## I. BACKGROUND
The incidence of breast ductal Carcinoma in Situ (DCIS) currently represents up to $20 - 25\%$ of all breast carcinomas but its management remains controversial (1). In ductal carcinoma in situ of the breast (DCIS), histological diagnosis obtained before surgical treatment carries the risk of under staging the disease if the presence of invasive cancer is then found postoperatively. These patients need a second operation to assess the nodal status. Assessing the risk of under staging DCIS patients with even small lesions presenting as a mass would be helpful for optimal treatment planning. This should be considered in the decision-making process with regards to the extent of surgical intervention (2).
The overall natural progression of DCIS to invasive malignancy is reported to range from 14 to $75\%$. Histopathologic diagnosis of breast cancer should be obtained before the definitive treatment using minimal-invasive investigations. However, when invasive ductal cancer (IDC) is subsequently found in postoperative specimens these patients often need a further operation, usually a sentinel node biopsy to assess the nodal status.
Among the radiological features a mammographic extent of more than $4 - 5\mathrm{cm}$ and the presence of architectural distortion, focal asymmetric density or mass on mammography are proven important risk factors of final histological upstaging of DCIS to invasive carcinoma. In some reports, a mass lesion visible on ultrasound can be significantly related to the risk of upstaging DCIS to invasive ductal carcinoma. The cut-off points of lesion size that makes a distinction between low and high risk DCIS are still subjects of debate (3).
In the majority (62%–98%) of cases, DCIS is detected due to the presence of calcifications at mammography. In 2%–23% of cases, DCIS may manifest as a mass or asymmetry. The ultrasonographic (US) features of DCIS can be subtle and nonspecific however advances in US technology have improved the ability not only to characterize mammographic masses and asymmetries, but also to detect calcifications. Alongside increased implementation of US for screening and for targeted evaluation of breast magnetic resonance (MR) imaging abnormalities, recognizing the US features of DCIS has become increasingly important (4).
Encysted papillary cancer (EPC) is a rare form of breast cancer accounting for approximately $(1 - 2\%)$ of all breast tumors, usually presenting in postmenopausal women. The prediction of the biological behavior of this rare form of breast cancer and the clinical outcome is associated with an overall favorable prognosis;
however, its consideration as a form of ductal carcinoma in situ with non-invasive nature is unclear as it has been shown to present histologically with invasion of basement membrane and even metastasis (5).
Both DCIS and encysted papillary carcinomas are pathologically non-invasive but clinically have a risk of metastasising to axillary lymph nodes, presumably due to a pathological sampling error. Therefore, offering sentinel lymph node biopsy (SLNB) to this pre invasive carcinomas category when performing breast conserving surgery is controversial. Initial SLNB should be considered for patients diagnosed with DCIS by needle biopsy when they have a high risk for harbouring invasive ductal cancer preoperatively (6). However, performing a sentinel lymph node biopsy also carries the risk of potentially unnecessary additional surgery with associated comorbidity.
## II. METHODS
The source of the data for the EPC was via a Winpath search from January 2014 to March 2019 using the term 'papillary carcinoma'. This produced 85 results, however from this a number of cases including micropapillary carcinoma and papilloma that were not EPC's were excluded. The results included two entries, one for the biopsy (EPC/B5a) and one for the subsequent excision. The search also found 3 cases of EPC on excision that initially were called papillary lesions/B3/B4. Additionally, 5 cases were excluded due to arising from background/neighbouring EPC's. In total 29 patients were identified to have EPC on biopsy.
The source of data for the DCIS again utilised a search on WinPath using the term 'DCIS' from January 2013 to April 2019 for all core biopsies including VABs. This generated a total of 637 patients of which any patients with invasive carcinoma or patients with a focus of invasion in the initial core biopsy were then excluded. This left 388 patients who were then inputted into an Excel spreadsheet. The following details were then included: referral route (screening, symptomatic or incidental), Clinical score (P score), Radiological score (R and U), imaging size (mm), presentation (mass forming DCIS or micro calcification only), high risk of occult invasion (mass forming or microcalcification of 40mm or more), date of surgery, details of surgery, excision diagnosis, final tumour size, SLNB performed, SLNB results. This data was collected from Maxims (hospital electronic patient record software).
## III. RESULTS
### a) Encysted papillary carcinoma results
Of the 29 patients found to have EPC on core, 16 (55%) were found to be EPC on excision, while 9 (31%) were found to be invasive carcinoma on excision. A further 4 (14%) had no further surgery after the core (Table 1).
Table 1
<table><tr><td>EPC on core</td><td>EPC on excision</td><td>Invasive carcinoma on excision</td><td>No further surgery after core</td></tr><tr><td>29</td><td>16 (55%)</td><td>9 (31%)</td><td>4(14%)</td></tr></table>
From the EPC patient group, $72\%$ of the patients were symptomatic on presentation while $28\%$ were found on screening. Of those presenting all were female and their ages ranged between 51 and 91. The median age of presentation was 71.
Of the EPC patients, 5 (21%) underwent a mastectomy, 19 (65%) had a local wide excision and 4 (14%) underwent no surgery. Within the patient group undergoing no surgical intervention for EPC, 3 (75%) had PET and 1 (25%) had no treatment.
Within the EPC patient group, 5 of the patients underwent mastectomy of which all had SLNB at the time of operation as per protocol. Of the remaining 20 patients, 5 had SLNB at the time of operation, 5 (40%) had SLNB at a later stage and 10 never underwent SLNB. Of the SLN biopsies for EPC, 2 of the 15 (13%) who underwent SLNB were found to have positive nodes (Figure 1) Of those who had mastectomy, 2 of the 5 (40%) had positive nodes. Of those who underwent breast conserving surgery, none of the patients had positive nodes.
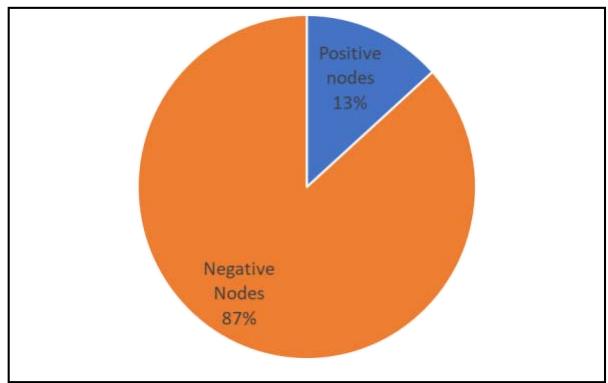 Figure 1: SLN results for EPC
### b) DCIS data analysis
Of the 396 patients with DCIS, 188 (48%) were found to be high risk, while 206 (52%) were found to be low risk. Within this group the age range was between 33 and 83 with a mean age of 66, all of which were female. 130 (69%) of the high risk patient group were found from sampling, 50 (27%) were symptomatic and a further 8 (4%) were incidental.
Of the high-risk patients, 114 (61%) underwent mastectomy, 60 (32%) had BCS and 14 (7%) had no surgical intervention. 75 (40%) of the total high risk patients had a simple mastectomy, 39 (21%) had a mastectomy and immediate Recon, 36 (19%) had a WLE and 19 (20%) had a total mastectomy.
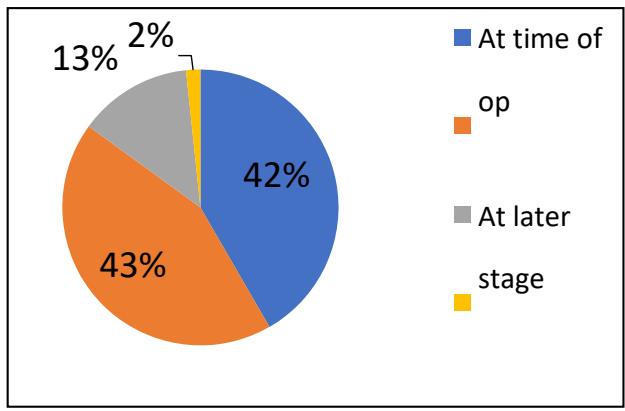 Figure 2: SLNB for high risk DCIS + BCS population
Within the patient group that underwent no surgical intervention, 6 (44%) received primary endocrine therapy (PET) either due to comorbidities or patient choice. Final excision histology for high risk DCIS (post operative) found that $38\%$ were found to have invasive disease, whereas $13\%$ of the low risk DCIS group were found to have invasive histology.
Within the high-risk DCIS group, 139 (80%) had a SLNB at the time of the operation and a further 8 (5%) had an SLNB later (Figure 4). Of the total high-risk patients (188) there were 114 who underwent mastectomy and thus had SNLB at the time of the operation. Of the 60 who had breast conserving surgery, 33 (55%) underwent SNLB either at the time of operation or at a later stage. This leaves however 26 patients (43%) who never received a SLNB (Figure 2).
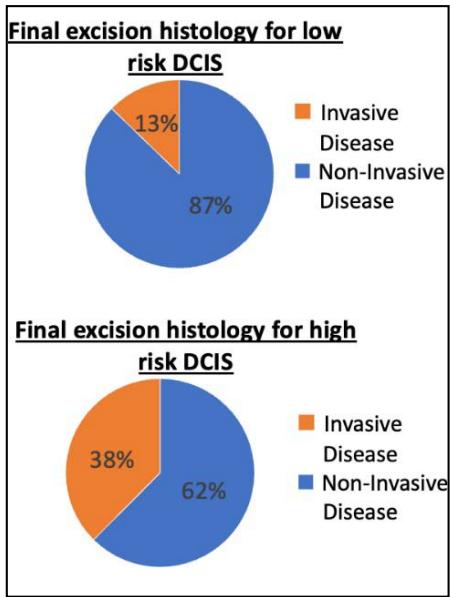 Figure 3: Post Histology results for DCIS groups
Of these patients, 23 of the 26 $(87\%)$ were found to have no invasive disease on final histology hence had no SLNB (Figure 3).
The results of the SNLB within the high-risk DCIS population revealed 15 patients had a positive SLNB which represents $7.9\%$ of the entire high risk patient population (Figure 3). Their age range was between 43 years to 81 years old (mean age 65 years). 9 patients presented symptomatic while a further 6 were screen detected. 11 patients were identified to have mass forming DCIS and 4 were found to have macrocalcification of more than $40\mathrm{mm}$ on investigation. These high-risk DCIS patient groups surgical procedures consisted of: 11 mastectomies, 3 WLE and one TM. 14 of these patients had SLNB at time of initial operation whereas one patient had a subsequent SLNB. This single patient was 58 years old and initially diagnosed with $8\mathrm{mm}$ mass forming DCIS on imaging and core biopsy. He was recorded as P1 R3 U4 and initially underwent WLE. This resulted in an excision diagnosis of G2 hence subsequent SLNB and positive results.
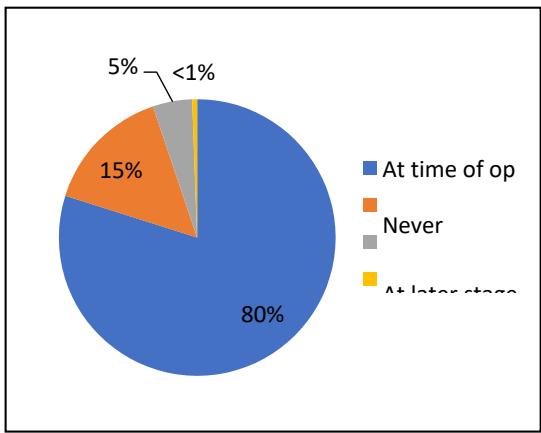 Figure 4: SLNB undertaken for high risk DCIS patient group
## IV. DISCUSSION
### a) EPC Analysis
EPC analysis from the pathology department Audit we found that the provisional diagnosis of encysted papillary carcinoma was upgraded to an invasive carcinoma on final histology in around a third of cases. This has an implication for surgical planning with regards to SLNB offering at the time of surgery. According to our Audit data, the age of patients with EPC ranges from 51- 91year-old women, with a mean age of 72, which is a little older than the literature suggests and gave an older mean age for patients with DCIS. EPC is most commonly found in postmenopausal women (7). In our Audit data we found $72\%$ were symptomatic and $28\%$ were screen detected and this is in agreement with other published data. Encysted papillary carcinoma presents as a palpable lump in 70- $80\%$ of cases and then less commonly as nipple discharge or incidental finding (8). In our Audit analysis we found $70\%$ were symptomatic with either lumps or nipple discharge. However, we found that nearly $30\%$ presented via screening.
Treatment of EPC consists of surgery (wide local excision or mastectomy) $\pm$ radiotherapy (RT) $\pm$ hormonal treatment. The question of an axillary procedure remains unanswered with some series reporting the metastatic potential of EPC and recommending sentinel lymph node biopsy (SLNB) (7). Our data analysis showed that, the surgical treatments for this group were mostly BCS $(65\%)$, mastectomy $(21\%)$ and $14\%$ had no surgery. The patients not undergoing surgery received primary endocrine treatment due to associated medical co-morbidities or had no active treatment as oestrogen/progesterone receptors were negative.
Patients with EPC require sentinel lymph node biopsy (SLNB) for staging of the axilla, only if there is evidence or suspicion of invasion in their core biopsy or final histology specimen or if they are undergoing a mastectomy as their primary treatment (9). When mastectomy was planned, SLNB's were offered to all 5 patients. Of the 20 patients who had BCS, 5 of them had a SLNB at the time of operation and another 5 had an SLNB at a later date (due to finding invasive components at final histology).
From these sentinel node biopsies, it was found that $87\%$ were negative for nodal metastasis and $13\%$ were positive for nodal metastasis. Of those undergoing BCS, 0 of the 9 patients had positive nodes.
### b) DCIS analysis
According to the considered literatures we defined the high risk of invasion in this group as follows: Extensive micro-calcification $>40\mathrm{mm}$, or any size mass forming DCIS. After applying this definition, we found that, 188 (48%) were high risk of invasion.
Within the group all were females, and their ages ranged from 33 to 88, with a mean age of 66. As expected, the majority were screen detected DCIS (69%), versus 27% symptomatic and only 4% were incidental at their presentation. We found that the surgical treatments in this group were mostly mastectomy in about 62% of cases including both simple mastectomies and immediate breast reconstruction. Breast conserving surgery made up around $32\%$, including wide local excision, therapeutic mammoplasty, quadrantectomy and excision biopsies for diagnosis and proofed therapeutic treatment. Finally, around $7\%$ had no surgical intervention, this because they had primary endocrine treatment for either co morbidity or patient's choice. On final post operative histology, we found, out of those who were classed as high-risk DCIS about $38\%$ were found to have invasive disease on final histology. This compares to $13\%$ of "low risk DCIS" that were found to have invasive disease on final histology.
According to the literature, initial SLNB should be considered for patients diagnosed with DCIS by needle biopsy when they have a high risk for harbouring invasive ductal cancer preoperatively (10). Patients in whom a preoperative diagnosis of DCIS is likely to be upgraded to invasive carcinoma will benefit from SLNB biopsy being performed with the initial surgery (11). Therefore, in these patients' group, we found 139 (80%) had SLNB at time of operation, 8 (5%) had SLNB at later stage, 26 (15%) never had a SLNB and 1 (less than 1%) declined the SLNB.
114 patients underwent mastectomy and had SLNB at time of operation as per protocol and guidelines. Of the 60 who had breast conserving surgery; 25 (42%) had SLNB at time of operation, 26 (43%) never had SLNB, 8 (13%) had SLNB at a later stage (all these patients were found to have invasive cancer on excision histology), only 1 patient (2%) declined the SLNB.
Despite a $23\%$ upstaging rate, the rate of clinically significant positive SLNs in patients treated with BCS is low, supporting omission of upfront SLNB (30). Similarly, we found in our study that among the patients who had an axillary assessment, their histology results were mostly negative $(92.1\%)$, however of the patients with high-risk DCIS, 15 $(7.9\%)$ had a Positive SLNB. 11 out of these 15 patients had a mastectomy as their operation. This indicated a large DCIS size subset that necessitates a mastectomy. With the new technique of partial breast reconstruction SLNB should be offered in those cases to be able to predict the risk of invasion and even involved lymph node with metastatic carcinoma. Further analysis of the high-risk group, either extensive microcalcification or mass forming DCIS showed, of those 113 who had extensive microcalcifications 88 had SLN $(78\%)$ and 4 had positive nodes $(4\%)$. Of those 82 who had mass forming, 40 had SLN $(49\%)$ and 4 had positive nodes $(4\%)$. The total percentage of positive lymph nodes in these groups were about $8\%$. Regarding mass forming DCIS, we found $65\%$ had SLNB and $35\%$ didn't have SLNB, of those who had SLNB, $5\%$ had positive nodes and just above $60\%$ had negative nodes and $34\%$ didn't have SLNB at all. Of the cohort that were screen detected masses, $33\%$ had SLNB and of those $5\%$ had a positive node. Additionally, patients who had high risk DCIS detected incidentally, $50\%$ had SLNB and none had positive nodes histologically.
## V. CONCLUSIONS
Within the encysted papillary carcinoma group the overall results showed that the provisional diagnosis of encysted papillary carcinoma is upgraded to an invasive carcinoma on final histology in around a third of cases. This may have an implication when deciding surgery and sentinel node biopsy should be offered at the time of therapeutic surgery. We should offer SLNB to patients with large sized EPC, which necessitate a mastectomy (tumour size is $40\mathrm{mm}$ or above on imaging).
In terms of the risk of invasive ductal carcinoma in situ (DCIS), in this Audit, we have defined a 'high risk' group of patients with pre-op diagnosis of non-invasive cancer undergoing BCS, who would benefit from SLNB at the time of the surgery. These are those with extensive microcalcifications of more than $40~\mathrm{mm}$ or any size mass forming DCIS. When counselling patients within this group the risk of them having invasive disease in final histology is up to $40\%$ but the risk of sentinel node involvement is approximately $8\%$.
## RECOMMENDATIONS
As a result of this audit, SLNB is offered at the time of initial surgery to the patients with high-risk DCIS and EPC to avoid further operations at later stage, which may result in increased patient emotional burden and higher service cost.
Audit Registration/Approval
Declarations of interest: None
Generating HTML Viewer...
References
10 Cites in Article
G Lissidini (2019). Breast Ductal Carcinoma In Situ (DCIS): Open controversies and Guidelines Update of the European Institute of Oncology (EIO).
Bartlomiej Szynglarewicz (2015). preoperatively diagnosed ductal cancers in situ of the breast presenting as even small masses are of high risk for the invasive cancer foci in postoperative specimen.
G Leonard,S Swain (2004). Ductal Carcinoma In Situ, Complexities and Challenges.
Lilian Wang,Megan Sullivan,Hongyan Du,Marina Feldman,Ellen Mendelson (2013). US Appearance of Ductal Carcinoma in Situ.
G Lissidini,G Curigliano,C Leonardi,B Bonanni,R Lo Gullo,P Veronesi,G Farante (2019). Breast Ductal Carcinoma In Situ (DCIS): Open controversies and Guidelines Update of the European Institute of Oncology (EIO), Milan.
Judicael Hotton,Julia Salleron,Philippe Rauch,Julie Buhler,Marion Pierret,Florian Baumard,Lea Leufflen,Frederic Marchal (2019). Predictive factors of axillary positive sentinel lymph node biopsy in extended ductal carcinoma in situ treated by simple mastectomy at once.
Y Uemoto,N Kondo,Y Wanifuchi-Endo,T Hisada,S Nishikawa,Y Katagiri,H Kato,S Takahashi,T Toyama (2019). Abstract P3-03-34: Sentinel lymph node biopsy is unnecessary in ductal carcinoma <i>in situ</i> patients diagnosed by biopsy.
Nicole Nicosia Esposito,David Md,M Dabbs,Rohit Bhargava,Md (2020). Are Encapsulated Papillary Carcinomas of the Breast In Situ or Invasive? A Basement Membrane Study of 27 Cases.
Melissa Pilewskie,Maria Karsten,Julia Radosa,Anne Eaton,Tari King (2016). Is Sentinel Lymph Node Biopsy Indicated at Completion Mastectomy for Ductal Carcinoma In Situ?.
Andrea Barrio,Kimberly Van Zee (2017). Controversies in the Treatment of Ductal Carcinoma in Situ.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Mona Sulieman. 2026. \u201cAn Audit on the Role of Sentinel Lymph Node Biopsy (SLNB) in High Risk of Invasion DCIS and Encysted Papillary Carcinoma\u201d. Unknown Journal GJMR-I Volume 23 (GJMR Volume 23 Issue I2): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.