Biochemical Changes of Mancozeb-Induced AlteBiochemical Changes of Mancozeb-Induced Alternations in Testes and Thyroid gland of Male Ratsrnations in Testes and Thyroid gland of Male Rats
Mancozeb manganese ethylene bis dithiocarbamate polymeric complex -with zinc salts is a very important protective non-systemic fungicide, classified as an ethylene bis dithiocarbamate fungicide. Mancozeb used for controlling fungal diseases for a wide variety of crops because of its broad-spectrum as fungicidal effects and high compatibility with agrochemicals. The objective of this study was to investigate the results of the fungicide mancozeb at different doses on some biochemical parameters, reproductive performance, and histological changes in testes and thyroid gland. The low amount equals 1/7 of LD 50 manczeb-d 1 , and the high amount equals 1/3.5 of LD 50 mancozeb-d 1 . Amounts of mancozeb (mancozeb-d1 and mancozeb-d 2 ) adjusted according to the rat’s body weights. The results showed that mancozeb decreased plasma testosterone level, sperm count, viability, motility, and significantly (P
## I. INTRODUCTION
Mancozeb manganese ethylene bis dithiocarbamate polymeric complex with zinc salts is a very important protective non-systemic fungicide, classified as an ethylene bis dithio-carbamate fungicide. Ethylene bis (dithio-carbamate)s (EBDCs) are the most widely used classes of organic fungicides in the world because it can effect on abroad types of fungus, and high compatibility with agrochemicals[1]. The EBDCs registered for food uses in the United States are mancozeb, and zineb. They were first introduced during the 1940s are widely used. These compounds have low water solubility, which results in remaining on the surface of treated crops as superficial deposits[2]. Mancozeb consists of a zinc-rich shell surrounding a central nucleus of polymer-structured EBDC. This structure is highly stable, and the low solubility of the zinc shell means EBDC can pass through this layer and be deposited on the leaf surface[3]. Mancozeb is unstable in water and decomposed by light, heat, and moisture-producing; ethylene thiourea (ETU) and ethylene bis (isothiocyanate) sulphide (EBIS) and other degradation products such as glycine and ethylene urea (EU), which is further to $\mathrm{CO}_{2}$ under aerobic conditions. ETU is relatively stable and has a high solubility in water, so it can contaminate groundwater. ETU and EBIS are the main responsible compounds for the toxic effects linked to this fungicide group. ETU has teratogenic, carcinogenic, immunotoxic, and mutagenic effects, and EBIS is toxic and causes peripheral paralysis and thyroid dysfunction[4].
Ethylene bis isothiocyanate sulfide (EBIS), converted to ethylene bis isothiocyanate (EBI) by UV light. Both EBIS and EBI are active toxicants and can interfere with and inactivate sulphydryl groups in enzymes and amino acids, leading to enzymatic disruptions, and inhibition of spore germination[3, 5].
Mancozeb and its metabolites are widespread in the environment and have toxic effects due to their ingestion, inhalation, and percutaneous absorption by non- target organisms. Exposures to mancozeb are associated with a neurotoxic, developmental disability, immunotoxic, and carcinogenic effects in humans and experimental animals. Recent toxicological evidence has shown an endocrine- disruptive effect of mancozeb. It can disrupt the pituitary gland leading to decreasing the release of stimulating thyroid hormone (TSH) and thyroid hormones triiodothyronine and tetraiodothyronine. Moreover, it decreases thyroid hormone synthesis or action by directly interacting with nuclear hormone receptors, inhibiting thyroid peroxidase, and inhibiting iodine uptake. Also, it has toxic effects on endocrine systems, ovary, testes, and epididymis[6].
Experiments conducted on rodents have established that mancozeb and ETU can cross the placental barrier with significant potential to disrupt reproductive performance, cause DNA damage, and initiate tumors in fetal cells[7].
## II. MATERIALS AND METHODS
### a) Animals, experimental design and sampling
This study was approved by the Ethical Committee of the Institutional Animals Care and Use, Alexandria, Egypt, and met all guidelines for their use.
Mancozeb (85%) was obtained from the central agriculture pesticide laboratory. Eighteen healthy adult male rats (Rattus norvegicus) with an average weight of $(180\pm 10)\mathrm{g}$ were obtained from animal house, Faculty of Medicine, Alexandria University, and acclimated for two weeks before the experiment. They were assigned to 3 groups and housed in Universal galvanized wire cages at room temperature $(22 - 25^{\circ}C)$ and in a photoperiod of $12\mathrm{h/day}$. Animals have been provided with a balanced commercial diet containing, $18\%$ crude protein, $14\%$ crude fiber, $2\%$ fat, and $2600\mathrm{Kcal / Kg}$ feed.
Animals were divided randomly into three groups (6 animals each). Animals were maintained on food and water ad libitum. Doses of mancozeb have been prepared by dissolving in carboxy methylcellulose and adjusted according to the rat's body weights and given orally by gavages approximately at the same time each morning, three times per weekday after day for four weeks. Group I (control) was orally administered with carboxy methyl cellulose. Group II was orally administered with a dose equal to $700\mathrm{mg / Kg}$ body weight of mancozeb (1/7 LD50). Group III was orally administered with an amount equivalent to $1400\mathrm{mg / Kg}$ body weight of mancozeb (1/3.5 LD50).
The doses were chosen based on the previous study [8]. At the end of the treatment period, rats were sacrificed, blood was collected from the heart venacava, in heparinized tubes, and was centrifuged at $1,000 \times g$ for 15 min. Blood plasma was separated in Eppendorf tubes and stored at $-80^{\circ} \mathrm{C}$ till further investigations. Testes were isolated, weighed, then washed with saline and preserved at $-80^{\circ} \mathrm{C}$ for further biochemical studies. Parts of testes and thyroid glands used for histological studies kept in formalin (10%). The crude homogenates of the testes were prepared according to Greer [9].
### b) Testes homogenate biochemical assay and blood plasma enzymes and hormonal assay
Acid phosphatase (ACP; EC 3.1.3.2) was determined according to Daniel[10], Fructose was determined according to Foreman[11]. Enzyme-linked immunosorbent assay (ELISA) of testosterone was determined according to Nash[12], while T3 and T4 were determined according to Thakur[13] and TSH was determined according to Liu[14]. Biochemicals and hormonal kits have been purchased from BioSystems Company.
### c) Sperm collection and analysis
Immediately after decapitation, the rat's testes and epididymis were removed, cleaned from accessory tissues, and sperm collection was performed according to Trošić [15]. Sperm viability has been assessed by the eosin Y stain and the motility of sperm was assayed by the number of sperm that could move in a line. The percentage of viable sperm and the motility of sperm were calculated according to Wyrobek and Bruce [16]. The integrity of the acrosome was assessed using the Tejada acridine orange method [17, 18].
### d) Histological examination
Parts of testes and thyroid glands were fixed in $10\%$ formalin solution, embedded in paraffin wax, and cut with a microtome for $5\mu$ thick sections. The sections were stained by Hematoxylin and Eosin (H&E) stains and microscopically studied to evaluate their morphology[19].
### e) Statistical Analysis
The data were analyzed using a one-way analysis of variance (ANOVA) followed by Duncan's multiple comparisons. $P < 0.05$ was statically significant according to Norusis[20].
## III. RESULTS
a) Effects of Mancozeb on acid phosphatase (ACP) in blood plasma and testes homogenate and fructose level in testes homogenate of male rat
- The present study showed that, treatment of rats with mancozeb- $\mathsf{d}_1$ increased ACP activity in blood plasma when compared to the control group, while in the group treated with Mancozeb- $\mathsf{d}_2$, ACP activity decreased when compared to control. Furthermore, in testes, homogenate treatment of rats with Mancozeb- $\mathsf{d}_1$ and Mancozeb- $\mathsf{d}_2$ decreased ACP activity and fructose level when compared to the control group (Table1).
b) Effects of Mancozeb on testosterone and sperm quality
- Treatment of rats with Mancozeb decreased testosterone when compared to the control group. Also, Mancozeb decreased sperm motility, viability, and increased the number of total abnormal sperm, altered acrosome, and abnormal DNA when compared to the control group (Table 2).
c) Effects of Mancozeb on thyroid hormones in blood plasma
Results presented in Table 3 showed that, treatment of rats with Mancozeb- $\mathbf{d}_1$ and Mancozeb- $\mathbf{d}_2$ decreased T3 and T4 when compared to the control group. Treatment of rats with Mancozeb- $\mathbf{d}_2$ decreased
TSH when compared to the control group, while treatment with Mancozeb-d1 increased TSH when compared to the control group.
Table 1: Effects of Mancozeb on acid phosphatase (ACP) in blood plasma and testes homogenate and fructose level in testes homogenate of male rat
<table><tr><td rowspan="2">Parameters</td><td colspan="3">Groups</td></tr><tr><td>Control</td><td>Mancozeb-d1</td><td>Mancozeb-d2</td></tr><tr><td>ACP (IU/L)</td><td>70.54±1.15b</td><td>76.60±2.17a</td><td>57.54±1.54c</td></tr><tr><td>ACP (IU/g tissue)</td><td>172.37±5.81a</td><td>160.39±10.60b</td><td>134.93±4.26c</td></tr><tr><td>Fructose (mg/g tissue)</td><td>151.95±2.65a</td><td>120.97±3.88b</td><td>122.91±4.90b</td></tr></table>
Results expressed as Mean $\pm$ SE, $n = 6$
In Tables, the values denoted by different letters within the same row represent significant differences $(P < 0.05)$.
Mancozeb dose-1 (700mg/Kg) and Mancozeb dose-2 (1400mg/Kg), respectively
Table 2: Effects of Mancozeb on testosterone in blood plasma and sperm quality
<table><tr><td rowspan="2">Parameters</td><td colspan="3">Groups</td></tr><tr><td>Control</td><td>Mancozeb-d1</td><td>Mancozeb-d2</td></tr><tr><td>Testosterone (μg/dL)</td><td>3.93±0.114a</td><td>3.20±0.093b</td><td>3.03±0.088b</td></tr><tr><td>Motility (%)</td><td>76.20±0.583a</td><td>66.70±2.12b</td><td>60.40±2.56c</td></tr><tr><td>Viability (%)</td><td>71.00±0.548a</td><td>61.00±2.074b</td><td>54.00±2.793c</td></tr><tr><td>Abnormal sperms(%)</td><td>5.30±0.2b</td><td>7.20±0.490a</td><td>8.20±0.583a</td></tr><tr><td>Altered acrosome(%)</td><td>5.40±0.245b</td><td>7.40±0.510a</td><td>7.20±0.374a</td></tr><tr><td>Abnormal DNA(%)</td><td>6.20±0.583b</td><td>7.20±0.374b</td><td>11.20±0.490a</td></tr></table>
Results expressed as Mean $\pm$ SE, $n = 6$
In Tables, the values denoted by different letters within the same row represent significant differences $(P < 0.05)$.
Mancozeb dose-1 (700mg/Kg) and Mancozeb dose-2 (1400mg/Kg), respectively
Table 3: Effects of Mancozeb on thyroid hormones in blood plasma
<table><tr><td rowspan="2">Parameters</td><td colspan="3">Groups</td></tr><tr><td>Control</td><td>Mancozeb-d1</td><td>Mancozeb -d2</td></tr><tr><td>T3 (μg/dL)</td><td>619.8±18.04a</td><td>490.00±14.26b</td><td>481.0±14.00b</td></tr><tr><td>T4 (μg/dL)</td><td>0.530±0.015a</td><td>0.214±0.006b</td><td>0.235±0.007b</td></tr><tr><td>TSH (μg/dL)</td><td>0.466±0.014b</td><td>0.807±0.024a</td><td>0.415±0.012c</td></tr></table>
Results expressed as Mean $\pm$ SE, $n = 6$
In Tables, the values denoted by different letters within the same row represent significant differences $(P < 0.05)$.
Mancozeb dose-1 (700mg/Kg) and Mancozeb dose-2 (1400mg/Kg), respectively
### d) Effects of Mancozeb on histological changes in testes
Microscopic examination of control testes of male rats showed; typical testicular structure, normal spermatogonium cells (Spg) with its regular basophilic differentiated nuclei cells, normal spermatogenic (Figure 1). On the other hand, testes tissue of male rats treated with Mancozeb- $\mathsf{d}_1$ (700 mg/kg); revealed large basal vacuoles in the cytoplasm cells, necrotic (N) spermatocytes (SPC) reduced volume of mature spermatozoa (SP) in some tubules. Necrosis was observed in some tubules (N)(Figure 2). Meanwhile, treatment rats with Mancozeb- $\mathsf{d}_2$ (1400mg/kg) showed, dispatching of the tubular epithelium from primary spermatogonial (PSG) layer and reduction of the primary (PSP) and secondary (SSP) spermatids, with the presence of basal vacuoles (BV) (Figure 3).
### e) Effects of mancozeb on histological changes in the thyroid gland
Microscopic examination of the control thyroid gland of male rats showed; typical architecture with follicular cells (F) with colloid (C), parafollicular cells (C-cells) (Figure 4). Meanwhile, the thyroid gland of male rats treated with Mancoeb- $\mathsf{d}_1$ $700~\mathrm{mg / kg})$;showed vacuoles (V) in follicular epithelium, the follicular appeared flattened with flattened nuclei, reduction of colloid is evident; hyperplasia of parafollicular cells (C
Cells). Moreover, desquamation (d) of the follicular epithelium into the lumen of the thyroid follicles (Figure 5). On the other hand, treatment of rats with Mancozeb $\mathsf{d}_2$ $(1400\mathsf{mg / kg})$ showed interstitial edema (O), vacuolated erythrocytes (V), with variable size strands with irregular contours of the follicles (F), reduction of colloid in most follicles, hyperplasia of the follicular epithelium (Figure 6).
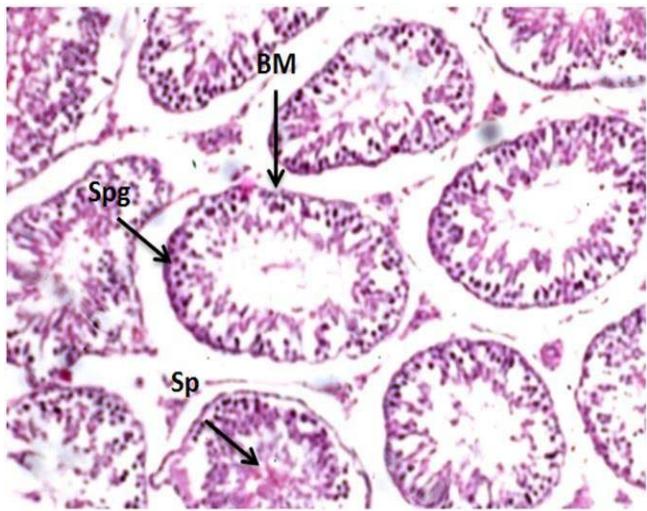 Figure (1): Photomicrographs of testes section of male rats control group (H&EX200)
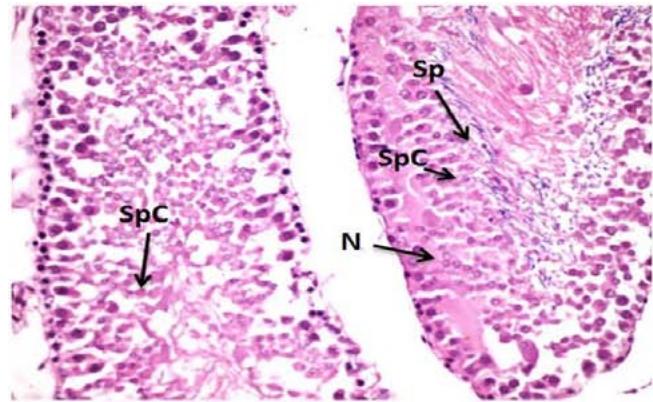 Figure (2): Photomicrographs of testes section of male rats treated with Manc-d1 (H&EX400)
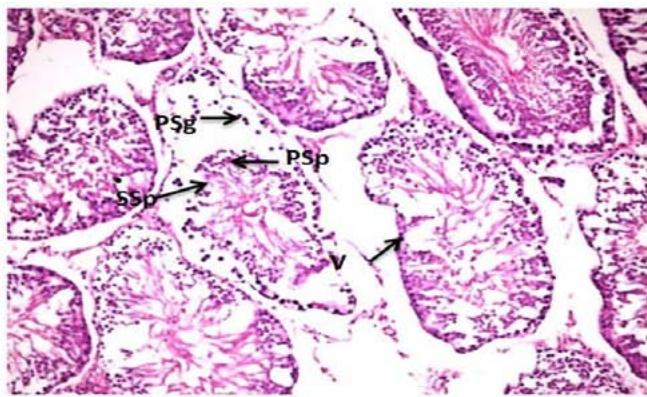 Figure (3): Photomicrographs of testes section of male rats treated with Manc-d2 (H&EX200)
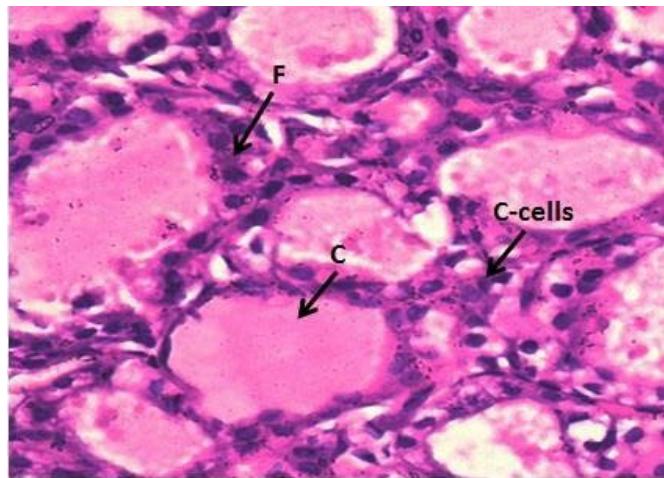 Figure (4): Photomicrographs of thyroid gland section of male rats control group (H&EX400)
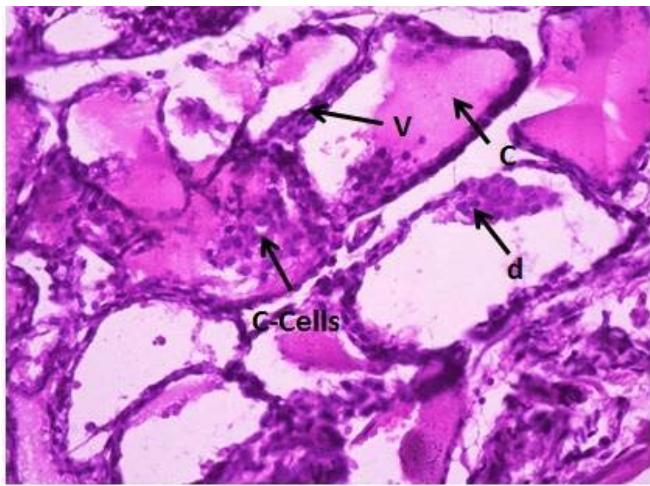 Figure (5): Photomicrographs of thyroid gland section of male rats treated with Manc-d1 (H&EX400)
## IV. DISCUSSION
Fructose has been used to indicate the seminal vesicle function, obstructive azoospermia, and inflammation of male accessory glands [21].
Increases in sperm concentration are often accompanied by a decrease in fructose level in seminal plasma, because sperm use fructose as the primary source of energy. However, other studies have also shown that fructose concentrations in seminal plasma of patients with oligozoospermia and azoospermia did not decrease as compared to ordinary men[22]. Fructose is produced endogenously within the human brain from glucose by the polyol pathway[23].
So, the present results revealed that, the decrease in fructose level means depression in the polyol pathway (fructose formation) and inactivity in cells due to treatment with mancozeb (manc).
Ananthan and Kumaran[24] showed that Mancozeb treatment (300 mg/kg body weight/day) for 60 days caused a significant increase in acid phosphatase in the testicular tissue of rats and increased activities in the serum. The decrease in acid phosphatase activity in the testes following the administration of Mancozeb could be attributed to either leakage of the enzyme into the extracellular fluid as a result of the disruption of the ordered lipid bilayer of the membrane or inhibition of the enzyme activity by this fungicide corresponding with the present results [24, 25, 26].
The decrease of testosterone might be responsible for the decreased sperm counts and motility and also morphological abnormality of testes. The insecticides may cause mitochondrial membrane impairment in Leydig cells and disrupt testosterone biosynthesis by diminishing the delivery of cholesterol into the mitochondria and decreasing the conversion of cholesterol to testosterone.
Acetamiprid-fed rats had fewer Leydig cells than regular diet-fed rats which may have been
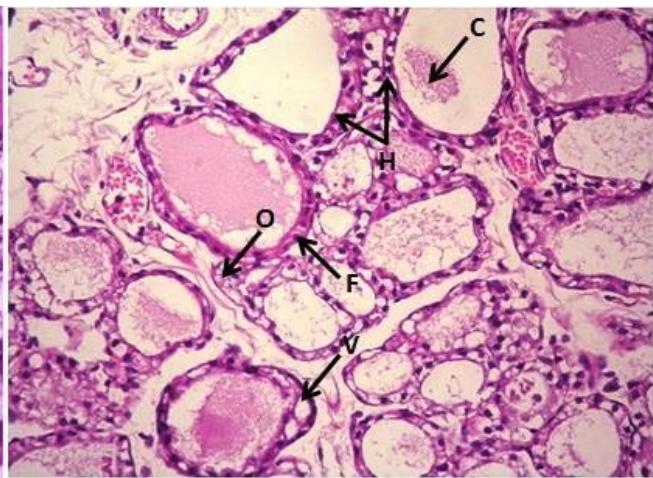 Figure (6): Photomicrographs of thyroid section of male rats treated with Manc-d2 (H&EX400) contributed to the reduction in testosterone biosynthesis[27].
Mancozeb and it's metabolites disturb endocrine gland action and its hormone secretion [6]. Treatment with mancozeb also changes the biochemical parameters of the reproductive tract. A fall in glycogen level may be due to interference in glucose metabolism. Fungicides induce inhibition of glycolytic enzymes, which affect the spermatozoa maturation and sperm motility. Inhibition of glycogen synthesis results in decreasing spermatogenesis process and reduction in serum testosterone[28].
Mancozeb has blocked the conversion of iodide to iodine. Inhibition in the iodide trapping and oxidizing process can lead to microscopic changes in the thyroid follicular cells and a reduced level of T4 [29]
The plasma TSH levels were increased upon chronic exposure to Mancozeb, indicating the usual negative feedback mechanism of the hypothalamus-pituitary- thyroid axis to low plasma T4 concentration. Chronic exposure to mancozeb has also been shown to reduce the synthesis and action of thyroid hormone through directly interacting with nuclear hormone receptors and inhibiting thyroid peroxidase and iodine uptake[6].
## V. CONCLUSION
Mancozeb has adverse effects on the hormonal system, showing thyroid hormones disruption and decreased testosterone level resulting in abnormal sperms, thus reducing fertility, also exposure to mancozeb causes histological changes in the testes and thyroid gland.
### ACKNOWLEDGEMENT
In the name of "Allah" the most beneficent and the most merciful, who gave me strength and knowledge to complete this dissertation
Firstly, I would like to express my sincere gratitude and appreciation to Prof. Dr. Nasser Gamal El din Mohamed, Chief. Researcher of Pathology, Animal Health Research Institute, Dokki, Giza. Nevertheless, I am also grateful to Prof. Dr. Fatma Mahmoud Darwish, Chief Researcher of Pathology, Animal Health Research Institute, Dokki, Giza, for their help and guidance in the histological studies throughout this study.
Generating HTML Viewer...
References
29 Cites in Article
O López-Fernández,E Pose-Juan,R Rial-Otero,J Simal-Gándara (2017). Effects of hydrochemistry variables on the half-life of mancozeb and on the hazard index associated to the sum of mancozeb and ethylenethiourea.
E Hwang,J Cash,M Zabik (2003). Determination of degradation products and pathways of mancozeb and ethylenethiourea (ETU) in solutions due to ozone and chlorine dioxide treatments.
M Gullino,F Tinivella,A Garibaldi,G Kemmitt,L Bacci,B Sheppard (2010). Mancozeb: past, present, and future.
Olalla López-Fernández,Remedios Yáñez,Raquel Rial-Otero,Jesús Simal-Gándara (2016). Kinetic modelling of mancozeb hydrolysis and photolysis to ethylenethiourea and other by-products in water.
Anna Pirozzi,Antonietta Stellavato,Annalisa La Gatta,Monica Lamberti,Chiara Schiraldi (2016). Mancozeb, a fungicide routinely used in agriculture, worsens nonalcoholic fatty liver disease in the human HepG2 cell model.
Daeho Kwon,Hwa-Kyoung Chung,Woon-Seob Shin,Yoon-Sun Park,Seong-Chun Kwon,Jae Song,Byong-Gon Park (2018). Toxicological evaluation of dithiocarbamate fungicide mancozeb on the endocrine functions in male rats.
Fiorella Belpoggi,Morando Soffritti,Marina Guarino,Luca Lambertini,Daniela Cevolani,Cesare Maltoni (2002). Results of Long‐Term Experimental Studies on the Carcinogenicity of Ethylene‐bis‐Dithiocarbamate (Mancozeb) in Rats.
Dennis Tierney (2005). Overview of Atrazine Interim Reregistration Eligibility Decision (IRED): Watershed Mitigation Components.
J Greer,J Foerster,J Lukens,Wintrobe (2003). s Clinical Hematology.
O Daniel,P Kind,E King (1954). Urinary Excretion of Acid Phosphatase.
Darhl Foreman,Lawrence Gaylor,Eric Evans,Cynthia Trella (1073). A modification of the Roe procedure for determination of fructose in tissues with increased specificity.
J Nash,B. Davail- Cuisset,S Bhattacharyya,H Suter,F Le Menn,D Kime (2000). An enzyme linked immunosorbant assay (ELISA) for testosterone, estradiol, and 17,20β-dihydroxy-4-pregenen-3-one using acetylcholinesterase as tracer: application to measurement of diel patterns in rainbow trout (Oncorhynchus mykiss).
C Thakur,T Saikia,R Yadav (1997). Total Serum Levels of Triiodothyronine (T3) Thyroxine (T4) and Thyrotropine (TSH) in School going Children of Dibrugarh District: An Endemic Goitre Region of Assam.
C Liu,D Hermsen,J Domberg,C Graeber,H Hautzel,Y Duan,K Xu,C Liu,X Mao,K Cupisti,W Scherbaum,M Schott (2008). Comparison of M22-based ELISA and Human-TSH-Receptor-based Luminescence Assay for the Measurement of Thyrotropin Receptor Antibodies in Patients with Thyroid Diseases.
Ivančica Trošić,Mirjana Mataušić-Pišl,Ivan Pavičić,Ana Marjanović (2013). Histological and Cytological Examination of Rat Reproductive Tissue After Short-Time Intermittent Radiofrequency Exposure.
A Wyrobek,W Bruce (1975). Chemical induction of sperm abnormalities in mice..
Rafael Tejada,J Mitchell,Amos Norman,Jaroslav Marik,Stanley Friedman (1984). A test for the practical evaluation of male fertility by acridine orange (AO) fluorescence.
J Erenpreiss,J Bars,V Lipatnikovai,J Erenpreisa (2001). Comparative study of cytochemical tests for sperm chromatin integrity.
R Drury,E Wallington (1980). Preparation and fixation of tissues.
M Norušis (2006). SPSS 14.0 guide to data analysis.
Ketaki Desai,Nilofer Moid,Pragnesh Patel,Hyacinth Highland (2016). Evaluation of Deltamethrin induced reproductive toxicity in male Swiss Albino mice.
N Trang,V Huyen,N Linh,T Sang (2018). Seminal Fructose Concentration in Man Infertility and the Fructose Test's Meaning In Diagnosis Reason of Azoospermia Man.
Janice Hwang,Lihong Jiang,Muhammad Hamza,Feng Dai,Renata Belfort-Deaguiar,Gary Cline,Douglas Rothman,Graeme Mason,Robert Sherwin (2017). The human brain produces fructose from glucose.
G Ananthan,B Kumaran (2013). Effects of mancozeb on the specific activities of testicular phosphatases and Protective role of vitamin C in Albino rats.
Y Adjrah,S Karou,A Agbonon,K Eklu-Gadegbeku,C De Souza,M Gbeassor (2013). Effect of mancozeb-treated lettuce (<i>Lactuca sativa</i>) on wistar rat liver.
E Yahia,M Aiche,A Chouabbia,M Boulakoud (2014). Subchronic mancozeb treatment induced liver toxicity via oxidative stress in male wistar rats.
Hesham Ibrahim,Sabah Banna,Ahmed Rafea (2010). Acetamiprid, Insecticide-Induced Oxidative Damage on Reproductive Parameters Male Rats.
S Joshi,N Gulati,A Gajraj (2005). Evaluation of toxic impacts of mancozeb on testis in rats.
Reena Kackar,Mithilesh Srivastava,Rajendra Raizada (1997). Induction of Gonadal Toxicity to Male Rats after Chronic Exposure to Mancozeb..
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Ahmed Abdelrafea. 2026. \u201cBiochemical Changes of Mancozeb-Induced AlteBiochemical Changes of Mancozeb-Induced Alternations in Testes and Thyroid gland of Male Ratsrnations in Testes and Thyroid gland of Male Rats\u201d. Global Journal of Medical Research - L: Nutrition GJMR-L Volume 22 (GJMR Volume 22 Issue L1).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.
Biochemical Changes of Mancozeb-Induced AlteBiochemical Changes of Mancozeb-Induced Alternations in Testes and Thyroid gland of Male Ratsrnations in Testes and Thyroid gland of Male Rats