This study assessed the effectiveness of different coagulants for treating leachates before their release into the environment. Three inorganic coagulants (ferric chloride, ferrous sulphate and alum) and one organic coagulant [Moringer Oleifera seed (MOS)] were used in a jar test to determine the optimum pH and dosage for the coagulants. Raw and treated leachates were analysed for physiochemical parameters such as pH, chemical oxygen demand, Total solids, Pb and Cr. The optimum pH for ferric chloride, ferrous sulphate, alum and MOS was 7, 7,6 and 10 respectively. While the optimum dosage for each coagulant was 3g/L, 3g/L, 5g/L and 5g/L respectively. The analysis of the raw leachate sample showed that it was highly polluted (Dry season: COD -3000mg/L, TSS -2369mg/L, Cr -0.075mg/L, Pb -0.25mg/L and Mn -0.29mg/L; Wet season: COD -3000mg/L, TSS -2369mg/L, Cr -0.075mg/L, Pb -0.25mg/L and Mn -0.29mg/L). Coagulants removal efficiency (RE) for COD ranges from 12% to 41% with ferric chloride having the highest removal efficiency.
## I. INTRODUCTION
The continuous growth in population and industrialization globally has led to increases in solid waste generation and the problem of its management. Solid waste collection and disposal are among the most serious threats to waste management in most cities in developing countries (Donevska et.al., 2006). Solid waste is any material, which is not in liquid form, and has no value to the person who is responsible for it (Zurbrugg, 2003). Babatola (2008) described waste as any material lacking direct value to the user and so must be disposed of.
The poor management of solid wastes constitutes a disaster for human health and leads to environmental degradation (Achankeng, 2003). One of the most important issues of concern in open dump or landfill waste disposal method is the issue of leachate generation and its potential for downgrading water resources systems (Sartaj et.al., 2010). Leachates are defined as the aqueous effluent generated as a consequence of rainwater percolation through wastes, biochemical processes in waste's cells and the inherent water content of wastes themselves (Lee et.al., 2012). The generated leachate can cause significant environmental damage, becoming a major pollution hazard when it comes into contact with the surrounding soil, ground or surface waters. This leachate often contains a high concentration of organic matter and inorganic ions, including ammoniacal nitrogen and heavy metals; posing great treat to human (Zouboulis et al., 2008).
The quality of leachate is affected by factors such as dumpsite age, precipitation, seasonal weather variation, waste type and composition. Treatment methods are highly dependent on leachate characteristics and tolerance of the method against changes in leachate quality such a variable nature along with other factors. The leachate treatments success depends also on the characteristics of the leachate and age of the landfill. Therefore, in order to avoid environmental damage, landfill leachate must be collected and appropriately treated before being discharged into any water body (Oh et.al., 2007).
Coagulation is widely used for wastewater treatment. This treatment is efficient to operate and the operating cost is low (Wang et.al., 2008). It has many factors that can influence the efficiency, such as the type and dosage of coagulant, pH, mixing speed and time and retention time. The optimization of these factors may influence the efficiency (Wangand Bank, 2007). Coagulation destabilizes the colloidal suspension of the particles with coagulants and then causing the particles to agglomerate with flocculants. After that, it will accelerate separation and thereby makes the effluents clearer (Gnandi et.al., 2005).
There are two kinds of coagulants; inorganic and organic coagulants. Inorganic coagulants (such as Alum, Ferric chloride etc.) are the most commonly used in coagulation treatment of leachate. The use of organic coagulants (M. oleifera seed, Phaseolus vulgaris seed, etc.) is not as common as the inorganic coagulants. The Moringa Oleifera tree grows in tropical and subtropical regions around the world and its seeds have been used in drinking water treatment in small scale in Sudan and India for generations. Coagulation studies are usually carried out using jar test equipment. The jar test has been the typical technique used in wastewater and drinking water industry to improve the addition of coagulant and flocculants (Silver et.al., 2004).
This paper seeks to investigate the efficiency of M. oleifera and compare the differences in the removal efficiency of alum, ferrous sulphate and ferric chloride to M. oleifera as coagulants in removing physicochemical parameters of leachate. Also to assess the effect of pH on the effectiveness of coagulants in leachate treatment and determine the pollution level of leachate samples by determination of water quality parameters.
Ogun State, South-West Nigeria. Abeokuta covers an approximate area of about $40.63\mathrm{km}^2$. Saje dumpsite lies between latitude $7^{\circ}09'$ N - $7^{\circ}19'$ N and longitudes $3^{\circ}29'$ E - $3^{\circ}41'$ E (Ufoebune et.al., 2008).
The Saje dumpsite (figure 1) established in 2006 was formerly a quarry, where mining was done over a long period of time for granites. In order to reclaim the site the state government decided to use the quarry as dumpsite. The dumpsite is the only major dumpsite used in Abeokuta metropolis and is about 4 ha in area. Saje area was formally an outskirt of Abeokuta town but due to increased population of the metropolis, houses have encroached the site of the dump site (Badejo et.al., 2013).
The location coordinate of the dumpsite was obtained with a hand held Global Positioning System (GPS, Garmin MAP 76CSx model made in Taipei County, Taiwan) with position accuracy of less than $3\mathrm{m}$. The choice of the sampling points within the dumpsite was considered using the following criteria: location, accessibility and availability of leachate.
## II. MATERIALS AND METHODS
### a) Study Area
The study area Saje is located in Abeokuta North Local Government of Abeokuta, the capital of
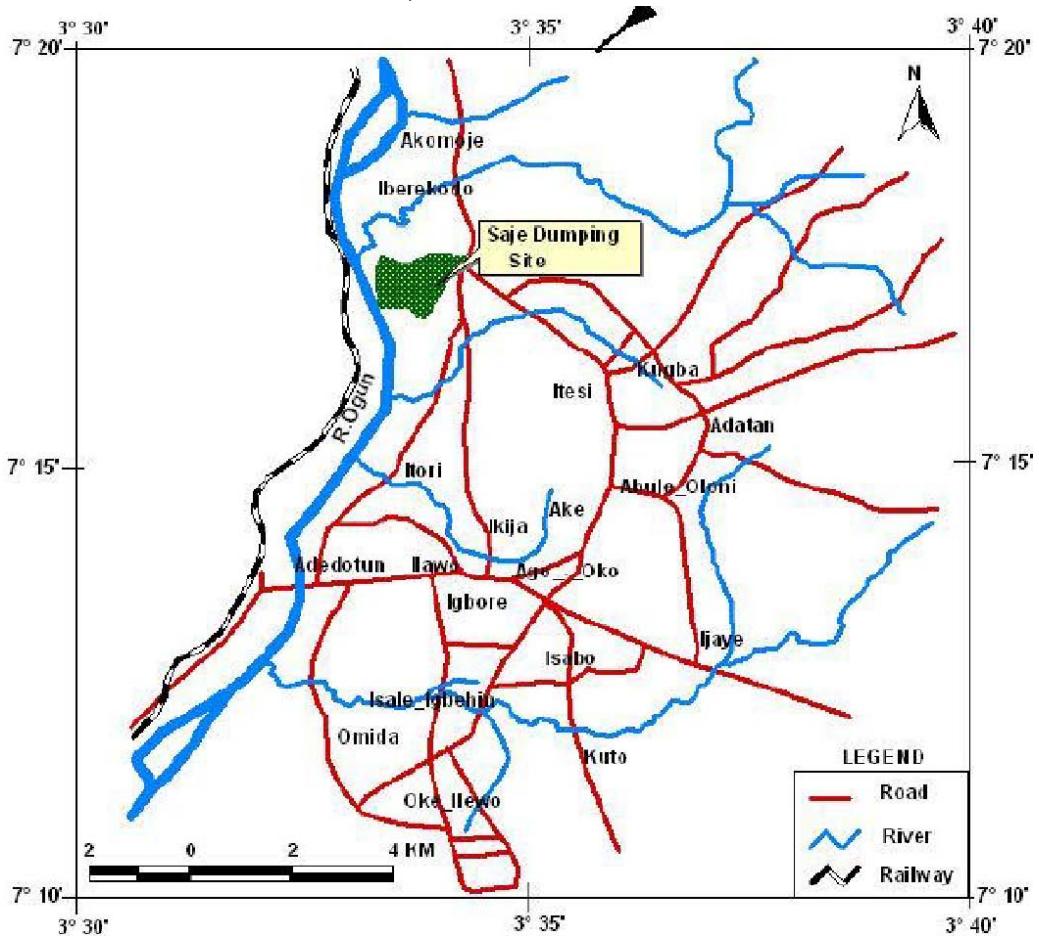 Figure 1: Saje Dumpsite Location within Abeokuta City, Ogun State, Nigeria.
### b) Sampling Procedures
Leachate: Leachate samples were collected from Saje dump site (figure 1) in Abeokuta, Ogun State during wet and dry seasons using standard methods. Samples were collected in $15\mathrm{L}$ plastic containers, transported to the laboratory and stored in a refrigerator at $4^{\circ}\mathrm{C}$. The samples were removed from the refrigerator and left under room temperature for at least 2 hours. Then, the samples were thoroughly stirred to agitate settled solids before any tests were conducted on the leachate samples.
 Figure 2: Leachate collection point from Saje dumpsite
### c) Reagents
In this study, Ferric Chloride $\left(\mathrm{FeCl}_{3} \cdot 6 \mathrm{H}_{2} \mathrm{O}\right)$, Ferrous Sulphate $\left(\mathrm{Fe} \left(\mathrm{SO}_{4}\right)_{3} \cdot 7 \mathrm{H}_{2} \mathrm{O}\right)$ and Aluminum Chloride $\left(\mathrm{Al}_{2} \left(\mathrm{SO}_{4}\right)_{3} \cdot 18 \mathrm{H}_{2} \mathrm{O}\right)$ were used as inorganic coagulants. Moringa oleifera Seed (MOS) was used as organic coagulant. MOS $10\%$ Stock solution was prepared daily by dissolving $30 \mathrm{~g}$ of MOS powder into $270 \mathrm{ml}$ of distilled water, and the solution was well mixed.
### d) Analysis Techniques
The physical and chemical parameters were determined using APHA Standard Methods (2005) for testing water and waste water. pH was assessed by glass electrode method with a calibrated pH meter, while temperature EC and TDS was determined using HM Digital Meter COM-100. Total alkalinity, total hardness, Acidity, chloride, were determined by titrimetric method. A total suspended solid was determined by gravimetric method. Chemical oxygen demand (COD) was determined by open reflux method. Nitrate Phosphate and Sulphate were measured by UV-Visible spectrophotometer. The heavy metal analysis was carried out using Atomic Absorption Spectrophotometer (AAS) Model 210 VGP of the Buck Scientific AAS series.
### e) Experimental Procedure
Chemical coagulation was performed using beakers and stirrer as Jar test apparatus. The experimental process consisted of three subsequent stages: initial rapid mixing at 160 rpm for 10 min, followed by slow mixing for 20 min at 30 rpm, the final settling time for 1 h.
First, the optimum pH was determined by varying the pH of the sample using HCl and NaOH at constant coagulant concentration. The pH with the highest removal efficiency was the optimum pH.
About 2L beakers of equal volume were used to examine the different coagulants at their respective optimum pH. A known mass of (1g, 2g, 3g, 4g and 5g) of each coagulants was added to a jar containing 1liter of leachate samples at optimum pH using the jar test procedure. To determine the efficiency of coagulant dose, the supernatant was withdrawn by using a pipette from a point about $2\mathrm{cm}$ below the top of liquid level of the beaker and the supernatant was assessed for TSS, COD, Mn, Pb and Cr.
### f) Data Analysis
Data collected were evaluated for descriptive and inferential statistics using the Statistical Package for Social Sciences (SPSS) for windows version 20. The removal efficiency (RE) of the coagulants was determined for each parameter by using the equation:
$$
\mathrm{RE}(\%) = \frac{\frac{C_i - C_f}{C_i}}{\times 100\frac{C_i - C_f}{C_i}} \times 100
$$
Where, $C_i$ and $C_t$ are the initial and final concentrations of the parameters.
## III. RESULTS AND DISCUSSION
### a) Characteristics of Landfill Leacahte
The results of the physiochemical analysis of the untreated leachate samples from the dumpsite during dry and wet seasons are presented in Table 1.
The Chemical Oxygen Demand (COD), Total Suspended Solids (TSS), Total Dissolved Solid (TDS),
Chloride, Chromium, Lead and Manganese of the untreated leachate samples from the dumpsites exceeded the limiting values recommended by the WHO and the FMENV. The values of all other parameters were within the allowable limits as specified. The high level of Pb and Mn is due to the dumping of metals such as cans, used batteries, iron etc in the dumpsite.
Table 1: Characteristic of raw leachate sample for wet and dry seasons
<table><tr><td>Parameters</td><td>Dry (mg/L)</td><td>Wet (mg/L)</td><td>T value</td></tr><tr><td>*pH</td><td>7.50±0.10</td><td>7.70±0.20</td><td>0.011</td></tr><tr><td>Alkalinity</td><td>47.00±1.00</td><td>49.00±1.00</td><td>0.007</td></tr><tr><td>COD</td><td>2900±101</td><td>3000.0±100</td><td>0.01</td></tr><tr><td>Acidity</td><td>50.00±10.10</td><td>47.00±3.0</td><td>0.006</td></tr><tr><td>Hardness</td><td>2240±201</td><td>2280.0±102</td><td>0.01</td></tr><tr><td>**EC</td><td>5790±120</td><td>8740.0±90.0</td><td>0.02</td></tr><tr><td>TDS</td><td>3110±110</td><td>4720.0±96.0</td><td>0.004</td></tr><tr><td>TSS</td><td>2333.0±120.00</td><td>2333.0±20.0</td><td>0.001</td></tr><tr><td>Chloride</td><td>870.0±65.00</td><td>910.0±202</td><td>0.01</td></tr><tr><td>Nitrate</td><td>0.73±0.00</td><td>0.31±0.00</td><td>0.01</td></tr><tr><td>Phosphate</td><td>0.49±0.00</td><td>0.32±0.00</td><td>0.012</td></tr><tr><td>Sulphate</td><td>204.51±2.52</td><td>174.84±10.0</td><td>0.01</td></tr><tr><td>Chromium</td><td>0.07±0.01</td><td>0.08±0.01</td><td>0.013</td></tr><tr><td>Lead</td><td>0.25±0.0</td><td>0.25±0.03</td><td>0.013</td></tr><tr><td>Manganese</td><td>0.29±0.02</td><td>0.30±0.02</td><td>0.014</td></tr></table>
### b) Coagulants optimum pH
Results for the optimum pH obtained from the coagulation of leachate samples using Ferric Chloride, Ferrous Sulphate, Aluminum Chloride and MOS at varying pH values (4,6,7,8, and 10) to evaluate COD and TSS concentrations in the samples are shown in Figure 3 and 4. All the coagulants were kept at $2\mathrm{g} / \mathrm{L}$ in all the runs. The pH with the highest removal efficiency (ER) was taken as the optimum pH for the coagulant. It was
Noticed that all the coagulants gave different results at different pH.$\mathrm{FeCl}_3$and$\mathrm{FeSO}_4$had optimum pH of 7, while Alum had optimum pH of 6, and MOS had its optimum pH at a pH value of 10.
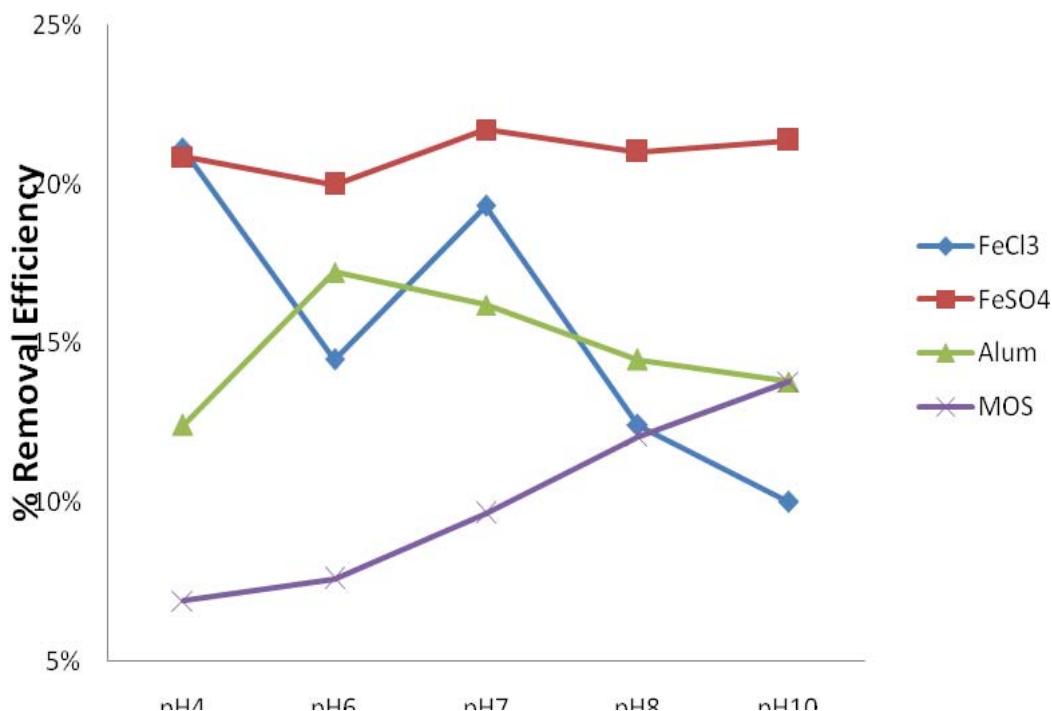
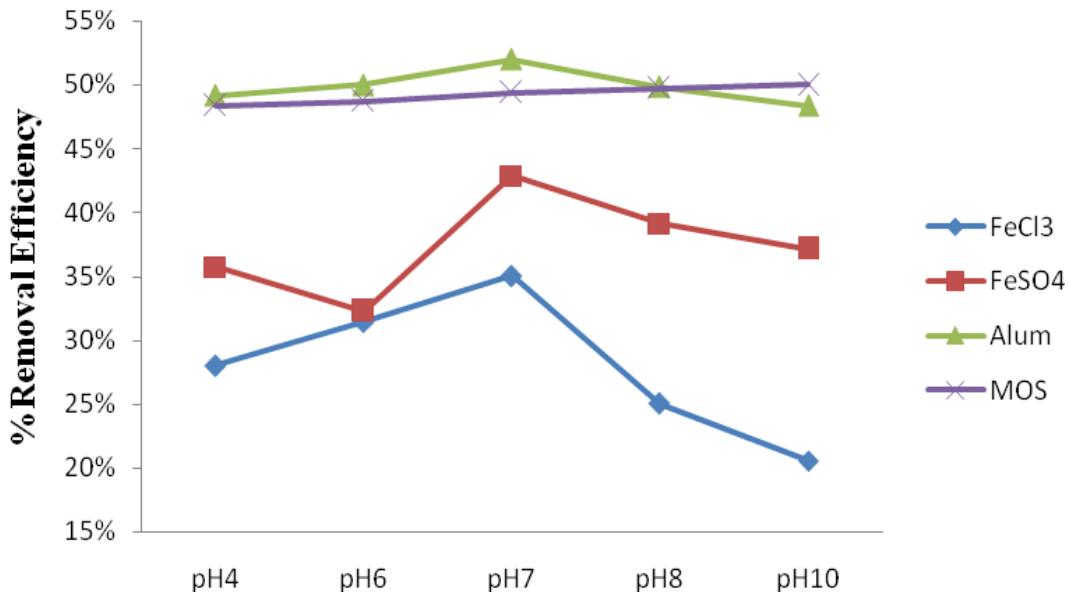 Figure 3: Effects of varying pH at constant coagulant dosage on COD
### c) Effects of Different Coagulant Concentrations in Coagulation Treatment
To observe the effect of coagulant dose, the experimental runs were conducted at different doses (1, 2, 3, 4 and $5\mathrm{g / L}$ ). The percentage removal efficiency at each dose was compared. Depending on the coagulants, the optimal dose varied with the various coagulants used.
$\mathrm{FeCl}_3$ removal efficiency for heavy metals ranged from $55\%$ to $85\%$ (Figure 5), this is in line with the reported work of Lee et.al. (2012) where $\mathrm{FeCl}_3$ was reported to remove $75\%$ of Pb. Amuda and Alade (2006) also gave a report in this range. $\mathrm{FeCl}_3$ removed Cr better than Pb and Mn. $\mathrm{FeCl}_3$ was not as efficient in removing
COD, the value ranged from $19\%$ to $40\%$ (Figure 6). Other studies also reported low RE of $\mathrm{FeCl}_3$ for COD (Ibrahim et.al., 2012; Lee et.al., 2012). The optimum dosage for $\mathrm{FeCl}_3$ was determined to be $3\mathrm{g} / \mathrm{L}$, the RE dropped beyond this dosage.
$\mathrm{FeSO}_4$ removal efficiency for heavy metals ranged from $65\%$ to $85\%$ as shown in Figure 7. $\mathrm{FeSO}_4$ also removed more of Cr when compared to Pb and Mn, following the trend of $\mathrm{FeCl}_3$. It was also not as efficient in removing COD, the value ranges from $21\%$ to $37\%$ (Figure 8) this was in accordance with the work of Ibrahim et.al. (2012). $\mathrm{FeSO}_4$ also had optimum dosage of $3\mathrm{g / L}$. Additional concentration above the optimum dosage reduced the efficiency of the coagulant.
Alum had a higher RE for heavy metals compared to $\mathrm{FeCl}_3$ and $\mathrm{FeSO}_4$. Its values ranged from $72\% - 94.28\%$. Like in other Coagulants, Cr has the highest RE of $94.28\%$. This was closely followed by Pb (92.8%) and Mn (87.9%) as presented in Figure 9. The maximum COD removal of $41.72\%$ (Figure 10) resulted at 5 g/L coagulant dose. The COD removal increased with alum dosage increase. Zazouli and Yousefi (2008), Bila et.al. (2005) also reported RE of Alum for heavy metals in the range of $71\% - 96\%$ and COD in the range of $27\% - 40\%$ in their reports. Meanwhile, Trebouet et.al. (2001) reported a much lower maximum RE of $66\%$ for heavy metals.
MOS had the best range of RE for heavy metals of all the four coagulants with a minimum of $72.4\%$ and maximum of $95.6\%$ (Figure 11). It removed more of $\mathsf{Pb}$, than Mn and Cr had the least RE. Ravikumar and Sheeja, (2013) reported a $93\%$ RE for $\mathsf{Pb}$ and $70\%$ RE for Cr in their work. Both Alum and MOS increased there RE for heavy metals with increase in concentration. Figure 12 showed that MOS treated samples had increased COD concentrations, giving a negative RE. This is similar to the report in previous studies (Arnoldsson and Bergman, 2007).
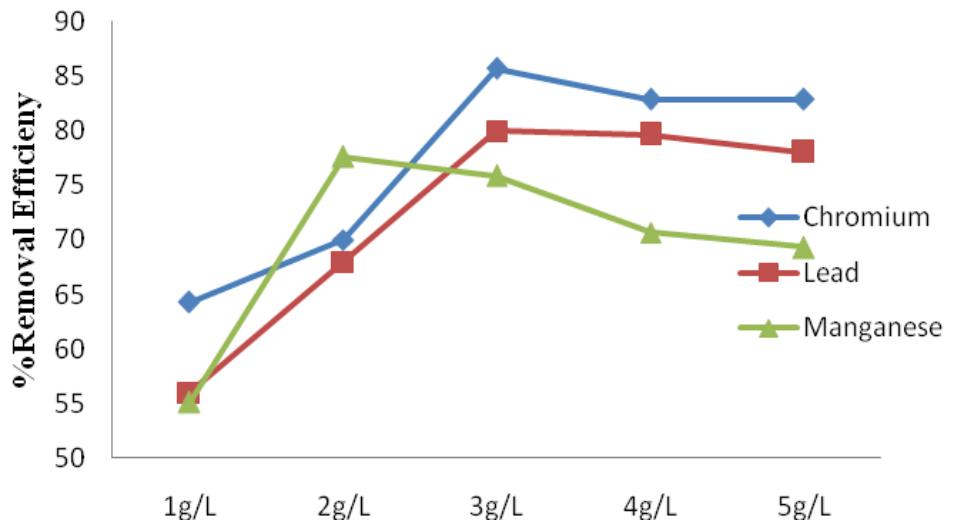 Figure 4: Effects of varying pH at constant coagulant dosage on TSS
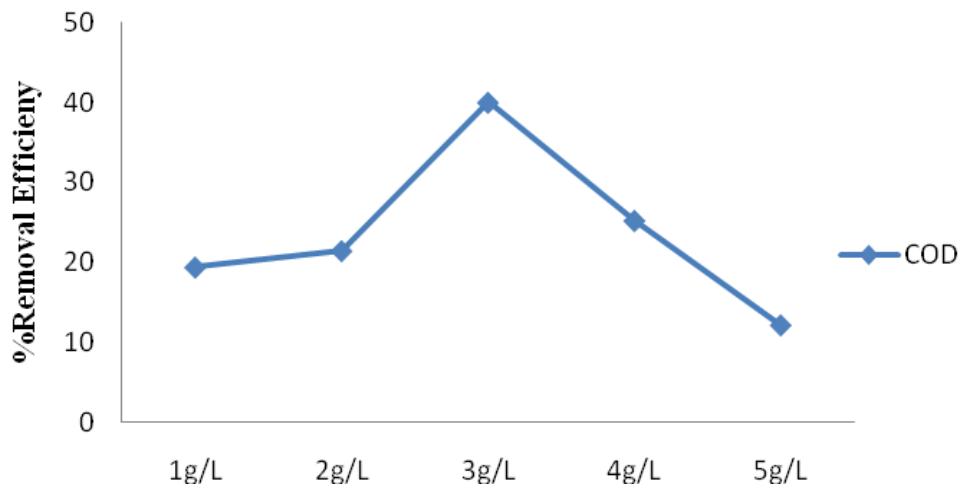 Figure 5: $\mathsf{FeCl}_3$ percentage removal efficiency for Cr, Pb and Mn
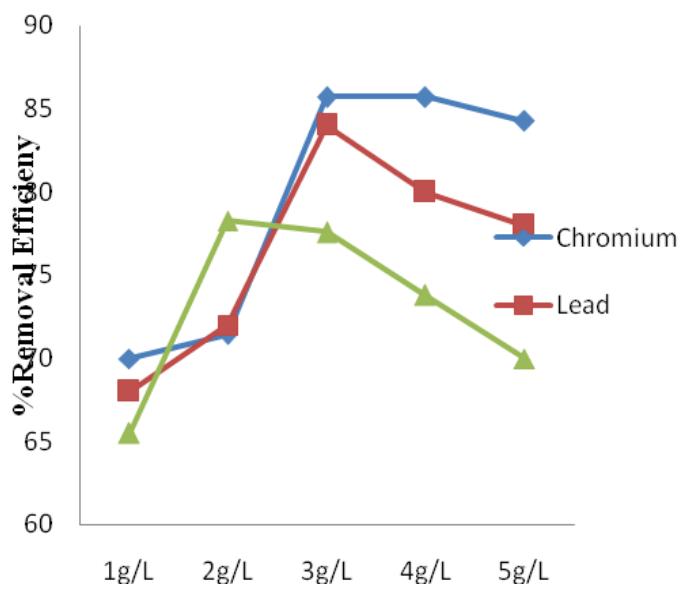 Figure 7: $\mathrm{FeSO}_4$ percentage removal efficiency for Cr, Pb and Mn
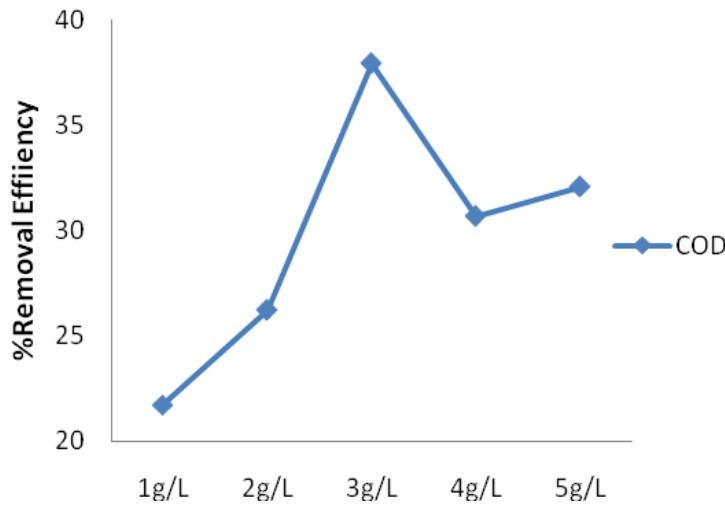 Figure 8: $\mathsf{FeSO}_4$ percentage removal efficiency for COD
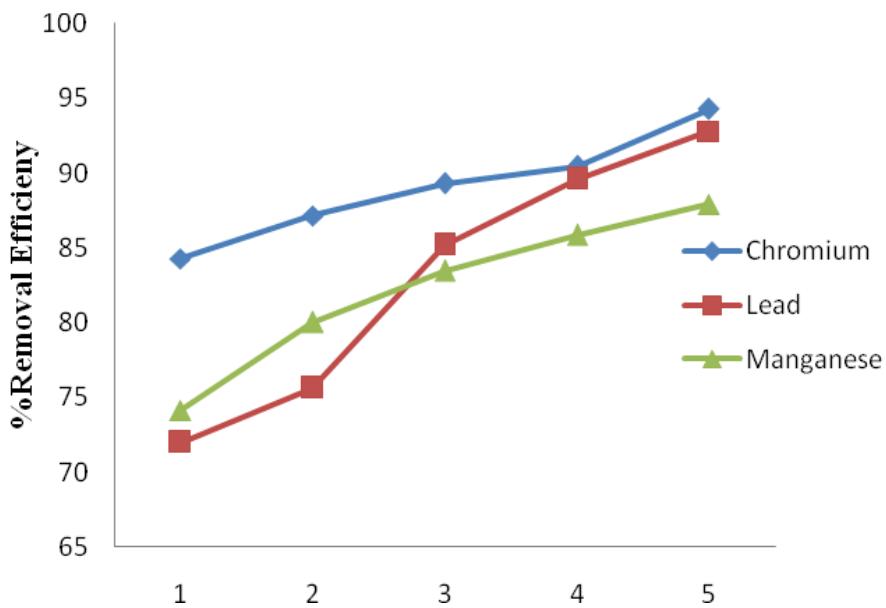 Figure 9: Alum percentage removal efficiency for Cr, Pb and Mn
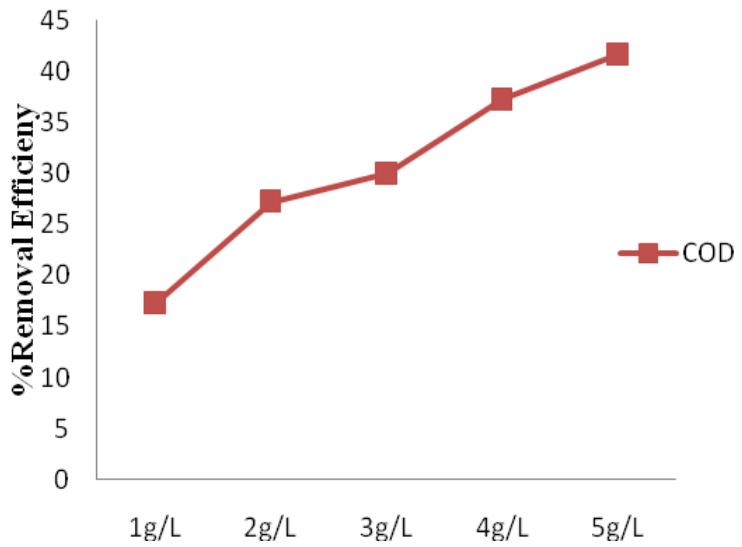 Figure 10: Alum percentage removal efficiency for COD
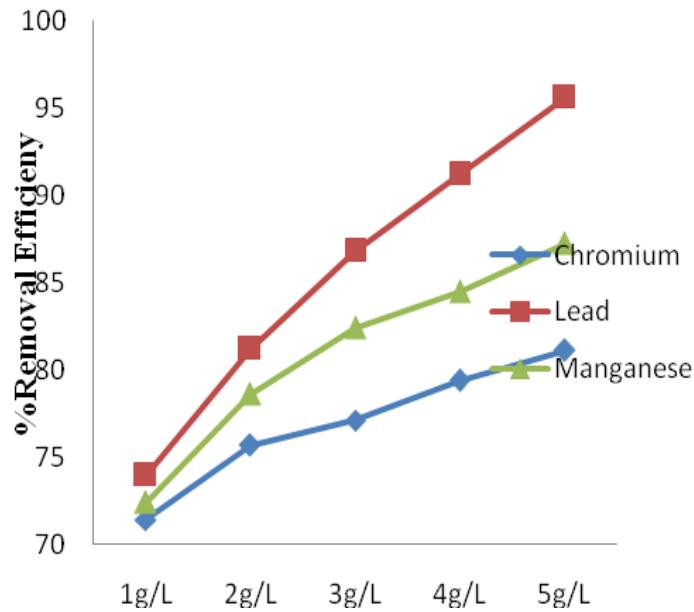 Figure 6: $\mathrm{FeCl}_3$ percentage removal for COD
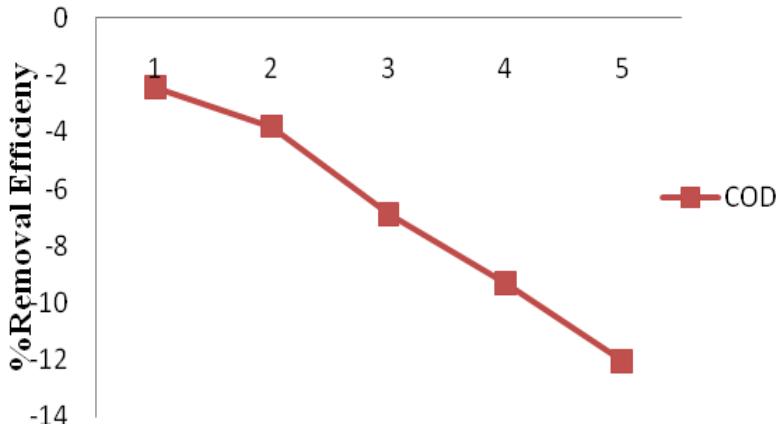 Figure 11: MOS percentage removal efficiency for Cr, Pb and Mn Figure 12: MOS percentage removal efficiency for COD
Alum exhibited a good performance in the sample parameters compare to other coagulants. Table 2 shows that the coagulants were able to reduce heavy metals from the leachates samples to level below standard limits but COD and TSS still had values higher than the recommended standards.
Table 2: Summary of comparison of coagulants optimum dosage parameters
<table><tr><td>Parameters</td><td>Raw</td><td>FeCl3(3g/L)</td><td>FeSO4(3g/L)</td><td>ALUM(5g/L)</td><td>MOS(5g/L)</td><td>WHO Standard</td><td>FMENV Limit For Discharge To The Environment</td></tr><tr><td>COD (mg/L)</td><td>3000</td><td>1770</td><td>1875</td><td>1730</td><td>0</td><td>60.9</td><td>60.9</td></tr><tr><td>TSS (mg/L)</td><td>2369</td><td>207</td><td>1637</td><td>368</td><td>1047.5</td><td>25</td><td>25</td></tr><tr><td>Chromium (mg/L)</td><td>0.0750</td><td>0.0105</td><td>0.0105</td><td>0.0045</td><td>0.0133</td><td>0.0500</td><td>0.0500</td></tr><tr><td>Lead (mg/L)</td><td>0.2500</td><td>0.0490</td><td>0.0395</td><td>0.0175</td><td>0.0110</td><td>0.0500</td><td>0.0500</td></tr><tr><td>Manganese (mg/L)</td><td>0.2950</td><td>0.0705</td><td>0.0645</td><td>0.0355</td><td>0.0365</td><td>0.0500</td><td>-</td></tr></table>
Over all, Alum was a better coagulant than the three other coagulants in reducing the physical and chemical parameters of leachates.
### d) Seasonal Variations of Leachate Concentration
The test was carried out both in the dry and wet seasons to determine effect of season on the efficiency of coagulants. Table 4 and 5 is a summary of the physical and chemical properties of the raw and treated leachate samples for dry and wet seasons.
It was shown that the physical and chemical properties of the leachates are higher in the wet season than in the dry season. This can be attributed to the fact that rainfall is a crucial factor in the formation of leachate and the characteristic of the leachate.
Season has no effect on the efficiency of coagulants. The trend of the removal efficiency of each coagulants tested in dry season is similar to that of the wet season.
Table 4: Summary of Physical and Chemical Parameters of Raw Leachates and Treated Leachate in the Dry Season
<table><tr><td></td><td>Raw Leachate</td><td>After treatment FeCl3</td><td>After treatment FeSO4</td><td>After treatment Alum</td><td>After treatment MOS</td></tr><tr><td>COD (mg/L)</td><td>2900.0±100.50b</td><td>2216.0±274.32a</td><td>2038.0±177.68a</td><td>2010.0±274.32a</td><td>3100.0±268.23b</td></tr><tr><td>TSS (mg/L)</td><td>2333.0±120.00c</td><td>1037.9±295.90a</td><td>1726.5±383.22b</td><td>717.56±295.90a</td><td>1246.5±159.15ab</td></tr><tr><td>Cr (mg/L)</td><td>0.07±0.01c</td><td>0.02±0.00b</td><td>0.01±0.01ab</td><td>0.01±0.00a</td><td>0.02±0.00b</td></tr><tr><td>Pb (mg/L)</td><td>0.25±0.03c</td><td>0.07±0.02b</td><td>0.06±0.02ab</td><td>0.04±0.02a</td><td>0.04±0.02a</td></tr><tr><td>Mn (mg/L)</td><td>0.29±0.02c</td><td>0.09±0.02b</td><td>0.08±0.02b</td><td>0.05±0.02a</td><td>0.06±0.02a</td></tr></table>
Table 5: Summary of Physical and Chemical Parameters of Raw Leachates and Treated Leachate in the Wet Season
<table><tr><td></td><td>Raw Leachate</td><td>After treatment FeCl3</td><td>After treatment FeSO4</td><td>After treatment Alum</td><td>After treatment MOS</td></tr><tr><td>COD (mg/L)</td><td>3000.0±100b</td><td>2350.0±288.57a</td><td>2170.0±163.25a</td><td>2122.0±288.57a</td><td>3270.00±279.52b</td></tr><tr><td>TSS (mg/L)</td><td>2333.0±20.0c</td><td>1074.2±295.98a</td><td>1591.0±136.38b</td><td>717.6±295.98a</td><td>1246.6±159.21ab</td></tr><tr><td>Cr (mg/L)</td><td>0.08±0.01c</td><td>0.02±0.00b</td><td>0.02±0.01ab</td><td>0.01±0.00a</td><td>0.02±0.00b</td></tr><tr><td>Pb (mg/L)</td><td>0.25±0.03c</td><td>0.07±0.02b</td><td>0.06±0.02ab</td><td>0.04±0.02a</td><td>0.04±0.02a</td></tr><tr><td>Mn (mg/L)</td><td>0.30±0.02c</td><td>0.09±0.02b</td><td>0.08±0.02b</td><td>0.05±0.02a</td><td>0.06±0.02a</td></tr></table>
## IV. CONCLUSION
The application of coagulation treatment for raw leachate collected from Saje dumpsite showed the leachate was characterized by low pH and high concentration of pollutants; especially that of organic matter as observed in the COD level and high level of heavy metals which are all above the WHO and the FMEv limit for waste water. The study showed that the leachate from the dumpsite is polluted and there is need for it to be treated before it is released into environment.
The study showed that coagulation treatment is efficient in ameliorating the polluting potential of dumpsite leachates. All the four coagulants; ferric chloride, ferrous sulphate, alum and MOS were able to reduce the heavy metals in the leachate by over $55\%$ and MOS removing as high as $95.6\%$. MOS was better than the other coagulants in terms of removal efficiency for heavy metal. The coagulants were not as effective against COD, with alum giving the highest removal efficiency of $41.7\%$ and MOS increased the COD concentration. None of the coagulants was able to bring the COD level down to below the FMEv standard limit.
This study also revealed pH as an important factor in coagulation. It was established that each coagulant has the pH at which it works best; to remove contaminants. This pH is referred to as the optimum pH. In this study the optimum pH for Ferric chloride and ferrous sulphate was 7.0, Alum was 6.0 and MOS was 10.0.
This study had determined the optimum dosage of each coagulant to get the best use of them. It was observed that the optimum dosage for ferric chloride, ferrous sulphate, alum and MOS were $3.0\mathrm{g / L}$, $3.0\mathrm{g / L}$, $5.0\mathrm{g / L}$ and $5.0\mathrm{g / L}$ respectively. From the results Alum was the best coagulant for treating leachates, closely followed by ferric chloride, MOS and ferrous sulphate in that order.
This study has shown little or no seasonal variation in the concentration of leachate. The season did not have significant effect on the efficiency of the coagulants
Moringa Oleifera showed good coagulating properties, and has many advantages compared to aluminium sulphate. It did not affect the pH, alkalinity or conductivity of the water, and it can be produced locally at low cost. Moringa oleifera is an environmentally-friendly natural coagulant that can be used to replace alum and other inorganic coagulants particularly in treating drinking water. It is a method that certainly can be considered as a good, sustainable and cheap solution for smaller waterworks, if the supply of Moringa seeds can be guaranteed.
Generating HTML Viewer...
References
22 Cites in Article
E Achankeng (2003). Globalization, Urbanization and Municipal Solid Waste Management in Africa.
(1999). American Public Health Association (APHA).
O Amuda,A Alade (2006). Coagulation/flocculation process in the treatment of abattoir wastewater.
E Arnoldsson,M Bergman (2007). Assessment of drinking water treatment using MoringaOleifera natural coagulant.
J Babatola (2008). A Study of Hospital Waste Generation and Management Practice in Akure, Nigeria.
A Badejo,A Taiwo,A Adekunle,B Bada (2013). Spatio-Temporal Levels of Essential Trace Metals around Municipal Solid Waste Dumpsite in Abeokuta, Nigeria.
D Bila,F Montalvão,A Silva,M Dezotti (2005). Ozonation of a landfill leachate: evaluation of toxicity removal and biodegradability improvement.
K Choi,S Kim,C Kim,J Park (2006). Removal efficiencies of endocrine disrupting chemicals by coagulation/flocculation, ozonation, powdered/granular activated carbon adsorption, and chlorination.
K Donevska,M Jovanovski,A Efremov,J Papic (2006). Impact Assessment of solid waste landfill in the Municipality of Center Zupa.
Ibrahim,Ahmed Nabil,M,Mahmoud,Mohamed (2012). Combined coagulation flocculation pretreatment unit for municipal wastewater.
M Lee,D Zawawi,A Abdul (2012). Treatment of leachate by Coagulation-Flocculation using different coagulants and polymer.
B Oh,J Lee,J Yoon (2007). Removal of contaminants in leachate from landfill by waste steel scrap and converter slag.
I Onwughara,I Nnorom,O Kanno (2010). Issues of Roadside Disposal Habit of Municipal Solid Waste, Environmental Impacts and Implementation of Sound Management Practices in Developing Country -Nigeria.
M Sartaj,M Ahmadifar,A Jashni (2010). Assessment of in-situ aerobic treatment of municipal landfill leachate at laboratory scale.
M Sartaj,M Ahmadifar,A Jashni (2010). Assessment of in-situ aerobic treatment of municipal landfill leachate at laboratory scale.
A Silva,M Dezotti,Sant'anna,G Jr (2004). Treatment and detoxication of a sanitary landfill leachate.
A Tatsi,A Zouboulis,K Matis,P Samaras (2003). Coagulation–flocculation pretreatment of sanitary landfill leachates.
D Trebouet,J Schlumpf,P Jaouen,F Quemeneur (2001). Stabilized landfill leachate treatment by combined physicochemical–nanofiltration processes.
G Ufoegbune,O Oguntoke,C Adeofun,A Salako (2008). Remote Sensing Techniques applied to Time Related Changes in the Landuse of Abeokuta and its Environs.
Z Wang,C Banks (2007). Treatment of a highstrength sulphate-rich alkaline leachate using an anaerobic filter.
M Zazouli,Z Yousefi (2008). Removal of Heavy Metals from Solid Wastes Coagulation-Floculation Process.
O Zerbock (2003). Urban Solid Waste Management: Waste reduction in developing nations (written for the requirements of CE 5993 Field Engineering in the Developing World).
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Oso, S. 2026. \u201cAbatement of Polluting Effects of Waste Dump Leachates using Different Coagulants\u201d. Global Journal of Human-Social Science - B: Geography, Environmental Science & Disaster Management GJHSS-B Volume 22 (GJHSS Volume 22 Issue B3).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.