## I. INTRODUCTION
Radiotherapy is one of the pillars of the treatment of lung cancer and it can be used as an ablative therapy alone in the early stages of the disease or combined with chemotherapy in more advanced stages (1). Despite its curative role in many settings, radiotherapy is not without side effects. One of the most unwanted side effects is radiation pneumonitis (RP). RP is an inflammatory response resulting from damage to the irradiated lung parenchyma (2) that typically occurs within six months of treatment completion (3).
Several factors appear to be associated with the risk of developing RP and its severity, including patient-related, tumor-related, and treatment-related dosimetric factors, as well as tumor size and location (4). Patients receiving chemoradiotherapy or with prior lung resection are also in the group of patients at high risk of developing RP (5-6). However, the analysis of all these variables in the calculating toxicity potentials is uncommonly performed due to the lack of suitable algorithms.
There is essential heterogeneity among patients receiving radiotherapy. Currently, the rate of lung cancer in non-smokers has been increasing synchronously with the increase in the diagnosis of adenocarcinoma and inversely with the diagnosis of squamous cell carcinoma and small cell lung cancer (7). In any case, the number of smokers and patients with previous lung diseases who develop lung cancer is high (8). Such diseases include chronic obstructive bronchopulmonary disease
(COBPD) and interstitial lung disease (ILD) and are significant risk factors for cancer. A study published in 2015 demonstrated that patients with COBPD had a higher incidence of pneumonitis associated with consolidation in the irradiated volume (52%) than those without COBPD (16%) (9). The benefits of radiotherapy are well known, but it is essential to pay attention to the particularities of patients to adjust their treatment to minimize pulmonary complications and improve clinical outcomes (10).
Lung densitometry is a method that can differentiate healthy tissue from emphysematous or fibrotic tissue (11, 12). This diagnostic method measures lung density and classifies the tissue according to its ability to attenuate X-rays in computed tomography (CT) studies. CT lung density measurements are expressed in Hounsfield units (HU) (13), and the different densities obtained characterize the tissue, reflecting the degree of lung damage. For example, decreased X-ray attenuation occurs in emphysema and cystic lung disease, whereas increased X-ray attenuation occurs in pulmonary fibrosis.
In patients with lung cancer who are candidates for radiotherapy treatment, densitometry provides additional information about the patient's clinical condition (14,15), in addition to the possibility of visually mapping the whole lung tissue and its different densities (figure 1).
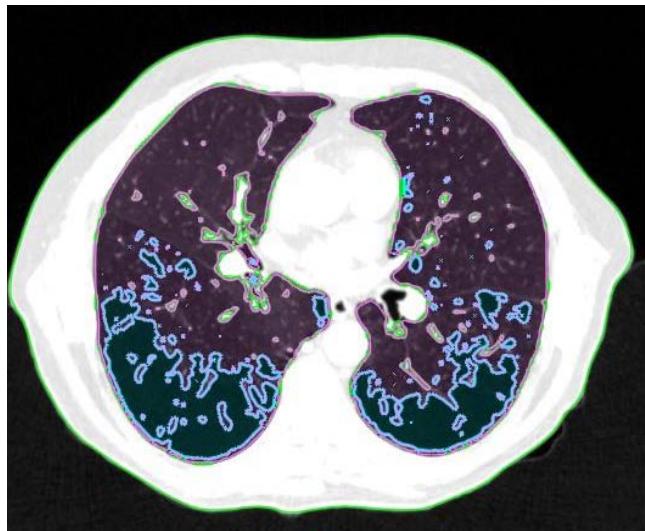
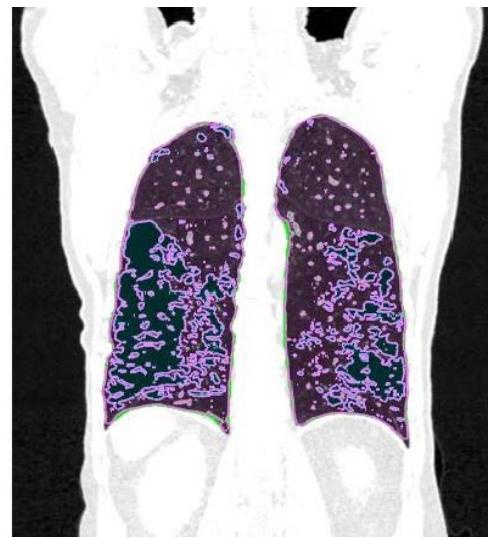 Figure 1: Visual representation of emphysema (blue), normal tissue (pink), and fibrosis (green)
## II. MATERIALS AND METHODS
Patients receiving lung stereotactic body radiotherapy (SBRT) for early-stage primary lung tumors (stages I and II) between 2017 and 2022 at Santa Casa de Porto Alegre, Brazil, were selected. Patients with CT scans with a millimetric slice thickness obtained at 120 kV and 80 mAS during forced inspiration were included.
All CT scans were examined for their lung density characteristics. Lung density measurements were made on radiotherapy planning CT scans, obtained with a 64-slice CT scanner (Ingenuity Core 64; Philips Healthcare, Cleveland, OH, USA). The structures of interest were outlined using the Eclipse radiotherapy planning system v15.6 (Varian Medical Systems, Palo Alto, CA, USA). The structures of interest were defined as "Right Lung," "Left Lung," and "Lungs" (both lungs drawn as a single structure) and automatically segmented, as were the structures corresponding to the different density ranges to be analyzed.
CT lung density measurements are expressed in HU, a quantitative scale for describing radiodensity divided into 2048 density values, where water is arbitrarily defined to be 0 HU, air is defined as -1000 HU, and bone density as 1000 HU (16). Quantitative indices of emphysema show low attenuation values, corresponding to the proportion of lung volume with attenuation between -1000 and -950 HU (17). Functional lung volume can be measured with attenuation between -950 and -700 HU, and for fibrosis, attenuation values range from -700 to -200 HU (18).
The structures corresponding to the attenuation ranges were obtained using the "Image Thresholding" tool (figure 2), which allows manual selection of HU values. They were defined as "Emphysema" for attenuation ranging from $-1000$ to $-950$ HU, "Normal Tissue" (functional lung volume) for attenuation ranging from $-949$ to $-700$ HU, and "Fibrosis" for attenuation ranging from $-699$ to $-200$ HU. The attenuation values of $-949$ HU and $-699$ HU were used to avoid data duplication. The volumes were then measured and recorded in a table.
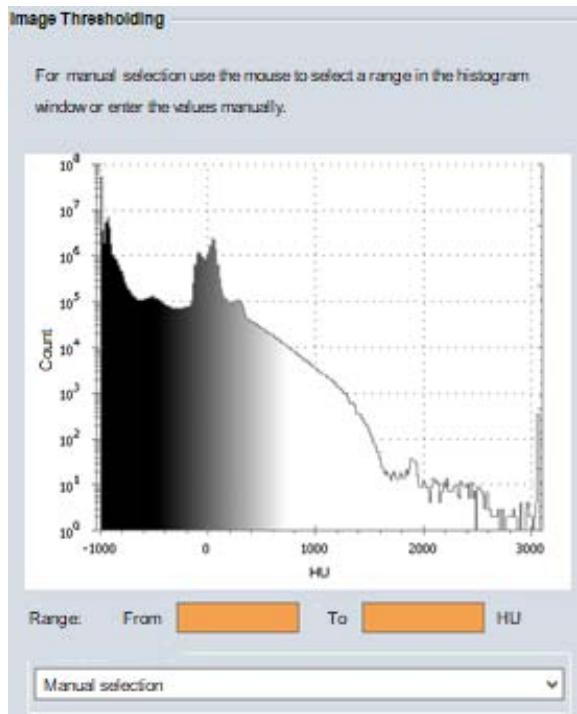 Figure 2: Image Thresholding Tool
Given its importance in the clinical and oncological treatment of patients with lung cancer, emphysema was divided into three groups. Emphysema was considered insignificant when it involved less than $5\%$ of the total lung volume, and severe emphysema was defined as involvement more significant than $15\%$ (19). Fibrosis was considered a single group.
Continuous variables are expressed as mean (SD) if normally distributed. Categorical variables are expressed as counts and percentages. Continuous variables were compared using t-tests or the Wilcoxon rank sum test. Data were analyzed in SPSS, version 29.0 (SPSS Inc., Chicago, IL, USA).
## III. RESULTS
CT data from 39 patients were analyzed. The mean patient age was 71.5 years, and most were female, accounting for $61.5\%$ of the sample. The most common histological type in biopsied patients was adenocarcinoma $(62.5\%)$, and the tumor site showed no predilection for any particular lobe.
Table 1: Baseline characteristics of the sample
<table><tr><td>Variable</td><td>n=39</td></tr><tr><td>Age (years) – mean ± SD [min – max]</td><td>71.7 ± 7.9 [55 – 87]</td></tr><tr><td>Sex – n(%)</td><td></td></tr><tr><td>Male</td><td>15 (38.5)</td></tr><tr><td>Female</td><td>24 (61.5)</td></tr><tr><td>Staging – n(%)</td><td></td></tr><tr><td>Primary tumor with pathological diagnosis</td><td>31 (79.5)</td></tr><tr><td>No pathological diagnosis, or inconclusive histological type – n(%)</td><td>8 (20.5)</td></tr><tr><td>Adenocarcinoma</td><td>20 (51.3)</td></tr><tr><td>Squamous cell carcinoma</td><td>12 (30.8)</td></tr><tr><td>No biopsy</td><td>7 (17.9)</td></tr><tr><td>BED (Gy) – mean ± SD [min – max]</td><td>152.2 ± 37.2 [85 – 180]</td></tr><tr><td>Tumor site – n(%)</td><td></td></tr><tr><td>RLL</td><td>9 (23.1)</td></tr><tr><td>LLL</td><td>9 (23.1)</td></tr><tr><td>RML</td><td>1 (2.6)</td></tr><tr><td>RUL</td><td>10 (25.6)</td></tr><tr><td>LUL</td><td>10 (25.6)</td></tr></table>
Of the total sample, 26 patients $(66.7\%)$ had no or insignificant emphysema (less than $5\%$ of the total lung volume), 7 $(17.9\%)$ had $5\%$ to $15\%$ of whole lung tissue with emphysema, being defined as mild-to-moderate emphysema, and 6 $(15.4\%)$ had severe emphysema, with greater than $15\%$ of the total lung volume being emphysematous. Tissue with attenuation areas corresponding to fibrosis was found in $7.5\%$ (SE, 1.21).
Table 2: Percentages of emphysema, normal tissue, and fibrosis on CT scans obtained at sustained maximal inspiration
<table><tr><td>Percentages</td><td>Inspiration
Mean ± SE</td></tr><tr><td>Emphysema</td><td>5.51 ± 1.30</td></tr><tr><td><5%</td><td>26 (66.7%)</td></tr><tr><td>5% to 15%</td><td>7 (17.9%)</td></tr><tr><td>>15%</td><td>6 (15.4%)</td></tr><tr><td>Normal</td><td>82.1 ± 1.56</td></tr><tr><td>Fibrosis</td><td>7.5 ± 1.21</td></tr></table>
## IV. DISCUSSION
There is consensus on the indication of radiotherapy in non-operated patients, both in the early and advanced stages of disease. A 5-year tumor control rate of $90\%$ can be achieved with ablative radiotherapy (20-22) or hypofractionated radiotherapy for early-stage tumors (23), generally with low toxicity, but not without toxicity, with reports of grade 5 toxicity (24), especially in more centrally located tumors. In the treatment of more advanced tumors, the 5-year local control rate is less than $30\%$ (25), with high rates of severe pneumonitis (grades 3 to 4) affecting one-third of patients when radiotherapy is combined with chemotherapy (26, 27), the standard of care for locally advanced tumors (28).
When evaluating radiotherapy planning, dose limits for irradiation of normal lung tissue are tabulated generically. Studies on lung toxicity in three-dimensional treatment suggest that the mean dose to both lungs, excluding tumor volume, should remain below 20 Gy. In comparison, it is prudent to limit the lung volume receiving 20 Gy to $30\%$, perhaps reaching $35\%$ $(V_{20Gy} \leq 30 - 35\%)$ (29), regardless of tumor stage. In ablative radiotherapy, the allowed doses have a different absolute number, but they also treat all patients with dose escalations, regardless of patients' pre-existing lung function and tumor stage. Such doses are $V_{20Gy} < 10\%$, not exceeding the limit of $15\%$. In addition, the doses must not exceed 12.5 Gy for the critical volume of 1500 cc and 13.5 Gy for 1000 cc (30-31). The use of $V_{X}$ -based dose analysis ("V" volume of normal lung receiving a dose of "X" Gy) is attractive because this metric can be readily obtained via the dose-volume histogram (DVH). However, as pointed out in the QUANTEC report on pulmonary toxicity, $V_{X}$ cutoffs may not be universal. The percent volume receiving a dose of "X" may depend on the treatment technique and does not allow the inclusion of other toxicity factors associated with the actual final toxicity (32).
In general, in the dose limits suggested in the literature, patients with severe lung disease have their normal tissue considered within the same dose limits as those for patients without any functional changes. The tissue volume considered normal is the tissue without tumor (32, 33), that is, excluding the target volume. Therefore, emphysematous or fibrotic areas, known to be entirely non-functional, are regarded as "normal" tissue in the volume.
For lung density analysis, we only included patients with a well-documented inspiration breath-hold technique. For this reason, we chose to analyze patients who would receive SBRT, as this group has a more detailed CT analysis than patients who receive conventional fractionation. It is known, however, that most patients receiving SBRT have clinical conditions unfavorable to surgery, which is still the standard treatment for early-stage tumors (34, 35), whether due to chronic lung disease or other comorbidities. Therefore, our sample has more patients with advanced lung disease than the group of all patients with lung cancer.
Despite the small sample size, the data corresponding to mild-to-moderate emphysema (17.9%) in patients with lung tumors are consistent with data from the literature (36). In the current study, severe emphysema was present in 15.4% of patients, which implies that these patients have limitations, that is, symptoms of their underlying lung disease. When receiving ablative or conventionally fractionated radiotherapy, these patients are more likely to develop clinical and radiological pneumonitis.
## V. CONCLUSION
Several studies have demonstrated that accurate quantification of lung density can be helpful in various clinical applications, such as the diagnosis and monitoring of lung diseases, medical procedure planning, and assessment of treatment response. In this study, it was possible to expand the use of tools currently available in the SBRT protocol, such as CT scan at inspiration and resources available in the planning system. The results based on lung densitometry provide essential information about the clinical characteristics of patients who are candidates for radiotherapy treatment, which can be helpful for future research and understanding specific lung conditions, allowing a personalized DVH assessment.
In conclusion, radiation dose-volume effects on the lung play an essential role in the developing pulmonary complications after radiotherapy. A personalized approach, considering risk factors and using advanced techniques, can help minimize these effects and, consequently, improve the quality of life of patients undergoing treatment.
Generating HTML Viewer...
References
33 Cites in Article
Shalini Vinod,Eric Hau (2020). Radiotherapy treatment for lung cancer: Current status and future directions.
Jeffrey Ryckman (2020). Correlation of dosimetric factors with the development of symptomatic radiation pneumonitis in stereotactic body radiotherapy.
Marek Konkol,Paweł Śniatała,Piotr Milecki (2022). Radiation-induced lung injury — what do we know in the era of modern radiotherapy?.
Yongmei Liu,Weili Wang,Kevin Shiue,Huan Yao,Alberto Cerra-Franco,Ronald Shapiro,Ke Huang,Douglas Vile,Mark Langer,Gordon Watson,Greg Bartlett,Huisi Ai,Francis Sheski,Jian-Yue Jin,Rich Zellars,Pingfu Fu,Tim Lautenschlaeger,Feng-Ming (spring) Kong (2021). Risk factors for symptomatic radiation pneumonitis after stereotactic body radiation therapy (SBRT) in patients with non-small cell lung cancer.
Vivek Verma,Charles Simone,Maria Werner-Wasik (2017). Acute and late toxicities of concurrent chemoradiotherapy for locally-advanced non-small cell lung cancer.
Eva Bongers,Angela Botticella,David Palma,Cornelis Haasbeek,Andrew Warner,Wilko Verbakel,Ben Slotman,Umberto Ricardi,Suresh Senan (2013). Predictive parameters of symptomatic radiation pneumonitis following stereotactic or hypofractionated radiotherapy delivered using volumetric modulated arcs.
Sarah Dubin,Daniel Griffin (2020). Lung cancer in non-smokers.
A Houghton,Mcgarry (2013). Mechanistic links between COPD and lung cancer.
Toshihiko Inoue,Hiroya Shiomi,Ryoong-Jin Oh (2015). Stereotactic body radiotherapy for Stage I lung cancer with chronic obstructive pulmonary disease: special reference to survival and radiation-induced pneumonitis.
Gerard Walls,Michael Mcmahon,Natasha Moore,Patrick Nicol,Gemma Bradley,Glenn Whitten,Linda Young,Jolyne O'hare,John Lindsay,Ryan Connolly,Dermot Linden,Peter Ball,Gerard Hanna,Jonathan Mcaleese (2023). Clinicoradiological outcomes after radical radiotherapy for lung cancer in patients with interstitial lung disease.
Jens Bakker (2021). Measuring pulmonary function in COPD using quantitative chest computed tomography analysis.
Harvey Coxson (2013). The presence and progression of emphysema in COPD as determined by CT scanning and biomarker expression: a prospective analysis from the ECLIPSE study.
Tami Denotter,Johanna Schubert,Hounsfield Unit (2019). FAIR Publishing Guidelines for Spectral Data and Chemical Structures.
Mario Mascalchi,Gianna Camiciottoli,Stefano Diciotti (2017). Lung densitometry: why, how and when.
Tomoki Kimura,Ikuno Nishibuchi,Yuji Murakami,Masahiro Kenjo,Yuko Kaneyasu,Yasushi Nagata (2012). Functional Image-Guided Radiotherapy Planning in Respiratory-Gated Intensity-Modulated Radiotherapy for Lung Cancer Patients With Chronic Obstructive Pulmonary Disease.
Bruno Hochhegger,Edson Marchiori,Klaus Irion,Hugo Oliveira (2010). Acurácia da mensuração do enfisema pulmonar na tomografia computadorizada: pontos importantes.
Fernanda Balbinot,Álvaro Da Costa Batista Guedes,Douglas Nascimento,Juliana Zampieri,Giordano Alves,Edson Marchiori,Adalberto Rubin,Bruno Hochhegger (2016). Advances in Imaging and Automated Quantification of Pulmonary Diseases in Non-neoplastic Diseases.
Ting Cheng,Yong Li,Shuai Pang,Huanying Wan,Guochao Shi,Qijian Cheng,Qingyun Li,Zilai Pan,Shaoguang Huang (2019). <p>Normal lung attenuation distribution and lung volume on computed tomography in a Chinese population</p>.
Craig Hersh,George Washko,Raúl Estépar,Sharon Lutz,Paul Friedman,Meilan Han,John Hokanson,Philip Judy,David Lynch,Barry Make,Nathaniel Marchetti,John Newell,Frank Sciurba,James Crapo,Edwin Silverman (2013). Paired inspiratory-expiratory chest CT scans to assess for small airways disease in COPD.
Robert Timmerman,Chen Hu,Jeff Michalski,Jeffrey Bradley,James Galvin,David Johnstone,Hak Choy (2018). Long-term Results of Stereotactic Body Radiation Therapy in Medically Inoperable Stage I Non–Small Cell Lung Cancer.
Robert Timmerman,Rebecca Paulus,Harvey Pass,Elizabeth Gore,Martin Edelman,James Galvin,William Straube,Lucien Nedzi,Ronald Mcgarry,Cliff Robinson,Peter Schiff,Garrick Chang,Billy Loo,Jeffrey Bradley,Hak Choy (2018). Stereotactic Body Radiation Therapy for Operable Early-Stage Lung Cancer.
Gregory Videtic,Chen Hu,Anurag Singh,Joe Chang,William Parker,Kenneth Olivier,Steven Schild,Ritsuko Komaki,James Urbanic,Hak Choy (2019). A Randomized Phase 2 Study Comparing 2 Stereotactic Body Radiation Therapy Schedules for Medically Inoperable Patients With Stage I Peripheral Non-Small Cell Lung Cancer: NRG Oncology RTOG 0915 (NCCTG N0927).
Jan Nyman,Andreas Hallqvist,Jo-Åsmund Lund,Odd-Terje Brustugun,Bengt Bergman,Per Bergström,Signe Friesland,Rolf Lewensohn,Erik Holmberg,Ingmar Lax (2016). SPACE – A randomized study of SBRT vs conventional fractionated radiotherapy in medically inoperable stage I NSCLC.
Andrea Bezjak,Rebecca Paulus,Laurie Gaspar,Robert Timmerman,William Straube,William Ryan,Yolanda Garces,Anthony Pu,Anurag Singh,Gregory Videtic,Ronald Mcgarry,Puneeth Iyengar,Jason Pantarotto,James Urbanic,Alexander Sun,Megan Daly,Inga Grills,Paul Sperduto,Daniel Normolle,Jeffrey Bradley,Hak Choy (2019). Safety and Efficacy of a Five-Fraction Stereotactic Body Radiotherapy Schedule for Centrally Located Non–Small-Cell Lung Cancer: NRG Oncology/RTOG 0813 Trial.
David Spigel (2022). Five-year survival outcomes from the PACIFIC trial: durvalumab after chemoradiotherapy in stage III non-small-cell lung cancer.
David Palma,Suresh Senan,Kayoko Tsujino,Robert Barriger,Ramesh Rengan,Marta Moreno,Jeffrey Bradley,Tae Kim,Sara Ramella,Lawrence Marks,Luigi De Petris,Larry Stitt,George Rodrigues (2013). Predicting Radiation Pneumonitis After Chemoradiation Therapy for Lung Cancer: An International Individual Patient Data Meta-analysis.
Saori Tatsuno,Hiroshi Doi,Wataru Okada,Eri Inoue,Kiyoshi Nakamatsu,Masao Tanooka,Masahiro Tanaka,Yasumasa Nishimura (2022). Risk factors for radiation pneumonitis after rotating gantry intensity-modulated radiation therapy for lung cancer.
Anne Aupérin (2010). Meta-analysis of concomitant versus sequential radiochemotherapy in locally advanced non-small-cell lung cancer.
Lawrence Marks,Soren Bentzen,Joseph Deasy,Feng-Ming (spring) Kong,Jeffrey Bradley,Ivan Vogelius,Issam El Naqa,Jessica Hubbs,Joos Lebesque,Robert Timmerman,Mary Martel,Andrew Jackson (2010). Radiation Dose–Volume Effects in the Lung.
(2009). RTOG. 2011 Feb 9 -RTOG 0236 Protocol Information, A phase II trial of stereotactic body radiation therapy (SBRT) in the treatment of patients with medically inoperable stage I/II non-small cell lung cancer.
A Bezjak (2012). ONCOLOGY RTOG 0813 Seamless Phase I/II Study of Stereotactic Lung Radiotherapy (SBRT) for early stage, centrally located, Non-Small Cell Lung Cancer (NSCLC) in Medically Inoperable Patients.
Vitali Moiseenko,Jimm Grimm,Ellen Yorke,Andrew Jackson,Anthony Yip,Minh-Phuong Huynh-Le,Anand Mahadevan,Kenneth Forster,Michael Milano,Jona Hattangadi-Gluth (2020). Dose-Volume Predictors of Radiation Pneumonitis After Lung Stereotactic Body Radiation Therapy (SBRT): Implications for Practice and Trial Design.
Yinnan Meng,Haihua Yang,Wei Wang,Xingni Tang,Caiping Jiang,Yichao Shen,Wei Luo (2019). Excluding PTV from lung volume may better predict radiation pneumonitis for intensity modulated radiation therapy in lung cancer patients.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Dalenogare, M.O.. 2026. \u201cAre Known Lung Dose Limits Valid for all Patients?\u201d. Global Journal of Medical Research - F: Diseases GJMR-F Volume 23 (GJMR Volume 23 Issue F8): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.