## I. INTRODUCTION
Amyotrophic Lateral Sclerosis (ALS) is a neurodegenerative disease characterized by loss of both upper and lower motor neurons with progressive muscle weakness. In the late stage of the disease, respiratory muscle weakness ensues, leading to hypercapnic respiratory failure, representing the most common cause of death in these patients. The condition has no definitive treatment, and its progression can be only partly modified by pharmacological intervention; when respiratory muscle involvement occurs, the only approach that has demonstrated an improvement in survival time and quality of life is the use of non-invasive ventilation (NIV). Early recognition of diaphragm dysfunction and respiratory muscle weakness is crucial to define the optimal timing for starting NIV [1-3]. The European Federation of Neurological Societies (EFNS) guidelines recommend initiating NIV when at least one respiratory clinical symptom plus forced vital capacity (FVC) $< 80\%$ of the predicted value[4]. However, the PFTs, which represent the most common and validated parameter to assess the respiratory volumes and the need for NIV, are challenging to perform in this subset of patients, because they require cooperation with the examiner and coordination during the maneuvers. On the other, diaphragmatic ultrasound (DU) is a noninvasive, radiation-free technique to assess the diaphragm function regarding of inspiratory displacement and thickening[5]. Hence, DU could provide clinically relevant information to detect patients at risk of diaphragmatic dysfunction for whom starting the NIV is indicated. Limited literature is available [6-8] on the correlation between the data obtained at DU with those obtained at PFTs in the evaluation of diaphragmatic dysfunction in ALS patients. These studies, however, did not take in to consideration the changes in lung volumes induced by changes in body position observed in ASL patients. The aims of our study in ALS patients were: 1) confirm the utility of DU in assessing diaphragmatic function, and 2) evaluate the correlation between DU and PFTs measurements in both standing and supine position.
## II. METHODS
### a) Study design and cohort
A case-control single-centre study conducted in Rome at Umberto I Hospital. During a nine months run-in period, from May 2021 to January 2022, twenty-nine ALS patients with spinal onset, and thirteen healthy controls were enrolled. The inclusion criteria were: (1) diagnosis of ALS according to El Escorial-Revised Diagnostic Criteria[9]; (2) age $>18$ years old; (3) ability and collaboration to perform respiratory tests. The exclusion criteria were (1) ALS with bulbar onset, because unable to correctly perform PFTs; (2) tracheostomy; (3) ventilatory support at the time of the enrolment (4) pregnancy; (5) breastfeeding (6) actual or previous lung diseases. Healthy controls were selected among age-comparable volunteers, with no biological relationship to patients. This study was conducted per the Declaration of Helsinki and Good Clinical Practice guidelines. The local ethics committee approved the study protocol0897/2021, and written informed consent was obtained from all the subjects in the study.
### b) Measurements
Demographic and anthropometric variables were collected in all patients at the time of enrolment.
### c) Pulmonary Function Testing (PFTs)
Lung function evaluation followed the American Thoracic Society (ATS) guidelines[10,11]. The best of at least three measurements was taken to assure reproducibility. The assessment of Vital Capacity (VC) and Forced Vital Capacity (FVC), Forced Expiratory Volume during the first second $(\mathrm{FEV}_1)$ was performed standing upright and lying in a $30^{\circ}$ supine position, using the automated equipment microQuark (Cosmed).
### d) Diaphragm ultrasound (DU)
DU evaluates diaphragm thickening and excursion [5,7,12]. It was assessed by the same observer during the run-in period using Biosound Esaote - AU5 Harmonic EPI, according to Sarwal et al.[13]:
1. Diaphragm thickness was evaluated both at FRC (Te) and at TLC (Ti) by using a B-Mode 7.5-10 Hertz linear probe. Te is the thickness at FRC, and Ti is the thickness at TLC. This technique measures, in a lying $30^{\circ}$ supine position, the diaphragmatic thickness in the zone of apposition to the rib cage between the middle and posterior axillary line. These body and probe positions are utilized to detect the thicker part of the muscle and to minimize possible measurement errors[5,14-16]. The diaphragmatic thickening is then calculated by using two different equations:
$\Delta$ Thickness $(\Delta T) = \text{Ti - Te}$
and
Thickening Fraction $(\mathrm{TF}) = [(T_i - T_e) \times 100]$
2. The real-time diaphragmatic excursion of the right hemidiaphragm was evaluated using a right anterior subxiphoid approach in a lying $30^{\circ}$ supine position during maximal inspiratory effort by M-Mode 3.5 Hertz convex probe[5,15].
### e) Statistical analysis
The data are presented for two groups, ALS patients and controls (Table 1). Categorical variables are presented as a number of patients and percentage and compared between groups with a chi-square test. Numerical data are presented as mean $\pm$ standard deviation (SD) or median [inte quartile range (IQR)], as appropriate. Shapiro-Wilk test was used to test the normal distribution of numerical data. Student's t-test or Wilcoxon test was used to compare numerical values between the two groups, as appropriate. The correlation between respiratory function tests and diaphragmatic ultrasonographic indices was performed using the Pearson test. A value of $p \leq 0.05$ was considered statistically significant. Receiver operating characteristic (ROC) curves were constructed to evaluate the performance of ultrasound-derived indices in predicting FVC $< 80\%$, one of the criteria for starting NIV. The optimal cut-point was determined as the maximal value of sensitivity plus specificity. The statistical analysis was performed with R statistical software version 4.1.
## III. RESULTS
Patient characteristics, PFTs as well as ultrasound-derived indices are shown in Table 1. No significant differences in age, sex, and BMI were observed in the two groups.
Table 1: Demographic, PFTs and ultrasonography evaluation of the study population
<table><tr><td></td><td>Spinal ALS
n=29</td><td>Controls
n=13</td><td>p value</td></tr><tr><td>Male/female n (%)</td><td>20/9 (68/32)</td><td>6/7 (46/54)</td><td>0.06</td></tr><tr><td>Age (year)</td><td>60 [54-72]</td><td>46 [37-68]</td><td>0.06$</td></tr><tr><td>BMI (Kg/m2)</td><td>24.2± 5.2</td><td>26.3 ± 4.3</td><td>p=0.16#</td></tr><tr><td>FVC standing up (%)</td><td>67.5 ± 23.4</td><td>110.7 ± 12</td><td>< 0.001#</td></tr><tr><td>FVC lying 30° supine position (%)</td><td>59.4 ± 27.3</td><td>106.9 ± 13</td><td>< 0.001#</td></tr><tr><td>VC standing up(%)</td><td>71 [54-89]</td><td>108 [105-121]</td><td>< 0.001$</td></tr><tr><td>VC lying 30° supine position (%)</td><td>64 [41-73.5]</td><td>109 [99-113]</td><td>< 0.001</td></tr><tr><td>ΔFVC (%)</td><td>10[5-26.7]</td><td>4 [2-5]</td><td>< 0.001$</td></tr><tr><td>ΔVC (%)</td><td>12 [4-26]</td><td>4 [1-7]</td><td>p=0.02$</td></tr><tr><td>Diaphragmatic excursion during deep breath (mm)</td><td>52.3[32.3-62]</td><td>62.3 [54.6-70.9]</td><td>p=0.04$</td></tr><tr><td>TF%</td><td>44.5 [34.5-79]</td><td>67 [53-105]</td><td>p=0.05$</td></tr><tr><td>ΔT</td><td>0.8 [0.6-1]</td><td>1.3 [1.1-1.9]</td><td>< 0.001$</td></tr><tr><td>Ti (mm)</td><td>2.3 [2-3.2]</td><td>3.3 [2.7-4.6]</td><td>p=0.01$</td></tr><tr><td>Te (mm)</td><td>1.5 [1.3-2]</td><td>1.7 [1.6-2.2]</td><td>p=0.05$</td></tr></table>
ALS: Amyotrophic Lateral Sclerosis; BMI: Body Mass Index; FRC: Functional Residual Capacity; FVC: Forced Vital Capacity; PCF: Peak Cough Flow; SD: Standard Deviation; TF: Thickening Fraction; TLC: Total Lung Capacity; VC: Vital Capacity; Te: thickness at FRC; $\Delta T$: Thickness; Ti: Thickness at TLC.
* $p \leq 0.05$ (in bold) indicates significant differences
- ${}^{\#}T$ test expressed as mean $\pm {SD}$ (standard deviation)
- Wilcoxon test expressed as median [IQR]
As expected, PFTs and DU values were significantly different between the two groups, the most striking differences being $\Delta T$, VC and FVC in a lying $30^{\circ}$ supine and standing up position and the changes in FVC and VC when moving from standing to the supine position (i.e., three-fold greater in ALS patients).
### a) Correlation between $\Delta T$ and PFTs in both ALS patients and controls
As shown in Fig.1, in ALS, $\Delta T$ significantly correlates with VC and FVC, in standing up $(r = 0.58$
$\mathrm{p = 0.001}$; $\mathrm{r = 0.61}$ $\mathrm{p < 0.001}$ and lying $30^{\circ}$ supine position $(\mathrm{r} = 0.59$ $\mathrm{p = 0.001}$; $\mathrm{r} = 0.62$ $\mathrm{p < 0.001}$, respectively. A weak correlation was observed in controls between $\Delta T$ with VC and FVC, in standing up and lying $30^{\circ}$ supine position.
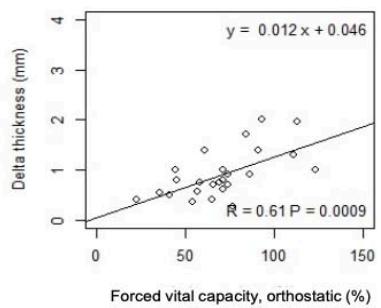
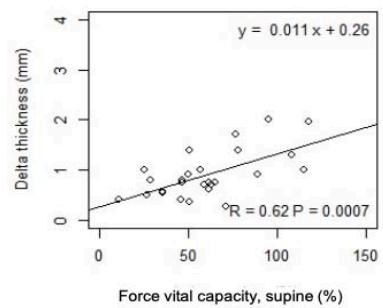
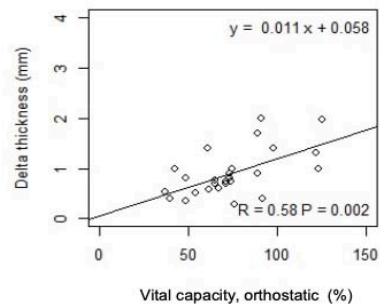
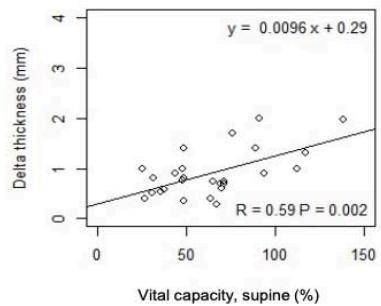
Fig.1: Correlation between $\Delta T$ and PFTs
### b) Correlation between TF and PFTs in both ALS patients and controls
As shown in Fig. 2, in ASL patients but not in controls, TF significantly correlates with VC and with
FVC in standing up (r=0.55 p=0.003; r=0.53 p=0.005) and lying $30^{\circ}$ supine position (r=0.55 p=0.003; r=0.51 p=0.007), respectively.
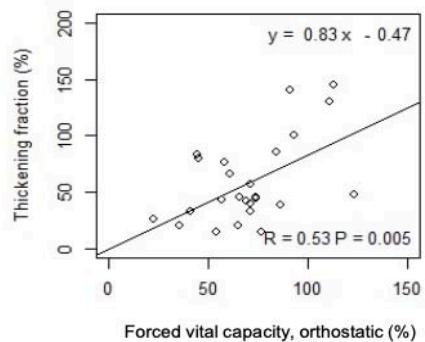
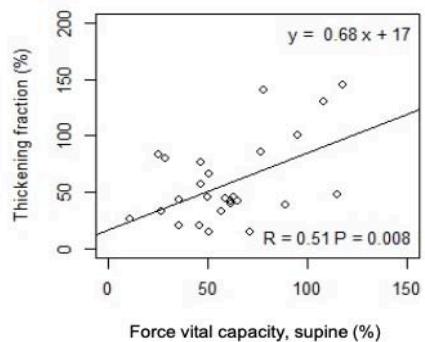
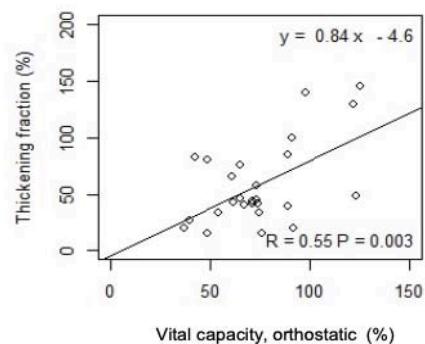
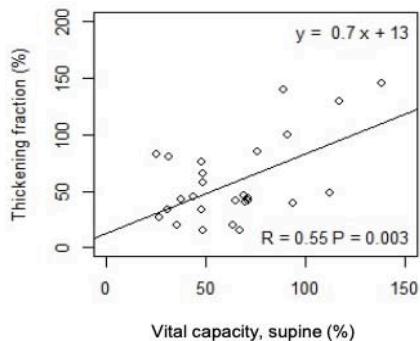
Fig. 2: Correlation between TF and PFTs
### c) Correlation between diaphragmatic excursion and PFTs in both ALS patients and controls
As shown in n Fig. 3, in ASL patients but not in controls, DE significantly correlates with VC and FVC in standing up (r=0.55 p= 0.007; r =0.65 p<0.001) and
lying $30^{\circ}$ supine position $(r = 0.59, p = 0.003$; $r = 0.63, p = 0.001)$, respectively. Moreover, in ALS patients, DE negatively correlated with the change in VC $(r = -0.54, p = 0.01)$ and FVC from standing up to lying $30^{\circ}$ supine position $(r = -0.50, p = 0.02)$.
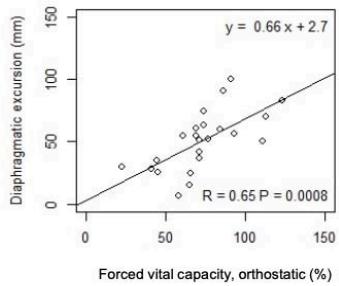
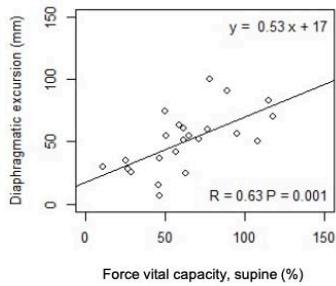
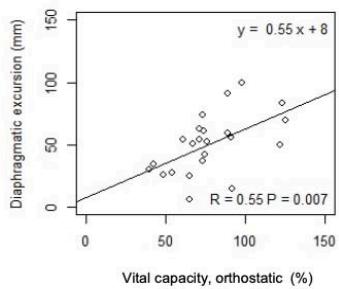
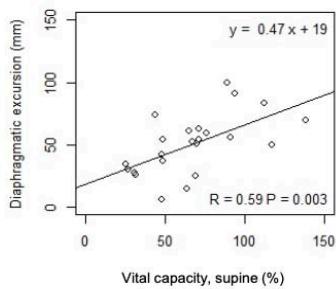
Fig. 3: Correlation between DE and PFTs
### d) ROC curve analysis: predictive value of DU indices for NIV timing initiation
In our cohort, 23 out of 29 patients had a standing up FVC $< 80\%$ of predicted. As shown in Fig.4, in those patients, the AUC for $\Delta T$, TF and
diaphragmatic excursion were 0.94 [0.87-1], 0.87 [0.69-1], 0.88 [0.72-1] respectively. The optimal cut-point found was $0.9 \mathrm{~mm}$ for $\Delta T$, $85\%$ for TF, and $56 \mathrm{~mm}$ for diaphragmatic excursion.
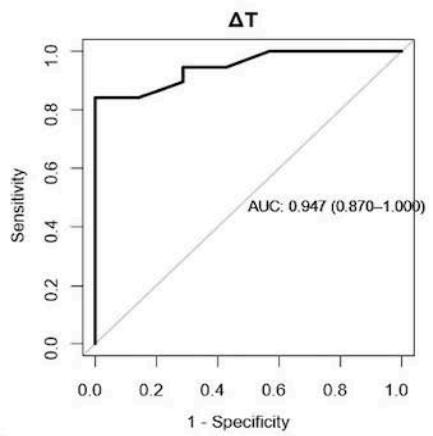
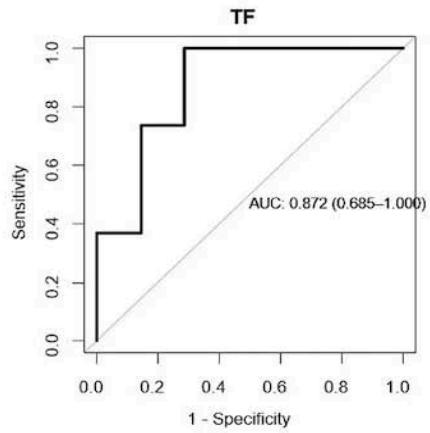
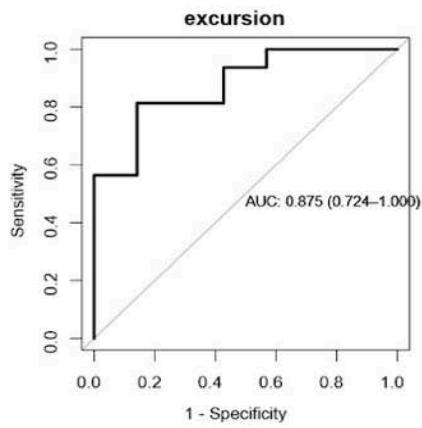
Fig. 4: ROC curve analysis: predictive value of DU indices for NIV timing initiation
## IV. DISCUSSION
Our study has demonstrated that VC and FVC measurements, in standing up and $30^{\circ}$ lying position, correlate with ultrasonographic diaphragmatic indices for assessing diaphragmatic dysfunction in patients with ASL and, perhaps more importantly, we found that DU indices predict with good accuracy the timing for NIV initiation. Of notice, neuromuscular diseases lead to an increased elastic load, increased alveolar surface tension and stiffness of the thoracic wall causing a reduction in maximal inspiratory strength and diaphragmatic fatigue. The best way to assess the mechanical properties of the diaphragm and its ability to generate pressure is the transdiaphragmatic pressure measured by magnetic phrenic nerve stimulation with simultaneous measurements of oesophageal and gastric pressures[17]. Because of invasiveness, $\Delta T$ measurement at DU has been proposed as an alternative non-invasive approach evaluating of diaphragmatic contractile reserve[18,19]. In fact, in line with Fantini et al. [8], we postulate that $\Delta T$ might express the maximal transdiaphragmatic pressure. Specifically, $\Delta T$ cannot measure directly the pressure generated by the diaphragm shortening, but can assess how the pressure generated by a maximum inspiratory effort can short the diaphragm itself. Although not significant, maybe because of the small sample, this trend of correlation is also preserved in controls. Otherwise, in our cohort of recently diagnosed ALS patients, although TF correlates well with lung volumes in both positions, we cannot consider it an index of early diaphragmatic fatigue[16]. TF may differ based on the presentation of the disease: in acute or subacute ALS, the diaphragm may have a preserved thickness with reduced excursion
due to prominent neuropathic damage[20]; on the contrary, with a more chronic course of the disease, the diaphragm becomes thin and atrophic with poor inspiratory thickening and may even become paradoxically thinner with inspiration[21]. Indeed, in our cohort, we found a reduced baseline diaphragm thickness (Te) compared to the control and, TF can vary greatly depending on the value of thickening at FRC. With this perspective, we can speculate that the role of TF is different between acute/subacute ALS, with early TF reduction and normal Te value, and chronic ALS, with a similar reduction in Ti and Te values until late stage, maintaining a normal TF value. According to our results, also Fantini et al. [16] show that TF is an index of late stage of diaphragmatic dysfunction, that is not useful for detecting early diaphragmatic dysfunction. Moreover, no significant correlation was found between TF and lung volumes in controls, because of the stability of lung volumes in both positions measured. We also have shown that diaphragmatic excursion measured during maximal inspiratory effort significantly correlates positively with all lung volumes and negatively in position changing. At the beginning of the disease, the diaphragmatic dysfunction expressed as muscle fatigue and reduced excursion, could be compensated by accessories respiratory muscle. In this way, if there is a diaphragmatic decreased capacity to generate force and reduced excursion, this is not translated into a reduction in respiratory function tests. When the disease progress in patients with ALS, the respiratory system elastic load is much higher than the muscle strength, leading to atelectasis, reduced lung elasticity, and, finally increased elastic load. Whit this scenario, there is a definitive patient deterioration with generalized muscle weakness and, finally, reduction of lung volumes. Based
on this finding, we could speculate that DU evaluation by transabdominal approach is a feasible and reproducible technique and integrate the transthoracic approach, better reflecting a direct and dynamic evaluation of this muscle, supporting physicians in an early subclinical diagnosis of diaphragmatic dysfunction in ALS patients. According to our results, Aliberti et al.[5] evaluated diaphragmatic excursion performed bedside confirming our data. Moreover, in ALS patients, the excursion is negatively correlated with the change in VC and FVC from standing up to lying $30^{\circ}$ supine position showing, confirming that more is the change between the two positions, less is the diaphragmatic excursion. In this study, we also investigated ROC analysis of all the DU indices (ΔT, TF, Excursion) to identify the cut-off point to predict FVC lower than $80\%$ of the predicted value, which is an indication for mechanical ventilation, following the current consensus guidelines[4,10]. Interestingly, when the ΔT is lower than $0.9 \mathrm{~mm}$ and the diaphragmatic excursion during a deep breath is lower than $56 \mathrm{~mm}$ predicts diaphragmatic dysfunction in patients who are not able or cooperative to perform respiratory functional tests, and could guide clinicians in the decision to start NIV. An important limitation of our study is the absence of a comparison between the DU measurement and the gold standard method used to assess its contractility, transdiaphragmatic pressure measured by phrenic nerve stimulation, because it was invasive and not easy to perform in routine clinical practice. Another considerable limitation is the small sample size, there was not easy to recruit vulnerable patients because of their physical and emotional frailty. Another limitation was the lack of inter and intravariability analysis of the DU performed. Finally, the absence of solid literature references about our indices needs further research to confirm and corroborate the feasibility of our findings.
## V. CONCLUSION
In summary, the relevance of finding out an index of early diaphragmatic dysfunction is mandatory in patients with neuromuscular diseases who cannot perform traditional lung function tests. If TF is affected by the baseline value of diaphragm thickness, the DU indices $\Delta T$ and diaphragmatic excursion are independent of this value and could provide specific information about muscle fatigue and an early diaphragmatic dysfunction before the exhausting muscle phase become clinically evident. This finding thus opens new perspectives on the use of this imaging technique in the early diagnosis and follow-up of diaphragmatic function impairment.
Acknowledgements: We are grateful to all the patients that accepted to participate in this study.
Conflict of Interest: None of the authors have potential conflicts of interest to be disclosed should appear in the paper.
Data Availability Statement: The data that support the findings are available in the Supplementary material.
Human/Animal Ethics Approval Declaration: The local ethics committee approved study protocol number 0897/2021 and written informed consent was obtained from all the subjects in the study.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Generating HTML Viewer...
References
21 Cites in Article
Lewis Rowland,Neil Shneider (2001). Amyotrophic Lateral Sclerosis.
Lewis Rowland (1998). Diagnosis of amyotrophic lateral sclerosis.
C Morelot-Panzini,G Bruneteau,J Gonzalez-Bermejo (2019). NIV in amyotrophic lateral sclerosis: The 'when' and 'how' of the matter.
P Andersen,S Abrahams,G Borasio,M De Carvalho,A Chio (2012). EFNS guidelines on the Clinical Management of Amyotrophic Lateral Sclerosis (MALS) -revised report of an EFNS task force.
Stefano Aliberti,Grazia Messinesi,Andrea Gramegna,Lucio Tremolizzo,Emanuela Susani,Alberto Pesci (2013). Diaphragm ultrasonography in the management of patients with amyotrophic lateral sclerosis.
Stephen Bourke,Mark Tomlinson,Tim Williams,Robert Bullock,Pamela Shaw,G Gibson (2006). Effects of non-invasive ventilation on survival and quality of life in patients with amyotrophic lateral sclerosis: a randomised controlled trial.
Y Hiwatani,M Sakata,H Miwa (2013). Ultrasonography of the diaphragm in amyotrophic lateral sclerosis: Clinical significance in assessment of respiratory functions.
Riccardo Fantini,Jessica Mandrioli,Stefano Zona,Federico Antenora,Andrea Iattoni,Marco Monelli,Nicola Fini,Roberto Tonelli,Enrico Clini,Alessandro Marchioni (2016). Ultrasound assessment of diaphragmatic function in patients with amyotrophic lateral sclerosis.
Benjamin Brooks,Robert Miller,Michael Swash,Theodore Munsat (2000). El Escorial revisited: Revised criteria for the diagnosis of amyotrophic lateral sclerosis.
M Miller,R Crapo,J Hankinson,V Brusasco,F Burgos,R Casaburi,A Coates,P Enright,C Grinten,P Gustafsson,R Jensen,D Johnson,N Macintyre,R Mckay,D Navajas,O Pedersen,R Pellegrino,G Viegi,J Wanger (2005). General considerations for lung function testing.
J Wanger,J Clausen,A Coates,O Pedersen,V Brusasco,F Burgos,R Casaburi,R Crapo,P Enright,C Van Der Grinten,P Gustafsson,J Hankinson,R Jensen,D Johnson,N Macintyre,R Mckay,M Miller,D Navajas,R Pellegrino,G Viegi (2005). Standardisation of the measurement of lung volumes.
Y Yoshioka,A Ohwada,M Sekiya,F Takahashi,J Ueki,Y Fukuchi (2007). Ultrasonographic evaluation of the diaphragm in patients with amyotrophic lateral sclerosis.
A Sarwal,F Walker,M Cartwright (2013). Neuromuscular ultrasound for evaluation of the diaphragm: Ultrasound of Diaphragm.
Dimitrios Matamis,Eleni Soilemezi,Matthew Tsagourias,Evangelia Akoumianaki,Saoussen Dimassi,Filippo Boroli,Jean-Christophe M. Richard,Laurent Brochard (2013). Sonographic evaluation of the diaphragm in critically ill patients. Technique and clinical applications.
Riccardo Fantini,Roberto Tonelli,Ivana Castaniere,Luca Tabbì,Maria Pellegrino,Stefania Cerri,Francesco Livrieri,Francesco Giaroni,Marco Monelli,Valentina Ruggieri,Nicola Fini,Jessica Mandrioli,Enrico Clini,Alessandro Marchioni (2019). Serial ultrasound assessment of diaphragmatic function and clinical outcome in patients with amyotrophic lateral sclerosis.
B Higgs,Of,Pressure (1989). MEASUREMENT OF TRANSDIAPHRAGMATIC PRESSURE.
C Roussos,P Macklem (1977). Diaphragmatic fatigue in man.
F Bellemare,B Bigland-Ritchie (1987). Central components of diaphragmatic fatigue assessed by phrenic nerve stimulation.
Andrea Boon,Kais Alsharif,C Harper,Jay Smith (2008). Ultrasound‐guided needle EMG of the diaphragm: Technique description and case report.
Andrea Boon,Cullen Oʼgorman (2016). Ultrasound in the Assessment of Respiration.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
F. Viccaro. 2026. \u201cAgreement Between Diaphragm Ultrasound and Pulmonary Function Testing in the Evaluation of Diaphragm Function in Patients with ALS\u201d. Global Journal of Medical Research - F: Diseases GJMR-F Volume 23 (GJMR Volume 23 Issue F4): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.