## I. INTRODUCTION
Modern world requires people to adapt to the increased stress on the psyche associated with economic and political instability, social problems, and man-made and environmental factors, which together lead to the development of urban stress, accompanied by fatigue, irritability, tension and even unmotivated hatred and aggression [1].
When the body is under the influence of extreme environmental factors, both physiological shifts and psychological changes of varying degrees of severity can occur, with a pattern of manifestations commonly including a kind of "blockade" of cognitive processes, in which the volume of perception narrows, synthesis processes in thinking are disrupted, and purposeful behaviour becomes disorganized [2].
Accordingly, the discovery, development and use of drugs that increase stress resistance, resistance to pathogenic factors and work capacity and that activate mental activity, the ability to concentrate and learn, with minimal side effects, is an urgent task in pharmacology[8].
The domestic drug Seroxidol, injected as a 50 mg/ml solution, has pronounced antioxidant, antihypoxic and membranotropic effects.
Purpose of the study: This study pursued the development of a method for the quantitative determination of ethyl methyl hydroxypyridine succinate by UV spectrophotometry of the drug Seroxidol in a 50 mg/ml solution for injection and its bioequivalence.
## II. MATERIALS AND METHODS
Class A volumetric glassware was used in the work: conical flasks of $50 \mathrm{ml}$ and $100 \mathrm{ml}$, volumetric pipettes, an AS-220/X ser # B635963283 analytical balance (Ohaus, Germany), a SHIMADZU UV-1900UV spectrophotometer, and cuvettes with a thickness of 10 mm.
The object of the study was injection solutions corresponding to the TPA Seroxidol, a $50\mathrm{mg / ml}$ solution for injection. Determination was carried out by UV spectrophotometry.
The acute toxicity of the preparations was studied in 60 white mice of both sexes, weighing 19-21 g. From the compared preparations Seroxidol produced by LLC "MEDIOFARM", Uzbekistan and "Mexidol®" produced by LLC "Ellara", Russia, a $2.5\%$ solution was prepared and administered to the mice once intravenously at doses of $150 \mathrm{mg/kg}$, $175 \mathrm{mg/kg}$, $200 \mathrm{mg/kg}$, $225 \mathrm{mg/kg}$ and $250 \mathrm{mg/kg}$ (0.12-0.2 ml) [3].
The animals were kept under continuous observation during the first hour, then under hourly observation throughout the first day of the experiment and once a day for the next 13 days of the experiment.
As indicators of the functional state of animals, the general condition of the mice and their behaviour, the intensity and nature of the motor activity, the presence of seizures, coordination of movements, reaction to external stimuli and tone of skeletal muscles, appetite, body weight, and number and consistency of faecal masses were monitored[7].
During the experiment, the clinical state of the animals was monitored: the presence/absence of signs of poisoning, the time of their appearance, and death.
All experimental animals were kept under standard conditions on a common diet with free access to water and food [3].
After completion of the experiment, the average lethal doses (LD50) were determined [5].
## III. RESULTS AND DISCUSSION
Two millilitres of the medicine were placed in a volumetric flask with a capacity of $100\mathrm{ml}$, and the volume of the solution was brought up to the mark with 0.01 mol/l hydrochloric acid and stirred.
Next, $1.0 \mathrm{ml}$ of the resulting solution was placed in a $100 \mathrm{ml}$ volumetric flask, and the volume of the solution was brought to the mark with $0.01 \mathrm{~mol} / \mathrm{L}$ hydrochloric acid and mixed (test solution).
The optical density of the resulting solution was measured on a spectrophotometer at the absorption maximum at a wavelength of $297\mathrm{nm}$ in a cuvette with a layer thickness of $10\mathrm{mm}$, using $0.01\mathrm{mol/L}$ hydrochloric acid as a reference solution.
$$
X = \frac{D_{1}\alpha_{0}\times1\times100\times100\times P\times(100-W)}{D_{0}\times2\times1\times100\times1\times100\times100} = \frac{D_{1}\alpha_{0}\times P\times(100-W)}{D_{0}\times20000},
$$
where:
$D_{1}$ optical density of the test solution;
$D_{o}$ - optical density of the working standard solution of ethyl methyl hydroxypyridine succinate;
$A_{\mathrm{o}}$ - weighed portion of WS of ethyl methyl hydroxypyridine succinate, g;
P- content of the main ingredient ethyl methyl hydroxypyridine succinate in the WS of ethyl methyl hydroxypyridine succinate, $\%$;
W- moisture content in the WS of ethyl methyl hydroxypyridine succinate, in%.
The content of C12H47NO5 (ethyl methyl hydroxypyridine succinate) in 1 ml of the preparation should be from 0.045 to 0.055 g.
Preparation of WS of ethyl methyl hydroxypyridine succinate: Approximately 0.1 g (accurately weighed) of ethyl methyl hydroxypyridine succinate was placed in a volumetric flask with a capacity of $100 \, \text{ml}$, and $50.0 \, \text{ml}$ of $0.01 \, \text{mol/l}$ hydrochloric acid solution was added.
After the complete dissolution of the sample, the volume of the solution was brought to the mark with the same solvent and mixed. Then, $1.0 \mathrm{ml}$ of the resulting solution was placed in a $100 \mathrm{ml}$ volumetric flask, the volume of the solution was brought to the mark with the same solvent, and the solution was mixed. One millilitre of WS contained approximately $0.00001 \mathrm{~g}$ of ethyl methyl hydroxypyridine succinate.
The solution must be freshly prepared. The data obtained are presented in Figures 1, 2 and 3.
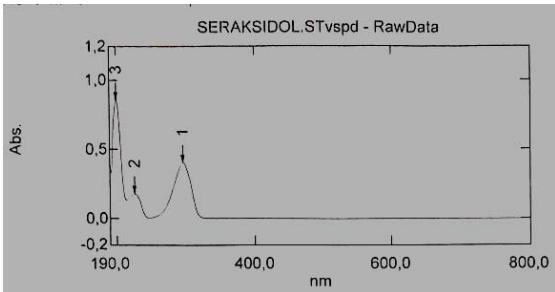
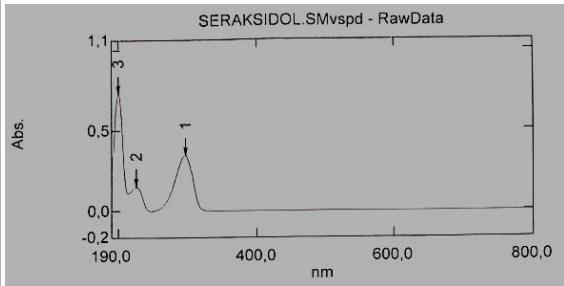
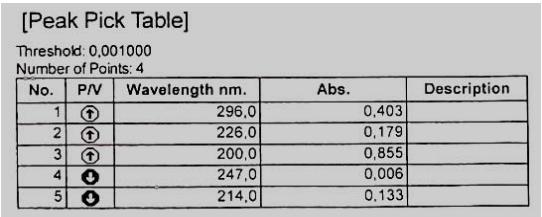
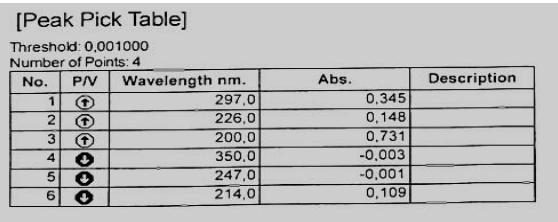 Fig. 1: Typical spectrum of a standard sample of the tested succinate Fig. 2: Spectrum of the product ethyl methyl hydroxypyridine, Seroxidol
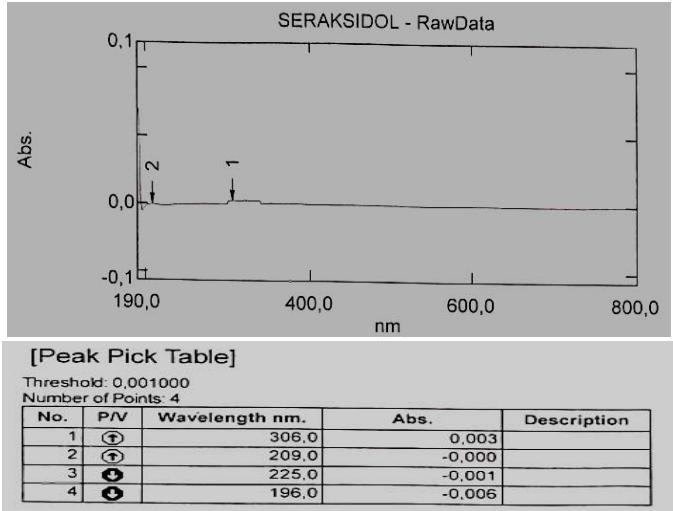 Fig. 3: Spectrum of the "placebo" solution
Table 1: The results of the quantitative determination of the medicine Seroxidol
<table><tr><td>X1,%</td><td>X̄,%</td><td>f</td><td>S2</td><td>S</td><td>Δ X̄</td><td>ε.%</td></tr><tr><td>X1=0.049</td><td>0.049</td><td>4</td><td>0.0000023</td><td>0.00152</td><td>0.0042</td><td>3.812</td></tr><tr><td>X2=0.047</td><td></td><td></td><td></td><td></td><td></td><td></td></tr><tr><td>X3=0.051</td><td></td><td></td><td></td><td></td><td></td><td></td></tr><tr><td>X4=0.050</td><td></td><td></td><td></td><td></td><td></td><td></td></tr><tr><td>X5=0.050</td><td></td><td></td><td></td><td></td><td></td><td></td></tr></table>
Experiments have shown that after a single intravenous administration of the Seroxidol preparation, produced by MEDIOFARM LLC, Uzbekistan, and Mexidol®, produced by Ellara LLC, Russia, at a dose of 150 mg/kg, no visible changes were observed in the behaviour and functional state of the animals.
All mice were active and responded to external stimuli, and food and water consumption was normal. No pathological changes in the hair and skin, diuresis, no diuresis, and no changes in the consistency and amount of faeces were observed. No signs of intoxication were observed. In this group, until the end of the experiment, no deaths were observed among the animals.
When the drugs were administered at a dose of $175\mathrm{mg/kg}$, the mice developed clonic-tonic seizures, 1 mouse died in the generic group, and 2 mice died in the comparison group. [4] When the drugs were administered at a dose of $200\mathrm{mg / kg}$, a decrease in motor activity, rapid breathing, impaired coordination of movements, a weakening of the reaction to external stimuli, and a decrease in food and water consumption were observed in experimental animals. Three mice died in the comparison group.
At a dose of $225\mathrm{mg / kg}$, the animals ceased to respond to external stimuli, and no food or water consumption was observed. During the experiment, 5 individuals in each group died [4].
The administration of a dose of $250 \, \mathrm{mg/kg}$ of either Seroxidol produced by LLC "MEDIOFARM",
Uzbekistan, or "Mexidol®" produced by LLC "Ellara", Russia, caused the immediate death of some animals after the administration of the medicine. By the end of the experiment, the condition of the surviving animals returned to normal as the signs of intoxication decreased. The LD50 of the drug Seroxidol produced by LLC "MEDIOFARM", Uzbekistan, was 200.0 (188.7 ÷ 211.1) mg/kg. The LD50 of the drug "Mexidol®" produced by LLC "Ellara", Russia, was 193.9 (178.5 ÷ 209.6) mg/kg. The acute toxicities of the compared drugs are shown in Table 2.
Table 2: Determination of the acute toxicity (LD50) of Seroxidol preparations produced by MEDIOFARM LLC, Uzbekistan, and Mexidol® manufactured by Ellara LLC, Russia
<table><tr><td rowspan="3">No animals</td><td colspan="5">Seroxidol produced by LLC "MEDIOFARM", Uzbekistan</td><td colspan="5">"Mexidol®" produced by LLC "Ellara", Russia</td></tr><tr><td rowspan="2">weight, g</td><td colspan="2">dose</td><td rowspan="2">method of administration</td><td rowspan="2">lethality</td><td rowspan="2">weight, g</td><td colspan="2">dose</td><td rowspan="2">way introduction</td><td rowspan="2">lethality</td></tr><tr><td>mg/kg</td><td>ml</td><td>mg/kg</td><td>ml</td></tr><tr><td>1</td><td>21</td><td rowspan="6">150</td><td>0.13</td><td rowspan="6">i/v</td><td>No</td><td>21</td><td rowspan="6">150</td><td>0.13</td><td rowspan="6">i/v</td><td>No</td></tr><tr><td>2</td><td>20</td><td>0.12</td><td>No</td><td>20</td><td>0.12</td><td>No</td></tr><tr><td>3</td><td>21</td><td>0.13</td><td>No</td><td>19</td><td>0.11</td><td>No</td></tr><tr><td>4</td><td>20</td><td>0.12</td><td>No</td><td>21</td><td>0.13</td><td>No</td></tr><tr><td>5</td><td>20</td><td>0.12</td><td>No</td><td>21</td><td>0.13</td><td>No</td></tr><tr><td>6</td><td>21</td><td>0.13</td><td>No</td><td>19</td><td>0.11</td><td>No</td></tr><tr><td>1</td><td>20</td><td rowspan="6">175</td><td>0.14</td><td rowspan="6">i/v</td><td>No</td><td>20</td><td rowspan="6">175</td><td>0.14</td><td rowspan="6">i/v</td><td>No</td></tr><tr><td>2</td><td>19</td><td>0.13</td><td>death</td><td>20</td><td>0.14</td><td>death</td></tr><tr><td>3</td><td>19</td><td>0.13</td><td>No</td><td>21</td><td>0.15</td><td>No</td></tr><tr><td>4</td><td>19</td><td>0.13</td><td>No</td><td>19</td><td>0.13</td><td>death</td></tr><tr><td>5</td><td>20</td><td>0.14</td><td>No</td><td>20</td><td>0.14</td><td>No</td></tr><tr><td>6</td><td>21</td><td>0.15</td><td>No</td><td>19</td><td>0.13</td><td>No</td></tr><tr><td>1</td><td>21</td><td rowspan="6">200</td><td>0.17</td><td rowspan="6">i/v</td><td>death</td><td>21</td><td rowspan="6">200</td><td>0.17</td><td rowspan="6">i/v</td><td>death</td></tr><tr><td>2</td><td>20</td><td>0.16</td><td>death</td><td>20</td><td>0.16</td><td>No</td></tr><tr><td>3</td><td>19</td><td>0.15</td><td>No</td><td>21</td><td>0.17</td><td>death</td></tr><tr><td>4</td><td>19</td><td>0.15</td><td>death</td><td>19</td><td>0.15</td><td>No</td></tr><tr><td>5</td><td>20</td><td>0.16</td><td>No</td><td>21</td><td>0.17</td><td>No</td></tr><tr><td>6</td><td>20</td><td>0.16</td><td>No</td><td>21</td><td>0.17</td><td>death</td></tr><tr><td>1</td><td>19</td><td rowspan="6">225</td><td>0.17</td><td rowspan="6">i/v</td><td>death</td><td>20</td><td rowspan="6">225</td><td>0.18</td><td rowspan="6">i/v</td><td>death</td></tr><tr><td>2</td><td>19</td><td>0.17</td><td>No</td><td>21</td><td>0.19</td><td>death</td></tr><tr><td>3</td><td>20</td><td>0.18</td><td>death</td><td>20</td><td>0.18</td><td>death</td></tr><tr><td>4</td><td>20</td><td>0.18</td><td>death</td><td>21</td><td>0.19</td><td>No</td></tr><tr><td>5</td><td>21</td><td>0.19</td><td>death</td><td>19</td><td>0.17</td><td>death</td></tr><tr><td>6</td><td>21</td><td>0.19</td><td>death</td><td>21</td><td>0.19</td><td>death</td></tr><tr><td>1</td><td>20</td><td rowspan="6">250</td><td>0.20</td><td rowspan="6">i/Av</td><td>death</td><td>21</td><td rowspan="6">250</td><td>0.21</td><td rowspan="6">i/v</td><td>death</td></tr><tr><td>2</td><td>1921</td><td>0.19</td><td>death</td><td>2120</td><td>0.21</td><td>death</td></tr><tr><td>3</td><td>20</td><td>0.21</td><td>death</td><td>19</td><td>0.20</td><td>death</td></tr><tr><td>4</td><td>2019</td><td>0.20</td><td>death</td><td>2121</td><td>0.19</td><td>death</td></tr><tr><td>5</td><td></td><td>0.20</td><td>death</td><td></td><td>0.21</td><td>death</td></tr><tr><td>6</td><td></td><td>0.19</td><td>death</td><td></td><td>0.21</td><td>death</td></tr><tr><td colspan="2">\(LD_{50}\)</td><td colspan="4">200.0 (188.7÷211.1) mg/kg</td><td colspan="5">193.9 (178.5÷209.6) mg/kg</td></tr></table>
## IV. CONCLUSION
This method was carried out in accordance with the requirements of the TPA. One millilitre of the drug Seroxidol contained 0.049 mg of ethyl methyl hydroxypyridine succinate.
Thus, the data obtained show that the preparations Seroxidol (50 mg/ml solution for injection) produced by "MEDIOFARM" LLC (Uzbekistan) and "Mexidol®" (50 mg/ml solution for injection) produced by "Ellara" LLC, (Russia) at 5 ml each are biologically equivalent in terms of acute toxicity.
Generating HTML Viewer...
References
8 Cites in Article
A Barkovskaya,Yu,M Nazarova (2014). Stress factors in the socio-cultural space of a modern large kind // Izvestia Volg GTU.
A Barkovskaya,Yu,M Nazarova (2014). Stress factors in the socio-cultural space of the modern large genus.
G Zaitsev (2014). Living Under Extreme Conditions.
G Zaitsev (2014). Extreme conditions of professional activity: concept, content and classification.
D Sarvarova,N Yunuskhodjaeva,R Mavlanov Sh Study of the bioequivalence of the drug "Seroxidol". Infection, immunity and pharmacology.
Q Ubaydullayev,A Mukhitdinov (2017). Series Page.
Z Marchenko,M Balcezhin (2014). Methods of spectrophotometry in the UV and visible regions in inorganic analysis.
S Stanner,J Hughes,Cnm Kelly,J Buttriss (2004). A review of the epidemiological evidence for the ‘antioxidant hypothesis’.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Surayyo Z. Yuldasheva. 2026. \u201cQuantitative Determination of Ethyl Methyl Hydroxypyridina Succinate in the Preparation Seroxidol and its Bioequivalence\u201d. Global Journal of Medical Research - B: Pharma, Drug Discovery, Toxicology & Medicine GJMR-B Volume 22 (GJMR Volume 22 Issue B2).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.