## I. INTRODUCTION
Power consumption of refrigeration plants makes $48\%$ to $60\%$ from total power consumption of retail store.
Because of ozone depletion and global warming risks, we ought to use new ecological friendly refrigerants, which require improving refrigeration components, processes and cycle architecture to increase energy efficiency.
We need to use methods that could help to improve energy efficiency based on scientific methods.
There are some methods that used for thermodynamic analysis - energy method, exergy method and entropic and statistical method.
Energy method of analysis based on first law of thermodynamic and does not allow getting information about energy losses by system components. Exergy and entropic and statistical method based both on first and second laws of thermodynamic, but usage of entropic and statistical methods for system analysis used for cold generation is preferable.
In this paper introduced usage of entropic and statistical method of analysis for refrigeration plants operation.
## II. ENTROPIC AND STATISTICAL ANALYSIS OF REFRIGERATION PLANTS
### a) Entropic and Statistical Method of Analysis
Real processes in refrigeration plants are irreversible and nonequilibrium.
The reason of irreversibility is finite difference of mass streams potentials (temperature difference, pressure difference etc.) due to processes nonequilibrium.
The measure of irreversibility is entropy production. According to second law of thermodynamics:
$$
d S \geq \frac {d Q}{T} \quad \text {E q . (1)}
$$
The entropy has property of additivity e.g. total entropy production of the system equal to the sum of entropy change of each sub-system.:
$$
\Delta\mathcal{S} = \sum_{j} \Delta\mathcal{S}_{j} \quad \text{Eq.}(2)
$$
where $\sum_{i} S\Delta_{j}$ - entropy production in sub-systems, $i$ - sub-systems quantity.
According to Gyui-Stodola's theorem, for it is necessary to perform work for entropy production compensation. This work will be transferred to environment as heat. The total work spent for refrigeration plant operation will equal:
$$
L _ {t o t} = L _ {m i n} + \Delta L \quad \text {E q .} \tag {3}
$$
$L_{\text {min }}$ - minimum work for ideal refrigeration cycle with total reversible processes.
Distribution of energy expenses for entropy production compensation for one stage refrigeration cycle is shown on Figure 1.
Ideal Carnot cycle (c1-c2-c3-c4) is totally reversible, it means that work, which is spent for cold generation, will be minimum and is expressed by the area c1-c2-c3-c4. Real cycle is expressed by the area 1-2-3-4-5-6. Work, which is necessary for entropy production compensation in compressor, is expressed by area d-e-e3-e2. Area e3-2-3-4-e1 or equal to it in size e-f-e4-e3 expresses work, which is necessary for entropy production compensation in condenser. Work,
which is necessary for entropy production compensation in evaporator, is expressed by area c-d-e2-c2, in throttling devices – by area a-b-c3-e1.
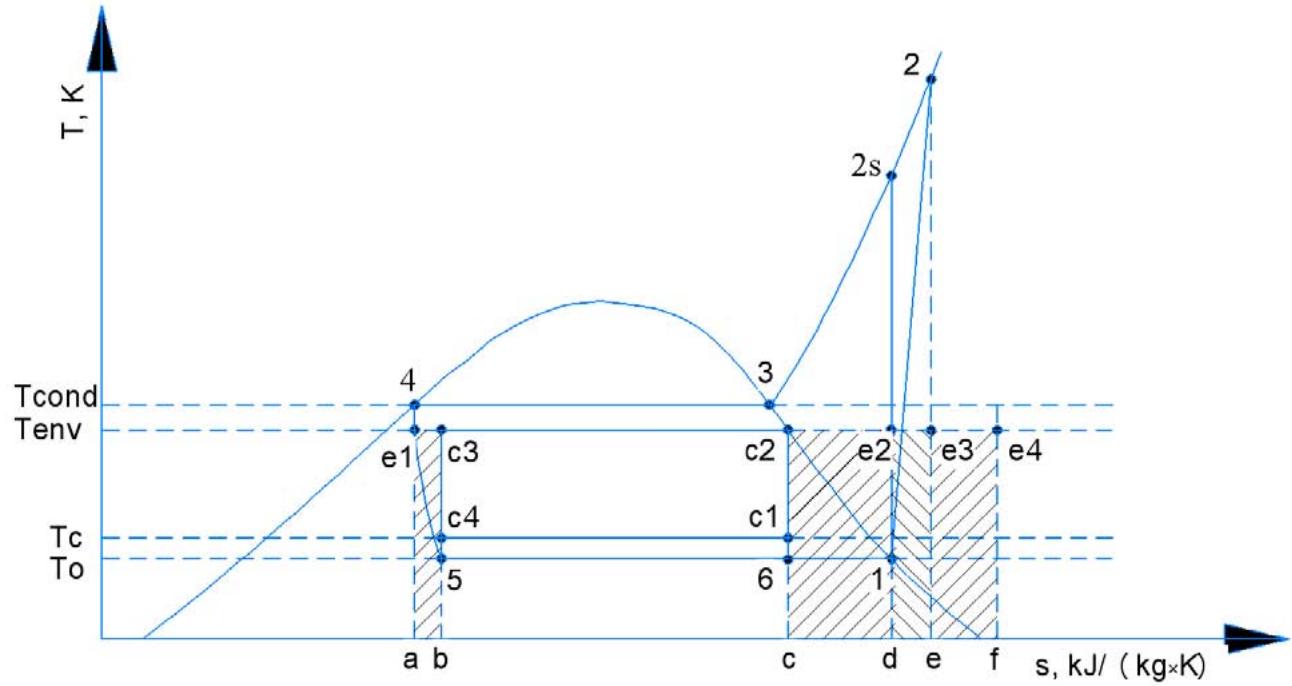 Figure 1: Distribution of Energy Expenses for Entropy Production Compensation by Elements of Single Stage Refrigeration Cycle
Total real work for single stage refrigeration cycle is expressed by the sum of areas.
$$
L_{tot} \propto (c1 - c2 - c3 - c4) + (d - e - e3 - e2) + (e - f - e4 - e3) + (c - d - e2 - c2) + (a - b - c3 - e1) = (1 - 2 - 3 - 4 - 5 - 6) \propto \left(h_{2} - h_{1}\right) \tag{Eq. (4)}
$$
Analysis of the single stage refrigeration cycle analysis will be carried out in the following sequence.
Specific mass cooling capacity at evaporation temperature.
$$
q _ {O} = h _ {1} - h _ {4} = h _ {1} - h _ {5} \quad \text {E q . (5)}
$$
Minimum specific work which is necessary for cold generation.
$$
I _ {\text{min}} = q _ {O} \times \frac{T _ {\text{env}} - T _ {C}}{T _ {C}} \tag{Eq. (6)}
$$
Adiabatic compression work, calculated by using h-lgp diagram:
$$
I _ {S} = h _ {2 S} - h _ {1} \tag {Eq. (7)}
$$
Actual specific compression work.
$$
I_{comp} = q_{cond} - q_{o} = h_{2} - h_{4} - (h_{1} - h_{4}) = \frac{I_{s}}{\eta_{s}} \quad \text{Eq.} \tag{8}
$$
Degree of thermodynamic efficiency.
$$
\text{therm} = \frac{\text{I} _ {\text{min}}}{\text{I} _ {\text{comp}}} \tag{Eq. (9)}
$$
COP at adiabatic compression.
$$
\mathfrak{S} = \frac{q_{O}}{I_{S}} \tag{Eq. (10)}
$$
Actual value of COP.
$$
\text{sact} = \frac{q _ {O}}{I _ {\text{comp}}} \tag{Eq. (11)}
$$
Part of compression work, which is necessary for entropy production compensation in condenser, is the sum of parts of compression works for entropy production compensation in gas cooling process $\Delta I_{sh}$ and condensation process $\Delta I_{cd}$:
$$
\Delta I _ {\text{cond}} = \Delta I _ {\text{s h}} + \Delta I _ {\text{c d}} \quad \text{E q .} \tag{12}
$$
where
$$
\Delta I _ {s h} = \left(h _ {2 s} - h _ {3}\right) - T _ {\text{env}} x \left(s _ {2 s} - s _ {3}\right) \quad \text{E q .} \tag{13}
$$
$$
\Delta I_{cd} = T_{env} \times \left(h_{3} - h_{4}\right) \times \left(\frac{1}{T_{env}} - \frac{1}{T_{cond}}\right) \tag{Eq. (14)}
$$
Part of compression work, which is necessary for entropy production compensation in throttling process.
$$
\Delta I_{thr} = T_{env} \times \left(s_{5} - s_{4}\right) \quad \text{Eq. (15)}
$$
Part of compression work, which is necessary for compensation of entropy production in heat transferring processes from cooling object to refrigerant (evaporation).
$$
\Delta I _ {e. e V a p} = \left(h _ {1} - h _ {5}\right) \times T _ {e n V} \times \frac{T _ {c} - T _ {o}}{T _ {o} \times T _ {c}} \quad \text{Eq.} \tag{16}
$$
Part of compression work, which is necessary for compensation of entropy production in heat transferring processes from cooling object to refrigerant (total).
$$
\Delta I_{evap} = \Delta I_{e.evap} \quad \text{Eq. (17)}
$$
Specific adiabatic compression work is a sum of parts of compression work for compensation of entropy production in all processes of refrigeration cycle.
$$
I _ {s. c a l c} = I _{min} + \Delta I _ {\text{cond}} + \Delta I _ {\text{thr}} + \Delta I _ {\text{evap}} \tag{Eq. (18)}
$$
Energetic losses in compressor.
$$
\Delta I _ {\text{comp}} = I _ {\text{comp}} - I _ {\text{s . calc}} \quad \text{E q .} \tag{19}
$$
Rated compression work.
$$
I _ {\text{comp . calc}} = I _ {\text{s . calc}} + \Delta I _ {\text{comp}} \quad \tag{Eq. (20)}
$$
By using analysis results, the chart of distribution losses on system elements can be constructed.
### b) Refrigeration Plant Analysis in Case of Condensation Pressure Algorithm Change
One of the goals, which is very important to refrigeration equipment end user, is to evaluate results of implementation of new technical solution (usage of more efficiency compressors, heat exchangers, new control algorithms etc).
The goal of refrigeration plant optimization (it is located in Moscow, Russia) was energy consumption reduction. This plant work with R404A as refrigerant and used for 10 medium temperature consumers. Design date is shown in Table 1.
Table 1: Design Date of Refrigeration Plant
<table><tr><td>Cycle</td><td>Single Stage</td></tr><tr><td>Refrigerant</td><td>R404A</td></tr><tr><td>Evaporation temperature, °C</td><td>-10</td></tr><tr><td>Condensation temperature, °C</td><td>+45</td></tr><tr><td>Suction gas superheat, K</td><td>15</td></tr><tr><td>Liquid subcooling, K</td><td>0</td></tr><tr><td>Condenser type</td><td>Air cooled</td></tr><tr><td>Cooling capacity, kW</td><td>33,8</td></tr><tr><td>Quantity of compressors, pcs.</td><td>2</td></tr><tr><td>Compressor type</td><td>scroll</td></tr><tr><td>Model</td><td>ZB76KCE-TFD - 1 pcs.
ZBD76KCE-TFD - 1 pcs.</td></tr><tr><td>Consumers quantity, pcs.</td><td>10</td></tr><tr><td>Thermal processing</td><td>Storage</td></tr><tr><td>Product type</td><td>Egg (1 consumer), milk products (6 consumers), sausages (2 consumers), fish (1 consumer)</td></tr></table>
There were two measurement periods - 4.5 days with fixed condensing set point (condensation pressure was keeping on the same value) and 4.5 days with floating condensing set point (condensation pressure was changing depending on ambient temperature).
Initial date (average values for considered period) is given in Table 2, changing of operating parameters is shown on Figs. 2 and 3, where System 1 is refrigeration plant before algorithm change, System 2 is refrigeration plant after algorithm change.
Table 2: Initial date for Analysis
<table><tr><td>Temperatures, °C</td><td>System 1</td><td>System 2</td></tr><tr><td>Evaporation</td><td>-9.0</td><td>-9.07</td></tr><tr><td>Condensation</td><td>+34.79</td><td>+19.33</td></tr><tr><td>Ambient</td><td>+8.51</td><td>+8.18</td></tr><tr><td>Air in cooling volume</td><td>+1.95</td><td>+2.1</td></tr><tr><td colspan="3">Adiabatic efficiency was taken from selection software of compressor manufactures</td></tr><tr><td>Adiabatic efficiency</td><td>0.7117</td><td>0.6661</td></tr></table>
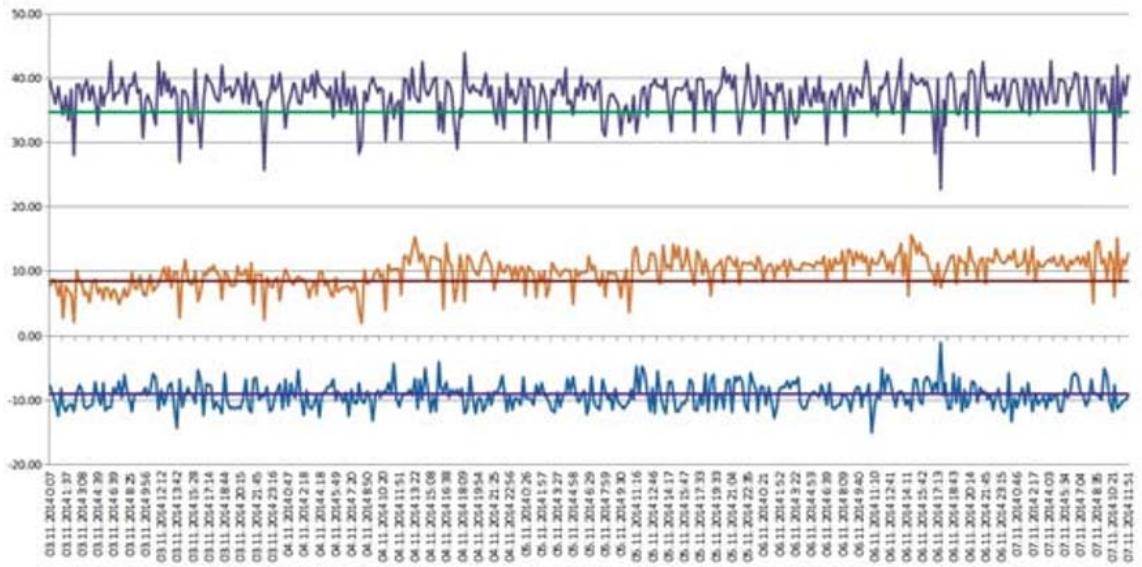
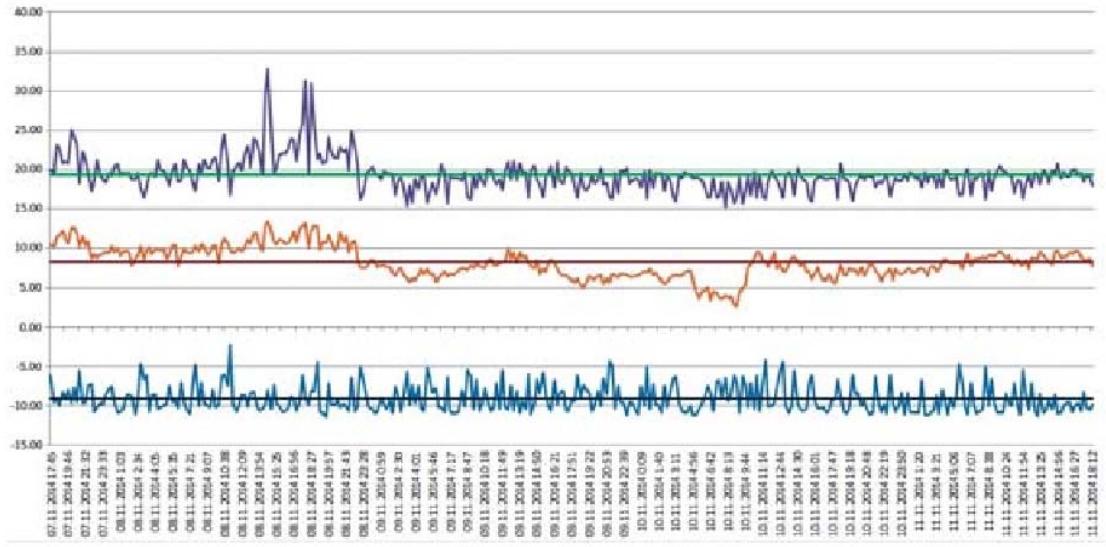 Figure 3: Schedules of Change of Condensation Temperature (Top Schedule), Ambient Temperature (Middle Schedule), Evaporation Temperature (Lower Schedule) and Their Average Values (Horizontal Lines) For System 1
Because there was not possibility to measure all necessary values by using regular devices, some values were taken according to statistical information:
- Total superheat on suction line 12 K;
- Calculation of energetic losses in evaporator was done by using average value of cooling air temperature in all cooling room.
 Figure 4: Schedules of Change of Condensation Temperature (Top Schedule), Ambient Temperature (Middle Schedule), Evaporation Temperature (Lower Schedule) and their Average Values (Horizontal Lines) for System 1

Analysis results are given in Table 3 and on Fig.
5.
Table 3: Analysis Results
<table><tr><td>-</td><td>System 1</td><td>System 2</td></tr><tr><td>q0, kJ/kg</td><td>119.71</td><td>144.52</td></tr><tr><td>Imin, kJ/kg</td><td>2.88</td><td>3.19</td></tr><tr><td>Is.calc, kJ/kg</td><td>27.96</td><td>19.32</td></tr><tr><td>Icomp, kJ/kg</td><td>39.24</td><td>29.00</td></tr><tr><td>ηtherm</td><td>0.0795</td><td>0.1184</td></tr><tr><td>εact</td><td>3.05</td><td>4.98</td></tr><tr><td>ΔIcond,%</td><td>34.45</td><td>23.98</td></tr><tr><td>ΔIthr,%</td><td>16.47</td><td>10.29</td></tr><tr><td>ΔIevap,%</td><td>12.91</td><td>21.32</td></tr><tr><td>ΔIcomp,%</td><td>28.83</td><td>33.4</td></tr></table>
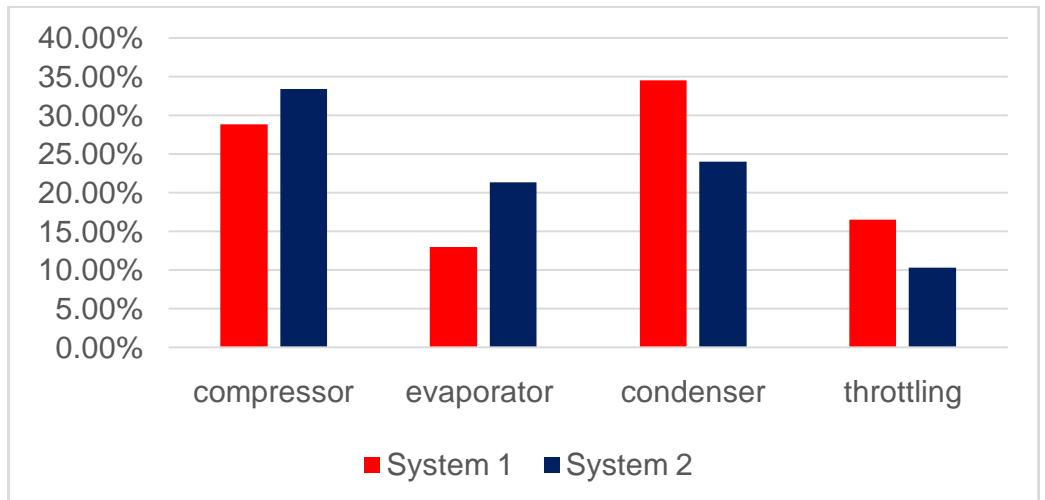 Figure 5: Part of Compression Work (Energetic Losses) which is Necessary for Compensation of Entropy Production in System Elements for Refrigeration Plant before and after Optimization,% from Compression Work
### c) Efficiency Analysis at Refrigerant Plant Operation with Different Working Cycles
One more and, perhaps, more relevant task is the analysis of various systems with different working cycles.
Comparison of two systems is given below. System 3 works by using single stage refrigeration cycle, located in Moscow. System 4 works by using refrigeration cycle with economizer, located in Volgsky, Russia. Design date is shown in Table 4.
Table 4: Design Date of Refrigeration Plants
<table><tr><td></td><td>System 3</td><td>System 4</td></tr><tr><td>Refrigerant</td><td>R404A</td><td>R404A</td></tr><tr><td>Refrigeration cycle</td><td>Single stage</td><td>With economizer</td></tr><tr><td>Evaporation temperature, °C</td><td colspan="2">-35</td></tr><tr><td>Condensation temperature, °C</td><td colspan="2">+40</td></tr><tr><td>Suction superheat, K</td><td colspan="2">15</td></tr><tr><td>Liquid subcooling, K</td><td colspan="2">0</td></tr><tr><td>Temperatures, °C</td><td></td><td></td></tr><tr><td>evaporation</td><td>-33.46</td><td>-32.58</td></tr><tr><td>condensation</td><td>+36.75</td><td>+31.35</td></tr><tr><td>ambient</td><td>+10.58</td><td>+15.98</td></tr><tr><td>air in cooling volume</td><td>-15.14</td><td>-16.25</td></tr><tr><td>Average superheat in evaporators, K</td><td>13.67</td><td>9.73</td></tr><tr><td>Adiabat</td><td></td><td></td></tr></table>
As in the previous case, operation parameters were taken from monitoring system for specified period. Initial average date for analysis is given in Table 5.
Analysis results are given in Table 6 and on Fig.
6.
Table 6: Analysis Results
<table><tr><td></td><td>System 3</td><td>System 4</td></tr><tr><td>q0, kJ/kg</td><td>101.89</td><td>167.31</td></tr><tr><td>Imin, kJ/kg</td><td>10.73</td><td>22.17</td></tr><tr><td>Is.calc, kJ/kg</td><td>50.67</td><td>61.91</td></tr><tr><td>Icomp, kJ/kg</td><td>75.31</td><td>97.97</td></tr><tr><td>ηtherm</td><td>0.149</td><td>0.214</td></tr><tr><td>εact</td><td>1.35</td><td>1.71</td></tr><tr><td>ΔIcond,%</td><td>19.59</td><td>14.64</td></tr><tr><td>ΔIthr,%</td><td>22.58</td><td>9.41</td></tr><tr><td>ΔIevap,%</td><td>10.88</td><td>13.2</td></tr><tr><td>ΔIcomp,%</td><td>32.7</td><td>36.78</td></tr><tr><td>ΔIeco,%</td><td>-</td><td>3.34</td></tr></table>
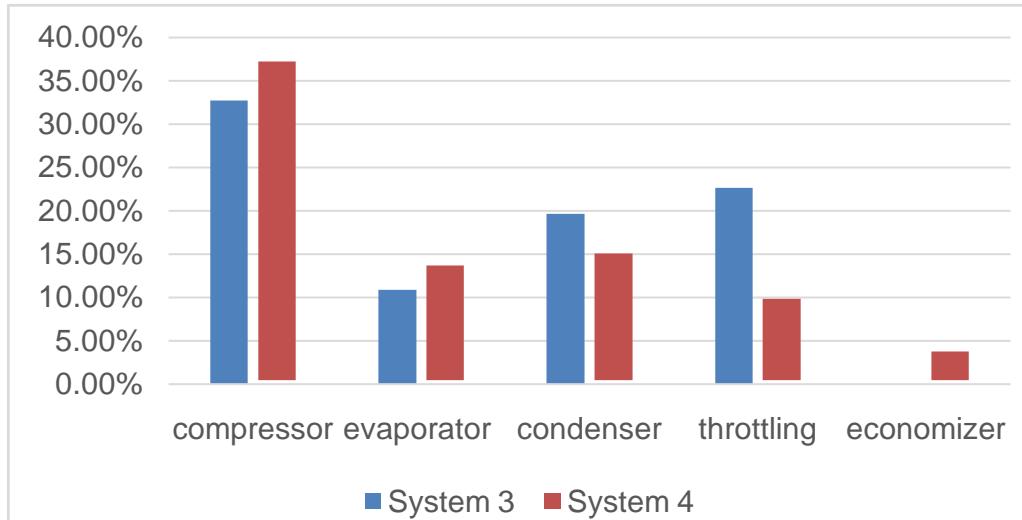 Figure 6: Part of Compression Work (Energetic Losses) which is Necessary for Compensation of Entropy Production in System Elements for Refrigeration Plant before and after Optimization,% from Compression Work.
## III. CONCLUSIONS
Application of entropic and statistical method of the analysis during cooling plant operation allows increasing energy efficiency because we can get information about losses in different refrigeration plant components and take measures to increase their operation efficiency. It is possible to use regularly installed sensors.
The most appropriate value for comparison of energy efficiency different cooling plants is the degree of thermodynamic efficiency.
Changing control algorithm of condensation pressure from maintenance of the fixed setting to floating setting allow to increase the degree of thermodynamic efficiency on $32.9\%$ and decrease power consumption on $36.7\%$. Power consumption was measured by using special sensors.
The degree of thermodynamic efficiency refrigeration plant working with refrigeration cycle with economizer is higher than the degree of thermodynamic efficiency refrigeration plant working with single stage refrigeration cycle on $30.37\%$. Date was received during operation of real refrigeration plants.
Generating HTML Viewer...
References
10 Cites in Article
A Arkharov (2014). Fundamentals of cryology. Entropy and statistical analysis of low-temperature systems.
A Arkharov,V Shishov (2014). Analysis of lowtemperature refrigeration cycles using the and statistical analysis.
V Shishov,V Portyanikhin,M Talyzin (2015). Statistical Entropy Analysis of Subcritical Refrigeration Cycles with Ejector as an Expansion Device.
A Arkharov,V Shishov,M Talyzin (2016). Entropy and statistical analysis of low-temperature refrigeration cycles and based on it choice of an optimal cold supply system for a shop.
G Paniagua,J Braun,T Meyer,V Athmanathan,S Roy (2008). AN OASIS OF PURE AEROTHERMAL DILEMMAS:INTEGRATING TURBINES WITH ROTATING DETONATION COMBUSTOR.
A Arkharov,V Shishov,M Talyzin (2017). Сomparison of CO2 transcritical cycles with conventional refrigerants cycles applied for cold supply of mercantile business using an entropy and statistical analysis.
M Zdobnov,N Lavrov,V Shishov (2018). Analysis of losses in the mixing section of the central air conditioner using the entropy-statistical method.
A Arkharov,V Shishov,M Talyzin (2017). Entropy and statistical analysis of low-temperature transcritical cycles on СО2.
A Arkharov,V Semenov,S Krasnonosova (2016). An Entropy-Statistical Analysis of a Natural Gas Liquefaction Plant with External Nitrogen Cooling Cycle.
A Arkharov,V Semenov,S Krasnonosova,S Lebedev,V Kul’byakin (2016). Statistical Entropy Analysis and Experimental Study of LNG Plant with Precooling at –70°C.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Talyzin Maksim Sergeevich. 2026. \u201cEntropic and Statistical Analysis of Existed Refrigeration Plant for Retail\u201d. Global Journal of Research in Engineering - A : Mechanical & Mechanics GJRE-A Volume 23 (GJRE Volume 23 Issue A4).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.