## I. INTRODUCTION
Brunner glands (BG), first described by the swiss anatomist Johann Conrad Brunner in 1688, $^{1}$ are located predominantly in the submucosa of the proximal duodenum and are composed of cells with columnar to cuboidal cytoplasm and basal nuclei, arranged in lobules separated by delicate fibrous septa. They secrete alkaline mucus (composed of mucin glycoproteins, bicarbonate and various additional factors including epidermal growth factor, trefoil peptides, bactericidal factors, proteinase inhibitors, and surface-active lipids) that protect the epithelium from digestive enzymes. $^{2}$ The exact classification of Brunner glandlesions (BGL) is evolving and hyperplasia (BGH), adenoma (BGA), hamartoma and brunneroma, have been used as descriptors. In general, lesions smaller than $0.5\mathrm{cm}$ are considered hyperplasia and not true neoplasias. $^{3}$ Historically, BGL/BGH were first classified by Feyrter into three types: type 1 (diffuse nodular hyperplasia with sessile projections extending beyond the duodenal bulb), type 2 (nodular or sessile hyperplasia confined to the duodenal bulb), and type 3 (pedunculated or sessile adenoma forming a mass). $^{4}$ However, a clear pathologic distinction based on clinical pathologic correlation has not been developed and the malignant potential of these benign lesions remains uncertain. $^{5}$
BGL represent less than $1\%$ of primary tumors in the small intestine, and approximately $10\%$ of duodenal neoplasms. Rare reports have documented possible progression to carcinoma, $^{6}$ and presenting symptoms vary widely according to the size of the lesions. They tend to be asymptomatic until growing beyond $1.5 \mathrm{~cm}$, while tumors larger than $2 \mathrm{~cm}$ may manifest with upper gastrointestinal bleeding and obstruction. $^{3}$ The literature on genetic alterations in BGL is sparse and devoid of KRAS hits. We report the case of a 64-year-old male with a $1.5 \mathrm{~cm}$ polyp in the duodenum corresponding to a BGH type 3/BGA with a point mutation (G12D) in KRAS.
## II. CASE REPORT
A 64-year-old male with history of heart failure with reduced ejection fraction, chronic renal failure stage 5, coronary artery disease, diabetes mellitus, hypertension, stroke, benign prostatic hyperplasia and tobacco use disorder, presented to the hospital for a nephrology follow up visit. Detection of combined iron deficiency and chronic disease anemia (decreased hemoglobin $6.6\mathrm{g / dl}$, hematocrit $20.6\%$ and iron 34 ug/dL; with normal MCV 91.7 fL, and ferritin 69 ng/ml) prompted upper endoscopy and colonoscopy. The upper endoscopy showed a $1.5\mathrm{cm}$, pink-tan polyp in the duodenum, which was resected. The histopathological examination revealed BGA/BGH type 3 without dysplasia or malignancy (Figure 1 & 2). Immunohistochemistry revealed intact expression of DNA mismatch repair proteins (MLH1, MSH2, MSH6 and PMS2) supporting lack of microsatellite instability. Due to the rarity of the lesion next generation sequencing (Oncomine Focus, ThermoFisher) was performed on extracted DNA revealing a KRAS G12D genetic alteration. In addition, the colonoscopy revealed three tubular adenomas (one $0.4\mathrm{cm}$ pedunculated polyp in the ascending colon and two sessile polyps ranging from 0.3 to $0.5\mathrm{cm}$ in the transverse colon). Of note, the patient died three months later due to respiratory failure from SARS-CoV-2 infection.
## III. DISCUSSION
BGL present most commonly as an incidental endoscopic finding in asymptomatic patients during their fifth or sixth decade of life without a predilection towards gender or race. $^{7}$ Although BG proliferations are traditionally considered benign they can be premalignant $^{8}$ and develop dysplasia and even invasive carcinoma in $\sim 2\%$ and $0.3\%$ of cases, respectively. $^{9}$ However, the exact molecular pathogenesis is unknown. Brosens et al. reported a BG hamartoma in one patient with juvenile polyposis syndrome harboring a germline mutation in SMAD4/DPC4, a highly conserved transcription factor activated by TGF- $\beta$.[^10] Loss of the tumor suppressor LRIG1 (a transmembrane protein that interacts with EGFR family proteins) has been associated with increased proliferation of Brunner glands in mice and intestinal adenomatous polyps in humans.[^11] In addition, Levi et al. described BGH smaller than 1 cm in 20% of 10 patients with Cowden syndrome (CS),[^12] suggesting a pathogenic role for PTEN mutations, which are present in $\sim 85\%$ of patients affected by this syndrome.
Mutations in the Kirsten rat sarcoma viral oncogene homologue (KRAS) have not been previously reported in BGL to the best of our knowledge. This well-known proto-oncogene encodes a protein that acts as a molecular switch transducing extracellular signals from membrane receptors (like EGRF) to the cytosolic MAPK and PI3K/mTOR pathways, ultimately leading to activation of nuclear transcription controlling cell proliferation, differentiation, and survival.[13] KRAS is mutated in approximately $25\%$ of human tumors, representing one of the most commonly altered genes associated with cancer.[14] Missense mutations in KRAS stabilize an active GTP-bound form of the protein promoting oncogenesis. The G12D point mutation we identified in a BGA is a well-recognized and powerful cancer driver mutation with impaired GTPase catalytic activity. $^{15}$ It is also the most prevalent alteration in human cancer, which is present in $4.2\%$ of cases in the American Association of Cancer Research public database. $^{16}$ Interestingly, KRAS G12D is embryonic lethal in mouse models, but is sufficient to initiate transformation of fibroblasts in cell culture and to induce preneoplastic epithelial hyperplasias in the lung and gastrointestinal tract. $^{17}$ Because there is crosstalk between the MAPK, PTEN/PI3K and TGF- $\beta/$ BMP pathways, $^{18}$ it is possible that other altered genes (SMAD4, LRIG1 and PTEN) described in BGL may act in concert with KRAS to promote neoplasia. The classical adenoma-carcinoma sequence (with mutations in APC, KRAS, and p53) plays an important role in duodenal carcinogenesis when adenomatous change/dysplasia is present. $^{19}$ However, the validity of this paradigm in BG neoplasia is unclear. Of note, BG adenocarcinoma arising from BGH has been associated with GNAS mutations arising in foveolar metaplasia. $^{20}$ The BGA presented here did not show dysplasia or metaplasia, and raises a potential role for KRAS in the regulation of BG proliferation, which deserves further studies.
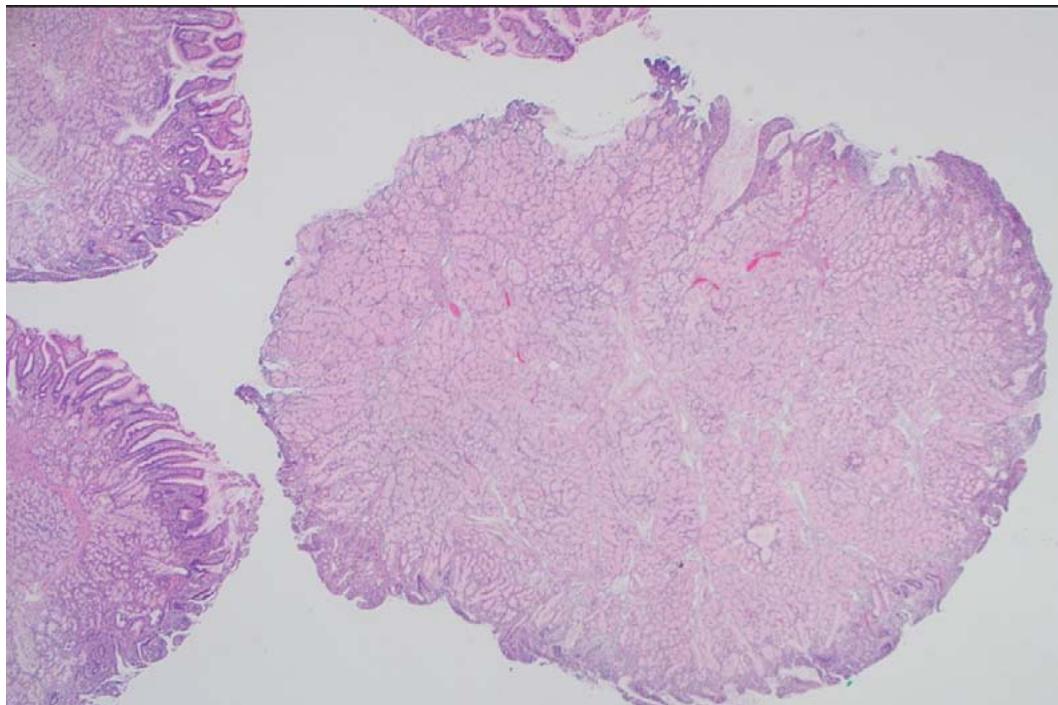 Figure 1: Photomicrograph of Brunner gland nodule (H & E x20)
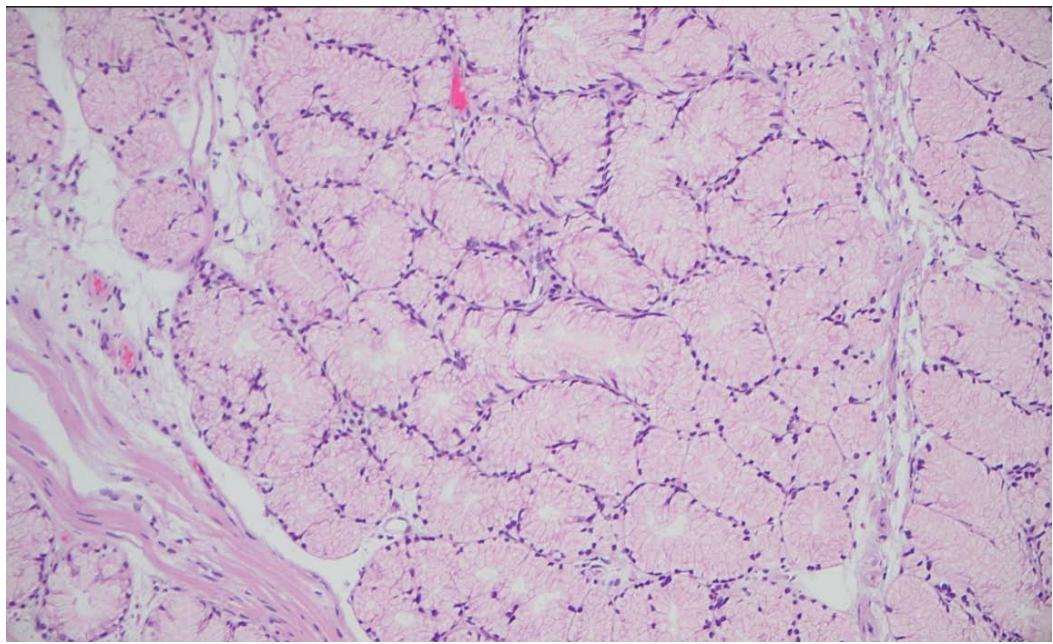 Figure 2: Photomicrograph of Brunner gland nodule (H & E x200)
Generating HTML Viewer...
References
20 Cites in Article
Rocco (2006). Large Brunner's gland adenoma: Case report and literature review.
William Krause (2000). Brunner's Glands: A Structural,Histochemical and Pathological Profile.
N Patel,A Levy,A Mehrotra,L Sobin (2006). Brunner's gland hyperplasia and Hamartoma: Imaging features with Clinicopathologic Correlation.
Woong Lee,Hyeon Yang,Yun Lee,Sung Jung,Gi Choi,Hoon Go,Anna Kim,Sang Cha (2008). Brunner's Gland Hyperplasia: Treatment of Severe Diffuse Nodular Hyperplasia Mimicking a Malignancy on Pancreatic-Duodenal Area.
M Zhu,H Li,Y Wu (2021). Brunner's gland hamartoma of the duodenum: A literature review.
M Koizumi,N Sata,K Yoshizawa (2007). Carcinoma arising from Brunner's gland in the duodenum after 17 years of observation -A case report and literature review.
Lcf De Nes,F Ouwehand,Sha Peters,M Boom (2007). A Large Brunner’s Gland Hamartoma Causing Gastrointestinal Bleeding and Obstruction.
M Brookes,S Manjunatha,C Allen,M Cox (2003). Malignant potential in a Brunner’s gland hamartoma.
Takaki Sakurai,Hiromi Sakashita,Gen Honjo,Ippei Kasyu,Toshiaki Manabe (2005). Gastric Foveolar Metaplasia With Dysplastic Changes in Brunner Gland Hyperplasia.
Laa Brosens (2011). Juvenile polyposis syndrome.
Y Wang,C Shi,Y Lu,E Poulin (2015). Loss of LRIG1 leads to expansion of Brunner glands followed by duodenal adenomas with gastric metaplasia.
Z Levi,H Baris,I Kedar (2011). Upper and lower gastrointestinal findings in PTEN mutation-positive Cowden syndrome patients participating in an active surveillance program.
Erika Martinelli,Floriana Morgillo,Teresa Troiani,Fortunato Ciardiello (2017). Cancer resistance to therapies against the EGFR-RAS-RAF pathway: The role of MEK.
R Burge,G Hobbs (2022). Not all RAS mutations are equal: A detailed review of the functional diversity of RAS hot spot mutations.
S Vatansever,B Erman,Z Gümüş (2019). Oncogenic G12D mutation alters local conformations and dynamics of K-Ras.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Nava, Victor E.. 2026. \u201cBrunner Gland Adenoma with a KRAS G12D Point Mutation\u201d. Global Journal of Medical Research - K: Interdisciplinary GJMR-K Volume 22 (GJMR Volume 22 Issue K6).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.