Antimeiotic Properties of the Aqueous Extracts of Leaves, Fruits and Roots of the Muskmelon Cucumis melo L. (Cucurbitaceae) in the Pest Grasshopper Zonocerus variegatus L. (Pyrgomorphidae)
The Muskmelon, Cucumis melo L., is a Cucurbitaceae widely cultivated in Cameroon for its nutritional and ethnomedicinal benefits. Species of Cucurbitaceae are known to contain several bioactive molecules that include the terpenoid cucurbitacins, which has been shown to cause significant molting defects and mortality in a variety of Coleoptera insect species such as Leperesinus fraxini PANZ(Coleoptera, Scolytidae) Stereonychus fraxini DE GEER (Coleoptera, Curculionidae). This study was designed to determine if an aqueous extract of Muskmelon, C. melo var. Cantaloupensis Americana, could profoundly affect the meiotic process in the Orthoptera grasshopper Zonocerus variegatus, a veritable food crop pest in Africa south of the Sahara. Different concentrations (0 µg/ml, 5µg/ml, 10 µg/ml, 20 µg/ml, 30 µg/ml, and 40 µg/ml) of aqueous extract of the leaves, fruits, and roots of C. melo were, respectively injected using the intraperitoneal method (into the hemocoel) of new reproductive and adult male individuals of Z. variegatus. Cytogenetic analysis revealed that Muskmelon extracts significantly reduced meiotic indexinduced meiotic chromosome abnormalities and significantly reduced chiasma frequency.
## I. INTRODUCTION
The Muskmelon, Cucumis melo L. (Cucurbitaceae), is variously known in Cameroon as the melon. In addition to Citrulus lanatus (Thunb.) (water melon), C. melo is one of the important cultivated cucurbits in Cameroon. C. melo L. is an annual creeping plant with long stems tendrils, large rounded heart-shaped green leaves, as well as large and round fruits that may be embroidered with white spots [1,2]. The muskmelon is cultivated especially in the North West, and West Regions where whole fruits and dried seeds are commonly sold on the Cameroonian markets [3]. The mesocarp of the fruitis eaten, the seeds used as a thickener in many Cameroonian soups. The species is an important and valuable vegetable crop in Cameroon and several tropical countries. It is widely consumed for its nutritional value and used for a wide variety of traditional medicinal properties [4-6]. The family Cucurbitaceae is an economically significant group of plants that contains bioactive phytochemicals such as Glycosides, Terpenoids, Saponins, Tannins, Steroids, and Carotenoids [7]. The terpenoids contain the bitter-tasting bioactive principle, cucurbitacins, compounds that have curative and several biological activities [3,8-15]. Cucurbitacins are essential for their therapeutic use in cancer treatment and other ethnomedicinal activities. They have also been linked with controlling several beetle pests of Cucurbitaceae plants. Cucurbitacins are very effective in natural plant defense against herbivores [16]. Cucurbitacin B, a variety of this principle, has been shown to significantly reduce the adult longevity and fecundity in the melon aphid, Aphis gossypii[17,15]. It has also been shown to have potent antifeeding properties for insects not adapted to exploiting cucurbits. Four beetles, Popillia japonica Newman, Ceratoma trifurcata (Forster), Leptinotarsadecemlineata (Say), and Trichoplusia ni (Hubner), were reported to stop feeding on application of cucurbitacin B to appropriate sources [18]. Cucurbitacin has been shown to affect oviposition in the moths Ostrinianubilalis (Hubner), and Spodopteraexigua (Hubner) females [18]. Available literature indicates that the bio-pesticidal activities of cucurbitacin have been extensively investigated for beetles of the Coleoptera order. On the other hand, information on the pesticidal effects of cucurbitacin on other pest species, especially the Orthoptera grasshoppers, a vital pest group in Cameroon and Africa, is not available in the literature. Zonocerus variegatus L., has been variously shown to
be a veritable pest of both food and cash crops in Cameroon and several Central, East, and West African countries. This grasshopper is a severe problem because of its wide host ranges, and its economic and ecological cost. Thus far, this grasshopper pest has been controlled solely with chemical pesticides such as Malathion. Therefore, the search for effective bioactive substances in the control of the pest continues. The present study was designed to determine the effects of the bioactive compounds in C. melo on the meiotic process in the pest grasshopper Z. variegatus L. It is expected that the extract will affect the meiotic process in the grasshopper which lead to the disruption of the reproduction process in this grasshopper pest species and hence valuable to controlling the pest population.
## II. MATERIALS AND METHODS
### a) Raw Materials and Extraction
The leaves, fruits, and roots of C. melo used for this study were collected from a farm in Baessing, a village in the Menoua Division of the West Region in Cameroon. The farm was located at latitude $5^{\circ}30'2''\mathrm{N}$ and longitude $10^{\circ}14'39''\mathrm{E}$. It was free of the use of fertilizers and pesticides. Leaves, ripe fruits and roots were collected only from mature plants. The species and variety which was easily recognizable were authenticated by Dr. NGANSOP Eric of the National Herbarium in Yaoundé with reference N080613/SRF/ Cam of 20/04/2022. The plant materials collected were taken to the laboratory, and washed of dust and ground before extraction. The different plant parts were individually chopped into small bits to accelerate drying and then dried in an oven at $60^{\circ}\mathrm{C}$ until there was no weight change. These specimens were next ground into powder. To prepare the aqueous extracts, $100\mathrm{g}$ of each sample was individually macerated in distilled water and stored at room temperature while constantly stirring at regular intervals with a spatula. After $36\mathrm{h}$, the mixtures were filtered using a sieve of $150~\mu \mathrm{m}$ in diameter and then with a coffee filter paper no.
4. The filtrates obtained were treated to $60^{\circ}\mathrm{C}$ in an oven to obtain whitish powders with total dry weights of $34.18\%$, $34.69\%$ and $33.56\%$ for leaves, ripe fruits, and roots, respectively. These were used to prepare stock solutions for leaves, ripe fruits and roots, respectively. From the stock solutions, micro dilutions of $5\mu \mathrm{g / ml}$, $10~\mu \mathrm{g / ml}$, $20~\mu \mathrm{g / ml}$, $30~\mu \mathrm{g / ml}$, and $40~\mu \mathrm{g / ml}$ were prepared by the addition of distilled water.
### b) Experimental Animals
Eighty (80) adult males of Zonocerus variegatus collected on campus in the University of Dschang (West Region of Cameroon) were brought to the Laboratory of the Research Unit of Biology and Applied Ecology (RUBEA) of the University of Dschang. Before the animal studies, the grasshoppers were reared in mineral water bottle cages and fed with fresh leaves (4 g per day per individual) of bitter leaves (Vernonia amygdalina), two days.
### c) Administration of Extract
The grasshoppers were divided into groups of five individuals and labeled I, II, III, IV, V & IV. The grasshoppers in groups I, II, III, IV, & V were respectively used for the evaluation of the aqueous extracts of leaves, fruit, and roots of C. melo, while those in group IV were used as the control. Each of these groups was further divided into five subgroups groups of A, B, C, D & E, each containing five (05) insects. Grasshoppers in groups A, B, C, D & E were respectively injected peritoneally (in hemocoel) with $0.1\mathrm{ml}$ of $5\mu \mathrm{g / ml}$, $10\mu \mathrm{g / ml}$, $20\mu \mathrm{g / ml}$, $30\mu \mathrm{g / ml}$, and $40\mu \mathrm{g / ml}$ of aqueous extract. The treated grasshoppers were allowed an incubation period of $96\mathrm{h}$ before being anesthetized and dissected for the testes. Grasshoppers in group F were the control.
### d) Preparation and Analysis of Chromosome Smears
The chromosome smears were prepared using the method of [19], and the smears were examined with the help of 10X, and the 40X objectives of the Fisher binocular compound light microscope. The meiotic smears obtained were examined for abnormalities that included laggards, bridges, sticky chromosomes, vagrant chromosomes, and breakages.
### e) Meiotic Index
The slides prepared for the extracts were observed under the microscope to record the Number of non-dividing and dividing cells. The Meiotic index was calculated using the formula\[20\]:
$$
\text{Meiotic Index} (\%) = \frac {\text {Number of dividing cells recorded}}{\text {Total number of cells examined}} \times 100
$$
### f) Photographs
Photographs of chromosome aberrations present were made using a Techno, Camon 16 phone mounted with a 48M AIQUAD Camera.
### g) Statistical Analysis of Data Collected
The Python 3.1 statistical software Pandas package was used for this analysis. The mean of the different types of chromosomal abnormalities as well as the other in-depth meiotic parameters, were subjected to the one-way ANOVA test followed by the Tukey posthoc test (HSD) at the level of significance of $p < 0.05$ [21].
## III. RESULTS AND DISCUSSION
### a) Cytogenetic Analysis
After staining testicular follicles of Z. variegatus treated with aqueous extracts of C. melo, the Orceinstained cells were analyzed with the 10X and 40X objectives of the compound light microscope. The results on the meiotic index, meiotic behavior of chromosomes, and chiasma frequency obtained are discussed in this section.
### b) Meiotic Index
Fig. 1 revealed that the meiotic index decreased with increase in the concentration of extract. The meiotic index for treatments with the leaves of C. melo was not much compared to that observed for fruits and roots. A significant decrease in the meiotic index was recorded for the highest concentration (40 $\mu$ g /ml) of extract of roots.
Table 1: Effects of aqueous extracts of leaves, fruits, and roots of C. melo on the meiotic index of the cells of Z. variegatus
<table><tr><td>Treatments</td><td>Meiotic Index (MI) 100%</td></tr><tr><td>Tape water (Control)</td><td>14.25±4.69a</td></tr><tr><td>Leaves</td><td>9.45±3.45c</td></tr><tr><td>Fruits</td><td>8.80±3.28b</td></tr><tr><td>Roots</td><td>8.00±2.2b</td></tr></table>
Values are means ± SEM. A number of trials n=5. Groups that have no letters in common differ significantly different from the control group (Distilled water), applying one-way ANOVA followed by Tukey's post-test (HSD)
The mean meiotic indices obtained for leaves, fruits, and roots of C. melo (Table 1) were subjected to
ANOVA. This analysis revealed that the mean meiotic indices obtained were significantly lower than the control. However, the meiotic indices induced by the extracts of fruits and roots were not different. The antimeiotic activity of the extracts can be linked to the distribution of cucurbitacins (the bioactive principle) in the leaves, fruits, and roots C. melo. Cucurbitacins have been reported to be more concentrated in the stem and roots of Cucurbitaceae plants than in other parts of the plant [2], and in the fruits [22]. These results indicated that the aqueous extracts from the leaves, fruits, and roots of C. meloat certain concentrations, have antimeiotic properties.
### c) Meiotic Behaviour of Chromosomes
With the objective to investigate the effect of the extracts on the meiotic behaviour of the chromosomes in Z. variegatus, the chromosome smears prepared were examined for abnormal behaviour. This was important because the behavior of the chromosomes would determine whether treatment with the extracts would produce normal or abnormal spermatozoa and hence affect the meiotic reproductive. An analysis of the smears revealed various degrees of abnormalities depending more or less on concentrations of the extract applied. Several types of chromosome abnormalities that included sticky Metaphase 1, bridges, and lagging chromosomes in Anaphase 1, and chromosome fragments were recorded (Fig. 2a - e).
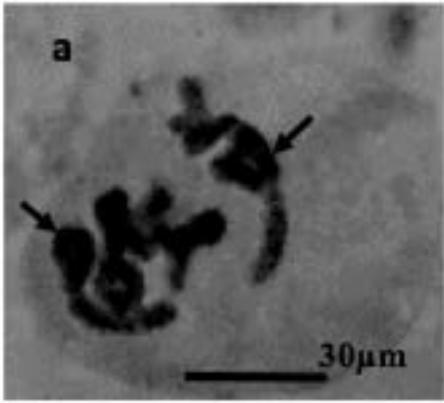
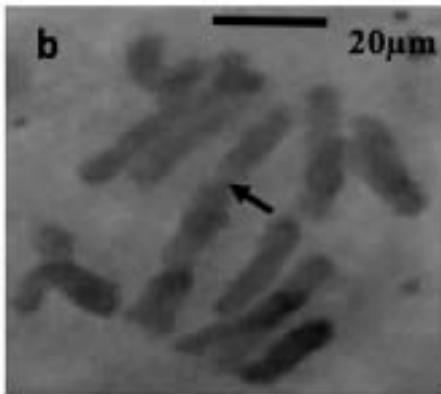
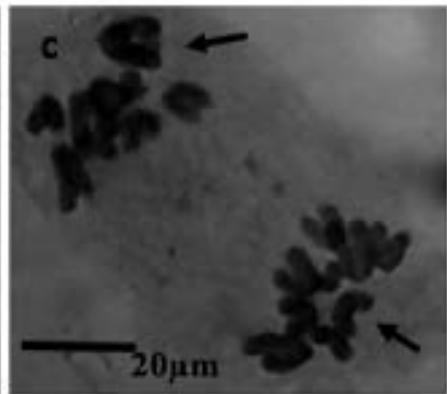
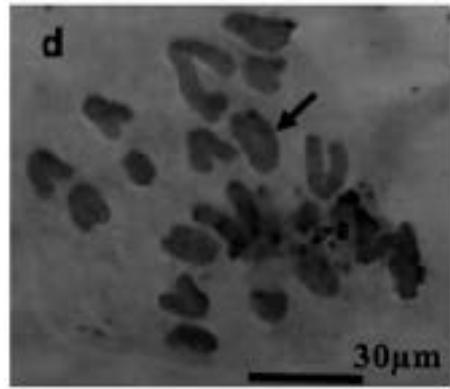
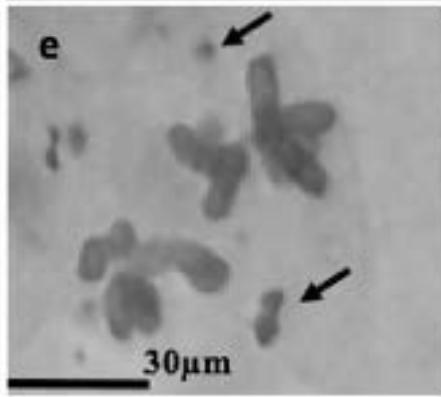
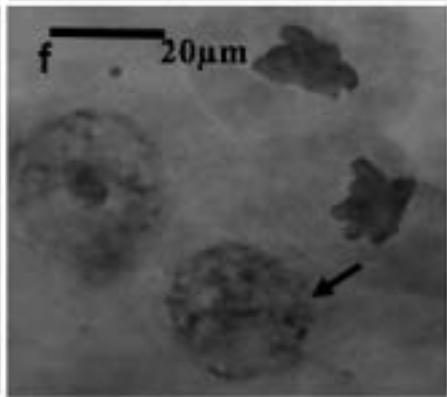 Figure 2: Various chromosome deformities in Z. variegatus on treatment with aqueous extracts of C. melo
(a= Sticky chromosomes; b= Anaphase I with bridge; c= laggard arrowed; d= laggard arrowed; e= Chromosomes break; f= Ghost cell)
It is essential to mention here that sticky chromosomes and bridges could result in chromosome breakages in the course of meiosis and hence produce acentric fragments. Acentric chromosome fragments lack centromeres that are essential for the division and the retention of the chromosome in the cell. Acentric chromosomes fragments are therefore lost when the cell divides. The loss leads to unbalanced gametes and could result in infertility. Lagging chromosomes contribute to the uneven distribution of chromosomes and, therefore, to the formation of cells with the abnormal numbers of chromosomes. These abnormalities, called aneuploidy, could be amongst the major causes of infertility in animals and plants [23-25] and could be used in the biocontrol of pest grasshoppers. Many translucent cells were observed in various frequencies during this study. The outline of the cells that occurred singly was visible, but the nucleus and cytoplasmic structures were not stainable (Fig. 2f). As per the definition of [26] such cells could be described as ghost, shadows or translucent cells. They have been variously recorded in human samples and associated with cancers. They are often swollen or enlarged cells that do not have nuclei [26]. Records of ghost cells in grasshopper species were not available in cytogenetic literature. Therefore, this report is a pioneer record for ghost cells in Orthoptera. There is no knowledge about their origin, nature, significance and relation to meiosis. During this study, the frequency of ghost cells was observed to be concentration dependent. Hence it is suggested that their presence is an indication of high-level cytotoxicity of the extract. In conclusion, meiotic abnormalities lead to morphological and genetic variations, which bring about not only evolution but also intraspecific reproductive barriers. Such reproductive barriers could be exploited for pest control.
### d) Chiasma Frequency
The importance of chiasmata in a population cannot be overemphasized. Individuals with high chiasma frequencies are considered robust, while those with low chiasma frequencies are unstable and can be easily affected by the environmental changes. Therefore, small changes in climate affect populations with low chiasma frequencies, and can lead to, drastic reduction in population size. Chiasma formation in control was normal. The individuals had mean chiasma frequencies of 11.90, which is normal and was not at variance with the observations of [27]. On the other hand, chiasma frequencies in individuals in the treatments experienced significant reductions (Fig. 3); Chiasma frequency was, therefore, inversely proportional to the concentration of extract. These observations are at variance with the report of [28], who recorded an increase in mean chiasma frequency in individuals of $Z$ variegatus treated with aqueous extracts of Annona muricata. The present data could not explain this difference.
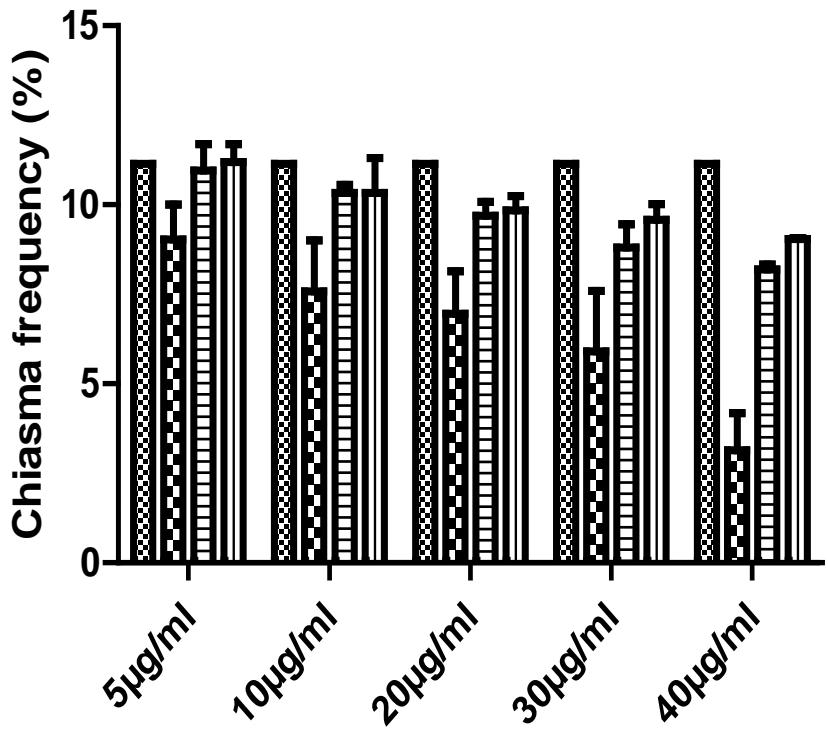
 Control
 Fruits
 Leaves
 Figure 3: Chiasma frequency in Z. Variegatus treated witch extracts of C. melo Roots
Table 2: Effect of aqueous extracts of leaves, fruits, and roots of C. melo on chiasma frequency of the cells of Z. variegatus
<table><tr><td>Treatments</td><td>Chiasma frequency (%)</td><td>Mean percent chiasma frequency per bivalent</td></tr><tr><td>Tape water (Control)</td><td>11.90±1.70a</td><td>1.32±0.168a</td></tr><tr><td>Leaves</td><td>10.01±0.3b</td><td>1.11±0.28a</td></tr><tr><td>Fruits</td><td>8.20±1.19d</td><td>0.91±0.88c</td></tr><tr><td>Roots</td><td>9.11±0.86c</td><td>1.01±0.73b</td></tr></table>
Analysis of the mean chiasma frequencies recorded (Table 2) revealed that the different extracts induced the formation of chiasmata differently, with the extract of fruits causing the lowest mean chiasma frequency. In all the treatments, induction was significantly lower than for the control. It is worth noting that chiasmata are essential for the attachment of homologous chromosomes in bivalents and hence subsequent segregation to the poles at Anaphase 1. Therefore, chiasmata are crucial for producing normal and genetically balanced spermatozoa and hence reproductive success in a population [29]. Available evidence shows that populations with decreased chiasma frequencies are less stable and are unable to adequately withstand sudden changes in the environment as compared to the populations with increased chiasma frequencies that can adapt to sudden changes in the environment [30,31]. The effect that decreased chiasma frequency in this study was difficult to assess from these results.
## IV. CONCLUSION
The aqueous extracts of the Muskmelon, C. melo can induce cytogenotoxic changes in the meiotic cells of the pest grasshopper Zonocerus variegatus, and hence affect fertility. At high concentrations of the aqueous extract, cytogenotoxic changes induced could be drastic and a reduction lead to reduction in future populations. Therefore, at sufficiently high concentrations, extracts of C. melo could be used in the formulation of biopesticides to control the pest grasshopper Z. variegatus. The following research will test these extracts on a few ovarian and testis cancers induced in Wistar rats.
Abbreviations list
Not used
Declarations
Ethical approval and consent to participate
All experimental studies on plants and grasshoppers have complied with relevant institutional, national and international guidelines and legislation.
Consent to publication
Availability of data and material
Competing interests
The authors have no competing interests to declare that are relevant to the content of this article.
Funding
No funding was obtained for this study.
## ACKNOWLEDGEMENTS
Our sincere thanks go to Professor WATCHO Pierre, the Head, Reproduction and Sexual Disorders Sub-Unit, Physiology, and Pr. MAYAKA Théodore Bileng, the Head, Applied Ecology Laboratory (LABEA), Faculty of Science, University of Dschang for providing laboratory facilities.
Author Contributions
- N.A. and S.R.A. conceived and conducted research experiments,
- N.A. and D.T.I. analysed data and conducted statistical analyses,
- N.A. and S.R.A wrote the manuscript,
- S.R.A. reviewed the manuscript. All authors read and approved the manuscript.
Generating HTML Viewer...
References
31 Cites in Article
I Tahir,M Oluoch (2004). Cucumis melo L. Plant resources of tropical Africa.
P Milind,S Kulwant (2011). International Research Journal Of Pharmacy.
Xiaojuan Wang,Mine Tanaka,Herbenya Peixoto,Michael Wink (2017). Cucurbitacins: elucidation of their interactions with the cytoskeleton.
K Vishal,K Jeetendra,K Prabhat (2017). Pharmacological importance of Cucumis melo L.: An overview.
Sema Kale,Bertrand Matthäus,Fahad Aljuhaimi,Isam Ahmed,Mehmet Özcan,Kashif Ghafoor,Elfadıl Babiker,Magdi Osman,Mustafa Gassem,Hesham Alqah (2020). A comparative study of the properties of 10 variety melon seeds and seed oils.
Z Xudong,B Yuzhuo,W Yun,W Chunlan,F Jianhua,G Longlan,L Yu,F Jingbin,K Mallappa,Y Maddipatla,R Gudepalya,P Boregowda,D Yue (2020). Anticancer Properties of Different Solvent Extracts of Cucumis melo L. Seeds and Whole Fruit and Their Metabolite Profiling Using HPLC and GC-MS.
R Rajasree,P Sibi,F Femi (2016). LAS VEGAS SANDS CORP., a Nevada corporation, Plaintiff, v. UKNOWN REGISTRANTS OF www.wn0000.com, www.wn1111.com, www.wn2222.com, www.wn3333.com, www.wn4444.com, www.wn5555.com, www.wn6666.com, www.wn7777.com, www.wn8888.com, www.wn9999.com, www.112211.com, www.4456888.com, www.4489888.com, www.001148.com, and www.2289888.com, Defendants..
Bolleddula Jayaprakasam,Navindra Seeram,Muraleedharan Nair (2003). Anticancer and antiinflammatory activities of cucurbitacins from Cucurbita andreana.
J Rio,M Recio (2005). Medicinal plants and antimicrobial activity.
S Shweta,S Saboo,K Priyanka,G Thorat,S Ganesh,L Tapadiya,F Khadabadi (2013). Ancient and Recent Medical Uses Of Cucurbitaceae Family.
Hajer Arjaibi,Mahmoud Ahmed,Fathi Halaweish (2017). Mechanistic investigation of hepato-protective potential for cucurbitacins.
Domenico Montesano,Gabriele Rocchetti,Predrag Putnik,Luigi Lucini (2018). Bioactive profile of pumpkin: an overview on terpenoids and their health-promoting properties.
Ahmad Faraz,Mohammad Faizan,Fareen Sami,Husna Siddiqui,John Pichtel,Shamsul Hayat (2019). Nanoparticles: biosynthesis, translocation and role in plant metabolism.
Agata Beata,O (2020). Vegetables from the Cucurbitaceae family and their products: Positive effect on human health.
Chenchen Zhao,Chao Ma,Junyu Luo,Lin Niu,Hongxia Hua,Shuai Zhang,Jinjie Cui (2021). Potential of Cucurbitacin B and Epigallocatechin Gallate as Biopesticides against Aphis gossypii.
Guillaume Chomicki,Hanno Schaefer,Susanne Renner (2019). Origin and domestication of Cucurbitaceae crops: insights from phylogenies, genomics and archaeology.
Hafiz Yousaf,Tisheng Shan,Xuewei Chen,Kangsheng Ma,Xueyan Shi,Nicolas Desneux,Antonio Biondi,Xiwu Gao (2018). Impact of the secondary plant metabolite Cucurbitacin B on the demographical traits of the melon aphid, Aphis gossypii.
Douglas Tallamy,Jason Stull,Nathan Ehresman,Piotr Gorski,Charles Mason (1997). Cucurbitacins as Feeding and Oviposition Deterrents to Insects.
R Seino,Y Manjeli,D Focho,D Shambo (2007). The B chromosome in the meiotic process of the African pest grasshopper <i>Taphronota thaelephora</i> STAL. (Orthoptera: Pyrgomorphidae)..
Kelli Zortéa,Ana Rossi,Angelo Cordeiro,Nilo Sander,Elisa Cardoso,Carolina Silva (2022). Pollen morphology, meiotic index and pollen viability in individuals of Vochysia divergens Pohl. native to the Amazon and the Pantanal.
B Scherrer (2020). Unknown Title.
A Usmangani,N Dally,Savaliram (2022). Simultaneous detection of anticancer compounds (Cucurbitacin I, B and E) and some pharmacological properties of Indian Blastania species.
Marie Janicke,Loren Lasko,Rudolf Oldenbourg,James Lafountain (2007). Chromosome Malorientations after Meiosis II Arrest Cause Nondisjunction.
C Joydeb,B Sayan,K Pulok (2020). Determination of cucurbitacin E in some selected herbs of ayurvedic importance through RP-HPLC.
K Rupinderpal,D Yumnam,K Ravindra,G Saurabh,S Mohd,V Wani (2022). Genotoxic effect of fruit extract of wild and cultivated cucurbits using Allium cepa assay.
E Rajesh,Sudha Jimson,K Masthan,N Balachander (2015). Ghost cell lesions.
R Seino,D Alain,D Tonleu,M Yacouba (2013). Karyotype and meiosis analysis of four species of Cameroonian Pyrgomorphidae (Orthoptera).
R Seino,Y Manjeli,D Focho,D Shambo (2014). The B chromosome in the meiotic process of the African pest grasshopper <i>Taphronota thaelephora</i> STAL. (Orthoptera: Pyrgomorphidae)..
Pablo Parra-Nunez,Mónica Pradillo,Juan Santos (2019). Competition for Chiasma Formation Between Identical and Homologous (But Not Identical) Chromosomes in Synthetic Autotetraploids of Arabidopsis thaliana.
E Sanchez-Moran,S Armstrong,J Santos,F Franklin,G Jones (2002). Variation in Chiasma Frequency Among Eight Accessions of <i>Arabidopsis thaliana</i>.
Yukinobu Hirose,Ren Suzuki,Tatsunori Ohba,Yumi Hinohara,Hirotada Matsuhara,Masashi Yoshida,Yuta Itabashi,Hiroshi Murakami,Ayumu Yamamoto (2011). Chiasmata Promote Monopolar Attachment of Sister Chromatids and Their Co-Segregation toward the Proper Pole during Meiosis I.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Ngnaniyyi Abdoul. 2026. \u201cAntimeiotic Properties of the Aqueous Extracts of Leaves, Fruits and Roots of the Muskmelon Cucumis melo L. (Cucurbitaceae) in the Pest Grasshopper Zonocerus variegatus L. (Pyrgomorphidae)\u201d. Global Journal of Science Frontier Research - G: Bio-Tech & Genetics GJSFR-G Volume 22 (GJSFR Volume 22 Issue G2): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
The Muskmelon, Cucumis melo L., is a Cucurbitaceae widely cultivated in Cameroon for its nutritional and ethnomedicinal benefits. Species of Cucurbitaceae are known to contain several bioactive molecules that include the terpenoid cucurbitacins, which has been shown to cause significant molting defects and mortality in a variety of Coleoptera insect species such as Leperesinus fraxini PANZ(Coleoptera, Scolytidae) Stereonychus fraxini DE GEER (Coleoptera, Curculionidae). This study was designed to determine if an aqueous extract of Muskmelon, C. melo var. Cantaloupensis Americana, could profoundly affect the meiotic process in the Orthoptera grasshopper Zonocerus variegatus, a veritable food crop pest in Africa south of the Sahara. Different concentrations (0 µg/ml, 5µg/ml, 10 µg/ml, 20 µg/ml, 30 µg/ml, and 40 µg/ml) of aqueous extract of the leaves, fruits, and roots of C. melo were, respectively injected using the intraperitoneal method (into the hemocoel) of new reproductive and adult male individuals of Z. variegatus. Cytogenetic analysis revealed that Muskmelon extracts significantly reduced meiotic indexinduced meiotic chromosome abnormalities and significantly reduced chiasma frequency.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.
Antimeiotic Properties of the Aqueous Extracts of Leaves, Fruits and Roots of the Muskmelon Cucumis melo L. (Cucurbitaceae) in the Pest Grasshopper Zonocerus variegatus L. (Pyrgomorphidae)