## I. INTRODUCTION
The novel Coronavirus 2019 disease (COVID-19) has become a global crisis and a challenge to public health owing to its fast rate of dissemination and increased mortality rate. Although the disease was first observed in December 2019 in the Hubei Province of China, soon it circulated hastily around the world. On March 2020, the disease has been declared as a 'pandemic emergency' by the World Health Organization. The outbreak is responsible for more than 608,328,548 confirmed cases and 6,501,469 deaths worldwide till September 2022 [1].
The incubation period of disease ranges from 1-14 days with fever, cough, shortness of breath or difficulty in breathing, or fatigue as the most common presenting symptoms. Less common features, such as headache, loss of taste or smell, sore throat, diarrhea, and nausea or vomiting, may also be present [2]. The severity of symptoms varies from person to person as it depends upon the time of exposure to the virus, the patient's age and gender as well as the coexisting diseases.
It was found that the coronavirus invades human cells with the help of receptors known as Angiotensin-converting enzyme 2 (ACE 2) and transmembrane protease serine 2 (also called transmembrane serine protease or TMPRSS2) [3]. Among these two, the ACE 2 receptor is found mainly in the cells of the lung, liver, kidney, gastrointestinal (GI) and even on the salivary glands and dorsum of the tongue of the oral cavity [4]. These cells with the receptors act as host cells for the virus through which, the virus invades these cells of the body and starts an inflammatory response in these organs.
Previously, it was assumed that COVID-19 lacks oral manifestations unlike other viral exanthema but after some years, SARS-CoV-2 was detected from the saliva of the patients suggesting a possibility that oral manifestations could be clinical characteristics of COVID-19. Also, the presence of the ACE 2 receptor in some specific organs of the oral cavity such as the tongue and salivary glands confirms the possibility of the involvement of the oral cavity in coronavirus infection. The frequency of oral manifestations among COVID-19 patients is unknown but some previous studies have tried to provide the incidence and prevalence of these manifestations. A huge study conducted by Nuno-Gonzalez on 666 patients suggests that oral cavity findings are present in $25.65\%$ of cases [5]. The commonly occurring oral manifestations found in a case series conducted by Sinadinos and Shelswell were blisters, ulcerations, and desquamative gingivitis [6]. In the oral cavity, the most commonly involved sites in COVID-19 disease are the palate and tongue followed by the gums and the lips [7]. In the tongue, the ulcerations are quite common specifically on the dorsum surface or sides of the tongue. Rarely, only in $15\%$ of patients, the ulcerations develop on the ventral surface [8]. Additionally, multiple pinpoint yellowish ulcers and white plaque can also be present on the tongue [3]. The occurrence of white plaque on the dorsal surface of the tongue is due to the occurrence of fungal infections which is also one of the common oral manifestations of SARS-CoV-2, probably caused by lower immunity. Dima et al reported a case of a neonate with COVID-19 having oral cavity candidiasis [9]. These oral manifestations are accompanied with pain in $75\%$ of cases [7]. In another study, $25\%$ of patients reported impaired taste, $15\%$ had burning sensations, and $20\%$ had difficulty in swallowing. Ageusia was observed in $24\%$ of patients, hypogeusia in $35\%$, and dysgeusia in $38\%$ of the COVID-19 patients. Taste disorders are more common in women than men [10].
A proper understanding of the dentist regarding oral manifestations of COVID-19 is very important as it helps in the early diagnosis of the disease and hence prevents transmission. The present systematic review aims to summarize the findings of the available past literature regarding oral manifestations of COVID-19 to highlight the role of the dentist in intervening the severity of this deadly pandemic disease.
## II. METHODS
### a) Outcome
The primary outcome of this study was the systematic evaluation and characterization of currently reported cases and studies of oral manifestations associated with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. This review followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [11].
### b) Eligibility criteria
The clinical evidence was searched in the form of original peer-reviewed journal articles which include observational and cross-sectional studies investigating the prevalence of oral disorders in patients with COVID-19. Apart from these, case reports and case series were also included in the systematic review. The range of data publication was limited from January 1, 2020, to June 30, 2022. Conference papers, book reviews, book chapters, letters to the editor and replies, newspaper and newsletter articles, expert opinions, and theses or dissertations were not used. Articles that are not published in English were also excluded.
### c) Data sources and search strategy
We conducted an extensive literature search in several electronic bibliographic databases (PubMed, Scopus, Science direct, Litcovid) and retrieved all articles published from January 1, 2020, to June 30, 2022. Observational cross-sectional, case-control and cohort studies, case reports/ series reporting MIS-C, and letters to the editor were screened. The surveillance studies were also included.
#### PUBMED [search until 30.06.2022]
(((((((((((((((((((((((oral manifestations) OR (oral involvement)) OR (oral lesions)) AND (gingival lesions)) OR (gingival bleeding)) OR (gingival ulcer)) AND (buccal mucosa involvement)) OR (buccal ulcer)) AND (lip ulcer)) OR (lip mucosa involvement)) AND (tongue lesions)) OR (tongue ulcer)) OR (tongue involvement)) OR (ageusia)) OR (hypogeusia)) OR (dysgeusia)) OR (loss of taste)) OR (altered taste)) OR (gustatory impairment)) AND (burning sensation)) AND (aphthous stomatitis)) AND (ulcers on palate)) AND (COVID-19)) OR (coronavirus)) OR (corona pandemic)) OR ((SARS-CoV-2)
#### LITCOVID [search until 30.06.2022]
"Oral manifestations in COVID-19" OR "tongue lesions in coronavirus" OR "gingival involvement in coronavirus pandemic" OR "buccal mucosa involvement in COVID-19"
#### SCOPUS [search until 30.06.2022]
(tongue lesions OR mucosal lesions OR oral lesions AND COVID-19 OR coronavirus)
SCIENCE DIRECT [search until 30.06.2022]
Oral manifestations in COVID-19 OR oral involvement in coronavirus
### d) Data collection
The inclusion of studies was done in two phases. In phase I, the titles of all the studies were screened first, followed by the screening of abstracts, based on the inclusion and exclusion criteria. Articles eligible based on the title and abstract would then be read in full and judged for their eligibility.Duplicates were eliminated and irrelevant articles were excluded from the systematic review. The full-text articles of all potential studies were obtained and evaluated for inclusion. In phase II, the references of all the included studies, case reports, and case series were again screened to search for any new potentially eligible studies.
All studies that reported orofacial manifestations in patients with laboratory-confirmed COVID-19 were included. Studies with suspected COVID-19 cases were excluded. To provide a systematic overview of these studies, we evaluated the included studies in terms of their demographic details such as author, year, country, study type, sample size, gender, age, study duration, medical history, admission in the ICU, and severity of the disease. Apart from this, the details related to the oral manifestations such as the site affected, onset of the orofacial manifestations, general symptoms, any special investigations performed, treatment of oral lesions, and outcome of the disease were also recorded. In the end, a total of 75 articles were included in this systematic review. The article inclusion flow diagram is shown in figure 1
### e) Outcome measures
The primary outcome was estimating the prevalence of oral manifestations among COVID-19 patients. The secondary outcome was the most common type of oral manifestation and site affected in the oral cavity.
### f) Statistical analysis
Qualitative data were resynthesized by grouping and comparing information reported in newly included studies. Oral and mucosal conditions were summarized in schematic representations.
## III. RESULTS
A lot of studies along with the literature reviews have evaluated the oral mucosa involvement in COVID-19. We got 268 articles on the preliminary search, out of them, 75 articles were included in the final analysis [5,6,9,10,12-82].
The demographic characteristics of the included studies $(n = 75)$ are shown in table 1. We retrieved the data from 75 studies [5,6,9,10,12-82] including 11,321 patients with individual sample sizes of studies ranging from 14 [47] to 1172 [35]. All the articles $(n = 75)$ were published in year 2020 [6,9,12-27,56-69], 2021 [5,28-43,70-79], and 2022[44-55,80-82] but most of the articles $(n = 33)$ were from 2020 followed by the year 2021 $(n = 31)$ and 2022 $(n = 15)$.
The 75 articles included data from 27 countries around the globe. Most of the studies were conducted in Italy $(n = 13)$ [16,19,20,22,23,28,29,36,37,58,66,72,81], followed by Brazil $(n = 7)$ [10,47,49,52,56,75,77], India $(n = 7)$ [17,33,44,45,50,55,80], Egypt $(n = 5)$ [34,38,41,68,71], Turkey $(n = 5)$ [26,39,42,48,64], Iran $(n = 4)$ [25,54,62,79], Spain $(n = 4)$ [5,53,59,60], USA $(n = 4)$ [27,57,65,67], Saudi Arabia $(n = 3)$ [40,46,51], New York $(n = 2)$ [30,31], China $(n = 2)$ [15,35], Iraq $(n = 2)$ [12,43], Israel $(n = 2)$ [13,74], Europe $(n = 2)$ [18,70], Copenhagen $(n = 1)$ [14], California $(n = 1)$ [21], France $(n = 1)$ [24], Qatar $(n = 1)$ [32], Romania $(n = 1)$ [9], United Kingdom $(n = 1)$ [6], Colombia $(n = 1)$ [61], Norway $(n = 1)$ [63], Indonesia $(n = 1)$ [69], Afghanistan $(n = 1)$ [73], Czech Republic $(n = 1)$ [76], Ukraine $(n = 1)$ [78], and Poland $(n = 1)$ [82].
Among 75 studies, most of them were cross-sectional studies (n=36) [5,12-26,29,30,33,34,37-42,44-46,48-51,53-55] followed by case report (n=20) [10,61-75,78-81], case series (n=10) [6,9,56-60,76,77,82], retrospective studies (n=6) [27,28,35,36,47,52], prospective study (n=2) [31,43], and case-control study (n=1) [32].
A total of 11,321 patients were evaluated in the systematic review, of which 6581 (58.1%) were females. Three studies did not report the gender of the patients in their studies [5,22,35]. The age group of most of the studies (n=39) was in the range of more than 50 years followed by less than 30 years (n=12). Three studies did not report the age group of the patients [5,34,35] and one study was conducted among newborns [9]. The detailed age distribution is shown in figure 2.
Only 33 studies have reported the medical history of the patient. Among those studies, most patients were having hypertension $(n = 25)$, diabetes $(n = 19)$, respiratory disease and asthma $(n = 7)$, cardiovascular disease $(n = 6)$, allergy $(n = 4)$, and others. Regarding hospitalization, 21 studies reported that
COVID-19 patients were admitted to the hospital, 11 studies reported hospitalization in ICU, and 5 of the studies also showed that the patients were ventilated (Table 1).
The oral manifestations were divided into 12 categories: taste alteration, tongue manifestation, xerostomia, red and white lesions, vesiculobullous lesions, periodontal changes, burning sensation, bleeding disorders, lip lesions, TMJ disorders, salivary gland disorders, and fungal infections. Most of the patients were having multiple signs and symptoms. Hence, we have calculated the oral manifestation individually. Taste alteration $(n = 43;57.3\%)$ was found in most people followed by vesiculobullous lesions $(n = 41;54.6\%)$ and tongue manifestations $(n = 26;34.6\%)$. The detailed results of the type of oral manifestation is shown in figure 3.The most common sites of involvement in descending order were the tongue $(n = 62)$ followed by oral mucosa $(n = 27)$ and lips $(n = 19)$ (Figure 4). Descriptive characteristics of the oral lesions are shown in Table 2.
Taste Disorders and tongue manifestation: The prevalence of taste disorders and tongue manifestation was assessed with data from 43 (57.3%) and 26 (34.6%) studies respectively. Loss of taste or ageusia and was reported by 21 studies [12,14,20,23,35,40-42,44,49, 53,55,56,58,63,65,67,70,72,74]. Taste alteration or dysfunction was reported by [5,13,16-19,21,22,29, 36,38,46,51,54,66,78], Amblygeustia [15], dysgeusia [24,28,37,50,54,59,60]
Three studies [29,36,51] reported geographic tongue and one study [30] reported strawberry tongue among the COVID-19 patients. Other tongue manifestations are shown in table 2.
Vesiculobullous lesions: Forty one (54.6%) included studies and case reports or case series were having the vesiculobullous lesions [5,6,10,26,27,29,31,33,34,37-44,46-51,53,55,56,57,59-62,64,69,70,75-77,79,81,82].
Xerostomia: Xerostomia was noted in 18cross-sectional studies [13,15,28,34,37,38,40,42,44,45,46,48,50-55] and 2 case reports[60,78] constituting $26.6\%$ of the included studies.
Red and white lesions: Eighteen studies(24%) reported red and white lesions in COVID-19 positive patients [9,24-26,29,33,36,37,39,43,49,51,53,57,61,68,80,82].
Periodontal involvement: Twelve studies (16%) were found having the involvement of gingiva and periodontium among the patients of COVID-19 of the present systematic review [6,26,29,32,38,39,42,44, 51,55,59,78].
Burning sensation: Eleven studies (14.6%) reported the complaint of burning sensation [5,33,38,42-44,53,55, 57,60,78].
Bleeding disorders: A total of six studies (8%) reported the incidence of bleeding disorders in COVID-19-positive patients [29,51-53,64,82].
Other findings: Salivary gland disorders (4%) [37,38,52], fungal infection (2.6%) [43,82], and dental pain (2.6%) [34,42] was found in three, two, and two studies respectively.
The latency time between the appearance of systemic symptoms and oral lesions was between 2 weeks before to 10 days after the onset of systemic symptoms. In most of the studies $(n = 14)$, general symptoms followed the oral symptoms (Table 2).
General treatment, as well as the treatment for the oral lesions among COVID-19 patients, is given in supplementary table 1. Oral lesions healed between 7 and 21 days after appearance. Different types of therapies including chlorhexidine mouthwash, nystatin, oral fluconazole, topical or systemic corticosteroids, systemic antibiotics, systemic acyclovir, artificial saliva, and photobiomodulation therapy (PBMT) were prescribed for oral lesions depending on the severity and etiology.
## IV. DISCUSSION
COVID-19 has become a public health problem around the world. Initially, it was thought that the lack of involvement of the oral mucosa is a differentiating feature of COVID-19 as compared to other viral infections but in April 2020, a case report published by Chaux-Bodard et al have shown the association of COVID-19 with the oral mucosa in a 45-year-old female having painful inflammation of the papilla of the tongue, which ultimately healed as an asymptomatic ulcer in 10 days without a scar along with a skin lesion in the toe and was tested positive on Day 8 [83]. Since then, many observational studies and case reports were published in the literature depicting the involvement of oral mucosa among COVID-19 patients. The present systematic review was conducted with the same intent to elaborate the association between COVID-19 and the oral cavity with the help of previously published studies.
SARS-CoV-2 invades human cells of the lower respiratory system with the help of receptors known as ACE 2 and transmembrane protease serine 2 [3]. Among these two, the ACE 2 receptor is found mainly in the cells of the lung, liver, kidney, gastrointestinal (GI) and even on the cells of nasal epithelium and oral mucosa [4]. These cells act as host cells for the virus through which, the virus invades these cells of the body and starts an inflammatory response in these organs which, in turn, causes the smell and taste dysfunctions early in the course of the disease [15]. Hence, the mechanism of development of oral lesions can be directly through the effects of the replicating virus in these cells (lesions will be SARS-CoV-2-specific) and indirectly as a sequel of possible drug reactions that may develop during the latency period, viral exanthem, through physical and psychological stress of the COVID-19 or its treatment, or co-infection with other bacterial infections enhancing the severity of COVID-19[59]. According to Amorim dos Santos et al [4], the deterioration of the general health of COVID-19 patients along with the longer period of hospitalization and several treatment procedures also predispose the occurrence of oral lesions. Chaux-Bodard et al. hypothesized that oral lesions may arise as a sequel of various inflammatory reactions that induce vascular inflammation [83]. Previously published reports of the Italy and United Kingdom stated the temporary association of pediatric inflammatory multisystem syndrome with SARS-COV-2 cases [84]. Various diseases like Kawasaki disease and erythema multiforme can itself predispose to oral manifestations. Hence, we have excluded such conditions from our systematic review.
Talking about the oral lesions, the most common sites of involvement in descending order were the tongue (n=62), oral mucosa (n=27), lips (n=19), entire oral cavity (palate (n=12), gingiva and periodontium (n=12), and salivary gland (n=3). Sousa et al found the palate and tongue followed by the gums and the lips as the most commonly involved sites in COVID-19 patients [7]. Description of the oral manifestations among COVID-19 patients are as follows:
### a) Taste disorders
According to the various published studies, smell and taste changes are the early indicators of the COVID-19 pandemic which are effective in early diagnosis and decision making. Though these symptoms are not life-threatening, they may hamper the quality of life of the patient. Prof C. Hopkins, President of the British Rhinological Society has stated that loss of smell/taste can be the only symptom of COVID-19 [85]. Several public health surveillance organizations such as the European Centre for Disease Prevention and Control, Centre for Disease Control and Prevention (CDC), WHO [86], and Public Health England included the sudden onset of anosmia, ageusia, or dysgeusia in the list of main clinical criteria for the case definition of COVID-19 [87]. In our systematic review also, general symptoms followed the oral symptoms, especially loss of taste. The possible explanation for the taste disturbances in COVID-19 patients is the higher expression of ACE 2 receptor in the tongue as compared to the buccal and gingival tissues resulting in the damage to mucosal epithelial cells of the oral cavity [88].
In the present systematic review, the incidence of taste disorders and tongue manifestation was assessed with data from 43 (57.3%) and 26 (34.6%) studies respectively. Al-Zaidi et al in their cross-sectional study found the overall prevalence of taste dysfunction in $83.08\%$ of COVID-19 patients. The taste recovers at one week for $50\%$ of the participants followed by less than a week $(25\%)$, within 2 weeks $(18.75\%)$, and within 3 weeks $(6.25\%)$ [12]. Amorim dos Santos et al in their living systematic review (LSR) found taste disorder as the most prevalent oral symptom in this population with a prevalence of $45\%$ [4] but in their second LSR, the prevalence decreased to $38\%$. They stated that the prevalence of taste disorders among COVID-19 patients varies from $14\%$ in Africa to $49\%$ in Europe [89]. Yan CH et al found taste loss in $71\%$ of COVID-19-positive subjects and the association was strongly associated with COVID-19 positivity (OR 10.2; $95\%$ CI, 4.74-22.1) [21]. A total of $52\%$ reported changes in taste sensation in Biadsee A et al study with 52 patients reporting a change in spicy taste perception, 54 in salty taste, 53 in sour taste, and 61 patients in sweet taste [13]. Bodnia NC et al reported a total loss of taste in $70\%$ of patients which resolved within 1-3 weeks for $78\%$ and 3-6 weeks for $22\%$ [14]. A meta-analysis conducted by Tong JY et al have shown that gustatory changes are noted in $43.93\%$ of these individuals [90]. The prevalence of taste alterations was estimated to be around $54.73\%$ [95% CI: 46.28-63.04%] in another meta-analysis conducted by Nijakowski K et al [91].
Three studies conducted by Favia G et al [29], Bardellini E et al [36], and Binmadi NO et al [51] reported geographic tongue in our systematic review. Bardellini E et al conducted a pediatric retrospective cohort study and reported oral pseudomembranous candidiasis $(n = 2)$, coated tongue $(n = 2)$, taste alteration $(n = 3)$, and geographic tongue $(n = 1)$ as the most common oral lesions of which, geographic tongue appeared concurrently with the high fever according to the patient's mother [36]. The etiopathogenesis of the geographic tongue is still unclear but some authors reported the association of several non-genetic multifactorial factors, including viral infections [92]. A study conducted by Halepas S et al [30] reported strawberry tongue among COVID-19 patients. Other tongue manifestations are plaque-like changes in the tongue [13,52,61], macroglossia [24,32], ulcerations on tongue [10,24,26,56,73,76,80,81], fissured tongue [29,44,73], lingual papillitis, white tongue and glossitis with patchy depapillation [5], burning sensation in tongue [42,68,78], white coat, numbness, and black discoloration of tongue [43], depapillation of tongue [45,60], hairy tongue [46], Greasy tongue coat [71], and smooth tongue and mycosis of the tongue [82].
### b) Vesiculobullous lesions
Forty one included studies and case reports/case series (54.6%) have mentioned the incidence of vesiculobullous lesions [5,6,10,26,27, 29,31,33,34,37-44,46-51,53,55,56,57,59-62,64,69,70,75- 77,79,81,82]. Favia G et al widely describes the histological aspect of oral SARS-CoV-2-related lesions and found ulcers $(52.8\%)$ as the most detected oral manifestation [29]. Presas CMC et al reported three cases with ulcers, of which one was infected by the SARS-CoV-2 virus and two were suspected patients infected by the SARS-CoV-2 virus. The lesions resemble herpes simplex infection but were not confirmed by biopsies [59]. Painful oral ulcers were the most common orofacial manifestations in patients with COVID-19 in a review conducted by Halboub E et al [93]. Tapia ROC et al [57] and Dalipi ZS et al [70] found bullous and lesions on the palate and oral mucosa respectively. Riad A et al found multiple ulcers (1 and 7 ulcers per patient) with their size ranging between 1 and $5\mathrm{mm}$, of which the majority $(92.3\%)$ were not bleeding, and all of them $(100\%)$ were manifested on the dorsum or side of the tongue. They found a statistically significant association between the number of ulcers and gender, onset, duration, Ct value, and pain score [8]. A systematic review conducted by Orilisi G stated that oral ulcers, cheilitis, and tongue lesions were more common in patients before hospitalization, while perioral pressure ulcers, macroglossia, blisters, and oral candidiasis were more recurrent in patients during hospitalization [94].
Regarding the mechanism of the formation of the ulcer, it was proposed that an increased level of tumor necrosis factor (TNF)- $\alpha$ in COVID-19 patients can lead to chemotaxis of neutrophils to oral mucosa and the development of aphthous-like lesions. Stress and immunosuppression secondary to COVID-19 infection could be other possible reasons for the appearance of such lesions in COVID-19 patients [10].
Red and white lesions: Eighteen studies (24%) reported red and white lesions in COVID-19 positive patients [9,24-26,29,33,36,37,39,43,49,51,53,57,61,68,80,82].
The various types of red and white lesions reported in the included studies are cheilitis and oral lichenoid reaction [24], white plaques on the intraoral mucous layer [25,37,43,68,80], rash and erythema [26], candidiasis [9,29,36,49,51,53], reddish-white spots on the palate [33,57], erythema and lichen planus [39], angular cheilitis [53,82], and reddish plaques on the lower lip [61].
Xerostomia Xerostomia was noted in 18 cross-sectional studies [13,15,28,34,37,38,40,42,44,45,46,48,50-55] and 2 case reports [60,78] with $26.6\%$ of studies reporting the complaint of dry mouth. In a study conducted by Biadsee et al, $56\%$ of patients reported xerostomia which was assessed by the question "Do you feel the need to drink more (dry mouth)?" [13]. In the updated version of the LSR performed by dos Santos et al [89], xerostomia was the most prevalent oral symptom identified in patients with COVID-19, whereas, taste disturbances were the predominant feature in the original LSR [4]. In a meta-analysis conducted Nijakowski K et al, xerostomia was prevalent
- among $37.58\%$ [95% CI: 26.35-49.53%] of the COVID-19 patients [91].
- Periodontal involvement: Twelve studies (16%) [6,26,29,32,38,39,42,44,51,55,59,78] have shown the prevalence of gingivitis and periodontitis. The gingival manifestations found among the COVID-19 patients of our systematic review were the gingivitis [29], desquamative gingivitis [6,59], ulceronecrotic gingivitis [29], and gingival bleeding [6,38,42,44,55,78]. Periodontitis [32] and necrotizing periodontal disease [51] were reported in two studies.
- Other findings: Red and/or swollen lips was observed by Halepas S et al in $48.9\%$ of patients [30]. Pale lips [33], reddish plaques on the lower lip [61], nodule in the lower lip [10], and reddish macules [42] were the findings related to lip involvement in COVID-19 patients. Reddish-white spots on the palate [33], ulcerations on the palate [6,39,56,59,60,81,82], white coat of the palate [43,68], bulla on the left and right palatal mucosa [57], an erythematous surface on the hard palate [64], and angioma type lesion on the right side of the palate [82] were the palatal findings among COVID-19 patients. Eleven studies $(14.6\%)$ included in the systematic review found the complaint of burning sensation also [5,33,38,42-44,53,55,57,60,78]. Biadsee A et al found a statistically significant strong association between burning mouth and taste change $(p = 0.002$, $p = 0.009$, respectively) [13].
- Although we have tried to summarize the findings of the studies reporting oral manifestations among COVID-19 patients, one of the biggest limitations of this systematic review was the lack of temporal dimension. We were not able to say that these oral manifestations are directly connected to COVID-19, or due to indirect manifestations of other factors such as stress, immunosuppression, and/or medications. Another shortcoming was the lack of definitive diagnosis as most of the included cases have not undergone biopsy for confirmation of the diagnosis.
## V. CONCLUSION
Our systematic review shows a higher prevalence of oral manifestation, specifically taste alteration (57.3%) followed by vesiculobullous lesions (54.6%), and tongue manifestations (34.6%). COVID-19 patients show various oral manifestations that may help clinicians with early identification of the disease. Recognition of signs and symptoms of COVID-19 is critical for early diagnosis and better prognosis. Dental practitioners can play an important role not only in the prevention of COVID-19 transmission but also in breaking the chain of COVID-19 disease. Raising awareness of these symptoms is important to initiate early diagnosis and treatment of this deadly COVID-19 disease.
- Declarations
- Ethics approval and consent to participate-Not required.
- Availability of data and material-Can be made available whenever required.
- Competing interests- None
- Funding- None
- Authors' contributions- All authors contributed equally for the article.
- Acknowledgements- None
- study of 713 patients. Dent Res J (Isfahan). 2021; 18: 67.
34. Abubakr N, Salem ZA, Kamel AHM. Oral manifestations in mild-to-moderate cases of COVID-19 viral infection in the adult population. Dent Med Probl. 2021; 58(1):7-15. doi: 10.17219/dmp/130814.
35. Song J, Deng YK, Wang H, Wang ZC, Liao B, Ma J, et al. Self-reported Taste and Smell Disorders in Patients with COVID-19: Distinct Features in China. Current Medical Science. 2021; 41(1): 14-23. DOI https://doi.org/10.1007/s11596-021-2312-7
36. Bardellini E, Bondioni MP, Amadori F, Veneri f, Lougaris V, Meini A, et al. Non-specific oral and cutaneous manifestations of Coronavirus Disease 2019 in children. Med Oral Patol Oral Cir Bucal. 2021 Sep; 26(5): e549-e553.
37. Gherlone EF, Polizzi E, Tetè G, De Lorenzo R, Magnaghi C, Querini RP, et al. Frequent and Persistent Salivary Gland Ectasia and Oral Disease After COVID-19. Journal of Dental Research.2021; 100(5): 464-47.
38. El Kady DM, Gomaa EA, Abdella WS, Hussien RA, Abd EIAziz RH, Khater AGA. Oral manifestations of COVID-19 patients: An online survey of the Egyptian population. Clin Exp Dent Res.2021; 7: 852-860.
39. Fidan V, Koyuncu H, Akin O. Oral Lesions in COVID-19 Positive Patients. Am. J. Otolaryngol. 2021; 42: 102905.
40. Natto Z.S, Afeef M, Khalil D, Kutubaldin D, Dehaithem M, Alzahrani A, et al. Characteristics of Oral Manifestations in Symptomatic Non-Hospitalized COVID-19 Patients: A Cross-Sectional Study on a Sample of the Saudi Population. Int. J. Gen. Med. 2021; 14: 9547-9553.
41. Elamrousy W.A.H, Nassar M, Issa D.R. Prevalence of Oral Lesions in COVID-19 Egyptian Patients. J. Int. Soc. Prev. Community. Dent. 2021; 11: 712-720.
42. Bulut D.G, Turker N, Serin S, Ustaoglu G. The Effect of Severe Acute Respiratory Syndrome Coronavirus 2 on the Health of Oral Tissue: A Survey-Based Study. J. Oral Health Oral Epidemiol. 2021; 10: 43-49.
43. Naser Al, Al-Sarraj MN & Deleme ZH. Oral and Maxillofacial Lesions in COVID 19 Infection from Mosul Hospital in Iraq: Epidemiological Study and Approach to Classification and Treatment. J Oral Res 2021; 10(6): 1-14.
44. Muthyam AK, Padma RM, Suhas K, Adepu S, Kommuri S, Dantala S. Oral manifestations in COVID-19 patients. An observational study. Journal of Family Medicine and Primary Care: March 2022; 11(3): 1000-1005. doi: 10.4103/jfmpc.jfmpc_1264 21
45. Ganesan A, Kumar S, Kaur A. Oral Manifestations of COVID-19 Infection: An Analytical Cross-Sectional Study. J. Maxillofac. Oral Surg.
2022\. https://doi.org/10.1007/s12663-021-01679-x
46. El Tantawi M, Sabbagh HJ, AlKhateeb NA, Quritum M, Abourdan J, Qureshi N, et al. Oral manifestations in young adults infected with COVID-19 and impact of smoking: a multi-country cross-sectional study. Peer J. 2022; 10:e13555 DOI 10.7717/peerj.13555
47. Soares CD, Souza LL, de Carvalho MGF, Pontes HAR, DDS, Mosqueda-Taylor A, Hernandez-Guerrero JC, et al. Oral Manifestations of Coronavirus Disease 2019 (COVID-19) A Comprehensive Clinicopathologic and Immunohistochemical Study. Am J Surg Pathol 2022; 46: 528-536.
48. Tuter G, Yerebakan M, Celik B, Kara G. Oral manifestations in SARSCoV-2 infection. Med Oral Patol Oral Cir Bucal. 2022; 1;27 (4):e330-9.
49. Schwabhtp G, Palmierib M, Zerbinatia RM, Sarmento DJS, Reisb T, Ortega KL. Lack of direct association between oral mucosal lesions and SARS-CoV-2 in a cohort of patients hospitalised with COVID-19. Journal of Oral Microbiology. 2022; 14(1): 1-7. https://doi.org/10.1080/20002297.2022.2047491
50. Chawla J, Navaneeth Y, Bakshi SS, Kalidoss VK, Yadav S, Polineni S, et al. Oral manifestations associated with COVID-19 disease: An observational cross sectional study. Journal of Oral Biology and Craniofacial Research. 2022; 12: 279-283.
51. Binmadi NO, Aljohani SR, Alsharif MT, Almazrooa SA, Sindi AM. Oral Manifestations of COVID-19: A Cross-Sectional Study of Their Prevalence and Association with Disease Severity. J. Clin. Med. 2022; 11(15): 4461; https://doi.org/10.3390/jcm11154461
52. de Paula Eduardo F, Bezinelli LM, Gobbi MF, Bergamin LG, de Carvalho DLC, Corrêa L. Oral lesions and saliva alterations of COVID-19 patients in an intensive care unit: A retrospective study. Spec Care Dentist.2022; 1-9.
53. Villarroel-Dorrego M, Chacon L, Rosas R, Barrios V, Pernia Y, Vélez H. Hallazgos bucales en pacientes COVID-19. Actas Dermosifiliogr. 2022;113:183-186
54. Manifar S, Koopaie M, Karimi Farani A, Davoudi M, Kolahdouz S. Assessment of Oral Manifestations and Oral Health in Hospitalized Patients with COVID-19: Machine Learning and Statistical Analysis. Ann Mil Health Sci Res.2022; 20(1):e121764. doi: 10.5812/amh-121764.
55. Bhuyan R, Bhuyan SK, Mohanty JN, Panda NR, Bhuyan V, Ojha P, Priyadarshini P, Sahoo G. A Preliminary Survey on the Oral Manifestation of COVID-19 in the First and Second Waves in Bhubaneswar, City of Odisha, India. Natl J Community Med 2022; 13(5): 294-297. DOI: 10.55489/njcm.130520221617
56. Brandao TB, Gueiros LA, Melo TS, Prado-Ribeiro AC, Nesrallah ACF, Prado GVB, et al. Oral lesions in
- patients with SARS-CoV-2 infection: could the oral cavity be a target organ?.Oral Surg Oral Med Oral Pathol Oral Radiol. 2020;000:e1-e7
57. Tapia ROC, Labrador AJP, Guimaraes DM, Valdez LHM. Oral mucosal lesions in patients with SARS-CoV-2 infection. Report of four cases. Are they a true sign of COVID-19 disease? Spec Care Dentist. 2020; 1-6.
58. Vaira LA, Deiana G, Fois AG, Pirina P, Madeddu G, De Vito A, et al. Objective evaluation of anosmia and ageusia in COVID-19 patients: Single-center experience on 72 cases. Head & Neck. 2020; 42: 1252-1258.
59. Presas CMC, Amaro Sánchez J, López-Sánchez AF, Jané-Salas E, Somacarrera Pérez ML. Oral vesiculobullous lesions associated with SARS-CoV-2 infection. Oral Dis.
2020\. https://doi.org/10.1111/odi.13382
60. Rodríguez MD, Romera JR, Villarroel M. Oral manifestations associated with COVID-19. Oral Dis. 2020 Aug 17: 10.1111/odi.13555.
61. Corchuelo J, Ulloac FC. Oral manifestations in a patient with a history of asymptomatic COVID-19: Case report. International Journal of Infectious Diseases. 2020; 100 (2020):XXX-XXXs
62. Zarch RE, Hosseinzadeh P. COVID-19 from the perspective of dentists: A case report and brief review of more than 170 cases. Dermatologic Therapy. 2021; 34:e14717.
63. Hjelmeseth J. Loss of smell or taste as the only symptom of COVID-19. Tidsskr Nor Laegeforen. 2020 Apr 3; 140(7). doi: 10.4045/tidsskr.20.0287.
64. Kahraman FC, Caskurlu H. Mucosal involvement in a COVID-19-positive patient: a case report. Dermatol Ther. 2020 Jul;33(4):e13797. doi: 10.1111/dth.13797.
65. Smith AC, Hodges J, Pratt M, Porter IM. COVID-19 Patient With Chief Complaint of Anosmia and Ageusia; a Unique Perspective on Atypical Symptomatology and Management in the Military. Military Medicine. 2020; 185: 11/12: 2176
66. Maniaci A, Iannella G, Vicini C, Pavone P, Nunnari G, Falsaperla R, et al. A Case of COVID-19 with Late-Onset Rash and Transient Loss of Taste and Smell in a 15-Year-Old Boy. Am J Case Rep. 2020; 21: e925813
67. Melley LE, Bress E, Polan E. Hypogeusia as the initial presenting symptom of COVID-19. BMJ Case Rep 2020; 13:e236080. doi: 10.1136/bcr-2020-236080
68. Riad A, Gad A, Hockova B, Klugar M. Oral Candidiasis in Non-Severe COVID-19 Patients: Call for Antibiotic Stewardship. Oral Surg. 2020 Sep 21;10.1111/ors.12561. doi: 10.1111/ors.12561.
69. Putraa BE, Adiartoa S, Dewayantib SR, Juzara DA. Viral exanthem with "Spins and needles sensation" on extremities of a COVID-19 patient: A self
- reported case from an Indonesian medical frontliner. International Journal of Infectious Diseases. 2020; 96: 355-358.
70. Dalipi ZS, Dragidella F, Dragidella DK. Oral Manifestations of Exudative Erythema Multiforme in a Patient with COVID-19. Case Reports in dentistry.
2021\. Article ID 1148945. https://doi.org/10.1155/2021/1148945
71. Eita AAB. Parosmia, Dysgeusia, and Tongue Features Changes in a Patient with Post-Acute COVID-19 Syndrome. Case Rep Dent. 2021; 2021: 3788727.
72. Cirillo N, Colella G. Self-reported smell and taste alteration as the sole clinical manifestation of SARS-CoV-2 infection. Oral Surg Oral Med Oral Pathol Oral Radiol. 2021 Apr; 131(4):e95-e99.
73. Nejabi MB, Noor NAS, Raufi N, Essar MY, Ehsan E, Shah J. et al. Tongue ulcer in a patient with COVID-19: a case presentation. BMC Oral Health. 2021; 21: 273.
74. Klein H, Karni N, Israel S, Gross M, Muszkat M, Niv MY. Reversible Taste Loss in a COVID-19 Patient With Preexisting Chronic Smell Impairment. Journal of Investigative Medicine High Impact Case Reports. 2021; 9: 1-3.
75. Ramires M.C.C.H, Mattia M.B, Tateno R.Y, Palma L.F, Campos L. A Combination of Phototherapy Modalities for Extensive Lip Lesions in a Patient with SARS-CoV-2 Infection. Photodiagnosis Photodyn. Ther. 2021; 33: 102196.
76. Hockova B, Riad A, Valky J, Šulajová Z, Stebel A, Slavik R, et al. Oral Complications of ICU Patients with COVID-19: Case-Series and Review of Two Hundred Ten Cases. J Clin Med. 2021 Feb; 10(4): 581.
77. Teixeira I.S, Leal F.S, Tateno R.Y, Palma L.F, Campos L. Photobiomodulation Therapy and Antimicrobial Photodynamic Therapy for Orofacial Lesions in Patients with COVID-19: A Case Series. Photodiagnosis Photodyn. Ther. 2021; 34: 102281.
78. Emelyanova N, Isayeva G, Komir I, Shalimova A, Buriakovska O, Vovchenko M. Changes in the Oral Cavity of a Patient after Suffering from Coronavirus Infection COVID-19: A Clinical Case. Acta Med. Mediterr. 2021; 37: 827-831.
79. Fathi Y, Hoseini E.G, Mottaghi R. Erythema Multiform-like Lesions in a Patient Infected with SARS-CoV-2: A Case Report. Future Virol. 2021; 16: 157-160.
80. Shenoy P, Archana P, Chatra L, Veena KM, Prabhu R. Tongue Ulcer as an Oral Manifestation of COVID-19: A Case Report. International Journal of Research and Reports in Dentistry. 2022; 5(1): 8-11. Article no.IJRRD.81119
81. Palaia G, Pernice E, Pergolini D, Impellizzeri A, Migliau G, Gambarini G, et al. Erythema Multiforme as Early Manifestation of COVID-19: A Case Report.

- Pathogens 2022; 11: 654. https://doi.org/10.3390/pathogens11060654
82. Rafałowicz B, Wagner L, Rafałowicz J. Long COVID Oral Cavity Symptoms Based on Selected Clinical Cases. Eur J Dent. 2022 May; 16(2): 458-463.
83. Chaux-Bodard AG, Deneuve S, Desoutter A. Oral manifestation of covid-19 as an inaugural symptom? J Oral Med Oral Surg. 2020; 26(2): 18. https://doi.org/10.1051/mcbc/2020011
84. Viner RM, Whittaker E. Kawasaki-like disease: emerging complication during the COVID-19 pandemic. The Lancet. 2020; 395(10239): 1741-1743. https://doi.org/10.1016/s0140-6736(20)31129-6
85. Hopkins C, Kumar N. Loss of sense of smell as marker of COVID-19 infection. www.entuk.org/sites/default/files/files/Loss%20of%2 0sense%20of%20smell%20as%20marker%20of%20 COVID.pdf. Accessed on $24^{\text{th}}$ July 2022.
86. Cirillo N. Taste alteration in COVID-19: a rapid review with data synthesis reveals significant geographical differences. medRxiv.
2020\. https://doi.org/10.1101/2020.09.11.20192831. Accessed on 28th July 2022.
87. Public Health England. COVID-19: investigation and initial clinical management of possible cases. Updated October 2, 2020. Available at: https://www.gov.uk/government/publications/wuhan-novel-coronavirus-initial-investigation-of-possiblecases/investigation-and-initial-clinical-management-of-possiblecases-of-wuhan-novel-coronavirus-wn-cov-infection#criteria. Accessed on $2^{\text{nd}}$ August 2022.
88. Jafek BW, Murrow B, Michaels R et al. Biopsies of human olfactory epithelium. Chem Senses 2002;27:623-631
89. Amorim Dos Santos J, Normando AGC, Carvalho da Silva RL, et al. Oral Manifestations in Patients with COVID-19: A 6-Month Update. J Dent Res. 2021; 1-9. https://doi.org/10.1177/00220345211029637
90. Tong JY, Wong A, Zhu D, Fastenberg JH, Tham T. The prevalence of olfactory and gustatory dysfunction in COVID-19 patients: a systematic review and meta-analysis. Otolaryngol Head Neck Surg. 2020; 163: 3-11.
91. Nijakowski K, Wyzga S, Singh N, Podgórski F, Surdacka A. Oral Manifestations in SARS-CoV-2 Positive Patients: A Systematic Review. J. Clin. Med. 2022; 11: 2202. https://doi.org/10.3390/jcm11082202
92. Majorana A, Bardellini E, Flocchini P, Amadori F, Conti G, Campus G. Oral mucosal lesions in children from 0 to 12 years old: ten years' experience. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2010; 110:e13-8.
93. Halboub E, Ali Al-Maweri S, Alanazi Rh, Qaid Nm, Abdulrab S. Orofacial Manifestations Of Covid-19: A Brief Review Of The Published Literature. Braz. Oral Res. 2020; 34: E124.
94. Orilisi G, Mascitti M, Togni L, Monterubbianesi R, Tosco V, Vitiello F, et al. Oral Manifestations of COVID-19 in Hospitalized Patients: A Systematic Review. Int. J. Environ. Res. Public Health. 2021; 18: 12511. https://doi.org/10.3390/ijerph182312511
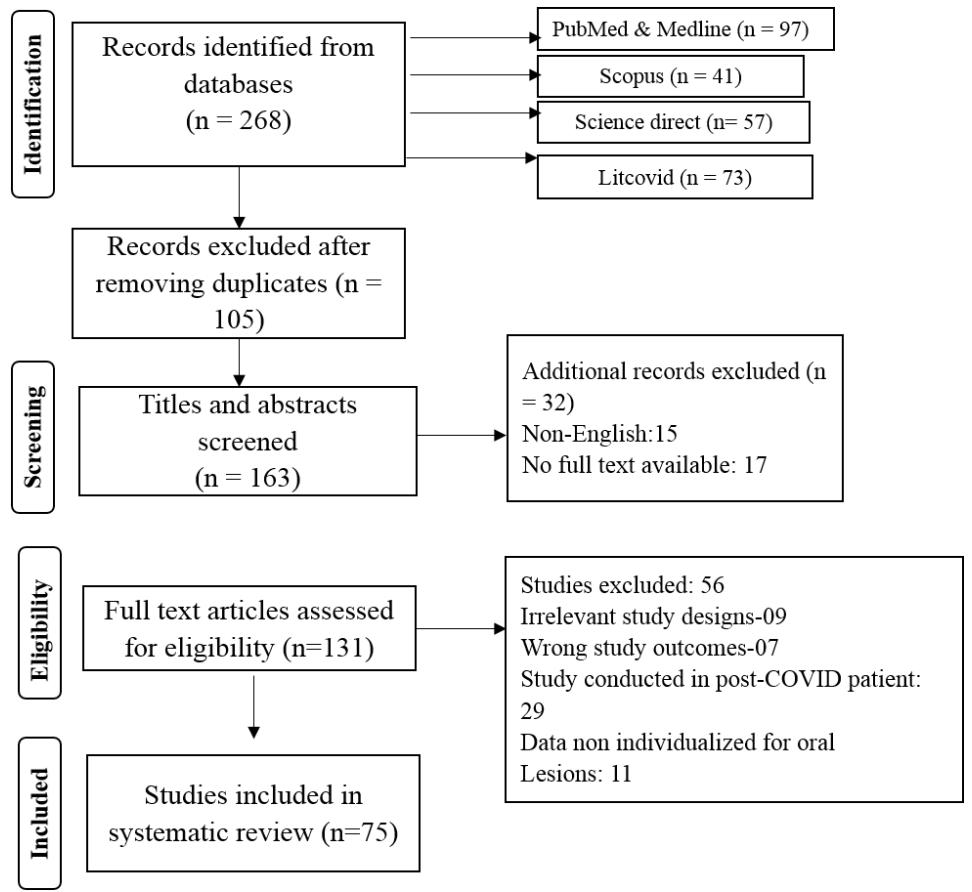
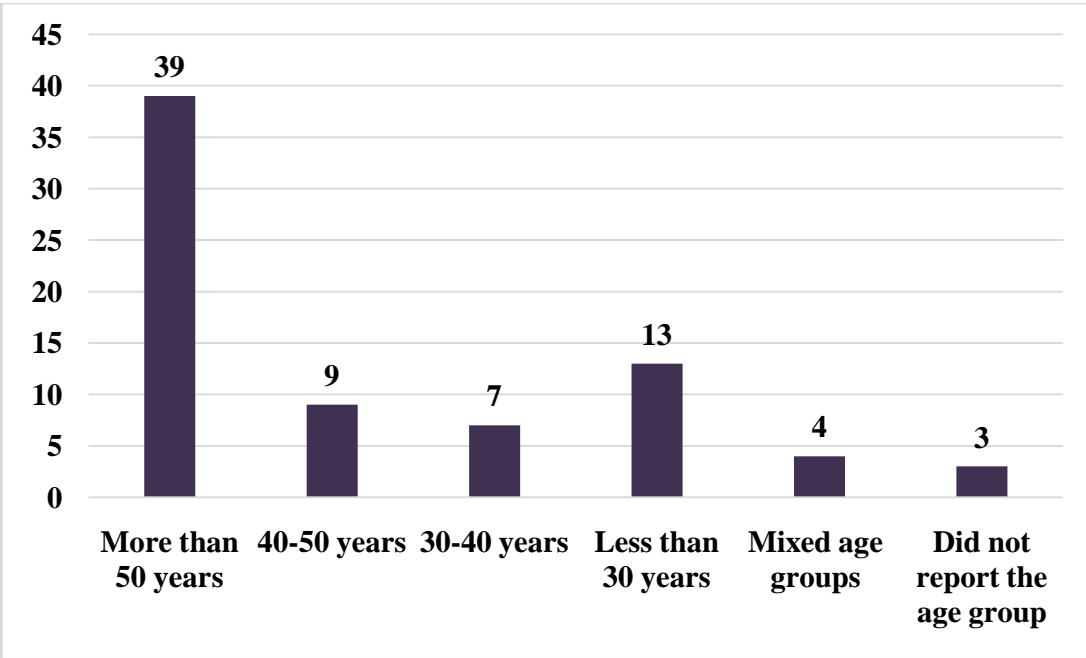
Figure 1: Flow diagram of literature search and selection criteria of the included studies (n=75)
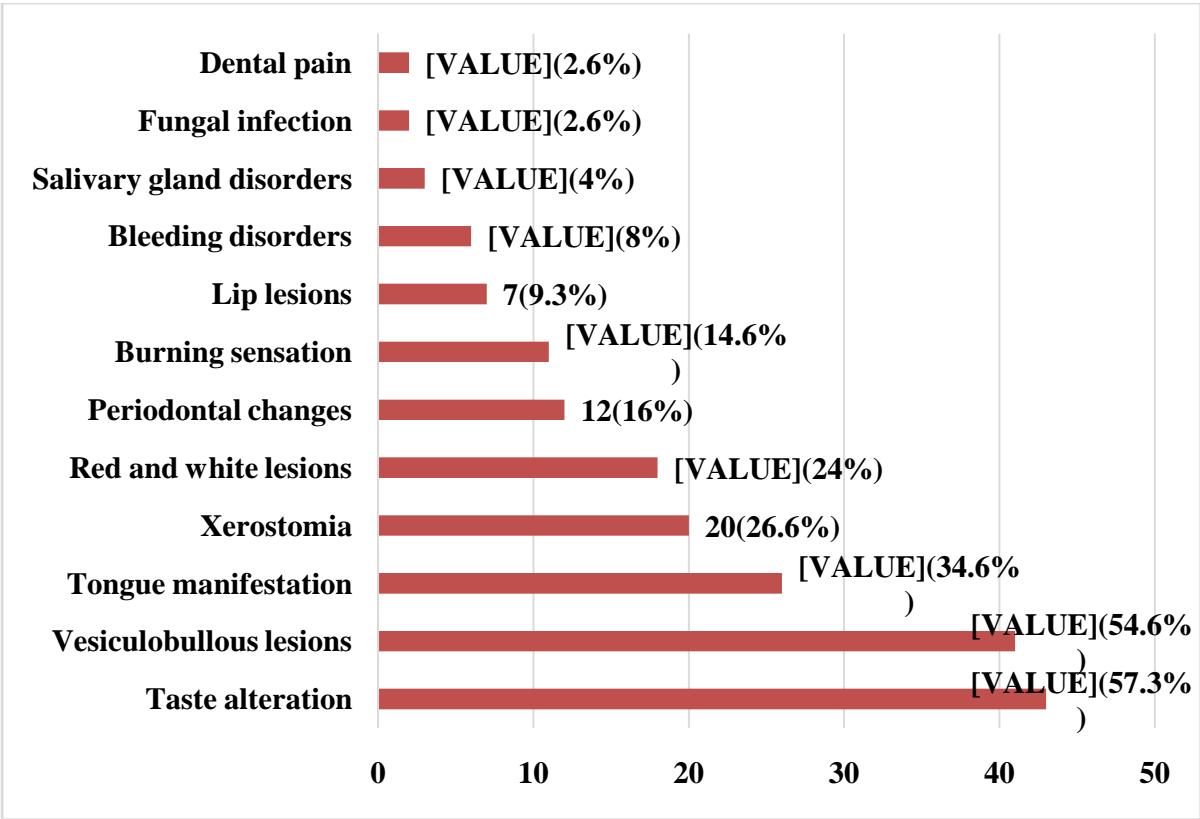
Figure 3: Categories of oral manifestation among the patients with COVID-19
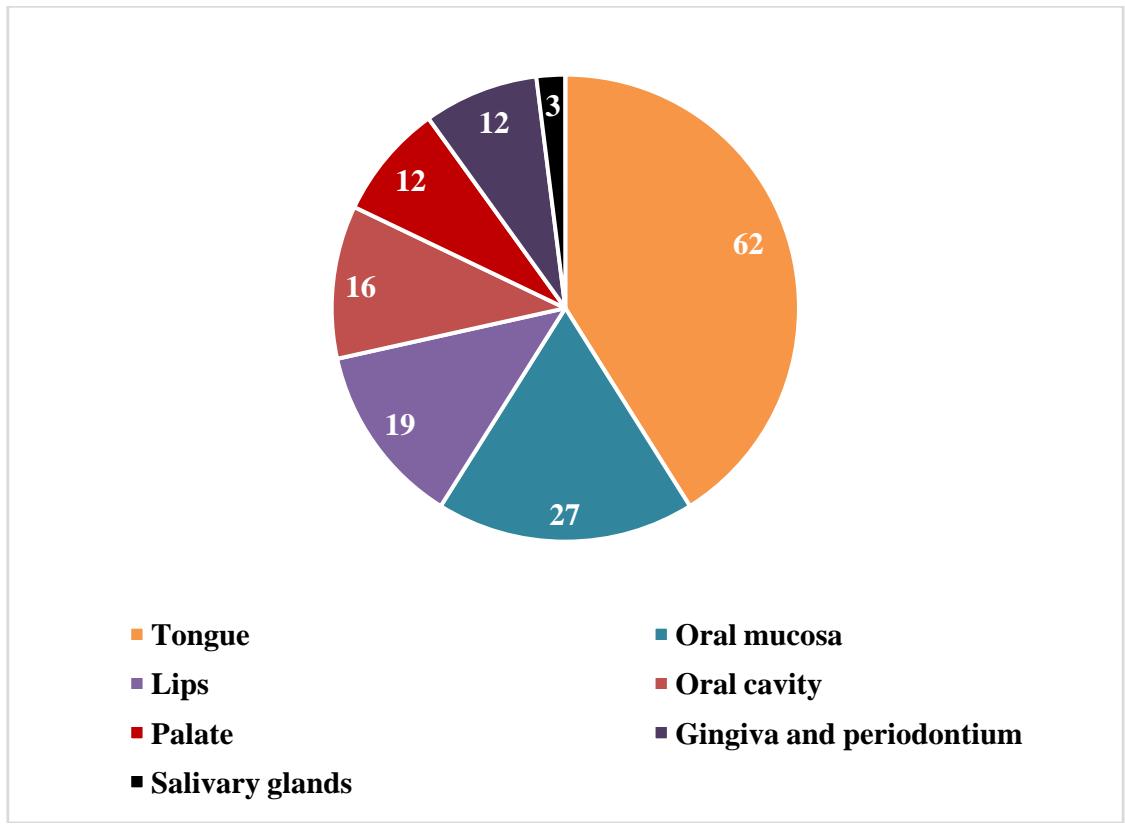 Figure 4: Intraoral sites of involvement among COVID-19 patients
Table 1: Demographic characteristics of the included studies (n=75)
<table><tr><td>S. No.</td><td>Author & Year</td><td>Study location</td><td>Study design</td><td>Sample size</td><td>Gender</td><td>Age</td><td>Study duration</td><td>Medical history</td><td>Admission in the ICU</td><td>Severity of the disease</td></tr><tr><td>1.</td><td>Al-Zaidi and Badr (2020) [12]</td><td>Iraq</td><td>Cross-sectional</td><td>65</td><td>M-41.6% F-58.4%</td><td>41.2 yrs</td><td>5 April 2020 - 17 May 2020</td><td>-</td><td>-</td><td>Moderate</td></tr><tr><td>2.</td><td>Biadsee et al (2020) [13]</td><td>Israel</td><td>Cross-sectional</td><td>140</td><td>M-58; F-70</td><td>36.5 yrs</td><td>March 25, 2020, and April 15, 2020.</td><td>-</td><td>-</td><td>Mild</td></tr><tr><td>3.</td><td>Bodnia and Katzenstein (2020) [14]</td><td>Copenhagen</td><td>Cross-sectional</td><td>65</td><td>F-22; M-28</td><td>45 yrs</td><td>March 2020</td><td>-</td><td>-</td><td>Mild</td></tr><tr><td>4.</td><td>Chen L et al (2020) [15]</td><td>China</td><td>Cross-sectional</td><td>31</td><td>M-15 F-16</td><td>60.6 yrs</td><td>28 February 2020 to 4 March 2020</td><td>-</td><td>-</td><td>-</td></tr><tr><td>5.</td><td>Dell V et al (2020) [16]</td><td>Italy</td><td>Cross-sectional</td><td>355</td><td>M-54%</td><td>50 yrs</td><td>March10 to 30, 2020</td><td>Cardiovascular disease, allergic (Sinusitis)</td><td>-</td><td>Mild to Moderate</td></tr><tr><td>6.</td><td>Kumar L et al (2020) [17]</td><td>India</td><td>Cross-sectional</td><td>141</td><td>M-58.9%; F-41.1%.</td><td>15.2 yrs</td><td>May to August 2020</td><td>-</td><td>-</td><td>Mild to Moderate</td></tr><tr><td>7.</td><td>Lechien JR et al (2020) [18]</td><td>Europe(Multi Centre)</td><td>Cross-sectional</td><td>417</td><td>F-263; M-154</td><td>36.9 ± 11.4 yrs</td><td>-</td><td>Allergic Rhinitis Asthma Hypertension Hypothyroidism</td><td>Hospitalisation of severe cases</td><td>Mild Moderate Severe</td></tr><tr><td>8.</td><td>Paderno A et al (2020) [19]</td><td>Italy</td><td>Cross-sectional</td><td>508</td><td>M-56% F-44% (55±15 years)</td><td>55±15 years</td><td>March 27 to April 1, 2020</td><td>-</td><td>Hospitalisation of severe cases</td><td>Mild Moderate Severe</td></tr><tr><td>9.</td><td>Rizzo PB et al (2020) [20]</td><td>Italy</td><td>Cross-sectional</td><td>202</td><td>F-55.1% M-44.9%</td><td>56 yrs</td><td>March 19 and March 22, 2020</td><td>-</td><td>-</td><td>Mild</td></tr><tr><td>10.</td><td>Yan CH et al (2020) [21]</td><td>California</td><td>Cross-sectional</td><td>59 and 203 (Covid +ve and -ve)</td><td>M&F-49.2% (Covid +ve); M-34%, F-65% (covid -ve)</td><td>54 yrs</td><td>March 3, 2020, and March 29, 2020</td><td>Allergic Rhinitis, immuno comprosised state, hypertenson, DM, Cardiac Disorders, Cancer CLD, History of Head Trauma, Neurological disease</td><td>Hospitalisation of severe cases</td><td>Mild Moderate Severe</td></tr><tr><td>11.</td><td>Sinjari B et al (2020) [22]</td><td>Italy</td><td>Cross-sectional</td><td>20</td><td>-</td><td>-</td><td>May 2020 to June 2020)</td><td>DM, Cardiov vascular condi tions</td><td>-</td><td>Mild to Moderate</td></tr><tr><td>12.</td><td>Giacomelli A et al (2020) [23]</td><td>Italy</td><td>Cross-sectional</td><td>59</td><td>M-40% F-60%</td><td>60 yrs</td><td>March 19,2020</td><td>-</td><td>-</td><td>Mild</td></tr><tr><td>13.</td><td>Mascitti H et al (2020) [24]</td><td>France</td><td>Cross-sectional</td><td>59</td><td>M:F-3:1</td><td>Median age (IQR) was 57.6 (49.4-69.1) years.</td><td>March 31, 2020</td><td>-</td><td>-</td><td>Mild</td></tr><tr><td>14.</td><td>Salehi M et al (2020) [25]</td><td>Iran</td><td>Cross-sectional</td><td>53</td><td>M-43.4%; F-56.6%</td><td><50 yrs-20.7%; ≥50 yrs-79.3%</td><td>1 March 2020 to 30 April 2020</td><td>Cardiov vascular diseas s (52.83) and DM (37.7%; Chronic kidney disease-20.7%</td><td>-</td><td>Mild to Moderate</td></tr><tr><td>15.</td><td>Askin O et al (2020) [26]</td><td>Turkey</td><td>Cross-sectional</td><td>210</td><td>M-58.6%; F-41.4%</td><td>7.44 ± 17.259 yrs</td><td>April 2020.</td><td>Comorbidities</td><td>29 in ICU 129 in wards</td><td>Moderate and Severe</td></tr><tr><td>16.</td><td>Katz J et al (2020) [27]</td><td>USA</td><td>Retrospective study</td><td>889</td><td>F-509 M-386</td><td>18-34 yrs-66%</td><td>Registry Study</td><td>-</td><td>-</td><td>-</td></tr><tr><td>17.</td><td>Fantozzi PJ et al (2021) [28]</td><td>Italy</td><td>Retrospective study</td><td>326</td><td>M-52.3% F-47.7%</td><td>Median age-57 (48-67) days</td><td>6 March to 30 April 2020</td><td>Hypertension (n = 29), chronic pulmon ary disease (n = 11), DM (n = 10), cardiovascular disease (n = 9), cancer (n = 5)</td><td>Hospitalized (median no of days-12.5 days)</td><td>Moderate and severe</td></tr><tr><td>18.</td><td>Favia G et al (2021) [29]</td><td>Bari, Italy</td><td>Cross-sectional</td><td>123</td><td>M:F ratio 1.3:1</td><td>Median age 72 yrs</td><td>October 2020 to December 2020</td><td>-</td><td>History of Hospitaliza tion and ICU</td><td>Moderate and severe</td></tr><tr><td>19.</td><td>Halepas S et al (2021) [30]</td><td>New York</td><td>Cross-sectional</td><td>47</td><td>M-51.1%; F-48.9%</td><td>9.0 ± 5.0 yrs</td><td>March 15 through June 1, 2020</td><td>-</td><td>History of Hospitaliza tion, ICU</td><td>Mild Moderate Severe</td></tr><tr><td>20.</td><td>Rekhtman S et al (2021) [31]</td><td>New York</td><td>Prospective cohort study</td><td>296</td><td>M-71% F-29%</td><td>Median age- 64 (57-77)</td><td>May 11, 2020 and June 15, 2020</td><td>CAD- 23%; Congestive heart failure- 14%; Asthma 9%; COPD- 14%; DM- 34%; Hypertension- 71%</td><td>History of Hospitalization</td><td>Moderate and Severe</td></tr><tr><td>21.</td><td>Maraouf N et al (2021) [32]</td><td>Qatar</td><td>Case control</td><td>Cases- 40; Control- 528</td><td>Cases-M- 50%; F- 50% Controls- M-54.9%; F-45.1%</td><td>Cases- 53.6 yrs Controls -41.5 yrs</td><td>February and July 2020</td><td>DM- Cases- 42.5% Controls -27.8%</td><td>Hospitalization and ICU admission</td><td>Mild Moderate Severe</td></tr><tr><td>22.</td><td>Nuno-Gonzalez A et al [5](2021)</td><td>Spain</td><td>Cross-sectional</td><td>666</td><td>-</td><td>55.7yrs</td><td>10 and 25 April 2020</td><td>-</td><td>History of Hospitalisa tion</td><td>Mild Moderate</td></tr><tr><td>23.</td><td>Subramania m T et al (2021) [33]</td><td>India</td><td>Cross-sectional</td><td>713</td><td>M:F-6:3</td><td>69 yrs</td><td>May 2020 and June 2020</td><td>DM Hyperte nsion</td><td>-</td><td>Mild Moderate</td></tr><tr><td>24.</td><td>Abubakr N et al (2021) [34]</td><td>Egypt</td><td>Cross-sectional</td><td>573</td><td>408 females and 165 males.</td><td>36.19 ±9.11 years</td><td>May 1, 2020 to July 1, 2020</td><td>-</td><td>-</td><td>Mild Moderate</td></tr><tr><td>25.</td><td>Song J et al (2021) [35]</td><td>China</td><td>Retrospective</td><td>117 2</td><td>-</td><td>-</td><td>December 2019</td><td>-</td><td>History of Hospitaliza tion</td><td>Mild</td></tr><tr><td>26.</td><td>Bardellini E et al (2021) [36]</td><td>Italy</td><td>Retrospective</td><td>27</td><td>M:F-19:8</td><td>4.2 yrs</td><td>March to April 2020</td><td>-</td><td>-</td><td>Mild</td></tr><tr><td>27.</td><td>Gherlone EF et al (2021) [37]</td><td>Italy</td><td>Cross-sectional</td><td>122</td><td>M-75.4% F-24.6%</td><td>62.5 yrs</td><td>July 23, 2020 to September 7, 2020</td><td>CAD, DM, Chronic kidney disease, active neoplasia a COPD</td><td>History of Hospitaliza tion and ICU and Ventilation</td><td>Moderate Severe</td></tr><tr><td>28.</td><td>El Kady DM et al (2021) [38]</td><td>Egypt</td><td>Online survey</td><td>58</td><td>M-53.4%; F-46.6%</td><td>18-46 yrs</td><td>May 15 to June 10, 2020</td><td>-</td><td>History of Hospitalisa tion</td><td>Mild</td></tr><tr><td>29.</td><td>Fidan V et al (2021) [39]</td><td>Turkey</td><td>Cross-sectional</td><td>74</td><td>M-66.2% F-33.8%</td><td>51.4 ± 6.3 yrs</td><td>April to October 2020</td><td>-</td><td>Hospitalize d</td><td>Mild Moderate</td></tr></table>

Figure 2: Age distribution of the patients included in the systematic review (n=75)
<table><tr><td>30.</td><td>Natto ZS et al (2021) [40]</td><td>Saudi Arabia</td><td>Cross-sectional study</td><td>109</td><td>M-67% F-33%</td><td>39.3±1 2.4 yrs</td><td>July-October 2020</td><td>DM (10.1%); hypertensive (7.3%); asthma and arthritis (1.7%)</td><td>-</td><td>-</td></tr><tr><td>31.</td><td>Elamrousy WAH et al (2021) [41]</td><td>Egypt</td><td>Cross-sectional</td><td>124</td><td>M-74.2%; F-25.8%</td><td>50.32 ± 12.47 yrs</td><td>2 September 2020, to 10 June 2021</td><td>DM (n = 52), hypertensive (n = 16), cardiac disease (n = 8), renal disease (n = 4), liver disease (n = 4)</td><td>Hospitalized</td><td>Severe (58.1%)</td></tr><tr><td>32.</td><td>Bulut DG et al (2021) [42]</td><td>Turkey</td><td>Cross-sectional</td><td>200</td><td>M-75 F-125</td><td>20-30 yrs: 89 (62/27), 31-40: 65 (43/22), 41-50: 27 (14/13), 51-60: 15 (4/11), 61-70: 4 (2/2)</td><td>September 2020 to March 2021</td><td>-</td><td>Hospitalized (11.5%)</td><td>Moderate Severe</td></tr><tr><td>33.</td><td>Naser Al et al (2021) [43]</td><td>Iraq</td><td>Prospective study</td><td>338</td><td>M-59%; F-41%</td><td>Mean age-45 yrs</td><td>August 2020 to March 2021</td><td>Respiratory diseases, DM, hypertension, heart disease, urogenital disease, hematological disease, gastrointestinal disease</td><td>Hospitalized</td><td>critical admitted cases-38.6%</td></tr><tr><td>34.</td><td>Muthyam AK et al (2022) [44]</td><td>India</td><td>Cross-sectional</td><td>100</td><td>M-51% F-49%</td><td>More than 35 yrs-54%; Less than 35 yrs-46%</td><td>-</td><td>Immuno compromised state, Multidrug therapy</td><td>Hospitalation</td><td>Mild to Moderate</td></tr><tr><td>35.</td><td>Ganesan A et al (2022) [45]</td><td>India</td><td>Cross-sectional</td><td>500</td><td>M-73.4% F-26.6%</td><td>53.46 ± 17.50 years</td><td>-</td><td>-</td><td>-</td><td>-</td></tr><tr><td>36.</td><td>El Tantawi M et al (2022) [46]</td><td>Multicountry study (Saudi Arabia)</td><td>Cross-sectional</td><td>434</td><td>M-41.5% F-58.5%</td><td>18-23 yrs</td><td>August 2020 to January 2021</td><td>Cancer and COPD</td><td>-</td><td>Mild</td></tr><tr><td>37.</td><td>Soares CD et al (2022) [47]</td><td>Brazil</td><td>Retrospective</td><td>14</td><td>M-71.5% F-38.5%</td><td>58 yrs</td><td>-</td><td>-</td><td>-</td><td>-</td></tr><tr><td>38.</td><td>Tuter G et al (2022) [48]</td><td>Turkey</td><td>Cross-sectional</td><td>204</td><td>M-37.3% F-62.7%</td><td>53.3 yrs</td><td>February 2021 to March 2021</td><td>DM hypertensive Immuno supressi on</td><td>Hospitalisa tion ICU</td><td>Mild Moderate Severe</td></tr><tr><td>39.</td><td>Schwab G et al (2022) [49]</td><td>Brazil</td><td>Cross-sectional</td><td>154</td><td>M-59.7% F-40.3%</td><td>54.60 ± 13.93 y ears</td><td>January 13 to May 28 of 2021</td><td>-</td><td>Hospitalisa tion ICU Ventilation</td><td>Moderate to Severe (discharged /death)</td></tr><tr><td>40.</td><td>Chawla J et al (2022) [50]</td><td>India</td><td>C Cross-sectional</td><td>217</td><td>M-70% F-30%</td><td>50-60 yrs</td><td>September and December 2020</td><td>DM, hypertensive CAD, bronchi al Asthma</td><td>-</td><td>Mild Moderate</td></tr><tr><td>41.</td><td>Binmadi NO et al (2022) [51]</td><td>Saudi Arabia</td><td>Cross-sectional</td><td>195</td><td>M-25% F-75%</td><td>33% were 18 to 24 years old and 33% were 25 to 34 years old.</td><td>March of 2020 and March of 2022</td><td>Immuno supressi on, hormonal modulat ion</td><td>Hospitalisa tion ICU Ventilation</td><td>Mild Moderate Severe Critical</td></tr><tr><td>42.</td><td>de Paula Eduardo F et al (2022) [52]</td><td>Brazil</td><td>Retrospective</td><td>519</td><td>M-68.2% F-31.8%</td><td>51-80 yrs</td><td>May 2020 to February 2021</td><td>-</td><td>ICU</td><td>Severe</td></tr><tr><td>43.</td><td>Villarroel-Dorrego M et al (2022) [53]</td><td>Spain</td><td>Cross-sectional</td><td>55</td><td>M-54.5% F-45.5%</td><td>51 ± 23.24 y</td><td>-</td><td>-</td><td>-</td><td>-</td></tr><tr><td>44.</td><td>Manifar S et al (2022) [54]</td><td>Iran</td><td>Cross-sectional</td><td>140</td><td>M-44.2% F-55.8%</td><td>53.78 ± 17.44 yrs</td><td>1 September 2020 to 17 October 2020</td><td>-</td><td>Hospitalisa tion</td><td>Moderate Severe</td></tr><tr><td>45.</td><td>Bhuyan R et al (2022) [55]</td><td>India</td><td>Cross-sectional</td><td>169 (first wave) 211 (2ndwave)</td><td>1st wave-M-35.5%; F-64.5% 2ndwave-M-45.5%; F-55.5%</td><td>63 ±17 and 57 ± 18 (1st and 2nd wave)</td><td>-</td><td>Comorb idities</td><td>Hospitalisa tion ventilator</td><td>Mild Moderate Severe</td></tr><tr><td>46.</td><td>Brandao TB et al (2020) [56]</td><td>Brazil</td><td>Case series</td><td>08</td><td>M-05; F-03</td><td>53 yrs</td><td>-</td><td>Hyperte nision COPD (case 1); DM, obesity,</td><td>Hospitalisa tion</td><td>Mild Moderate Severe-Critical</td></tr><tr><td></td><td></td><td></td><td></td><td></td><td></td><td></td><td></td><td>renalFailure, bariatric surgery, fibromyalgia (case 2); obesity, Parkinson on disease, hypertension, COPD (case 3) DM and Hypertension (case 4)</td><td></td><td></td></tr><tr><td>47.</td><td>Dima M et al (2020) [9]</td><td>Romania</td><td>Case series</td><td>03</td><td>M:F-2:1</td><td>Newbor ns</td><td>May 2020</td><td>Diaper erythema</td><td>Neonatology Ward</td><td>Mild</td></tr><tr><td>48.</td><td>Tapia ROC et al (2020) [57]</td><td>Latin America</td><td>Case series</td><td>04</td><td>F:M-3:1</td><td>47.2 ± 6.8 yrs</td><td>-</td><td>-</td><td>Case 2-Hospitalised</td><td>Mild (case 1 & 3); hospitalized (case 2); moderate (case 4)</td></tr><tr><td>49.</td><td>Vaira LA et al (2020) [58]</td><td>Italy</td><td>Case series</td><td>72</td><td>M-27; F-45</td><td>49.2 yrs</td><td>March 31, 2020 and April 6, 2020.</td><td>History of head trauma, allergic rhinitis, chronic rhino sinusitis, and psychiatric or neurological disorders.</td><td>-</td><td>Mild Moderate</td></tr><tr><td>50.</td><td>Presas CMC et al (2020) [59]</td><td>Spain</td><td>Case series</td><td>03</td><td>M:F-2:1</td><td>59 yrs</td><td>last week of March and the first week of April 2020</td><td>DM & hypertensive (case 2); Obesity and Hypertension (case 3)</td><td>Case 3-Hospitalisation</td><td>Case 1-Mild Case 3-Moderate to severe</td></tr><tr><td>51.</td><td>Sinadinosand Shelswell (2020) [6]</td><td>United Kingdom</td><td>Case series</td><td>03</td><td>M:F-2:1</td><td>58 yrs</td><td></td><td>DM and Hypertensive (case 2); obesity (case 3)</td><td>-</td><td>Mild to Moderate</td></tr><tr><td>52.</td><td>Rodriguez MD et al (2020) [60]</td><td>Spain</td><td>Case series</td><td>03</td><td>F:M-2:1</td><td>53 yrs</td><td>-</td><td>-</td><td>Case 1 - Home quarantine Case 2 & 3-Hospitalisa</td><td>Case 1-Mild Case 2-Moderate Case 3-Moderate</td></tr><tr><td></td><td></td><td></td><td></td><td></td><td></td><td></td><td></td><td></td><td>tion</td><td></td></tr><tr><td>53.</td><td>Corchuelo and Ulloa (2020) [61]</td><td>Colombia</td><td>Case report</td><td>01</td><td>Female</td><td>40 yrs</td><td>-</td><td>-</td><td>-</td><td>Moderate</td></tr><tr><td>54.</td><td>Dos Santos et al (2020) [10]</td><td>Brazil</td><td>Case report</td><td>01</td><td>Male</td><td>67 yrs</td><td>March 31, 2020</td><td>CAD, autosomal dominant polycystic kidney disease, and kidney transplants, immuno suppres sion, venous thrombo embolism</td><td>Hospitalisation in ICU</td><td>Severe</td></tr><tr><td>55.</td><td>Zarch and Hosseinzadeh (2020) [62]</td><td>Iran</td><td>Case report</td><td>01</td><td>Female</td><td>56 yrs</td><td>October 2020</td><td>-</td><td>-</td><td>-</td></tr><tr><td>56.</td><td>Hjelmeseth J (2020) [63]</td><td>Norway</td><td>Case report</td><td>01</td><td>Female</td><td>60 yrs</td><td>-</td><td>-</td><td>-</td><td>-</td></tr><tr><td>57.</td><td>Kahraman and Çaşkurlu (2020) [64]</td><td>Turkey</td><td>Case report</td><td>01</td><td>Male</td><td>51 yrs</td><td>18 March 2020</td><td>-</td><td>-</td><td>Moderate</td></tr><tr><td>58.</td><td>Smith AC et al (2020) [65]</td><td>United States</td><td>Case report</td><td>01</td><td>Male</td><td>21 yrs</td><td>March 19, 2020</td><td>-</td><td>-</td><td>Mild</td></tr><tr><td>59.</td><td>Maniaci A et al (2020) [66]</td><td>Italy</td><td>Case report</td><td>01</td><td>Male</td><td>15 yrs</td><td>-</td><td>-</td><td>-</td><td>Mild</td></tr><tr><td>60.</td><td>Melley LE et al (2020) [67]</td><td>USA (Pennsylvania)</td><td>Case report</td><td>01</td><td>Female</td><td>59 yrs</td><td>May 2020</td><td>-</td><td>-</td><td>-</td></tr><tr><td>61.</td><td>Riad A et al (2020) [68]</td><td>Egypt</td><td>Case report</td><td>01</td><td>Female</td><td>47 yrs</td><td>-</td><td>Cardiovascular disease DM</td><td>-</td><td>Moderate</td></tr><tr><td>62.</td><td>Putra BE et al (2020) [69]</td><td>Indonesia</td><td>Case report</td><td>01</td><td>Male</td><td>29 yrs</td><td>-</td><td>Cardiovascular diseases</td><td>-</td><td>Moderate</td></tr><tr><td>63.</td><td>Dalipi ZS et al (2021) [70]</td><td>Europe</td><td>Case report</td><td>01</td><td>Male</td><td>17 yrs</td><td>-</td><td>-</td><td>-</td><td>-</td></tr><tr><td>64.</td><td>Eita AAB et al (2021) [71]</td><td>Egypt</td><td>Case report</td><td>01</td><td>Female</td><td>31 yrs</td><td>-</td><td>Irritable Bowel Syndrome</td><td>-</td><td>Severe</td></tr><tr><td>65.</td><td>Cirillo and Colello (2021) [72]</td><td>Italy</td><td>Case report</td><td>01</td><td>Female</td><td>36 yrs</td><td>March 2020</td><td>-</td><td>-</td><td>Mild</td></tr><tr><td>66.</td><td>Nejabi MB et al (2021) [73]</td><td>Afghanistan</td><td>Case report</td><td>01</td><td>Male</td><td>62 yrs</td><td>-</td><td>-</td><td>-</td><td>Mild</td></tr><tr><td>67.</td><td>Klein H et al (2021) [74]</td><td>Israel</td><td>Case report</td><td>01</td><td>Female (pregnant)</td><td>40 yrs</td><td>-</td><td>-</td><td>-</td><td>Mild</td></tr><tr><td>68.</td><td>Ramires MCCH et al (2021) [75]</td><td>Brazil</td><td>Case report</td><td>01</td><td>Female</td><td>50 yrs</td><td>-</td><td>Obesity, hypertension, and type2 DM</td><td>Hospitalisation Ventilation</td><td>Severe</td></tr><tr><td>69.</td><td>Hocková B et al (2021) [76]</td><td>Czech Republic</td><td>Case series</td><td>03</td><td>M:F-3:0</td><td>62 yrs</td><td></td><td>Arterial hypertension, hypercholesterolemia, GERD (case 1); Arterial hypertension, history of MI and septic shock (case 2)</td><td>ICU</td><td>Severe</td></tr><tr><td>70.</td><td>Teixeira IS et al (2021) [77]</td><td>Brazil</td><td>Case series</td><td>04</td><td>M:F-1:3</td><td>57 yrs, 84 yrs, 70 yrs, 64 yrs</td><td>-</td><td>Hypertension, hypothyroidism, and rectal tumor (case 2); hypertension, hypothyroidism (case 3); bipolar disorder (case 4)</td><td>-</td><td>-</td></tr><tr><td>71.</td><td>Emelyanova N et al (2021) [78]</td><td>Ukraine</td><td>Case report</td><td>01</td><td>Female</td><td>38 yrs</td><td>-</td><td>-</td><td>-</td><td>-</td></tr><tr><td>72.</td><td>Fathi Y et al (2021) [79]</td><td>Iran</td><td>Case report</td><td>01</td><td>Female</td><td>22 yrs</td><td>April 2020</td><td>-</td><td>Hospitalization (2nd day)</td><td></td></tr><tr><td>73.</td><td>Shenoy P et al (2022) [80]</td><td>India</td><td>Case report</td><td>01</td><td>Female</td><td>55 yrs</td><td>-</td><td>-</td><td>-</td><td>-</td></tr><tr><td>74.</td><td>Palaia G et al (2022) [81]</td><td>Italy</td><td>Case report</td><td>01</td><td>Female</td><td>30 yrs</td><td>-</td><td>-</td><td>-</td><td>-</td></tr><tr><td>75.</td><td>Rafałowicz B et al (2022) [82]</td><td>Poland</td><td>Case series</td><td>06</td><td>M-4 F-2</td><td>43 yrs, 72 yrs, 53 yrs, 48 yrs, 66 yrs; 71 yrs</td><td>January-June 2021</td><td>Hypertension and insulin resistance (case 2)</td><td>No</td><td></td></tr></table>
Table 2: Oral manifestations of the included studies (n=75)
<table><tr><td>S.No.</td><td>Author & year</td><td>Oral manifestation</td><td>Site</td><td>Type of oral manifestation</td><td>Occurrence/duration of oral manifestation</td><td>Systemic manifestation</td></tr><tr><td>1.</td><td>Al-Zaidi and Badr (2020) [12]</td><td>Loss of taste (83%)</td><td>Tongue</td><td>Taste alterations</td><td>1 week before systemic symptoms</td><td>Fever (63.08%), cough (60.00%), dyspnea (47.69%), sore throat, diarrhea (32.31%), chest pain (30.77%).</td></tr><tr><td>2.</td><td>Biadsee et al (2020) [13]</td><td>Taste alteration (n=67), dry mouth (72), plaque-like changes in the tongue (9), swelling in the oral cavity (10)</td><td>Tongue, oral cavity</td><td>Taste alteration, tongue manifestation, xerostomia</td><td>Along with systemic symptoms</td><td>Cough and runny nose (p = 0.018), olfactory dysfunction</td></tr><tr><td>3.</td><td>Bodnia and Katzenstein (2020) [14]</td><td>Total loss of taste (70%)</td><td>Tongue</td><td>Taste alterations</td><td>1-3 weeks (78%), 3-6 weeks (22%)</td><td>Fatigue, headache, fever, dry cough and disturbance of the sense of smell</td></tr><tr><td>4.</td><td>Chen L et al (2020) [15]</td><td>Amblygeustia (47.2%), dry mouth (11.1%)</td><td>Tongue, oral cavity</td><td>Taste alteration, xerostomia</td><td>Along with systemic symptoms</td><td>Submandibular lymph node enlargement (1); cough (21); fever (20); diarrhea (04); chest tightness (13)</td></tr><tr><td>5.</td><td>Dell V et al (2020) [16]</td><td>Taste disorders (65.5%)</td><td>Tongue</td><td>Taste alterations</td><td>Mean duration: 10 days</td><td>Fever (72.1%); cough (47.9%); fatigue (40.3%); dyspnea (21.7%); diarrhea (19.7%)</td></tr><tr><td>6.</td><td>Kumar L et al (2020) [17]</td><td>Taste Dysfunction (28.4%)</td><td>Tongue</td><td>Taste alterations</td><td>Duration: 2-15 days</td><td>Malaise (14.2%), sore throat (19.9%), cough (20.6%), fever (48.2%), diarrhea (5.7%), nasal discharge (3.5%) headache (5.7%)</td></tr><tr><td>7.</td><td>Lechien JR et al (2020) [18]</td><td>Gustatory dysfunction (88.8%)</td><td>Tongue</td><td>Taste alterations</td><td>Mean duration: 9.2 ± 6.2 days</td><td>Olfactory dysfunction (85.6%)</td></tr><tr><td>8.</td><td>Paderno A et al (2020) [19]</td><td>Gustatory dysfunction (group a-51.9%) Group b-78.9%) Partial-36.8% Total-60.1% Unable to assess-3.1%</td><td>Tongue</td><td>Taste alterations</td><td>First symptom in 11.9% (group a) and 10.2% (group b) Mean duration: 9.2 ± 5.4</td><td>Olfactory dysfunction, fever, cough, headache, dyspnea, asthenia, diarrhea, nausea, nasal congestion, phangodynia</td></tr><tr><td>9.</td><td>Rizzo PB et al (2020) [20]</td><td>Loss of taste (n=113)</td><td>Tongue</td><td>Taste alterations</td><td>Mean duration: 9.5 days</td><td>Dry cough, fever, headache, sore throat, chest pain, nausea, abdominal pain</td></tr><tr><td>10.</td><td>Yan CH et al (2020) [21]</td><td>Gustatory impairment -71% (p<0.001)</td><td>Tongue</td><td>Taste alterations</td><td>-</td><td>Fatigue (81%), fever (70%), anosmia (68%), myalgia or arthralgia (63%), diarrhea (48%), nausea (27%).</td></tr><tr><td>11.</td><td>Sinjari B et al (2020) [22]</td><td>Impaired taste (25%), burning sensation (15%), difficulty in swallowing (20%), dry mouth (30%) (p=0.02)</td><td>Oral cavity, tongue</td><td>Taste alterations</td><td>-</td><td>-</td></tr><tr><td>12.</td><td>Giacomelli A et al (2020) [23]</td><td>Dysgeusia (8.5%) Ageusia (1.7%)</td><td>Tongue</td><td>Taste alterations</td><td>Before hospitalization (91%)</td><td>Fever (72.8%), cough (37.3%), dyspnea (25.4%), sore throat (1.7%), arthralgia (5.1%), headache (3.4%), asthenia (1.7%), abdominal symptoms (8.5%)</td></tr><tr><td>13.</td><td>Mascitti H et al (2020) [24]</td><td>Oral lichenoid reaction 32.5%; oral enanthema 27.5%; macroglossia 25.0% cheilitis 12.5%; ageusia-20.5%; extensive ulcerations of the tongue2.5%</td><td>Lips, tongue, oral cavity and oral mucosa</td><td>Red and white lesions, tongue manifestation, taste alteration, vesiculobullous lesion</td><td>-</td><td>Macular exanthema (80%), face edema (32%), livedo (13%), urticarial rash (8%), purpura (5%), oral lichenoid lesions (33%), and conjunctivitis (18%)</td></tr><tr><td>14.</td><td>Salehi M et al (2020) [25]</td><td>White plaques on the intraoral mucous layer</td><td>Mucous membrane</td><td>Red and white lesions</td><td>-</td><td>-</td></tr><tr><td>15.</td><td>Askin O et al (2020) [26]</td><td>Necrosis on maxillary arch (1 case); aphous stomatitis 5.8%; rash and erythema; apthous lesion on side of tongue</td><td>Mucous membrane and tongue</td><td>Vesiculobullous lesion, tongue manifestation, red and white lesions, periodontal changes</td><td>-</td><td>Cutaneous findings (36.1%)</td></tr><tr><td>16.</td><td>Katz J et al (2020) [27]</td><td>Recurrent aphthous stomatitis-0.64%</td><td>Oral mucosa</td><td>Vesiculobullous lesion</td><td>-</td><td>-</td></tr><tr><td>17.</td><td>Fantozzi PJ et al (2021) [28]</td><td>Dry mouth-45.9%; swallowing difficulties,-39.2%; dysgeusia-59.5%</td><td>Tongue; oral cavity</td><td>Taste alteration, xerostomia</td><td>First symptom (xerostomia)-19.6%; dysgeusia (87.9%) Duration (xerostomia) -7 days; dysgeusia 6 days</td><td>Fever (90.9), cough (46.8), dyspnea (34.3), diarrhea (4.5), sore throat (3.6), fatigue (3.6), myalgia/arthralgia (2.7), vomiting (2.7)</td></tr><tr><td>18.</td><td>Favia G et al (2021) [29]</td><td>Geographic tongue (n=7); fissured tongue (5); ulcerative lesion (65); blisters (19); hyperplasia of papillae (48); angina bullosa (11); candidiasis (28); ulceronecrotic gingivitis (7) Petechiae (14); oral haemorrhage (1) Taste disorders (90%)</td><td>Tongue, oral mucosa, lips</td><td>Tongue manifestation, vesiculobullous lesion, red and white lesions, periodontal changes, bleeding disorders, taste alteration</td><td>Together with general symptoms (26.2%); Duration: one week (41%) After 1 week of general symptoms (32.6%)</td><td>Fever, anosmia, cough, sore throat, congestion, runny nose, nausea or vomiting, muscle and body aches, dermatologic manifestation, pneumonia, dyspnea, hypoxia (spo2<90%)</td></tr><tr><td>19.</td><td>Halepas S et al (2021) [30]</td><td>Red and/or swollen lips (48.9%); strawberry tongue (10.6%)</td><td>Lips, tongue</td><td>Lip lesions, tongue manifestation</td><td>-</td><td>Fever</td></tr><tr><td>20.</td><td>Rekhtman S et al (2021) [31]</td><td>Rashes on lips and tongue-5.7% and 2.9%; ulcers on lips and tongue</td><td>Lips and tongue</td><td>Tongue manifestation, vesiculobullous lesion, lip lesions</td><td></td><td>Generalized rashes and vesiculobullous lesions present</td></tr><tr><td>21.</td><td>Maraouf N et al (2021) [32]</td><td>Periodontitis-258/568</td><td>Periodontium</td><td>Periodontal changes</td><td>-</td><td>-</td></tr><tr><td>22.</td><td>Nuno-Gonzalez A et al [5](2021)</td><td>Oral mucosal changes (11.7%), transient anterior U-shaped lingual papillitis (11.5%), tongue swelling (6.6%), aphthous stomatitis (6.9%), burning sensation in the mouth (5.3%), mucositis (3.9%), glossitis with patchy depapillation (3.9%), white tongue (1.6%), and enanthema (0.5%), taste disturbances</td><td>Tongue, oral mucosa</td><td>Tongue manifestation, vesiculobullous lesion, burning sensation</td><td>-</td><td>-</td></tr><tr><td>23.</td><td>Subramaniam T et al (2021) [33]</td><td>Ulcers on oral mucosa (case 1); burning mouth and mucositis on lower labial mucosa (cases 2.5); papillary atrophy (case 3); reddish-white spots on the palate (case 4); ulcers on lower lip (cases 6,7,8); pallor of lip (case 9)</td><td>Oral mucosa, palate, lips, tongue</td><td>Tongue manifestation, vesiculobullous lesion, burning sensation, red and white lesions, lip lesions</td><td>-</td><td>Fever, cough, dyspnea, runny nose, chest tightness, loss of smell</td></tr><tr><td>24.</td><td>Abubakr N et al (2021) [34]</td><td>Dental pain (23%), pain in jaw bones or joint (12.0%), halitosis (10.5%), ulcerations (20.4%), and dry mouth (47.6%)</td><td>Teeth, jaw bones, oral cavity</td><td>Vesiculobullous lesion, xerostomia, pain in teeth and jaw, tmj disturbances</td><td>-</td><td>Fever, myalgia, dysphagia, and hyposmia, loss of smell, nasal itching</td></tr><tr><td>25.</td><td>Song J et al (2021) [35]</td><td>Loss of taste (20.6%; median score, 6)</td><td>Tongue</td><td>Taste alteration</td><td>First symptom (0.4%) Recovery time-7 days</td><td>Nasal obstruction (8.6%), rhinorrhea (10.3%), nasal itching (4.9%), sneezing (11.0%), loss of smell (11.4%)</td></tr><tr><td>26.</td><td>Bardellini E et al (2021) [36]</td><td>Oral pseudomembranous candidiasis (7.4%), geographic tongue (3.7%), coated tongue (7.4%); taste alteration (11.1%)</td><td>Tongue, oral mucosa</td><td>Red and white lesions, tongue manifestation</td><td>-</td><td>Fever, cough, rhinorrhoea, breathing difficulty</td></tr><tr><td>27.</td><td>Gherlone EF et al (2021) [37]</td><td>Salivary gland ectasia-38%; dry mouth-30%; dysgeusia-17%; white plaque-28%; oral ulcers-12%</td><td>Salivary glands, tongue, oral mucosa, oral cavity</td><td>Salivary gland disorders, xerostomia, red and white lesions, vesiculobullous lesions, taste alteration</td><td>-</td><td>-</td></tr><tr><td>28.</td><td>El Kady DM et al (2021) [38]</td><td>Dry mouth 39.7%; loss of salt sensation-34.5%, loss of sweet sensation-29.3%, altered food taste-</td><td>Tongue, salivary glands, gingiva, oral mucosa</td><td>Xerostomia, taste alteration, periodontal changes, salivary gland disorders, vesiculobullous</td><td>-</td><td>-</td></tr><tr><td></td><td></td><td>25.9%; tongue redness 8.8%; gingival bleeding 7%; salivary glands infection 22.4%; swellings in the salivary gland or cheek 13.8%; pain or swelling below mandible-10.8%; burning mouth sensation-22.4%; ulcers-17.2%</td><td></td><td>lesions, burning sensation</td><td></td><td></td></tr><tr><td>29.</td><td>Fidan V et al (2021) [39]</td><td>Aphthous-like ulcer (36.5), erythema (25.7), lichen planus (16.2); tongue (31.8), oral mucosa (27.0), gingiva (14.9), palate (5.4)</td><td>Tongue/oral mucosa/gingiva/palate-39.7%/34.5%/18.9%/6.9%</td><td>Vesiculobullous lesions, red and white lesions, periodontal changes</td><td>Oral lesions prior covid-19 diagnosis</td><td>-</td></tr><tr><td>30.</td><td>Natto ZS et al (2021) [40]</td><td>Loss of taste-43.4%; erythema/desquamaed tingivitis and coated tongue (7.3%); ulcers/blisters (6.4%); pain and soreness (2.8%); dry mouth (0.9%)</td><td>Tongue; gingiva; oral mucosa, oral cavity</td><td>Vesiculobullous lesions, taste alteration, xerostomia</td><td>After systemic symptoms</td><td>Cough, fever, sore throat, runny nose, muscle pain, headaches, nausea, and diarrhea</td></tr><tr><td>31.</td><td>Elamrousy WAH et al (2021) [41]</td><td>Oral ulcers (92.8%); dry mouth (84%); loss of taste (55%); hemorrhagic ulcers with crust on lips</td><td>Lip/tongue/labial mucosa-42.3%/38.5%/34.6%</td><td>Vesiculobullous lesions, lip lesions, taste alteration</td><td>-</td><td>Asthenia (67.7), breath problems (67.7), cough (67.7), fatigue (19.4), abdominal symptoms (12.9)</td></tr><tr><td>32.</td><td>Bulut DG et al (2021) [42]</td><td>Taste loss (53%), halitosis (21%), oropharyngeal wound and pain (18%), pain in the chewing muscles (16%), pain in the temporomandibular joint (17.5%), gum bleeding (17.5%), dry mouth (38%, after recovery 12.0), aphthous ulcer (14.5%), sensitivity and/or pain in teeth (12%), herpes labialis (8.5%), burning in the tongue (7.5%)</td><td>Tongue, gingiva, lips, oral cavity</td><td>Taste alteration, TMJ disturbances, xerostomia, burning sensation, vesiculobullous lesion, periodontal changes, teeth pain</td><td>-</td><td>Presence of symptoms (87.5)</td></tr><tr><td>33.</td><td>Naser Al et al (2021) [43]</td><td>Burning sensation (6%), numbness or tingling of the tongue (2%), white coat of the tongue, gingiva, palate (31.6%, 22.4%, 15.6%), loss of taste (79.5%), aphthous ulcers (24.8%), black discoloration of oral cavity, lips and tongue (4.7%, 6.8%), yellow coating on lips (5.3%)</td><td>Tongue, palate, lips, oral mucosa, oral cavity</td><td>Burning sensation, tongue manifestation, red and white lesions, vesiculobullous lesion, fungal infection, lip lesions</td><td>-</td><td>-</td></tr><tr><td>34.</td><td>Muthyam AK et al (2022) [44]</td><td>Dry mouth (44%) followed by swallowing difficulty, mouth ulcerations, chewing problems, gum bleeding, and burning sensation, altered taste (72%); fissured tongue, halitosis, and loss of taste-2%</td><td>Gums, tongue, oral mucosa, oral cavity</td><td>Xerostomia, vesiculobullous lesions, taste alteration, periodontal changes, tongue manifestation, burning sensation</td><td>Altered taste lasted more than 1 week-53%</td><td>Weakness (8%), cough and cold (4%), and body pain (2%)</td></tr><tr><td>35.</td><td>Ganesan A et al (2022) [45]</td><td>Gustatory disturbance-51.2; dry mouth=28%; erythema, ulcers and depapilation of tongue-15.5% A statistically significant correlation between oral manifestations and disease severity (p≤0.001).</td><td>Tongue, oral mucosa</td><td>Xerostomia, tongue manifestation, taste alteration</td><td>-</td><td>-</td></tr><tr><td>36.</td><td>El Tantawi M et al (2022) [46]</td><td>Dry mouth (11.1% vs 7.5%, p = 0.009) and change in taste (11.5% vs 2.7%, p < 0.001) were greater in COVID-19 person; leukoplakia-4.6%; ulcers & hairy tongue-2.3%;gingival inflammation-13.1%</td><td>Oral cavity, tongue, gingiva</td><td>Vesiculobulous lesions, xerostomia</td><td>-</td><td>-</td></tr><tr><td>37.</td><td>Soares CD et al (2022) [47]</td><td>Lesions in the palate/tongue/lips or palate-57.1%, 29%/14.3%.</td><td>Tongue, lips, palate</td><td>Vesiculobulous lesions</td><td>-</td><td>Anosmia, fever, and headache.</td></tr><tr><td>38.</td><td>Tuter G et al (2022) [48]</td><td>Dry mouth (44.2%); oral lesions (22.4%); oral mucosa (15.2%); tongue (10.8%).</td><td>Tongue, Oral mucosa</td><td>Vesiculobulous lesions, xerostomia</td><td>-</td><td>-</td></tr><tr><td>39.</td><td>Schwab G et al (2022) [49]</td><td>Ageusia - 11.0%; opportunistic oral infections such as pseudomembranous candidiasis and herpes simplex-4.5%</td><td>Tongue, oral mucosa</td><td>Vesiculobulous lesions, taste alteration, red and white lesions</td><td>-</td><td>Cough - 72.7%; dyspnoea - 63.0%; fever - 53.9%; anosmia - 14.3%</td></tr><tr><td>40.</td><td>Chawla J et al (2022) [50]</td><td>Dry mouth (38%) (p=0.03); Dysgeusia (32%) (p=0.04); Vesiculobulous lesion-13%; Oral ulcers-3.7%</td><td>Oral cavity, tongue</td><td>Vesiculobulous lesions, xerostomia, taste alteration</td><td>-</td><td>Cough/sore throat/shortness of breath/running nose-30%/20%/7%/11%</td></tr><tr><td>41.</td><td>Binmadi NO et al (2022) [51]</td><td>Distortion of taste-60%; dry mouth-42%, oral ulcerations-11%, gingivitis/petechiae/candidiasis-6%; necrotizing periodontal disease/vesiculobullous lesions/erythema migrans/geographic tongue-4%</td><td>Gingiva, tongue, oral mucosa, oral cavity</td><td>Vesiculobulous lesions, xerostomia, taste alteration, periodontal changes, red and white lesions, bleeding disorders, tongue manifestation</td><td>Concurrently-47%, after the general symptoms-43%, and before the general symptoms-9%</td><td>Fever (95%), headache (65%), fatigue (65%), cough (63%), myalgia/arthralgia (53%), loss of smell (53%), sore throat (50%), shortness of breath or dyspnea (40%), nausea or vomiting (21%), and diarrhea (15%).</td></tr></table>
<table><tr><td>42.</td><td>de Paula Eduardo F et al (2022) [52]</td><td>Saliva alterations-24.4%; dryness-9.9%; tongue coating-3%; sialorrhea-3.3%; petechiae-10.5%; oral bleeding-7.5%</td><td>Oral mucosa, tongue</td><td>Xerostomia, salivary gland disorders, bleeding disorders, tongue manifestation</td><td>-</td><td>-</td></tr><tr><td>43.</td><td>Villarroel-Dorrego M et al (2022) [53]</td><td>Hemorrhagic ulcerative lesions-7.3%; erythematous and pseudomembranous forms of candidiasis-12.7%; angular cheilitis-1.5%; total loss of taste-60%; burning mouth-36.4%; dry mouth-27.3%</td><td>Tongue, oral mucosa</td><td>Xerostomia, bleeding disorders, red and white lesions, vesiculobullous lesions, taste alteration, burning sensation</td><td>-</td><td>-</td></tr><tr><td>44.</td><td>Manifar S et al (2022) [54]</td><td>Dry mouth-68.6% (p<0.001) Dysgeusia-51.4% (p<0.001) Hypogoeusia-49.3%; halitosis-31.4%; metallic taste-29.3%</td><td>Tongue, oral cavity</td><td>Xerostomia, taste alteration</td><td>-</td><td>Gastrointestinal symptoms, smell defects, asthma, skin rashes, cough, malaise, myalgia, anorexia, respiratory distress, olfactory dysfunction</td></tr><tr><td>45.</td><td>Bhuyan R et al (2022) [55]</td><td>Burning sensation-2.4%; dry mouth-2.4%; loss of taste-31% (p<0.001); mouth ulcer-2.4%. Bleeding gum-2.4%.</td><td>Oral cavity, gums, tongue</td><td>Xerostomia, taste alteration, burning sensation, vesiculobullous lesion, periodontal changes</td><td>-</td><td>-</td></tr><tr><td>46.</td><td>Brandao TB et al (2020) [56]</td><td>Multiple aphthous-like ulcers covered with mucopurulent membrane in the Upper and lower lip mucosa and tongue (cases 1,2,4,5); ulcers on tongue and hard palate (case 3); ulcers on tongue and aguesia (cases 6,7,8)</td><td>Lips, tongue, palate</td><td>Vesiculobullous ulcers, taste alteration, tongue manifestation</td><td>06-10 days</td><td>Chest tightness, fever, cough (cases 1,5,7,8); cough, Fever, dyspnea (cases 2,6); Abdominal distension, fever, mild dyspnea (cases 3 and 4)</td></tr><tr><td>47.</td><td>Dima M et al (2020) [9]</td><td>Oral candidiasis</td><td>Oral mucosa</td><td>Red and white lesions</td><td>-</td><td>Epistaxis and diaper erythema (all 3 cases); palpebral edema (newborn 2)</td></tr><tr><td>48.</td><td>Tapia ROC et al (2020) [57]</td><td>Bulla on the hard palate (x6mm) (case 1); diffuse purple macule (x12mm) and papule-plaque (x8mm) on the left and right palatal mucosa (case 2); tongue enlarge-ment (case 3); Burning mouth sensation and reddish macules on hard palate (case 4)</td><td>Palate, oral mucosa, tongue</td><td>Vesiculobullous lesion, red and white lesions, burning sensation, tongue manifestation</td><td></td><td>Fever, myalgia, dysphagia, and hyposmia</td></tr><tr><td>49.</td><td>Vaira LA et al (2020) [58]</td><td>Hypogoeusia (33 cases) Complete ageusia (1 case)</td><td>Tongue</td><td>Taste alteration</td><td></td><td>Fever, cough, nasal obstruction, sore throat, hyposmia, anosmia, pneumonia</td></tr><tr><td>50.</td><td>Presas CMC et al (2020) [59]</td><td>Dysgeusia (case 1); multiple ulcers On palate (case 2); pain on tongue, blisters in lip mucosa and Desquamative gingivitis (case 3)</td><td>Tongue, lips,</td><td>Taste alteration, periodontal changes, vesiculobullous lesions</td><td>Along with systemic symptoms</td><td>Asthenia; hyposmia, and enlargement of lymph nodes in the neck (cases 1 and 3) Fever and diarrhea (case 2)</td></tr><tr><td>51.</td><td>Sinadinosand Shelswell (2020) [6]</td><td>Pain in palate (case 1); pain and ulcerations in palate (case 2), pain in tongue, blisters of the labial mucosa; desquamative gingivitis (case 3)</td><td>Palate, tongue, gums, lips</td><td>Tongue manifestation, periodontal changes, vesiculobullous lesions</td><td>-</td><td>Sore throat (case 1) Pneumonia (case 3)</td></tr><tr><td>52.</td><td>Rodriguez MD et al (2020) [60]</td><td>Dysgeusia, aphthous-like lesions, burning sensation, and tongue depapillation (case 1); burning mouth sensation and unilateral commissural fissures (case 2); dry mouth, lesions on the tongue, palate, and commissure (case 3)</td><td>Tongue, palate, oral mucosa</td><td>Taste alteration, xerostomia, vesiculobullous lesions, tongue manifestation, burning sensation</td><td>Before presentation (case 1); after discharge (case 2); with systemic symptoms (case 3)</td><td>Fever, malaise, and anosmia, diarrhea, and pneumonia (cases 1 and 3)</td></tr><tr><td>53.</td><td>Corchuelo and Ulloa (2020) [61]</td><td>Reddish plaques on the lower lip, dark brown pigmentation and aphthous ulcers in the gums, whitish area in tongue</td><td>Lower lips, gums, oral mucosa</td><td>Red and white lesions, vesiculobullous lesions, tongue manifestation</td><td>Mean duration: 8-10 days</td><td>-</td></tr><tr><td>54.</td><td>Dos Santos et al (2020) [10]</td><td>Hypogeusia, white plaque, multiple pinpoint yellowish ulcers in the tongue, nodule in lower lip (1cm)</td><td>Tongue, lower lip, oral mucosa</td><td>Taste alteration, vesiculobullous lesions, tongue manifestation</td><td>Mean duration: 14 days</td><td>Respiratory symptoms and progressive dyspnea on exertion, Fever and diarrhea.</td></tr><tr><td>55.</td><td>Zarch and Hosseinzadeh (2020) [62]</td><td>Vesicles on lower lip mucosa</td><td>Lip mucosa</td><td>Vesiculobullous lesions</td><td>2 days before systemic symptoms</td><td>High fever, Fatigue, and lack of appetite</td></tr><tr><td>56.</td><td>Hjelmeseth J (2020) [63]</td><td>Total ageusia</td><td>Tongue</td><td>Taste alteration</td><td>-</td><td>-</td></tr><tr><td>57.</td><td>Kahraman and Çaşkurlu (2020) [64]</td><td>Erythematous surface (hard palate), few petechiae in the midline and numerous pustular enanthema near the soft palate border</td><td>Palate</td><td>Bleeding disorders, vesiculobullous lesions</td><td>Mean duration: 10 days</td><td>Sore throat Fever, fatigue, severe dry cough, inability to taste or smell,</td></tr><tr><td>58.</td><td>Smith AC et al (2020) [65]</td><td>Loss of taste</td><td>Tongue</td><td>Taste alteration</td><td>Before general symptoms</td><td>Frontal headache, loss of smell, headache, loose stools</td></tr><tr><td>59.</td><td>Maniaci A et al (2020) [66]</td><td>Transient loss of taste</td><td>Tongue</td><td>Taste alteration</td><td>Mean duration: 12 days</td><td>Fever, sore throat, runny nose, presence of erythematous skin lesions on the lower limbs, asthenia</td></tr><tr><td>60.</td><td>Melley LE et al (2020) [67]</td><td>Loss of taste</td><td>Tongue</td><td>Taste alteration</td><td>1 week before systemic presentation</td><td>Shortness of breath, Fatigue, and loss of appetite</td></tr><tr><td>61.</td><td>Riad A et al (2020) [68]</td><td>Painful white patches on the dorsal surface of the tongue and palate, mild tongue pain</td><td>Tongue, palate, oral mucosa</td><td>Tongue manifestation, red and white lesions</td><td>2 weeks before diagnosis</td><td>Sore throat, generalised myalgia, and Fatigue with intermittent fever</td></tr><tr><td>62.</td><td>Putra BE et al (2020) [69]</td><td>Stomatitis aphthous</td><td>Oral mucosa</td><td>Vesiculobullous lesions</td><td>Day 7</td><td>Fever, back pain, myalgia, sore throat, dry cough, rhinorrhea, anosmia</td></tr><tr><td>63.</td><td>Dalipi ZS et al (2021) [70]</td><td>Loss of taste Bullous and erosive erythematous lesions of lips and oral mucosa</td><td>Tongue, lips</td><td>Taste alteration, vesiculobullous lesions, lip lesions</td><td>Loss of taste-2 weeks before diagnosis</td><td>Fever, cough, headache, muscle pain, loss of smell, dark red, purpuric, irregular maculopapular lesions on abdomen</td></tr><tr><td>64.</td><td>Eita AAB et al (2021) [71]</td><td>Dysgeusia and greasy tongue coat</td><td>Tongue</td><td>Taste alteration, tongue manifestation</td><td>Before systemic symptoms</td><td>Sore throat, fever (38°C), nasal congestion, conjunctivitis, nausea, Abdominal pain, diarrhea, fatigue, severe joint pain.</td></tr><tr><td>65.</td><td>Cirillo and Colello (2021) [72]</td><td>Loss of taste</td><td>Tongue</td><td>Taste alteration</td><td>1 week before presentation</td><td>Loss of smell, headache, fatigue, and muscle pain</td></tr><tr><td>66.</td><td>Nejabi MB et al (2021) [73]</td><td>Fissured tongue, white scars and painful erosive ulcer on the Dorsal surface of the tongue</td><td>Tongue</td><td>Tongue manifestation</td><td>After 1 week of general symptoms</td><td>Fever, cough, taste alterations, olfactory dysfunction, and chest tightness</td></tr><tr><td>67.</td><td>Klein H et al (2021) [74]</td><td>Loss of taste</td><td>Tongue</td><td>Taste alteration</td><td>From 4thto 6 weeks</td><td>Fever, dry cough, chest pain, sore throat, diarrhea, nausea, headache, and back pain.</td></tr><tr><td>68.</td><td>Ramires MCCH et al (2021) [75]</td><td>Crusted ulcers on lip vermilion (both upper and lower lips)</td><td>Lips</td><td>Vesiculobullous lesions</td><td>2 weeks after the onset of fever</td><td>Flu-like syndrome; evere and progressive dyspnea (spo2 = 88%)</td></tr><tr><td>69.</td><td>Hocková B et al (2021) [76]</td><td>Oral lesions at the dorsal surface of the tongue (case 1); multiple lesions located on the tongue dorsum and labial mucosa (case 2); lesions on upper and lower lip (case 3)</td><td>Tongue and lips</td><td>Vesiculobullous lesions</td><td>After the diagnosis (all 3 cases)</td><td>Headache, fever, dry cough, and dyspnoea</td></tr><tr><td>70.</td><td>Teixeira IS et al (2021) [77]</td><td>Painful vesiculobulous lip lesions</td><td>Lips</td><td>Vesiculobullous lesions</td><td>After 4 days (case 1); after 10 days (case 2); after 11 days (case 3); after 19 days (case 4)</td><td>Headache, myalgia, and dyspnea</td></tr><tr><td>71.</td><td>EmelyanovaN et al (2021) [78]</td><td>Unusual tongue appearance and burning sensation, intermittent bleeding of gums, severe dryness in the oral cavity and persistent distortion of taste</td><td>Tongue, gums, oral mucosa</td><td>Burning sensation, periodontal changes, taste alteration, tongue manifestation, xerostomia</td><td>Third day (dysgeusia) and fifth day (xerostomia) after systemic symptoms</td><td>Rhinorrhea, coughing and increased body temperature to 38.5°</td></tr><tr><td>72.</td><td>Fathi Y et al (2021) [79]</td><td>Oral pain, ulcerative lesions on oral mucosa, hemorrhagic crusts on lips</td><td>Oral mucosa and lips</td><td>Vesiculobullous lesions, lip lesions,</td><td>3rdday (oral pain)</td><td>Fever, abdominal pain, nausea and occasional vomiting</td></tr><tr><td>73.</td><td>Shenoy P et al (2022) [80]</td><td>Ulcer with irregular borders on the dorsum of the tongue surrounded by a scrapable whitish plaque</td><td>Tongue</td><td>Tongue manifestation, red and white lesions</td><td>Systemic symptoms-3 weeks prior</td><td>Fever, cough, chest tightness</td></tr><tr><td>74.</td><td>Palaia G et al (2022) [81]</td><td>Extensive erosions involving lips, ulcers on the hard palate, blisters and ulcers on the dorsal surface of the tongue cheek mucosa</td><td>Palate, lips, oral mucosa</td><td>Vesiculobullous lesions</td><td>7 days prior to general symptoms (duration of oral lesions-14 days)</td><td>Bilateral cutaneous lesions were also evident on the hands. Low-grade fever</td></tr><tr><td>75.</td><td>Rafałowicz B et al (2022) [82]</td><td>Unilateral aphthous-like lesions on the left side of the hard palate (case 1 & 5); hemorrhagic changes on the palate and cheilitis (case 2); smooth tongue with intensely red-purple mucosa (case 3); angioma type lesion on the right side of the palate (case 4); mycosis of the tongue, extensive lesions on the palate, spontaneous bleeding, cheilitis (case 6)</td><td>Hard palate, tongue; lips</td><td>Vesiculobullous lesions, red and white lesions, fungal infection, tongue manifestation, bleeding disorders</td><td>-</td><td>Fever, malaise, taste disorders, anosmia, and pneumonia (case 1); dyspnea, persistent diarrhea, and vomiting (case 2); loss of smell and taste and fever for 9 days (case 4 & 5)</td></tr></table>
Supplementary table 1: Treatment of oral lesions for COVID-19 patient
<table><tr><td>Author & year</td><td>Treatment given for oral lesions</td><td>Resolution time</td><td>General treatment for COVID-19</td><td>Outcome of COVID-19</td></tr><tr><td>Sinadinos and Shelswell (2020) [6]</td><td>Valaciclovir and topical antiseptics (chlorhexidine and hyaluronic acid)-cases 1 and 3; topical antiseptic mouthwash (case 2)</td><td>10 days (cases 1 and 3), 7 days (case 2)</td><td>-</td><td>All 3 recovered</td></tr><tr><td>Dima M et al (2020) [9]</td><td>Nystatin for the oral candidiasis.</td><td>15 days 21 days (newborn 3)</td><td>Vitamin D, topical cream for erythema (cases 1 and 2); Ampicillin (100 mg/kgc/d), Gentamicin (4 mg/kgc/d), aminophylline (3 0.3mL/d), and Fluconazole iv (6 mg/kgc) (newborn 3)</td><td>All 3 cases recovered</td></tr><tr><td>Dos santos et al (2020) [10]</td><td>Oral nystatin (100,000 IU/mL, 8/8h, for 30 days), chlorhexidine digluconate (0.12%) 1% hydrogen peroxide</td><td>44 days</td><td>Fluconazole 200mg/100mL, Meropeném-1000mg, 8/8hs, Sulfamethoxazole+Trim etropin-400mg +80mg, 1.5 ampule, 8/8hs for 10 days Enoxaparin sodium - 60mg/day.</td><td>Recovered</td></tr><tr><td>Favia G et al (2021) [29]</td><td>Hyaluronic acid gel and chlorhexidine 2% mouthwash, miconazole nitrate twice a day in patients with cytological diagnosis of candidiasis, tranexamic acid for local hemorrhages</td><td>14-21 days</td><td>-</td><td>Good recovery</td></tr><tr><td>Naser Al et al (2021) [43]</td><td>Chlorhexidine 0.2% mouthwash (35%), Nystatin (15%), Mycoheal (8%), Anginovag spray (22%) Fluconazole (13%), Kenalog spray (6%), Amphotericin B (1%)</td><td>7.7 days</td><td>-</td><td>98% responded to treatment</td></tr><tr><td>Soares CD et al (2022) [47]</td><td>Most patients with chronic ulcers were treated with topical corticosteroids and the lesions resolved after 1 to 2 weeks.</td><td>2 weeks</td><td>-</td><td>All recovered</td></tr><tr><td>Brandao TB et al (2020) [56]</td><td>Daily photobiomodulation therapy (PBMT)-10 days; 0.12% chlorhexidine mouthwash</td><td>11-14 days</td><td>Intravenous acyclovir-250 mg/m 2-3 times a day for 10 days.</td><td>Recovered (06 cases); critical condition (cases 2 & 4)</td></tr><tr><td>Tapia ROC et al (2020) [57]</td><td>Topical Mometasone furoate 0.1% in solution after oral hygiene and clorhexidine 0.12% mouthwash</td><td>14 days</td><td>Acetaminophen 500mg every 6 h for 4 days and Fexofenadine 180 mg every 24 h per 10 days (case 1,2); Dexamethasone 8mg (case 2)</td><td>Recovered</td></tr><tr><td>Presas CMC et al (2020) [59]</td><td>Topical Antiseptics, chlorhexidine and hyaluronic acid mouthwash (Cases 1 & 2); prednisolone 30 mg per day (case 3)</td><td>10 days</td><td>Valaciclovir 500 mg every 8 hr for 10 days (case 1); antibiotics, corticosteroids, and lopinavir 200 mg, ritonavir 50 mg, hydroxychloroquine 200 mg) (case 3)</td><td>Recovered</td></tr><tr><td>Rodríguez z MD et al (2022) [60]</td><td>triamcinolone acetonide 0.05% (case 1); ointment containing neomycin, nystatin, and triamcinolone acetonide and chlorhexidine mouthwash (case 2); Nystatin solution rinses (case 3)</td><td>10 days (cases 1 and 2); 15 days (case 3);</td><td>-</td><td>All 3 recovered</td></tr><tr><td>Corchuelo and Ulloa (2020) [61]</td><td>Nystatin oral suspension-3 ml (300,000 I.U) every 6 h; Chlorhexidine gluconate (0.12%)</td><td>14 days</td><td>-</td><td>Recovered</td></tr><tr><td>Kahraman and Çaszkurlu (2020) [64]</td><td>Antibiotherapy</td><td>14 days</td><td>Clarithromycin 500 mg b.i.d. immediately after the diagnosis</td><td>Recovered</td></tr><tr><td>Smith AC et al (2020) [65]</td><td>-</td><td>21days</td><td>Fluticasone, Loratadine, and nasal spray</td><td>Recovered</td></tr><tr><td>Melley LE et al (2020) [67]</td><td>-</td><td>15days</td><td>Hydroxychloroquine 400 mg, azithromycin 500 mg</td><td>Recovered</td></tr><tr><td>Riad A et al (2021) [68]</td><td>Topical antifungal, nystatin (Microstatin) and antibacterial mouthwash, chlorhexidine 0.2%, twice daily (case 1); topical antifungal miconazole (Daktarin Gel) (cases 2 and 3); systemic antifungal fluconazole (Flucoral) (case 3)</td><td>10 days (case 1); 4 days (case 2); 7 days (case 3)</td><td>Azithromycin (Zithromax, levofloxacin (Uniloxam), r ivaroxaban (Xarelto), and lactoferrin (Pravotin) (case 1)</td><td>All 3 recovered</td></tr><tr><td>Putra BE et al (2020) [69]</td><td>Oral hygiene regimen and mouthwash</td><td>21 days</td><td>Azithromycin 500 mg, hydroxychloroquine-400 mg (10 days), oseltamivir-75 mg (10 days), vitamin C-1000 mg (14 days), vitamin D-5000 IU (14 days) On day six, the lesions lessened</td><td>Recovered</td></tr><tr><td>Dalipi ZS et al (2021) [70]</td><td>Antibiotic therapy with penicillin and anticoagulant therapy with Fraxiparine solution for injection 0.4 mL, antiseptic mouthwash (0.2% chlorhexidine solution mouthwash twice per day for 14 days</td><td>21 days</td><td>Systemic corticosteroids, vitamins (C, B complex), and locally applied tablets (panthenol-calcium with pantothenic acid)</td><td>Recovered</td></tr><tr><td>Eita AAB (2021) [71]</td><td>0.12% chlorhexidine mouth rinse and nystatin oral drops 100.000 IU/ml 2-3 times daily). Daily vitamin C (1000 mg) and zinc (50 mg)</td><td>28 days</td><td>Azithromycin 500 mg/day, paracetamol 500 mg 2 tablets ×3 times daily, zinc (50 mg), and vitamin C (1000 mg) supplements</td><td>Recovered</td></tr><tr><td>Nejabi MB et al (2021) [73]</td><td>Intravenous Acyclovir 5 mg/kg for 7 days; PMBT-10 days; Chlorhexidine 0.12%, H2O2-1%)</td><td>14 days</td><td>Azithromycin 500 mg daily for one week and ceftriaxone 1g twice a day for 3 days. antifungal agents (fluconazole 200 mg tablets for 1 week)</td><td>Recovered</td></tr><tr><td>Ramires MCCH et al (2021) [75]</td><td>Photodynamic therapy for 2 days, methylene blue at 0.01% was applied over all lesions. After 5 min (time pre-irradiation), the laser device Therapy EC® was used at 660 nm. A total of 30 points were distributed throughout the affected areas: 20 points on the upper and 10 on the lower lip.</td><td>Healed after 4 days of therapy</td><td>-</td><td>Recovered</td></tr><tr><td>Teixeira IS et al (2021) [77]</td><td>For PBMT, a laser device Laser DUO® was used at 660 nm, on contact mode, point by point, with 100 mW, 33 J/cm2, 0.5 J, and 5 s per point. Then, aPDT technique was performed, with methylene blue at 0.01% applied over all lesions and after 3 min, the same laser parameters were used.</td><td>Marked improvement after 72 hrs</td><td>Azithromycin and ceftriaxone. dexamethasone</td><td>Recovered</td></tr><tr><td>Fathi Y et al (2021) [79]</td><td>Removal of dental plaque and chlorhexidine mouthwash (twice a day). Valacyclovir for 5 days</td><td>Resolved after 4 days</td><td>Metronidazole, ceftriaxone, meropenem, ribavirin and hydroxychloroquine were administered and supplemental oxygen was given</td><td>-</td></tr><tr><td>Shenoy P et al (2022) [80]</td><td>Tab Fluconazole, Clotrimazole mouth paint, Chlorhexidine 0.12% mouth rinse.</td><td></td><td>Anticoagulants, corticosteroids and multivitamins. Medications include Inj.Enoxararin, Inj. Pantop, Tab.Shelcal HD, Neb.Budecort, Inj.Predmet 40 mg, Cap.Meganeuron</td><td>Succumbed to disease.</td></tr><tr><td>Rafałowicz B et al (2022) [82]</td><td>Semiconductor laser therapy with the Smart bio stimulation function-five treatments at 3-day intervals (case 1); aliva-stimulating tablets SST (Sinclair Pharmaceuticals), Kserostemin (artificial saliva) (Aflofarm), and mouthwash with chlorhexidine three times a day for a period of 14 days (case 3); antifungal Nystatin (Teva) antibiotic was administered at a dose of 100,000 IU/mL four times a day along with vitamin A + E + F (Gorvita) ointment on the lips (case 6)</td><td>192 days (case 1); 2 weeks (case 3); 60 days (case 5)</td><td>-</td><td>-</td></tr></table>
Generating HTML Viewer...
References
94 Cites in Article
(2022). WHO Coronavirus Disease (COVID-19) Dashboard.
Nickolas Kipshidze,George Dangas,Christopher White,Nodar Kipshidze,Fakiha Siddiqui,Christopher Lattimer,Charles Carter,Jawed Fareed (2020). Viral Coagulopathy in Patients With COVID-19: Treatment and Care.
J Amorim Dos Santos,Agc Normando,R Carvalho Da Silva,A Acevedo,G De Luca Canto,N Sugaya,A Santos-Silva,E Guerra (2021). Oral Manifestations in Patients with COVID-19: A Living Systematic Review.
A Nuno‐gonzalez,P Martin‐carrillo,K Magaletsky,M Martin Rios,C Herranz Mañas,J Artigas Almazan,G García Casasola,E Perez Castro,A Gallego Arenas,A Mayor Ibarguren,M Feito Rodríguez,B Lozano Masdemont,M Beato,E Ruiz Bravo,P Oliver,M Montero Vega,P Herranz Pinto (2021). Prevalence of mucocutaneous manifestations in 666 patients with COVID‐19 in a field hospital in Spain: oral and palmoplantar findings.
Angela Sinadinos,Jonathan Shelswell (2020). Oral ulceration and blistering in patients with COVID-19.
Fac Garcia De Sousa,T Paradella (2021). Considerations on oral manifestations of COVID-19.
Abanoub Riad,Islam Kassem,Barbora Hockova,Mai Badrah,Miloslav Klugar (2020). Tongue ulcers associated with SARS‐CoV‐2 infection: A case series.
Mirabela Dima,Ileana Enatescu,Marius Craina,Izabella Petre,Emil Iacob,Daniela Iacob (2020). First neonates with severe acute respiratory syndrome coronavirus 2 infection in Romania.
J Amorim Dos Santos,Costa Normando,A,Carvalho Da Silva,R (2020). Oral mucosal lesions in a COVID-19 patient: New signs or secondary manifestations?.
David Moher,Alessandro Liberati,Jennifer Tetzlaff,Douglas Altman (2009). Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement.
Al-Zaidi Hmh,H Badr (2020). Incidence and recovery of smell and taste dysfunction in COVID-19 positive patients.
Ameen Biadsee,Ameer Biadsee,Firas Kassem,Or Dagan,Shchada Masarwa,Zeev Ormianer (2020). Olfactory and Oral Manifestations of COVID‐19: Sex‐Related Symptoms—A Potential Pathway to Early Diagnosis.
N Bodnia,T Katzenstein (2020). Acute loss of smell and taste among patients with symptoms compatible with COVID-19.
L Chen,J Zhao,J Peng,X Li,X Deng,Z Geng (2020). Detection of 2019-ncov in saliva and characterization of oral symptoms in covid-19 patients.
V Dell'era,F Farri,G Garzaro,M Gatto,P Valletti,M Garzaro (2020). Smell and taste disorders during COVID-19 outbreak: Cross-sectional study on 355 patients.
Lakshit Kumar,Namrata Kahlon,Avani Jain,Jasleen Kaur,Mitasha Singh,A Pandey (2021). Loss of smell and taste in COVID-19 infection in adolescents.
Jerome Lechien,Carlos Chiesa-Estomba,Daniele De Siati,Mihaela Horoi,Serge Le Bon,Alexandra Rodriguez,Didier Dequanter,Serge Blecic,Fahd El Afia,Lea Distinguin,Younes Chekkoury-Idrissi,Stéphane Hans,Irene Delgado,Christian Calvo-Henriquez,Philippe Lavigne,Chiara Falanga,Maria Barillari,Giovanni Cammaroto,Mohamad Khalife,Pierre Leich,Christel Souchay,Camelia Rossi,Fabrice Journe,Julien Hsieh,Myriam Edjlali,Robert Carlier,Laurence Ris,Andrea Lovato,Cosimo De Filippis,Frederique Coppee,Nicolas Fakhry,Tareck Ayad,Sven Saussez (2020). Olfactory and gustatory dysfunctions as a clinical presentation of mild-to-moderate forms of the coronavirus disease (COVID-19): a multicenter European study.
Alberto Paderno,Alberto Schreiber,Alberto Grammatica,Elena Raffetti,Michele Tomasoni,Tommaso Gualtieri,Stefano Taboni,Silvia Zorzi,Davide Lombardi,Alberto Deganello,Luca Redaelli De Zinis,Roberto Maroldi,Davide Mattavelli (2020). Smell and taste alterations in COVID‐19: a cross‐sectional analysis of different cohorts.
P Rizzo,D Borsetto,C Fabbris,G Spinato,D Frezza,A Menegaldo (2020). Evolution of Altered Sense of Smell or Taste in Patients With Mildly Symptomatic COVID-19.
Carol Yan,Farhoud Faraji,Divya Prajapati,Christine Boone,Adam Deconde (2020). Association of chemosensory dysfunction and COVID‐19 in patients presenting with influenza‐like symptoms.
Bruna Sinjari,Damiano D’ardes,Manlio Santilli,Imena Rexhepi,Gianmaria D’addazio,Piero Di Carlo,Piero Chiacchiaretta,Sergio Caputi,Francesco Cipollone (2020). SARS-CoV-2 and Oral Manifestation: An Observational, Human Study.
Andrea Giacomelli,Laura Pezzati,Federico Conti,Dario Bernacchia,Matteo Siano,Letizia Oreni,Stefano Rusconi,Cristina Gervasoni,Anna Ridolfo,Giuliano Rizzardini,Spinello Antinori,Massimo Galli (2020). Self-reported Olfactory and Taste Disorders in Patients With Severe Acute Respiratory Coronavirus 2 Infection: A Cross-sectional Study.
Hélène Mascitti,Benjamin Bonsang,Aurélien Dinh,Florence Assan,Véronique Perronne,Thibault Leblanc,Clara Duran,Frédérique Bouchand,Morgan Matt,Aurélie Le Gal,Julia N’guyen Van Thanh,Aymeric Lanore,Louis Jacob,Nicolas Kiavue,Soline Siméon,Simon Bessis,Pierre De Truchis,Stéphanie Landowski,Benjamin Davido,Frédérique Moreau,Marie-Anne Rameix-Welti,Elyanne Gault,Jean-Louis Gaillard,Anne-Laure Roux,Valérie Sivadon-Tardy,Elsa Salomon,Faten El Sayed,Robert Carlier,Jean-François Emile,Christian Perronne,Isabelle Bourgault-Villada (2020). Clinical Cutaneous Features of Patients Infected With SARS-CoV-2 Hospitalized for Pneumonia: A Cross-sectional Study.
Mohammadreza Salehi,Kazem Ahmadikia,Shahram Mahmoudi,Saeed Kalantari,Saeidreza Jamalimoghadamsiahkali,Alireza Izadi,Mohammad Kord,Seyed Dehghan Manshadi,Arash Seifi,Fereshteh Ghiasvand,Nasim Khajavirad,Saeedeh Ebrahimi,Amirhossein Koohfar,Teun Boekhout,Sadegh Khodavaisy (2020). Oropharyngeal candidiasis in hospitalised COVID‐19 patients from Iran: Species identification and antifungal susceptibility pattern.
Ozge Askin,Rozerin Altunkalem,Dursun Altinisik,Tugba Uzuncakmak,Umit Tursen,Zekayi Kutlubay (2020). Cutaneous manifestations in hospitalized patients diagnosed as <scp>COVID</scp> ‐19.
Joseph Katz,Sijia Yue (2020). Increased odds ratio for COVID‐19 in patients with recurrent aphthous stomatitis.
Paolo Fantozzi,Emanuele Pampena,Domenico Di Vanna,Eugenia Pellegrino,Daniele Corbi,Stefano Mammucari,Federica Alessi,Riccardo Pampena,Giuliano Bertazzoni,Salvatore Minisola,Claudio Mastroianni,Antonella Polimeni,Umberto Romeo,Alessandro Villa (2020). Xerostomia, gustatory and olfactory dysfunctions in patients with COVID-19.
Gianfranco Favia,Angela Tempesta,Giuseppe Barile,Nicola Brienza,Saverio Capodiferro,Maria Vestito,Lucilla Crudele,Vito Procacci,Giuseppe Ingravallo,Eugenio Maiorano,Luisa Limongelli (2021). Covid-19 Symptomatic Patients with Oral Lesions: Clinical and Histopathological Study on 123 Cases of the University Hospital Policlinic of Bari with a Purpose of a New Classification.
Steven Halepas,Kevin Lee,Aaron Myers,Richard Yoon,Wendy Chung,Scott Peters (2021). Oral manifestations of COVID-2019–related multisystem inflammatory syndrome in children: a review of 47 pediatric patients.
Sergey Rekhtman,Rachel Tannenbaum,Andrew Strunk,Morgan Birabaharan,Shari Wright,Nicole Grbic,Ashna Joseph,Stephanie Lin,Aaron Zhang,Eric Lee,Erika Rivera,Michael Qiu,John Chelico,Amit Garg (2021). Eruptions and related clinical course among 296 hospitalized adults with confirmed COVID-19.
Nadya Marouf,Wenji Cai,Khalid Said,Hanin Daas,Hanan Diab,Venkateswara Chinta,Ali Hssain,Belinda Nicolau,Mariano Sanz,Faleh Tamimi (2021). Association between periodontitis and severity of COVID‐19 infection: A case–control study.
T Subramaniam,M Nikalje,S Jadhav (2021). Oral manifestations among COVID-19: An observational study of 713 patients.
Nermeen Abubakr,Zeinab Salem,Amany Kamel (2021). Oral manifestations in mild-to-moderate cases of COVID-19 viral infection in the adult population.
Jia Song,Yi-Ke Deng,Hai Wang,Zhi-Chao Wang,Bo Liao,Jin Ma,Chao He,Li Pan,Yang Liu,Isam Alobid,De-Yun Wang,Ming Zeng,Joaquim Mullol,Zheng Liu (2021). Self-reported Taste and Smell Disorders in Patients with COVID-19: Distinct Features in China.
E Bardellini,Mp. Bondioni,F Amadori,F Veneri,V Lougaris,A Meini,A Plebani,A Majorana (2021). Non-specific oral and cutaneous manifestations of Coronavirus Disease 2019 in children.
E Gherlone,E Polizzi,G Tetè,R De Lorenzo,C Magnaghi,P Rovere Querini,F Ciceri (2021). Frequent and Persistent Salivary Gland Ectasia and Oral Disease After COVID-19.
El Kady,D Gomaa,E Abdella,W Hussien,R,Abd Elaziz,R Khater,Aga (2021). Oral manifestations of COVID-19 patients: An online survey ofthe Egyptian population.
Z Natto,M Afeef,D Khalil,D Kutubaldin,M Dehaithem,A Alzahrani (2021). Characteristics of Oral Manifestations in Symptomatic Non-Hospitalized COVID-19 Patients: A Cross-Sectional Study on a Sample of the Saudi Population.
Walid Elamrousy,Mohamed Nassar,Dalia Issa (2021). Prevalence of Oral Lesions in COVID-19 Egyptian Patients.
D Bulut,N Turker,S Serin,G Ustaoglu (2021). The Effect of Severe Acute Respiratory Syndrome Coronavirus 2 on the Health of Oral Tissue: A Survey-Based Study.
Alyaa Naser,Marwa Al-Sarraj,Ziad Deleme (2021). Oral and Maxillofacial Lesions in COVID 19 Infection from Mosul Hospital in Iraq: Epidemiological Study and Approach to Classification and Treatment.
Alka Muthyam,M Reddy,Suhas Kulkarni,Adepu Srilatha,Kommuri Sahithi,Dantala Satyanarayana (2022). Oral manifestations in COVID-19 patients: An observational study.
A Ganesan,S Kumar,A Kaur (2022). Oral Manifestations of COVID-19 Infection: An Analytical Cross-Sectional Study.
Maha El Tantawi,Heba Sabbagh,Nada Alkhateeb,Maryam Quritum,Joud Abourdan,Nafeesa Qureshi,Shabnum Qureshi,Ahmed Hamoud,Nada Mahmoud,Ruba Odeh,Nuraldeen Al-Khanati,Rawiah Jaber,Abdulrahman Balkhoyor,Mohammed Shabi,Morenike Folayan,Noha Gomaa,Raqiya Al_Nahdi,Nawal Mahmoud,Hanane El Wazziki,Manal Alnaas,Bahia Samodien,Rawa Mahmoud,Nour Abu Assab,Sherin Saad,Sondos Al-Hachim,Ali Alshaikh,Wafaa Abdelaziz (2022). Oral manifestations in young adults infected with COVID-19 and impact of smoking: a multi-country cross-sectional study.
Ciro Soares,Lucas Souza,Maria De Carvalho,Hélder Pontes,Adalberto Mosqueda-Taylor,Juan Hernandez-Guerrero,Sanderson Do Nascimento Medeiros,Alexandre De Oliveira Sales,Fábio Alves,Clóvis Lopes Pinto,Oslei De Almeida (2022). Oral Manifestations of Coronavirus Disease 2019 (COVID-19).
G Tuter,M Yerebakan,B Celik,G Kara (2022). Oral manifestations in SARS-CoV-2 infection.
G Schwabhttp,M Palmierib,R Zerbinatia,Djs Sarmento,T Reisb,K Ortega (2022). Lack of direct association between oral mucosal lesions and SARS-CoV-2 in a cohort of patients hospitalised with COVID-19.
Jitendra Chawla,Satvinder Bakshi,Vinoth Kalidoss,Shikha Yadav,Swapnika Polineni,Cheranjeevi Jayam (2022). Oral manifestations associated with COVID-19 disease: An observational cross sectional study.
Nada Binmadi,Suad Aljohani,Maha Alsharif,Soulafa Almazrooa,Amal Sindi (2022). Oral Manifestations of COVID-19: A Cross-Sectional Study of Their Prevalence and Association with Disease Severity.
Paula De,F Eduardo,L Bezinelli,M Gobbi,L Bergamin,Dlc De Carvalho,L Corrêa (2022). Oral lesions and saliva alterations of COVID-19 patients in an intensive care unit: A retrospective study.
M Villarroel-Dorrego,L Chacón,R Rosas,V Barrios,Y Pernía,H Vélez (2022). Hallazgos bucales en pacientes COVID-19.
Soheila Manifar,Maryam Koopaie,Ali Karimi Farani,Mansour Davoudi,Sajjad Kolahdouz (2022). Assessment of Oral Manifestations and Oral Health in Hospitalized Patients with COVID-19: Machine Learning and Statistical Analysis.
Ruchi Bhuyan,Sanat Bhuyan,Jatindra Mohanty,Nihar Panda,Varsha Bhuyan,Priyadarshini Ojha,Pallavi Priyadarshini,Gangadhar Sahoo (2022). A Preliminary Survey on the Oral Manifestation of COVID-19 in the First and Second Waves in Bhubaneswar, City of Odisha, India.
Thaís Brandão,Luiz Gueiros,Thayanara Melo,Ana Prado-Ribeiro,Ana Nesrallah,Gladys Prado,Alan Santos-Silva,Cesar Migliorati (2020). Oral lesions in patients with SARS-CoV-2 infection: could the oral cavity be a target organ?.
Roc Tapia,Ajp Labrador,D Guimaraes,Lhm Valdez (2020). Oral mucosal lesions in patients with SARS-CoV-2 infection. Report of four cases. Are they a true sign of COVID-19 disease?.
Luigi Vaira,Giovanna Deiana,Alessandro Fois,Pietro Pirina,Giordano Madeddu,Andrea De Vito,Sergio Babudieri,Marzia Petrocelli,Antonello Serra,Francesco Bussu,Enrica Ligas,Giovanni Salzano,Giacomo De Riu (2020). Objective evaluation of anosmia and ageusia in <scp>COVID</scp>‐19 patients: Single‐center experience on 72 cases.
M Rodríguez,J Romera,M Villarroel (2020). Oral manifestations associated with COVID-19.
Jairo Corchuelo,Francisco Ulloa (2020). Oral manifestations in a patient with a history of asymptomatic COVID-19: Case report.
R Zarch,P Hosseinzadeh (2021). COVID-19 from the perspective of dentists: A case report and brief review of more than 170 cases.
Jøran Hjelmesæth,Dagfinn Skaare (2020). Covid-19 med nedsatt lukte- og smakssans som eneste symptom.
F Kahraman,H Çaşkurlu (2020). Mucosal involvement in a COVID-19-positive patient: a case report.
L Smith,C Hodges,Molly Pratt,Ian Porter (2020). Case Report: COVID-19 Patient With Chief Complaint of Anosmia and Ageusia; a Unique Perspective on Atypical Symptomatology and Management in the Military.
A Maniaci,G Iannella,C Vicini,P Pavone,G Nunnari,R Falsaperla (2020). A Case of COVID-19 with Late-Onset Rash and Transient Loss of Taste and Smell in a 15-Year-Old Boy.
Lauren Melley,Eli Bress,Erik Polan (2020). Hypogeusia as the initial presenting symptom of COVID-19.
Abanoub Riad,Akram Gad,Barbora Hockova,Miloslav Klugar (2020). Oral candidiasis in non‐severe COVID‐19 patients: call for antibiotic stewardship.
Bayushi Putra,Suko Adiarto,Santi Dewayanti,Dafsah Juzar (2020). Viral exanthem with “Spins and needles sensation” on extremities of a COVID-19 patient: A self-reported case from an Indonesian medical frontliner.
Zana Dalipi,Fatmir Dragidella,Donika Dragidella (2021). Oral Manifestations of Exudative Erythema Multiforme in a Patient with COVID‐19.
Aab Eita,Parosmia (2021). Parosmia, Dysgeusia, and Tongue Features Changes in a Patient with Post‐Acute COVID‐19 Syndrome.
Nicola Cirillo,Giuseppe Colella (2021). Self-reported smell and taste alteration as the sole clinical manifestation of SARS-CoV-2 infection.
Mohammad Nejabi,Noor Noor,Nahid Raufi,Mohammad Essar,Ehsanullah Ehsan,Jaffer Shah,Asghar Shah,Arash Nemat (2021). Tongue ulcer in a patient with COVID-19: a case presentation.
Hadar Klein,Noam Karni,Sarah Israel,Menachem Gross,Mordechai Muszkat,Masha Niv (2021). Reversible Taste Loss in a COVID-19 Patient With Preexisting Chronic Smell Impairment.
M Ramires,M Mattia,R Tateno,Palma Campos,L (2021). A Combination of Phototherapy Modalities for Extensive Lip Lesions in a Patient with SARS-CoV-2 Infection.
Barbora Hocková,Abanoub Riad,Jozef Valky,Zuzana Šulajová,Adam Stebel,Rastislav Slávik,Zuzana Bečková,Andrea Pokorná,Jitka Klugarová,Miloslav Klugar (2021). Oral Complications of ICU Patients with COVID-19: Case-Series and Review of Two Hundred Ten Cases.
Inessa Teixeira,Fidel Leal,Ricardo Tateno,Luiz Palma,Luana Campos (2021). Photobiomodulation therapy and antimicrobial photodynamic therapy for orofacial lesions in patients with COVID-19: A case series.
N Emelyanova,G Isayeva,I Komir,A Shalimova,O Buriakovska,M Vovchenko (2021). Changes in the Oral Cavity of a Patient after Suffering from Coronavirus Infection COVID-19: A Clinical Case.
Yaser Fathi,Elaheh Hoseini,Reza Mottaghi (2021). Erythema multiform-like Lesions in a Patient Infected With SARS-CoV-2: A Case Report.
Prashanth Shenoy,P Archana,Laxmikanth Chatra,K Veena,Rachana Prabhu,Prathima Shetty (2022). Evaluation of Patterns of Mandibular Bone Invasion in CBCT of Patients with Oral Squamous Cell Carcinoma.
Gaspare Palaia,Elena Pernice,Daniele Pergolini,Alessandra Impellizzeri,Guido Migliau,Gianluca Gambarini,Umberto Romeo,Antonella Polimeni (2022). Erythema Multiforme as Early Manifestation of COVID-19: A Case Report.
Barbara Rafałowicz,Leopold Wagner,Juliusz Rafałowicz (2022). Long COVID Oral Cavity Symptoms Based on Selected Clinical Cases.
Anne-Gaëlle Chaux-Bodard,Sophie Deneuve,Aline Desoutter (2020). Oral manifestation of Covid-19 as an inaugural symptom?.
Russell Viner,Elizabeth Whittaker (2020). Kawasaki-like disease: emerging complication during the COVID-19 pandemic.
C Hopkins,N Kumar (2022). Loss of sense of smell as marker of COVID-19 infection.
N Cirillo (2020). Taste alteration in COVID-19: a rapid review with data synthesis reveals significant geographical differences.
A Lee (2020). Wuhan novel coronavirus (COVID-19): why global control is challenging?.
B Jafek,B Murrow,R Michaels (2002). Biopsies of human olfactory epithelium.
J Amorim Dos Santos,Agc Normando,Carvalho Da Silva,R (2021). Oral Manifestations in Patients with COVID-19: A 6-Month Update.
Jane Tong,Amanda Wong,Daniel Zhu,Judd Fastenberg,Tristan Tham (2020). The Prevalence of Olfactory and Gustatory Dysfunction in COVID‐19 Patients: A Systematic Review and Meta‐analysis.
Kacper Nijakowski,Sylvia Wyzga,Nisha Singh,Filip Podgórski,Anna Surdacka (2022). Oral Manifestations in SARS-CoV-2 Positive Patients: A Systematic Review.
Alessandra Majorana,Elena Bardellini,Pierangela Flocchini,Francesca Amadori,Giulio Conti,Guglielmo Campus (2010). Oral mucosal lesions in children from 0 to 12 years old: ten years' experience.
Esam Halboub,Sadeq Al-Maweri,Rawan Alanazi,Nashwan Qaid,Saleem Abdulrab (2020). Orofacial manifestations of COVID-19: a brief review of the published literature.
Giulia Orilisi,Marco Mascitti,Lucrezia Togni,Riccardo Monterubbianesi,Vincenzo Tosco,Flavia Vitiello,Andrea Santarelli,Angelo Putignano,Giovanna Orsini (2021). Oral Manifestations of COVID-19 in Hospitalized Patients: A Systematic Review.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Dr. Ankita Gupta. 2026. \u201cEstimating the Prevalence of Oral Manifestations in Covid-19 Patients: A Systematic Review\u201d. Global Journal of Medical Research - J: Dentistry & Otolaryngology GJMR-J Volume 23 (GJMR Volume 23 Issue J2).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.