## I. INTRODUCTION
About seven years ago, the author published a paper in GJSFR concerning the true nature of dark matter (DM), the gravitational source (mass) existing in the universe whose true nature is unknown [1]. The author showed that energy levels far lower than those derived based on quantum mechanics exist in the hydrogen atom [2]. Electrons at these ultra-low energy levels have negative mass, and exist near the atomic nucleus (proton).
The author has given the name "dark hydrogen atom (DHA)" to this unknown material composed of an electron with negative mass and a proton with positive mass. The name "dark matter" is used for matter consisting of atomic nuclei with positive mass, and multiple electrons with negative mass. DHAs have almost the same mass as ordinary hydrogen atoms, but are extremely small compared to hydrogen atoms. Therefore, they can attain a state of far higher density than ordinary matter. As a result, DHAs can serve as an enormous gravitational source.
Previously, the author has published several papers on DM, but the claims of the author have attracted almost no notice [3][4][5]. However, in the last seven years, the author has acquired new knowledge relating to DM. Thus, by incorporating the latest results into this paper, and paper [1] will be rewritten in a more compelling form.
## II. THE TRUE NATURE OF POTENTIAL ENERGY OF THE HYDROGEN ATOM
According to the famous virial theorem, if $K$ is taken to be the kinetic energy of the entire system, and $V$ is taken to be the potential energy of the entire system, then the following relation holds between $K$ and $V$:
$$
\left\langle K \right\rangle = - \frac {1}{2} \left\langle V \right\rangle . \tag {1}
$$
The time average of $K$ is equal to $-1/2$ the time average of $V$. Also, the sum of the time average $K$ of the kinetic energy of the entire system and the time average of the total mechanical energy $E$ of the entire system becomes 0. That is,
$$
\left\langle K \right\rangle + \left\langle E \right\rangle = 0. \tag {2}
$$
Next, if Eqs. (1) and (2) are combined, the result is as follows:
$$
\left\langle E \right\rangle = - \left\langle K \right\rangle = \frac {1}{2} \left\langle V \right\rangle . \tag {3}
$$
Now, consider the case where an electron at rest in an isolated system in free space absorbs energy and begins to move. Assume that the mass of the electron has increased to $m$ due to absorption of that energy.
Einstein and Sommerfeld defined the relativistic kinetic energy $K_{\mathrm{re}}$ as follows [6].
$$
K _ {\mathrm {r e}} = m c ^ {2} - m _ {0} c ^ {2}. \tag {4}
$$
Here, $m_0c^2$ is the rest mass energy of the body. And $mc^2$ is the relativistic energy.
The "re" subscript of $K_{\mathrm{re}}$ stands for "relativistic."
If this object is an electron, then Eq. (4) can be written as follows.
$$
K _ {\mathrm {r e}} = m c ^ {2} - m _ {\mathrm {e}} c ^ {2}. \tag {5}
$$
Here, $m_{\mathrm{e}}c^{2}$ is the rest mass energy of the electron.
Eq. (5) holds if the energy absorbed by the electron is all converted into kinetic energy of the electron. This can be regarded as a matter of course, but in the natural world the contrary phenomenon also exists. There are also cases where an electron acquires kinetic energy by releasing energy.
Next, consider the case where an electron at rest in an isolated system in free space is attracted by the electrostatic attraction of the proton (hydrogen atom nucleus), and forms a hydrogen atom. The electron at rest has a rest mass energy of $m_{\mathrm{e}}c^{2}$. When this electron is taken into the region of the hydrogen atom, it acquires an amount of kinetic energy equivalent to the emitted photon.
Both energy sources must satisfy the law of energy conservation. The energy source here has been thought to be potential energy. However, we did not deeply pursue the true nature of this potential energy. Potential energy is a name, but there is no corresponding real entity.
The only energy an electron has when at rest is rest mass energy. There is no possible source for supplying the photon emitted by the electron, and the acquired kinetic energy, aside from the rest mass energy of the electron.
Thus, the author regarded the decrease in rest mass energy of the electron as corresponding to the potential energy of the electron, and expressed this as follows [7].
$$
V (r) = - \Delta m _ {\mathrm {e}} c ^ {2}. \tag {6}
$$
If potential energy is defined in this way, then the reason why the electron is not absorbed into the atomic nucleus can be explained without relying on quantum mechanics.
It has been thought that the stability of the atom cannot be explained in classical physics. According to Maxwell's electromagnetism, an electron attracted by a proton (hydrogen atom nucleus) is captured instantly by the atomic nucleus while emitting electromagnetic waves.
This problem was solved by Bohr. He explained that the energy levels of the hydrogen atom are discontinuous due to the adoption of the quantum condition [8]. At the same time, he pointed out that there is a minimum value to the energy levels of the hydrogen atom, and explained the reason why the electron is not absorbed into the atomic nucleus (proton). However, the viewpoint of this paper differs from Bohr's explanation.
The potential energy of the hydrogen atom is given by the following formula.
$$
V (r) = - \frac {1}{4 \pi \varepsilon_ {0}} \frac {e ^ {2}}{r} = - m _ {\mathrm {e}} c ^ {2} \frac {r _ {\mathrm {e}}}{r}. \tag {7}
$$
Here, $r_{\mathrm{e}}$ is the following classical electron radius.
$$
r _ {\mathrm {e}} = \frac {e ^ {2}}{4 \pi \varepsilon_ {0} m _ {\mathrm {e}} c ^ {2}}. \tag {8}
$$
According to Eq. (7), the rest mass energy of the electron is exhausted by $r = r_{\mathrm{e}}$
Therefore, the formula for potential energy is applicable in the following region.
$$
V (r) = - m _ {\mathrm {e}} c ^ {2} \frac {r _ {\mathrm {e}}}{r}, \tag {9}
$$
No rest mass energy to emit another photon remains in an electron which has approached the atomic nucleus up to the position $r = r_{\mathrm{e}}$
In existing theory, this is explained as $V(r) = -\infty$ when $r \to 0$, but this viewpoint must be reexamined [9].
The proton radius is thought to be about $r_{\mathrm{e}} / 4$, and therefore based on classical considerations alone, it is possible to explain the reason why the electron is not absorbed into the atomic nucleus. That is, there is the following lower limit on the energy levels of the hydrogen atom.
$$
E = - K = \frac {1}{2} V (r) = - \frac {m _ {\mathrm {e}} c ^ {2}}{2}. \tag {10}
$$
However, this paper does not claim that the hydrogen atom has energy levels like those in Eq. (10).
Incidentally, Bohr derived the following formula as the energy levels of the hydrogen atom.
$$
E_{\mathrm{BO},n} = -\frac{1}{2} \left(\frac{1}{4\pi\varepsilon_{0}}\right)^{2} \frac{m_{\mathrm{e}} e^{4}}{\hbar^{2}} \cdot \frac{1}{n^{2}} = -\frac{\alpha^{2} m_{\mathrm{e}} c^{2}}{2 n^{2}}, \quad n = 1, 2, \dots . \tag{11}
$$
Here, $\alpha$ is the following fine-structure constant.
$$
\alpha = \frac {e ^ {2}}{4 \pi \varepsilon_ {0} \hbar c} = 7. 2 9 7 3 5 2 5 6 6 4 \times 1 0 ^ {- 3}. \tag {12}
$$
The following value is obtained by taking the ratio of Eqs. (10) and (11).
$$
\frac {E _ {\mathrm {B O} , 1}}{E} = \alpha^ {2} \approx 5. 3 2 5 \times 1 0 ^ {- 5}. \tag {13}
$$
It should be recognized that $E_{\mathrm{BO,1}}$ is an energy far higher than the lower limit value that is classically predictable.
## III. ENERGY-MOMENTUM RELATIONSHIP APPLICABLE TO THE ELECTRON IN A HYDROGEN ATOM
Einstein's energy-momentum relationship does not hold in the hydrogen atom, where potential energy is present. However, physicists have not treated this as much of a problem. This is because physicists have thought that the theory describing the behavior of the electron in the hydrogen atom is quantum mechanics rather than the special theory of relativity (STR).
However, Dirac's relativistic wave equation is derived by taking Einstein's relationship as a departure point.
The author will first look for an energy-momentum relationship applicable to the electron in a hydrogen atom, and then later consider that a relativistic wave equation satisfying that relationship should be derived. The author actually derived a relativistic wave equation to replace the Dirac equation [10]. If one wishes to discuss electron spin, that equation must be solved.
This paper first points out physicists' mistakes regarding electrons in the hydrogen atom.
In discussions at the classical quantum theory level, many physicists believe that the mass of the electron in motion within the hydrogen atom increases when the electron's velocity increases, as predicted by the STR.
However, this is completely mistaken. When an electron is taken into a hydrogen atom, the mass of the electron decreases. If $m_{n}$ is the relativistic mass of an electron whose principal quantum number is in the state $n$, then $m_{n} < m_{\mathrm{e}}$.
Attention must be paid to the fact that, inside the hydrogen atom, the relativistic mass of the electron $m_{n}$ is smaller than the rest mass $m_{e}$.
Care must be given to the fact that the relativistic kinetic energy of the electron in a hydrogen atom, $K_{\mathrm{re,n}}$ is given not by $m_{n}c^{2} - m_{\mathrm{e}}c^{2}$ but by $m_{e}c^{2} - m_{n}c^{2}$ [11]. That is,
$$
K_{\mathrm{re},\mathrm{n}} \neq m_{n} c^{2} - m_{\mathrm{e}} c^{2}.
$$
$$
K_{\mathrm{re,n}} = -E_{\mathrm{re,n}} = m_{\mathrm{e}} c^{2} - m_{n} c^{2}.
$$
Physicists can regard $K_{\mathrm{re,n}}$ as completely mistaken. In the discussion in this paper, Eqs. (6) and (15) are decisively important. If Eqs. (6) and (15) are accepted, then the formulas derived later can be easily understood.
According to the STR, the following relation holds between the energy and momentum of a body moving in free space [12].
$$
(m c ^ {2}) ^ {2} = (m _ {0} c ^ {2}) ^ {2} + c ^ {2} p ^ {2}. \tag {16}
$$
The relationship of Einstein holds in an isolated system in free space. If the object of concern is an electron, its rest mass energy becomes $m_{\mathrm{e}}c^{2}$.
The author previously derived an energy-momentum relationship applicable inside the hydrogen atom using three methods. First, this relationship was derived mathematically [13].
With the following method, this relation was derived through considerations using an ellipse [14]. This is a wonderful derivation method, but it is hard to solve. This paper introduces a third method which is comparatively easy to understand [15].
Now, Eq. (16) is rewritten as follows.
$$
\left(m c ^ {2}\right) ^ {2} = m _ {0} ^ {2} c ^ {4} + \left(m ^ {2} c ^ {4} - m _ {0} ^ {2} c ^ {4}\right) = \left(m _ {0} c ^ {2}\right) ^ {2} + c ^ {2} p ^ {2}. \tag {17}
$$
Comparing Eqs. (16) and (17), the relativistic momentum $p_{\mathrm{re}}$ can be defined as follows.
$$
p _ {\mathrm {r e}} ^ {2} = m ^ {2} c ^ {2} - m _ {0} ^ {2} c ^ {2}. \tag {18}
$$
Hence,
$$
p _ {\mathrm {r e}} ^ {2} = \left(m + m _ {0}\right) \left(m c ^ {2} - m _ {0} c ^ {2}\right). \tag {19}
$$
The following relation holds due to Eqs. (4) and (19).
$$
K _ {\mathrm {r e}} = \frac {p _ {\mathrm {r e}} ^ {2}}{m + m _ {0}}. \tag {20}
$$
In classical physics, the following relation holds between momentum $p$ and kinetic energy $K$.
$$
K = \frac {1}{2} m _ {0} v ^ {2} = \frac {p ^ {2}}{2 m _ {0}}. \tag {21}
$$
The relativistic kinetic energy of an electron in a hydrogen atom is defined as follows by referring to Eq. (20).
$$
K_{\mathrm{re},\mathrm{n}} = \frac{p_{\mathrm{re},\mathrm{n}}^{2}}{m_{\mathrm{e}} + m_{\mathrm{n}}}. \tag{22}
$$
Here, $m_{n}c^{2}$ is the relativistic energy of the electron when the principal quantum number is in the state $n$. Also, $p_{\mathrm{re,n}}$ indicates the relativistic momentum of the electron.
This paper defines $E_{\mathrm{re,n}}$ as the relativistic energy levels of the hydrogen atom. (The quantum number used here is just the principal quantum number. Therefore, $E_{\mathrm{re,n}}$ is not a formula which predicts all the relativistic energy levels of the hydrogen atom.)
However, the term "relativistic" used here does not mean based on the STR. It means that the expression takes into account the fact that the mass of the electron varies due to velocity.
According to the STR, the electron's mass increases when its velocity increases. However, inside the hydrogen atom, the mass of the electron decreases when the velocity of the electron increases.
The following formula can be derived from Eqs. (15) and (22).
$$
\frac {p _ {\mathrm {r e} , n} ^ {2}}{m _ {\mathrm {e}} + m _ {n}} = m _ {\mathrm {e}} c ^ {2} - m _ {n} c ^ {2}. \tag {23}
$$
Rearranging this, the following relationship can be derived.
$$
\left(m _ {n} c ^ {2}\right) ^ {2} + c ^ {2} p _ {\mathrm {r e}, n} ^ {2} = \left(m _ {\mathrm {e}} c ^ {2}\right) ^ {2}. \tag {24}
$$
Equation (24) is the energy-momentum relationship applicable to the electron in a hydrogen atom.
Here, we will reconfirm that, in Eqs. (16) and (24), the energy of an electron initially at rest is $m_{\mathrm{e}}c^{2}$. (This paper does not discuss the issues of whether an electron can be completely at rest.) Also, it will be confirmed, regarding the fact that Eq. (24) is applicable in the hydrogen atom, that $r_{\mathrm{e}} \leq r$, just like the scope of application of the formula for potential energy. There are limits on the application of Eq. (24). It is not the case that Eq. (24) is applicable in all regions within the hydrogen atom.
## IV. SOLUSION OF EQUATION (24)
In the past, Dirac derived the following negative solution from Eq. (16).
$$
E = \pm m c ^ {2} = \pm m _ {0} c ^ {2} \left(1 - \frac {v ^ {2}}{c ^ {2}}\right) ^ {- 1 / 2}. \tag {25}
$$
If the same logic is applied to Eq. (24), then the following formula can be derived.
$$
E_{\mathrm{ab},n} = \pm m_{n} c^{2} = \pm m_{\mathrm{e}} c^{2} \left(1 + \frac{v_{n}^{2}}{c^{2}}\right)^{-1/2}.
$$
$E_{\mathrm{ab},n}$ defined here are the energy levels describing the energy possessed by the electron on an absolute scale.
However, Eq. (26) does not incorporate the discontinuity peculiar to the micro world. Therefore, Eq. (26) must be rewritten into a relationship where energy is discontinuous.
Thus, the author derived discontinuous energy levels using the following relationship [16](Appendix).
$$
\frac{v_{n}}{c} = \frac{\alpha}{n}.
$$
Now, using the relationship in Eq. (27),
$$
E _ {\mathrm {a b}, n} ^ {\pm} = \pm m _ {n} c ^ {2} = \pm m _ {\mathrm {e}} c ^ {2} \left(1 + \frac {\alpha^ {2}}{n ^ {2}}\right) ^ {- 1 / 2} \tag {28a}
$$
$$
= \pm m_{\mathrm{e}} c^{2} \left(\frac{n^{2}}{n^{2} + \alpha^{2}}\right)^{1/2}.
$$
$E_{\mathrm{re},n}$ are the energy levels when $m_{\mathrm{e}}c^{2}$ is taken to be the energy of an electron at rest at a position infinitely far from a proton. The relationship of $E_{\mathrm{ab},n}$ and $E_{\mathrm{re},n}$ is as follows.
$$
E _ {\mathrm {r e}, n} = E _ {\mathrm {a b}, n} - m _ {\mathrm {e}} c ^ {2} = m _ {n} c ^ {2} - m _ {\mathrm {e}} c ^ {2} < 0. \tag {29}
$$
Also, the relationship of these energies can be illustrated as follows (see Fig.1).
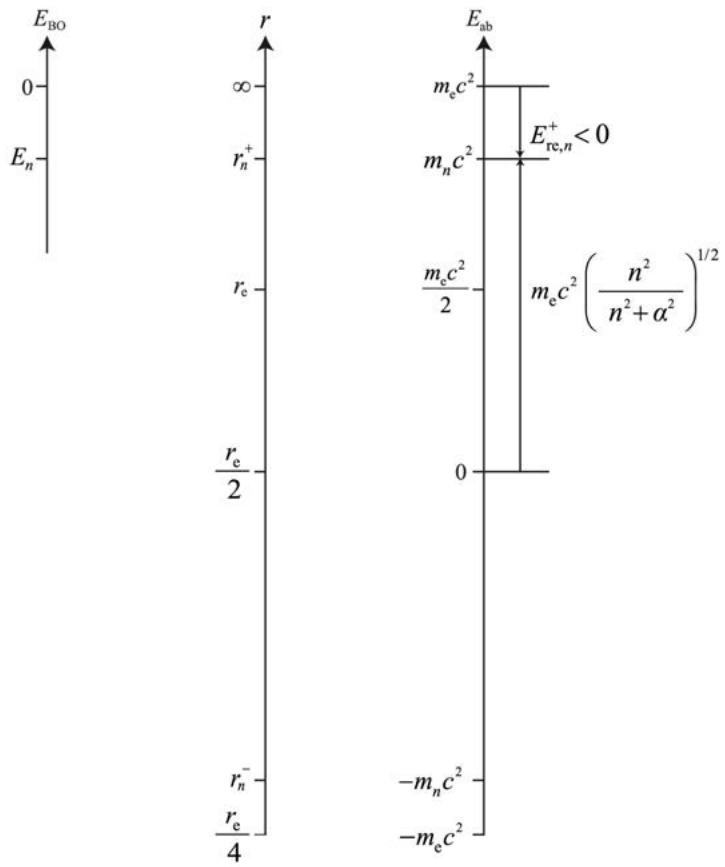 Fig. 1: Relationship of $E_{\mathrm{re},n}$ and $E_{\mathrm{ab},n}$ defined in Eq. (29). $E_{\mathrm{re},n}$ corresponds to the decrease in rest mass energy of the electron, and $E_{\mathrm{ab},n}$ ( $m_n c^2$ ) corresponds to the remaining part.
Using the definition in Eq. (29), $E_{\mathrm{re},n}$ is as follows.
$$
\begin{array}{l} E _ {\mathrm{r e}, n} ^ {+} = m _ {\mathrm{e}} c ^ {2} \left(\frac{n ^ {2}}{n ^ {2} + \alpha^ {2}}\right) ^ {1 / 2} - m _ {\mathrm{e}} c ^ {2} \\= m _ {\mathrm{e}} c ^ {2} \left[ \left(\frac{n ^ {2}}{n ^ {2} + \alpha^ {2}}\right) ^ {1 / 2} - 1 \right]. \tag{30} \\\end{array}
$$
$E_{\mathrm{re},n}^{+}$ are the energy levels of an ordinary hydrogen atom, and thus $E_{\mathrm{re},n}$ are omitted unless particularly necessary.
To simplify the discussion in this paper, the only quantum number addressed is $n$.
Next, when the part of Eq. (30) in parentheses is expressed as a Taylor expansion,
$$
\begin{array}{l} E _ {\mathrm {r e}, n} \approx m _ {\mathrm {e}} c ^ {2} \left[ \left(1 - \frac {\alpha^ {2}}{2 n ^ {2}} + \frac {3 \alpha^ {4}}{8 n ^ {4}} - \frac {5 \alpha^ {6}}{1 6 n ^ {6}}\right) - 1 \right] \\\approx - \frac {\alpha^ {2} m _ {\mathrm {e}} c ^ {2}}{2 n ^ {2}}. \tag {31} \\\end{array}
$$
This shows that Eq. (11) for the energy levels of the hydrogen atom derived by Bohr is an approximation of Eq. (30).
Now, the total mechanical energy of the hydrogen atom is given by the following formula.
$$
E _ {\mathrm {r e}, n} = K _ {\mathrm {r e}, n} + V \left(r _ {n}\right) = - K _ {\mathrm {r e}, n}. \tag {32}
$$
Also, if the formula for potential energy is used, then $E_{\mathrm{re},n}$ can be written as follows.
$$
E_{\mathrm{re},n} = \frac{1}{2} V\left(r_{n}\right) = -\frac{1}{2} \frac{1}{4\pi\varepsilon_{0}} \frac{e^{2}}{r_{n}} = -\frac{1}{2} m_{\mathrm{e}} c^{2} \frac{r_{\mathrm{e}}}{r_{n}} = - m_{\mathrm{e}} c^{2} \left(\frac{r_{\mathrm{e}} / 2}{r_{n}}\right). \tag{33}
$$
From Eq. (33), $m_{n}c^{2}$ is:
$$
m _ {n} c ^ {2} = E _ {\mathrm {a b}, n} = m _ {\mathrm {e}} c ^ {2} + E _ {\mathrm {r e}, n} = m _ {\mathrm {e}} c ^ {2} - m _ {\mathrm {e}} c ^ {2} \left(\frac {r _ {\mathrm {e}} / 2}{r _ {n}}\right) = m _ {\mathrm {e}} c ^ {2} \left(\frac {r _ {n} - r _ {\mathrm {e}} / 2}{r _ {n}}\right). \tag {34}
$$
Her the following energy is obtained if $r_{\mathrm{e}} / 2$ and $r_{\mathrm{e}} / 4$ are substituted for $r_{n}$ in Eq. (34).
When $r = \frac{r_{\mathrm{e}}}{2}$, $E_{\mathrm{ab}} = 0$ and when $r = \frac{r_{\mathrm{e}}}{4}$, $E_{\mathrm{ab}} = -m_{\mathrm{e}}c^{2}$ (35)
How should we think about emission of a photon by an electron that has consumed all its rest mass energy?
In another paper, the author proposed a method of solving that problem. According to the author's hypothesis, for an electron in the state $E_{\mathrm{ab}} = 0$ the photon energy and negative energy cancel out. The absolute amount of the respective energies cannot be definitely determined. However, if it is assumed that the electron approaches to a distance of $r = r_{\mathrm{e}} / 4$ from the center of the atomic nucleus, then the state with $E_{\mathrm{ab}} = 0$ can be regarded as the state where the photon energy of $m_{\mathrm{e}}c^{2}$ and the negative energy of $-m_{\mathrm{e}}c^{2}$ have canceled each other out. This negative energy can be thought of as the energy the electron latently possesses, and it is predicted that this energy cannot be removed from the electron.
Using this hypothesis, the energy can have a negative mass due to emission of a photon. More detailed explanation will be left to another paper [14].
## V. ORBITAL RADIUS OF AN ELECTRON IN A HYDROGEN ATOM
The following equation holds due to Eqs. (28b) and (34).
$$
\frac {n ^ {2}}{n ^ {2} + \alpha^ {2}} = \left(\frac {r _ {n} - r _ {\mathrm {e}} / 2}{r _ {n}}\right) ^ {2}. \tag {36}
$$
From this, the following quadratic equation is obtained.
$$
r _ {n} ^ {2} - \left(\frac {n ^ {2} + \alpha^ {2}}{\alpha^ {2}}\right) r _ {\mathrm {e}} r _ {n} + \left(\frac {n ^ {2} + \alpha^ {2}}{\alpha^ {2}}\right) \frac {r _ {\mathrm {e}} ^ {2}}{4} = 0. \tag {37}
$$
If this equation is solved for $r_n$,
$$
r _ {n} ^ {\pm} = \frac {r _ {\mathrm {e}}}{2} \left(1 + \frac {n ^ {2}}{\alpha^ {2}}\right) \left[ 1 \pm \left(1 + \frac {\alpha^ {2}}{n ^ {2}}\right) ^ {- 1 / 2} \right]. \tag {38}
$$
Next, if the electron orbital radii corresponding to the energy levels in Eq. (28b) are taken to be, respectively, $r_n^+$ and $r_n^-$,
$$
r _ {n} ^ {+} = \frac {r _ {\mathrm {e}}}{2} \frac {\left(n ^ {2} + \alpha^ {2}\right) ^ {1 / 2}}{\left(n ^ {2} + \alpha^ {2}\right) ^ {1 / 2} - n}. \tag {39}
$$
$$
r _ {n} ^ {-} = \frac{r _ {\mathrm{e}}}{2} \frac{\left(n ^ {2} + \alpha^ {2}\right) ^ {1 / 2}}{\left(n ^ {2} + \alpha^ {2}\right) ^ {1 / 2} + n}.
$$
Also, Eqs. (39) and (40) can be written as follows [3].
$$
r _ {n} ^ {+} = \frac {r _ {\mathrm {e}}}{2} \left[ 1 + \frac {n}{\left(n ^ {2} + \alpha^ {2}\right) ^ {1 / 2} - n} \right]. \tag {41}
$$
$$
r _ {n} ^ {-} = \frac {r _ {\mathrm {e}}}{2} \left[ 1 - \frac {n}{\left(n ^ {2} + a ^ {2}\right) ^ {1 / 2} + n} \right]. \tag {42}
$$
In Eq. (42), the electron approaches toward $r_{\mathrm{e}} / 4$ as $n$ increases. Therefore,
$$
\frac {r _ {\mathrm {e}}}{4} < r _ {n} ^ {-} \leq \frac {r _ {\mathrm {e}}}{2}. \tag {43}
$$
$$
\frac {r _ {\mathrm {e}}}{2} \leq r _ {n} ^ {+}. \tag {44}
$$
In this paper, $r_n^+$ is called the orbital radius, as is customary. However, a picture of the motion of the electron cannot be drawn, even if that motion is discussed at the level of classical quantum theory. The electron in a hydrogen atom is not in orbital motion around the atomic nucleus. The domain of the ordinary hydrogen atom that we all know starts from $r = r_{\mathrm{e}} / 2(E_{\mathrm{ab}} = 0)$ (The equality sign holds in Eqs. (43) and (44) when $n = 0$ ).
With the aid of quantum mechanics, $r_n^+$ and $r_n^-$ can be regarded as the locations (positions) where presence of the electron has maximum probability.
The next compares the orbital radii of an electron in a hydrogen atom $r_n^+$ and the orbital radii of an electron with a negative mass $r_n^-$.
The following ratio is obtained from Eqs. (39) and (40).
$$
\frac {r _ {n} ^ {-}}{r _ {n} ^ {+}} = \frac {\left(n ^ {2} + \alpha^ {2}\right) ^ {1 / 2} - n}{\left(n ^ {2} + \alpha^ {2}\right) ^ {1 / 2} + n}. \tag {45}
$$
Here, if we set $n = 1$
$$
\frac {r _ {1} ^ {-}}{r _ {1} ^ {+}} = \frac {\left(1 + \alpha^ {2}\right) ^ {1 / 2} - 1}{\left(1 + \alpha^ {2}\right) ^ {1 / 2} + 1} = 1. 3 3 1 2 4 8 4 1 6 8 \times 1 0 ^ {- 5} \approx \frac {1}{7 5 1 2 0}. \tag {46}
$$
The author pointed out that an electron with negative mass forming DHA exists near the atomic nucleus (proton) [4][17].
## VI. EVIDENCE OF THE EXISTENCE OF ULTRA-LOW ENERGY LEVELS
Is there an experiment demonstrating the existence of electrons at ultra-low energy levels? The author believes that the experiment of triplet production demonstrates the author's hypothesis.
This is a phenomenon where two electrons and one positron are produced from a gamma ray of energy $4m_{\mathrm{e}}c^{2}$ (2.044MeV). The author has pointed out that one of the electrons produced here is an electron with the negative energy which formed the DHA. However, it was difficult to determine which was better, the author's interpretation or the existing explanation. The author has previously discussed this problem a number of times, so that will not be repeated here. Please refer to the author's other papers [18].
Recently, the author has discovered a new formula which gives the energy levels of the hydrogen atom [19]. The formula previously derived by the author was as follows.
$$
E _ {\mathrm{re},n} ^ {+} = m _ {n} c ^ {2} - m _ {\mathrm{e}} c ^ {2} = m _ {\mathrm{e}} c ^ {2} \left[ \left(\frac{n ^ {2}}{n ^ {2} + \alpha^ {2}}\right) ^ {1 / 2} - 1 \right]. \tag{47}
$$
However, in recent papers, the author has shown that the energy levels of the hydrogen atom can also be described with the following formula.
$$
E_{\mathrm{re},n}^{+} = -K_{\mathrm{re},n} = -c p_{\mathrm{re},n} \left[ \frac{\left(n^{2} + \alpha^{2}\right)^{1/2} - n}{\left(n^{2} + \alpha^{2}\right)^{1/2} + n} \right]^{1/2}. \tag{48}
$$
Taking Eqs. (39) and (40) into account, Eq. (48) can be described as follows.
$$
E _ {\mathrm {r e}, n} ^ {+} = - c p _ {\mathrm {r e}, n} \left(\frac {r _ {n} ^ {-}}{r _ {n} ^ {+}}\right) ^ {1 / 2}. \tag {49}
$$
In Eq. (49), $r_n^-$ of electrons at ultra-low energy levels contribute to the energy levels of an ordinary hydrogen atom.
In this paper, Eq. (49) is derived using a method different from that in the previous paper.
Now, it is known that Eq. (24) can be written as follows if Eq. (28b) is taken into consideration.
$$
\left(m _ {\mathrm {e}} c ^ {2}\right) ^ {2} \left(\frac {n ^ {2}}{n ^ {2} + \alpha^ {2}}\right) + c ^ {2} \left(m _ {\mathrm {e}} c\right) ^ {2} \left(\frac {\alpha^ {2}}{n ^ {2} + \alpha^ {2}}\right) = \left(m _ {\mathrm {e}} c ^ {2}\right) ^ {2}. \tag {50}
$$
From this, the following formula can be derived.
$$
p_{\mathrm{re},n} = m_{\mathrm{e}} c \left(\frac{\alpha^{2}}{n^{2} + \alpha^{2}}\right)^{1/2}.
$$
Incidentally, the following equation can be derived from Eqs. (24) and (51).
$$
\left(m _ {n} c ^ {2}\right) ^ {2} = c ^ {2} p _ {\mathrm{r e}, n} ^ {2} \left[ \frac{m _ {\mathrm{e}} ^ {2} c ^ {2}}{p _ {\mathrm{r e} , n} ^ {2}} - 1 \right]. \tag{52}
$$
Substituting the value of Eq. (51) for $p_{\mathrm{re},n}^2$ in parentheses in Eq. (52),
$$
\begin{array}{l} \left(m _ {n} c ^ {2}\right) ^ {2} = c ^ {2} p _ {\mathrm {r e}, n} ^ {2} \left[ \left(\frac {n ^ {2} + \alpha^ {2}}{\alpha^ {2}}\right) - 1 \right] \\= c ^ {2} p _ {\mathrm {r e}, n} ^ {2} \frac {n ^ {2}}{\alpha^ {2}}. \tag {53} \\\end{array}
$$
$$
m _ {n} ^ {\pm} c ^ {2} = \pm c p _ {\mathrm {r e}, n} \frac {n}{\alpha}. \tag {54}
$$
Incidentally, according to Maxwell's electromagnetism, there is the following relationship between the energy and momentum of light.
$$
E = c p. \tag {55}
$$
In light of Eq. (55), Eq. (54) was written as a formula including $cp$.
Using Eq. (54) the energy levels of an ordinary hydrogen atom can be written as follows.
$$
\begin{array}{l} E _ {\mathrm {r e}, n} ^ {+} = m _ {n} c ^ {2} - m _ {\mathrm {e}} c ^ {2} \\= c p _ {\mathrm {r e}, n} \frac {n}{\alpha} - m _ {\mathrm {e}} c ^ {2} \\= - c p _ {\mathrm {r e}, n} \left(\frac {m _ {\mathrm {e}} c}{p _ {\mathrm {r e} , n}} - \frac {n}{\alpha}\right). \tag {56} \\\end{array}
$$
Using Eq. (51) again here,
$$
E _ {\mathrm{r e}, n} ^ {+} = - c p _ {\mathrm{r e}, n} \left[ \left(\frac{n ^ {2} + \alpha^ {2}}{\alpha^ {2}}\right) ^ {1 / 2} - \frac{n}{\alpha} \right]. \tag{57}
$$
Incidentally, the author can write the fine-structure constant as follows.
$$
\alpha = \left[ \left(n ^ {2} + \alpha^ {2}\right) ^ {1 / 2} + n \right] ^ {1 / 2} \left[ \left(n ^ {2} + \alpha^ {2}\right) ^ {1 / 2} - n \right] ^ {1 / 2}. \tag {58}
$$
Using Eq. (58), Eq. (57) becomes as follows.
$$
\begin{array}{l} E _ {\mathrm {r e}, n} ^ {+} = - c p _ {\mathrm {r e}, n} \cdot \frac {\left[ \left(n ^ {2} + \alpha^ {2}\right) ^ {1 / 2} - n \right] ^ {1 / 2} \left[ \left(n ^ {2} + \alpha^ {2}\right) ^ {1 / 2} - n \right] ^ {1 / 2}}{\left[ \left(n ^ {2} + \alpha^ {2}\right) ^ {1 / 2} + n \right] ^ {1 / 2} \left[ \left(n ^ {2} + \alpha^ {2}\right) ^ {1 / 2} - n \right] ^ {1 / 2}} \\= - c p _ {\mathrm {r e}, n} \left[ \frac {\left(n ^ {2} + \alpha^ {2}\right) ^ {1 / 2} - n}{\left(n ^ {2} + \alpha^ {2}\right) ^ {1 / 2} + n} \right] ^ {1 / 2} \\= - c p _ {\text {r e}, n} \left(\frac {r _ {n} ^ {-}}{r _ {n} ^ {+}}\right) ^ {1 / 2}. \tag {59} \\\end{array}
$$
Actually, Eq. (59) is not used to calculate the energy levels of the hydrogen atom. However, Eq. (59) is an important formula that allows us to have confidence that ultra-low energy levels exist in the hydrogen atom.
## VII. CONCLUSION
The author has previously pointed out that ultra-low energy levels, not predictable in quantum mechanics, exist in the hydrogen atom. An electron in such a state has negative mass. The author adopted the name DHA for a material formed from such an electron and a proton with positive mass. The thought process whereby the author arrived at this conclusion is summarized below.
1. The reduction in rest mass energy of the electron corresponds to the potential energy of the hydrogen atom. If the reduction in rest mass energy of the electron is expressed as $-\Delta m_{\mathrm{e}}c^{2}$, then potential energy of the hydrogen atom is given by the following formula.
$$
V \left(r _ {n}\right) = - \Delta m _ {\mathrm {e}} c ^ {2}. \tag {60}
$$
Also, the relativistic kinetic energy $K_{\mathrm{re},n}$ of the electron in a hydrogen atom is given by the following equation.
$$
K _ {\mathrm {r e}, n} = - E _ {\mathrm {r e}, n} = m _ {\mathrm {e}} c ^ {2} - m _ {n} c ^ {2}. \tag {61}
$$
Taking Eqs. (60) and (61) as a departure point, it is possible to derive the various surprising formulas in this paper.
2. The energy-momentum relationship applicable to the electron in a hydrogen atom, where potential energy is present, is not the relationship of Einstein but rather the following relationship.
$$
\left(m _ {n} c ^ {2}\right) ^ {2} + c ^ {2} p _ {\mathrm {r e}, n} ^ {2} = \left(m _ {\mathrm {e}} c ^ {2}\right) ^ {2}. \tag {62}
$$
3. Dirac predicted that there is a negative solution to Einstein's relationship, but using the same argument, it is evident that Eq. (62) has the following positive and negative solutions.
$$
E _ {\mathrm {a b}, n} ^ {\pm} = \pm m _ {n} c ^ {2} = \pm m _ {\mathrm {e}} c ^ {2} \left(\frac {n ^ {2}}{n ^ {2} + \alpha^ {2}}\right) ^ {1 / 2}. \tag {63}
$$
$E_{\mathrm{ab}}$ is the absolute energy of the electron. $E_{\mathrm{ab}}$ becomes zero when the position $r$ of the electron is $r = r_{\mathrm{e}} / 2$.
Also, the $r$ 's of the electron corresponding to these two energy levels are, respectively, as follows.
$$
r _ {n} ^ {+} = \frac {r _ {\mathrm {e}}}{2} \left[ 1 + \frac {n}{\left(n ^ {2} + \alpha^ {2}\right) ^ {1 / 2} - n} \right]. \tag {64}
$$
$$
r _ {n} ^ {-} = \frac {r _ {\mathrm {e}}}{2} \left[ 1 - \frac {n}{\left(n ^ {2} + \alpha^ {2}\right) ^ {1 / 2} + n} \right]. \tag {65}
$$
This shows that the region in an ordinary hydrogen atom starts not from $r = 0$ but from $r = r_{\mathrm{e}} / 2$.
4. The author has previously derived the following formula for the energy levels of a hydrogen atom including only the principal quantum number $n$.
$$
E _ {\mathrm {r e}, n} ^ {+} = m _ {n} c ^ {2} - m _ {\mathrm {e}} c ^ {2} = m _ {\mathrm {e}} c ^ {2} \left[ \left(\frac {n ^ {2}}{n ^ {2} + \alpha^ {2}}\right) ^ {1 / 2} - 1 \right]. \tag {66}
$$
However, recently the author noticed that the energy levels of the hydrogen atom can also be described with the following formula.
$$
E _ {\mathrm {r e}, n} ^ {+} = - c p _ {\mathrm {r e}, n} \left(\frac {r _ {n} ^ {-}}{r _ {n} ^ {+}}\right) ^ {1 / 2}. \tag {67}
$$
Furthermore, Eq. (67) was derived by the author using two methods. Eq. (67) is believed to be compelling evidence that ultra-low energy levels exist in the hydrogen atom.
### ACKNOWLEDGMENTS
I would like to express my thanks to the staff at ACN Translation Services for their translation assistance. Also, I wish to express my gratitude to Mr. H. Shimada for drawing figures.
#### APPENDIX
Bohr's orbital radius $r_{\mathrm{BO},n}$ is normally described with the following formula.
$$
r _ {\mathrm {B O}, n} = 4 \pi \varepsilon_ {0} \frac {\hbar^ {2}}{m _ {\mathrm {e}} e ^ {2}} n ^ {2}. \tag {A1}
$$
Bohr thought the following quantum condition was necessary to find the energy levels of the hydrogen atom.
$$
m _ {c} v _ {n} \cdot 2 \pi r _ {\mathrm {B O}, n} = 2 \pi \eta h. \tag {A2}
$$
In Bohr's theory, the energy levels of the hydrogen atom is treated non-relativistically, and thus here the momentum of the electron is taken to be $m_{\mathrm{e}}v$. Also, the Planck constant $h$ can be written as follows [20].
$$
\hbar = \frac {h}{2 \pi} = \frac {m _ {\mathrm {e}} c \lambda_ {\mathrm {C}}}{2 \pi}. \tag {A3}
$$
$\lambda_{\mathrm{C}}$ is the Compton wavelength of the electron.
When Eq. (A3) is used, the fine-structure constant $\alpha$ can be expressed as follows.
$$
\alpha = \frac {e ^ {2}}{4 \pi \varepsilon_ {0} h c} = \frac {e ^ {2}}{2 \varepsilon_ {0} m _ {\mathrm {e}} c ^ {2} \lambda_ {\mathrm {C}}}. \tag {A4}
$$
Also, the classical electron radius $r_{\mathrm{e}}$ is defined as follows.
$$
r _ {\mathrm {e}} = \frac {e ^ {2}}{4 \pi \varepsilon_ {0} m _ {\mathrm {e}} c ^ {2}}. \tag {A5}
$$
If $r_{\mathrm{e}} / \alpha$ is calculated here,
$$
\frac {r _ {\mathrm {e}}}{\alpha} = \frac {\lambda_ {\mathrm {C}}}{2 \pi}. \tag {A6}
$$
If Eq. (A1) is written using $r_{\mathrm{e}}$ and $\alpha$, the result is as follows.
$$
r _ {\mathrm {B O}, n} = 4 \pi \varepsilon_ {0} \frac {\hbar^ {2}}{m _ {\mathrm {e}} e ^ {2}} n ^ {2} = \frac {e ^ {2}}{4 \pi \varepsilon_ {0} m _ {\mathrm {e}} c ^ {2}} \left(\frac {4 \pi \varepsilon_ {0} \hbar c}{e ^ {2}}\right) ^ {2} n ^ {2} = \frac {r _ {\mathrm {e}}}{\alpha^ {2}} n ^ {2}. \tag {A7}
$$
Equation (A7) containing $r_{\mathrm{e}}$ is superior to Eq. (A1) from a physical standpoint.
Next, if $\hbar$ in Eq. (A3) and $r_{\mathrm{BO},n}$ in Eq. (A7) are substituted into Eq. (A2),
$$
m _ {\mathrm {e}} v _ {n} \cdot 2 \pi \frac {r _ {\mathrm {e}}}{\alpha^ {2}} n ^ {2} = 2 \pi n \frac {m _ {\mathrm {e}} c \lambda_ {\mathrm {C}}}{2 \pi}. \tag {A8}
$$
If Eq. (A6) is also used, then Eq. (A8) can be written as follows.
$$
m _ {\mathrm {e}} v _ {n} \cdot 2 \pi \frac {r _ {\mathrm {e}}}{\alpha^ {2}} n ^ {2} = 2 \pi n \frac {m _ {\mathrm {e}} c r _ {\mathrm {e}}}{\alpha}. \tag {A9}
$$
From this, the following relationship can be derived.
$$
\frac {v _ {n}}{c} = \frac {\alpha}{n}. \tag {A10}
$$
Due to Eq. (A10), it is possible to identify discontinuous states that are permissible in terms of quantum mechanics in the continuous motions of classical theory.
Generating HTML Viewer...
References
18 Cites in Article
Koshun Suto (2015). Presentation of Strong Candidates for Dark Matter.
Koshun Suto (2014). Previously Unknown Ultra-low Energy Level of the Hydrogen Atom Whose Existence can be Predicted.
Koshun Suto (2017). Previously Unknown Ultra-low Energy Level of the Hydrogen Atom Whose Existence can be Predicted.
Koshun Suto (2017). Presentation of Dark Matter Candidates.
K Suto Unknown Title.
A Sommerfeld (1923). Atomic Structure and Spectral Lines.
Koshun Suto (2009). True nature of potential energy of a hydrogen atom..
N Bohr (1913). On the Constitution of Atoms and Molecules.
K Suto (2018). Potential Energy of the Electron in a Hydrogen Atom and a Model of a Virtual Particle Pair Constituting the Vacuum.
Koshun Suto (2018). Derivation of a Relativistic Wave Equation more Profound than Dirac’s Relativistic Wave Equation.
Koshun Suto (2019). Derivation of a Relativistic Wave Equation more Profound than Dirac’s Relativistic Wave Equation.
A Einstein (1961). Relativity.
Koshun Suto (2011). An energy-momentum relationship for a bound electron inside a hydrogen atom.
Koshun Suto (2020). Theoretical Prediction of Negative Energy Specific to the Electron.
K Suto (2020). Dark Matter and the Energy-Momentum Relationship in a Hydrogen Atom.
K Suto (2021). The Quantum Condition That Should Have Been Assumed by Bohr When Deriving the Energy Levels of a Hydrogen Atom.
Koshun Suto (2021). The Quantum Condition That Should Have Been Assumed by Bohr When Deriving the Energy Levels of a Hydrogen Atom.
Koshun Suto (2021). Dark Matter Has Already Been Discovered.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Koshun Suto. 2026. \u201cA Compelling Formula Indicating the Existence of Ultra-low Energy Levels in the Hydrogen Atom\u201d. Global Journal of Science Frontier Research - A: Physics & Space Science GJSFR-A Volume 22 (GJSFR Volume 22 Issue A5).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.