Introduction-What could humanity have done better in fighting the SARS-CoV-2 pandemic? From a financial and scientific point of view, it has done many things right, but a crucial ethical question has remained rather unexamined. In this paper, I argue that controlled human infection studies (HIS) would have been ethically justifiable and the right way forward in developing a vaccine against Covid-19. The phase 2/3 trials of the vaccines from AstraZeneca, Pfizer/Biontech and Moderna took between 112 and 196 days. Human challenge trials would have taken much less time, about 30 days. In retrospect, these three vaccines could have been launched 82 to 166 days earlier than they actually were. If this had happened, hundreds of thousands of deaths and millions of hospitalisations worldwide could have been avoided due to the cumulative effect. In terms of preparatory measures for the next pandemic, the ethical discussion on HIS is of utmost relevance for the well-being of future generations.
### INTRODUCTION
What could humanity have done better in fighting the SARS-CoV-2 pandemic? From a financial and scientific point of view, it has done many things right, but a crucial ethical question has remained rather unexamined. In this paper, I argue that controlled human infection studies (HIS)[^1] would have been ethically justifiable and the right way forward in developing a vaccine against Covid-19. The phase 2/3 trials of the vaccines from AstraZeneca, Pfizer/Biontech and Moderna took between 112 and 196 days. Human challenge trials would have taken much less time, about 30 days. In retrospect, these three vaccines could have been launched 82 to 166 days earlier than they actually were. If this had happened, hundreds of thousands of deaths and millions of hospitalisations worldwide could have been avoided due to the cumulative effect. In terms of preparatory measures for the next pandemic, the ethical discussion on HIS is of utmost relevance for the well-being of future generations.
Phase 1 study: the first use of vaccines on humans In order to understand the ethical issues surrounding HIS, it is necessary to understand how vaccines are tested on humans. Once vaccine
developers have tested a certain agent against an infectious disease in animals ('preclinical studies') and these creatures have been successfully immunised, the next step is the first application in humans. The immune system of humans is so fundamentally different from that of even the animals most similar to us, that the approval of an investigational vaccine solely on the basis of animal experiments is not an option. Depending on the number of test persons and the exact question, a distinction is usually made between three phases (and occasionally a phase 4 after approval) in human application. For human volunteers, phase 1 (first in human), poses the greatest risk.
How would one have proceeded in a human infection study?
Regulatory authorities need data on the efficacy of vaccine candidates beyond the results of the phase 1 trial for their decisions. Let us assume that HIS were ethically permissible. In such a scenario, the process to SARS-CoV-2 vaccine could have consisted as described in chart 1.
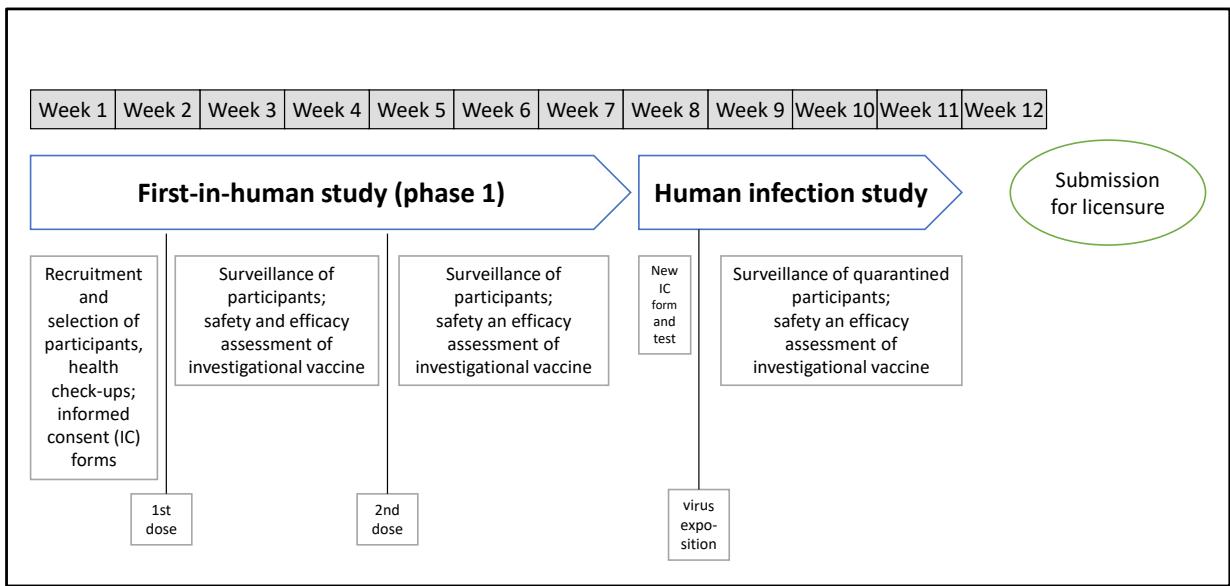 Chart 1: Process to SARS-CoV-2 vaccine licensure, including a phase 1 and a human infection study (hypothetical)
How approved vaccines against Covid-19 were actually tested in the field?
Time is the decisive factor in protecting future generations from new pandemics. As HIS studies can replace phase 2/3 studies (but not phase 1 studies), it is important to know exactly how long the phase 2/3 studies lasted. The following table shows the relevant data for the first vaccines approved in the EU and the USA, i.e. those from Pfizer Biontech, Moderna and AstraZeneca, as well as for the CureVac vaccine CVnCoV.
Table 1: Duration of phase 2/3 studies in the process to SARS-CoV-2 vaccine licensure (de facto)
<table><tr><td></td><td>Duration of the phase 2/3 study</td><td>Participants</td><td>Infected persons in the active agent group</td><td>Infected persons in the placebo group</td><td>Effectiveness of the vaccine</td></tr><tr><td>BNT162b2
(Pfizer/
BioNTech)</td><td>115 days
27.07.2020 - 18.11.2020</td><td>43.448</td><td>8</td><td>162</td><td>95%</td></tr><tr><td>mRNA-1273
(Moderna)</td><td>112 days
27.07.2020 - 15.11.2020</td><td>30.420</td><td>11</td><td>185</td><td>94,1%</td></tr><tr><td>ChAdOx
(AstraZeneca)</td><td>196 days
23.04.2020 - 4.11.2020</td><td>23.848</td><td colspan="3">The values of these columns are not comparable, as the phase 3 study was divided into two sub-studies, and the summation of the values was strongly criticised within the scientific community.</td></tr><tr><td>CVnCOV6
(CureVac)</td><td>123 days
11.12.2020 - 12.04.2021</td><td>39.680</td><td>83</td><td>145</td><td>48,2%</td></tr></table>
The phase 3 trials of the first vaccines approved in the EU and the USA took between 112 (Moderna) and 196 (AstraZeneca) days, depending on the vaccine. Human infection studies would have taken significantly less time, about 30 days. In retrospect, therefore, the vaccines that were gradually approved could have been on the market 82 to 166 days earlier than they actually were. Indeed, a large number of deaths and hospitalisations could have been avoided if HIS had been used instead of the usual phase 2/3 trials.
Without HIS, the following adversities occur. The stronger the protective measures (i.e. lock-down), the more months are lost. The crucial question, how many infected people must there be before the regulatory authorities are satisfied is an opaque process. This is where vaccine manufacturers and regulatory authorities have to come to an agreement. Ultimately, these are negotiation processes that are hidden from the public. Different actors - the government, the regulatory authorities, the public - have different ideas, which can lead to tensions. An example from Turkey: "The Turkish researchers, speaking alongside Health Minister FahrettinKoca, said 26 of the 29 people who were infected during the trial were given placebos, adding the trial would continue until 40 people become infected.
(...)" Health Minister Koca said Ankara would now – this was on 24 Dec 2020 –approve the vaccine, although "researchers initially planned to announce the results after 40 people were infected." 7 Now, how decides here at what point the vaccine candidate is safe? Incidentally, the vaccine in question was China's Sinovac vaccine, and the vaccine effectiveness of $91.25\%$ calculated on the basis of the small number of cases, which the Turkish health minister communicated to the public, is doubtful. However, this is also true for the decimal places in the vaccine efficacy calculated by e.g. Pfizer Biontech or Moderna from the low infection cases of their respective studies. Waiting to see when 10, 20, 30, 40, 60, 80 or 100 vaccinated people will 'accidentally' be infected is gruelling when the whole world is waiting for a vaccine. And the small numbers lead to unsatisfying data about vaccine effectiveness.
#### Ethical requirements for HIS in general
Uncontrolled pandemics are among the existential risks for future generations. The potential of HIS to reduce this risk is undisputed and this potential was once again highlighted by the WHO in 2020 during the first wave of the Corona pandemic: "Well designed human challenge studies provide one of the most efficient and scientifically powerful means for testing vaccines, especially because animal models are not adequately generalizable to humans. Challenge studies could thus be associated with substantial public health benefit in so far as they (a) accelerate vaccine development, (b) increase the likelihood that the most effective (candidate) vaccines will ultimately become available, (c) validate tests of immunity, and (d) improve knowledge regarding SARS-CoV-2 infection and transmission." 8
Can the worst effects of pandemics be avoided in general, i.e. also in the future, if humanity relies on HIS? That depends on many virological-medical factors. From an ethical point of view, one cannot come to a simple yes or no conclusion in respect to HIS. The following factors and framework conditions play a role in determining the answer:
#### 1. Nature of the pathogen - benefit of a vaccine
HIS have helped in the early research with smallpox, yellow fever and malaria that eventually changed the course of global public health. And HIS have recently helped, for example, to accelerate the development of vaccines against typhoid and cholera.[9] Whether vaccines help in the long term depends also on the ability of a virus to generate immune escape variants. The ability to mutate varies from the genetically stable smallpox virus at one end of the scale to the very rapidly mutating influenza viruses at the other. SARS-CoV-2 is somewhat in the middle.
#### Variant-adapted vaccines
### BA.2.12.1 and BA.4/5 are now increasing in prevalence
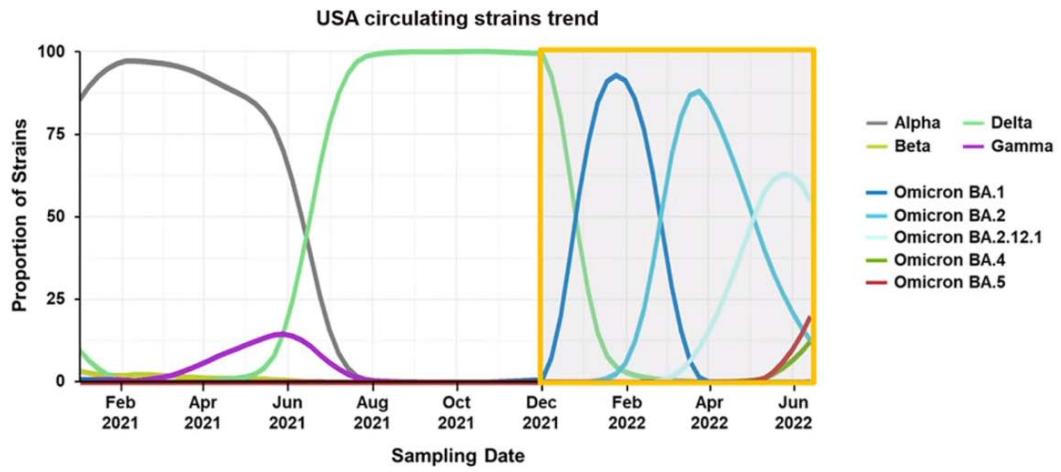 Chart 2: Circulating strain trends in the USA GISAID Initiative database: https://www.gisaid.org/ (accessed May 31, 2022).
This means vaccination is a useful but not a perfect remedy. This is the case for most infectious diseases. All experts agree: If mankind had failed to develop vaccines against SARS-CoV-2, the death toll would have been much higher. Georg Schmidt, chairman of the Working Group of Medical Ethics Committees in Germany, is of the opinion that one can consider conducting a HIS only if the risk is manageable and a social catastrophe is imminent. This was, according to Mr. Schmidt, not the case in the Covid-19 situation.[^10] Not a catastrophe? Peer-reviewed global estimates of excess deaths indicate 18.2 million people may have died because of the COVID-19 pandemic until 31 Dec 2021.[^11] The global Corona pandemic was a catastrophe, especially for the most vulnerable members of society. In addition to the millions of deaths and long-haul Covid cases we should not forget all the liberty rights restrictions due to lockdown measures, and the lost livelihoods due to economic depression. Undoubtedly, the sheer size of a catastrophe matters. The more a pathogen poses an existential risk to humanity or its potential, the more HIS are justified. In a scenario in which a new pandemic would put not millions, but billions of people at risk, many more ethicists would throw overboard their concerns.
#### 2. Benefits of HIS for vaccine research with regard to vulnerable subgroups
The best possible design of vaccine trials, including how many sequential trials there should be, varies from pandemic to pandemic. However, the tendency is that HIS can generate extremely important data for vaccine development. In the case of the SARS-CoV-2 pandemic, the objection to HIS was that the data obtained in young, healthy volunteers could not be transferred to the vulnerable group of people over 70. The WHO disagrees: "Prioritizing the safety of participants is standard in modern challenge studies and acceptable in so far as studies with low-risk participants nevertheless produce useful results (for example, that would help to identify the most promising vaccine candidates or validate correlates of protection)."12
#### 3. Health risks for the test persons
The lower the health risks associated with HIS, the more likely they are to be ethically permissible. A specific assessment is always required. In the case of SARS-CoV-2, there were still many uncertainties in the initial phase regarding the pathogenicity or lethality of the virus. There were also no effective drugs or therapies against SARS-CoV-2 in 2020-2021. Unlike, for example, malaria, influenza, typhoid and cholera - diseases for which controlled infection studies have been and are being conducted. The WHO states: "Challenge studies have a long history, including early research with smallpox, yellow fever and malaria that changed the course of global public health. In the last 50 years, challenge studies have been performed safely in tens of thousands of consenting adult volunteers under the oversight of research ethics committees. These studies have recently helped, for example, to accelerate the development of vaccines against typhoid and cholera, and to determine correlates of immune protection against influenza." ${}^{13}$
Generally, the risks to the subjects are reduced when there is excellent diagnostics so that action can be taken within a sufficiently long incubation period before the disease becomes life-threatening. This was not the case with SARS-CoV-2. And as there was no effective therapy, the health risks for HIS test persons in early 2020 were high.
But it should be noted that in any case there are ethical dilemmas in vaccine development. This is because when HIS are not used, tens of thousands of people must be involved in the phase 2 and phase 3 which then become necessary. And the much larger numbers alone may cause harm to subjects or third parties. In the phase 3 trial to develop Moderna's mRNA-1273 vaccine, 15,210 people received the drug and the same number received a placebo. 30 study participants in the placebo group became seriously ill, and one person died.[^14] The WHO emphasises: "Although challenge studies involve the additional risk associated with being infected with a challenge strain (compared to vaccine field trials, which do not increase the probability of infection), it is ethically salient to assessments of risk that challenge studies involve fewer participants, who are more closely monitored and provided with immediate treatment."[^15]
The comparison between the risks of phase 1-participants and HIS-participants is of particular interest. To be able to draw analogies, one real phase 1 study during the process to SARS-CoV-2 vaccine testing is explained in detail in the box below:
Table 1: Unsolicited Adverse Effects (AEs), Serious AEs, Medically Attended AEs and AEs of Special Interest
<table><tr><td></td><td>Relation- ship</td><td>2μg</td><td>4μg</td><td>6μg</td><td>8μg</td><td>12μg</td><td>Placebo</td></tr><tr><td></td><td>N=</td><td>47</td><td>48</td><td>46</td><td>44</td><td>28</td><td>32</td></tr><tr><td rowspan="2">Unsolicited</td><td>Any</td><td>22 (47)</td><td>32 (67)</td><td>29 (63)</td><td>28 (64)</td><td>21 (75)</td><td>14 (44)</td></tr><tr><td>Related</td><td>7 (15)</td><td>19 (40)</td><td>15 (33)</td><td>18 (41)</td><td>12 (43)</td><td>4 (13)</td></tr><tr><td rowspan="2">SAEs</td><td>Any</td><td colspan="6">3 (1.2)a</td></tr><tr><td>Related</td><td colspan="6">0</td></tr><tr><td>Medically attended AEs</td><td>Any</td><td>2 (4.3)</td><td>1 (2.1)</td><td>4 (8.7)</td><td>3 (6.8)</td><td>4 (14.3)</td><td>6 (18.8)</td></tr><tr><td>AESI</td><td>Any</td><td>0</td><td>0</td><td>0</td><td>0</td><td>0</td><td>0</td></tr><tr><td colspan="8">Italicised numbers are percentagesaThe 3 unrelated SAEs are not shown by group to maintain blind for this interim analysis</td></tr></table>
The three SAEs were classified as unrelated. It was obvious that these cases – one of which was, for example, a broken arm in a bicycle accident $^{21}$ – were not caused by the vaccination. In order to keep a complete account, they are nevertheless recorded in the study protocol. The conclusion of this phase 1 safety/tolerability study of CVnCoV, according to the project leader, Peter Kremsner: "There were no vaccine-related serious adverse effects for the participants. Dose-dependent increases in the frequency and severity of solicited AEs were mainly mild or moderate and of transient duration $^{22}$. This pleasing result was probably not recorded in all of the more than one hundred 'first in human' studies conducted in the 2020 race for the best vaccine to protect mankind against SARS-CoV-2. $^{23}$ Many of the vaccine development projects that were undertaken in 2020 were terminated when their unsolicited AEs were too severe.
#### Final ethical assessment
One can only make serious ethical judgements about empirical facts after one has properly understood them. The case study about organisation of phase 1 trials show that everything is done to protect volunteers from serious harm. But severe illnesses or even deaths are always possible. Nevertheless, ethicists have never objected to this kind of trials – as they are the lesser evil compared to having vaccines too late or not having them at all while a pandemic is raging. This sheds light on the ethical evaluation of HIS. In both cases, phase 1 trials and HIS, the financial compensation of subjects is of relevance for the final ethical assessment. In HIS, the subjects can become contagious after being infected with the virus, therefore there is the need to quarantine them for two weeks minimum. If one only reimburses loss of earnings for this time, one arrives at sums that invite polemical reporting. And unfortunately this really happened as the following case shows:
In England, during the Corona pandemic, there was a HIS to find out what is the minimum viral load that can lead to infection. So the goal here was not to directly accelerate vaccine development. Rather, the study goal was to find out what is the smallest possible amount of virus that can cause Covid-19 disease.[^24] The study was commented on in a ZEIT article as follows: "5,770 Euros please? Great. All you need is 17 days of time. You get your own room, a comfortable bed, video games, books, three meals a day and ideal medical care. And yougetinfectedwith Corona."[^25] To avoid this kind of polemic, I think it would be best to pay for accommodation and meals during isolation time, but refrain from paying any more compensation or an honorarium. Since monetary incentives (including for kidney donation, etc.) are more attractive to poorer people than to wealthier people, there is no real free choice. Given the great willingness to end the Corona pandemic as quickly as possible by participating in vaccination studies, enough volunteer participants would have been found.[^26]
Let us assume (counterfactually) that HIS would have taken place during summer 2020 to speed up vaccine licensure. It would have been mandatory from an ethical point of view to provide excellent care for Covid-19 treatment, including priority for any scarce lifesaving resources, in state-of-the-art facilities.[^27] In concrete terms, this would have meant that scarce resources, e.g. for the drug Remdisivir or artificial lungs, would have been kept ready for this group.
According to Shah et al, "for SARS-CoV-2 controlled human infection studies to be ethically permissible, risks to participants, study personnel, and third parties should be minimized, reasonable in relation to the social value of the research, and below the upper limit of acceptable risk."[^28] It is debatable if one should go one step further to protect the health of the HIS subjects. The very idea of phase 1 trials of Biontec, Moderna, AstraZeneca and other companies was to test efficacy of their vaccine candidates (next to tolerability). In the CVnCoV case study described above, as hoped, the antibodies against the spike protein (iGG) in the test persons' bodies rose sharply up to 10,000.[^29] It could be assumed that the subjects of the described phase 1 study (apart from the placebo group) had a certain individual vaccination protection as of August 2020.
Now imagine that only those few people that were somewhat protected against Covid-19 would be admitted for human infection studies (if they volunteered) - without installing an 'unvaccinated' control group. Scientifically usable data could be collected from such a HIS even if the establishment of a placebo group was completely dispensed with. The medical-scientific gain in knowledge would then not be as high as it could be. But it would still be many times higher than in the current procedure, where tens of thousands of people are recruited and then wait for a few in both subgroups to become infected.
Another important criterion would have been the 'informed consent' of the test persons. De facto, at least a basic virological-medical knowledge is necessary to be able to calculate the personal risk. Altruism, as commendable as it is, must be supplemented with knowledge. A written or oral test would have been the best way to check whether the test persons had really acquired a certain basic knowledge. But what if, in the case of a new virus, there are still no corroborated facts on pathogenicity (and thus on risk)? The test persons can also give their consent to participate in an HIS in which medicine or science does not yet know many variables. This only needs to be clearly communicated. With regard to the SARS-CoV-2 pandemic, it was known in the summer of 2020 that the virus triggers a much more severe course of disease in older people than in younger people. But many details were still unknown.
All in all, human autonomy should be the deciding argument. In particular, it is incomprehensible why our society legally allows phase 1 trials in vaccine development, but not subsequent human infection trials. As made clear in the case study below, the phase 1 trial subjects also take a risk. As long as someone can assess the risk to themselves, they should be allowed to act altruistically, even at the risk of their health or even their life.
It remains to be stated that no general judgment is possible about HIS to accelerate vaccine developments or approvals. In the case of SARS-CoV-2, the author considers it justified in retrospect, but this ethical judgment has no anticipatory effect on the next pandemic, when circumstances may again be quite different.
In any case, there should be a more open debate than before. The autonomy argument probably justifies human infection studies in more pandemic and endemic situations than previously assumed; there is again no obligation of hospitals etc. to provide the infrastructure for this.
--- In spring 2020, Tremmel participated in the phase 1 trial of the CureVac vaccine licensure process $^{30}$ as a subject and received $8\mu \mathrm{g}$ of the investigational vaccine (CVnCoV) twice.
[^1]: Synonyms are Human Challenge Studies (HCS) or Human Challenge Trials (HCT). _(p.1)_
[^10]: Reich 2021. _(p.4)_
[^11]: Wang 2022. _(p.4)_
[^14]: Baden et al 2021: 403. _(p.4)_
[^15]: WHO 2020: 6. _(p.4)_
[^24]: Gallagher 2021. _(p.6)_
[^25]: Reich 2021. _(p.6)_
[^26]: See, for instance: www.1daysooner.org/. _(p.7)_
[^27]: Eyal et al. 2020: 1753. _(p.7)_
[^28]: Shah et al. 2020: 833. _(p.7)_
[^29]: For details, see Kremsner 2021a. _(p.7)_
[^2]: ${}^{2}$ Tremmel 2021. _(p.2)_
[^3]: This refers to the large trial study with thousands of participants. In practice, this is not always referred to as Phase 3, but also as Phase 2/3, Phase 2a/3 or Phase 2b/3, depending on the circumstances. _(p.2)_
[^4]: Johnson & Johnson is not included here because only one dose was administered here. This automatically reduces the time for the clinical trials. As it turned out, however, the immune protection also suffered. _(p.2)_
[^5]: Baden et al 2021; Polack et al 2020; Voysey et al 2021; Kremsner et al 2021b. _(p.2)_
[^12]: WHO 2020: 14. _(p.4)_
[^13]: WHO 2020: 2 _(p.4)_
[^16]: Kremsner et al. 2021a: _(p.5)_
[^17]: Kremsner et al. 2021a: 932. _(p.5)_
[^18]: There were individual cases of severe health damage in 'first in human' studies in medical history, cf. Attarwala 2010. _(p.5)_
[^19]: Local: pain at the injection site, redness, swelling and itching. Systemic: headache, fatigue, chills, muscle or joint pain, nausea/vomiting and diarrhoea. _(p.5)_
[^20]: US Department of Health and Human Services, Food and Drug Administration (FDA) 2007. _(p.5)_
[^21]: Kremsner et al. 2021a: 935. _(p.6)_
[^22]: Kremsner et al. 2021a: 932. _(p.6)_
[^23]: A list of projects is continuously updated by WHO: https://www.who.int/publications/m/item/draft-landscape-of-covid-19-candidate-vaccines. _(p.6)_
[^30]: Kremsner et al. 2021a. _(p.7)_
[^932]: "The study was monitored by an internal safety review committee (iSRC) and a data safety monitoring board (DSMB)." _(p.5)_
Generating HTML Viewer...
References
15 Cites in Article
Lindsey Baden,Hana El Sahly (2021). Efficacy and Safety of the mRNA-1273 30 Kremsner et al. 2021a. SARS-CoV-2 Vaccine.
N Eyal,M Lipsitch,P Smith (2020). Human Challenge Studies to Accelerate Coronavirus Vaccine Licensure.
Laura Gallagher (2021). call-for-investigation-of-rodneys-death-feb-17-1987-1-p.
Tuvan Gumrukcu,Ali Kucukgocmen (2020). Turkey says China's Sinovac COVID vaccine 91.25% effective in late trials.
Euzebiusz Jamrozik,Michael Selgelid (2021). Human Challenge Studies in Endemic Settings.
Kremsner Peter,G Guerrero,Rodolfo Arana-Arri,Eunate (2021). Efficacy and safety of the CVnCoV SARS-CoV-2 mRNA vaccine candidate in ten countries in Europe and Latin America (HERALD): a randomised, observer-blinded, placebo-controlled, phase 2b/3 trial.
Stephan Reich (2021). http://ljournal.ru/wp-content/uploads/2016/08/d-2016-154.pdf.
Seema Shah (2020). Ethics of controlled human infection to address COVID-19.
Jörg Tremmel (2021). Unknown Title.
(2007). FDA Issues Guidance for Collection of Race and Ethnicity Data in Clinical Trials for FDA Regulated Products.
Merryn / Voysey,Suea Clemens,Shabir Madhi (2021). Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: an interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK.
Haidong Wang (2022). Australian COVID-19 pandemic: A Bradford Hill Analysis of Iatrogenic Excess Mortality.
(2020). Key criteria for the ethical acceptability of Covid-19 human challenge studies.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Jörg Tremmel. 2026. \u201cHuman Infection Studies and the SARS-CoV-2 Pandemic\u201d. Global Journal of Medical Research - F: Diseases GJMR-F Volume 23 (GJMR Volume 23 Issue F3).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.