Cancer disease results from mutations leading to apoptosis failure and immune system dysfunction. Because one in two people in developed counties will be diagnosed with cancer in their lifetimes, cancer and metastasis prevention should be ahead of therapies. The immune system in cancer patients is compromised and can be fixed with a reboot. The major oncofetal protein -alpha-fetoprotein -can deliver toxins instead of nutrients to the immune suppressor cells and kill them. The death of myeloid suppressor cells unleashes the immune attack on cancer cells, cancer stem cells, and metastases. Injectable and oral formulations of alphafetoprotein with toxins provide an opportunity to prevent and treat the disease.
"The scientific man does not aim at an immediate result. He does not expect that his advanced ideas will be readily taken up. His work is like that of the planter—for the future. His duty is to lay the foundation for those who are to come and point the way."
## I. INTRODUCTION
Surgery, radiation, and chemotherapy can cure about half of cancer patients; nevertheless, in the United States alone, nearly 600,000 people died last year. The US cancer death rate has fallen $33\%$ since 1991, partly due to advances in treatment, early detection, and less smoking [1]. It is better to prevent cancer before than to cure the disease after. A healthy environment and lifestyle can prevent only $29\%$ of cancer-causing mutations [2], while everyone needs cancer prophylactics. Each day apoptosis and the immune system erase billions of mutants and expired cells. The immune system is confused in cancer patients. There is an opportunity to prevent early-stage cancer or metastases by rebooting the immune system with a unique delivery vehicle—alpha-fetoprotein (AFP) and toxins.
From the fertilized egg, trillions of cells grow through duplications. Stem cells are deposited on the way. When stimulated to increase, a stem cell is undergoing an "asymmetric division" [3]. The proliferating daughter cell continues to divide and proceed down the tissue hierarchy, from stem cell to progenitor cell, before becoming a fully differentiated mature tissue cell. Multiple types of stem cells have been identified in a wide range of tissue, sharing multipotency characteristics.
In the bone marrow, hematopoietic stem cells (HSC) exist undifferentiated. They are at the peak of a blood cell differentiation hierarchy. The white blood cell ratio is neutrophils $(70\%)$ lymphocytes $>$ monocytes $>$ eosinophils $>$ basophils. Myeloid-derived suppressor cells (MDSCs) are a small heterogeneous cell population of immature myeloid progenitors of granulocytes, macrophages, and dendritic cells (DCs) at different stages of differentiation generated from a common HSC [4].
MDSCs can leave the bone marrow and spread throughout the body, becoming immune response calmers during pregnancy, cancer, regeneration, stress, autoimmune and infectious diseases, obesity, age, etc.[5]. Besides the bone marrow, other sites generate MDSCs: the placenta and umbilical cord, the tumor site, and the spleen [6].
MDSCs exist in an undifferentiated state at the peak of the immune cell's hierarchy. They affect innate and adaptive immunity cells directly and indirectly. MDSCs inhibit natural killer (NK) cells, DCs, and T-cells, induce regulatory T cells (T regs) and modulate macrophages, etc. [7-9].
In 1862 Rudolf Virchow was the first to link the origin of cancers from otherwise normal cells correctly: "every cell arises from another cell." Indeed, $5\%$ of tumor-causing mutations are inherited, and tumor cells are activated later in life, while $66\%$ of tumor-causing mutations appear during duplications [2]. According to the hierarchical model, the tumor grows from a single cell. Like in embryogenesis, a core group of stem cells exists at the top of the tumor hierarchy, from which other more differentiated cells are formed. Descending from the undifferentiated cells to the most mature cells that comprise the bulk of the tumor mass. Cancer stem cells (CSCs) are cancer cells with characteristics associated with normal stem cells; specifically, give rise to all cell types found in a particular cancer sample. Approximately $73\%$ of current CSC surface markers appear on embryonic or adult stem cells and are rarely expressed on normal tissue cells. It is believed that the elimination of CSCs could eradicate whole cancer [10].
Pregnancy is a natural phenomenon that ensures the survival of the species. An embryo turns off a critical pathway required for the immune system to attack intruders. The suppression of the immune response in pregnancy is robust since the embryo cells have half the father's proteins that the mother's immune system should recognize as foreign. Moreover, even surrogate motherhood is possible, with no genetic relationship to the embryo. The immunology of pregnancy and cancer is similar [11]. Embryo and cancer cells exploit the mechanisms that allow them to grow despite the host's immune system attacks. During pregnancy, the mother's anti-embryo immune response is neutralized by the oncofetal proteins that re-appear during cancer development.
Some of the oncofetal proteins are AFP, AFP receptor (AFPR), human chorionic gonadotropin (HCG), carcinoembryonic antigen (CAE), and pregnancy-associated protein A. A few molecules regulate the immune tolerance of the mother - AFP, HCG, glycodelin, and pregnancy-specific $\beta 1$ -glycoprotein [12]. Cancers express oncofetal proteins at "a wrong time in a wrong place" to withstand immune system attacks.
Forty years ago, oncofetal proteins were used for cancer patients' vaccination. The placenta is the first organ that forms after conceiving—before any baby's organs even take shape. Immunotherapy with placental proteins has demonstrated a $77.1\%$ 5-year survival rate and a $65.4\%$ 10-year in 35 terminal patients [11]. (The handful of cancer immunotherapy drugs available today have demonstrated robust and durable results only in a minority of patients).
AFP is the major oncofetal protein secreted in early post-implantation embryos of mammals; the ability to synthesize AFP is restricted to the visceral endoderm cells around the embryonic region of the egg cylinder [13]. Later in development, AFP is produced by the yolk sac, liver, and gastrointestinal tract and penetrates the mother's blood (\<200 ng/ml) (Fig. 1).
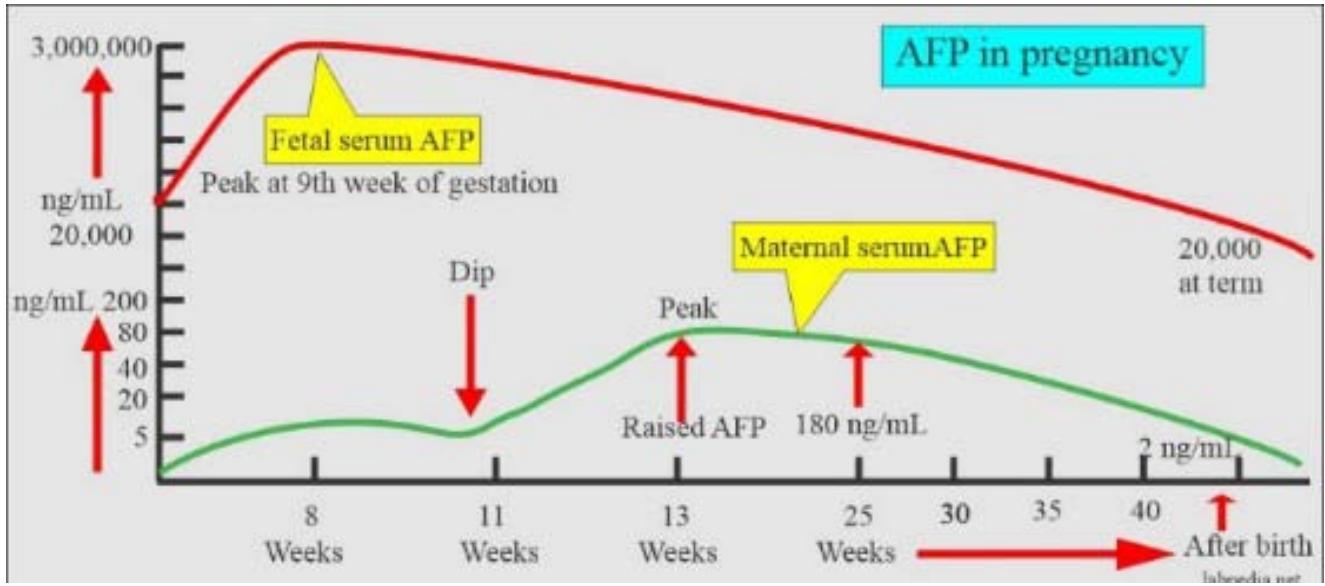 Figure 1: AFP generated by embryo cells penetrates the mother's blood.
AFP can be re-expressed in adult life in a few cancers: hepatomas, germ cell tumors, yolk sac tumors of the ovary, and gastrointestinal cancers. The AFP structure, functions, and clinical applications are well covered in the literature [14-18].
AFP and its peptides have immunomodulatory properties [19-21]. For example, AFP selectively induced a rapid downregulation of surface MHC class II antigens in their expression on human monocytes, thereby making embryo/tumor cells "invisible" to the immune system. MHC class II antigens on monocytes are the key molecules in antigen presentation. They differentiate between a self-cell and an "alien" embryo or cancer cell. AFP neither alters the expression of MHC I, CD4, CD18, CD45, and Fc receptors for IgG on the surface of monocytes/macrophages nor affects the functional maturation of the macrophages Fc receptors or the ability to express antibody-dependent cell-mediated cytolytic activity. AFP may, by reducing the antigen-presenting capacity of monocytes/ macrophages, function as an essential factor in maintaining a fetal allograft, as well as participate in the downregulation of the entire immune system in cancer [22].
Nevertheless, the primary immune regulatory impact is AFP ligands because AFP delivers dozens of molecules within 3-5 days of its half-life. Moreover, the AFP-binding monocytes play a fundamental role in regulating the immune response.
The AFP's primary function is nutrient delivery in a shuttle manner (like oxygen delivery by hemoglobin). AFP binds different ligands, delivers them to the AFPR-positive cells, and releases them inside the cell compartment with an acidic pH. The 69 kDa AFP can hide 1-2 molecules (\<2 kDa) in its hydrophobic cavity [23]. For example, docosahexaenoic acid (DHA) is not synthesized by the mother, who should take essential nutrients with food. AFP grabs DHA from albumin and transports it through the placenta [24]. Polyunsaturated fatty acids are necessary for many purposes, for example, the myelination of nerve fibers in the fast-growing embryo's brain.
Embryo cells and normal and malignant peripheral monocytes/macrophages use the autocrine AFP/AFPR system for nutrient supply (Fig. 2, A) [25].
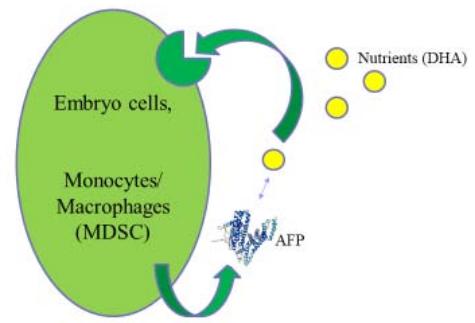 A
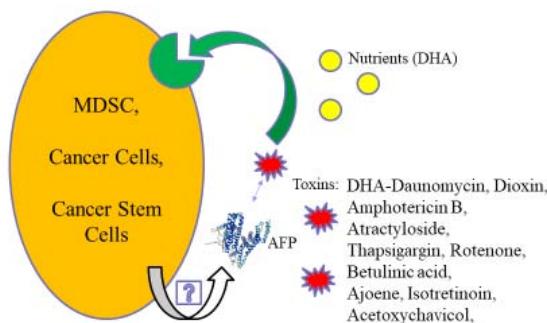 B Figure 2: AFP/AFPR shuttle delivery system. A: AFP delivers nutrients to AFPR-positive cells. B: AFP delivers toxins. Abbreviations: AFP – alpha-fetoprotein, DHA – docosahexaenoic acid, MDSC – myeloid-derived suppressor cell, AFP secretion is unknown.
Low-differentiated lymphocytes use AFPR during blast transformation [26]. Monocytes/macrophages have specific 62 and 65 kDa AFP-binding receptors, which are involved in the physiological regulation of the immune response [27].
Unlike AFP, many cancers express AFPR [28], which should be considered the tumor marker and oncofetal protein #1. The AFP gene-knockout rodent models have demonstrated that AFP is not obligatory for full-term delivery [29]. The AFPR gene and structure are unknown yet to perform the gene-knockout experiment. In any case, the AFP/AFPR duo is vital for the embryo (and cancer).
The fact that the mother is tolerable to the embryo for nine months demonstrates that from the very beginning, AFP (Fig. 1) delivers nutrients to MDSCs, "corrupting" them to generate a protective shield over an embryo. MDSCs suppress NK cells which are "spontaneous cytotoxic cells" involved in surveillance against tumor cells. NK cells attack "aliens," low differentiated embryos, stem, cancer cells, and CSCs [30, 31]. NK cells can erase cancer at the earlier stages and solve the urgent problem of metastases.
## II. INJECTABLE AFP-TOXINS
In cancer, MDSCs are "corrupted" also [32]. The effective way to ruin corruption is to eliminate the head. For this purpose, instead of DHA, AFP can deliver toxins to MDSCs (Fig. 2, B). The MDSCs death unleashes NK cells, and the whole immune system, enabling it to recognize and attack cancer the way it does other diseases. In addition, AFP-toxin kills AFPR-positive cancer cells and possibly CSCs (Fig. 2, B).
Like AFP-DHA, AFP complexes with hydrophobic/amphiphilic toxins hidden in the hydrophobic cavity are stable in the bloodstream. They release the toxin only inside the AFPR-positive cells.
After toxin unloading, AFP can work as a shuttle and deliver additional toxins. DHA-daunomycin conjugate binds AFP and inhibits tumor growth in AFP-producing mice [33]. Most cancers do not produce AFP, but $>80\%$ are AFPR-positive; hence, exogenous AFP with toxins kills them. Thus, the treatment with AFP and amphotericin B in the excess (1:60-100) for shuttling demonstrated a response in 6 out of 8 cancer patients and increased quality of life [34]. The cytokine storm-like reaction, sometimes observed during the AFP with amphotericin B infusions, preceded cancer cells' death, and it can indicate the consequences of MDSCs death [18]. The potent AFP-binding toxins are expected to provide even better than AFP-amphotericin B response in cancer treatments.
Cancer cells can activate an AFP/AFPR autocrine loop (Fig. 2, B) [35, 36]. MDSCs are progenitor cells with only a few duplication steps from stem and embryo cells, and, as well as CSCs; they should retain an AFP-mediated nutrient delivery system. These need research. At least, AFP is absorbed by MDSCs, stimulating their suppressive activity [37, 38].
AFP attracts MDSCs and T regs through AFP-binding C-C chemokine receptor type 5 (CCR5) [39-41]. These regulatory cells migrate, accumulate, and suppress the immune attack on cancer. Targeting CCR5 reboots immunosuppressive myeloid cells [42, 43].
AFP binds to the neonatal Fc receptor (FcRn) [44, 45]. The FcRn is found in MDSCs in pancreatic cancer monocytes. MDSCs and DCs are elevated in pancreatic cancer patients compared to non-cancer donors [46]. They can be targeted through AFPR and/or FcRn by AFP-toxin drugs.
AFPR, CCR5, and FcRn are valuable MDSCs markers, at least for a transitory period.
Many current drugs do not eliminate CSCs, which may be why many cancers regrow after treatment. Immunotherapy does not act directly on cancer but works on the immune system. Checkpoint inhibitors and CAR T-cells are too unsafe for early-stage cancer; complexity and cost also prevent their application. Dissemination of cancer cells from the primary tumor into distant body tissues and organs is the leading cause of death in cancer patients. While most clinical strategies aim to reduce or impede the growth of the primary tumor, no treatment to eradicate metastatic cancer exists at present [47]. The MDSCs- and CSCs-targeting drugs have a bright future as they are critical in tumor and metastasis prevention [48, 49].
More than 100 years ago, Paul Ehrlich proposed a "magic bullet" that kills cancer cells, sparing the healthy ones. Nevertheless, this approach did not elevate the survival rate of cancer patients. The additional target outside of cancer cells should be hit. This "magic target" is a myeloid suppressor cell. Combining "magic bullets" and the "magic target" approach can cure cancer.
"Magic bullet" can kill "magic target" MDSC. Paclitaxel hits both cancer cells and MDSCs [50], and AFP potentiates its direct cytotoxic and immunotherapy action [51]. Thapsigargin is a more potent toxin than paclitaxel. AFP-thapsigargin complex (ACT-902) depletes MDSCs and tumor-associated macrophages. In mice, chemotherapy using ACT-902 and AFP with paclitaxel demonstrated superior efficacy and safety compared to chemotherapy alone. ACT-902 has led to the complete regression of five out of six highly resistant to chemotherapy POP-92 xenografts by day seven of treatment with no further growth after this period in mice [52]. Or the AFP-maytansine conjugate combines both immunotherapy and targeted chemotherapy with undetectable bone marrow toxicity. It has shown $100\%$ survival with no tumor re-growth after in the mice models [53]. AFP-toxin conjugates might pave a new road to the cancer cure [54-58]. On the other hand, unlike complexes, artificial conjugates have the risk of immune response to themselves.
The neuroblastoma cells may re-express embryonal or fetal antigens, suggesting some reversion towards an earlier stage of differentiation, and they can incorporate AFP [59, 60]. AFP-maytansine conjugate can kill brain tumor cells and CSCs found in human brain tumors [61].
Glioblastoma is the most aggressive, malignant primary brain tumor in adults. Myeloid cells are critical regulators of immune and therapeutic responses to glioblastoma [62]. In the glioblastoma microenvironment, M-MDSCs represent the predominant subset [63]. M-MDSCs can be depleted by AFP-toxin conjugate [54].
It has been found that MDSCs account for approximately $30 - 50\%$ of the tumor mass in gliomas. MDSCs are increased following conventional chemotherapy treatments [64]. Targeting MDSCs in combination with other therapies has shown promising therapeutic effects in brain cancer [65], and AFP-toxin drugs can be one of these therapies.
## III. ORAL INSTEAD OF INJECTABLE
"Let food be thy medicine, and let medicine be thy food."
Hippocrates
MDSCs can be affected by ingredients from herbs and supplements. For example, withaferin A – a promising anti-cancer constituent of the Ayurvedic medicinal plant Withania somnifera-reduces MDSCs function [66]. Nevertheless, pregnant women should avoid Withania somnifera tonic as it may induce abortion at high doses [67]. In ancient Rome and Greece, women used silphium, an oral herbal contraceptive. This valuable herb is seen on a coin with a crab that once was a cancer disease name (Fig. 3).
 Figure 3: A coin of Magas of Cyrene c. 300-282/75 BC. Reverse: silphium and small crab symbols.
Was silphium used not only for pregnancy prevention but for cancer treatment too? There is no silphium in Nature anymore to check the hypothesis. Still, Artemisia absinthium, also used in Roman times for birth control, contains artemisinin that prevents early embryo implantation in animal models. Supposedly, AFP shuttles silphium ingredients or artemisinin to MDSCs, decreases their immunosuppressive activities, and leads to pregnancy or cancer prevention.
AFP wins the competition with excess albumin for binding embryo toxins such as diethylstilbestrol, dioxin, warfarin, etc., and can lead to pregnancy prevention or loss [18]. It can also be true for orally administrated thalidomide, miltefosine, etc. Toxins directly affect embryo cells, and AFP with toxins activate the mother's immune system like paclitaxel.
Traditional medicines and spices often contain anti-cancer agents, such as withaferin A, ajoene, acetoxychavicol, capsaicin, curcumin, quercetin, all-trans retinoic acid, sinigrin, artemisinin, astaxanthin, scutebarbatine A, etc. Small amounts of AFP naturally existing in the body potentiate their anti-cancer activity by the mechanism discussed earlier. To activate the immune system significantly, AFP-binding anti-cancer agents should be used together with exogenous AFP [68, 69].
Porcine AFP and betulinic acid (1:2) gavage inhibited mouse tumor growth. Tumor inhibition was potentiated by the excess of betulinic acid [70]. The beneficial effect of an extra amount of agent for tumor growth inhibition was supported by the experiment with the porcine AFP-ajeene (1:2) complex and ajoene in excess [18].
In suboptimal doses, the oral porcine AFP- attractyloside (1:2) complex has shown a response in six of twelve metastatic colorectal cancer patients [71]. Additional spices, herbs, or supplements having anticancer properties could potentiate the treatment. In another trial, a woman with stage IV ovarian cancer took elevated doses of the porcine AFP-attractyloside complex and survived over ten years [18].
AFP-toxin complex absorption from the gastrointestinal tract into the lymph nodes needs research. It can be like IgG-antigen complex absorption through the FcRn of the gut enterocytes [18].
AFP with AFP-binding toxins promises helpful in disease prevention, as they require low concentrations of AFP and toxins, like pregnancy prevention. Injectable recombinant AFP is safe in doses higher than in pregnant mothers' blood (0.3 - 0.5 $\mu$ g/mL) [72]. Porcine AFP can be taken orally in high doses safely.
## IV. CONCLUSION
AFP-toxin conjugates or AFP with AFP-binding toxins injections deplete MDSCs, reboot the immune system, and can prevent or treat cancer and metastases. As a shuttle delivery vehicle, AFP can potentiate the anti-cancer activity of the AFP-binding toxins or drugs. Oral administration of AFP with herbs or supplements with anti-cancer properties is possible. Like embryo toxins do not hurt the mother but prevent pregnancy, the simultaneous presence of AFP and AFP-binding toxins in the bloodstream can safely prevent early-stage cancers or metastases. Oral preparations do not need high AFP purity, and porcine AFP can be used instead of human protein. Taken once or twice a year course, AFP with AFP-binding toxins can reboot the immune system and prevent cancer and metastasis. No conflict of interests
Generating HTML Viewer...
References
70 Cites in Article
R Siegel,K Miller,N Wagle (2023). Cancer statistics.
C Tomasetti,B Vogelstein (2015). Variation in Cancer Risk among Tissues Can Be Explained by the Number of Stem Cell Divisions.
S Chhabra,B Booth (2021). agents, such as withaferin A, ajoene, acetoxychavicol, capsaicin, curcumin, quercetin, alltrans retinoic acid, sinigrin, artemisinin, astaxanthin, scutebarbatine A, etc. Small amounts of AFP naturally existing in the body potentiate their anti-cancer activity by the mechanism discussed earlier. To activate the immune system significantly, AFP-binding anti-cancer agents should be used together with exogenous.
V Bronte,S Brandau,S Chen,M Colombo,A Frey,T Greten (2016). Recommendations for myeloidderived suppressor cell nomenclature and characterization standards.
G Pawelec,C Verschoor,S (2019). Myeloid-Derived Suppressor Cells: Not Only in Tumor Immunity.
Nikoleta Bizymi,Andreas Matthaiou,Angelos Matheakakis,Ioanna Voulgari,Nikoletta Aresti,Konstantina Zavitsanou,Anastasios Karasachinidis,Irene Mavroudi,Charalampos Pontikoglou,Helen Papadaki (2022). New Perspectives on Myeloid-Derived Suppressor Cells and Their Emerging Role in Haematology.
Filippo Veglia,Emilio Sanseviero,Dmitry Gabrilovich (2021). Myeloid-derived suppressor cells in the era of increasing myeloid cell diversity.
Yoshiaki Nagatani,Yohei Funakoshi,Hirotaka Suto,Yoshinori Imamura,Masanori Toyoda,Naomi Kiyota,Kimihiro Yamashita,Hironobu Minami (2021). Immunosuppressive effects and mechanisms of three myeloid-derived suppressor cells subsets including monocytic-myeloid-derived suppressor cells, granulocytic-myeloid-derived suppressor cells, and immature-myeloid-derived suppressor cells.
Mohd Idris,R Mussa,A Ahmad,S Al-Hatamleh,Mai Hassan,R,Tengku Din,Tadaa (2022). The Effects of Tamoxifen on Tolerogenic Cells in Cancer.
Won-Tae Kim,Chun Ryu (2017). Cancer stem cell surface markers on normal stem cells.
V Govallo (1993). Immunology of Pregnancy and Cancer.
S Zamorina,Y Troynich,N Loginova,Y Charushina,K Shardina,V Timganova (2021). Pregnancy-Associated Proteins as a Tool in the Therapy of Autoimmune Diseases and Alloimmune Disorders (Review).
M Dziadek,E Adamson (1973). Localization and synthesis of alphafoetoprotein in post-implantation mouse embryos.
Gerald J. Mizejewski (1987). Alpha-fetoprotein: Immunomodulation in autoimmune diseases during pregnancy and puerperium stages.
V Chereshnev,Rodionov Syu,V Sherkasov (2004). Alpha-fetoprotein.
A Terentiev,N Moldogazieva (2013). Alpha-fetoprotein: a renaissance.
N Lakhi,M Moretti (2016). Alpha-Fetoprotein: Functions and Clinical Application.
V Pak (2021). Alpha-fetoprotein and Its Receptor in Fixing the Cancer Brakes.
Paul Munson,Juraj Adamik,Lisa Butterfield (2022). Immunomodulatory impact of α-fetoprotein.
Susanna Sologova,Sergey Zavadskiy,Innokenty Mokhosoev,Nurbubu Moldogazieva (2022). Short Linear Motifs Orchestrate Functioning of Human Proteins during Embryonic Development, Redox Regulation, and Cancer.
G Mizejewski (2022). Alpha-Fetoprotein as an Immunoregulator and Immune Response Modifier: Historical Background and Current Update.
Katrin Laan-Pütsep,Hans Wigzell,Paul Cotran,Magnus Gidlund (1991). Human α-fetoprotein (AFP) causes a selective down regulation of monocyte MHC class II molecules without altering other induced or noninduced monocyte markers or functions in monocytoid cell lines.
Alexander Terentiev,Nurbubu Moldogazieva,Olga Levtsova,Dmitry Maximenko,Denis Borozdenko,Konstantin Shaitan (2012). MODELING OF THREE DIMENSIONAL STRUCTURE OF HUMAN ALPHA-FETOPROTEIN COMPLEXED WITH DIETHYLSTILBESTROL: DOCKING AND MOLECULAR DYNAMICS SIMULATION STUDY.
J Hsia,H Deutsch (1987). An in Vitro Model of Placental Transfer of Polyunsaturated Fatty Acids: The Albumin-Alpha-Fetoprotein Exchange System.
C Esteban,J Trojan,A Macho,Z Mishal,C Lafarge-Frayssinet,Uriel (1993). Activation of an Alpha-Fetoprotein/Receptor Pathway in Human Normal and Malignant Peripheral Blood Mononuclear Cells.
J Torre,J Laborda,J Naval,N Darracq,M Calvo,Z Mishal,J Uriel (1989). Expression of alpha-fetoprotein receptors by human T-lymphocytes during blastic transformation.
Y Suzuki,C Zeng,E Alpert (1992). Isolation and partial characterization of a specific alpha-fetoprotein receptor on human monocytes..
Hebatallah Sedky,Soha Youssef,Doaa Gamal,Heba Houssein,Walaa Elsalakawy (2020). First report of the unique expression of RECAF (receptor for alfa feto-protein) in adult B-NHL/CLL patients.
Christelle De Mees,Jean-François Laes,Julie Bakker,Johan Smitz,Benoît Hennuy,Pascale Van Vooren,Philippe Gabant,Josiane Szpirer,Claude Szpirer (2006). Alpha-Fetoprotein Controls Female Fertility and Prenatal Development of the Gonadotropin-Releasing Hormone Pathway through an Antiestrogenic Action.
Anahid Jewett,Janko Kos,Kawaljit Kaur,Tahmineh Safaei,Christine Sutanto,Wuyang Chen,Paul Wong,Artin Namagerdi,Changge Fang,Yuman Fong,Meng-Wei Ko (2019). Natural Killer Cells: Diverse Functions in Tumor Immunity and Defects in Pre-neoplastic and Neoplastic Stages of Tumorigenesis.
Nicola Tumino,Francesca Besi,Stefania Martini,Anna Di Pace,Enrico Munari,Linda Quatrini,Andrea Pelosi,Piera Fiore,Giulia Fiscon,Paola Paci,Francesca Scordamaglia,Maria Covesnon,Giuseppe Bogina,Maria Mingari,Lorenzo Moretta,Paola Vacca (2022). Polymorphonuclear Myeloid-Derived Suppressor Cells Are Abundant in Peripheral Blood of Cancer Patients and Suppress Natural Killer Cell Anti-Tumor Activity.
Francesco De Sanctis,Annalisa Adamo,Stefania Canè,Stefano Ugel (2022). Targeting tumour-reprogrammed myeloid cells: the new battleground in cancer immunotherapy.
H Deutsch,T Tsukada,T Sasaki,H Hirai Cytotoxic Effects of Daunomycin-Fatty Acid Complexes on Rat Hepatoma Cells.
V Pak,N Pak,S Reshetnikov,S Nikonov,A Ogirenko (2001). Method of treatment of malignant neoplasms and complex preparation having antineoplastic activity for use in such treatment.
Juan Torres,Maurice Geuskens,Jose Uriel (1991). Receptor‐mediated endocytosis and recycling of alpha‐fetoprotein in human B‐lymphoma and T‐leukemia cells.
Cristina Esteban,Maurice Geuskens,José Uriel (1991). Activation of an alpha‐fetoprotein (AFP)/receptor autocrine loop in HT‐29 human colon carcinoma cells.
N Belyaev,Bogdanov Ayu,Savvulidi Phg (2008). The influence of alpha-fetoprotein on natural suppressor cell activity and Ehrlich carcinoma growth.
S Zamorina,K Shardina,V Timganova,M Bochkova,S Uzhviyuk,M Raev,V Chereshnev (2021). Effect of Alpha-Fetoprotein on Differentiation of Myeloid Supressor Cells.
Aurélie Atemezem,Elisabeth Mbemba,Renée Marfaing,Jenny Vaysse,Michel Pontet,Line Saffar,Nathalie Charnaux,Liliane Gattegno (2002). Human α-fetoprotein binds to primary macrophages.
Carlos Oliveira,Julie Oda,Roberta Losi Guembarovski,Karen Oliveira,Carolina Ariza,Jamil Neto,Bruna Banin Hirata,Maria Watanabe (2014). CC Chemokine Receptor 5: The Interface of Host Immunity and Cancer.
Elias Hawila,Hila Razon,Gizi Wildbaum,Carolin Blattner,Yair Sapir,Yuval Shaked,Viktor Umansky,Nathan Karin (2017). CCR5 Directs the Mobilization of CD11b+Gr1+Ly6Clow Polymorphonuclear Myeloid Cells from the Bone Marrow to the Blood to Support Tumor Development.
Yi Ban,Junhua Mai,Xin Li,Marisa Mitchell-Flack,Tuo Zhang,Lixing Zhang,Lotfi Chouchane,Mauro Ferrari,Haifa Shen,Xiaojing Ma (2017). Targeting Autocrine CCL5–CCR5 Axis Reprograms Immunosuppressive Myeloid Cells and Reinvigorates Antitumor Immunity.
Serena Zilio,Silvio Bicciato,Donald Weed,Paolo Serafini (2022). CCR1 and CCR5 mediate cancer-induced myelopoiesis and differentiation of myeloid cells in the tumor.
Michal Pyzik,Kine Sand,Jonathan Hubbard,Jan Andersen,Inger Sandlie,Richard Blumberg (2019). The Neonatal Fc Receptor (FcRn): A Misnomer?.
R Blumberg,S Baker,M Pyzik,A Gandhi (2020). Methods to manipulate alpha-fetoprotein (AFP).
Justin Thomas,Molly Torok,Kriti Agrawal,Timothy Pfau,Trang Vu,Justin Lyberger,Hsiaochi Chang,Alyssa Castillo,Min Chen,Bryan Remaily,Kyeongmin Kim,Zhiliang Xie,Mary Dillhoff,Samuel Kulp,Gregory Behbehani,Zobeida Cruz-Monserrate,Latha Ganesan,Dwight Owen,Mitch Phelps,Christopher Coss,Thomas Mace (2022). The Neonatal Fc Receptor Is Elevated in Monocyte-Derived Immune Cells in Pancreatic Cancer.
Shams Twafra,Chana Sokolik,Tal Sneh,Kolluru Srikanth,Tomer Meirson,Alessandro Genna,Jordan Chill,Hava Gil-Henn (2022). A novel Pyk2-derived peptide inhibits invadopodia-mediated breast cancer metastasis.
Andrew Law,Fatima Valdes-Mora,David Gallego-Ortega (2020). Myeloid-Derived Suppressor Cells as a Therapeutic Target for Cancer.
Alexander Cole,Adetunji Fayomi,Vivian Anyaeche,Shoumei Bai,Ronald Buckanovich (2020). An evolving paradigm of cancer stem cell hierarchies: therapeutic implications.
Alexandra Sevko,Tillmann Michels,Melissa Vrohlings,Ludmila Umansky,Philipp Beckhove,Masashi Kato,Galina Shurin,Michael Shurin,Viktor Umansky (1950). Antitumor Effect of Paclitaxel Is Mediated by Inhibition of Myeloid-Derived Suppressor Cells and Chronic Inflammation in the Spontaneous Melanoma Model.
L Kotra,C Paige,A Bello,I Sherman (2016). Drug complexes comprising alpha-fetoprotein.
Igor Sherman,Rebecca Boohaker,Karr Stinson,Patricia Griffin,Wendy Hill (2022). An alpha-fetoprotein-maytansine conjugate for the treatment of AFP receptor expressing tumors..
Nikolai Belyaev,Nurshat Abdolla,Yuliya Perfilyeva,Yekaterina Ostapchuk,Vladimir Krasnoshtanov,Aikyn Kali,Raikhan Tleulieva (2018). Daunorubicin conjugated with alpha-fetoprotein selectively eliminates myeloid-derived suppressor cells (MDSCs) and inhibits experimental tumor growth.
K Shardina,S Zamorina,M Raev,V Chereshnev (2020). APPLICATION OF ALPHA-FETOPROTEIN IN IMMUNOPHARMACOLOGY – BACKGROUND.
Bo Lin,Xu Dong,Qiujiao Wang,Wei Li,Mingyue Zhu,Mengsen Li (2021). AFP-Inhibiting Fragments for Drug Delivery: The Promise and Challenges of Targeting Therapeutics to Cancers.
O Gulevskyy (2021). URRENT CONCEPT OF THE STRUCTURAL AND FUNCTIONAL PROPERTIES OF ALFA-FETOPROTEIN AND THE POSSIBILITIES OF ITS CLINICAL APPLICATION.
Mariya Sokol,Nikita Yabbarov,Mariia Mollaeva,Margarita Chirkina,Murad Mollaev,Artur Zabolotsky,Sergey Kuznetsov,Elena Nikolskaya (2022). Alpha-Fetoprotein Mediated Targeting of Polymeric Nanoparticles to Treat Solid Tumors.
M Hajeri-Germond,J Naval,J Trojan,J Uriel (1985). The uptake of alpha-foetoprotein by C-1300 mouse neuroblastoma cells.
M Hajeri-Germond,J Naval,A Trojan,L Jay,T Castillo,A Ly,P Penagos (2017). Brainfrom development to neoplasia, and gene therapy solution.
S Singh,I Clarke,M Terasaki,V Bonn,C Hawkins,J Squire,P Dirks (2003). Identification of a cancer stem cell in human brain tumors.
De Leo,A Ugolini,A Veglia,F (2020). Myeloid Cells in Glioblastoma Microenvironment.
Salemizadeh Parizi,M,Salemizadeh Parizi,F Abdolhosseini,S Vanaei,S Manzouri,A Ebrahimzadeh,F (2021). Myeloid-derived suppressor cells (MDSCs) in brain cancer: challenges and therapeutic strategies.
Senthilnath Lakshmanachetty,Joselyn Cruz-Cruz,Eric Hoffmeyer,Allison Cole,Siddhartha Mitra (2021). New Insights into the Multifaceted Role of Myeloid-Derived Suppressor Cells (MDSCs) in High-Grade Gliomas: From Metabolic Reprograming, Immunosuppression, and Therapeutic Resistance to Current Strategies for Targeting MDSCs.
Pratima Sinha,Suzanne Ostrand-Rosenberg (2013). Withaferin A, a potent and abundant component of Withania somnifera root extract, reduces myeloid-derived suppressor cell function (P2103).
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Vladimir N. Pak. 2026. \u201cA Cancer Prevention and Treatment Opportunity\u201d. Global Journal of Medical Research - F: Diseases GJMR-F Volume 23 (GJMR Volume 23 Issue F2): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Cancer disease results from mutations leading to apoptosis failure and immune system dysfunction. Because one in two people in developed counties will be diagnosed with cancer in their lifetimes, cancer and metastasis prevention should be ahead of therapies. The immune system in cancer patients is compromised and can be fixed with a reboot. The major oncofetal protein -alpha-fetoprotein -can deliver toxins instead of nutrients to the immune suppressor cells and kill them. The death of myeloid suppressor cells unleashes the immune attack on cancer cells, cancer stem cells, and metastases. Injectable and oral formulations of alphafetoprotein with toxins provide an opportunity to prevent and treat the disease.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.