrm{l}$.
2. Figure 2 shows the relative contents of Co, Ni, and Fe in the film during deposition from the electrolyte solution when the concentration of each component was $0.083\mathrm{mol} / \mathrm{l}$. There is a weak dependence on the current density at a value of more than $25\mathrm{mA} / \mathrm{cm}^2$; in the Co-Ni-Fe film, the nickel content is $19 - 22\%$, the iron content is $28 - 33\%$, and the cobalt content has a value of $55 - 47\%$. With a lower current density, the composition of the Co-Ni-Fe film varies greatly; the nickel content decreases from 57 to $19\%$, the iron content increases from 5 to $28\%$, and the cobalt content ranges from $42 - 55 - 48\%$.
Fig. 2: Dependence of the composition of films obtained from a three-component solution on the electric current density of $3.6 \div 40 \mathrm{~mA} / \mathrm{cm}^2$. The solution contained $\mathrm{CoCl}_2$, $\mathrm{NiCl}_2$, $\mathrm{FeCl}_2$, and the concentration of each component was $0.083 \mathrm{~mol} / \mathrm{l}$. The solution included the following additives: boric acid, saccharin, hydrochloric acid.
Comparison of the dependences on the current density of the composition of films obtained from a three-component solution of $\mathrm{CoCl}_2$, $\mathrm{NiCl}_2$, $\mathrm{FeCl}_2$ in which the concentration of the components was either $0.48~\mathrm{mol / l}$ or $0.083~\mathrm{mol / l}$ showed that the dependencies were similar. Increasing the current density led to a decrease in the nickel content and an increase in the iron content, and the predominant cobalt deposition persisted. Their dependence of the composition of the films on the current density was stable within a certain area.
At a current of more than $25\mathrm{mA/cm^2}$, the ratio of metal concentrations in the film to the metal concentrations in the electrolyte was 1.44 for cobalt, 0.97 for iron, and 0.54 for nickel, which is almost the same as the values at an electrolyte concentration of 0.48 mol/l, but closer to the concentrations in the electrolyte. The relative contents of Co, Ni, and Fe in the film differ from the composition of the electrolyte and are highly dependent on the current density. It is not possible to control the deposition with the current density to obtain a film composition equal to the composition of the electrolyte.
3. Electrochemical deposition of Co-Ni-Fe films was performed in an electrolyte solution in which the Co, Ni, Fe chloride concentrations were all 0.00625 mol/l. The solution also contained $30\%$ hydrochloric acid $(0.3\mathrm{ml} / \mathrm{l})$, and the temperature was $70^{\circ}\mathrm{C}$. Redeposition was carried out from the same electrolyte solution, but with the addition of saccharin and boric acid, the results are shown in Table 1.
Table 1: Thickness H and composition of Co-Ni-Fe films after electrochemical deposition from an electrolyte solution in which each component had a concentration of $0.00625 \, \text{mol/l}$. The solution also contained added boric acid and saccharin. Process parameters: pH of the electrolyte, voltage between the anode and cathode U, electric current I, deposition time t, film growth rate V.
<table><tr><td>#</td><td>Ph</td><td>U, V</td><td>I, Ma</td><td>T, Min</td><td>H, μ</td><td>V, Nm/Min</td><td>Co,%</td><td>Ni,%</td><td>Fe,%</td><td>Electrolyte Additive</td></tr><tr><td>1</td><td>2,62</td><td>6,8</td><td>500</td><td>30</td><td>4,34</td><td>145</td><td>42</td><td>13,5</td><td>44</td><td></td></tr><tr><td>2</td><td>2,25</td><td>5,8</td><td>500</td><td>30</td><td>3,19</td><td>106</td><td>34</td><td>35</td><td>31</td><td>Saccharin 3 g/l</td></tr><tr><td>3</td><td>2,8</td><td>8,1</td><td>570</td><td>30</td><td>4,36</td><td>177</td><td>51</td><td>20</td><td>30</td><td></td></tr><tr><td>4</td><td>2,2</td><td>8,55</td><td>570</td><td>30</td><td>5,31</td><td>145</td><td>45,3</td><td>17,8</td><td>36,9</td><td>boric acid 20 ml/l</td></tr><tr><td>5</td><td>2,35</td><td>7,75</td><td>534</td><td>30</td><td>4,5</td><td>150</td><td>44,5</td><td>24</td><td>31,5</td><td>Saccharin 1,5 g/l</td></tr><tr><td>6</td><td>2,7</td><td>10,4</td><td>610</td><td>10</td><td>1,9</td><td>190</td><td>35,9</td><td>29,7</td><td>33,6</td><td></td></tr><tr><td>7</td><td>2,55</td><td>8,3</td><td>610</td><td>10</td><td>2,8</td><td>88</td><td>50,6</td><td>13,7</td><td>35,7</td><td>Saccharin 1,5 g/l boric acid 20 ml/l</td></tr></table>
The results of the study on the composition of the films are shown in Figure 3.
Without electrolyte additives the compositions of Co-Ni-Fe films show with markers connected by lines. The Co content is indicated by rectangular markers and a dashed line. The Ni content is indicated by triangular markers and a dotted line. The Fe content is indicated by round markers and a solid line. For the
samples that contained additives to the electrolyte (saccharin at concentrations of $3\mathrm{g / l}$ (at $10\mathrm{mA / cm}^2$ ) and $1.5\mathrm{g / l}$ (at $10.7\mathrm{mA / cm}^2$ ) and boric acid at a concentration of $20\mathrm{g / l}$ (at $11.4\mathrm{mA / cm}^2$ ), the composition values are presented in the form of individual points. Arrows indicate changes in the composition of films for the selected current in electrolyte solutions containing the appropriate additives.
As seen in the figure, the compositions of the films depend on the current, and at a current density of $12.2\mathrm{mA} / \mathrm{cm}^2$, the compositions of the metals in the Co-Ni-Fe films are close to $33\%$ molar content of $\mathrm{CoCl}_2$, $\mathrm{NiCl}_2$, $\mathrm{FeCl}_2$ salts in the electrolyte, i.e., congruent
Fig. 3: Dependence on the current density $J$ in the range of $10 - 12.8 \, \text{mA/cm}^2$ percentage of the components of films C(Co; Ni; Fe), obtained from an electrolyte with an equal $33\%$ molar content of salts $\text{CoCl}_2$, $\text{NiCl}_2$, $\text{FeCl}_2$, each with a concentration of $0.00625 \, \text{mol/l}$.
The thickness of the concentrated films was measured using an MSA-500 microsystem analyzer. The study of the composition of the films was carried out using a PhilipsXL 40 energy dispersion X-ray microanalyzer.
The dependences of the composition of Co-Ni-Fe films and their magnetic properties on the conditions of electrochemical deposition were studied.
Figure 4 shows the dependences of the voltage between the anode and cathode on the stabilized current densities of $5 - 20\mathrm{mA} / \mathrm{cm}^2$ at equal concentrations of $\mathrm{CoCl}_2$, $\mathrm{NiCl}_2$, and $\mathrm{FeCl}_2$ salts in the electrolyte.
electrochemical deposition of the triple-component Co-Ni-Fe alloy is observed.
Additives to the electrolyte change the composition of the films and disrupt the congruence of deposition in various ways. Saccharin increases the content of Co and Ni and reduces the content of Fe. With a decrease in the additive from 3 to $1.5\mathrm{g / l}$, the change in composition decreases. Boric acid at $20\mathrm{g / l}$ increases the content of Co and Fe and reduces the content of Ni.
The simultaneous addition of saccharin (1.5 g/l) and boric acid (20 ml/l) increases the content of Co, slightly reduces the content of Ni and greatly reduces the content of Fe.
The voltage drop across the interelectrode space varies almost linearly with increasing current density from 5 to $20\mathrm{mA/cm^2}$, i.e. the ohmic conductivity of the electrolyte determines the current in the operating mode. In the galvanostatic mode of electrochemical deposition from the chloride electrolyte with equal concentrations of each component, a decrease of the concentration leads to an increase of the voltage drop across the interelectrode space due to a decrease of the amount of ions in the electrolyte.
Fig. 4: Voltage between the anode and cathode as a function of current density of $5 - 20\mathrm{mA / cm}^2$ at the concentrations of $\mathrm{CoCl}_2$, $\mathrm{NiCl}_2$, and $\mathrm{FeCl}_2$ salts in the chloride electrolyte, M: (1) 0.5, (2) 0.08, and (3) 0.0074.
The growth rates of Co-Ni-Fe films in Fig. 5 differ little for the electrochemical deposition from the chloride electrolytes with the concentration of each component, M: 0.0074; 0.08; 0.5, i.e. when the concentration differs by 70 times. The growth rate of Co
Fig. 5: Dependence of growth rate $V$ of Co-Ni-Fe films on the cathode current density $J$ calculated by the Faraday's law (4) and the experimental curves for the electrolytes with the concentration of each component, M: 0.0074 (3); 0.08 (2); and 0.5 (1).
The results, which were obtained using the electrochemical deposition from the chloride electrolyte with the concentration of each component, M: 0.0074 (3); 0.08 (2); and 0.5 (1), the development of the procedure for the electrolyte preparation with the spectrum control, and the deposition at a temperature of $70^{\circ}\mathrm{C}$ enable one to obtain a low coercive force of Co-Ni-Fe films in a rather wide range of iron content in the film.
Ni-Fe films increases with increasing current density; however, it only slightly depends on the concentration of the components. The growth rates of Co-Ni-Fe films, which are calculated by the Faraday's law, exceed the experimental values by approximately two times.
## III. MAGNETIZATION AND COERCIVE FORCE OF CO-NI-FE FILMS
The magnetization and coercive force of the Co-Ni-Fe films were determined by the hysteresis loop of the magnetic field flux on the magnetic properties analyzer. The composition of the films on the plates was determined using an energy-dispersive X-ray microanalyzer. The results of measurements of magnetic parameters: specific magnetization $B / h$, coercive force $H_c$ and composition of films of triple alloy Co-Ni-Fe are presented in Figure 6.
Fig. 6: Dependence of B/h magnetization (1) and the coercive force Hc (2) of Co-Ni-Fe films on the content of Fe.
Magnetization of Co-Ni-Fe films of $130\mathrm{nWb} / \mu$ at a Fe content of $13\%$ to $23\%$. The coercive force of Co-Ni-Fe films has a minimum value of 1.25 Oe with a Fe content of $16\%$.
## IV. DISCUSSION OF THE RESULTS
The contents of the components in the film, which were fabricated by the electrochemical deposition from the three-component solutions of $\mathrm{FeCl}_2$, $\mathrm{CoCl}_2$, and $\mathrm{NiCl}_2$ with equal concentrations of the components, do not correspond to the composition of the electrolyte. At a high current density, when cobalt and iron are predominantly deposited, and a fraction of nickel in the deposit is small, the dependence of the film composition on the current density at the cathode is stabilized.
Fe-Co-Ni films were deposited by the electrochemical method from a sulfate chloride electrolyte [4], containing, mol/l: $\mathrm{NiSO}_4\cdot 0.304$, $\mathrm{NiCl}_2\cdot 0.084$, $\mathrm{CoSO}_4$ 0.1, $\mathrm{FeSO}_4$ 0.036, $\mathrm{H}_3\mathrm{BO}_3$ 20 g/l, 2 g/l stabilizer, 4 g/l tartaric acid, 4 g/l bleach, 0.1 g/l wetting agent and tartaric acid additives. Optimum conditions for obtaining high-quality films of $\mathrm{Fe}_{15.6-20.6}\mathrm{Co}_{43.8-61.9}\mathrm{Ni}_{22.5-40}$ current density $4\mathrm{A} / \mathrm{dm}^2$, temperature $40^{\circ}\mathrm{C}$, pH 2.3-3.2, tartaric acid concentration 8-12 g/l, molar $\mathrm{Co}^{2+} / \mathrm{Ni}^{2+}$ ratio = 0.26-0.4. The composition of the film depends on the current density, electrolyte temperature, and pH. The ratio of the content of elements in the electrolyte and the film is observed — CRL for Fe, Co = 3, and for Ni = 0.5. Cobalt and iron precipitate with a concentration greater than in the electrolyte, and less nickel.
For electrochemical deposition of iron, the abnormal deposition is characteristic. Iron is deposited more intensively than cobalt and nickel. Cobalt is deposited more intensively than nickel. [4].
Estimating the deposition rate based on electrochemical potentials assumes a normal deposition of nickel, but there are many factors in the processes that determine metal deposition.
The concept of congruent electrochemical deposition determines the equality of the electrolyte and sludge compositions.
The composition of Co-Ni-Fe films formed by electrochemical deposition depends on the temperature, pH, concentration and electrolyte composition [5-6]. The congruence of deposition is achieved by deposition from electrolytes with low concentrations of components and without additives. The same is true of the congruent deposition of Ni-Fe films [7-9].
The choice of current density used to obtain the desired composition of films is widely used in electrochemical deposition. The mechanism of the influence of the cathode current density on the composition of the resulting alloys is not defined.
One can ask the following: why does the composition of the Co-Ni-Fe film in our experiment depend on the density of the current, and at a high current density, why does the composition cease to change?
Here are some of the specifics of the processes. The resistance of the electrolyte in the electrode space is crucial for the ion conduction of dissolved salts.
The total current of the ions discharged on the cathode is much smaller than the current that is set during the process. This difference is due to the large current of ions that do not participate in deposition.
The nickel ions deposited on the cathode are formed on the anode at a high concentration and have little mobility. Iron ions are formed on the anode at a low concentration and have mobility due to the many ions of nickel. Cobalt ions are formed on the anode at a medium concentration but have greater mobility than iron atoms. These are the ions that are involved in the electrochemical reaction occurring during metal deposition. It is possible that such ions are hydroxide species formed by the hydrolysis of CoClOH, NiClOH, and FeClOH.
Current leakage is a continuous process, and the discharge of positive ions on the cathode should be accompanied by the formation of positive ions on the anode. The rate of deposition practically does not depend on the concentration of salts but depends on the density of the current, i.e., the number of positive ions created on the anode and discharged on the cathode. The mobility of active ions varies according to the different natures of the salts.
## V. CHEMICAL AND ELECTROCHEMICAL REACTIONS IN ELECTROLYTE
### a) Hydrogen index of $\mathrm{CoCl}_2$, $\mathrm{NiCl}_2$, $\mathrm{FeCl}_2$ solutions In solutions of $\mathrm{CoCl}_2$, $\mathrm{NiCl}_2$, $\mathrm{FeCl}_2$, a hydrogen pH was measured using a testo 206 meter [6]. The dependence of pH on the concentration of one-component solutions in the concentration range of 0.01 - 1 mol/l is shown in Figure 7. At a concentration of 1 mol/l, dissolving $\mathrm{NiCl}_2$ gives $\mathrm{pH} = 5.8$, $\mathrm{CoCl}_2$ gives $\mathrm{pH} = 4.7$, $\mathrm{FeCl}_2$ gives $\mathrm{pH} = 2.7$. The pH value characterizes the ion balance: the concentration of hydrogen, hydroxyl, acidity or alkalinity of water. Consequently, the hydrolysis of ferric chloride reduces the concentration of hydrogen during dissociation of water the most and increases the amount of hydroxyl.
A change in pH during dilution of the solution is not monotonous, but there are local peaks and a change in slope in the dependence. This means that a decrease in the concentration of the impurity is accompanied by a change in the degree of ionization and the value of the charge of ions. Local peaks are located in the dependences at different salt concentrations: $\mathrm{NiCl}_2 - 0.5\mathrm{mol} / \mathrm{l}$; $\mathrm{CoCl}_2 - 0.03\mathrm{mol} / \mathrm{l}$;
$\mathrm{FeCl}_2 - 0.02 \, \mathrm{mol} / \mathrm{l}$. To obtain the same level of salt ionization, it is necessary to have solutions of each of the salts or of the order of $1 \, \mathrm{mol} / \mathrm{l}$, or $0.01 \, \mathrm{mol} / \mathrm{l}$.
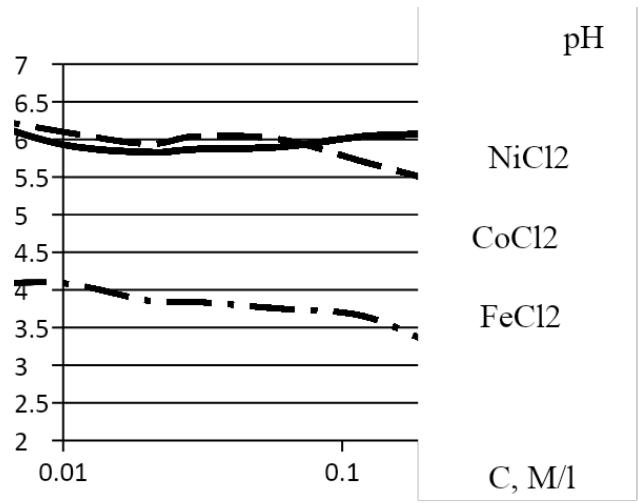 Fig. 7: The dependence of the pH of solutions of $\mathrm{CoCl}_2$, $\mathrm{NiCl}_2$, $\mathrm{FeCl}_2$ on concentration C.
Prior to the peaks, the dependences of pH on salt concentration are linear. After peaks, there is practically no dependence of pH on the concentration of salts of $\mathrm{NiCl}_2$, $\mathrm{CoCl}_2$, but for $\mathrm{FeCl}_2$ there is a change, but at a slower rate than in the linear section.
### b) Hydrolysis Reaction and Electrode Reactions
The experimental features of electrochemical deposition are described by a sequence of chemical and electrochemical reactions. The dissolution of cobalt, nickel and iron chlorides is accompanied by a hydrolysis reaction [5].
$$
F e C l _ {2} + H _ {2} O \leftrightarrow F e C l O H + H ^ {+} + C l ^ {-}.
$$
$$
\mathrm {N i C l} _ {2} + \mathrm {H} _ {2} \mathrm {O} \leftrightarrow \mathrm {N i C l O H} + \mathrm {H} ^ {+} + \mathrm {C l} ^ {-}.
$$
$$
\mathrm {C o C l} _ {2} + \mathrm {H} _ {2} \mathrm {O} \leftrightarrow \mathrm {C o C l O H} + \mathrm {H} ^ {+} + \mathrm {C l} ^ {-}.
$$
On the anode, there is an electrochemical reaction, and in accordance with the size of the current and the potentials of ionization, positive ions with different concentrations are formed. At the nickel anode, chlorine dissolves the electrode when produced.
Graphite anodes are successfully used in the electrolysis of chloride salt solutions, and the anode potential at them is low. The products of the destruction of graphite anodes do not contaminate the cathode metal. On graphite anodes, chlorine is released.
$$
\mathrm {F e O H} ^ {+} + \mathrm {H} ^ {+} + \mathrm {C l} _ {2} \uparrow .
$$
$$
\mathrm {N i O H} ^ {+} + \mathrm {H} ^ {+} + \mathrm {C l} _ {2} \uparrow .
$$
$$
CoOH^{+} + H^{+} + Cl_{2} \uparrow.
$$
Under the influence of the electric field in the electrolyte, there is a drift in the positive ions of the metal hydroxides from the anode to the cathode at a speed determined by the magnitude of the mobility of the ions and the tension of the electric field.
On the cathode, there is an electrochemical reaction of metal discharge from hydroxide species and the formation of water molecules.
$$
F e O H ^ {+} + e ^ {-} + H ^ {+} = F e + H _ {2} O.
$$
$$
\mathrm {N i O H} ^ {+} + \mathrm {e} ^ {-} + \mathrm {H} ^ {+} = \mathrm {N i} + \mathrm {H} _ {2} \mathrm {O}.
$$
$$
\mathrm {C o O H} ^ {+} + \mathrm {e} ^ {-} + \mathrm {H} ^ {+} = \mathrm {C o} + \mathrm {H} _ {2} \mathrm {O}.
$$
On the cathode, there is an electrochemical reaction of metal discharge from hydroxide species and the formation of water molecules.
### c) Concentration Polarization
The change in the electrode potential due to a change in the concentration of reagents in the electrode space during the passage of current is called concentration polarization [10]. Electrochemical reactions on the electrodes lead to a significant change in the concentrations of substances due to the slow diffusion of reagents or the removal of reaction products. The difference in the diffusion coefficients or mobility of the ions of the electrolyte component determines the balance of partial ionic currents and the content of the components in the film from the current density.
Based on the concept of concentration polarization, our results on the dependence of the concentration of the components of the triple alloy Co-Ni-Fe on the current density can be interpreted taking into account the dependence of the mobility of the ions, and not the potential of the electrode. Polarization of the electrode - the change in its potential has the same value for the ions of the three metals, and the mobility of the metal ions differs significantly, as the measurements show with a dynamic change in the potential of the electrode.
It has been experimentally established [11] that an area with an increased pH value is formed near the cathode (Figure 8).
Fig. 8: pH profiles in the vicinity of cathode during Fe-Ni alloy deposition. $(\mathrm{Fe}^{2+}$ 0.2 M/L, $\mathrm{Ni}^{2+}$ 0.8 M/L)
An increase in pH to a value of 7 in the region of $0.5 \, \mathrm{mm}$ near the cathode indicates the release of water during a cathode electrochemical reaction. The greater the current density, the stronger the deviation of the composition of the electrolyte near the cathode from a uniform homogeneous one corresponding to the thermodynamic equilibrium occurs.
The ions of the electrolyte component have different diffusion coefficients during the diffusion mechanism of mass transfer and mobility values during drift of ions in an electric field, which determines the different dependence of the content of components in the film on the current density, which creates a layer of concentration polarization that limits the mass transfer of electroactive ions to the electrode. The formation of a layer with a high pH value in the rolling region of the electrolyte explains the dependence of the composition of the precipitate on the mixing of the electrolyte, which violates the layer of concentration polarization.
The thickness of the layer deposited by the electrolyte during an electrochemical reaction - SISEER is fairly large, on the order of a millimeter. A layer of this thickness has a significant effect on the mass transfer of ions to the electrodes.
The formation of a layer with a changed composition in the cathode region of the electrolyte explains the dependence of the composition of the sediment on the mixing of the electrolyte that destroys the concentration polarization layer.
The lower the concentration of the electrolyte is, the smaller the deviation of the composition of the films from the equilibrium state and the closer the composition of the films to the composition of the electrolyte. Therefore, congruent electrochemical deposition of Co-Ni-Fe alloy can be obtained only when the concentrations of the main components of the electrolyte are low and when boric acid and saccharin are not added.
The flow of ionic current disrupts the thermodynamic equilibrium in the electrolyte due to the release of the products of electrochemical reactions in the electrode regions, and the mixing of the electrolyte with mechanical or magnetic stirrers, when exposed to an ultrasonic field, during rotation or reciprocating movement of the cathode, erodes the electrode layers and evens out the distribution of salts in the electrolyte.
With a dynamic change in voltage at a speed of $10\mathrm{mV / s}$ and control of the current flowing through the electrode, the dynamic nature of the behavior of ion currents is manifested \[12\](Figure 9). The presence of current peaks in the dynamic mode of voltage change is associated with the kinetic properties of the ions. The positive ions formed on the anode when approaching the cathode change the potential near the working electrode and reduce the current of the electrode. The difference in the mobility of cobalt, iron and nickel ions determines the temporal and amplitude change in the electrode current.
Fig. 9: Dependence of electrode current in a three-electrode electrochemical cell on dynamically changing voltage in solutions: 1- NiSO4; 2- FeSO4; 3- CoSO4; 4- FeSO4 + NiSO4 +CoSO4.
The presence of current peaks in the dynamic mode of voltage change is associated with the kinetic properties of the ions. The positive ions formed on the anode when approaching the cathode change the potential near the working electrode and reduce the current of the electrode. The difference in the mobility of cobalt, iron and nickel ions determines the temporal and amplitude change in the electrode current.
The flow of ionic current disrupts the thermodynamic equilibrium in the electrolyte due to the release of the products of electrochemical reactions in the electrode regions, and the mixing of the electrolyte with mechanical or magnetic stirrers, when exposed to an ultrasonic field, during rotation or reciprocating movement of the cathode, erodes the electrode layers and evens out the distribution of salts in the electrolyte.
With a low cathode current density and a high concentration of salts in SISEER, a low concentration of residues of molecules arises, from which metal atoms are released and which limit the flow of ionic currents of all three metals. The composition of the film is determined by partial ion currents in accordance with the mobility of ions in the electrolyte with the initial concentration of components. The increase in current density leads to an increase in the content of molecule residues after the electrochemical reaction of metal release on the cathode. The contribution of partial currents of metal ions depends on the composition of the concentration polarization layer. There is a limitation in the deposited film of the nickel concentration alloy, which has a small amount of mobility. Iron-containing ions have more mobility in the concentration polarization layer. The concentration of iron in the film increases. The mobility of cobalt-containing ions in SISEER is high, which leads to a
strong increase in the relative content of cobalt in the film.
Stirring the salt solution disrupts SISEER. Carrying out the process of electrochemical precipitation without mixing reduces the nickel content and increases the iron content in the film, since there is no violation of CISER. With a high cathode current density and a low concentration of salts in the electrolyte region near the cathode, the flow of ionic currents of all three metals is not limited. As a result, congruent deposition of the Co-Ni-Fe alloy film is observed.
Establishes [13] that at a given speed of rotation of the cathode, it is possible to select the density of the deposition current to obtain a given film composition at a different ratio of nickel and iron in the electrolyte. Electrochemical precipitation of Ni-Fe alloys with a wide spectrum of composition was carried out from a sulfate-chloride electrolyte containing $\mathrm{Ni}^{2+} / \mathrm{Fe}^{2+}$ ions (nickel sulfate - iron chloride) in proportions of 5: 1, 10: 1, 15: 1, 20: 1, 25: 1, boric acid and ascorbic acids, saccharin, sodium lauryl sulfate (Figure 10).
Fig. 10: Deposition of $\mathrm{Ni}_{81}\mathrm{Fe}_{19}$ permalloy films from an electrolyte with a different ratio of the content of the main components of Ni: Fe at current density - j and platinum cathode displacement rate [13].
The processes were carried out at current densities (20 - 160 mA/cm2), room temperature 23oC, pH = 3.0. The cathode was a platinum disk moving at a speed of 0.0005 - 0.008 cm/s. The change in cathode speed and current density made it possible, as shown in Figure 10, for a given ratio of nickel and iron 10: 1, 15: 1, 20: 1, 25: 1 in the electrolyte to obtain the composition of the permalloy film $\mathrm{Ni}_{81}\mathrm{Fe}_{19}$.
In our experiment with electrolyte stirring, according to the terminology adopted in the book [14], the effect of diffusion on the rate of electrochemical processes is not basic. The transport of electroactive ions is determined by the migration of ions at a high current of the background electrolyte. It should be noted that in the book it is believed that the voltage drop in the electrolyte occurs near the electrodes, and the field strength in the electrolyte is very small, which contradicts the continuity of the current.
Based on the experimental results of obtaining Co-Ni-Fe films, a mechanism of electrochemical deposition based on the phenomena of salt hydrolysis, discharge of positive ions at the anode, drift of ions in the volume of the electrolyte and discharge of negative ions at the cathode with precipitation of metals and the formation of SISEER [15], has been proposed.
Experimental data show a difference between the deposition rate of the three components of the Co-Ni-Fe alloy and the metal content in the electrolyte. The determining factor is the equality of the charges of the ions that form the partial currents of the electroactive ions. For the Triple Alloy Co-Ni-Fe, such conditions are met at an electrolyte component concentration of 0.06 M/L and a current density of $12\mathrm{mA/cm^2}$, which gives congruent deposition.
## VI. CONCLUSION
The deposition of Co-Ni-Fe films was performed in a chloride electrolyte with a ratio of $C_{\mathrm{Co}}$: $C_{\mathrm{Ni}}$: $C_{\mathrm{Fe}} = 1$: 1: 1, and technology for preparing an electrolyte via filtration and performing deposition at a temperature of $70^{\circ}\mathrm{C}$ was developed. It was found that the desired 1:1:1 ratio of concentrations of the metals in the film was achieved at a current density of $12.5\mathrm{mA/cm}^2$ and a metal chloride concentration of 0.00625 M/L. Co-Ni-Fe films are obtained reproducibly with minimal mechanical stresses and with good adhesion to the nickel sublayer during electrochemical deposition. The mechanistic study of the electrochemical deposition of a Co-Ni-Fe alloy as the electrolyte concentration changes resulted in congruent deposition of the three-component alloy. The use of congruent electrochemical deposition will simplify the choice of electrolyte composition for obtaining films with a complex composition. A review of the literature [16-47] shows that none of the researchers used this method of research.
Generating HTML Viewer...
References
46 Cites in Article
R Tikhonov,A Cheremisinov,D Gorelov,Iu. Kasakov (2020). The Magnetic Properties of the Co-Ni-Fe Films were obtained by Electrochemical Deposition by Tikhonov's Method.
R Tikhonov,S Polomoshnov,V Amelichev,A Cheremisinov,A Kovalev (2021). CoNiFe Triple System Films Formation by Electrochemical Deposition.
Y Yang (2015). Preparation of Fe-Co-Ni ternary alloys with electrodeposition.
Robert Tikhonov (2021). Features of the Electrochemical Deposition of Films from a Triple System of CoNiFe.
Unknown Title.
R Tikhonov,A Cheremisinov,M Tikhonov (2021). Ion Discharge in Electrochemical Deposition of CoNiFe Films.
Robert Tikhonov (2020). Mechanism of the Electrochemical Deposition of a CoNiFe Alloy.
R Tikhonov (2020). Electrochemical Deposition of NiFe Alloy at a Temperature of 70°C.
Robert Tikhonov (2020). Normal Electrochemical Deposition NiFe.
Robert Tikhonov (2019). Electrochemical Deposition of NiFe Alloy at a Temperature of 70°C.
B Damaskin,O Petriy,G Cirlina (2015). Electrochemistry. Textbook for universities.
Hiroaki Nakano,Masayuki Matsuno,Satoshi Oue,Masaaki Yano,Shigeo Kobayashi,Hisaaki Fukushima (2004). Mechanism of Anomalous Type Electrodeposition of Fe-Ni Alloys from Sulfate Solutions.
I Hanafi,A Daud,S Radiman (2016). Potentiostatic Electrodeposition of Co-Ni-Fe Alloy Particles Thin Film in a Sulfate Medium.
Steven Leith,Shirley Ramli,Daniel Schwartz (1999). Characterization of Ni x Fe1 − x ( 0.10 < x < 0.95 ) Electrodeposition from a Family of Sulfamate‐Chloride Electrolytes.
Yuliy Gamburg,Giovanni Zangari (2011). Theory and Practice of Metal Electrodeposition.
R Tikhonov,A Cheremisinov,D Gorelov,Iu. Kasakov (2021). The Magnetic Properties of the Co-Ni-Fe Films were obtained by Electrochemical Deposition by Tikhonov's Method.
Tetsuya Osaka,Takahiro Sawaguchi,Fumio Mizutani,Tokihiko Yokoshima,Madoka Takai,Yutaka Okinaka (1999). Effects of Saccharin and Thiourea on Sulfur Inclusion and Coercivity of Electroplated Soft Magnetic CoNiFe Film.
N Zech,E Podlaha,D Landolt (1999). Anomalous Codeposition of Iron Group Metals: I. Experimental Results.
I Tabakovic,S Riemer,V Inturi,P Jallen,A Thayer (2000). Organic additives in the electrochemical preparation of soft magnetic CoNiFe films.
Ibro Tabakovic,Venkatesewara Inturi,Steve Riemer (2002). Composition, Structure, Stress, and Coercivity of Electrodeposited Soft Magnetic CoNiFe Films: Thickness and Substrate Dependence.
L Perez,K Attenborough,J De Boeck,J Celis,C Aroca,P Sánchez,E López,M Sánchez (2002). Magnetic properties of CoNiFe alloys electrodeposited under potential and current control conditions.
Daheum Kim,D-Y Park,B Yoo,P Sumodjo,N Myung (2003). Magnetic properties of nanocrystalline iron group thin film alloys electrodeposited from sulfate and chloride baths.
Q Huang,E Podlaha (2004). Simulation of Pulsed Electrodeposition for Giant Magnetoresistance FeCoNiCu/Cu Multilayers.
D-Y Park,B Yoo,S Kelcher,N Myung (2006). Electrodeposition of low-stress high magnetic moment Fe-rich FeCoNi thin films.
Tiffany Yun,Wen Jiang (2008). Effects of additives on magnetic properties of electroplated CoNiFe films// Master's Theses.
James Rohan,Bernadette Ahern,Ken Reynolds,Stephan Crowley,David Healy,Fernando Rhen,Saibal Roy (2009). Electroless thin film CoNiFe–B alloys for integrated magnetics on Si.
L,A Csik,K Vad,E Toth-Kadar,A Pekker,G Molnár (2010). On the composition depth profile of electrodeposited Fe-Co-Ni alloys//Chemistry.
K Sundaram,V Dhanasekaran,T Mahalingam (2011). Structural and magnetic properties of high magnetic moment electroplated CoNiFe thin films.
L Phua,N Phuoc,C Ong (2012). Effect of Ni concentration on microstructure, magnetic and microwave properties of electrodeposited NiCoFe films.
Jie Gong,Steve Riemer,Augusto Morrone,Venkatram Venkatasamy,Michael Kautzky,Ibro Tabakovic (2012). Composition Gradients and Magnetic Properties of 5–100 nm Thin CoNiFe Films Obtained by Electrodeposition.
Li Jian,- Mei (2013). Effect of boron/ phosphorus containing additives on electrodeposited CoNiFe soft magnetic thin films.
N Valko,W Gurtovoy (2013). Structure and properties of coatings Co-Ni-Fe, electrolyticall y besieged by X-ray radiation//Fisika tverdogo tela.
Z Kayani,S Riaz,S Naseem (2014). Structural and magnetic properties of FeCoNi thin films.
Hilal Kuru,Hakan Kockar,Ozen Demirbas,Mursel Alper (2013). Characterizations of Electrodeposited NiCoFe Ternary Alloys: Influence of deposition potential.
N Kourov,V Pushin,A Korolev,Yu. Knyazev,M Ivchenko,Yu. Ustyugov (2015). Peculiar features of physical properties of the rapid quenched AlCrFeCoNiCu high-entropy alloy.
S Romankov,Y Park,I Shchetinin (2015). Mechanical intermixing of elements and self-organization of (FeNi) and (CoFeNi) nanostructured composite layers on a Ti sheet under ball collisions.
T Yanai (2016). Еlectroplated Fe-Co-Ni films prepared from deep-eutectic-solvent-based plating baths.
K Eguchi,K Azuma,T Akiyoshi,T Yanai,M Nakano,H Fukunaga (2016). DC/Pulse Plating of Fe-Ni-Co Films.
T Yanai,K Koda,K Eguchi,K Takashima,T Morimura,M Nakano,H Fukunaga (2017). Effect of Ammonium Chloride in Plating Baths on Soft Magnetic Properties of Electroplated Fe–Ni Films.
D Li,E Podlaha (2017). Template-Assisted Electrodeposition of Fe-Ni-Co Nanowires: Effects of Electrolyte pH and Sodium Lauryl Sulfate.
T Yanai,K Koda,J Kaji,H Aramaki,K Eguchi,K Takashima,M Nakano,H Fukunaga (2018). Electroplated Fe-Co-Ni films prepared in ammonium-chloride-based plating baths.
S Romankov,Y Park,I Shchetinin (2018). Structural transformations in (CoFeNi)/Ti nanocomposite systems during prolonged heating.
Ibro Tabakovic,Venkatram Venkatasamy (2018). Preparation of metastable CoFeNi alloys with ultra-high magnetic saturation (Bs = 2.4–2.59 T) by reverse pulse electrodeposition.
D Li (2018). Fabrication of Fe-Ni-Co nanowires by electrodeposition coupled with hydrogen evolution reaction and electrochemical detection of pyocyanin.
H Cesiulis,N Tsyntsaru,E Podlaha,D Li,J Sort (2018). Electrodeposition of Iron-Group Alloys into Nanostructured Oxide Membranes: Synthetic Challenges and Properties.
V Long,U Saraç,M Baykul,L Trong,Ş Ţălu,D Trong (2022). Electrochemical Deposition of Fe-Co-Ni Samples with Different Co Contents and Characterization of Their Microstructural and Magnetic Properties//Materials Science.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
R.Tikhonov. 2026. \u201cEffect of Electrolyte Concentration in Electrochemical Deposition on Co-Ni-Fe Film Composition\u201d. Global Journal of Research in Engineering - J: General Engineering GJRE-J Volume 22 (GJRE Volume 22 Issue J4).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.