Drug databases have become a critical tool used by various stakeholders to make informed decisions about medication therapy. Globally, researchers have carried out primary studies that have documented and provided knowledge on the antiplasmodial activities of numerous plants. A central database designed to document vital efficacy and safety information on the most promising antimalarial-medicinal plants being recommended for advanced studies in the drug discovery and development pipeline is needed to serve as vital repository supporting the general public, researchers, academic institutions, pharmacological companies, policymakers, and patients relying on herbal antimalarials. Current malaria research programs require the use of advanced techniques in the development of new antimalarials.
## I. INTRODUCTION
The world has rich flora diversity, and various ethnopharmacological studies conducted have allowed for the documentation of a large number of plants used to treat malaria in the endemic areas of transmission, especially in Sub-Saharan Africa, parts of Asia, and South America. Information on herbal medicine research is currently documented in multiple databases designed for various purposes (Natural Medicines 2017; MedlinePlus 2017; National Center for Complementary and Integrative Health 2016; Memorial Sloan-Kettering Cancer Center 2017). Even so, a centralized database will better serve to promote cross-functional collaboration, facilitate information sharing among different teams and stakeholders involved in the drug development processes, support evidence-based practice, enhance overall productivity, and allow for effective dissemination of new updates. Therefore, there is a need for a central information system that will provide comprehensive data addressing efficacy and safety concerns in the discovery and development of effective herbal antimalarials that are better acceptable, and readily available to everyone.
Currently, conventional antimalarial drugs are still effective but are also threatened by increasing rate of drug resistance by the Plasmodium species. Safe and efficacious plant-based antimalarials offer important alternatives (Aracil and Green 2019). They are increasingly relied upon to combat malaria burdens in locally administered monotherapies, or as a mixture of plant formulations using two or more plant species (Omagha et al. 2021a Martey et al. 2013; Orabueze et al. 2018). Globally, researchers have carried out primary studies that have documented and provided knowledge on the antiplasmodial activities of numerous plant species. Researchers, in their commitment to providing data on clinical evidence to support plant use, and ultimately mitigate preventable deaths from malaria and its associated ailments, have also produced potent herbal treatments that significantly contributed to decrease in morbidity and mortality rates. Results have been encouraging. Between 2000 and 2023, an estimated 2.2 billion malaria cases and 12.7 million malaria deaths were averted worldwide, with 1.7 billion cases and 12 million deaths prevented in the WHO African Region alone (WHO 2024).
Research for alternative antimalarial drugs that are safe, inexpensive and readily available to people, particularly those in the developing countries has accelerated over the last two decades. Malaria drug discovery is undoubtedly challenging with additional hurdles to identify and develop compounds with properties that are predictive of good efficacy and safety in humans. Scientists are optimistic as they also have got various opportunities too (Wells et al., 2015). To continue the current rate of productivity will require new strategies for hunting out promising candidates and continued increases in efficiency test compounds and pick the winners early. It is therefore imperative to have a clear understanding of potential hazards associated with new drugs at earlier stages of drug discovery programs (Hodgson 2001). To achieve this drug safety screening goals, important strategies including computational approaches offers some cost-effective ways to identify new plant medicines and associated active ingredients, as well as methods to uncover patterns in phytochemical data and drive the discovery of new pharmaceutical products. These novel plant-based antimalarials that meet the required parameters for biological activity and are suitable for further studies can be selected and properly documented using a successfully published and properly maintained drug database site. The centralized database can also be used to provide evidences for herbal medicine to inform treatment choices.
## II. WHY SAFETY SCREENING AND TOXICOLOGICAL STUDIES FOR ANTIMALARIALS?
Firstly, finding novel antimalarials that are efficacious and safe is currently crucial to the reduction of widespread drug resistance, disease burden, and economic loss due to malaria. Promising antimalarials are expected to be safe, especially in children and pregnant women (Delete 2020). Also, since herbal antimalarials are popularly relied on, and have proven to contain various phytochemicals which have medicinal properties, there are calls to improve the quality of ethnobotanical and pharmacological studies, and provide clinical data on safety and efficacy that will guide their standardization and development (Omagha et al. 2020; 2021b; 2023a, b; Dawet et al. 2023; Ocan et al. 2023; Czygan 2019; Wachtel-Galor and Nenzie 2011; Noronha et al. 2020). Fortunately, with the rise in the number of promising antimalarial remedies, techniques in computational methods such as high throughput virtual screening, ligand docking tools, ADMET (absorption, distribution, metabolism, excretion and toxicity) profiling and other modern drug assessment tools and software are being applied in drug discovery, drug assessment and drug development to accelerate our goal to discover new antimalarials.
## III. THE PROMISE OF HIGH-THROUGHPUT AND MULTI-OMICS SCREENING FOR HERBAL ANTIMALARIALS
Toxicity of some popularly used antimalarial drugs has been questioned (WHO, 2003), and some plant-based combinations demonstrated antagonistic effects, reduced survival time and exerted multiple organ toxicity (Moronkejiet al. 2019; Orabueze et al. 2018; Martey et al. 2013). Currently, the investigation into herbal toxicity is limited by the lack of a good animal model, a passive reporting system, analytical methodology that is not well characterized, limited knowledge of active ingredients and chemical interactions, limited knowledge of the mechanism of action, variability in the preparation method, and interpatient variability. Therefore, screening of drug databases using bioinformatics, multi-omics technologies such as genomics, proteomics, and metabolomics and other high-throughput or virtual screening techniques is valuable to develop new antimalarials (O'Connor et al. 2023). In modern drug development pipeline, these computational approaches can be applied for various purposes including antimalarial research. They can offer a cost-effective method to eliminate unsafe products at the initial stages of safety assessment (Rusyn and Daston 2010). They can ultimately facilitate the discovery of desirable drugs to help cope with the continually demanding pharmaceutical needs, drugs that reduce disease symptoms without causing severe side effects in the patient, and are affordable yet profitable for drug companies (David et al. 2009; Drews and Ryser 1997), drugs that have a low chance of resistance (Davies and Davies 2010), and those that have low deleterious effect on the environment. It is our hope that safety assessments using these resources will help in the drug development process by documenting safety profiles, and ultimately facilitate the discovery of desirable drugs from plants.
Drug Discovery: With the help of computational tools, drug discovery process is moving towards greater automation of the research process (Siddharthan et al. 2016). The core components of the modern drug discovery process include molecular modelling, pattern discovery, chemical structure prediction, access to heterogeneous databases and system modelling.
Drug Bioavailability and Bioactivity: The key characteristics of drugs are Absorption, Distribution, Metabolism, Excretion, Toxicity and efficacy—in other words bioavailability and bioactivity. Assessment of ADMET is increasingly required earlier in the discovery process, at a stage when considered compounds are numerous.
Study Interesting Compounds: Researchers identify and study the lead compounds to discover and develop a chemical compound that has some activity against malaria. The chemical structure and properties of the potential drug are then optimized. This process can be enhanced using software tools (Sakkiah et al. 2012; Petersen and Christensen 2011).
Drug Target Screening and Identification: The number of potential targets for drug discovery processes is increasing exponentially. The identification of new targets and subsequent discovery of compounds that act through specific pathways are being considered based on their locations within the malaria parasite. Several promising targets for drug intervention have been revealed in recent years and are now available to drug discovery scientists. As more genes are identified, the drug discovery cycle becomes more data-intensive (Whittaker 2003; Chen 2008).
Drug Target Validation: The possibility of failure in the clinical testing and approval phases can be moderated by drug target validation. Validation helps researchers to identify any secondary target that the drug may bind to, which may lead to any sort of unwanted or adverse reaction. Ideally, the drug candidate should be such that it binds to a single target only, as binding to other targets, apart from the actual target leads to unwanted pharmacological actions.
Role of Artificial Intelligence (AI) in Drug Screening and Optimization Strategies: AI tools and methods can play important roles in optimizing these drug screening strategies by using algorithms to analyze vast datasets, to identify bioactive compounds and their interactions, to predict drug targets, to speed up identification of promising candidates, to optimize formulations, to enhance the therapeutic effects of drugs, and to facilitate herbal medicine development. Methods such as machine learning, deep learning, network pharmacology, bioinformatics, systems biology, chemical informatics, and computer vision, can link chemical composition, herbal medicine, drugs, targets, symptoms, and diseases. This can contribute to herbal medicine development by streamlining the processes, and saving time and resources required. However, certain considerations including ethical implications, quality and availability needs to be addressed in order to apply AI technologies for drug discovery and development (Wu et al. 2022; Zeeshan et al. 2025).
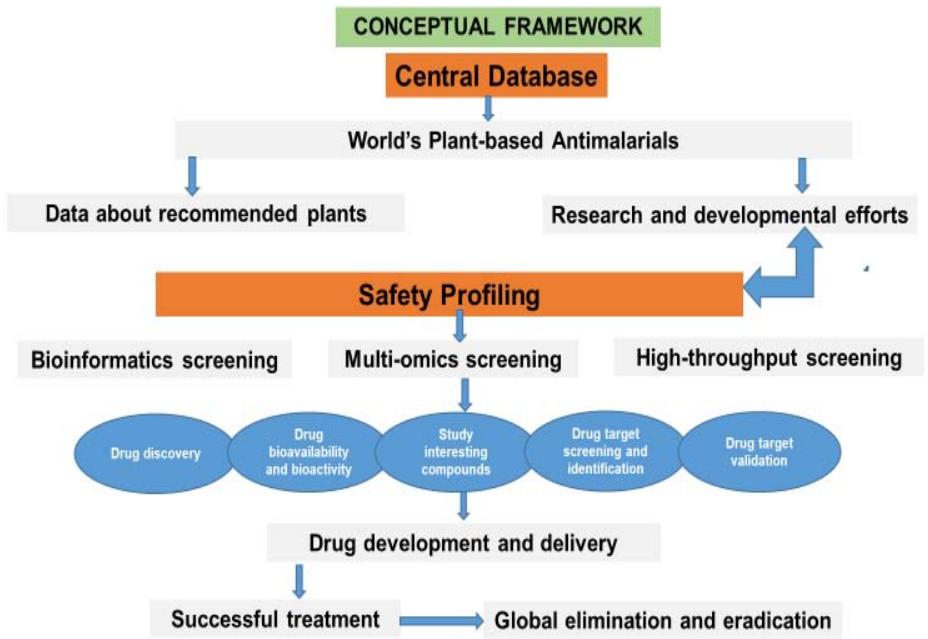 Figure 1: A framework highlighting research needs in the field of drug discovery and development in malaria endemic countries
## IV. HOW A CENTRALIZED DATABASE FOR NOVEL ANTIMALARIALS MIGHT HELP
information on their use to fight diseases such as malaria (Mojab 2012). Some databases of herbal medicines have been published (Natural Medicines 2017; MedlinePlus 2017; National Center for Complementary and Integrative Health 2016; Memorial Sloan-Kettering Cancer Center 2017). They include:
1. The natural medicines comprehensive database
2. The medicinal plant database of India
3. Herbmed
4. Dr. Duke's phytochemical and ethnobotanical database
5. Natmed pro (formerly natural medicines)
6. Micromedex
7. RXlist-Alternatives
Without a centralized system, data on medicinal plants in scientific and non-scientific reports are found scattered across different systems and locations, making it challenging to access, share, analyse, and recommend them. On the other hand, a global database of relevant information purposefully selected will help provide a single, centralized system where comprehensive data about promising plant medicines used to treat malaria can be managed and utilized. Providing this information in a single source will make it easier and faster to retrieve and analyze data. It will also facilitate better efficient data mining, reporting, and decision-making processes, as users do not have to navigate multiple systems or locations to gather the needed information. Building and maintaining a global database for plant-based antimalarials from across the world is expected to make outstanding contributions for the development of new drugs including:
1. To centralize data and facilitate reporting on the status of their efficacy in malaria endemic countries, clinical trials, research studies, manufacturing processes, and regulatory submissions.
2. To facilitate global collaboration, information sharing, and effective communication among various geographically dispersed teams and stakeholders involved in the drug development and manufacturing processes.
3. To provide guidance on their use based on recommendations following studies conducted in accordance with standard protocols.
4. To serve as excellent reference resource for educating health care professionals in various academic institutions who want to become knowledgeable advisers about plant-based medicines.
5. To serve as reliable information source that can be scaled and adapted when data needs evolves.
Putting together such a critical tool that will be efficient, reliable and up-to-date requires a broad range of skills and advanced tools. It will require prioritizing accuracy, comprehensiveness, clarity, and accessibility, while also addressing legal and ethical considerations. Information needs to be organized so it can be easily accessed, rapidly processed, and updated for the general public. A significant amount of resources and efforts are also needed.
## V. CONCLUSION
Research needs in the field of herbal medicines in malaria endemic countries where medicinal plants are commonly used for treatment are huge, but are balanced by the potential benefits. Publishing and maintaining a centralized database of promising antimalarials used in various countries will advise on their ability to produce desired results (efficacy), and the type and likelihood of adverse effects (safety). It will also bring about positive developmental changes that will benefit stakeholders including:
1. Pharmaceutical companies who use published findings to inform on drug discovery and development efforts.
2. Monitoring/regulatory agencies creating awareness and ensuring their safe and judicious use.
3. The scientific community and researchers.
4. Individuals seeking information of herbal antimalarial treatment options to locally meet their demands of malaria treatments.
5. Academic institutions who need properly documented resources for training.
Therefore, there is a need for a central database to document safety profiles of the world's antimalarials as a vital part of global surveillance and knowledge-sharing ecosystem. This important step ultimately aimed to improve patients' safety calls for support including funding and expert collaborations amongst passionate teams helping to ensure safe medicines for all.
### Declarations:
Scope Statement: This article provides valuable information highlighting the importance of a centralized database built for information on promising herbal antimalarial treatments used across the world as a strategy to contribute towards antimalarial drug discovery and development.
Ethics Approval and Consent to Participate: Not Applicable
Consent for Publication:The authors read and approved this paper for publication.
Availability of Data and Materials: Not Applicable
Competing Interests: The authors declare that there are no conflicts of interest.
Authors' Contributions: Omagha R.: conceptualization, resources, writing original draft, review and editing. Agbaje E.O., Alimba C.G., and Ajaegbu H.C.N.: review and editing.
Funding Statement: No financial support yet.
Generative AI disclosure: No Generative AI was used in the preparation of this manuscript.
Generating HTML Viewer...
References
39 Cites in Article
(2017). Explore key features of the most comprehensive database of its kind.
(2017). Drugs, herbs.
Memorial Sloan,-Kettering Cancer Center (2017). About herbs, botanicals & other products.
Aracil Amparo,Julia Green (2019). Plants with antimalarial properties: A systematic review of the current clinical evidence.
Rachel Omagha,Emmanuel Idowu,Chibuisi Alimba,Adetoro Otubanjo,Adeniyi Adeneye (2021). Survey of ethnobotanical cocktails commonly used in the treatment of malaria in southwestern Nigeria.
Onk Martey,O Shittah-Bay,J Owusu,Lkn Okine (2013). The antiplasmodial activity of an herbal antimalarial, AM 207 in Plasmodium bergheiinfected Balb/c Mice: absence of organ specific toxicity.
(2024). Addressing inequity in the global malaria response.
Xi Zhou,Yongquan Li,Xin Chen (2010). Computational identification of bioactive natural products by structure activity relationship.
T Belete (2020). Recent Progress in the Development of New Antimalarial Drugs with Novel Targets (2020).
R Omagha,E Idowu,C Alimba,A Otubanjo,E Agbaje,Hcn Ajaegbu (1948). Physicochemical and phytochemical screening of six plants commonly used in the treatment of malaria in Nigeria..
R Omagha,E Idowu,C Alimba,A Otubanjo,W Oyibo,E Agbaje (2021). In vivo antiplasmodial activities and acute toxicity assessment of two plant cocktail extracts commonly used among Southwestern Nigerians.
Rachel Omagha,Emmanuel T Idowu,Chibuisi G Alimba,Adetoro O Otubanjo,Esther O Agbaje,Wellington A Oyibo (1948). Clinico-biochemical and histopathological alterations in sub-chronically exposed mice (Mus Musculus) to polyherbal antimalarials.
Rachel Omagha,Emmanuel T Idowu,Chibuisi G Alimba,Adetoro O Otubanjo,Esther O Agbaje,Wellington A Oyibo (1947). Alterations in testis histology, reproductive hormones and abnormal sperm morphology in mice treated with polyherbal antimalarials.
F-C Czygan (2019). The role of medicinal plants as an important part in modern medicine (2019). AdvHorticSci 1990:56-60. A brief review of traditional plants as sources of pharmacological interests.
M Ocan,Loyce Ojiambo,K (2023). Efficacy of antimalarial herbal medicines used by communities in malaria affected regions globally: a protocol for systematic review and evidence and gap map.
S Wachtel-Galor,I Benzie (2011). Herbal medicine: an introduction to its history, usage, regulation, current trends, and research needs IFF.
(2011). Herbal Medicine: Biomolecular and Clinical Aspects.
Timothy Wells,Rob Van Huijsduijnen (2015). Ferroquine: welcome to the next generation of antimalarials.
Monica Noronha,Vishakha Pawar,Anil Prajapati,R Subramanian (2020). A literature review on traditional herbal medicines for malaria.
M Frenkel,E Arye (2001). The growing need to teach about complementary and alternative medicine: questions and challenges.
Osvaldo Almeida (2003). <i>Atlas: Mental Health Resources in the World 2001</i>. Edited by S. Saxena. (Pp. 55; available free from WHO, CH1211 Geneva 27, Switzerland.) World Health Organization: Geneva. 2001..
A Moronkeji,G Eze,M Igunbor,A Ogbonna,A Moronkeji (2019). Histomorphological and biochemical evaluation of herbal cocktail used in treating malaria on kidneys of adult wistar rats.
C Orabueze,A Sunday,O Duncan,C Herbert (2018). In vivoantiplasmodial activities of four Nigerian plants used singly and in polyherbal combination against Plasmodium berghei infection.
O Martey,O Shittah-Ba,J Owusu,L Okine (2013). The Antiplasmodial Activity of an Herbal Antimalarial, AM 207 in Plasmodium berghei-infected Balb/c Mice: Absence of Organ Specific Toxicity.
I Rusyn,G Daston (2010). Computational toxicology: realizing the promise of the toxicity testing in the 21st century.
E David,T Tramontin,R Zemmel (2009). Pharmaceutical R&D: the road to positive returns.
Jürgen Drews,Stefan Ryser (1997). Drug Development: The role of innovation in drug development.
J Davies,D Davies (2010). Origins and evolution of antibiotic resistance.
Lance O'connor,Blake O'connor,Su Lim,Jialiu Zeng,Chih Lo (2023). Integrative multi-omics and systems bioinformatics in translational neuroscience: A data mining perspective.
N Siddharthan,M Raja Prabu,B Sivasankari (2016). Bioinformatics in Drug Discovery a Revi.
S Shaikh,B Jayaram (2007). A Swift All-Atom Energy-Based Computational Protocol to Predict DNA-Ligand Binding Affinity and Δ T m.
Rasmus Petersen,Kathrine Christensen,Andreana Assimopoulou,Xavier Fretté,Vassilios Papageorgiou,Karsten Kristiansen,Irene Kouskoumvekaki (2011). Pharmacophore-driven identification of PPARγ agonists from natural sources.
P Whittaker (2003). What is the relevance of bioinformatics to pharmacology?.
Y Chen,F Chen (2008). Identifying targets for drug discovery using bioinformatics.
F Mojab (2012). Antimalarial natural products: a review.
M Fowler (2006). Plants, medicines and man.
A Dawet,D Yakubu,R Omagha,J Gushit (2023). Isolation of the active ingredients of antimalarial activity of the stem bark of Pseudocedrelakotschyi (Dry zone cedar).
C Wu,J Chen,Lai-Han Leung,E,Chang,Wang (2022). Editorial: Artificial Intelligence in Traditional Medicine.
Zeeshan Afsar,R Nethaji,K Vimal,K Shantiya,M Manjunatha,Rajendra Prasad,M,Babu Ganesan (2025). Modern Concepts of Integrating Artificial Intelligence with Traditional and Herbal Medicine: A Review.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Dr. Omagha Rachel. 2026. \u201cBuilding a Global Central Database for Promising Herbal Antimalarials from Various Countries: Strategies for Safe and Effective Drug Development\u201d. Global Journal of Medical Research - B: Pharma, Drug Discovery, Toxicology & Medicine GJMR-B Volume 25 (GJMR Volume 25 Issue B1): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Drug databases have become a critical tool used by various stakeholders to make informed decisions about medication therapy. Globally, researchers have carried out primary studies that have documented and provided knowledge on the antiplasmodial activities of numerous plants. A central database designed to document vital efficacy and safety information on the most promising antimalarial-medicinal plants being recommended for advanced studies in the drug discovery and development pipeline is needed to serve as vital repository supporting the general public, researchers, academic institutions, pharmacological companies, policymakers, and patients relying on herbal antimalarials. Current malaria research programs require the use of advanced techniques in the development of new antimalarials.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.