## I. INTRODUCTION
Obesity is a complex metabolic disorder with a multifactorial origin. Increasing evidence suggests that oxidative stress is a critical factor linking obesity to its metabolic complications [1].
Indeed, excessive accumulation of body white adipose tissue (WAT), particularly visceral WAT, stimulates a prooxidant and proinflammatory state, increasing the risk of a large number of pathological events such as insulin resistance, diabetes, cardiovascular complications, liver failure, sleep disorders and oncological problems [2].
Sustained nutrient overflow, especially in obesity states, can dysregulate adipose tissue storage capacity leading to adipocyte hypertrophy and tissue hypoxia [3]. It has been proposed that a relative oxygen deficit due to reduced blood flow in adipose tissue, contributes to adipocyte dysfunction and may impact systemic metabolic homeostasis [4]. Hypoxia affects several biological functions in adipose tissue, such as adipocyte differentiation, angiogenesis, apoptosis, inflammation, and insulin resistance [5]. Additionally, mitochondrial reactive oxygen species (ROS) production, especially superoxide anions and hydrogen peroxide, have been shown to activate signaling cascades, being responsible for the propagation of the hypoxic signal (Lempesis et al, 2020). Hypoxia-inducible transcription factor-1 (HIF-1) is an important regulatory signaling molecule that mediates the inflammatory responses induced by hypoxia [6]. This transcription factor, a heterodimer formed by an oxygen-sensitive $\alpha$ -subunit and a constitutively expressed $\beta$ -subunit (HIF-1 $\beta$ ), is accumulated during hypoxia resulting in the activation of HIF-1 $\alpha$ target genes [7]. The expression of the HIF-1 $\alpha$ subunit has been used as a marker of hypoxia states and several studies suggest that HIF-1 $\alpha$ inactivation could protect against obesity and insulin resistance [7, 8].
In recent decades, the increase in the global prevalence of overweight and obesity has prompted a wide range of strategies to reduce their impact on health. The development of functional foods represents a valuable opportunity as a non-pharmacological alternative for the prevention and/or treatment of obesity [9]. It involves the discovery of new bioactive compounds, as well as the understanding of the mechanisms by which these food ingredients exert their anti-obesity effects.
Prebiotics are a group of nutrients that selectively stimulate the growth and activity of beneficial bacteria in the human gut [10]. Fructooligosaccharides (FOS) and inulin, two non-digestible fructose polymers with different degrees of polymerization, are gaining great importance as functional ingredients given their multiple health benefits and pharmaceutical applications [11]. Due to the presence of a $\beta-(2\rightarrow 1)$ glycosidic bond, these oligosaccharides resist enzymatic hydrolysis in the mammalian digestive system and are metabolized by the gut microbiota into short-chain fatty acids (SCFAs). The SCFAs influence energy intake and weight gain improving obesity [11, 12].
Most of the commercially available inulin comes from Cichorium intybus (chicory) roots, and Agave tequilana Weber var. azul stem and leaves [13] FOS are naturally found in Allium spp. (onion, garlic, and leek), and Musa paradisiaca (banana) [14]. Smallanthus sonchifolius (Poepp. & Endl.) H. Robinson (yacon) tubers, native specie of the Andean regions of South America, are another abundant source of FOS with health-promoting benefits [15, 16]. In a previous study, we established that yacon flour restored serum lipid profile and attenuated weight gain in animal models and humans by modulating signaling pathways that regulate adipogenesis [17, 18]. Although the beneficial effects of yacon on metabolism are known, the effects on adipose tissue pathology and its associated parameters are not fully clarified.
In this context, we now analyzed the effects of yacon rich in FOS on hypoxia, oxidative stress, inflammation, and differentiation of visceral WAT in HFD-fed rats. Also, we compared these effects with those from agave inulin, to achieve a better characterization of the prebiotic potential of these oligofructans as functional ingredients.
## II. MATERIALS AND METHODS
### a) Plant Material
Smallanthus sonchifolius flour was obtained according to [18]. Briefly, the roots were carefully washed, peeled, sliced, and dried at $60^{\circ}\mathrm{C}$ in a forced air circulation oven to reduce water content. The dried material was then pulverized to obtain yacon roots flour and stored at $4^{\circ}\mathrm{C}$ until use. The content of FOS was estimated as previously [17] and daily intake levels of yacon flour were calculated concerning the amount of FOS using a dose equivalent to $680\mathrm{mg}$ FOS/Kg b.w./day corresponding to $1.57\mathrm{g}$ of yacon flour/kg b.w./day. To compare the metabolic effects of yacon flour and inulin, Organic Inulin Pure Prebiotic Powder from Blue Agave, was purchased from NOW Foods (USA) and administered to the animals at a dose of $680\mathrm{mg}$ FOS/Kg b.w./day.
### b) Animals, diets, and experimental protocol
Male Wistar rats weighing 180-200 g were obtained from the colony bred at the INSIBIO (CONICET-UNT), Tucumán, Argentina. Rats were kept in a breeding room with a controlled environment (temperature: $23 \pm 1^{\circ} \mathrm{C}$, relative humidity: $60 \pm 5\%$, and
12 h light-dark cycle). Rats were maintained on a 12-h light-dark cycle with free access to food and water. All experiments were performed following the Guide for the Care and Use of Laboratory Animals (Institute of Laboratory Animal Resources, Commission on Life Sciences, National Research Council, National Academy Press, Washington, DC) and approved by the local Animal Care Committee from the Universidad Nacional de Tucumán (No. 004/2017). The experimental animals were randomly divided into two groups: the control diet group (CD, n=6) was fed a standard chow diet (Asociación Cooperativas Argentinas S.E.N.A.S.A., containing 12.08 kJ/g calories: 69.5% from carbohydrates, 5.6% from fat, and 24.9% from protein) and a high-fat-diet group (HFD, n=18) which have free access to a special diet [18], containing 17.40 kJ/g: 35.0% from carbohydrates, 41.0% from fat, and 24.0% from protein). Both animal groups were maintained on each diet for 12 weeks. Water and solid food were available ad libitum. At this time, animals fed HFD reached obesity status.
### c) Experimental Groups
After 12 weeks on HFD or standard chow, the animals were randomly divided into the following groups according to the treatment with or without the addition of yacon flour or inulin, as a dietary supplement for 8 weeks.
i. HFDY group (n=6), rats fed a high-fat diet plus a tablet of yacon flour (equivalent to 680mg FOS/kg b.w). ii. HFDI group (n=6), rats fed a high-fat diet plus a tablet of inulin (680 mg/kg b.w.) iii. HFD group $(n = 6)$, rats fed a high-fat diet. iv. CD group $(n = 6)$, rats fed a standard diet.
Throughout the experimental period, animals were weighed (g) weekly and food intakes were recorded daily. Abdominal circumference (immediately anterior to the forefoot) and thoracic circumference (immediately behind the foreleg), and were determined according to [19]. The body weight and body length (nose-anus length) were used to determine the following morphometric parameters: Body mass index (BMI) = body weight (g)/length2 (cm2); Lee index = cube root of body weight (g)/nose-anus length (cm). Nutritional parameters were calculated based on food and energy intake: energy intake (kJ/day) = mean food consumption x dietary metabolizable energy.
### d) Tissue Sampling
The animals were sacrificed in the fasted state through cardiac puncture under anesthesia with 1:1 xylazine-ketamine. Collected blood samples were allowed to clot at $37^{\circ}\mathrm{C}$ and serum was separated by centrifugation at $3000\mathrm{g}$ for $10\mathrm{min}$. The visceral WAT (mesenteric) was quickly collected and rinsed thoroughly with ice-cold saline, blotted, weighed, and fixed in $4\%$ formaldehyde saline for histological analysis. The remaining tissues were frozen immediately and stored at $-80^{\circ} \mathrm{C}$ until analyzed.
### e) Biochemical Determinations
Blood glucose was measured using a blood glucometer and strips (Roche Accu-Chek Active, Mannheim, Germany). Serum levels of triglycerides (TG), total cholesterol (TC), and low-density lipoprotein cholesterol (LDL-c) were measured using commercial kits (Wiener lab Group, Argentina) according to the manufacturer's instructions.
### f) Oral glucose tolerance test (OGTT) and insulin tolerance test (ITT)
The OGTT and ITT were developed at week 12 (data not shown) and at the end of the experimental period (week 20). For OGTT, baseline blood glucose (time 0) was determined in fasted animals and then a glucose solution (2 g/kg b.w. glucose) was administered via gavage, followed by the determination of blood levels at 15, 30, 60, and 120 min with a Glucometer (AccuCheck; Roche Diagnostics, GmbH, Mannheim, Germany). For ITT the animals were injected intraperitoneally with 0.75 IU/kg b.w. of porcine Insulin (Betasint, BETA laboratory) after a 4-hour fast. Blood glucose concentration was measured with blood from tail-tip bleedings at 0, 15, 30, and 60 min. The areas under the curve of glucose were calculated [18].
### g) Histology and immunofluorescence microscopy analysis
Samples from fixed visceral WAT were dehydrated, embedded in paraffin, and cut into $4\mu \mathrm{m}$ thick sections at $50~{\mu\mathrm{m}}$ intervals. The sections were stained with hematoxylin and eosin (H&E) for histological study as previously described [18]. Ten randomly selected fields in two slides of each rat were evaluated by two investigators blinded to the origin of the slides. Adipocyte size was determined using ImageJ 1.8.0 software (National Institutes of Health, Bethesda, MD).
Tissue was blocked in $5\%$ goat serum in PBS for 1 h and stained with anti-F4/80 (Abcam, Cambridge, MA) or NF- $\kappa$ B p65 (eBioscienceTM) antibody overnight at $4^{\circ}\mathrm{C}$. After washing with PBS, tissue was incubated with an Alexa 488-conjugated anti-rabbit secondary antibody (Invitrogen) for 1 h at room temperature. The tissue was then imaged at $40\times$ magnification using a Leica DM2500 Microscope and analyzed using ImageJ software. A total number of F4/80-expressing cells was counted in ten randomly selected fields and expressed as positive cells/field.
### h) Redox State Markers
Visceral WAT samples were homogenized (1:10 w/v) in a 0.1 M phosphate buffer solution pH 7.4 at $4^{\circ}\mathrm{C}$. One part was homogenized to measure the activity levels of antioxidant enzymes and malondialdehyde (MDA). The second part was homogenized in a cold solution of 0.1 M HClO4 2mM EDTA for reduced glutathione (GSH) estimation.
Lipid peroxidation in WAT was analyzed by determining the formation of MDA [20]. GSH levels were determined through the spectrophotometric method [21]. The enzymatic antioxidant capacity was assayed by estimating superoxide dismutase (SOD) [22], catalase (CAT) [23], and glutathione-S-transferase (GST) [24] activities.
### i) RNA Extraction and RT PCR Amplification
RNA from adipose tissue was isolated using Trizol (Thermo Fisher Scientific, Netherlands) followed by purification with an RNeasy Mini Kit (Qiagen, Basel, Switzerland), according to the manufacturer's instruction including a DNase treatment with an RNase-free DNase Set. $0.5\mu \mathrm{g}$ of RNA were reverse transcribed into cDNA using M-MLV Reverse Transcriptase (Promega, USA). Gene expression was evaluated using a Mastercycler personal instrument (Eppendorf, Germany) according to [18], with an optimized concentration of primer sets for HIF-1α (NM024359.1) forward primer 5'-CACAGCTGACCAGTTACGATTG-3' and reverse primer 5'-CACAGACAACAACAACAACTGAAC-3'; TGF-β1 (NM021578.2) forward primer 5'-GTGGCTGAACCAAGGAGACG-3' and reverse primer 5'-GGTGTTGAGCCCTTTCCAGG-3'; MCP-1 (NM031530.1) forward primer 5'-TCAACCCTAAGGACTTCAGCAC-3' and reverse primer 5'-AGGCATCACATTCCAAATCACAC-3'; IL-1β (NM031512.2) forward primer 5'-CACCTCTCAAGCA-GAGCACA-3' and reverse primer 5'-GACCTGACTT-GGCAGAGGAC3'; TNF-α (NM012675.3) forward primer 5'-CTCAAGCCCTGGTATGAGCC-3' and reverse primer 5'-GGCTGGGTAGAGAACGGATG-3'; IL-6 (NM01-2589.2) forward primer 5'-ATTGTATG-AACA-GCGATGATGCAC3' and reverse primer 5'-CCAGGTAGAAACGGAACTCCAGA-3'; IL-10 (NM01-2854.2) forward primer 5'-ACTGCAGGACTTTAAGGGTTACTTG3' and reverse primer 5'-TAGACACCTTTGTCTTGGA GCTTA-3'; Wnt5a (NM022631.2) forward primer 5'-AGGACTTACCTCGGGACTGG-3' and reverse primer 5'-CGACCTGCTT-CATTGTTGTG-3; Scl2a4 (NM012751.1) forward primer 5'-CCTCCAGGATGAAGGAACA-3' and reverse primer 5'-GGGAGAAAAGCCCATCTAGG-3'; β-actin (NM-007393) forward primer 5'-CCGGCTTCGCGGGCGACG-3' and reverse primer 5'-TCCCGGCCAGCCAGGTCC-3'. mRNA levels were standardized against β-actin mRNA levels in the same sample. The mean value for the CD was set at 100%.
### j) Western Blotting
Protein levels of five Wnt-3a were measured with western blot as described previously [18]. Briefly, $20~\mu \mathrm{g}$ of total protein per sample was used for SDS-PAGE and transferred to a nitrocellulose membrane (Hybond-C super; Amersham, Buckinghamshire, UK). Blots were blocked with $5\%$ (w/v) fat-free milk dissolved in phosphate-buffered saline containing $0.05\%$ (v/v) Tween-20 (TBST), incubated with mouse anti-Wnt3a (1:100 dilution; Cell Signaling Technology Inc. Danvers, USA) as primary antibody and ECL anti-mouse IgG (Thermo Fisher Scientific) as a secondary antibody. Protein levels were detected with a biotin-extrAvidin-peroxidase system (SIGMA Aldrich, USA). Band intensities were quantified by ImageJ software.
### k) Statistical Analysis
All results are presented as the mean $\pm$ standard deviation and were analyzed with GraphPad Prism®, version 8 (GraphPad Software, Inc., La Jolla, CA, USA). Comparisons of all the variables between the groups of each experiment were performed through the use of unpaired Student's t-tests and by one-way ANOVA followed by Tukey's post-test. A probability level of $p < 0.05$ was considered statistically significant.
## III. RESULTS
### a) Effects of yacon on morphological and nutritional parameters
As shown in Table 1, animals in the HFD group significantly increased body weight compared to the CD group $(p < 0.05)$. Supplementation with yacon flour for 8 weeks significantly reduced body weight in obese HFD-fed animals $(p < 0.05)$. Consistent with these results, yacon or inulin supplementation significantly $(p < 0.05)$ reduced food intake in HFDY and HFD animals, resulting in lower energy intake $(p < 0.05)$. A significant $(p < 0.05)$ improvement in the morphometric parameters of the animals supplemented with each of the prebiotics at the dosetested was also observed. Table 1 also shows the effects of yacon flour on the relative weights of liver, and visceral WAT. HFD-fed rats exhibited an increase in the hepatic index that was reversed by both yacon and inulin supplementation. In addition, yacon supplementation significantly $(p < 0.05)$ reduced the visceral WAT index promoted by HFD-feeding, especially decreasing the deposition of visceral WAT in the mesenteric pad. However, inulin supplementation showed no difference $(p > 0.05)$ in adipose tissue deposition.
### b) Effects of yacon on adipose tissue hypoxia
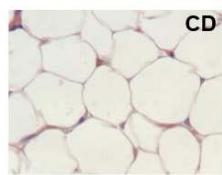
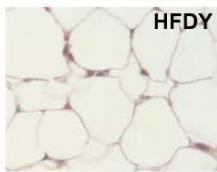
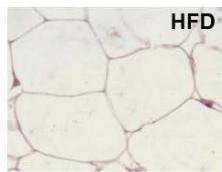
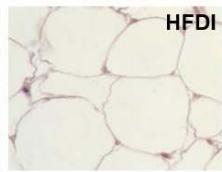
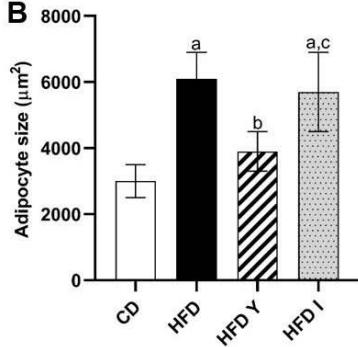
During obesity, adipocyte hypertrophy is associated with local adipose tissue hypoxia [25]. According to the above results, H&E staining revealed the presence of a higher number of large adipocytes in the visceral WAT of HFD and HFDI animals, while smaller adipocytes predominated in HFDY supplemented animals (Figure 1A, B). Then, we analyzed the expression of HIF- $1\alpha$, the inducible subunit of HIF-1, as a key transcription factor mediating hypoxic responses [7]. The results showed that HIF- $1\alpha$ gene expression was significantly $(p < 0.05)$ higher in the visceral WAT of the HFD group compared to the CD group. Yacon supplement significantly $(p < 0.05)$ reduced the expression of this factor compared to the HFD group. However, no effect on HIF-1 $\alpha$ expression was observed in the visceral WAT of the HFDI group (Figure 1C).
### c) Effects of yacon on oxidative stress of adipose tissue
Long-term exposure of fat cells to hypoxia can provoke oxidative stress in human and animal WAT [26]. Indeed, visceral WAT from HFD-rats showed increased levels of MDA and reduced GSH $(p < 0.05)$. Yacon consumption in HFDY significantly $(p < 0.05)$ reduced MDA and increased GSH in visceral WAT after an 8-wk feeding (Figure 2A, B). Moreover, yacon increased the GST and CAT enzyme activities in HDFY compared to the HFD group (Figure 2C, D). However, GPx activity did not show a significant difference between both groups $(p > 0.05)$ (Figure 2E). Inulin supplementation also improved the redox status of visceral WAT (Figure 2A-E), although to a lesser degree, except for GSH levels, where it showed a greater effect (Figure 2B).
### d) Effects of yacon on the inflammatory response of adipose tissue
The state of hypoxia promotes inflammation of adipose tissue in obesity through the activation of two main transcription factors, HIF-1 $\alpha$ and nuclear factor- $\kappa$ B (NF- $\kappa$ B), which in turn activate the transcription of several angiogenic and/or pro-inflammatory genes [7]. To study the inflammation response in yacon or inulin-supplemented HFD-rats, we quantified inflammation gene expression in visceral WAT (Figure 3). We found that pro-inflammatory adipocytokines were significantly $(p < 0.05)$ increased in the HFD compared to the CD group $(p < 0.05)$ (Figure 3A, B). Interestingly, yacon treatment improved the inflammatory status of adipose tissue, reducing the expression of MCP-1, TNF- $\alpha$, TGF- $\beta$ 1, IL-1 $\beta$, and IL-6 in the HFDY group $(p < 0.05)$. The anti-inflammatory cytokine IL-10 was significantly elevated in HFDY rats $(p < 0.05)$ (Figure 3A, B). Inulin treatment significantly $(p < 0.05)$ decreased the TGF- $\beta$ 1 and IL-6 expression in the visceral WAT similar to yacon, but increased IL-10 to a lesser extent (Figure 3B). Macrophage infiltration in WAT contributes to inflammation, extracellular matrix remodeling, and obesity-related metabolic dysfunction [27]. Macrophage marker F4/80 staining of visceral WAT sections revealed that macrophage infiltration to WAT decreased significantly $(p < 0.05)$ in HDFY animals (Figure 3C, D). Consistent with this finding, lower levels of NF- $\kappa$ B p65 in these adipocytes were detected (Figure 3C, E).
### e) Effects of yacon on adipogenesis-related genes
Hypertrophic WAT provides an optimal environment for increasing the differentiation rate of adipocyte progenitors, due to a decreased glucose uptake and higher plasma glucose levels [25]. So, we decided to analyze the expression of the paracrine factors Wnt3a and Wnt5a involved in adipocyte differentiation, as well as the glucose transporter SCL2A4 in the visceralWAT after an 8-wk HFD feeding. (Figure 4). Western blot assay showed that Wnt3a protein levels were lower in the visceral WAT of HFDrats. However, yacon significantly $(p < 0.05)$ increased this paracrine factor in HFD-animals (Figure 4A). In addition, while HFD-feeding up-regulated Wnt5a and down-regulated SCL2A4 mRNA expression in the visceral WAT $(p < 0.05)$; yacon supplementation restored the expression of both mRNAs to their CD levels $(p < 0.05)$. Inulin intake up-regulated SCL2A4 mRNAs levels in the visceral WAT showing a similar effect to yacon; however, no effects on SCL2A4 mRNAs expression were observed in these animals. $(p > 0.05)$ (Figure 4B).
### f) Effects of yacon on systemic glucose and lipids homeostasis
To compare the efficacy of both prebiotics on the systemic metabolic and oxidative status, we determined the effects of yacon flour and inulin supplementation on glucose and lipid homeostasis and oxidative stress markers. As shown in Table 2, the HFDY group showed reduced $(p < 0.05)$ fasting glucose levels and a significant $(p < 0.05)$ improvement in glycemic response after OGTT and ITT compared to the animals in the HFD group. Similar effects were observed in the group supplemented with inulin. Both yacon and inulin consumption improves the lipid profile of HFD animals. TG levels were similar in both groups, however, the HFDY group showed lower levels of TC and LDLc, compared to HFDI $(p < 0.05)$. In addition, the animals that consumed yacon showed lower postprandial TG values than inulin $(p < 0.05)$, but there were no significant differences in postprandial levels of cholesterol in both groups $(p > 0.05)$. The analysis of the oxidative stress markers MDA and GSH indicated that MDA was lower in the HFDY group compared to the HFDI group, while the level of GSH was further improved after inulin supplementation $(p < 0.05)$.
## IV. DISCUSSION
During the development of obesity, adipose tissue undergoes a remodeling process as a consequence of adipocyte enlargement and recruitment of adipogenic precursor cells, to adjust excess nutrients and avoid peripheral lipotoxicity [28]. Diet modification is one of the most relevant strategies against obesity in the long term management. In this sense, the identification of natural and safe ingredients to incorporate in functional foods destined to reduce weight is necessary. In this work, we analyzed the functional effect of yacon flour in the visceral WAT pathology of HFD-induced obese rats, and compare its efficacy versus commercial agave inulin.
Our results showed that both yacon flour, rich in FOS, and inulin were effective in reducing body weight and improving anthropometric parameters. This could be related to the ability of fructans to modulate gastrointestinal peptides such as ghrelin and peptide YY (PYY), involved in the control of food intake [18, 29]. Furthermore, there is evidence that these oligosaccharides increase the level of GLP-1 in the proximal colon, with an impact on metabolic regulation that contributes to reducing weight in obesity [12, 16, 18].
Overnutrition increases adiposity, leading to an adipocyte dysfunctional phenotype associated with local tissue hypoxia [3]. Growing data suggest that this chronic condition plays a key role in macrophage chemotaxis, adipocytokine dysregulation, and impaired insulin signaling in visceral WAT [4, 27]. Our data thus suggest that yacon supplementation ameliorates WAT tissue oxidative stress and inflammation, by alleviating hypoxia state, and improving visceral fat mass in HFD-fed rats. Indeed, here we show for the first time that HFD-induced overexpression of HIF-1α in visceral WAT was reduced by yacon supplementation. Indeed, here we show for the first time that HFD-induced HIF-1α overexpression in visceral WAT was reduced by yacon supplementation, but not by inulin. The action of yacon on HIF-1α could be related to the general weight loss induced by FOS. However, the absence of an effect on HIF-1α by inulin allows us to suggest that the differences found could be due to the presence of other components in the yacon flour more than the different degrees of polymerization [30] of the fructans under study. Further studies are needed to clarify this aspect.
HIF-1α factor induction is considered an early initiator for WAT dysfunction and it has been suggested that HIF-1α inhibition could ameliorate the negative effects of fat expansion associated with dietary-induced obesity [31]. So, the yacon flour supplement may constitute an effective food therapeutic option in the context of metabolic dysfunction. HIF-1α is a transcription factor that responds to a low concentration of oxygen [4, 5]. Under hypoxic conditions, mitochondria increase the production of ROS, leading to inhibition of prolyl hydroxylase activity with subsequent stabilization of HIF1-α protein [32]. HIF-1α is then translocated to the nucleus to stimulate the transcription of target genes involved in the regulation of oxygen homeostasis via angiogenesis and metabolic reprogramming [6, 33]. In addition, HIF-1α targeted a wide range of cellular functions, including apoptosis, autophagy, redox homeostasis, inflammation, fibrosis, and self-renewal [5, 7]. In line with this, we showed that yacon flour supplementation improved the redox state reducing lipoperoxidation and strongly improving antioxidant enzyme activities in visceral WAT. Moreover, yacon reduced NF-κB p65 signal in visceral WAT, which was related to reduced transcription of inflammatory cytokine MCP-1, TNF- $\alpha$, IL-1 $\beta$, and IL-6, and is consistent with the reduced macrophage infiltration demonstrated. Some data indicate that TGF- $\beta$ can increase the HIF-1 $\alpha$ gene transcription and promote its accumulation and activity by increasing protein stability [34]. So, the improvement in inflammatory parameters caused by yacon consumption and, particularly, the decrease in TGF- $\beta$ expression in the visceral WAT could also affect the transcription of the HIF-1 $\alpha$ gene and cooperate with the decreased bodyweight to reduce HIF-1 $\alpha$ levels. Furthermore, Zhou et al., [32] showed that TGF- $\beta$ 1 could also be a molecular target of HIF-1 $\alpha$ and ROS under hypoxic conditions, promoting tissue fibrosis. The authors also reported that suppression of HIF- $\alpha$ prevented upregulation of TGF- $\beta$ 1 mRNA. These data reinforce the extraintestinal effects of yacon by showing action on HIF-1 $\alpha$, ROS, and TGF- $\beta$.
TGF- $\beta 1$ has also been shown to play an important role in adipogenesis during the maturation stage of progenitor cells showing an inhibitory action on adipocyte differentiation [35]. This fact could imply an additional role for yacon in adipogenesis through the modulation of TGF- $\beta 1$ expression. In addition, Wnt5a, another paracrine factor belonging to the Wingless/Wnt family, is known to activate adipogenesis through a noncanonical pathway, promoting preadipocyte proliferation and PPAR- $\gamma$ activation [36]. On the other hand, Wnt3a maintains preadipocytes in an undifferentiated state through adipogenic inhibition of C/EBP $\alpha$ and PPAR- $\gamma$ [37]. Previous results indicated that yacon flour downregulates these key transcription factors, enhancing insulin sensitivity, and lowering blood lipid concentrations [18]. Kang et al. [38] evidenced an antagonistic effect of both factors during adipogenesis. They demonstrated that Wnt3a induction can block Wnt5a expression, inhibiting adipogenesis and restoring adipose tissue function. In concordance, our results showed that yacon down-regulates Wnt5a and up-regulates Wnt3a transcription in visceral WAT, evidencing the participation of Wnt paracrine factors in the effects of yacon on adipose tissue. The role of Wnt5a in promoting inflammation and metabolic dysfunction in obesity has also been described [36]. Previous work has demonstrated that yacon flour can improve glucose tolerance and insulin sensitivity in both animals and humans, modulating adiponectin and leptin levels [18]. Long-term yacon supplementation also improves glucose uptake ameliorating abnormal insulin signaling cascades in the visceral WAT of HFD rats by enhancing the p-Akt/Insulin ratio. Glucose transport in insulin-resistant adipocytes from obese and diabetic subjects correlates with reduced SCL2A4 mRNA and protein expression [39]. Here we demonstrated that both yacon and inulin supplementation induced a robust upregulation of SCL2A4 glucose transporter in visceral WAT of HFD-rats. These data suggest an additional role for both ingredients in maintaining insulin-dependent glucose homeostasis. Moreover, different studies have suggested that enhancement of SLC2A4 gene expression could represent a good effective approach not only to increase plasma membrane GLUT4 and WAT's ability to clear blood glucose [40] but also to improve fasting triglyceridemia [39].
## V. CONCLUSION
Functional ingredients are a trend in the market due to the possibility of manufacturing processed products with greater health benefits and pleasant sensory characteristics, adding value to products. The goal of the present study was to compare yacon flour rich in FOS and agave inulin regarding their effects on the oxidative stress and inflammatory response induced by lipid overload and hypoxia in adipose tissue. Although both fructans show a positive effect on the remodeling of adipose tissue, this research demonstrated new healthy properties of the bioactive compounds present in yacon flour vs. inulin, expanding the possibilities of its use by the food industry, in the development of new healthy products (dairy, bakery, meat and beverage products).
Conflicts of Interest
The authors declare that there are no conflicts of interest.
### ACKNOWLEDGMENTS
This research was supported by PICT 2017 (no. 3941) (ANPCyT, Argentina), PIP 2015 (no. 183) (CONICET Argentina), and PIUNT D619 (SCAIT-UNT, Argentina), grants to SMH and SSS.
SMH and SSS: CONICET Researcher Career of CONICET. MVG and ENDM: Recipient of CONICET Fellowship.
Table 2: Effect of yacon flour intake on glucose and lipids homeostasis
<table><tr><td></td><td>CD</td><td>HFD</td><td>HFDY</td><td>HFDI</td></tr><tr><td>Fasting glucose (mg/dL)</td><td>98.0±6.0</td><td>129.0±6.0a</td><td>116.0±3.0a,b</td><td>116.0±3.0a,b</td></tr><tr><td>OGTT AUC (mg/dL/min)</td><td>17799±457.2</td><td>21778±942.5a</td><td>18519±444.2b</td><td>18558±837.4b</td></tr><tr><td>ITT AUC (mg/dL/min)</td><td>3574±140.0</td><td>5866±570.1a</td><td>3574±785.6b</td><td>5324±231.8a,c</td></tr><tr><td>TG (mg/dL)</td><td>32.0±4.3</td><td>76.4±3.8a</td><td>46.8±5.1b</td><td>41.4±12.6b</td></tr><tr><td>TC (mg/dL)</td><td>63.2±6.9</td><td>98.3±11.1a</td><td>68.8±4.0b</td><td>81.4±3.4a</td></tr><tr><td>LDL-c (mg/dL)</td><td>25.2±0.9</td><td>39.0±4.2a</td><td>19±2.6a,b</td><td>29.3±2.1b,c</td></tr><tr><td>Postprandial TG AUC (mg/dL/min)</td><td>5180±227.7</td><td>12540±472.2a</td><td>10189±445.3a,b</td><td>6540±815.7a,b,c</td></tr><tr><td>GSH (mg/mL)</td><td>0.025±0.0027</td><td>0.018±0.0003a</td><td>0.021±0.0006a,b</td><td>0.020±0.0001b</td></tr><tr><td>MDA (nmol/mL)</td><td>0.34±0.04</td><td>0.90±0.05a</td><td>0.50±0.02a,b</td><td>0.65±0.02a,b,c</td></tr></table>
 A
 B
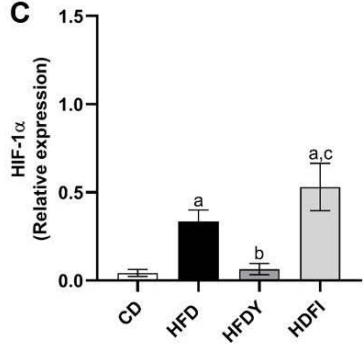 C Figure 1: Effects of yacon flour and inulin on visceral adipose tissue remodeling of HFD-fed rats. (A) H&E stained sections from visceral WAT (magnification $400\mathrm{x}$ ). (B) Adipocyte size. (C) Relative mRNA expressions of HIF-1α. Data were normalized to Actin mRNA and expressed as fold change over the CD rats. The results are expressed as the mean ± SD of triplicate RT-PCR analysis. (n=6/group). One-way ANOVA test (significant p < 0.05). a p\<0.05 vs. CD; b p\<0.05 vs. HFD; c p\<0.05 vs. HFDY
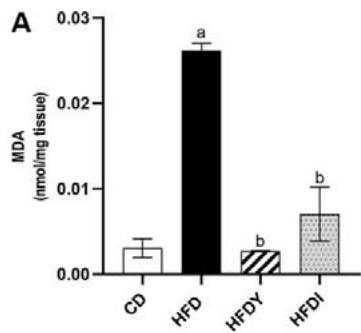
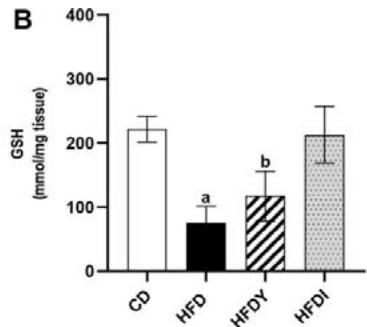
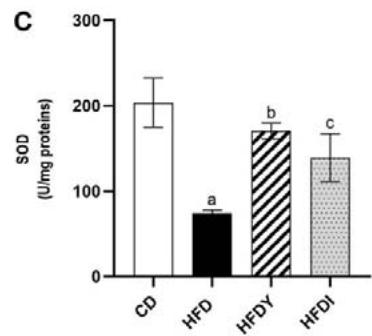
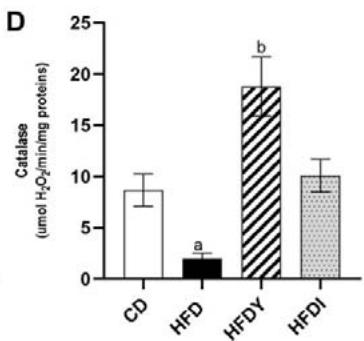
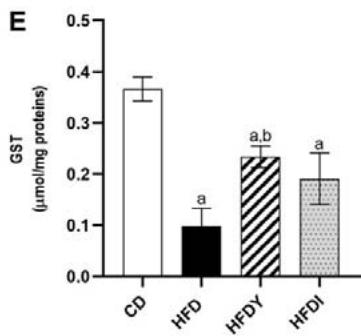 Figure 2: Effects of yacon flour and inulin on oxidative stress markers and antioxidant activity of visceral WAT of HFD-fed rats. (A) Malondialdehyde (MDA) and (B) reduced glutathione (GSH) levels; (C) superoxide dismutase (SOD); (D) catalase, and (E) glutathione S-transferase (GST) activities. The results are expressed as the mean $\pm$ SD. $(n = 6 / \text{group})$. $^a p < 0.05$ vs. CD; $^b p < 0.05$ vs. HFD; $^c p < 0.05$ vs. HFDY.
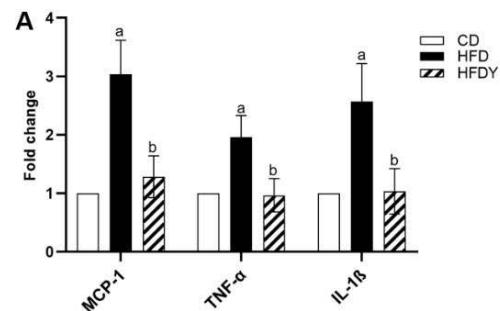
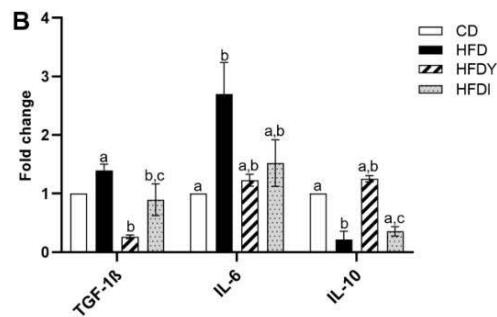
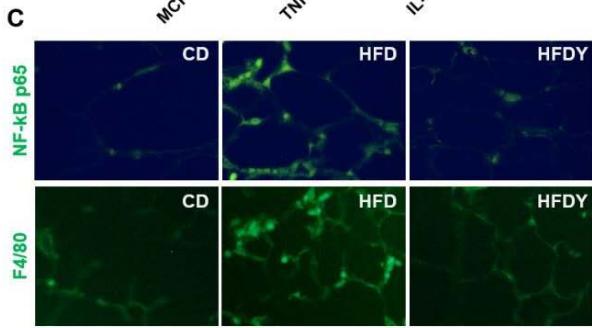
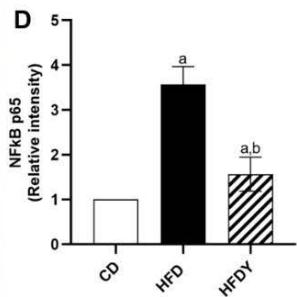
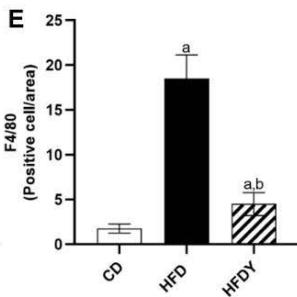 Figure 3: Effects of yacon flour and inulin in visceral adipose tissue inflammation of HFD-fed rats. Relative mRNA expressions of pro-inflammatory and anti-inflammatory cytokines (A) MCP-1, TNF- $\alpha$, IL-1 $\beta$, (B) TGF-1 $\beta$, and IL-6 IL-10. Data were normalized to Actin mRNA and expressed as fold change over the CD rats of triplicate RT-PCR analysis. (C) NF- $\kappa$ B p65 and F4/80 immunostaining in visceral WAT (magnification 400x). (D) NF- $\kappa$ B p65 relative intensity. (E) F4/80 positive cells count in visceral WAT. The results are expressed as the mean $\pm$ SD ( $n = 6$ /group). One-way ANOVA test (significant $p < 0.05$ ). a $p < 0.05$ vs. CD; b $p < 0.05$ vs. HFD; c $p < 0.05$ vs. HFDY
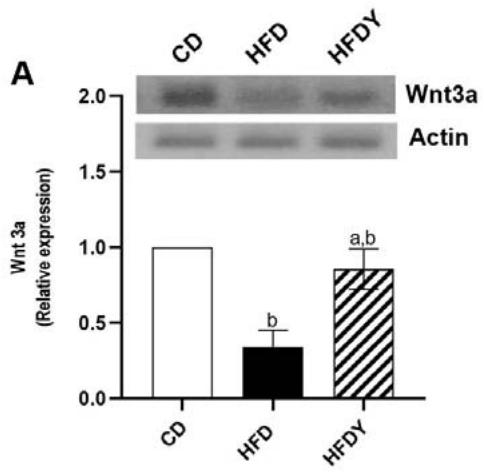
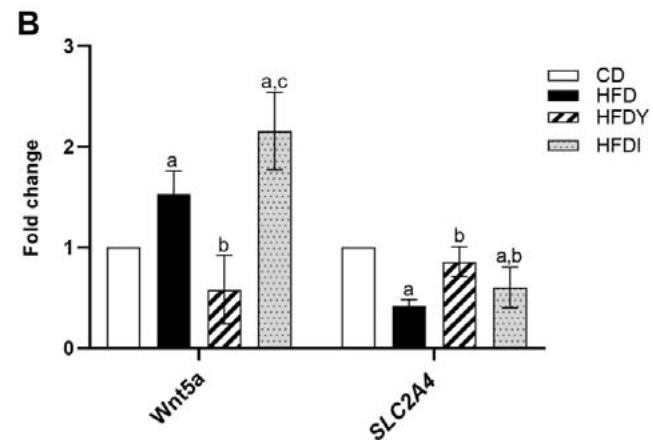 Figure 4: Effects of yacon flour and inulin on adipogenesis-related genes in HFD-fed rats. (A) Wnt3a protein expression in visceral fat by western blotting. (B) Relative mRNA expressions of Wnt5a paracrine factor and glucose transporter SLC2A4. Data were normalized to Actin mRNA and expressed as fold change over the CD rats. The results are expressed as the mean $\pm$ SD deviation of triplicate RT-PCR analysis ( $n = 6$ /group). One-way ANOVA test (significant $p < 0.05$ ).<sup>a</sup> $p < 0.05$ vs. CD; <sup>b</sup> $p < 0.05$ vs. HFD; <sup>c</sup> $p < 0.05$ vs. HFDY
Generating HTML Viewer...
References
43 Cites in Article
Zoi Michailidou,Mario Gomez-Salazar,Vasileia Alexaki (2021). Innate Immune Cells in the Adipose Tissue in Health and Metabolic Disease.
Yawen Zhou,Huige Li,Ning Xia (2021). The Interplay Between Adipose Tissue and Vasculature: Role of Oxidative Stress in Obesity.
J Rutkowski,J Stern,P Scherer (2015). The cell biology of fat expansion.
I Lempesis,R Meijel,K Manolopoulos,G Goossens (2019). Oxygenation of adipose tissue: A human perspective.
Atilla Engin (2017). Adipose Tissue Hypoxia in Obesity and Its Impact on Preadipocytes and Macrophages: Hypoxia Hypothesis.
S Kierans,C Taylor (2020). Regulation of glycolysis by the hypoxia‐inducible factor (HIF): implications for cellular physiology.
F Gonzalez,C Xie,C Jiang (2018). The role of hypoxiainducible factors in metabolic diseases.
C Jiang,A Qu,T Matsubara,T Chanturiya,W Jou,O Gavrilova,Y Shah,F Gonzalez (2011). Disruption of Hypoxia-Inducible Factor 1 in Adipocytes Improves Insulin Sensitivity and Decreases Adiposity in High-Fat Diet-Fed Mice.
Melina Konstantinidi,Antonios Koutelidakis (2019). Functional Foods and Bioactive Compounds: A Review of Its Possible Role on Weight Management and Obesity’s Metabolic Consequences.
D Davani-Davari,M Negahdaripour,I Karimzadeh,M Seifan,M Mohkam,S Masoumi,A Berenjian,Y Ghasemi (2019). Prebiotics: Definition, Types, Sources, Mechanisms, and Clinical Applications.
Yu-Ling Tsai,Tzu-Lung Lin,Chih-Jung Chang,Tsung-Ru Wu,Wei-Fan Lai,Chia-Chen Lu,Hsin-Chih Lai (2019). Probiotics, prebiotics and amelioration of diseases.
T Teferra (2021). Possible actions of inulin as prebiotic polysaccharide: A review.
A Márquez-Aguirre,R Camacho-Ruíz,Y Gutiérrez-Mercado,E Padilla-Camberos,M González-Ávila,F Gálvez-Gastélum,Díaz-Martínez Ne,D Ortuño-Sahagún (2016). Fructans from Agave tequilana with a Lower Degree of Polymerization Prevent Weight Gain, Hyperglycemiaand Liver Steatosis in High-Fat Diet-Induced Obese Mice.
La Rosa,S Kachrimanidou,V Buffetto,F Pope,P Pudlo,N Martens,E Rastall,R Gibson,G Westereng,B (2019). Wood-Derived Dietary Fibers Promote Beneficial Human Gut Microbiota.
Susana Genta,Wilfredo Cabrera,Alfredo Grau,Sara Sánchez (2005). Subchronic 4-month oral toxicity study of dried Smallanthus sonchifolius (yacon) roots as a diet supplement in rats.
Natalia Habib,Stella Honoré,Susana Genta,Sara Sánchez (2011). Hypolipidemic effect of Smallanthus sonchifolius (yacon) roots on diabetic rats: Biochemical approach.
S Genta,W Cabrera,N Habib,J Pons,I Carillo,A Grau,S Sánchez (2009). Yacon syrup: Beneficial effects on obesity and insulin resistance in humans.
Stella Honoré,Maria Grande,Jorge Gomez Rojas,Sara Sánchez (2018). <i>Smallanthus sonchifolius</i> (Yacon) Flour Improves Visceral Adiposity and Metabolic Parameters in High-Fat-Diet-Fed Rats.
E Novelli,Y Diniz,C Galhardi,G Ebaid,H Rodrigues,F Mani,A Fernandes,A Cicogna,J Novelli Filho (2007). Anthropometrical parameters and markers of obesity in rats.
J Beuge,S Aust (1978). The thiobarbituric acid assay.
G Ellman (1959). Tissue sulfhydryl groups.
Hiroyuki Ukeda,Susumu Maeda,Toshinao Ishii,Masayoshi Sawamura (1997). Spectrophotometric Assay for Superoxide Dismutase Based on Tetrazolium Salt 3′-{1-[(Phenylamino)-carbonyl]-3,4-tetrazolium}-bis(4-methoxy-6-nitro)benzenesulfonic Acid Hydrate Reduction by Xanthine–Xanthine Oxidase.
H Aebi,Catalase (1974). Methods of Enzymatic Analysis.
William Habig,Michael Pabst,William Jakoby (1974). Glutathione S-Transferases.
Ana Andrei,Anca Berbecaru-Iovan,Felix Din-Anghel,Camelia Stanciulescu,Sorin Berbecaru-Iovan,Ileana Banita,Catalina Pisoschi (2017). Interplay between Hypoxia, Inflammation and Adipocyte Remodeling in the Metabolic Syndrome.
Paul Trayhurn (2014). Hypoxia and Adipocyte Physiology: Implications for Adipose Tissue Dysfunction in Obesity.
S Mcmahon,M Charbonneau,S Grandmont,D Richard,C Dubois (2006). Transforming growth factor beta1 induces hypoxia-inducible factor-1 stabilization through selective inhibition of PHD2 expression.
Mi-Jeong Lee (2018). Transforming growth factor beta superfamily regulation of adipose tissue biology in obesity.
Qi Tang,Chang Chen,Yan Zhang,Minjia Dai,Yichen Jiang,Hang Wang,Mei Yu,Wei Jing,Weidong Tian (2018). Wnt5a regulates the cell proliferation and adipogenesis via MAPK‐independent pathway in early stage of obesity.
Twan De Winter,Roeland Nusse (2021). Running Against the Wnt: How Wnt/β-Catenin Suppresses Adipogenesis.
Xihai Kang,Yubing Xie,Douglas Kniss (2005). Adipose Tissue Model Using Three-Dimensional Cultivation of Preadipocytes Seeded onto Fibrous Polymer Scaffolds.
João Esteves,Caio Yonamine,Ubiratan Machado (2020). <i>SLC2A4</i> Expression and its Epigenetic Regulation as Biomarkers for Insulin Resistance Treatment in Diabetes Mellitus.
Veeraraghava Sharma,Manfred Keller,Andreas Weiler,Dieter Hunkler,Horst Prinzbach (1996). Von Pagodanen zu nichtpentagonalen (Homo)Dodecahedranen – der Undecacyclo[10. 10.10.0.0<sup>2, 20</sup>.0<sup>3, 10</sup>.0<sup>4, 19</sup>.0<sup>5, 9</sup>.− 0<sup>6, 18</sup>.0<sup>7, 15</sup>.0<sup>8, 13</sup>.0<sup>19, 22</sup>.0<sup>16, 21</sup>]docosan‐Käfig.
R Cossetti,S Tyldesley,C Speers,K Gelmon (2013). Abstract P3-12-05: The BC provincial experience on FEC-D vs. AC-taxane protocol as adjuvant treatment for breast cancer.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Maria Virginia Grande. 2026. \u201cYacon Flour: A Functional Food Ingredient with Impact on the Adipose 2 Tissue Remodeling of Obese Rats\u201d. Global Journal of Medical Research - L: Nutrition GJMR-L Volume 22 (GJMR Volume 22 Issue L1).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.