The analysis of the methodological principles on which pyrometry is based is carried out. Special attention is paid to the main methodological postulate and its consequences. The roots of its formation are considered. It is shown that the large methodological errors characteristic of pyrometry are a direct consequence of the system of priorities arising from this postulate. A new basic methodological postulate of pyrometry is formulated, it is shown that the development of the ideas contained in it will reduce the number of methodological errors by an order of magnitude or more, and the necessary and sufficient conditions for this reduction are formulated.
## I. INTRODUCTION
The rapid development of microelectronics and microprocessor technology in the last quarter of the 20th century made it possible to bring instrument engineering to a qualitatively higher level. In many industries, the instrumental errors of measuring instruments have decreased to fractions of a percent. Pyrometers are no exception here.
But at the same time, as is known, any of the pyrometry methods has inherent methodical errors[^1], the magnitude of which can reach 10... 15%, i.e. an order of magnitude or more exceeding the instrumental ones. There are still no ways to guarantee their reduction to the level of 1-2%. And the most significant thing is that over the past half century, it has not been possible to reduce the magnitude of these methodological errors in relation to any material whose non-contact temperature measurement may be in demand. And the reason for this, of course, is not at all due to dishonesty or low qualifications of researchers.
## II. ON THE NEED TO ANALYZE THE METHODOLOGY OF PYROMETRY
The author of this work argues that the problems of pyrometry are methodological in nature. Their solution requires analysis and possible revision of the methodological principles of pyrometry. The work [1] is devoted to this analysis.
For applied science, methodology is understood as a system (complex, interconnected set) of postulates and principles of research activity, which a scientist relies on in the course of obtaining and developing knowledge within a given specific scientific discipline or several scientific disciplines ([^2]).
Obviously, there are methodological postulates, principles and approaches common to all branches of technical sciences (the obligation of mathematical calculations or modeling, the correspondence of calculated data to experimental data, etc.), and there are also particular, specific ones that apply only to individual industries or to one specific industry. It is quite obvious that it is not methodological principles and postulates common to all branches of technical sciences that slow down the development of pyrometry, because even in related fields (for example, in contact methods of temperature control) there are no problems of large methodical errors. Therefore, the source of irreducible methodical errors should be sought in specific methodological postulates and principles of pyrometry.
However, the methodological postulates and principles specific to pyrometry have not yet been clearly formulated. The reason is that most specialists in this field have not yet realized that the problems of incessant huge methodical errors are methodical in nature. The desire to formulate methodological principles and postulates that determine the course of development of a particular industry arises only after realizing the futility of trying to solve the problem within the framework of existing knowledge. Half a century of stomping in place on the issue of reducing methodical errors from a 10...20 percent level to units or fractions of a percent just suggests that there is a need to revise the methodological postulates and principles of pyrometry.
## III. METHODOLOGICAL PRINCIPLES AND APPROACHES IN PYROMETRY
The principles and approaches most characteristic of modern pyrometry are listed below ([1]):
1. Regular resumption of attempts to find the temperature of the measured object only by its radiation, based only on Planck's or Wien's laws, without taking into account its radiative properties.
2. Consideration of the emissivity ${}^{2}$ as a minor, secondary, and even interfering factor,both in theoretical constructions and in the practical implementation of pyrometry methods.
3. The use of only those reference means during verification[^3] and calibration that perfectly implement the laws of radiation ("absolutely black bodies", BB).
4. The lack of reference means and measuring instruments of spectral emissivity.
5. The almost universal disregard of the dependence of the spectral emissivity characteristic of most objects on the temperature of the object and on the state of its surface (roughness, the presence of liquids, oil films, etc.).
6. Reduction (both in theoretical calculations and in practice) of the complex influence of emissivity to a one-dimensional effect described by a simple numerical coefficient, with complete disregard for the fact that emissivity is not a coefficient, but a function of at least two variables.
7. The use of various adjustment organs in almost all modern pyrometers, which make it possible to adjust the measurement results in any direction within a fairly wide range.
8. The lack of developed algorithms for determining the actual temperature of an object by its pseudotemperature (brightness, partial radiation, radiation or spectral ratio), taking into account the temperature dependence of the emissivity characteristic of most objects.
As for paragraphs 3, 4 and 7, they are obvious. The statement of paragraph 2 also becomes obvious when analyzing almost all books published over the past 50 years, the authors of which try to cover pyrometry as a whole, rather than highlight certain selected issues. In these books, the laws of Planck, Wien, Stefan-Boltzmann, Rayleigh-Jeans, Kirchhoff, Lambert are usually described in detail first, and only after that the concept of emissivity is introduced, characterizing the difference between the radiation of real objects and the radiation of the BB.
According to claim 1, measurements in polarized light can be noted [^3], the use of multiband spectral-ratio pyrometers with narrow spectral bands [^4], the use of spectrometers [^5], etc.
As a confirmation of what was said in paragraphs 5, 6 and 8, the following can be cited.
In the known relations present in almost all books on pyrometry, linking the actual temperature of an object $T_{\mathrm{d}}$ with its brightness or radiation temperature, the emissivity appears in the form of constants $\varepsilon_{\lambda}, \varepsilon_{\mathrm{s}}$:
$$
\frac{1}{T _ {\mathrm{d}}} = \frac{1}{T _ {\mathrm{b}}} + \frac{\lambda}{c _ {2}} \ln \varepsilon_ {\lambda} \tag{1}
$$
where $T_{\mathrm{d}}$ is the actual temperature, K; $T_{\mathrm{b}}$ is the brightness temperature measured by the pyrometer, K; $c_{2} = 1.4380 \cdot 10^{-2} \mathrm{~m} \cdot \mathrm{K}$; $\lambda$ is the operating wavelength of the monochromatic brightness pyrometer, m; $\varepsilon_{\lambda}$ is the radiation coefficient of the object at the wavelength $\lambda$.
$$
T_{\mathrm{d}} = T_{\mathrm{r}} / \sqrt[^4]{\varepsilon_{s}}
$$
where $T_{\mathrm{d}}$ is the actual temperature, K; $T_{\mathrm{r}}$ is the radiation temperature measured by the pyrometer, K; $\varepsilon_{s}$ is the integral radiation coefficient.
However, if we take into account that $\varepsilon_{\lambda}$ and $\varepsilon_{s}$ are not constants, but functions of wavelength $\lambda$ and temperature $T_{\mathrm{d}}$, and instead of constants substitute functions $\varepsilon (\lambda,T_{\mathrm{d}})$ in (1) and $\varepsilon_{\mathrm{s}}(T_{\mathrm{d}})$ in (2), then simple calculation relations (1) and (2) turn into equations unsolvable in analytical form. There are no algorithms for solving these equations in general.
## IV. THE MAIN METHODOLOGICAL POSTULATE OF PYROMETRY
The analysis of the above methodological principles and approaches characteristic of modern pyrometry allows us to identify something common to all of them without exception. This is the implicitly postulated priority of radiation laws in this industry over all other laws and patterns used to determine the temperature of heated bodies by their radiation. It is she who is today the main methodological postulate specific to pyrometry, which hinders its development.
This methodological postulate has a historical origin, since the laws of radiation were formulated back in the XIX century, and there is still no theory that would link the radiative characteristics of a substance with its physico-chemical constants, and at the same time would not diverge from experimental data in the entire spectral range.
Of particular importance is the fact that this priority is postulated implicitly, by stealth. None of the researchers claims that finding the exact temperature of an object by its radiation without knowing its radiative properties is possible. But in practice, all modern research in pyrometry is aimed precisely at finding the temperature of heated objects without knowing their radiative characteristics. After all, if this succeeds, it will be possible to forget about the dreary measurements of the emissivity, depending on both the state of the object's surface and its temperature. From this point of view, the game is worth the candle, since there are still no devices for measuring emissivity, and experimental installations that allow this to be done are large, expensive, low-mobility, measurements on them require high qualifications and a lot of time. Therefore, the prospect of learning how to measure temperature by radiation without knowing the radiative properties looks very tempting.
The most likely solution to this problem seems to be using spectrometers, so today most research is conducted in this area [^5]. However, the possibility of such a solution for any predetermined material is not yet obvious.
If we return to the pyrometers, then the following should be noted. The above-mentioned emissivity characterizes the difference between the radiation of a real object and the radiation of an BB. If the differences are small, then the measurement error with a pyrometer calibrated according to the BB is also small. But for many objects that have to be measured with pyrometers, the differences in their radiation spectrum are quite large from the spectrum of the radiation of the BB.
An BB-calibrated pyrometer, by definition, cannot correctly measure the temperature of an object that does not emit as an BB. The error that occurs during such measurements is the main methodical error, it is determined not by the quality of calibration, but by the problem of the measurement method (i.e., the need to measure an object that emits differently from the sample from which the pyrometer was calibrated). How can such an error be reduced or eliminated altogether?
In today's practice, pyrometers are equipped with regulators, with the help of which a certain coefficient can be entered into them, usually taking a value from 0.1 to 0.99...1. This coefficient is usually called the "blackness coefficient", "degree of blackness" or "radiation coefficient". Using this coefficient, the operator can change the measurement result. It is assumed that he knows the "correct" value of this coefficient, and by setting it, he will correct the pyrometer readings and eliminate the mentioned methodical error.
To understand the negative consequences of this approach, you need to ask yourself - where do these coefficients come from? In the best case, measurements once made under these conditions are usually quite rough, with a small number of samples, without fully taking into account all factors affecting the result, without estimating the error. But more often - from literary sources compiled according to the same measurement results, performed by unknown people, unknown when, and with the same disadvantages.
The main thing here is that with this approach, the correction value is not calculated, but determined experimentally by selecting the radiation coefficient for the value at which the pyrometer will show the correct result (or one that is considered correct for one reason or another)[^4]. Let's add to this that manufacturers do not provide information about what the algorithm for correcting the measurement results of the radiation coefficient entered into this pyrometer is. The latter completely excludes the possibility of correctly accounting for the effect on the pyrometer measurement result of the difference between the radiation spectrum of the measuring object and the frequency response spectrum, the dependence of this difference on the temperature of the object itself, and on the spectral range of the pyrometer, and on the width of the range, and on the state of the surface of the object, and a number of other parameters. As a result, fitting to the expected result remains the only way to correct. In production practice, this leads to the fact that the technologist does not know which of the radiation coefficients to choose from the abundance available in various sources. As a result, the selection is made "by eye" so that the measurement result corresponds to the expected one. This is where users have measurement errors with pyrometers up to $10 - 20\%$.
That is, the user is trying to eliminate the methodical error, but the method used today to exclude it does not guarantee its reduction. With a successful combination of circumstances, it can decrease to the level of $1 - 2\%$, and if unsuccessful, it can remain at the same level of $10 - 20\%$. And at the same time, the instrumental errors of modern pyrometers often do not exceed $0.2\ldots 0.5\%$. That is, the improvement of pyrometers in terms of further reducing the instrumental error at this stage is meaningless, because it does not lead to an increase in measurement accuracy. Improving the accuracy of measurements in pyrometry has run into a barrier of methodical errors. How to overcome it?
## V. THE NEED TO ISOLATE THE PLANCK COMPONENT FROM THE TOTAL RADIATION OF THE OBJECT
To overcome this barrier, it is necessary to realize what happens when measuring when we neglect the influence of the radiative properties of the measured object. Let's turn to Fig. 1. Here are the Spectral Radiance (SR) of transformer steel when heated to $1127^{\circ}\mathrm{C}$ in a nitrogen-hydrogen atmosphere (1) and high-alumina firebrick heated in air to the same temperature (2). The results are obtained based on the data given in [^4]. Here, for comparison, the SR of the source of ideal Planck radiation - BB (3) at the same $1127^{\circ}\mathrm{C}$ is given. Obviously, the isolation of the Planck component[^5] 3 from dependencies 1 and 2 is an operation completely unobvious, none of the radiation temperature measuring devices is designed to solve this problem.
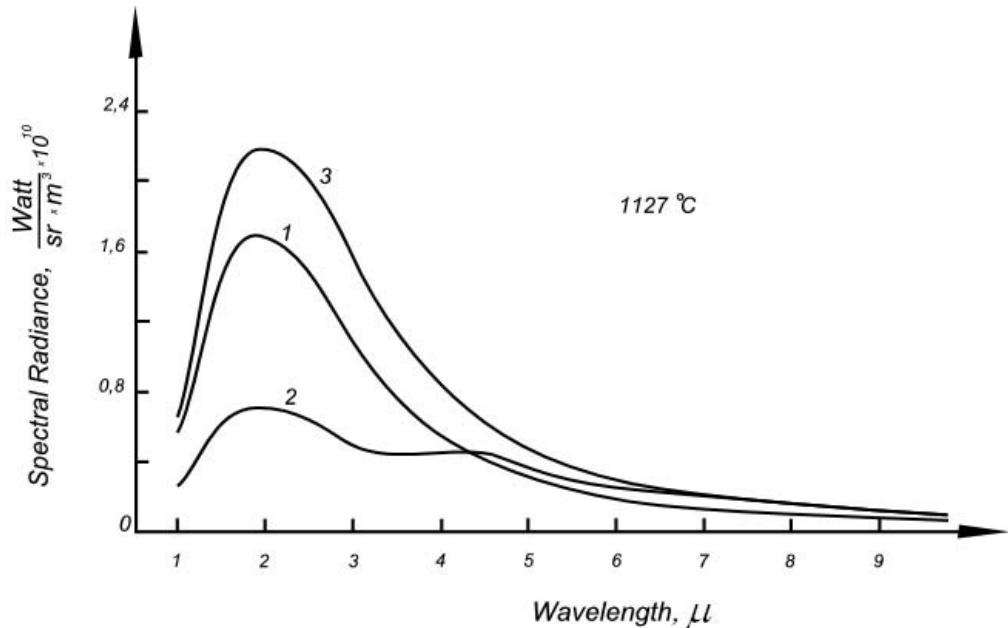 Figure 1: Spectral Radiance (SR) of transformer steel when heated to
$1127^{\circ}\mathrm{C}$ in a nitrogen-hydrogen atmosphere (1), high-alumina firebrick heated in air to the same temperature (2) and BB at $1127^{\circ}\mathrm{C}$ (3)
The need to isolate the Planck component from the complete SR of an object arose at the dawn of the development of practical pyrometry. This turned out to be necessary because pyrometers are universally calibrated by BB, and after such calibration they can correctly measure the temperature of only those objects that emit as BB - "black" and "gray". When measuring other objects, it is that part of their radiation that distinguishes it from the radiation of the BB, and introduces an additional error, which we call methodical. Therefore, in order to exclude it, one way or another, its banking component must be isolated from the entire radiation of the object. Or somehow exclude the influence of non-Plank component.
## VI. SPECTRAL EMISSIVITY AND ITS ROLE IN ELIMINATING METHODOCAL ERRORS
The spectral emissivity can be defined as the result of the functional division of the SR of a real object into the SR of an BB (hereinafter, functional division is understood to be the division of the ordinate of the function-divisible by the ordinate of the divisor function for the same abscissa for the set of all possible abscissae).
Figure 2 shows the dependences on the wavelength of the spectral emissivity of transformer steel (1) and high alumina firebrick (2), corresponding to a temperature of $1127^{\circ}\mathrm{C}$ [^4]. As noted, we call these functions spectral emissivity in order to distinguish them from the coefficients introduced into energy pyrometers and still called by inertia by many users of pyrometers and authors of articles on pyrometry "emissivity". Let's add that the spectral emissivity is also a function of the temperature of the object.
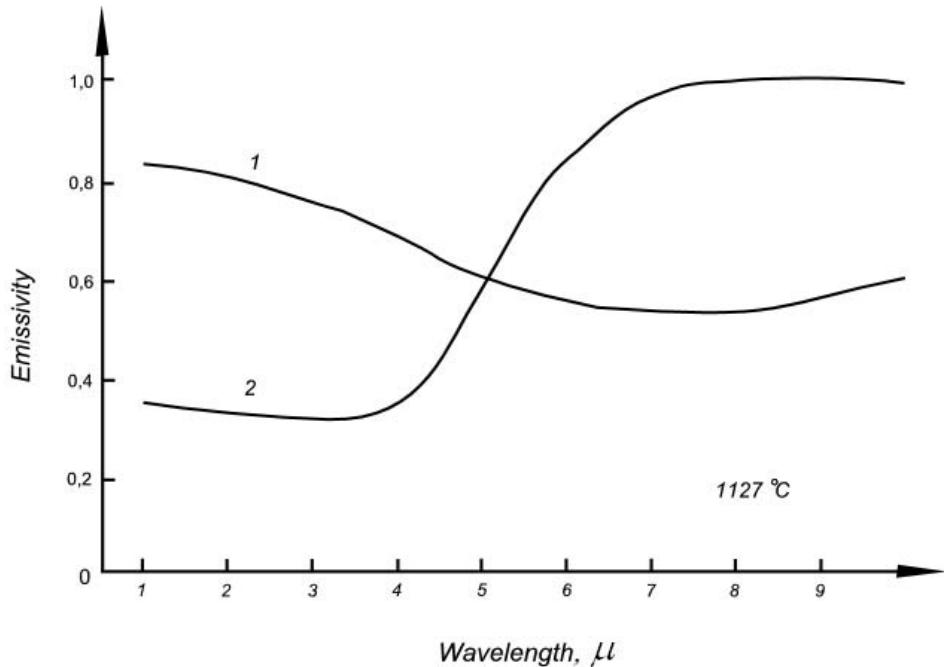 Figure 2: Spectral emissivity of transformer steel when heated to
$1127^{\circ}\mathrm{C}$ in a nitrogen-hydrogen atmosphere (1), and high-alumina firebrick heated in air to the same temperature (2)
Thus, the spectral emissivity is a function that contains the difference between the SR of a real object and the SR of an BB having an equal temperature with the object. Its non-accounting (or incorrect accounting) does not make it possible to correctly convert the SR of the measured object into a Planck curve of equal temperature with the object, according to which this temperature can be measured without any systematic errors using an BB-calibrated pyrometer.
One of the ways of such a separation of the Planck component from the total radiation spectrum is the functional division of the SR of a real object into its real spectral emissivity. However, this is possible only if we have at our disposal almost complete spectral dependences of the SR and emissivity, i.e. lying in the wavelength range where the values of the Planck curves for these objects exceed $0.5 - 1\%$ of their maxima. This is typical for spectral pyrometry, which is at the initial stage of its development. But pyrometers are not spectrometers, and such a functional division is impossible for them. Nevertheless, knowledge of the spectral emissivity is also necessary for classical energy pyrometers[^7] and pyrometers of spectral ratio (at least in the range of spectral sensitivity of these measuring instruments). However, it should be used in the allocation of the Planck component in a slightly different way. Here is one of the options for such a selection.
First, the full view of the object's SR is calculated for all measured temperatures (for example, with a given step within the entire measurement range). To do this, for each of the temperatures, its spectral emissivity is functionally multiplied by its Planck function. Next, using the obtained SR, a set of pseudotemperatures (brightness, or radiation, or spectral ratio - depending on which type of devices the correction is performed) is calculated. This calculation can be performed using calibration $^8$ functions. Then a table is formed in which the actual temperature is assigned to each of the obtained pseudo-temperatures - the one whose Planck curve was used to calculate this pseudotemperature. And at the last stage, the actual temperature of the measured object is determined based on the result of the pyrometer measurement using the above-mentioned recalculation table ([^6]). When using the real spectral emissivity in this algorithm, methodical errors are excluded, and the error is determined only by the instrumental errors of the pyrometer used.
In fact, in this case, we modify the scale determining function (inverse of the calibration function) of the pyrometer so that it takes into account the difference between the SR of the measured object and the SR of the BB. Which is essentially equivalent to separating the Planck component from the total flux of its radiation.
Once again, I would like to draw readers attention to an important statement - if we want to measure temperature with a pyrometer without methodical errors, we must somehow isolate the aforementioned Planck component from all the radiation that came to the pyrometer. Its measurement with an BB-calibrated pyrometer will give the desired result. Or, one way or another, exclude the influence of a non-Planck component, which will lead to the same measurement result.
And how do we allocate the Planck component today? Few people have thought about this - the extraction operation, which is the result of a complex mathematical calculation, has been replaced by correction using coefficients introduced into pyrometers, most often determined experimentally. The task of isolating the Planck component in today's pyrometry has been simplified to the limit - the radiation flux that came to the energy pyrometer is actually simply divided by the radiation coefficient introduced into it. A complex functional transformation is ultimately reduced to division by a constant taken from tables, which very often have a very distant relation to the measured object. Hence the methodical errors, which are absolutely independent of the pyrometer's own instrumental error.
Naturally, classical pyrometers are not spectrometers, they do not measure the SR with some kind of normalizing coefficient, but its integral value in the form of a signal at the output of their radiation receiver. Therefore, the functional division mentioned above is not a task for them. If we are talking about energy pyrometers, then correction by the radiation coefficient is all they can do. But then the procedure for isolating the Planck component should somehow "migrate" at least to the calculation of the radiation coefficient. Such a calculation of the radiation coefficient, taking into account the spectral emissivity $\varepsilon (\lambda,T)$, is described below (see (4)). However, it has certain limitations, which will be described later. Therefore, the task of comprehensively eliminating the influence of temperature-spectral emissivity on the pyrometer measurement result is still relevant. Taking into account all the above, it will be formulated and specified in the last subsection of this article.
It is safe to say that devices for remote temperature measurement will continue to be calibrated according to BB in the future. Consequently, the task of isolating the Planck component from the full SR, according to which these devices will measure temperature, will also remain. And its solution is impossible without knowledge of the spectral emissivity. And the more precisely it is determined (as well as the more accurately the calibration of the measuring instrument used is carried out), the more accurately the temperature of the measured object will be determined. In a different way, using some averaged coefficients introduced into pyrometers, it will not be possible to get rid of methodical errors.
## VII. ABOUT THE NEW BASIC
### METHODOLOGICAL POSTULATE OF
#### PYROMETRY
All of this means the need to rethink the basic methodological postulate mentioned above, which is specific to pyrometry, which consists in the fact that to determine the temperature of an object by its radiation, it is enough to know and use only the laws of radiation, ignoring accumulated or still missing knowledge about the radiative properties of specific objects. It should be replaced by a postulate proclaiming that the exclusion of methodical errors in non-contact measurement of its temperature is impossible without knowledge of the real (not generalized or averaged!) the spectral emissivity of a particular measured object, and its correct accounting. Attempts to deceive nature and continue to ignore the need to accumulate knowledge about the radiative properties of objects will leave unchanged methodical errors that have hindered the development of pyrometry for more than half a century.
However, this is not all. Since until now there has not been a theory that adequately connects the spectral emissivity with the physico-chemical constants of the object's material, it will be necessary to obtain the necessary information about the spectral emissivity experimentally. At the same time, it should be noted that at the moment there are no specialized measuring instruments for spectral emissivity on the market. Nevertheless, in the works of the author ([7, 8] the technical possibility of creating such measuring instruments is demonstrated, two such devices are described, one of which is protected by a patent of the Russian Federation.
The author also argues the need to have a verification scheme for such devices, as well as a currently missing standard of emissivity, which will stand at the top of this verification scheme ([9, 10]).
## VIII. ABOUT WHAT ELSE IS NEEDED
However, knowledge of the spectral emissivity is only a necessary condition for reducing or completely eliminating large methodical errors inherent in pyrometry methods. It is not sufficient for this reduction, since algorithms and methods for minimizing/eliminating these errors with full consideration of temperature-spectral emissivity are either insufficiently developed or absent. Therefore, it is necessary to solve the following scientific problems.
1. As is known, when measuring "non-gray" objects with spectral-ratio pyrometers, they have a methodical error, the mechanism of which is discussed in detail in[11]. The ratio(3)is known, which allows (knowing the spectral emissivity) to compensate for this methodical error:
$$
\frac {1}{T _ {d}} - \frac {1}{T _ {s p . r e l}} = \ln \frac {\varepsilon_ {\lambda_ {1}}}{\varepsilon_ {\lambda_ {2}}} \frac {1}{c _ {2}} \frac {1}{\frac {1}{\lambda_ {1}} - \frac {1}{\lambda_ {2}}} \tag {3}
$$
where $T_{d}$ is the actual temperature, K; $T_{sp,rel}$ is the temperature of the spectral ratio measured by the pyrometer, K; $c_{2} = 1.4380 \cdot 10^{-2} \mathrm{~m} \cdot \mathrm{K}$; $\lambda_{1}$ and $\lambda_{2}$ are the operating wavelengths of the narrowband pyrometer of the spectral ratio, m; $\varepsilon_{\lambda_{1}}$ and $\varepsilon_{\lambda_{2}}$ are the emission coefficients at wavelengths $\lambda_{1}$ and $\lambda_{2}$.
However, this ratio is valid only for pyrometers with narrow (no more than 10...20nm) spectral bands.
At the same time, the vast majority of spectral-ratio pyrometers produced today are broadband, the width of the spectral bands of their sensitivity is tens or even hundreds of nanometers. As a result, ratio (3) is essentially inapplicable for the absolute majority of pyrometers used in practice, and has more theoretical than practical value. Therefore, a universal method is needed to correct the methodical error that occurs when measuring the temperature of "non-gray" objects with any pyrometers of spectral ratio. This method is developed and described in [6, 12]. They present an algorithm for machine calculation of the temperature of the spectral ratio of "non-gray" objects using a calibration function, and an experimental study of the method is carried out. However, the proposed method (as well as in the ratio (3)) does not take into account the temperature dependence of the spectral emissivity. Therefore, the method needs to be improved, taking into account this dependence.
Thus, the problem can be formulated as follows: the above-mentioned universal method for correcting pyrometers of the spectral ratio must be improved in such a way as to take into account the temperature dependence of the spectral emissivity. After that, it, together with information about the spectral emissivity, will become necessary and sufficient conditions for minimizing/eliminating methodical errors in the method of pyrometry of the spectral ratio.
The solution of this problem is described by the author in [13].
2. In contrast to the spectral ratio pyrometry method, the emissivity correction is fundamentally necessary in the energy pyrometry method. To do this, before
measuring, a correction factor is introduced into the energy pyrometer, which in this work is called the radiation coefficient. The radiation coefficients introduced into pyrometers are almost universally determined experimentally, by adjusting this coefficient to the value at which the result of temperature measurement using a pyrometer is close to the result of measurement by contact methods. Once selected in this way, the radiation coefficient is then usually transferred to all pyrometers that have to measure such an object. The measurement errors caused by such a transfer are described in [14]. And, moreover, with this approach, it is impossible to correctly take into account not only the spectral range of the pyrometer used, but also the temperature dependence of the emissivity. And this in turn leads to the appearance of additional methodical errors described in [15].
In [16], a ratio is given that allows the recognition of the spectral emissivity and spectral sensitivity characteristics of a photodiode pyrometer to correctly determine the radiation coefficient:
$$
\varepsilon_ {\lambda , T} = \frac {\int_ {\lambda_ {1}} ^ {\lambda_ {2}} \varepsilon (\lambda , T) S (\lambda) E (\lambda , T) d \lambda}{\int_ {\lambda_ {1}} ^ {\lambda_ {2}} S (\lambda) E (\lambda , T) d \lambda} \tag {4}
$$
where $\varepsilon_{\lambda, T}$ is the radiation coefficient at wavelength $\lambda$ for temperature $T$, $\varepsilon(\lambda, T)$ is the spectral emissivity of the object; $S(\lambda)$ is the spectral characteristic of the pyrometer sensitivity; $E(\lambda, T)$ is the Planck function; $\lambda_1$ and $\lambda_2$ are the lower and upper limits of spectral sensitivity.
Since (4) represents the ratio of two definite integrals that are practically insoluble analytically, its use in practice by metrologists and technologists of enterprises is hardly possible – for this, a specialist must have a legally purchased package such as MathCad or Mathlab and be able to use it. Therefore, it is necessary to develop simple and freely distributed programs with which users with minimal computer skills could determine the radiation coefficient according to (4). One of the variants of the set of such programs is given in [17].
Further, since $\varepsilon (\lambda,T)$ and $E(\lambda,T)$ depend on the temperature of the object, the coefficient $\varepsilon_{\lambda,T}$ also depends on temperature. That is, the radiation coefficient found using (4) depends on the temperature. In this case, a vicious circle arises - in order to measure the temperature correctly with an energy pyrometer, you need to enter the correct value of the radiation coefficient into it. But to find the correct value of the radiation coefficient, you need to know the temperature of the object, which we are still only going to measure. The established practice of adjusting the radiation coefficient to the correct result, if this correct result is unknown in advance, does not solve the problem.
It follows from the above that for the accurate correction of energy pyrometers for emissivity, not only the ratio (4) is required, but also a preliminary knowledge of the temperature to be measured, because without this it is impossible to correctly select those $\varepsilon (\lambda,T)$ and $E(\lambda,T)$ that are necessary to calculate $\varepsilon_{\lambda,T}$ according to (4).
Therefore, the task can be formulated as follows: for energy pyrometers, it is necessary to develop a method of correction for emissivity, different from the one currently used, in which there is no need for prior knowledge of the temperature to be measured in order to correctly use the temperature-dependent radiation coefficient $\varepsilon_{\lambda,\mathrm{T}}$ i.e. it is necessary to break this vicious circle when you need to know its correct value to measure temperature, and in order to calculate it correctly in accordance with (4), we need to know this temperature, which is still unknown to us. The current method of correction is not capable of breaking it without some additional information.
It is the above-mentioned method of correction, which differs from the currently used one, in combination with knowledge of temperature-dependent spectral emissivity, that will be the necessary and sufficient means to minimize/exclude methodical errors in the method of energy pyrometry.
The solution of the mentioned problem is planned by the author to be published in one of the next issues of one of the periodicals covering measuring topics. A general approach to solving this problem is formulated in [18].
The implementation of solutions to the formulated tasks will dramatically reduce the methodical errors in pyrometry to a level comparable to the level achieved by instrumental errors.
## IX. CONCLUSION
1. The main methodological postulate specific to pyrometry is formulated – the implicitly postulated priority of radiation laws in this branch over all other laws and patterns used to determine the temperature of heated bodies. It is shown that it is the unconscious adherence to this postulate that does not allow for half a century to solve the problem of reducing/eliminating methodical errors in non-contact temperature control methods.
2. A new, alternative to the above, basic methodological postulate specific to pyrometry is formulated. He proclaims that without knowledge and use of the real (not generalized or averaged!) the temperature-spectral emissivity of a particular measured object it is impossible to exclude
methodical errors in the non-contact measurement of its temperature.
3. Since to date there has not been a theory that adequately connects the temperature-spectral emissivity with the physico-chemical constants of the object's material, it is argued that it will be necessary to obtain the necessary information about the spectral emissivity experimentally. At the same time, it should be noted that at the moment there are no specialized measuring instruments for spectral emissivity on the market. Nevertheless, in a number of the author's works, the technical possibility of their creation is demonstrated, two such devices are described, one of which is protected by a patent of the Russian Federation.
4. However, knowledge of the spectral emissivity is only a necessary condition for reducing or completely eliminating large methodical errors inherent in pyrometry methods. For sufficiency, it is necessary to develop algorithms and methods for accounting for the effect on the spectral emissivity of the temperature of the measured object, which are now either insufficiently developed or completely absent.
5. References are given to the algorithms developed by the author of this article for taking into account the influence of temperature on the spectral emissivity of an object used in the methods of spectral ratio pyrometry and energy pyrometry.
6. The algorithms noted in paragraph 5 (recognition of temperature-spectral emissivity), when implemented, will reduce the methodical errors of pyrometry methods to a level comparable to the level achieved by instrumental errors.
[^2]: Further, everywhere by the emissivity of an object we will understand its spectral $\varepsilon (\lambda)$ or temperature-spectral $\varepsilon (\lambda,T)$ emissivity _(p.2)_
[^3]: In Russia, verification is the confirmation by one of the accredited state metrological centers of the declared metrological characteristics of the device being verified _(p.2)_
[^4]: This is a problem that many still do not realize. Correction by experimentally selected coefficients causes very serious complaints from the point of view of metrology. This can be explained using such a simple example. Let's assume that we measure small voltage values in a printed circuit assembly with a DC microvoltmeter. As is known, when the copper probe of the device comes into contact with the Kovar pin of the chip, a fairly significant contact potential difference occurs, about $30\mathrm{mV}$ at room temperature. It is quite obvious that if, instead of subtracting this potential difference from the measurement result (adjusted, moreover, taking into account the temperature of the output of the chip), we smoothly "tweak" the gain of the microvoltmeter to the value that, according to someone once made estimates, gives the correct value of the measured value, then not only about the unity of measurements in radio engineering, but also their accuracy can be forgotten. It is unacceptable to exclude errors by the method of "fitting an experimentally selected coefficient to the correct result", without relying on the measurement of the influencing quantity and on knowledge of its dependencies on certain environmental parameters. _(p.3)_
[^5]: Here and further, under the Planck component (Planck curve), we will understand the CR BB. _(p.4)_
[^7]: Energy pyrometers are understood to be all pyrometers having only one radiation receiver, which determine the temperature by the magnitude of the signal from the receiver, i.e. by the magnitude of the energy flow that came to it _(p.5)_
[^6]: In this paper, the author calls this coefficient the "radiation coefficient", not emissivity. _(p.4)_
Generating HTML Viewer...
References
19 Cites in Article
A Frunze (2014). Metrological problems of modern energy pyrometry.
A Ya,N Baskakov,Tulenkov (2004). Methodology of scientific research: Textbook.
D Ya,Svet (1968). Objective methods of high-temperature pyrometry with a continuous radiation spectrum.
A Belenky,M Dubinsky,M Ladygichev,V Lisienko (2007). Temperature measurement: theory, practice, experiment.
A Magunov (2012). Spectral pyrometry.
A Frunze (2010). A numerical method of determining the spectral-ratio temperature.
(2014). Do We Need to Postulate Self-Objecl Differentiation in the First Year.
V Glebovskaya,S Tkachev,S Ivanov,A Nazarenko,A Rasulov,S Gordeev,P Bulychkin,N Besova,U Barsukov,O Trofimova,T Borisova,E Timoshkina (2017). Hyperthermia is an additional option in the treatment of squamous cell cancer of the anal canal.
Kovnev A. V.,Goffman V. G.,Gorokhovskii A. V.,Gorshkov N. V.,Kompan M. E.,Telegina O. S.,Tret’yachenko E. V.,Sleptsov V. V.,Baranov A. M. (2011). Impedance spectroscopy of potassium polytitanate modified with cobalt salts.
A Frunze (2012). Personality traits relating to childhood experiences may create addictive personality.
A Frunze (2018). Metrological Problems of Modern Spectral Pyrometry.
A Frunze (2013). About one little-known feature of spectral ratio pyrometers today.
M Samoilov,A Frunze (2010). Improving the accuracy of temperature measurements of "nongray bodies" with a broadband pyrometer of spectral ratio.
A Frunze (2023). Improving the accuracy of temperature measurement by a spectral ratio pyrometer.
A Frunze (2018). Metrological problems of modern energy-controlled pyrometry.
A Frunze (2012). Methodological errors of modern pyrometers and ways to minimize them.
D Ya,Svet (1982). Optical methods for measuring true temperatures.
A Frunze,R Gorbunov,D Simakov (2021). A set of low-level programs for calculating the temperature of the spectral ratio when measuring the temperature of "non-gray" bodies with two-spectral broadband pyrometers, In the collection: Current problems and prospects for the development of radio engineering and infocommunication systems.
A Frunze (2014). An algorithm for determining the actual temperature of an object, taking into account the temperature dependence of its emissivity, In the collection: Energy-saving technologies in industry.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Alexandr Frunze. 2026. \u201cThe Main Methodological Postulate of Pyrometry and the Need for Its Revision\u201d. Global Journal of Research in Engineering - A : Mechanical & Mechanics GJRE-A Volume 24 (GJRE Volume 24 Issue A1).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.