## I. INTRODUCTION
Osteoarthritis (OA) is a common disorder in humans and animals, and its prevalence increases with age. It can reach more than $80\%$ of animals eight years of age or older [1]. It is a debilitating and painful disease, involving local tissue damage as well as local and systemic inflammatory responses. It leads to decreased mobility and impacts the quality of life of both dogs and owners [2-4]. Preventing cartilage damage and oxidative stress, and maintaining a balanced immune response, are therefore key to disease prevention and to slowing down its progression. Management of OA in dogs usually involves nonsteroidal anti-inflammatory drugs despite their side effects. Many efforts have been put over the last decades into finding new active ingredients and nutraceuticals to maintain joint health and mobility [3,5].
Movoflex® Soft Chew (Virbac, USA) is a nutritional supplement that can help support dogs' mobility and joint health [2]. It contains a unique combination of ingredients that are well tolerated by dogs and that have shown effectiveness in supporting joint health, either individually or in combination: eggshell membrane (ESM), astaxanthin, hyaluronic acid (of high molecular weight), Boswellia serrata extract, and vitamin D3 [2]. Now, the advanced formula also contains hyaluronic acid (HA) of low molecular weight and krill oil as a source of omega-3 fatty acids.
Eggshell membrane contains different molecules found in joints, including glucosamine and glycosaminoglycans, elastin, collagen, and hyaluronic acid. It also contains different amino acids (mainly proline, glutamic acid, and glycine) that can help support protein synthesis [6]. Eggshell membrane has shown beneficial effects on joints and mobility, in humans with osteoarthritis [7-14] and in dogs [2,15,16].
Astaxanthin is a naturally occurring carotenoid with potent antioxidant properties (more powerful than other carotenoids and 500 times more powerful than vitamin E) [17-19]. Astaxanthin limits the production of mitochondrial reactive oxygen species (ROS) and modulates their effects, scavenging free radicals and decreasing the development of nitric oxide [18,19]. Astaxanthin has also been shown to stimulate the immune response in animals and humans [20,21]. The astaxanthin in Movoflex® Soft Chews comes from Haematococcus pluvialis, a unicellular microalga, one of the safest and richest sources of natural astaxanthin [18,19].
Boswellia serrata extracts, from the resin of the tree, have been used for joint health for a long time, notably in ayurvedic medicine. Several studies have shown its good tolerance and beneficial effects on joints in humans and animals [22,23], including in dogs [24]. Boswellic acids like 3-O-Acetyl-11-keto-beta-boswellic acid (AKBA) and 11-keto-b-boswellic acid (KBA) would indeed help control the immune system by inhibiting the 5-lipoxygenase (5-LOX) and would help improve pain and joint function [22,23]. Other mechanisms may also be involved.
Cholecalciferol is the inactive form of vitamin D3. It requires sequential hydroxylations in the liver and kidney to reach its physiologically active form (calcitriol or 1,25-dihydroxyvitamin D3). It is a steroidal hormone known to regulate calcium and phosphorus homeostasis in the body, as well as bone metabolism, among other properties [25,26]. The blood level of vitamin D is positively correlated with good musculoskeletal health and negatively correlated with OA or mobility reduction [25-29]. It has been shown in some studies that vitamin D supplementation can help patients with OA [25,30,31]. In dogs, vitamin D supplementation can help maintain bone health [26,29].
Hyaluronic acid (HA) is a large glycosaminoglycan consisting of repeating disaccharide units of N-acetylglucosamine and glucuronate. It is found in several tissues, including in the articular cartilage and synovial fluid. It is highly involved in the lubrication and viscoelasticity of the synovial fluid and plays a significant role in joint mobility [32,33]. Therefore, administrating HA has shown beneficial effects in OA dogs [32,33]. Since OA is associated with HA degradation over time, the HA administered was, mainly, of high molecular weight (MW) and usually injected directly into the joint [33]. However, oral forms can also be used with good bioavailability and efficacy [34,35]. A study has shown that HA of high MW can be found in dogs' joints after oral administration [36]. On the other hand, lower MW HA, which should have a higher oral bioavailability than the high MW HA [34,37], could bring the elements required for the HA synthesis and help initiate the restorative processes [33,38]. A recent study in an animal model even showed that the lower molecular weight HA would be more effective than the higher molecular weight HA for cartilage repair [38].
Krill are shrimp-like crustaceans that are small (about the size of a paperclip) but form abundant and dense swarms in polar seas. The oil extracted from the main Antarctic species, Euphausia superba, is naturally rich in omega-3 polyunsaturated fatty acids (or PUFAs), mainly eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) [39]. Contrary to what is found in fish oil (mainly triacylglyceride-bound PUFAs), PUFAs from krill oil come primarily in the phospholipid (PLs)-bound form. Phosphatidylcholine is a major PL in krill oil. On top of bringing essential nutrients like choline, PLs also seem to be a more effective delivery form of PUFAs, with higher bioavailability [39,40]. The health benefits of krill oil have been demonstrated in several studies, including those focusing on joint health [39,41,42]. It has also been suggested that PLs, like those in krill oil, could increase the bioavailability of astaxanthin and hyaluronic acid [43,44]. Combining these three ingredients has shown beneficial effects on joint and inflammation in different studies [45,46] and is now commercially available for humans (FlexPro MD®, Valensa).
The krill used to make the oil in Movoflex® advanced Soft Chews (Virbac, USA) is fished using an eco-harvesting technology to harvest krill in a sustainable and environmentally friendly way. This technology reduces the risk of by-catch of other species to almost zero.
We hypothesized that adding krill oil and a lower molecular weight HA could increase the performance of Movoflex® Soft Chews in dogs and remain well tolerated. We first tested its tolerance, in kenneled dogs, before testing its acceptability and perception of effectiveness on mobility, with dog owners already using the current Movoflex® Soft Chews.
## II. MATERIAL AND METHODS
### Tolerance
The tolerance of the chew was tested in 16 beagle dogs (6 months to 2 years old, $>5\mathrm{kg}$ ) receiving either one or five times the daily recommended number of chews, according to Virbac's standard procedures. The project was approved by the Virbac Ethical Committee and by French Authorities under authorization number 2021011113595530#28915.
Eight dogs received one Movoflex® Soft Chew size S per day (as recommended) for small dogs $< 18$ kg (1x group), and eight dogs received five chews per day (5x group), during 28 days. Another group of four dogs did not receive any chew (control group). A 14-day acclimation phase was included before administering the chews on Day 0. Dogs were housed in pairs and fed Veterinary HPM® Adult Neutered Dog (Virbac). They were observed daily, and a complete clinical examination, including body weight and body temperature measurements, was performed once a week from Day -14 to 28. Food consumption was checked daily. An adjustment of the food ration (-10g of kibbles daily) was made for the dogs receiving five chews per day (6.27 kcal/chew). Feces were examined and scored daily based on a simple scale (1- molded stools, 2- not molded stools, 3- liquid stools) during the whole study period. Blood samples were taken at the start and the end of the study for standard hematology [RBC, HGB, HCT, MCV, MCH, MCHC, RETIC, WBC, NEU, EOS, BASO, LYM, MONO and PLT - ProCyteDx Hematology Analyser (IDEXX)] and blood chemistry [BUN; CREA; TP; ALB; GLOB; GLU; ALKP; ALT; AST; Calcium; Chloride; Potassium; Magnesium; Sodium; Phosphate - Catalyst Chemistry Analyser (IDEXX)] analysis. Urine samples were also taken on fasted and sedated animals by ultrasound-guided cystocentesis in females and by vesical catheterization in males, when possible (no empty bladder) at the beginning and the end of the study for pH analysis. Urinary pH was measured with a pHmeter (Mettler Toledo Seven Multi pH meter) quickly after urine collection.
Perceived effectiveness
Movoflex® Advanced Soft Chews were provided to 78 owners of dogs, in the USA, already using Movoflex® Soft Chews before the study. Selected dogs had to be five years or older and included 26 small dogs weighing less than $18\mathrm{kg}$ ( $<40\mathrm{Ibs}$ ), 26 medium dogs between 18 and $36\mathrm{kg}$ (40-80 Ibs) and 26 large dogs weighing more than $36\mathrm{kg}$ ( $>80$ Ibs). The participants tested the chew over 30 days (1 chew per day - chew size according to body weight: 2g, 4g, or 6g, respectively). The participants had to fill out an online survey on Days 0, 7, and 30 to assess the product's physical properties (versus the current Movoflex® Soft Chews), palatability, reactions of the dog, and perception of effectiveness on their dog's mobility.
Every day, the owner had to score the reaction of the dog when given the chew, according to the following scale: 1- the dog ate the chew all at once; 2 - the dog ate part of it, stopped, then ate the rest; 3 - the dog ate some of it only; 4 - the dog played with it but did not eat; or 5 - the dog would not touch it spontaneously. To assess their dog's mobility, the owners had to describe five parameters:
- Dog's interaction with family and friends (1 - Happy and playful/Jumped around; 2 - Happy but reserved; 3 - Neutral; 4 - Sad, but reacted when called; 5 - Sad and didn't react much when called; 6 - Tended to isolate from others; 7 - Became aggressive when disturbed)
- Dog's ability to stand up after resting/lying down for some time (1 - Normal, stood up without difficulty; 2 - Slow, with some difficulty; 3 - Slow, with a lot of difficulty; 4 - Needed help getting up)
- Dog's ability to walk (1 - Normal, walks with ease; 2 - Normal, but needs to stop often; 3 - Difficult, needs to be encouraged; 4 - Difficult, refuses to walk)
- The way the dog walks (1 - Walks normally; 2 - Walks with stiffness; 3 - Unsteady when walking; 4 - Walks with lameness)
- Dog's ability to climb steps (1 - Normal; 2 - Slow; 3 - Made regular stops; 4 - Needed to be encouraged; 5 - Needed help/holding; 6 - Did not climb steps)
They also had to score from 0 (not difficult) to 10 (very difficult) the dog's ability/difficulty to walk, and the dog's ability/difficulty to stand up after some rest.
Statistics: Due to the ordinal nature of the data, Friedman's tests were used with the results obtained on Days 0, 7, and 30, for each mobility criteria. In case of significance (p\<0.05), Wilcoxon's signed-rank tests were used, using Bonferroni's correction to compare the data obtained on Day 0 versus Day 7 or Day 30 (p\<0.025 for significance).
## III. RESULTS
Tolerance
All dogs in the group receiving five chews per day (5x group) ate all their chews every day, while five dogs in the group receiving one chew per day (1x group) did not consume it every day (no forced administration). These same dogs also generally had a low food consumption, suggesting a general lack of appetite. Four of these dogs (who did not eat their chew more than 15 times) were excluded from the analysis while the other one was kept for analysis (only one day with no consumption). All dogs remained healthy throughout the study, and no product-related clinical signs were observed. Mild signs (ocular discharge, mild induced cough, licking granuloma) could be observed in some dogs, in all groups, before and after product administration, and were therefore unrelated to product administration. Digestive signs (vomiting or bloody stools) were observed twice in the 1x group animals during the administration period, with no link to product administration. Indeed, these signs can be observed sporadically in dogs with no clinical significance and were not observed in the 5x group.
The mean dog body weight and body temperature remained stable during the administration period (Supp. Table 1).
Concerning blood analysis (hematology, biochemistry and electrolytes), all mean values were within physiological ranges except for the globulin and MCHC, which were slightly lower than the reference range in all groups and at both time points (Supp. Table 2-5). All values remained pretty stable throughout the study. Some individual values could be slightly out of range but with no link to product administration (observed before and after the chew administration and in all groups).
Urine samples could be taken at both time points (Day -14 and Day 28) for the four control dogs, three dogs in the 1x group (excluding those who did not eat the chew every day), and six dogs in the 5x group. In all individuals, urine pH was between 5.11 and 6.82, with no relevant change between Day -14 and 28 (Supp. Table 6).
Therefore, no product-related clinical signs were observed during the study. Movoflex® Advanced Soft Chews are well tolerated by dogs, even when given five chews per day for 28 days, instead of one as recommended.
Perceived effectiveness
Among the 78 owners recruited on Day 0, 69 completed the study by filling in the questionnaires on the three days (Days 0, 7, and 30). The 69 owners included 21 owners of small dogs (\<18 kg or \<40 lbs), 25 owners of medium dogs (18-36 kg, or 40-80 lbs), and 23 owners of large dogs (>36 kg or >80 lbs).
Most owners reported giving the chew by hand (57% and 51% on Day 7 and Day 30, respectively) or on the bowl with food (51% and 41%, respectively). The remaining owners mostly gave it on the floor (13% and 9%, respectively).
During the trial, depending on the days, between $90\%$ and $96\%$ of dogs completely ate the chew. In $89\%$ of cases (over a total of 2070 chew presentations), the dogs ate the chew all at once, and in $4\%$ of cases, the dog ate part of it at first and the rest later (total consumption in $93\%$ of cases). The chew was partially taken in $1\%$ of cases, and in $7\%$ of cases the dog would not take the chew spontaneously. Five dogs $(7\%)$ three small dogs and two medium dogs) partially took the chew or refused to take the chew spontaneously more than $50\%$ of the time and were excluded from the analysis on mobility criteria (but were kept for the general appreciation).
By Day 30, most owners thought the tested chew was as easy $(83\%)$, or easier $(13\%)$ to give as/ than the previous Movoflex® Soft Chew and that palatability was similar (75%) or even better (16%).
Most owners also found the size, smell, color or shape of the new test chew were similar to the previous product they used.
Concerning the dog's mobility, $63\%$ of the 64 owners analyzed considered there was an improvement in their dog's ability to move around by Day 30 (34% on Day 7).
- Interaction with family and friends: on Day 0, $55\%$ of owners considered their dogs happy but reserved (score 2), while $42\%$ considered them happy and playful (score 1). On Day 30, this repartition changed to $34\%$ and $61\%$, respectively (Figure 1). The appreciation (score) given for this parameter was significantly different by Day 30 compared to Day 0 $(p < 0.01)$, with $23\%$ of owners giving a better (lower) score at the end of the study.
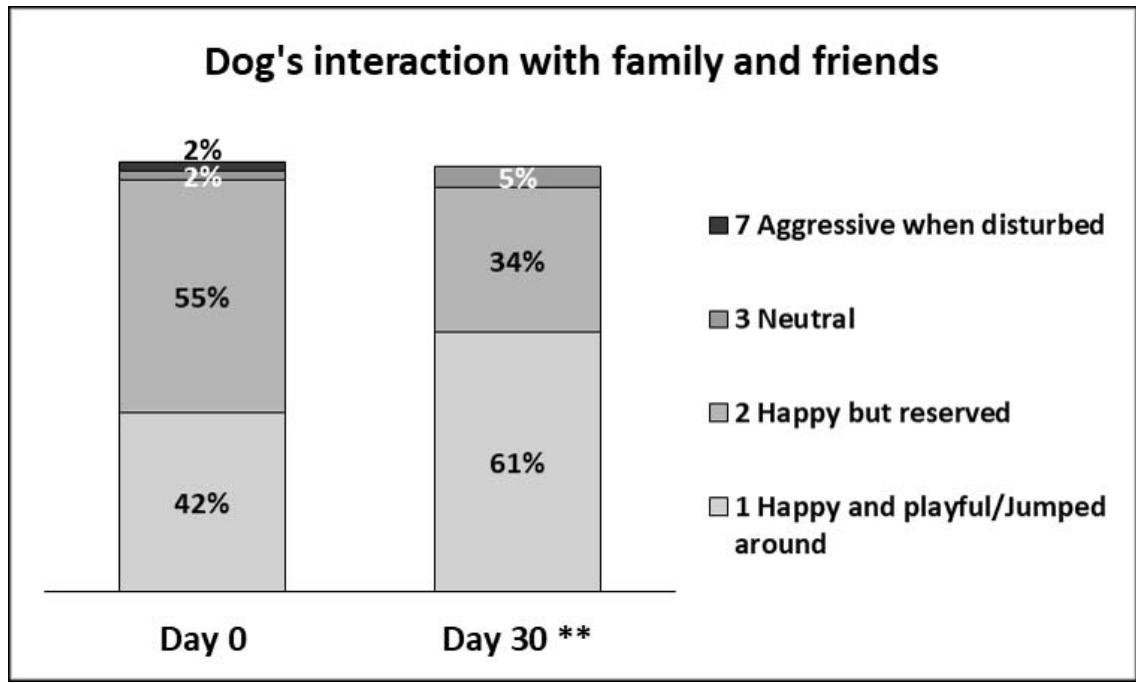 Figure 1: Dog's interaction with family and friends. The percentage of owners describing their dogs as: 1 - Happy and playful/Jumped around; 2 - Happy but reserved; 3 - Neutral; 4 - Sad, but reacted when called; 5 - Sad and didn't react much when called; 6 - Tended to isolate from others; or 7 - Became aggressive when disturbed, are depicted in this graph on day 0 (D0) and day 30 (D30). No owners reported their dogs to be 4, 5 or 6. A significant improvement was observed between Day 0 and Day 30 (\*\*, p\<0.01, Wilcoxon Signed rank test, n=64).
- Dogs walking ability
- Dog's ability to walk: most owners found their dog's ability to walk normal and with ease on Day 0 (61%) while some said their dogs needed to stop often (38%). By Day 30, more owners said their dogs walked with ease (78%, Figure 2). The appreciation (score) given for this parameter significantly improved by Day 30 ( $p < 0.01$ ), with $19\%$ of owners giving a better appreciation by then.
- The way of walking was described to involve stiffness by $55\%$ of owners on Day 0, while only $39\%$ said their dogs walked normally. By Day 30, more owners said their dogs walked normally (61%, Figure 2). The appreciation of this parameter significantly improved as of Day 7 ( $p < 0.01$; $20\%$ of owners gave a better appreciation) and was further improved by Day 30 ( $31\%$ of owners gave a better appreciation).
- When asked to score their dog's ability/difficulty to walk from 0 (very easy) to 10 (very difficult), the median (Q1-Q3) score significantly decreased from
3 (1-4) to 2 (0-3) between Day 0 and Day 30 $(p < 0.01$, Figure 2).
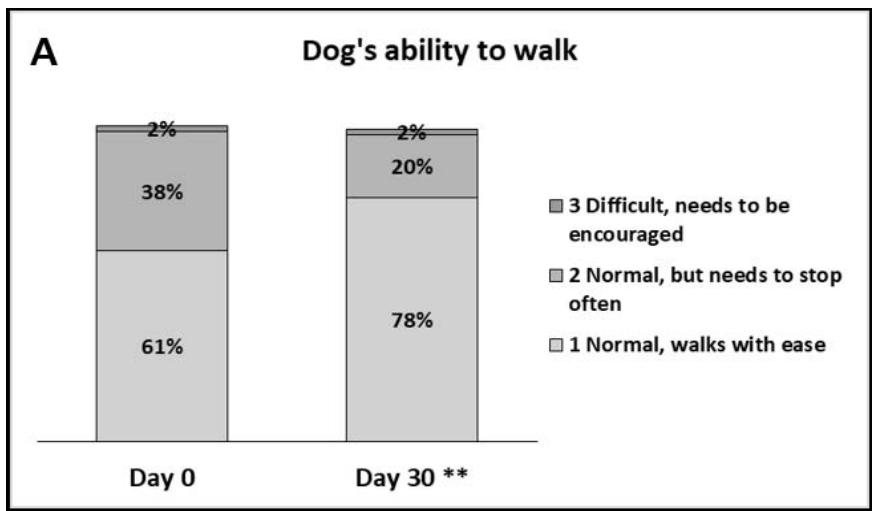
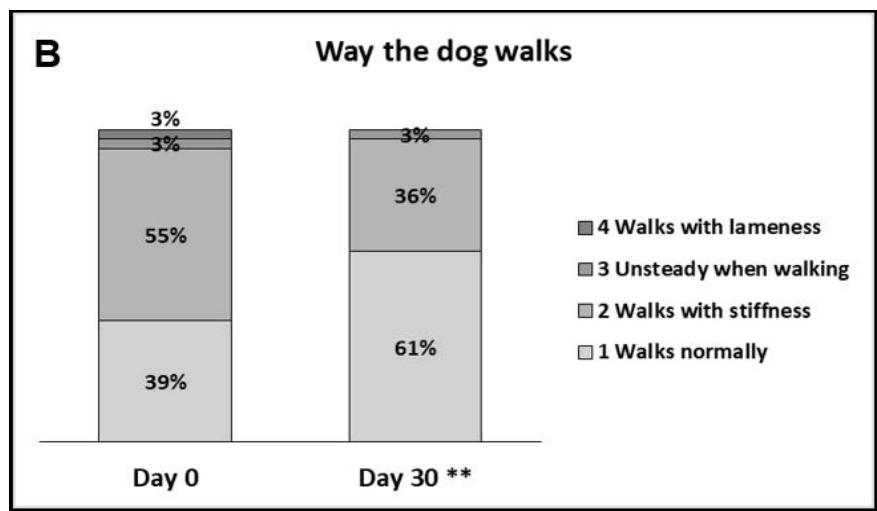
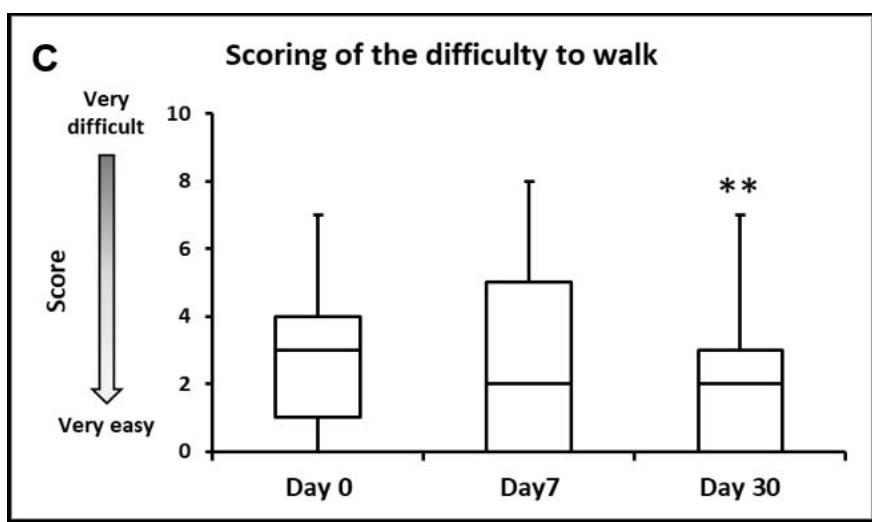 Figure 2: Dog's walking ability. A: Percentages of owners describing their dogs ability to walk as: 1 - Normal, walks with ease; 2 - Normal, but needs to stop often; 3 - Difficult, needs to be encouraged or 4 - Difficult, refuses to walk. B: Percentages of owners describing their dog's way of walking as: 1 - Walks normally; 2 - Walks with stiffness; 3 - Unsteady when walking or 4 - Walks with lameness. C: Box plot representing the median, Q1-Q3, min-max scores given by the owners for the dog's ability/difficulty to walk (on a scale from 0 - very easy to 10 - very difficult). For A,B,C: \*\*\*, p\<0.01.
- Dog's ability to stand up
- The dog's ability to stand after resting/lying also improved significantly as of Day 7 (p\<0.025; 20% of owners gave a better appreciation) and was further improved by Day 30 (31% of owners gave a better appreciation. p\<0.001). Indeed, on Day 0, 66% of owners described their dog's ability to stand as slow, with some difficulty, while on Day 30, this
number fell to $42\%$, and more owners felt their dogs could stand up with no difficulty (Figure 3)
- When asked to score their dog's ability/difficulty to stand from 0 (very easy) to 10 (very difficult), the median (Q1-Q3) scores given by owners significantly decreased from 4 (2-6) to 3 (2-5) and 2 (1-5) on Days 0, 7, and 30, respectively $(p < 0.01$, Figure 3).
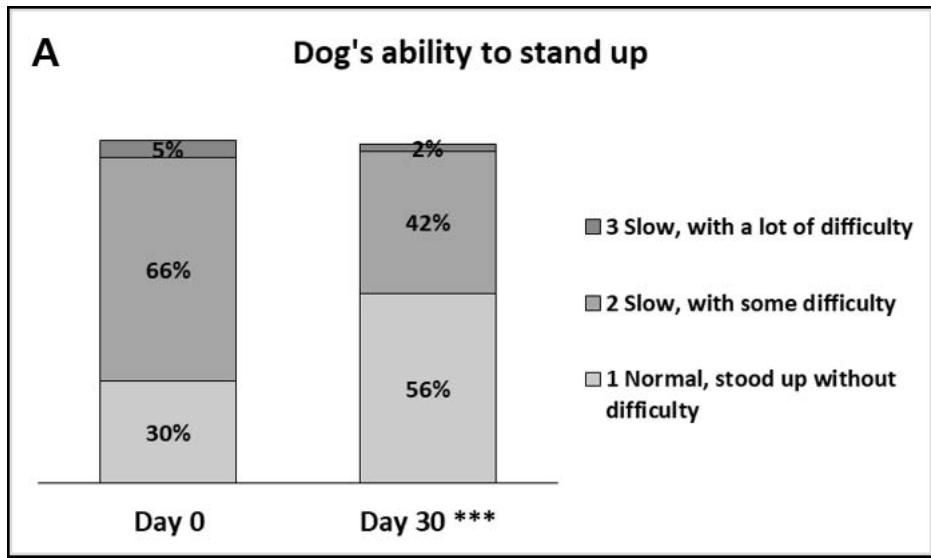
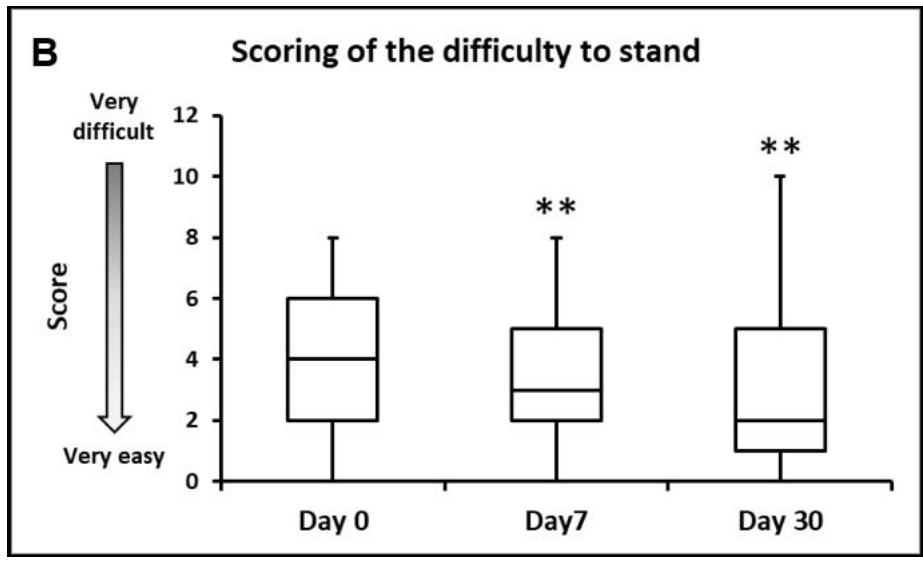 Figure 3: Dog's ability to stand up after resting/lying down for a period of time. A: Percentages of owners describing their dog's ability to stand as 1 - Normal, stood up without difficulty; 2 - Slow, with some difficulty; 3 - Slow, with a lot of difficulty; 4 - Needed help getting up. \*\*\*, p\<0.001 B: Box plot representing the median, Q1-Q3, min-max scores given by the owners for the dog's ability to stand up after resting (on a scale from 0 - very easy to 10 - very difficult). \*\*, p\<0.01.
- Dog's ability to climb steps: no significant improvement was observed for this parameter, based on the owner's appreciation (score). However, most owners described their dogs as slow when climbing steps on Day 0 (43%), while on Day 30, most owners (51%) described their dog's ability to climb as normal (Figure 4).
Finally, among the 69 owners who tested the new product, $87\%$ said they would definitely $(65\%)$ or probably $(22\%)$ buy the new Movoflex® Advanced Soft Chews. This purchase intention rate increased to $90\%$ $(70\%$ definitely and $20\%$ probably) if it is recommended by a veterinarian.
Dog's ability to climb steps
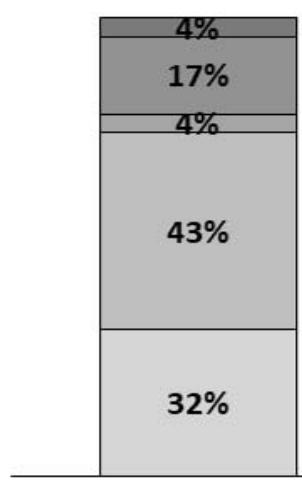 Day 0
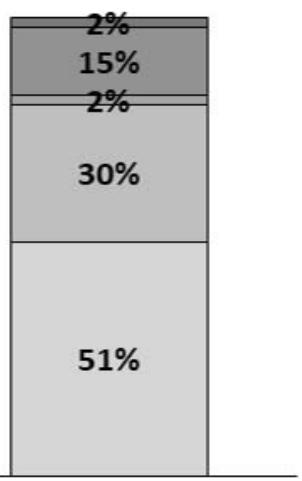 Figure 4: Dog's ability to climb steps. Percentages of owners describing their dog's ability to climb steps as 1 - Normal; 2 - Slow; 3 - Made regular stops; 4 - Needed to be encouraged; 5 - Needed help/holding; or 6 - Did not climb steps.
5 Needed help/holding 4 Needed to be encouraged 3 Made regular stops 2 Slow 1 Normal
Day 30
## IV. DISCUSSION
The advanced formula of Movoflex® Soft Chews (Virbac, USA), with low molecular weight HA and krill oil, was tested for its tolerance, palatability, and perceived effectiveness in dogs.
The tolerance evaluation showed that the chew was well tolerated by healthy dogs, even with five chews per day for 28 days. The dogs remained in good health, and no clinical signs linked to the product were observed. There was also no change in blood parameters or urine pH.
When the new chew formula was given for one month to 69 dogs who were previously taking the current Movoflex® Soft Chews, it was completely consumed in $93\%$ of cases, and $91\%$ of owners found the acceptability of the new chew was similar or even better than the previous one. According to dog owners, no change in the shape, aspect, or odor was observed compared to the current Movoflex® Soft Chews.
Since the dogs who tried the new formula previously received Movoflex® Soft Chews regularly, their mobility issues were mild but could still be improved. After seven days of daily administration, some mobility criteria were already improved, like the way of walking, and the ability to stand after lying down for some time. At this stage (Day 7), $34\%$ of owners considered their dog's mobility had improved. By Day 30, all mobility criteria but one (ability to climb steps) significantly improved, and $63\%$ of owners noticed the improvement in their dog's mobility.
These data suggest that adding krill oil and hyaluronic acid (HA) of low molecular weight to a supplement already containing ESM, astaxanthin, vitamin D3, Boswellia serrata extract, and HA of high molecular weight (Movoflex® Soft Chew) can improve its efficiency.
This finding could be explained by the omega-3 PUFA found in krill oil that can improve the bioavailability of astaxanthin and HA [43,44], enhancing their effects, on top of the known beneficial effects of omega-3 PUFA on joint health [41,42,47].
Hyaluronic acid can be found in joints, particularly in the synovial fluid. It has been shown that the concentration and molecular weight (MW) of HA in animals with OA were decreasing and that administering HA could help in maintaining joint health [33]. The high MW HA would be of interest to preserve the viscoelasticity of the synovial fluid, and the lower MW HA could help modulate the inflammation process and participate in cartilage repair [33,38].
Owners' rating of improvement remains a subjective way to evaluate the efficacy of supplements, and the findings need to be further supported by proper clinical studies involving veterinarians. However, owners remain the best persons to appreciate the progress of their dog's mobility on a day-to-day basis. Such studies, with less limited inclusion criteria and constraints, can include more participants and are performed in circumstances of evaluation closer to what is happening in real life.
Despite these limitations, the data suggest that the complex blend of ingredients found in Movoflex® Advanced Soft Chews can help improve dogs' mobility. Adding low MW HA and krill oil to the original formula led to improved benefits.
### ACKNOWLEDGMENTS
The authors would like to thank the Animal Unit's staff for taking good care of the dogs and their technical assistance.
## CONFLICT OF INTEREST
All authors are Virbac's employees and the study was financed by Virbac.
Generating HTML Viewer...
References
47 Cites in Article
W Marshall,B Bockstahler,D Hulse,S Carmichael (2009). A Review of Osteoarthritis and Obesity: Current Understanding of the Relationship and Benefit of Obesity Treatment and Prevention in the Dog.
C Muller,M Enomoto,A Buono,J Steiner,B Lascelles (2019). Placebo-Controlled Pilot Study of the Effects of an Eggshell Membrane-Based Supplement on Mobility and Serum Biomarkers in Dogs with Osteoarthritis.
Yves Henrotin,Christelle Sanchez,Marc Balligand (2005). Pharmaceutical and nutraceutical management of canine osteoarthritis: Present and future perspectives.
Zoe Belshaw,Rachel Dean,Lucy Asher (2020). “You can be blind because of loving them so much”: the impact on owners in the United Kingdom of living with a dog with osteoarthritis.
F Comblain,S Serisier,N Barthelemy,M Balligand,Y Henrotin (2016). Review of Dietary Supplements for the Management of Osteoarthritis in Dogs in Studies from 2004 to 2014.
Susan Hewlings,Douglas Kalman,Luke Schneider (2019). A Randomized, Double-Blind, Placebo-Controlled, Prospective Clinical Trial Evaluating Water-Soluble Chicken Eggshell Membrane for Improvement in Joint Health in Adults with Knee Osteoarthritis.
Erena Quintana,Marisa Fenaux,Manuel Nuez,Andrea Molero,Andres Aguirre (2018). Short-Term Effects of Ovomet®, Eggshell Membrane, in Joint Pain. A Double-Blind and Placebo Study.
Kevin Ruff,Anne Winkler,Robert Jackson,Dale Devore,Barry Ritz (2009). Eggshell membrane in the treatment of pain and stiffness from osteoarthritis of the knee: a randomized, multicenter, double-blind, placebo-controlled clinical study.
Kevin Ruff,D Devore,M Leu,M Robinson (2009). Eggshell membrane: A possible new natural therapeutic for joint and connective tissue disorders. Results from two open-label human clinical studies.
E Brunello,A Masini (2016). NEM ® Brand Eggshell Membrane Effective in the Treatment of Pain and Stiffness Associated with Osteoarthritis of the Knee in an Italian Study Population.
Gitte Jensen,Miki Lenninger,Joni Beaman,Robert Taylor,Kathleen Benson (2015). Support of Joint Function, Range of Motion, and Physical Activity Levels by Consumption of a Water-Soluble Egg Membrane Hydrolyzate.
Ulrich Danesch (2014). NEM Brand Eggshell Membrane Effective in the Treatment of Pain Associated with Knee and Hip Osteoarthritis: Results from a Six Center, Open Label German Clinical Study.
Kevin Ruff,Dennis Morrison,Sarah Duncan,Matthew Back,Cem Aydogan,Jason Theodosakis (2018). Beneficial effects of natural eggshell membrane versus placebo in exercise-induced joint pain, stiffness, and cartilage turnover in healthy, postmenopausal women.
Kevin Ruff,Kenneth Kopp,Pamela Von Behrens,Mark Lux,Matthew Mahn,Matthew Back (2016). Effectiveness of NEM<sup>® </sup>brand eggshell membrane in the treatment of suboptimal joint function in dogs: a multicenter, randomized, double-blind, placebo-controlled study.
A Aguirre,E Gil-Quintana,M Fenaux,N Sanchez,C Torre (2018). The Efficacy of Ovopet in the Treatment of Hip Dysplasia in Dogs.
W Miki (1991). Biological Functions and Activities of Animal Carotenoids.
Fredric Pashkow,David Watumull,Charles Campbell (2008). Astaxanthin: A Novel Potential Treatment for Oxidative Stress and Inflammation in Cardiovascular Disease.
Siti Oslan,Joo Tan,Siti Oslan,Patricia Matanjun,Ruzaidi Mokhtar,Rossita Shapawi,Nurul Huda (2021). Haematococcus pluvialis as a Potential Source of Astaxanthin with Diverse Applications in Industrial Sectors: Current Research and Future Directions.
Matej Baláž,Elena Boldyreva,Dmitry Rybin,Stefan Pavlović,Daily Rodríguez-Padrón,Tihana Mudrinić,Rafael Luque (2020). State-of-the-Art of Eggshell Waste in Materials Science: Recent Advances in Catalysis, Pharmaceutical Applications, and Mechanochemistry.
Jean Park,Bridget Mathison,Michael Hayek,Stefan Massimino,Gregory Reinhart,Boon Chew (2011). Astaxanthin stimulates cell-mediated and humoral immune responses in cats.
Jean Park,Jong Chyun,Yoo Kim,Larry Line,Boon Chew (2010). Astaxanthin decreased oxidative stress and inflammation and enhanced immune response in humans.
Ganpeng Yu,Wang Xiang,Tianqing Zhang,Liuting Zeng,Kailin Yang,Jun Li (2020). Effectiveness of Boswellia and Boswellia extract for osteoarthritis patients: a systematic review and meta-analysis.
J Reichling,H Schmökel,J Fitzi,S Bucher,R Saller (2004). Dietary support with Boswellia resin in canine inflammatory joint and spinal disease.
Thomas Mabey,Sittisak Honsawek (2015). Role of Vitamin D in Osteoarthritis: Molecular, Cellular, and Clinical Perspectives.
Jonathan Stockman,Cecilia Villaverde,Ronald Corbee (2021). Calcium, Phosphorus, and Vitamin D in Dogs and Cats.
Gregorio Bevilacqua,Faidra Laskou,Michael Clynes,Karen Jameson,Barbara Boucher,Kate Noonan,Cyrus Cooper,Elaine Dennison (2021). Determinants of circulating 25-hydroxyvitamin D concentration and its association with musculoskeletal health in midlife: Findings from the Hertfordshire Cohort Study.
B Boucher (2012). The Problems of Vitamin D Insufficiency in Older People.
F Ichikawa,K Katagiri,Y Higuchi,S Takeda,K Saito (2000). 1Alpha-Hydroxyvitamin D3 Prevents the Decrease of Bone Mineral Density in Lactating Beagles.
Pacharee Manoy,Pongsak Yuktanandana,Aree Tanavalee,Wilai Anomasiri,Srihatach Ngarmukos,Thanathep Tanpowpong,Sittisak Honsawek (2017). Vitamin D Supplementation Improves Quality of Life and Physical Performance in Osteoarthritis Patients.
Divya Sanghi,Abhishek Mishra,Amar Sharma,Ajai Singh,S Natu,Sarita Agarwal,Rajeshwar Srivastava (2013). Does Vitamin D Improve Osteoarthritis of the Knee: A Randomized Controlled Pilot Trial.
Keiichi Kuroki,James Cook,John Kreeger (2002). Mechanisms of action and potential uses of hyaluronan in dogs with osteoarthritis.
Claudio Serra Aguado,Juan Ramos-Plá,Carme Soler,Sergi Segarra,Víctor Moratalla,José Redondo (2021). Effects of Oral Hyaluronic Acid Administration in Dogs Following Tibial Tuberosity Advancement Surgery for Cranial Cruciate Ligament Injury.
Lajos Balogh,Andras Polyak,Domokos Mathe,Reka Kiraly,Juliana Thuroczy,Marian Terez,Gyozo Janoki,Yaoting Ting,Luke Bucci,Alexander Schauss (2008). Absorption, Uptake and Tissue Affinity of High-Molecular-Weight Hyaluronan after Oral Administration in Rats and Dogs.
Nozomi Hisada,Hideo Satsu,Akira Mori,Mamoru Totsuka,Jun-Ichi Kamei,Takashi Nozawa,Makoto Shimizu (2008). Low-Molecular-Weight Hyaluronan Permeates through Human Intestinal Caco-2 Cell Monolayers<i>via</i>the Paracellular Pathway.
V Iturriaga,B Vásquez,T Bornhardt,M Del Sol (2021). Effects of Low and High Molecular Weight Hyaluronic Acid on the Osteoarthritic Temporomandibular Joint in Rabbit.
L Burri,L Johnsen (2015). Krill Products: An Overview of Animal Studies.
Lei Liu,Nana Bartke,Hans Van Daele,Peter Lawrence,Xia Qin,Hui Park,Kumar Kothapalli,Anthony Windust,Jacques Bindels,Zhe Wang,J Brenna (2014). Higher efficacy of dietary DHA provided as a phospholipid than as a triglyceride for brain DHA accretion in neonatal piglets.
Michelle Ierna,Alison Kerr,Hannah Scales,Kjetil Berge,Mikko Griinari (2010). Supplementation of diet with krill oil protects against experimental rheumatoid arthritis.
Yoshio Suzuki,Minoru Fukushima,Keishoku Sakuraba,Keisuke Sawaki,Kazuaki Sekigawa (2016). Krill Oil Improves Mild Knee Joint Pain: A Randomized Control Trial.
Johanna Mercke Odeberg,Åke Lignell,Annette Pettersson,Peter Höglund (2003). Oral bioavailability of the antioxidant astaxanthin in humans is enhanced by incorporation of lipid based formulations.
S.-L Huang,P.-X Ling,T.-M Zhang (2007). Oral absorption of hyaluronic acid and phospholipids complexes in rats.
D Park,R Ko,S Kwon,B Min,S Yun,M Kim,J Minatelli,S Hill,S Lee,M Flexpro (2016). a Mixture of Krill Oil, Astaxanthin, and Hyaluronic Acid, Suppresses Lipopolysaccharide-Induced Inflammatory Cytokine Production Through Inhibition of NF-κB.
Min Park,Jae Jung,Stephen Hill,Elizabeth Cartwright,Margaret Dohnalek,Min Yu,Hee Jun,Sang Han,Jin Hong,Dong Son (2020). FlexPro MD®, a Combination of Krill Oil, Astaxanthin and Hyaluronic Acid, Reduces Pain Behavior and Inhibits Inflammatory Response in Monosodium Iodoacetate-Induced Osteoarthritis in Rats.
Marieke Loef,Jan Schoones,Margreet Kloppenburg,Andreea Ioan-Facsinay (2019). Fatty acids and osteoarthritis: different types, different effects.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Nicolas CS. 2026. \u201cAdding Krill Oil and Low Molecular Weight Hyaluronic Acid to Movoflex® Soft Chews Helps Improve Dog’s Mobility\u201d. Global Journal of Medical Research - G: Veterinary Science & Medicine GJMR-G Volume 22 (GJMR Volume 22 Issue G1).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.