Antihyperlipidemic Property of a Dietary Supplement of Moringa Oleifera Leaves and Pleurotus Ostreatus in Wistar Rats Stressed by Combination of Ethanol-Paracetamol.
## I. INTRODUCTION
Alcoholism and other serious health issues are brought on by excessive alcohol usage, including alcoholic liver damage (ALD). Alcoholism has been linked to several illnesses, and it is currently one of the most challenging health issues with substantial medical, social, and economic repercussions. (Pari and Karthikesan, 2001; Sivaraj et al., 2010). Alcohol abuse leads to significant illnesses such as hyperglycemia, cirrhosis, cardiovascular disease, pancreatic inflammation, and alcoholic fatty liver. (Ponnappa et al., 2000). Oxidative stress is one of the elements that are crucial in numerous pathways of alcohol-induced harm. The creation of ROS in our bodies is abnormally increased by our unhealthy eating habits and our way of life (smoking, drinking, obesity, and strenuous activity). When organisms experience oxidative stress brought on by free radical damage, antioxidants aid in coping. Antioxidant defenses come from two different sources: the diet, which includes fruits and vegetables, which are rich in vitamins C and E, carotenoids, ubiquinone, polyphenols, and lipoic acid. The other is endogenous and is made up of proteins, enzymes, or tiny molecules such as glutathione, uric acid, superoxide dismutase, and glutathione peroxidase (ferritin, transferrin, etc.). Additionally, some elements that are significant cofactors include selenium, copper, and zinc. (Pincemail et al., 2009).
We were particularly interested in the plant Moringa oleifera and the edible fungus Pleurotus ostreatus because they contain significant antioxidant content. The plant Moringa oleifera Lam. (Moringaceae), also known as Nebeday in Senegal, is of Indian ancestry and is now common throughout Asia and Africa. The leaves are utilized in traditional African medicine and are commonly consumed as a legume. They are an excellent source of protein (19–35% dry matter) (Kane et al., 2017; Makkar et al., (1996); Abou-Elezz et al., (2012) and are rich in metabolizable energy (2273–2978kcal/kg DM) (Makkar et al., (1996); Olugbemi et al., 2010). They are also rich in vitamins (A, B, C, and E), minerals (0.6-
11.2% dry Matter). The South African ecotype of the plant has been observed to contain 19.3% crude protein (Moyo., 2011). Traditional Chinese medicine uses M. oleifera leaves to treat diabetes, headaches, fever, and malnourishment (Ndong et al., 2007; Kerrarho., 1994).
Preview studies have shown the health and nutritional interest of edible mushrooms (Zhang et al., 2016; Alam et al., 2008; Pornariya and Kanok, 2009). P. ostreatus has been demonstrated for its antitumor effects, antioxidant properties, antihyperlipidemic effects and antidiabetic effects, (Zhang et al., 2016; Abrams et al., 2011; Alam et al., 2008; Elmastas et al., 2007; Jayakumar et al., 2007; Jayakumar et al., 2006). A daily intake of $15\mathrm{g}$ of dried oyster mushrooms would have an anti-hyperlipidemic effect on the subjects, it would also cover up to $50\%$ of the recommended daily intakes of macronutrients and minerals, according to research on Pleurotus ostreatus nutritional value and antihyperlipidemic effects on HIV-positive individuals taking ARVs (Abrams et al., 2011, Alam et al., 2008; Manzi et al., 2001; Alam et al., 2009; Manzi et al., 1999, Kane et al., 2017).
Given the rich nutrient, phytochemical, and organoleptic potential of M. oleifera and P. ostreatus, we designed this study to determine the antihyperlipidemic effect of Moringa oleifera leaves and Pleurotus ostreatus in Wistar rats stressed by a combination of Ethanol-paracetamol. In this paper, wewill code the dietary supplement by FMP16.
## II. MATERIALS AND METHODS
a) Plant Material and Preparation of the mixture of Leaves from M. oleifera and P. ostreatus
The fresh leaves of M. oleifera were harvested at the botanical garden of the University Cheikh Anta Diop (UCAD) of Dakar, Senegal and identified at the botanical department (UCAD). The leaves were cleaned immediately after harvest, cut into small pieces, and dried in the shade for two weeks. The dried material was ground into a powder using a manual homogenizer. P. ostreatus were obtained by cultivation at the biotechnological laboratory of the University Cheikh Anta Diop of Dakar. The Moringa oleifera and Pleurotus ostreatus powders were combined in a 2:1 ratio to create the dietary supplement. The mixture was created following Kane et al., instructions (2017). The combination was dissolved in $0.01\%$ starch paste before being fed to the rats.
### b) Animals and grouping
Wistar rats strain to weigh 150 to $200\mathrm{g}$ were obtained from the Animal House of the National Institute of Youth and Sports in Yaounde. They were placed in plastic cages under standard laboratory conditions (temperature 20 to $30^{\circ}\mathrm{C}$, relative air humidity 45 to $55\%$, and 12/12h light/dark cycle). The rats were fed with a basal diet and water ad libitum. The feed was a standard rat chow composed of carbohydrates (52%), protein (22%), fat (6.5%), water (12%), ash (6%), and fiber (4.5%). Every two days for 21 days, between 10:00 and 11:00 am, before the mixture administration of Moringa oleifera and Pleurotus ostreatus in proportion 2:1, made as reported by Kane et al., the amount of food and water ingested by each group of rats as well as body weights were recorded (2017). The experiments were performed during the day (09am-03pm).
### c) Experimental design
Thirty (30) adult male and female Wistar rats weighing 150 to $200\mathrm{g}$ were separated into five groups of six after two weeks of acclimatization:
- Group 1 (TG): a stress-free control group that consumed only their regular diet of water, food, and vehicle (starch paste) once daily for 21 days,
- Group 2 (TP): a control group that received paracetamol 12 hours after ethanol administration, was supplied in five sequential doses of $2 \mathrm{~g}. \mathrm{kg} - 1$ using an orogastric tube to stress the group. For 21 days, they consumed the standard diet of water and food at their leisure in addition to the vehicle starch paste,
- Group 3 (D1P): a group that received $500 \mathrm{mg} / \mathrm{kg}$ of FMP16 and was stressed by ethanol in five sequential doses of $2 \mathrm{~g}. \mathrm{kg}^{-1}$, administered through an orogastric tube; then received paracetamol $12 \mathrm{~h}$ after the last dose of ethanol. They received the standard diet (water and food ab-libitum) and the vehicle starch paste once a day for 21 days,
- Group 4 (D2P): a group that received $1000 \mathrm{mg} / \mathrm{kg}$ of FMP16 and was stressed by ethanol in five sequential doses of $2 \mathrm{~g}. \mathrm{kg}^{-1}$, administered through an orogastric tube; then received paracetamol $12 \mathrm{~h}$ after the last amount of ethanol. They received the normal diet (water and food ab-libitum) and the vehicle starch paste once a day for 21 days,
- Group 5 (D3P): a group that received $1500 \mathrm{mg} / \mathrm{kg}$ of FMP16 and was stressed by ethanol infive sequential doses of $2 \mathrm{~g}. \mathrm{kg}^{-1}$, administered through an orogastric tube; then received paracetamol $12 \mathrm{~h}$ after the last dose of ethanol. They received the standard diet (water and food ab-libitum) and the vehicle starch paste once a day for 21 days. The rats were given full access to food and water, and were on 12-hour light cycle each day (dark $12 \mathrm{~h}-12 \mathrm{~h}$ light). They were force-fed FMP16 using a gastroesophageal catheter and weighed every day. They fasted for the entire day before the animal sacrifice.
On the 23rd day, the rats were given a night of rest before being slaughtered (while sedated with ether) by having their jugular veins cut. Organs such the liver, kidneys, brain, and testicles were collected along with blood. The liver, which was exclusively used in this study, was wrung out, weighed, and stored at $-20^{\circ}\mathrm{C}$ until processing day. It was then rinsed with ice-cold saline $(0.9\% \mathrm{NaCl})$ to eliminate any remaining blood.
d) Determination of the biochemicals parameters in liver
- Preparation of liver supernates
Prior to biochemicals analysis, each liver sample was homogenized using a Poterproctor placed on ice and $10\%$ homogenate was prepared using the KCL buffer solution $(1.15\%)$. The homogenates were centrifuged at $3000~\mathrm{rpm}$ for $30\mathrm{min}$ at $4^{\circ}\mathrm{C}$ to collect the supernatant used for analysis. The supernatant of each sample was aliquoted in $1.5\mathrm{ml}$ Eppendorf tubes to estimate the activity of antioxidant parameters (peroxidized lipids LPO, glutathione cellular GSH, catalase CAT). All liver parameters were expressed as activity per mg proteins. The proteins concentration in each fraction were determined by the method of (Gornall et al., 1949)
- Determination of biochemical parameters
- Using the method of thiobarbituric acid-reacting substances, the mean malondialdehyde (MDA) level (mol/mg protein), a measure of lipid peroxidation, was evaluated (Singh et al., 2014).
- The level of catalase activity was assayed by the method of Sinha (1972).
- The level of Glutathionecellular activity was evaluated by the method of Ellman (1959).
- Serum glutamyl oxaloacetate transaminase (SGOT) and serum glutamyl pyruvate transaminase (SGPT) activities were assayed by the method of Karmen et al., (1955) and measured by standard essay kits SGM Italia Rome, Via Eschilo, 10139, (2012).
- The albumin level was assayed by the method of Ferreria& Price (1974) and measured by standard essay kits Hospitex diagnostics, Via Arno, 4001010L, (2013). Creatinin level was assayed by the method of Bergmeyer (1987) and measured by standard essay kits Hospitex diagnostics, Via Arno, 4001621L (2014).
- Testosterone level was assayed by the method of Tietz, (1986) and measured by Kit ELISA (DRG Diagnostics, Germany, EIA- 1559, (2009).
- Total Cholesterol level was assayed by the method of Allain et al, (1974) and measured by standard essay kits Hospitex Diagnostics, Via Arno, 4001210L, (2011).
- HDL Cholesterol level was assayed by the method of Grove (1979) and measured by standard essay kits SGM Italia, 10176, (2009).
- Triglycerides level was assayed by the method of Babblok et al., (1988) and measured by standard essay kits Fortress Diagnostics, United kingdom, BXC0271, (2013).
### e) Statistical Analysis
IBM SPSS Statistics 20 software was used for statistical analysis and data processing. P-values less than 0.05 were regarded as significant in the statistical analysis, which was conducted using one-way analysis of variance (ANOVA) and Bonferroni's post-test for multiple comparisons. The results are presented as the mean and standard deviation (SD).
## III. RESULTS AND DISCUSSION
Results have shown that no significant difference was observed in final body weights (155- 173g) (Table1). Body weight gain ranged between 0.8 and $19\mathrm{g}$ for the four treatment groups. A decrease of $10\%$ in the weight of D3N group was observed. These results corroborate those of Alam et al., (2011, 2009) who found that a diet enriched with Pleurotus ostreatus decreases the body weight of animals. However, Bobek et al., (1998) have shown that it does not affectthe weight as well as Schneider et al., (2011) who worked on humans with a daily dose of $30\mathrm{g}$ of dried oyster mushrooms, found that this does not affect anthropometric data. Bénissan et al., 2012, showed that the daily intake of $30\mathrm{g}$ of Moringa oleifera leaves improves nutritional recovery in children suffering from malnutrition. Hanaa et al., (2014), showed that a dose of $600~\mathrm{mg / kg}$ of Moringa oleifera lowers the body mass index in obese subjects. Furthermore, the mixture of these species at a high dose of $1500~\mathrm{mg / kg}$ would explain the weight loss. This result was in contrast with those of Osman et al. (2012), who reported up to $14\%$ changes in body weight of rats given M. oleifera extract for 21 days, attributing these changes to the rich nutrient quality of the extract.
Results also have shown no significant difference in the amount of protein in the liver (figure1). Regarding lipid peroxidation (figure 2), results show no significant difference in the concentration of peroxidized lipids between the groups except between the unstressed control group (TG) and the $1500\mathrm{mg / kg}$ dose group where the concentration was $34\%$ higher. These results are not in agreement with those of Mladenovic et al, (2013); Patere et al, (2011); Johnsen et al, (2007). The effect of FMP16 on oxidative stress enzymes such as catalase and cellular glutathione was also studied. Our results showed an increase in catalase activity of $87\%$, $85\%$, $90\%$, $82\%$ respectively for the TG, D1P, D2P and D3P groups compared to the TP group (intoxicated and untreated). Also, catalase activity of the $1000\mathrm{mg / kg}$ dose (D2P) was $35\%$ and $43\%$ higher respectively compared to the 500 and $1500\mathrm{mg / kg}$ doses (figure 3 and 4). These results corroborate those of Lamou et al, (2015); Pornariya and Kanok, (2009); Elmastas et al, (2007); Mishra et al, (2011) and, they would be justified by the antioxidant capacity of both Moringa oleifera and Pleurotus ostreatus (Zhang et al., 2016; Elmastas et al., 2007; Makkar et al., 1996; Sholapur and Patil, 2013).
The liver damage caused by paracetamol, known as a hepatotoxic agent in case of overdose
(Séide, 2008), is frequently used to assess the hepatoprotective effects of medicinal plants (Lewerenz et al., 2003; Liu et al., 2011). The markers that are used to determine toxicity are usually transaminases (ALAT and ASAT), whose high concentration in the extracellular medium is synonymous with an alteration of the cells. In this study, our results showed that ALAT and ASAT activities were decreased in the FMP16 groups (Table 2). Thus, ALAT activity decreased by $29\%$ in the 1000 mg/kg dose group compared to the intoxicated control. ASAT activity was higher in the intoxicated group (TP) by $28\%$ and $26\%$ compared to the 1000 and $1500~\mathrm{mg / kg}$ doses. Compared to the results of the studies by Adedapo et al, (2009) and Alam et al, (2011), who instead found that a dose of $1600~\mathrm{mg / kg}$ of Moringa oleifera leaves rather increased ALAT and ASAT activity; and on the other hand that a diet supplemented with $5\%$ Pleurotus rather decreased transaminase activities, we could think that this explains the fact that FMP16 rather tends to regulate their activities due to the antagonistic effect that these two species have.
Furthermore, results showed that the administration of FMP16 did not cause any significant difference in albumin and testosterone levels (Table 2). These results are similar to those of Alam et al, (2011) and Adedapo et al, (2009) who found that administration of Pleurotus and Moringa oleifera had no effect on albumin levels. However, Prabsattro et al, (2015), Zade et al, (2013), Okolo et al, 2016 rather found in their studies that Moringa oleifera increases sexual performance and thus could be considered as a potential aphrodisiac.
Regarding creatinine, FMP16 administration decreased creatinine levels in the treated groups (D1P, D2P, D3P) compared to the untreated and stressed group (TP) (Table 2). These results corroborate those of Sirag, (2009), Adedapo et al., (2009), Kane et al., 2022 who showed the protective effect of Pleurotus ostreatus and Moringa oleifera on kidney damage.
Our results on lipid metabolism in rats revealed a significant decrease in total cholesterol in the 500, 1000 and $1500\mathrm{mg / kg}$ dose groups (figure 5). There was a $28\%$ $39\%$ $30\%$ and $38\%$ difference in TP, D1P, D2P and D3P compared to TG. In addition, a difference of $15\%$ and $14\%$ of D1P and D3P compared to TP. However, there was no significant difference in HDL cholesterol levels (Table 2). The results of the Triglycerides levels (Table 2) show a difference of $47\%$ and $41\%$ of the 500 and $1000\mathrm{mg / kg}$ dose compared to the TG control group. There was also a $28\%$ decrease in Triglycerides levels at the $500\mathrm{mg / kg}$ dose compared to the dose $1000\mathrm{mg / kg}$. In most of the studies on the effects of Pleurotus ostreatus and Moringa oleifera, they found a decrease in the concentration of LDL cholesterol which is more related to cardiovascular diseases (Bobek and Galbavy, 1999; Bobek et al., 1998; Hossain et al., 2003). These results are also in agreement with those of Alam et al., (2009), Schneider et al., (2011), Chumark et al., (2008), Kane et al., (2022). Our results (figure 6) and those of previous studies suggest that FMP16 would be an excellent cholesterol-lowering agent that could be recommended for the prevention and treatment of cardiovascular diseases.
Table 1: Effect of the dietary supplement on rat weights
<table><tr><td>GROUPES</td><td>Starting Body weight (g)</td><td>Final Body weight (g)</td><td>P-value*</td></tr><tr><td>TG</td><td>154 ± 3,34</td><td>173,67 ± 9,16a</td><td>0, 02</td></tr><tr><td>TP</td><td>154,33±3,44</td><td>177,33±4,84d</td><td>0,01</td></tr><tr><td>D1P</td><td>154±2,53</td><td>185,67±2,86e</td><td>0,01</td></tr><tr><td>D2P</td><td>153,20±2,68</td><td>168±4,14</td><td>0,6</td></tr><tr><td>D3P</td><td>153±3,03</td><td>157±7,14</td><td>0, 13</td></tr></table>
Table 2: Effects of dietary supplement FMP16 on serum transaminases, albumin, testosterone, HDL-cholesterol and triglycerides activity
<table><tr><td>GROUPS</td><td>ALAT (U/l)</td><td>ASAT (U/l)</td><td>ALBUMINE (g/dl)</td><td>TESTOS. (ng/dl)</td><td>HDL-C (mg/dl)</td><td>Triglyc. (mg/dl)</td></tr><tr><td>TG</td><td>32,74±7,09</td><td>160,92±30,02</td><td>1,53±0,20</td><td>0,31±0,06</td><td>39,07±2,18</td><td>52,03±0,68</td></tr><tr><td>TP</td><td>48,48±3,32</td><td>196,35±33,40</td><td>1,61±0,37</td><td>0,47±0,12</td><td>38,88±1,88</td><td>78,14±12,69</td></tr><tr><td>D1P</td><td>43,16±8,80</td><td>209,16±25,74</td><td>1,80±0,10</td><td>0,53±0,08</td><td>36,30±2,89</td><td>71,02±18,92</td></tr><tr><td>D2P</td><td>34,31±4,55</td><td>141,86±11,19</td><td>1,60±0,20</td><td>0,44±0,04</td><td>35,08±2,53</td><td>98,18±7,64</td></tr><tr><td>D3P</td><td>43,66±2,13</td><td>145,86±22,20</td><td>1,75±0,18</td><td>0,43±0,03</td><td>34,17±5,47</td><td>87,83±21,97</td></tr></table>
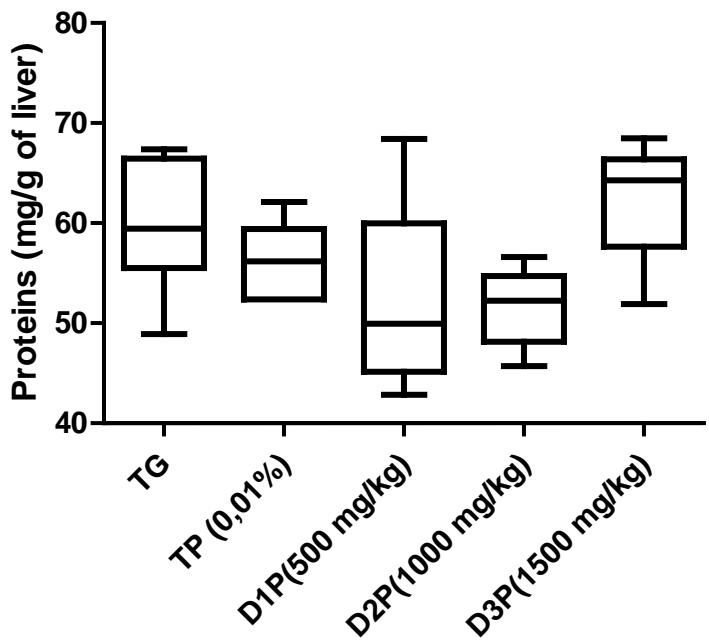 Figure 1: Total protein concentrations (mg/g of liver) in groups stressed by swimming and the Ethanol+ paracetamol combination
The values are expressed as mean ± SD. TG: Control group rats with food and water ad libitum, TP: stressed rats without treatment, D1P: dose of 500 mg/kg, D2P: dose of 1000 mg/kg, D3P: dose of 1500 mg/kg.
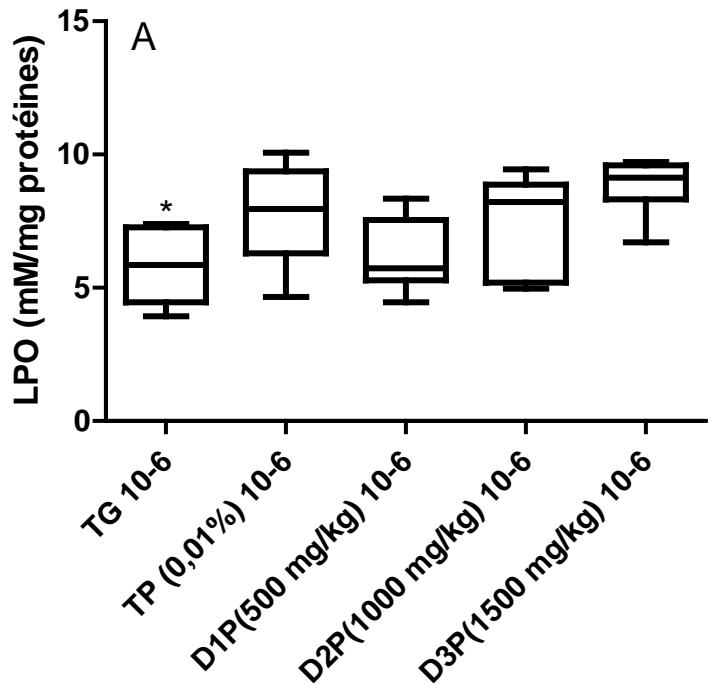 Figure 2: Effect of the dietary supplement on concentrations of peroxidized lipids (LPO)
The values are expressed as mean ± SD. TG: Control group rats with food and water ad libitum, TP: stressed rats without treatment, D1P: dose of 500 mg/kg, D2P: dose of 1000 mg/kg, D3P: dose of 1500 mg/kg. 10-6


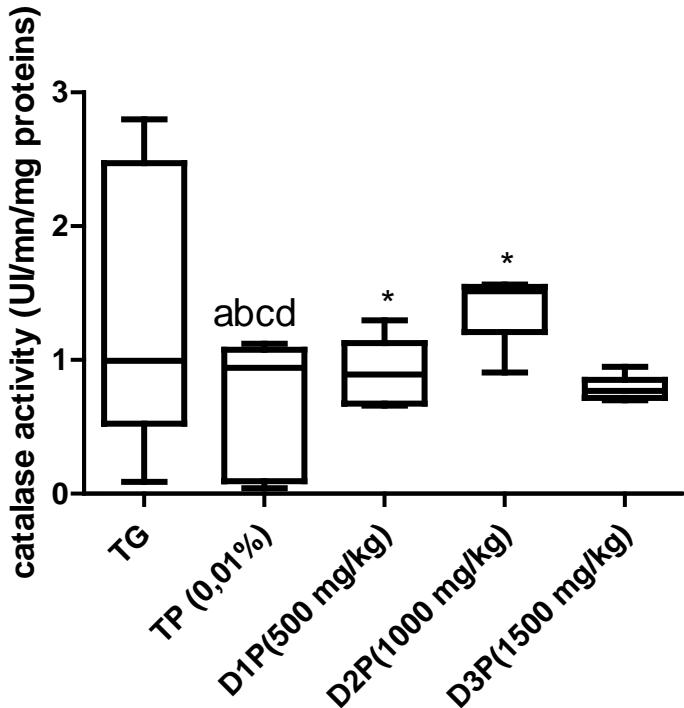 Figure 3: Effect of the dietary supplement on catalase activity (UI/mn/mg proteins)
- The values are expressed as mean ± SD. TG: Control group rats with food and water ad libitum, TP: stressed rats without treatment, D1P: dose of 500 mg/kg, D2P: dose of 1000 mg/kg, D3P: dose of 1500 mg/kg.
- a, b, c, d, mean statistically different with TG, D1P, D2P et D3P à $p < 0.05$ (Test de Bonferroni) \*, +, mean statistically different with D2P and D3P à $p < 0.05$ (Test de Bonferroni)
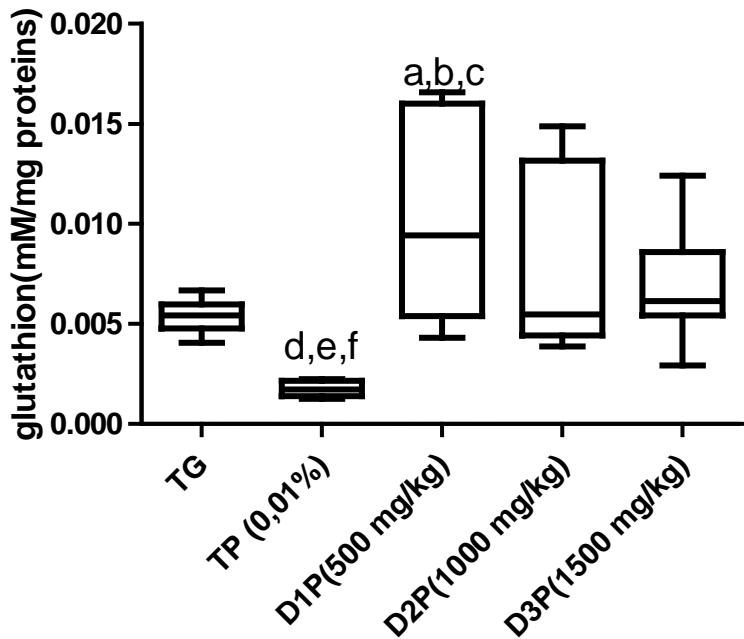 Figure 4: Effect of the dietary supplement on glutathion cellular activity (mM/mg proteins) Values are means $\pm$ SD.
- a, b, c, d mean statistically different with TG, D1P, D2P et D3P à $p < 0.05$ (Test de Bonferroni)
- e, f, g mean statistically different with TG, D2P et D3P à $p < 0.05$ (Test de Bonferroni)
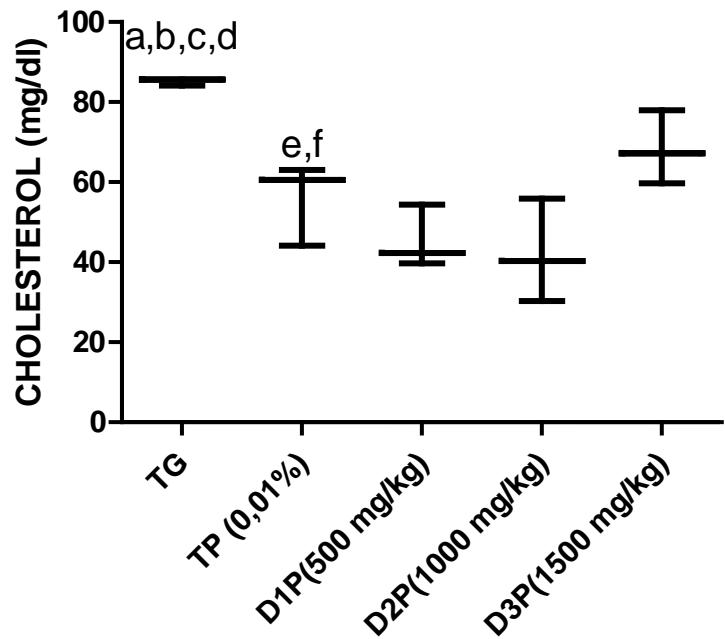 Figure 5: Total cholesterol level (mg/dl) in serum of rats stressed by the combination of ethanol (30% - 2g/kg) and paracetamol (750mg/kg)
The values are expressed as mean $\pm$ SD. TG: Control group rats with food and water ad libitum, TP: stressed rats without treatment, D1P: dose of 500 mg/kg, D2P: dose of 1000 mg/kg, D3P: dose of 1500 mg/kg. a, b,c, d statistically different mean compared to TP, D1P, D2P and D3P at $p < 0.05$ (LSD test) e, f mean statistically different from D1P and D3P at $p < 0.05$ (LSD test)
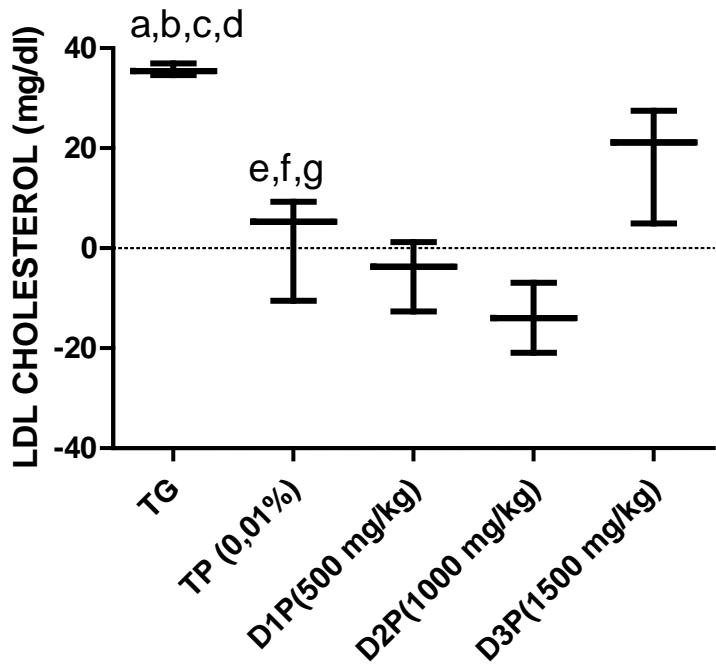 Figure 6: LDL cholesterol levels (mg/dl) in serum of rats stressed by the combination of ethanol (30% - 2g/kg) and paracetamol (750mg/kg)
The values are expressed as mean $\pm$ SD. TG: Control group rats with food and water ad libitum, TP: stressed rats without treatment, D1P: dose of 500 mg/kg, D2P: dose of 1000 mg/kg, D3P: dose of 1500 mg/kg. a,b,c,d statistically different mean compared to TP, D1P, D2P and D3P at $p < 0.05$ (LSD test) e, f, g mean statistically different from D1P, D2P and D3P at $p < 0.05$ (LSD test)
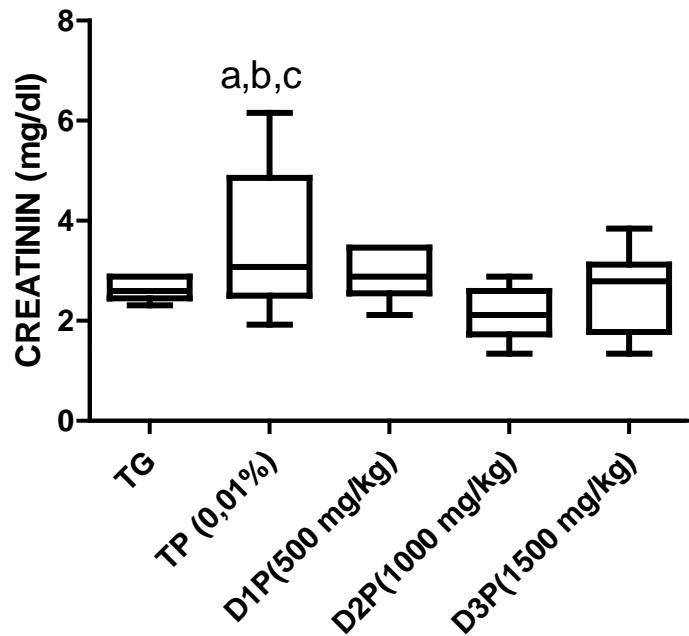 Figure 7: Effect of the dietary supplement on creatinin activity (mg/dl) in the serum of rats stressed by the combination of ethanol (30% - 2g/kg) and paracetamol (750mg/kg)
The values are expressed as mean ± SD. TG: Control group rats with food and water ad libitum, TP: stressed rats without treatment, D1P: dose of 500 mg/kg, D2P: dose of 1000 mg/kg, D3P: dose of 1500 mg/kg.a, b mean statistically different from D2P and D3P at p\<0.05 (Bonferroni test)
## IV. CONCLUSION
A dietary supplement of Moringa oleifera leaves and Pleurotus ostreatus in wistar rats shows that the powders of M. oleifera leaves and P. ostreatus mixture have an antihyperlipidemic effect as it significantly lowers total and LDL cholesterol levels in rats stressed by combination of ethanol and paracetamol. The dose $1000\mathrm{mg / kg}$ is most appropriate for chemically stressed animals. FMP16 would have no effect on albumin and testosterone levels.
### ACKNOWLEDGEMENTS
The authors would like to thank the Biotechnology Laboratory of the University Cheikh Anta Diop of Dakar, Senegal, and the Faculty of Science, the FODRUS-LAPHER-Biotech of the University of Yaoundé I, Cameroon. The project was part of a scholarship provided to Fatou Corka KANE through Africa for Innovations, Mobility, Exchange, Globalization and Quality (AFIMEGQ) program sponsored by the European Commission's EACEA program.
#### Conflict of Interests
The authors declare that there is no conflict of interests regarding the publication of this paper.
Generating HTML Viewer...
References
51 Cites in Article
Mary Njenga,James Gitau,Miyuki Iiyama,Ramni Jamnadassa,Yahia Mahmoud,Nancy Karanja (2004). Innovative Biomass Cooking Approaches for sub-Saharan Africa.
Abou-Elezz Fouad Mohammed,L Sarmiento-Franco,R Santos-Ricalde,J Solorio,Sanchez (2012). The nutritional effect of Moringa oleifera fresh leaves as feed supplement on Rhode Island Red hen egg production and quality.
Donald Abrams,Paul Couey,Starley Shade,Mary Kelly,Nnemdi Kamanu-Elias,Paul Stamets (2011). Antihyperlipidemic effects of Pleurotus ostreatus (oyster mushrooms) in HIV-infected individuals taking antiretroviral therapy.
A Adedapo,O Mogbojuri,Emikpe B O (2009). Safety evaluations of the aqueous extract of the leaves of Moringa oleifera in rats.
H Akhtar,K Ahmad (1995). Anti-ulcerogenic evaluation of the methanolic extracts of some indigenous medicinal plants of Pakistan in aspirin-ulcerated rats.
Nuhu Alam,Ruhul Amin,Asaduzzaman Khan,Ismot Ara,Mi Shim,Min Lee,Tae Lee (2008). Nutritional Analysis of Cultivated Mushrooms in Bangladesh -<i>Pleurotus ostreatus</i>,<i>Pleurotus sajor-caju</i>,<i>Pleurotus florida</i>and<i>Calocybe indica</i>.
H Azaizeh,S Fulder,K Khalil,O Said (2003). Ethnobotanical knowledge of local Arab practitioners in the Middle Eastern region.
M Badalyan (2014). Potential of mushroom bioactive molecules to develop healthcare biotech products.
Lillian Barros,Paula Baptista,Isabel Ferreira (2007). Effect of Lactarius piperatus fruiting body maturity stage on antioxidant activity measured by several biochemical assays.
B Moyo,P Masika,A Hugo,V Muchenje (2011). Nutritional characterization of Moringa (Moringa oleifera Lam.) leaves.
D Tallec (2008). The, café: aliments ou médicaments.
Mahfuz Elmastas,Omer Isildak,Ibrahim Turkekul,Nuri Temur (2007). Determination of antioxidant activity and antioxidant compounds in wild edible mushrooms.
(2015). The State of food insecurity in the world.
H Foidl,K Makkar,Becker (2001). The potential of Moringa oleifera for agricultural and industrial uses.
G Hao,C Zhang,W Cao,J Hao (2014). Effects of intragastric administration of five oyster components on endurance exercise performance in mice.
Luiz Gazzaneo,Reinaldo De Lucena,Ulysses De Albuquerque (2005). Knowledge and use of medicinal plants by local specialists in an region of Atlantic Forest in the state of Pernambuco (Northeastern Brazil).
Natalia Hanazaki,Jorge Tamashiro,Hermógenes Leitão-Filho,Alpina Begossi (2000). Diversity of plant uses in two Caiçara communities from the Atlantic Forest coast, Brazil.
S Handa,A Sharma,K Chakraborti (1986). Natural products and plants as liver protecting drugs.
H Makkar,K Becker (1996). Nutrional value and antinutritional components of whole and ethanol extracted Moringa oleifera leaves.
T Jayakumar,E Ramesh,P Geraldine (2006). Antioxidant activity of the oyster mushroom, Pleurotus ostreatus, on CCl4-induced liver injury in rats.
T Jayakumar,P Aloysiusthomas,P Geraldine (2007). Protective effect of an extract of the oyster mushroom, Pleurotus ostreatus, on antioxidants of major organs of aged rats.
J Fuglie Unknown Title.
M Olson (2001). Introduction to Moringa family.
J Kerrharo (1992). La pharmacopée Africaine, plantes médicinales et toxiques 1974.
Kane,L Souk Tounkara,D Kimassoum,M Guewo-Fokeng,A Diop,Wilfred Mbacham (2017). Nutritional value of a dietary supplement of Moringa oleifera and Pleurotus ostreatus.
F Kane,D Kimassoum,S Brice,M Paul,W Mbacham (2022). Antioxidant Property of a Dietary Supplement of Moringa oleifera Leaves and Pleurotus ostreatus in Wistar Rats Subjected to Forced Swimming Endurance Test.
A Khatun,A Hossain,M Islam,A Hossain,K Munshi (2012). Huque R Effect of gamma radiation on antioxidant marker and microbial safety of fresh bitter gourd (Momordica charantia L.).
Bonoy Lamou,Germain Taiwe,André Hamadou,Abene,Justin Houlray,Mahamat Atour,Paul Tan (2015). Antioxidant and Antifatigue Properties of the Aqueous Extract of <i>Moringa oleifera</i> in Rats Subjected to Forced Swimming Endurance Test.
Lev (2006). Ethno-diversity within current ethnopharmocology as part of Israeli traditional medicine-A review.
C Lieber,Y Wu,K Salmela (1994). Microsomal acetaldehyde oxidation is negligible in the presence of ethanol.
Lijun You,Mouming Zhao,Joe Regenstein,Jiaoyan Ren (2011). In vitro antioxidant activity and in vivo anti-fatigue effect of loach (Misgurnus anguillicaudatus) peptides prepared by papain digestion.
L Fuglie Introduction to the multiple uses of Moringa (7-10).
L Fuglie (2002). Nutrition naturelle sous les tropiques.
Lin-Zhang Huang,Bao-Kang Huang,Qi Ye,Lu-Ping Qin (2011). Bioactivity-guided fractionation for anti-fatigue property of Acanthopanax senticosus.
Moussa Ndong,Amadou T.G,Rokhaya Dg (2007). Valeur Nutritionnelle du Moringa Oleifera, Étude de la Biodisponibilité du fer, Effet de L'enrichissement de Divers Plats Traditionnels Sénégalais Avec la Poudre des Feuilles.
Nahid Richter,Perumal Siddhuraju,Klaus Becker (2003). Evaluation of nutritional quality of moringa (Moringa oleifera Lam.) leaves as an alternative protein source for Nile tilapia (Oreochromis niloticus L.).
Okwulehie, I. C,Urama Judith,Okorie, D. O. (2014). Chemical Composition and Nutritional Value of Mature and Young Fruiting-Bodies of Pleurotus Pulmonarius Produced On Andropogon Gayanus Straw and Khaya Ivorensis Sawdust.
H Osman,M Shayoub,E Babiker (2012). The effect of Moringa oleifera leaves on blood parameters and body weights of albino rats and rabbits.
L Pari,K Karthikesan (2007). Protective role of caffeic acid against alcohol-induced biochemical changes in rats.
S Pei (2001). Ethnobotnical approaches of traditional medicine studies: Some experiences from Asia.
B Ponnappa,Rubin (2000). Animal Models in Alcohol Research.
C Pornariya,O Kanok (2009). Amino acids and antioxidant properties of the oyster mushrooms, Pleurotus ostreatus and Pleurotus sajorcaju.
R Elias,S Kellerby,E Decker (2008). Antioxidant activity of proteins and peptides.
Silvia Rossato,Hermógenes De Leitão-Filho,Alpina Begossi (1999). Ethnobotany of caiçaras of the Atlantic Forest coast (Brazil).
Scott Powers,Karyn Hamilton (1999). ANTIOXIDANTS AND EXERCISE.
A Sivaraj,P Vinothkumar,S Palani,K Devi,E Elumalai,B Senthilkumar (2010). Preventive effect of aqueous leaf extract of Alternanthera sessilis L. on CCl4 induced hepatic damage in albino mice.
A Sivaraj,P Vinothkumar,K Sathiyaraj,K Devi,B Senthilkumar (2010). Hepatoprotective and antioxidant properties of Coccinia grandis aqueous leaf extract on ethanol induced liver toxicity in albino rats.
T Olugbemi,S Mutayoba,F Lekule (2010). Effect of Moringa (Moringa oleifera) Inclusion in Cassava Based Diets Fed to Broiler Chickens.
P Vinothkumar,A Sivaraj,K Devi,B Senthilkumar (2010). Hepatoprotective and antioxidant properties of aqueous rhizome extracts of Picrorhiza kurroa on CCl4 induced liver toxicity in albino rats.
(2010). Report on the global situation of non-communicable diseases.
Yan Zhang,Tao Hu,Hongli Zhou,Yang Zhang,Gang Jin,Yu Yang (2016). Antidiabetic effect of polysaccharides from Pleurotus ostreatus in streptozotocin-induced diabetic rats.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Fatou Corka Kane. 2026. \u201cAntihyperlipidemic Property of a Dietary Supplement of Moringa Oleifera Leaves and Pleurotus Ostreatus in Wistar Rats Stressed by Combination of Ethanol-Paracetamol.\u201d. Global Journal of Medical Research - L: Nutrition GJMR-L Volume 22 (GJMR Volume 22 Issue L2).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.
Antihyperlipidemic Property of a Dietary Supplement of Moringa Oleifera Leaves and Pleurotus Ostreatus in Wistar Rats Stressed by Combination of Ethanol-Paracetamol.