Background: Hypovitaminosis D is commonly prevalent in older population world over leading to increased levels of bone resorption, reduced bone mass, often resulting inosteoporosis and increased chances of falls and fractures. In children, vitamin D deficiency is known to cause rickets due to hypomineralization of bone.
## I. INTRODUCTION
Hypovitaminosis D is widespread and represents a serious health problem globally. Approximately one billion people worldwide suffer from vitamin
D deficiency and nearly $50\%$ of world population is vitamin D insufficient [1] It was discovered about a 100 years ago that ultra violet radiations of the sun stimulates the conversion of cholesterol into vitamin D. Being a prohormone, vitamin D plays a vital role in controlling calcium an phosphorus metabolism, and thus is essentially crucial detriment of bone health in people of all age groups. Aging is closely associated with changes in lifestyle, biological and socio-economic changes. Studies have indicated that dermal capacity to synthesize the vitamin in people after 65 years is estimated to be about $25\%$ of that in people of ages 20 - 30 years on exposure to the same amount of sunlight [2]. This reduction is related to the reduction in the concentration of skin 7- dehydrocholesterol. Other secondary factors which contribute to lower concentration of vitamin D, in elderly people, include decreased physical activity, reduced exposure to sun as more time is spent indoors, or increased use of sunscreen lotions [3].
The role of vitamin D as mineral supplier in normal bone and dental development is well established [4]. Calcium is absorbed from the gut when active vitamin D metabolite $1,25(\mathrm{OH})_{2}\mathrm{D}$ opens up calcium channels in the gut, stimulating the formation of calcium binding protein in the intestinal cells (Fig. 1). Once sufficient vitamin D and calcium are available, optimal circumstances for bone mineralization are created. However, when there is vitamin D deficiency the $1,25(\mathrm{OH})_{2}\mathrm{D}$ concentration drops, as a result, less calcium is available for bone mineralization (Fig.2). the parathyroid hormone(PTH) level increases resulting in hydroxylation of $25(\mathrm{OH})\mathrm{D}$ in the kidney to $1,25(\mathrm{OH})_{2}\mathrm{D}$. Increased PTH level in turn stimulates bone turnover, leading to bone loss [5]. When there is prolonged vitamin D deficiency, bone loss is increased, thus leading to osteoporosis. Higher turnover bone means more osteoid tissue (the bone which has not yet mineralized) which causes more than normal remodeling on the bone surface. As a result, the mineralized bone contains less mineral as mineral accumulation takes place upto two years after osteon formation. In case of severe prolonged vitamin D deficiency the volume of osteoid tissue accumulates to more than $5\%$ leading to osteomalacia. Generalized and progressive decrease of skeletal muscle mass and strength are the major musculoskeletal consequences associated with vitamin D deficiency. Hypovitaminosis in
older individuals directly interferes with changes in musculoskeletal system [6]
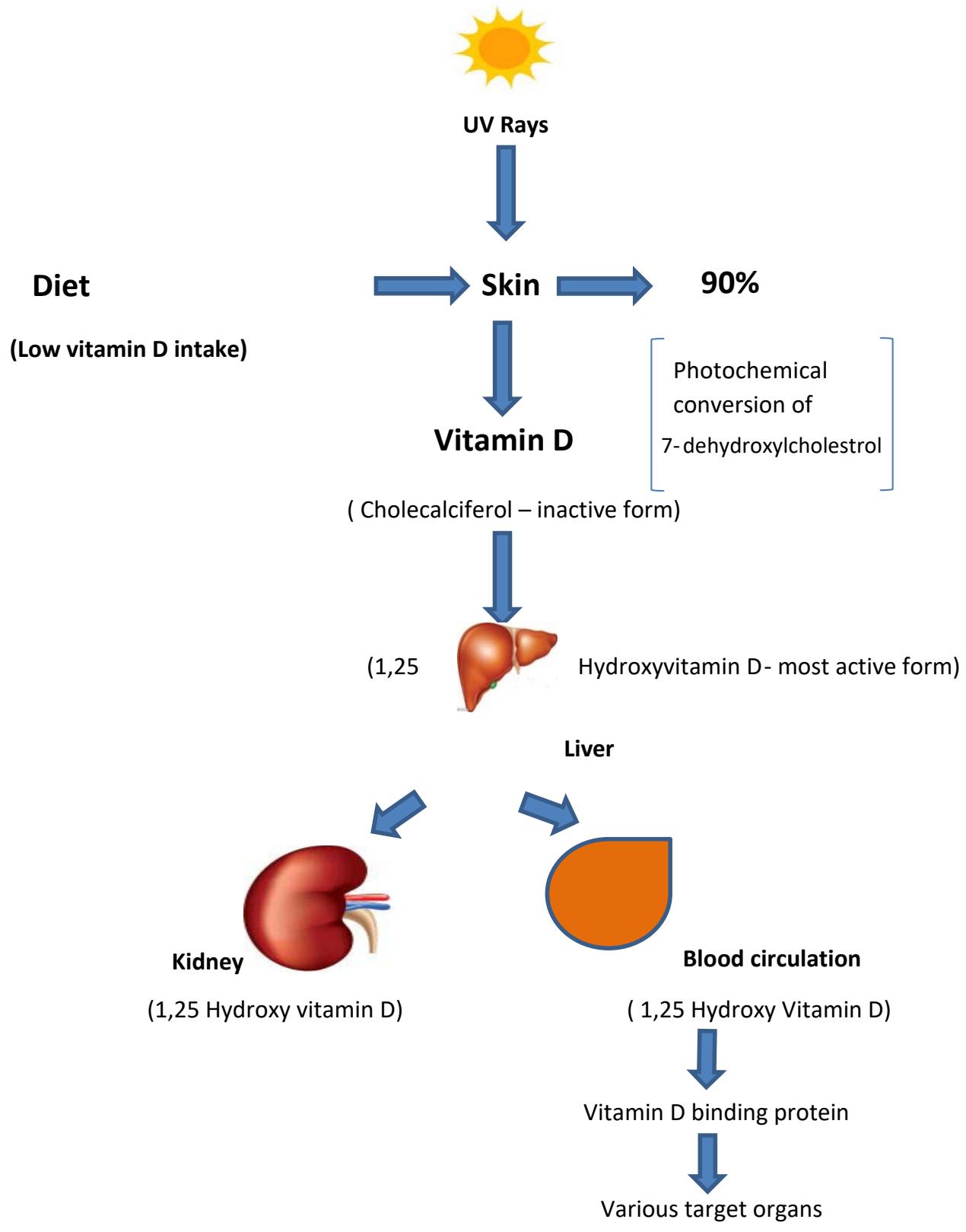 Figure 1: Vitamin D synthesis in the body in normal healthy adult.
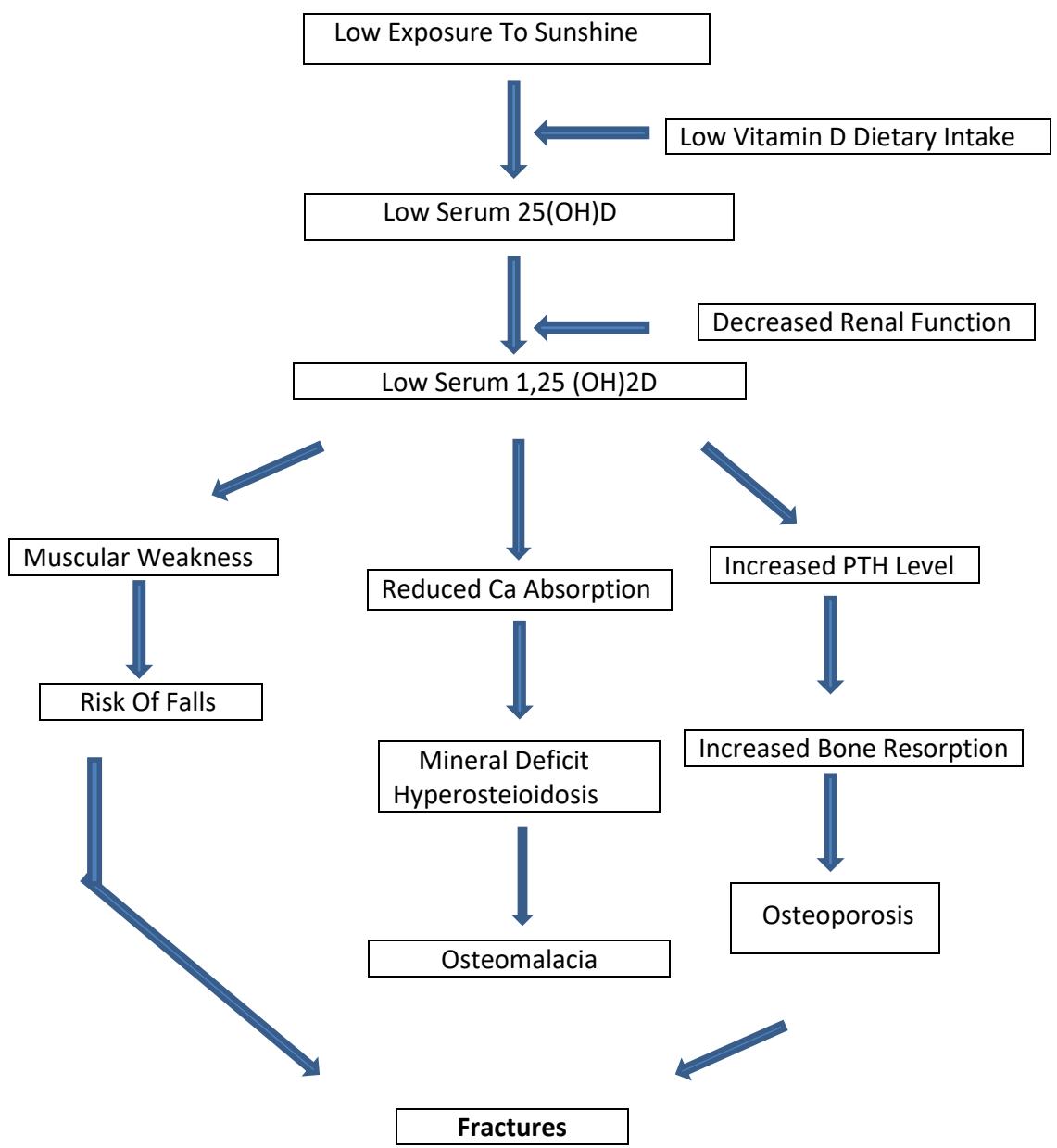 Figure 2: Pathophysiologic pathways of vitamin D deficiency causing osteoporosis, osteomalacia, falls and fractures.
Among children, the most severe form of vitamin D associated disease is Rickets. Rickets occurs when hypocalcemia and/or hypophosphatemia affect the development of the epiphyseal growth plate and is most common in infancy. Nutritional rickets is most common in children in the Middle East, Africa, and South Asia and its prevalence is now increasing in high-income countries, largely governed by inflow of immigrant populations.
## II. PURPOSE OF REVIEW
Osteoporosis, osteomalacia and rickets are common musculoskeletal diseases associated with vitamin D deficiency among aging global populations and children irrespective of race, region, sex, age and ethnicity. Changing population demographics, ill planned implementation strategies, and weak prevention policies are the chief hurdles in overcoming the vitamin D associated complications. This article reviews sustainable prevention strategies and identifies areas of future research.
## III. MATERIALS AND METHODS
We searched PubMed, EMBASE, Cochrane Library and Google Scholar databases using MeSH terms "vitamin D", "cholecalciferol", "osteoporosis", "osteomalacia", "Rickets" using logical operators "AND" or "OR". We selected observational studies and RCTs of risk of fractures and change in BMD associated with vitamin D supplementation Vs. placebo or recommended dose Vs. high dose alone or in combination with calcium. Literature search was conducted from January 2022 to May 2022. Two reviewers were involved in data acquisition, two were involved in quality assessment for their inclusion in proposed observational studies and one reviewer combined the results of earmarked studies to obtain the summary of effects.
### a) Vitamin D and Osteoporosis
Global statistics indicate that more than 200 million people are currently suffering from osteoporosis [7], a metabolic bone disease with low bone density and weak musculoskeletal architecture increasing the risk of fractures due to inadequate bone mineralization and decreased bone strength [8]. With more than 8.9 million fractures per year, osteoporosis is a worldwide concern. Although osteoporosis is typically associated with postmenopausal women, it also occurs in men with low BMD [9]. One in five American males have osteoporosis or low BMD [10]. The condition often remains undiagnosed due to being asymptomatic until it exhibits a fracture of the hip, pelvis, humerus (proximal), spine or wrist requiring hospitalization [11]. In addition, elderly people can also become bedridden thus leading to serious outcomes including mortality [12].
Primary Osteoporosis: Primary osteoporosis is associated with age and sex hormone deficiency. Age related osteoporosis is the resultant of continuous deterioration trabeculae in bone. Moreover, there is a significant increase in bone loss due to the reduced production of estrogen in post-menopausal women. In men, as aging occurs, testosterone and estrogen are inactivated by sex hormone binding globulin, thereby contributing to decrease in BMD with time [10,12,13,14]
Secondary osteoporosis: Several comorbid diseases and medications cause secondary osteoporosis. This may be attributed to the imbalance in regulatory mechanisms of calcium, vitamin D and hormones[10,15]. In addition, when patients are suffering from some inflammatory diseases such as rheumatoid arthritis, they are put on long-term glucocorticoid therapy and have been associated with secondary osteoporosis [14,16]. For men, excessive use of alcohol and hypogonadism are more commonly associated with osteoporosis [17]. Whereas, osteoporosis in $32.4\%$ women was attributed to hypercalcaemia, malabsorption of calcium, hyperparathyroidism, Cushing's disease, and hypocalciurichypercalcemia [18]. However, disorders of calcium metabolism and hyperparathyroidism contributed to $78\%$ of secondary causes [19].
### b) Vitamin D and Rickets
During childhood and adolescence hypovitaminosis D is mainly attributed to poor and unbalanced diet. During the initial years of life, absence of adequate sun exposure or vitamin D supplementation is an important risk factor for vitamin D deficiency [20], while, in adolescence, fast food and junk food are the relevant risk factors. Signs and symptoms associated with rickets include bowing of legs, knock knees, rachitic rosary, muscle weakness, seizures, tetany and cardiomyopathy. Radiographic findings reveal cupping, fraying and splaying of metaphysis near the epiphyseal growth plate. Muscle weakness and muscle pain has also been described in vitamin D deficiency associated bone disease [21,22,23].
90% of adult bone mineralization is accumulated by the end of adolescence [24]. Furthermore, vitamin D status in adolescence may be of great significance because 40% of adult bone mineralization occurs within this time of peak bone growth velocity [25]
## IV. RESULTS AND DISCUSSION
Vitamin D status is related to BMD in vitamin D deficient as well as vitamin D insufficient subjects. In this review article, several studies exploring the relationship of serum 25(OH)D and BMD have been described and analyzed. In addition, the cross sectional studies conducted to find the effect of vitamin D supplementation on bone turnover and prevention of fractures have also been discussed to affirm the effect of vitamin D supplementation on bone health in children as well as in the aging population. Some meta-analysis show that calcium should be added to vitamin D in order to be effective in reducing the risk of hip fracture [26]. Whereas, another meta-analysis of calcium supplementation studies suggested that co supplementation may increase the risk of cardiovascular disease. [27].
According to one study [28], men and women between ages 50-55 years were supplemented with vitamin D3 (2000 IU/day for 2 years) without calcium, compared with placebo did not significantly benefit bone density or structure in this study. Supplemental vitamin D3 did not increase BMD or prevent bone loss at the spine, hip or whole body. It also did not improve or adversely affect total, trabecular, or cortical BMD, cortical thickness or bone strength at the radius or tibia as compared to placebo.
Another RCT study [29] consisting of $53\%$ men and $47\%$ women with average age of 62.2 years were administered with vitamin D for 3 years at a dose of 4000 IU/day or 10000 IU/day compared with 400 IU/day resulted in statistically significant lower radial BMD, tibial BMD was significantly lower only with 10000 IU/day dose. No significant differences were reported in bone strength at either the radius or tibia. These findings do not support the benefit of high dose of vitamin D supplementation for bone health.
According to still another RCT conducted in England [30], three different doses of vitamin D (12000, 24000, 48000 IU/ month), were tested for their effect on BMD on individuals $>70$ years of age for one year. No difference in BMD between three doses of vitamin D suggests no effect of the intervention. However, parathyroid hormone concentrations decreased in all three groups, with significantly greater decrease in 48000 IU group $(p < 0.01)$. The treatment was safe and effective in increasing plasma 25(OH)D concentrations with no dose related adverse effects. Further, two RCTs that assessed very high annual doses of vitamin D, both showed an increase in the rise of fractures and falls among those allocated to vitamin D group [31,32] reinforcing the conclusion that intermittent dosing regimens with high dose of vitamin D cause toxic effects.
A number of observational studies of blood 25(OH)D concentration and risk of fracture demonstrated that higher blood 25(OH)D concentrations were associated with lower risks of any fracture and hip fracture. An increase of $10.0\mathrm{ng / ml}$ in 25(OH)D concentration was associated with a $7\%$ lower risk of any fracture and $20\%$ lower risk of hip fracture [33]. The same comparative study of RCTs demonstrated that vitamin D and calcium demonstrated a marginally significant reduction in the risk of any fracture of $6\%$ and hip fracture of $16\%$. However, $95\%$ CIs indicated some uncertainty of these estimates. As with RCTs of vitamin D supplementation alone, this study demonstrated no beneficial association with risk of fracture. However, elucidation of the results of these RCTs is restricted by their small sample size, short treatment duration, high risk of bias (chiefly because of incomplete follow up of outcomes), fragmented dosing regimen of vitamin D, and failure to achieve adequate differences in 25(OH)D concentrations.
Randomized controlled trials of vitamin D supplementation to optimize bone health have been performed in adolescents and mostly in females. Two review studies (34, 35) by the same authors concluded that vitamin D supplementation demonstrated no significance effect on total body mineral content (BMC) or bone mineral density of hip or fore arm. Another study in girls who were less than 2 years past menarche showed improvement in total body and lumbar spine BMC with vitamin D supplementation [36].
Another area of potential consequence of vitamin D deficiency in pediatric bone health is the risk for fracture. One study has examined the potential association of vitamin D deficiency and risk of fractures in children [37], while a cross-sectional study of 10 to 16 years old children, those with upper limb fracture, lower limb fracture and no fracture demonstrate no significance difference in 24(OH)D status [38]. In a case control study of 5 to 9 years old African American children, compared to 74 controls, the 76 cases exhibited 3.64 (95% CI 1.09 - 10.94) higher odds of vitamin D deficiency [39]. A study of children under 2 years of age who were admitted with fractures, 11 of 79 demonstrated hypomineralization on skeletal findings. For every 10 point increase in vitamin D status, the adjusted odds of hypomineralization were reduced 0.3 (95% CI 0.17 - 0.82) [40]. According to a recent RCT study findings, vitamin D sufficient children, at the age of 6 months, from mothers receiving high dose of vitamin D supplementation (2800 IU/day) during pregnancy had a $60\%$ reduced incidence of fractures compared with vitamin D insufficient children from mothers receiving standard dose (400IU/day) [41]. Same study also found that serum 25(OH)D concentration indicate whole body mineralization was higher in vitamin sufficient children at age 6 years, with the greater effect in vitamin D children from mothers receiving high dose of vitamin D. Thus indicating that childhood vitamin D sufficiency improved bone mineralization and in combination with pre-natal high dose vitamin D supplementation reduced the risk of fractures.
## V. CONCLUSION
Randomized clinical trials have demonstrated that vitamin D with or without calcium can increase BMD, decrease bone turnover and subsequently decrease fracture incidence. It is not known whether vitamin D is effective in vitamin D deficient or insufficient older individuals, or in complete older population. The dose response relationship is unclear. Meta-analysis of ongoing studies assessing the effects of higher daily doses of vitamin D on fracture risk are needed before making recommendations on the use of vitamin D for prevention of fractures. Some meta analysis favor the addition of calcium to vitamin D regimen. However further trials are needed to assess the efficacy and safety of higher daily doses of vitamin D with calcium for fracture prevention among older individuals with frailty or among other high risk groups with low vitamin D status. Due to the availability of limited data of the association of vitamin D with osteoporosis, osteomalacia and fracture risk, further exploration is required especially for vitamin D deficient high fracture risk population. Another question that needs to be addressed is whether vitamin D should be prescribed to all elderly or tailored to risk groups. Having said that, the optimal dose may differ between individuals i.e in case of different genetic polymorphism, chronic diseases and co- medication.
As the amount of research investigating vitamin D needs of children, unfortunately, the answers have become more unclear. Vitamin D deficient rickets is a disease with severe morbidity that responds well to vitamin D supplementation. Osteomalacia and bone hypomineralization, not of the magnitude of rickets, is more difficult to be diagnosed, therefore study of its response to vitamin D supplementation is challenging. Observational studies in children point to at least $10\mu \mathrm{g}/$ day vitamin D supplementation to achieve optimal bone health, but results of RCTs have not been clear. Vitamin D have been found to play a significant role in immune function especially in auto immune functions, infections, and allergic disease, but again the trials of its supplementation have little to prove the hypothesis. Due to these study results and other related issues, national and international guidelines are being restructured to reflect this ambiguity and provide less injunction regarding vitamin D supplementation after infancy. Attention to standard 25(OH)D concentration and investigation of genetic or other individual variations in vitamin D metabolism hopefully will identify the cause of these discrepancies in research results.
Funding: The funding organizations played no role in the study design; in the collection, analysis, and interpretation of data; in writing the review; or in the decision to submit the review for publication.
Conflict of Interest: None declared.
Ethical Approval: Not required.
Generating HTML Viewer...
References
41 Cites in Article
J Maclaughlin,M Holick (1985). Aging decreases the capacity of human skin to produce vitamin D3..
M Holick,L Matsuoka,J Wortsman (1989). Age, vitamin D, and solar ultraviolet.
M Holick (1994). Mccollum award lecture, 1994: Vitamin D -new horizons for the 21 st Century.
(1924). Scientific Opinion on the substantiation of a health claim related to vitamin D and contribution to normal bone and tooth development pursuant to Article 14 of Regulation (EC) No 1924/2006.
P Lips (2001). Vitamin D Deficiency and Secondary Hyperparathyroidism in the Elderly: Consequences for Bone Loss and Fractures and Therapeutic Implications.
M Urbanska,P Pludowski,E Suchowierska (2021). Vitamin D deficiency in older patients -Problems of sarcopenia, drug interactions, management in deficiency.
Nicole Wright,Anne Looker,Kenneth Saag,Jeffrey Curtis,Elizabeth Delzell,Susan Randall,Bess Dawson-Hughes (2014). The Recent Prevalence of Osteoporosis and Low Bone Mass in the United States Based on Bone Mineral Density at the Femoral Neck or Lumbar Spine.
(2004). Executive Summary: Mental Health: A Report of the Surgeon General.
Russel Burge,Bess Dawson-Hughes,Daniel Solomon,John Wong,Alison King,Anna Tosteson (2007). Incidence and Economic Burden of Osteoporosis-Related Fractures in the United States, 2005–2025.
L Raisz (2005). Pathogenesis of osteoporosis: concepts, conflicts, and prospects.
F Cosmon,S De Beur,M Leboff,F Lewiecki,B Tanner,S Randall (2014). Clinician's guide to prevention and treatment of osteoporosis.
M Jeremiah,B Unwin,M Greenawald,E Vincent (2015). Diagnosis and Management of osteoporosis.
(2010). Management of osteoporosis in postmenopausal women: 2010 Position Statement of The North American Menopause Society.
D Hunter,P Sambrook (2000). Bone Loss: Epidemiology of bone loss.
Zh Belaya (2015). WEEK OF OSTEOPOROSIS AND OTHER METABOLIC BONE DISEASES AT ENDOCRINOLOGY RESEARCH CENTER RUSSIAN MINISTRY OF HEALTH.
Lenore Buckley,Gordon Guyatt,Howard Fink,Michael Cannon,Jennifer Grossman,Karen Hansen,Mary Humphrey,Nancy Lane,Marina Magrey,Marc Miller,Lake Morrison,Madhumathi Rao,Angela Byun Robinson,Sumona Saha,Susan Wolver,Raveendhara Bannuru,Elizaveta Vaysbrot,Mikala Osani,Marat Turgunbaev,Amy Miller,Timothy Mcalindon (2017). 2017 American College of Rheumatology Guideline for the Prevention and Treatment of Glucocorticoid‐Induced Osteoporosis.
Ral Sutton,L Dian,P Guy (2011). Osteoporosis in men: an under recognised and undertreated problem.
C Tennenbaum,J Clark,K Schwartzman,S Wallenstein,R Lapinski,D Meier (2002). Yield of laboratory testing to identify secondary contributors to osteoporosis in otherwise healthy women.
K Tu,J Lie,Hyun (2018). Osteoporosis: A review of treatment options.
Madhusmita Misra,Danièle Pacaud,Anna Petryk,Paulo Collett-Solberg,Michael Kappy (2008). Vitamin D Deficiency in Children and Its Management: Review of Current Knowledge and Recommendations.
P Lips,K Cashman,C Lamberg -Allardt,H Bischoff -Ferrari,B Obermayer -Pietsch,M Bianchi (2019). Current vitamin D status in European and Middle East countries and strategies to prevent vitamin D deficiency; a position statement of the European Calcifed Tissue Society.
Richard Lewis,Emma Laing (2015). Conflicting reports on vitamin D supplementation: Evidence from randomized controlled trials.
Rebecca Moon,Justin Davies,Cyrus Cooper,Nicholas Harvey (2020). Vitamin D, and Maternal and Child Health.
Sarah Taylor (2020). Vitamin D in Toddlers, Preschool Children, and Adolescents.
Donald Bailey,Alan Martin,Heather Mckay,Susan Whiting,Robert Mirwald (2000). Calcium Accretion in Girls and Boys During Puberty: A Longitudinal Analysis.
Steven Boonen,Paul Lips,Roger Bouillon,Heike Bischoff-Ferrari,Dirk Vanderschueren,Patrick Haentjens (2007). Need for Additional Calcium to Reduce the Risk of Hip Fracture with Vitamin D Supplementation: Evidence from a Comparative Metaanalysis of Randomized Controlled Trials.
M Bolland,G Avenell,G Gamble (2010). Effect of calcium supplements on risk if myocardial infarction and cardiovascular events: meta -analysis.
S Meryl,M Leboff,H Sharon,M Chou,E Murata,C Donlon (2020). Effects of supplemental vitamin D on bone health outcomes in women and men in the vitamin D and Omeg A-3 trial (VITAL).
L Burt,E Billington,M Rose,D Raymond,D Hanley,S Boyd (2019). Effect of high dose vitamin D supplementation on volumetric bone density and bone strength: A randomized controlled trial.
Terry Aspray,Thomas Chadwick,Roger Francis,Elaine Mccoll,Elaine Stamp,Ann Prentice,Alexander Von Wilamowitz-Moellendorff,Inez Schoenmakers (2019). Randomized controlled trial of vitamin D supplementation in older people to optimize bone health.
K Sanders,A Stuart,E Williamson,J Simpson,M Kotowicz,D Young (2010). Annual high -dose oral vitamin D and falls and fractures in older women: A randomized control trial.
H Smith,F Anderson,H Raphael,P Maslin,S Crozier,C Cooper (2007). Effect of annual intramuscular vitamin D on fracture risk in elderly men and women a population-based, randomized, double-blind, placebo-controlled trial.
Pang Yao,Derrick Bennett,Marion Mafham,Xu Lin,Zhengming Chen,Jane Armitage,Robert Clarke (2019). Vitamin D and Calcium for the Prevention of Fracture.
T Winzenberg,S Powell,K Shaw,G Jones (2011). Effects of vitamin D supplementation on bone density in healthy children: systematic review and meta-analysis.
Tania Winzenberg,Sandi Powell,Kelly Shaw,Graeme Jones (2010). Vitamin D supplementation for improving bone mineral density in children.
A Khadilkar,M Sayyad,N Sanwalka,D Bhandari,S Naik,V Khadilkar (2010). Vitamin D supplementation and bone mass accrual in underprivileged adolescent Indian girls.
Dimitri Ceroni,Rebecca Anderson De La Llana,Xavier Martin,Léopold Lamah,Geraldo De Coulon,Katia Turcot,Victor Dubois-Ferrière (2012). Prevalence of vitamin D insufficiency in Swiss teenagers with appendicular fractures: A prospective study of 100 cases.
Leticia Ryan,Stephen Teach,Steven Singer,Rachel Wood,Robert Freishtat,Joseph Wright,Robert Mccarter,Laura Tosi,James Chamberlain (2012). Bone Mineral Density and Vitamin D Status Among African American Children With Forearm Fractures.
L Anderson,W Heong,Y Chen,K Thorpe,K Adeli,A Howard,E Sochett,C Birken,P Parkin,J Maguire (2017). 10: Vitamin D and Fracture Risk in Early Childhood: A Case-Control Study.
Sabah Servaes,Lisa States,Joanne Wood,Samantha Schilling,Cindy Christian (2020). Rachitic change and vitamin D status in young children with fractures.
Nicklas Brustad,Bo Chawes,Jonathan Thorsen,Martin Krakauer,Jessica Lasky-Su,Scott Weiss,Jakob Stokholm,Klaus Bønnelykke,Hans Bisgaard (2022). High-dose vitamin D supplementation in pregnancy and 25(OH)D sufficiency in childhood reduce the risk of fractures and improve bone mineralization in childhood: Follow-up of a randomized clinical trial.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Vikramaditya Rai. 2026. \u201cMusculoskeletal Complications Associated with Vitamin D Deficiency and Review of Current Interventions\u201d. Global Journal of Medical Research - H: Orthopedic & Musculoskeletal System GJMR-H Volume 23 (GJMR Volume 23 Issue H1).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.