This study aimed to evaluate the possible histological and ultrastructural changes of liver induced by methomyl pesticide exposure and estimation the possible protective effect of ginger extract for hepatic damage in rats. Ginger is used worldwide primarily as a spicy condiment and of the herbal sources of natural protection from contamination and oxidative stress thus play an important role in chemoprevention of liver diseases. Methomyl is one of the most frequently prescribed pesticides, that are used as a pesticide. It is characterized as a highly toxic compound and has been reported to cause multiple organs damage. Fifteen male albino rats were allocated into 5 main groups. (3 rats in each); one served as control and the four remained groups were for different treatments. GC-MS analysis of ginger extract revealed the content of gingerol, quercetin (3.20%), limonene and zingiberene. Treatment of methomyl treated rats with ginger extract maintained serum ALP, AST& ALT levels and reduced the damage effect with protective efficacy against pesticide induced hepatotoxicity, which appears also more effective than its therapeutic application.
## I. INTRODUCTION
Plants have been the major source of drugs for the treatment of various diseases in many ancient systems of medicine in the world. Ginger is an underground rhizomes of plant Zingiber officinale belonging to the family Zingiberaceae which is widely consumed as spice for the flavoring of foods (Ajith et al., 2007). It has been reported that ginger and its extracts possess some pharmacological activities including hypoglycemic, insulinotropic and hypolipidemic in human and in experimental animals (Kondeit et al., 2005).
The antioxidants in ginger include gingerols, shogaols, monoterpenes, sequiterpenes, some phenolic derivatives and other phytochemicals which are responsible for their pharmacological activities (Li et al., 2001).
Many previous studies investigated the hepatoprotective effects of ginger extract against liver toxicity induced by ethanol, carbon tetrachloride, bromobenzene and acetaminophen with significant decrease in the level of ALT and AST (Mallikarjuna et al., 2008; El-Sharaky et al., 2009).
The present work was conducted to study the effect of ginger on the liver tissues& serum profile enzymes.
## II. MATERIALS & METHODS EXPERIMENTAL DESIGN
Fifteen male albino rats were allocated into 5 main groups. 3 rats in each,
Group 1: served as control were administered $1 \mathrm{ml}$ once daily of Tween 80, vegetable oil and distilled water.
Group 2: received ginger extract in a dose $1 / 10 \, \text{LD}_{50}$ (1250 mg/kg /day orally) Zaki et al (2022).
Group 3: animals received methomyl orally in a dose $1 / 10\ \mathrm{LD}_{50}$ (1.25 mg/kg /dayorally) Araki et al (1982).
Groups 4: animals received ginger extract simultaneously with methomyl after two hours in the previous doses.
Group 5: (treatment) received ginger extract for 2 hours after methomyl exposure
The treatments were given for rats for 28 days, then, rats were sacrificed by ether anesthesia and specimens from liver were taken for light and electron microscopic examination. Gas chromatography-mass spectrometry (GC-MS) analysis. The chemical composition of water extract of ginger roots was performed using Trace_GC Ultra-ISQ mass spectrometer(Thermo-Scientific, Waltham, MA) with a direct capillary column TG-5MS (30m_0.25mm_0.25 mm film thickness). The column oven temperature was initially held at 40_C, and then increased by 5_C/min to 280_C. The injector and detector (MS transfer line) temperatures were kept at 250_C. Helium was used as a carrier gas at a constant flow rate of 1 ml/min. Extract derivatization was done using BSTFA/TMCS (80:20, v:v) for 1 h at 70_C, after evaporation to dryness of dichloromethane/methanol mixture. The resulting solution was dried and then dissolved in hexane. The solvent delay was 2 min and diluted samples of 1 ml were injected automatically using Auto sampler AS3000 (Thermo-Scientific, Waltham, MA) coupled with GC in the split less mode. El mass spectra were collected at 70 eV ionization voltages over the range of m/z 50–650 in full scan mode. The ion source and quadrupole temperatures were set at 200_C and 150_C, respectively. The components were identified by comparison of their retention times (Rt) and mass spectra with those of WILEY 09 (Flavor & Fragrance Natural & Synthetic Compounds) and NIST 11 (National Institute of Standards and Technology, Gaithersburg, MD) Mass Spectral databases.
### a) Plant material
The ginger rhizomes were collected from the Faculty of Pharmacy, Heliopolis University for Sustained Development, Medicinal Plant Reserve. It was identified and authenticated by, Medicinal plants branch at the national research center (Latin name: Zingiber officinale; plant part: Rhizome).
### b) Methomyl
Technical grade methomyl (90% active ingredient) was obtained from (Tabouk) Pesticide Company, Egypt.
### c) Animals used
Healthy adult male albino rats (120-180 gm) were obtained from laboratory animal breeding unit (Faculty of Medicine, Zagazig University). The rats were kept in metal cages during the whole experimental period under hygienic condition, fed on well balanced ration and provided water ad-libitum, through the experiment. The light system was 12/12 hrs. dark/night cycle. The rats were accommodated to laboratory conditions for two weeks before the experiment.
Aqueous ginger extract was prepared from available ginger (Zingiber officinale) roots. The ginger roots were peeled on crushed ice, and $(100\mathrm{gm})$ of ginger was cut into small pieces and homogenized in 75 ml of cold, sterile $0.9\%$ NaCl in the presence of crushed ice. The homogenization was carried out in a blender at high-speed bursts for total 15 minutes.
## i. Blood samples collection and preparation of plasma
The blood was collected by the retina and allowed to clot for 30 min. at room temperature. The clotted blood was then centrifuged at 3500 RCF for 30 min. The serum was separated and stored at $-25^{\circ}\mathrm{C}$ until protein and enzyme analyses were performed.
### a. Determination of alanine amino transferase (ALT) and aspartate amino transferase (AST)
ALT and AST were determined colorimetrically according to the method of Reitman, (1957). The reaction mixture consisted of $1\mathrm{ml}$ of a mixture of phosphate buffer $(\mathsf{pH}7.2)$, $0.2\mathrm{mM}\alpha$ -ketoglutaric and $200\mathrm{mM}$ L-aspartate. Incubate for exactly $30\mathrm{min}$, add $1\mathrm{ml}$ of $0.001\mathrm{M}2,4$ -dinitrophenyl hydrazine, wait for at least $30\mathrm{min}$, and then $10\mathrm{ml}$ of $0.4\mathrm{N}$ NaOH were added. The optical density of the produced brown color is measured after 5 minutes, using a spectrophotometer at $520\mathrm{nm}$.
# b. Determination of
The activities of acid and alkaline phosphatases were determined using the method of Powell and Smith, (1954).
In this method, the phenol released by enzymatic hydrolysis of disodium phenylphosphate reacts with 4-aminoantipyrine,and by the addition of potassium ferricyanide, the characteristic brown color is produced. The reaction mixture consisted of $1\mathrm{ml}$ carbonate buffer $(\mathsf{pH}4.5$ & 10.4), $1\mathrm{ml}$ of $0.01\mathrm{M}$ disodium phenyl phosphate (substrate), and $0.1\mathrm{ml}$ sample, and then incubate for exactly $30\mathrm{min}$. At the end of incubation period $0.8\mathrm{ml}$ of $0.5\mathrm{N}$ NaOH was added to stop the reaction. Then add $1.2\mathrm{ml}$ of $0.5\mathrm{N}$ $\mathrm{NaHCO}_3$, followed by the addition of $1\mathrm{ml}$ of 4-aminoantipyrine solution ( $1\%$ ) and $1\mathrm{ml}$ potassium ferricyanid ( $0.5\%$ ). The produced color was measured immediately at $510~\mathrm{nm}$. The enzyme activity is expressed by unit (U), where 1 unit hydrolyze $1.0\mu$ mole of p-nitrophenyl phosphate per minute at $37^{\circ}\mathrm{C}$, and pH 10.4.
### d) Statistical analysis
The significance differences were determined by analysis of variance (ANOVA). The significance of various treatments was evaluated by Duncan's multiple range tests $(p < 0.05)$. Data were subjected to statistical analyses using the software package Costat® Statistical Software (2005) a product of Cohort Software, Monterey, California, USA. The values of each measurement of the tested parameters were recorded as mean of five readings i standard error. Statistical analysis was carried out using simple one-way analysis of variance (ANOVA) test, using spss software windows version 17 (SAS, 2001). A probability of $P < 0.05$; and $P < 0.0$ as the level of significance unless stated otherwise. Statistical significant differences among all treatments were carried out by least significant differences (LSD).
## III. RESULTS
Chemical composition of GE The GC-MS analysis of GE revealed that it contains phenolics, alkaloids, flavonoids, tannins, anthraquinones, terpenoid, and steroids. The identified bioactive components of GE are listed in Table 1, with their respective RT and percent composition (area%), where the most important substances are gingerol, quercetin, DL-limonene, ar-curcumene, zingiberene, b-sesqui
phellandrene, linalool, pyrazine, and 1,8-terpin hydrate. The major identified compound was the gingerol $(7.09\%)$, while from total ion chromatogram (TIC), we note that the flavonoid quercetin has been appeared at multiple RTs with total percent composition of $3.2\%$.
Table (1): Bioactive components of aqueous extract of ginger roots as determined by using gas chromatography-mass spectroscopy (GC-MS)
<table><tr><td>Compound name</td><td>Molecular formula</td><td>Molecular weight</td><td>Retention time</td><td>Area%</td></tr><tr><td>Nerolidol</td><td>C15H26O</td><td>222</td><td>29.3</td><td>0.05</td></tr><tr><td>Lucenin</td><td>C27H30O16</td><td>610</td><td>33.75</td><td>0.06</td></tr><tr><td>Clionasterol</td><td>C40H56</td><td>414</td><td>33.55</td><td>0.09</td></tr><tr><td>Curcumene</td><td>C15H22</td><td>202</td><td>28.38</td><td>0.09</td></tr><tr><td>Cineole</td><td>C10H18O</td><td>154</td><td>24.86</td><td>0.10</td></tr><tr><td>Phenol</td><td>C9H12O2</td><td>152</td><td>22.88</td><td>0.11</td></tr><tr><td>Sesquiphellandrene</td><td>C15H24</td><td>204</td><td>29.39</td><td>0.12</td></tr><tr><td>Zingiberene</td><td>C15H24</td><td>204</td><td>28.69</td><td>0.18</td></tr><tr><td>Isochiapin</td><td>C19H22O6</td><td>348</td><td>41.21</td><td>0.18</td></tr><tr><td>Dihydrostilbene</td><td>C16H18O4</td><td>275</td><td>41.97</td><td>0.20</td></tr><tr><td>Quercetin</td><td>C18H16O7</td><td>344</td><td>43.03</td><td>3.41</td></tr><tr><td>Pyrazine</td><td>C5H6N2</td><td>95</td><td>8.35</td><td>12.71</td></tr><tr><td>Hydroxylinalool</td><td>C10H18O2</td><td>170</td><td>23.52</td><td>1.15</td></tr><tr><td>Terpinhydrate</td><td>C10H20O2</td><td>171</td><td>24.29</td><td>1.09</td></tr><tr><td>Rosifoliol</td><td>C15H24O</td><td>220</td><td>36.34</td><td>0.74</td></tr><tr><td>Limonene</td><td>C10H16</td><td>140</td><td>15.17</td><td>0.27</td></tr><tr><td>Gingerol</td><td>C17H26O4</td><td>294</td><td>45.17</td><td>0.79</td></tr></table>
Table (10): Effect of tested materials on the liver enzymes esterase of albino rats
<table><tr><td></td><td>G1 Control</td><td>G2 Ginger</td><td>G3 Methomyl</td><td>G4 Ginger& methomyl</td><td>G5 Methomyl & ginger</td></tr><tr><td>ALT (U/L)</td><td>134.16 ±1.211</td><td>124.12 ±5.21</td><td>152.15 ±1.379*</td><td>142.4 ±1.455**</td><td>144.27 ±1.205</td></tr><tr><td>AST (U/L)</td><td>35.45 ±0.684</td><td>40.56* ±5.96</td><td>73.32 ±1.215*</td><td>43.77 ±0.997**</td><td>65.55 ±1.990*</td></tr><tr><td>ALP (U/L)</td><td>375.16 ±0.921</td><td>395.22 ±11.54*</td><td>655.65 ±1.655*</td><td>511.28 ±1.568*</td><td>579.52 ±4.403*</td></tr></table>
The serum enzyme activities in rats fed supplemented with ginger extract; hot and cold for 28 days are given in Table 3. All treated groups showed significant decrease of serum aspartate and alanine aminotransferase, gamma glutamyl transferase and alkaline transferase activities as compared with control group.
The liver enzymes levels were reduced by the same amount after pesticide withdrawal with or without GE treatment, suggesting that GE does not exhibit metal-chelating ability and does not provide hepato protection against methomyl accumulation. Additionally, the markers of liver function remained persistently high, particularly AST, at early exposure periods in rats treated with methomyl, which may be indicative of hepatic damage (Omobowale et al., 2014). GE initially improved transaminases activities (Farag et al., 2010) but failed to restore their activity to normal towards the end of the exposure period. Thus, GE could protect against the early methomyl induced hepatotoxicity. Administration of ginger improved liver function s as it reduced liver enzymatic activities. These results were in accordance with that of Mallikarjuna et al. (2008) who showed that administration of ginger ethanolic extract $(200\mathrm{mg / kg})$ orally from day 15 to day 21 along with country-made Liquor (CML) produced significant lowering of AST, ALT, ALP and tissue lipid peroxide levels. Treatment of ginger significantly decreased serum urea and increased serum creatinine concentration since, ginger contain polyphenols and flavonoids that influence removing certain waste products from plasma. These results agree with Ajith et al., (2007) who reported that the presence of polyphenols and flavonoids in ginger extract might be responsible for the antioxidant nephroprotective activities and the reduction of serum urea and creatinine levels.
### a) Histopathological Work Up
Fixation and tissue processing: The formalin preserved liver, specimen was processed in an automated tissue processor. The processing consisted of an initial 2 step fixation and dehydration. Fixation comprising tissue immersion in $10\%$ buffered formalin for 48 hours, followed by removal of fixative in distilled water for 30 minutes. Dehydration was then carried out by running the tissues through a graded series of alcohol (70%, 90%, and $100\%$ ). The tissue was initially exposed to $70\%$ alcohol for 120 minutes followed by $90\%$ alcohol for 90 minutes and then two cycles of absolute alcohol, each for one hour. Dehydration was then followed by clearing the samples in several changes of xylene. It consisted of tissue immersion for an hour in a mixture comprising
50% alcohol and 50% xylene, followed by pure xylene for one and a half hour. Samples were then impregnated with molten paraffin wax, then embedded and blocked out. Paraffin sections (4-5 um) were stained with hematoxylin and eosin, (Suvara et al., 2012) Stained sections were examined for circulatory disturbances, inflammation, degenerations, apoptosis, necrosis, and any other pathological changes in the examined tissues.
### b) Histopathologic Finding
## i. Group 1. (Control free)
Liver: Examined serial sections from liver revealed normal hepatic parenchyma with preserved lobular pattern, portal triades and associated structures (portal vein, hepatic artery, hepatic vein, bilsductiles and lymphatics). Central vein sinusoids and hepatic cords were apparently normal. Hepaticlobules were separated by fine fibrous stromal connective tissue.(Fig.1).
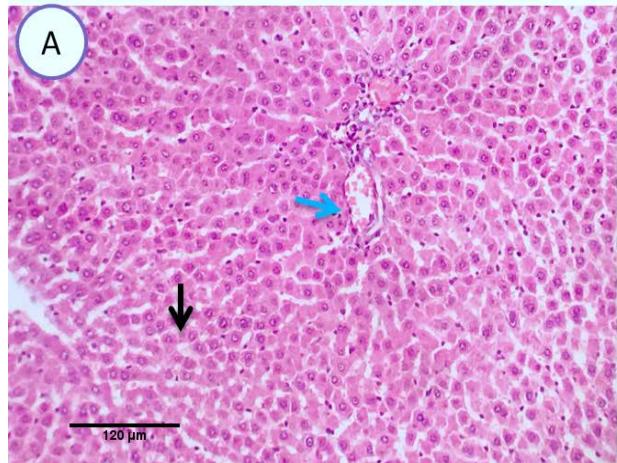
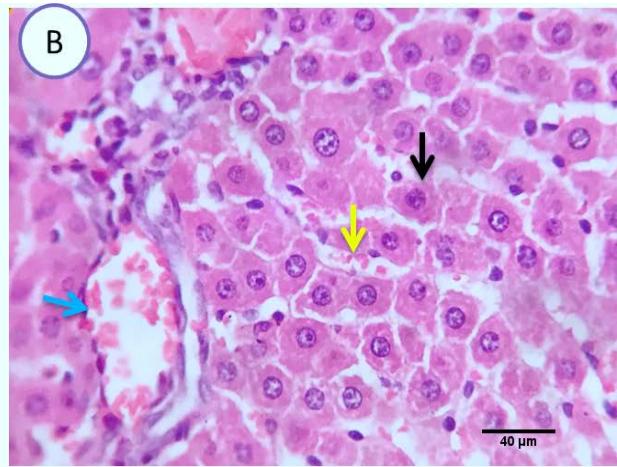 Fig. 1: Photo-micrograph from liver, control group, showing normal hepatic parenchyma with preserved lobular pattern, central veins (blue arrow), Sinusoids and hepatic cords (black arrow). Scale bars 120, 40 um
## ii. Group 2 Ginger administration
Liver: Examined serial sections denoted apparently normal hepatic parenchyma, lobular arrangement, portal triads structures, vascular tributaries and biliary tree, however some sections showed vascular dilatation and mild portal round cells aggregation. (Fig.4).
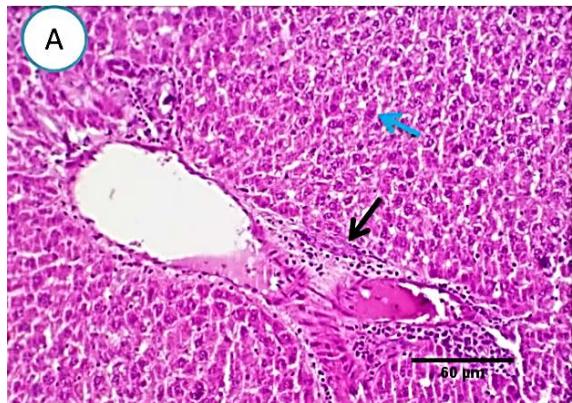
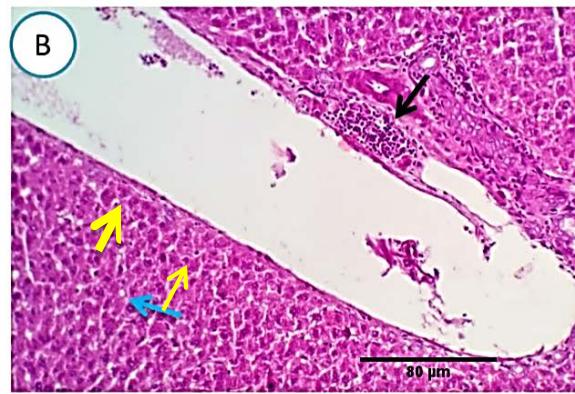 Fig. 2: Photomicrograph of liver (Group.G) showing normal hepatic parenchyma(blue arrows), vascular dilatation(yellow arrows)and mild portal round cells aggregation(black arrows). Scale bars 60, 80 um
Here, as a proof of concept, we used a mouse model to show that orally from ginger extracts resulted in protecting mice against methomyl-induced liver damage.
### c) Group3 (Methomyl pesticide)
Liver: Examined sections from liver of this group denoted moderate portal biliary proliferation, congestion of portal blood vessels, round cell infiltration, multifocal interstitial lymphocytic and macrophages aggregations replacing previous necrotic patches beside degenerative changes in a few hepatocytes. (Fig.4).
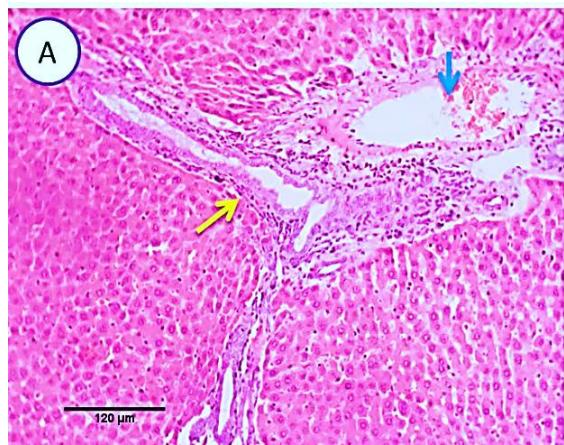
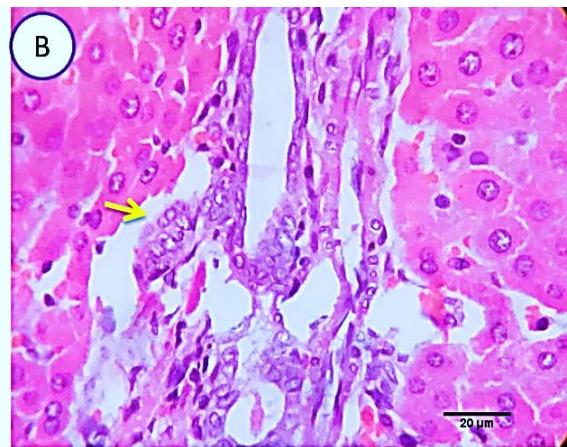 Fig. 3: Photo-micrograph from liver, group (2), showing, portal biliary proliferation (yellow arrow), congestions of portal blood vessels (blue arrow) and round cell infiltration beside degenerative changes in a few hepatocytes. Scale bars 120, 20 um.
### d) Group 4 (Ginger protection followed by methomyl administration)
Liver: Sections from liver of this group denoted moderate portal biliary proliferation, congestion of portal blood vessels, round cell infiltration, multifocal interstitial lymphocytic and macrophages aggregations replacing previous necrotic patches beside degenerative changes in a few hepatocytes. (Fig.10)
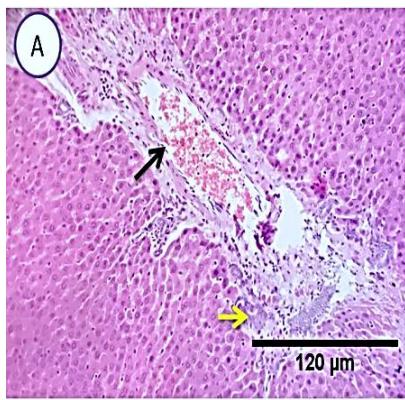
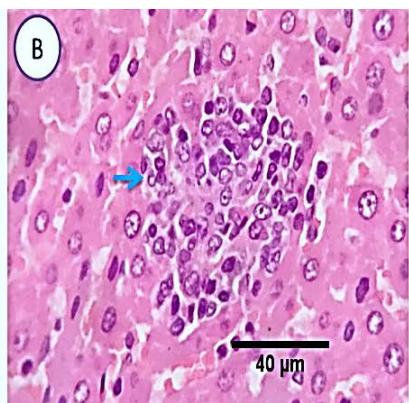
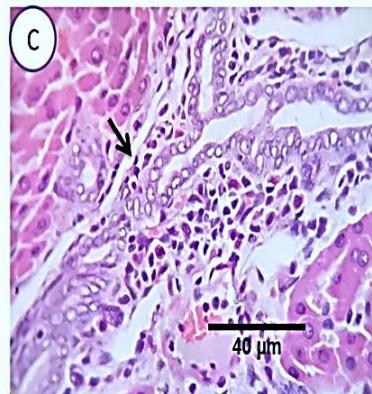 Fig. 4: Photo-micrograph from liver, group (4), showing, portal biliary proliferation (A, yellow arrow, B, black arrow), congestions of portal blood vessels(A, black arrow), round cell infiltration and interstitial lymphocytic and macrophages aggregations replacing previous necrotic patches (B, blue arrow). Scale bars 120, 40, 40 um.
### e) Group 5 (Ginger treatment after methomyl administration)
Liver: Examined sections from liver of this group denoted moderate portal biliary proliferation, congestion of portal blood vessels, round cell infiltration, multifocal interstitial lymphocytic and macrophages aggregations replacing previous necrotic patches beside degenerative changes in a few hepatocytes.(Fig.7).
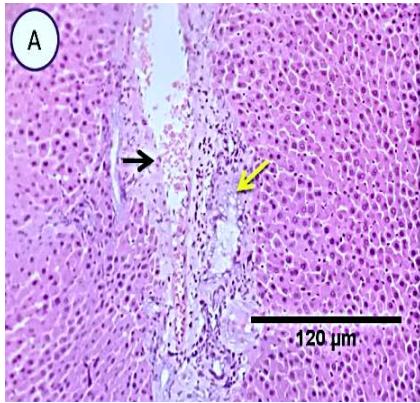
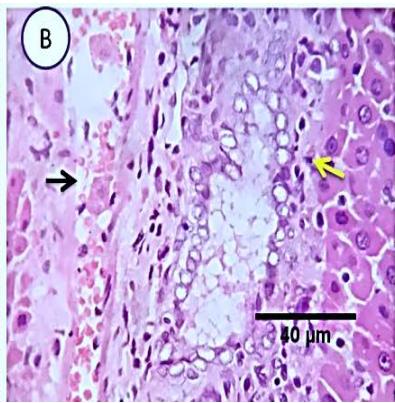
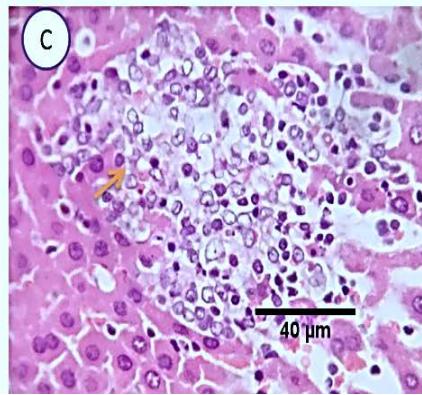 Fig. 5: Photo-micrograph from liver, group (3), showing, portal biliary proliferation (yellow arrow), congestions of portal blood vessels (black arrow) and round cell infiltration and interstitial lymphocytic and macro-phages aggregations replacing previous necrotic patches (orange arrow). Scale bars 120, 40, 40 um.
## IV. CONCLUSION
Ginger extract appear to be highly effective in improving the toxic effects caused by pesticide, and the use of ginger was beneficial in lowering liver enzymes and has hepatoprotective effect.
Generating HTML Viewer...
References
21 Cites in Article
Suvarna Kim,S,Christopher Layton,Bancroft John,D (2012). Bancroft's Theory and Practice of Histological Techniques.
(2022). Unknown Title.
T Ajith,V Nivitha,S Usha (2007). Zingiber officinale Roscoe alone and in combination with α-tocopherol protect the kidney against cisplatin-induced acute renal failure.
W Li,X Zhang,Y Wu (2001). Antiinflammatory effect and mechanism of proanthocyanidine from grape seeds.
K Mallikarjuna,P Sahitya Chetan,K Sathyavelu Reddy,W Rajendra (2008). Ethanol toxicity: Rehabilitation of hepatic antioxidant defense system with dietary ginger.
Stanley Reitman,Sam Frankel (1957). A Colorimetric Method for the Determination of Serum Glutamic Oxalacetic and Glutamic Pyruvic Transaminases.
C Huang,J Horng,Yin Mc (2004). Antioxidative and antiglycative effects of six organosulfur compounds in low density lipoprotein and plasma.
Temidayo Omobowale,Ademola Oyagbemi,Akinleye Akinrinde,Adebowale Saba,Oluwabusola Daramola,Blessing Ogunpolu,James Olopade (2014). Failure of recovery from lead induced hepatoxicity and disruption of erythrocyte antioxidant defence system in Wistar rats.
R Kondeti,M Korivi,N Kesireddy,C Kuo,S Kiesireddy (2011). Protective effect of dietary ginger on antioxidant enzymes and oxidative damage in experimental diabetic rat tissues.
Oecd Guideline For Testing Of Chemicals (2001). Test No. 425: Acute Oral Toxicity: Up-and-Down Procedure.
A Farag (2005). Deterimenral effects of certain pesticides on white albino rat (Rattus norvrgicus Boric).
(2005). Microcomputer Program Analysis Version.
A El-Sharaky,A Newairy,M Kamel,S Eweda (2009). Protective effect of ginger extract against bromobenzene-induced hepatotoxicity in male rats.
M Powell,M Smith (1954). The Determination of Serum Acid and Alkaline Phosphatase Activity with 4-Aminoantipyrine (A.A.P.).
Giacomo Certini (2005). Effects of fire on properties of forest soils: a review.
Yun-An Chen,Pao-Wen Grace Liu,Liang-Ming Whang,Yi-Ju Wu,Sheng-Shung Cheng (2020). Effect of soil organic matter on petroleum hydrocarbon degradation in diesel/fuel oil-contaminated soil.
Dilfuza Egamberdieva,Stephan Wirth,Undine Behrendt,Elsayed Abd_Allah,Gabriele Berg (2016). Biochar Treatment Resulted in a Combined Effect on Soybean Growth Promotion and a Shift in Plant Growth Promoting Rhizobacteria.
Dagmar Etkin (2001). ANALYSIS OF OIL SPILL TRENDS IN THE UNITED STATES AND WORLDWIDE.
S Gan,E Lau,H Ng (2009). Remediation of soils contaminated with polycyclic aromatic hydrocarbons (PAHs).
G Giovannini,S Lucchesi,M Giachetti (1988). EFFECT OF HEATING ON SOME PHYSICAL AND CHEMICAL PARAMETERS RELATED TO SOIL AGGREGATION AND ERODIBILITY.
R Haynes,R Swift (1989). Effect of rewetting air-dried soils on pH and accumulation of mineral nitrogen.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Marwa. M. Zaki. 2026. \u201cPossible Protective Role of Ginger Extract on Methomyl Induced Hepatotoxicity in Adult Male Albino Rats\u201d. Global Journal of Science Frontier Research - B: Chemistry GJSFR-B Volume 23 (GJSFR Volume 23 Issue B1).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Subject: Global Journal of Science Frontier Research - B: Chemistry
Authors:
Marwa. M. Zaki, Amir. E. Abo Elhassan, Eman. S. M. El Zahid, Ahmed, Ahmed. M. I. Hejab, Ayman. H. Kamel, Nedal M. Fahmy, Tarek. R. Amin, Saad. S.M. Hassan (PhD/Dr. count: 0)
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.