## I. BACKGROUND
Lymphangiomas (Lms) or, as recently preferred (according to ISSVA classification for vascular anomalies 2018), "lymphatic malformations" consist of lymphatic channels anastomosis and cystic spaces, as a result of abnormal connections between the lymphatic and venous systems or abnormal development or location of lymphatic vessels. Their incidence is 1/12000 births, accounting for $6\%$ of all benign lesions of infancy and childhood. Cervico-facial lesions represent $75\%$ of the cases and $80 - 90\%$ become symptomatic within the first 2 years of life (both sexes equally affected) because of their progressive enlargement with growth. Many of them are congenital and often associated with other vascular or chromosomal abnormalities (e.g. Turner syndrome), without a clear familial tendency; however, they may arise or increase in size due to trauma, inflammation or lymphatic obstruction. Spontaneous regression within 18-24 months is documented in $1.6 - 16\%$ of cases. Classification into macro-cystic, micro-cystic and mixed is based upon the diameter of the cysts. Presentation varies from asymptomatic, soft, not compressible, transilluminant neck tumefaction to dysphagia, malocclusion, sleep disordered breathing, respiratory distress and recurrent infections [1]. Since they are not encapsulated, they show infiltrative growth and often are not dissociable from airway, nerves and blood vessels [2].
Histology is similar to hamartomas, although some state a lymphangiectasic or neoplastic nature, without any malignant potentiality [3]. Ultrasonography documents mono- or multiloculate fluid-superfluid lesions with a substantial lack of flow with Doppler mode (figure/patient 1-2). Computer Tomography (CT) describes low attenuation masses with occasional fluid level and minimum septal and peripheral enhancement. Magnetic resonance imaging (MRI) detects iso- or hyposignal on T1-weighted sequences and hyper-signal on T2-weighted sequences, a halo of enhancement around the septa of macro-cystic lesions and peri lesional lymphoedema (figure/patient 2-3) [4].
While small and asymptomatic lesions might benefit from purely conservative management (e.g.
compression, analgesia), larger and symptomatic (e.g. dysphagia, dyspnoea) malformations would require non-conservative treatment; this implies a multidisciplinary approach (surgical, radiological, physician's) which varies according to each patient's characteristics and lesions, considering the lack of univocal indications for treatment as well as all those issues related to the young age of the patient (psychological or parental concern). Traditional surgical treatment has been considered for many years the gold standard for lymphatic malformations and is still considered the most definitive solution; however, as lymphangiomas are infiltrative, a complete eradication is often impossible [5]. This explains the relatively high percentage of recurrences (up to $27\%$ ) and intraoperative risks, with a mortality rate of $2 - 6\%$. At the present, surgery is indicated for lesions larger than $3\mathrm{cm}$ (sometimes for debulking, followed by sclerotherapy), with progressive growth, bone erosion, dyspnoea or dysphagia [6]. It is also advocated for the resection of remaining fibrotic tissue after sclerotherapy or as a first-line therapy for lesions outside the cervico-facial region, associated with few intraoperative risks or aesthetic sequelae [7]. Minor complications include lymphorrhea, keloids, dehiscences, fistulas, and the need of a prolonged post-operative drainage from the wound, with an associated risk of infection[8].
The aim of the study was to review and discuss the non-surgical strategies for the management of cervical lymphangiomas, with particular attention to the sclerosing agents.
## II. MATERIAL AND METHODS
All relevant articles including "cervical lymphangiomas" or/and "sclerotherapy" were searched on PubMed, Cochrane and Embase platforms. Inclusion criteria were: (1) studies published mostly within the last fifteen years, (2) studies including paediatric population, (3) studies reporting advantages, success rate, recurrence rate, complications and dose for each and every sclerosing agent, (4) studies written in English. In addition, instructions for the procedure of ultrasound or CT-guided sclerotherapy and other alternative treatments were considered.
## III. RESULTS
As many as 47 articles dealing with sclerotherapy of cervical lymphangiomas in children were collected. Beside short information about the first therapeutic approaches, laser therapy, radiofrequency ablation and oral medications (e.g. Sirolimus) including the newest target-therapy, a much larger knowledge was obtained about sclerotherapy's rationale, steps and agents, from the most employed (OK-432, bleomycin/pingyangmycin, doxycycline, sodium tetradecyl-sulphate) to potential alternatives (such as ethanol) or occasionally-used substances (e.g., acetic acid, Ethibloc, Tissucol, Polidocanol).
## IV. DISCUSSION
Historically, the first non-surgical treatments for cervical lymphangiomas were with simple direct drainage, aspiration and radiation. A combined radiochemotherapeutic approach was performed in selected cases of hemolymphatic malformations [1].
Laser therapy is an alternative to surgery for small and superficial (cutaneous or oral) lymphangiomas or for debulking of invasive, large and non-excisable lymphangiomas. Traditional techniques consist of the resection and removal by photocoagulation with argon, carbon dioxide, Nd:Yag, KTP and diode lasers. Pain, oedema and swelling are associated with complete healing at 6-8 weeks, although a scar may persist for many years. Ten to $33\%$ of cases are complicated by intraoperative bleeding or nerve injury. Namour and coll suggested a peculiar therapeutic method for debulking of invasive and extensive lesions within oral soft tissues, preventing the patients from mutilation. A CO2 laser machine with output power of 2W in defocus and in non-contact mode for at least 3 min is used, with a distance between the laser handpiece and the tissular impact point around 6 cm, the delivered focal point at $0.3 \mathrm{~mm}$, the effective spot diameter range at tissue at about 2 cm (power density $= 0.63 \mathrm{~W} / \mathrm{cm}^{2}$ ) and the estimated energy density range at 114.65-191 J/cm2. This protocol does not cause disintegration/vaporisation but only overheating, with subsequent fibrous healing. Out of seventeen patients, only three (18%) experienced recurrence and no major complication (embolism, infection or mutilation) occurred. Other minor treatments include cryotherapy, diathermy and electro-cautery [9].
Another example of local treatment is offered by radio-frequency ablation (RFA) or hypothermic ablation, destroying lesions at lower temperatures $(40 - 70^{\circ})$ with subsequent lower damages within surrounding tissues. Micro-cystic lymphangiomas in mouth, throat, pharynx, retro-pharynx and tongue may benefit from RFA, which also helps to stop accidental or intra-operative bleedings [10].
Sclerotherapy is nowadays largely employed in case of macro-cystic or mixed lymphangiomas, where the size of the cysts allows them to be punctured and a wider distribution of the agent is achieved. However, OK-432, bleomycin and above all doxycycline have recently proved effective also with micro-cystic variety [2]. The rationale comes from the observation that lymphangiomas can reduce their size or regress after a spontaneous infection, probably due to the destruction of the epithelium, reduction of lymph production and collapse of the cyst: thus, the idea of adopting sclerosing agents to mimic such an effect. Puncture of the dominant cyst (or more, in case they are non-communicating) with a fine needle (20 G or more) is followed by aspiration (in order to enhance the SA effect) and injection, in one or more times, of the sclerosing agent, using the same amount as the aspirated fluid (if impossible, half of the lesion's volume). It can be repeated in case of partial response or whenever more administrations are chosen, each session being separated by weeks up to a month. General or loco-regional anaesthesia is preferred in children or uncooperative patients and in adults or small cystic malformations, respectively; the patient's position is changed many times to favour a uniform distribution of the agent and the treatment lasts up to two hours. A postoperative compressive bandage is advised in order to increment the time of contact between the solution and the cyst's wall and to prevent seroma formation, bleeding or effusion of the SA. The procedure can be performed under ultrasound or CT: the first is cheaper, more available, does not employ ionising radiations (thus being advisable in children and young adults), defines better the different components within the lesion, grants different cranio-caudal angles and reduces the risk of accidental puncture of large blood vessels; however, it is operator-dependent and offers a narrow field of view, with the risk of missing some important findings, especially at the post-procedure check. CT instead is easier to perform and provides a more panoramic view but implies radiation and the needle path to lie on the axial plane with its full length [1]; a detailed example of this procedure is offered in figure (patient) 4, whereas figures (patients) 5 and 6 show examples of successful outcomes comparing lymphangiomas before and after sclerotherapy. Fluoroscopic guidance is also reported, especially in case of the most superficial lesions, with the possibility to inject contrast medium into the lesion in order to highlight communications between the intra-lesional spaces and establish the amount of SA to be used [4]. Complications include intraoperative bleeding (due to their dysplastic nature), accidental injuries to nerves, vessels, organs and other tissues (due to extravasation), peri-lesional fibrosis and aesthetic sequelae (due to necrosis followed by second-intention reparation), dose-dependent cardio-pulmonary toxicity (especially with bleomycin) and acute respiratory insufficiency (with large lymphangiomas undergoing inflammation, necrosis and quick volumetric expansion); the latter could be managed with dexamethasone or, preferably, avoided by splitting the treatment in more sessions [7]. An example of follow-up program would consist of a clinical examination after one to three weeks, ultrasonography after six to twelve weeks and then (depending on the results of ultrasound) MRI, unless evidence of early recurrence or any other complication occurs [8]. A review by Adams et al didn't prove the superiority of sclerotherapy over surgery but showed it
was the treatment of choice in most major paediatric vascular anomaly centres: surgery was reserved for refractory cases, with sclerosing agents not improving either clinics or aesthetics, micro cystic lesions or those associated with life-threatening airway obstruction. Nowadays there are no worldwide-accepted guidelines and patient selection seems to guide the choice. In addition, neither surgery nor sclerotherapy can guarantee complete healing with just one session: it is actually advisable to perform multiple treatments or combine them[1].
When dealing with sclerotherapy, the first substances to be employed (with poor results) were boiling water, quinine, urethane, iodine tincture, nitromin, sodium morrhuate $5\%$ and acetic acid 40 to $50\%$, the last two with little more reference in literature [1]: sodium morrhuate is currently employed for orbital lymphangiomas; acid acetic causes an area of coagulative necrosis much wider than ethanol (with pain and tingling sensation) but with a faster and more complete regression [11]. Many others sclerosing agents (SA) were used by the years, but no guidelines are currently available due to the infrequency of the disease and the limited studies; therefore, the decision is based on Centres and operators according to their experience and the availability and side-effects of the agents [12].
OK-432 (Picibanil; table 1) was first used by Ogita in 1987 [13]. It comes from the lyophilisation of a mixture of Streptococcus pyogenes and G-Penicillin. It favours the production of IL-1, IL-2, IL-6, INF-Y and TNF, the activation of neutrophils, macrophages, NK and T lymphocytes, the apoptosis of the epithelium and increases the permeability of the endothelium, accelerating the lymphatic fluid drainage. Compared to others SA, OK-432 is associated with a lower risk of extravasation and subsequent peri-lesional fibrosis, systemic toxicity or/and aesthetic sequelae [14]. In addition, according to both Efe and Hazim (2016), the lesion is still feasible for surgery even after sclerotherapy failure [15,16]. Luzzato et al (2000) confirmed its usefulness for residual and recurrent lesions, as well as the low invasivity and scarring [17]. Sichel et al (2004) agreed with the lack of a significant peri-lesional fibrosis [18]. The percentage of success is $50 - 92\%$ (remaining high even with repeated injections), with $43\%$ of complete and $29.3\%$ of partial remissions [19], whereas the frequency of the recurrences is around $11\%$ [15] and the complications are rare and mostly local (pain, heat, induration, erythema, oedema, swelling, aesthetic sequelae, swallowing difficulty and odynophagia, infections) with sporadic fever, sepsis and shock, especially in patients with allergy for G penicillin [1]. Yoo et al (2009) stressed the safety of OK-432, reporting minor complications only and stating a high long-term efficacy [20]. Rebuffini et al (2012) also reported anaemia and a transitory increase of platelets' concentration [21]. The low systemic toxicity allows OK-432 to be used also with micro-cystic (where the percentage of success reaches nearly $50\%$ ) or intraparenchymal components, where the risk of tissue absorption is the most [22]. Ruiz et al (2004) confirmed its feasibility for micro-cystic lesions, as well as those associated with a risk of airway obstruction [23]. Ogita recommends 0,1 mg/10 cc with a maximum of 20 mL of solution or 0,2 mg of substance [24]. Despite all its advantages and proven effectiveness, however, OK-432 is less and less employed in the UK.
Bleomycin sulphate (table 2) is an antibiotic with antitumour action, inhibiting DNA synthesis. Tanigawa et al were the first to employ it as a sclerosing agent, reporting a lower recurrence rate than surgery and stating the possibility to use it in case of surgery failure [25]. Sung et al also used it for debulking of unresectable lesions [26]. It shows good responsivity $(88\%)$, a discrete frequency of complete remissions $(36-63\%)$ and a low recurrence rate $(15\%)$, as sclerosing agent [27]. Zulfiqar et al (1999) confirmed it safety and effectiveness, especially when dealing with macro-cystic lesions [28]; a decade later, Sanldas et al (2011) confirmed a higher success when treating unilocular malformations [29]. Analogous outcomes, along with similar success rates, were obtained by Kurmar (2012) [30], Jain (2013) [31], Porwal (2018) [32] and Hashmi (2020) [33]. Orford et al (1995), obtained minimal surgical scars and declared a low risk of potential injury of nerves and/or blood vessels. Local signs of inflammation (restricted movement of the neck, pain, swelling, induration, stridor, difficulty in breathing or swallowing, intra-luminal bleed, infections) subside spontaneously within few days. Systemic effects range from mild (vomiting, diarrhoea, flu, local hyperpigmentation, hyperkeratosis and thickening of the skin) to anaphylaxis [34]. Mathur et al (2005) declared no major complication or mortality [35]; the same conclusion was drawn by Rozman (2010) [22]. Sporadic cases of pulmonary fibrosis actually occur only when high doses are employed (total administration of $400\mathrm{U}$ or single administration of $30\mathrm{mg/mm}^2$ ), as during systemic chemotherapy or with renal clearance under $25-35\mathrm{mL/min}$. Follow-up to monitor for pulmonary fibrosis is left to exercise tolerance and patients are also advised to avoid live vaccine for 3 months. The suggested dose is between 0,5 and $1\mathrm{mg/kg}$ with a maximum of $5\mathrm{mg/kg}$ [35].
Pingyangmycin or bleomycin A5 (table 4) is similar to common bleomycin (A2) but it is cheaper and determines less peri-lesional fibrosis and complications, with possible hair loss, gastrointestinal reactions, fever, rash. The recommended dose is $1\mathrm{mL} / \mathrm{cm}^2$ and must be lower than $8\mathrm{mg}$ per single injection and $40\mathrm{mg}$ in total. Jia et al. (2014) treated orbital and peri-orbital lymphatic malformations with PYM, with a mean volume decrease of $84\%$ after a median number of 2 injections, and no recurrences at 8 months were observed [12]. Gao (2002) employed it also with oral, maxillofacial and cervical lesions, considering it as a potential primary therapy [14].
Doxycycline (table 3) is a bacteriostatic antibiotic, which inhibits angiogenesis through the blockage of the production of metalloproteinases and vascular endothelial growth factor (VEGF) [10]. It is inexpensive, widely available and it has minimal side effects, including dental discolouration in children, allergic reactions [2], haemolytic anaemia, hypoglycaemia, neurological complications and rare cases of methaemoglobinaemia [36]. Pain, swelling, haemorrhage and cellulitis may occur, as well as scarring, skin excoriation and Horner's syndrome [2]. Complete or near complete response was achieved from the very beginning of its use, as reported from the experience of Cordes (2007) [37] and Nehra (2008) [38]. Later, Jeffrey Cheng (2015) performed sclerotherapy with doxycycline on a larger cohort of subjects, reporting an efficacy of $84.2\%$, and insufficient responses or recurrences in $33\%$ [2]. The recommended dose varies between 20 and $150\mathrm{mg}$ at a concentration of 10 to 20 mg/mL [36]. Despite many Authors, such as Cahill (2011) [39] and Farnoosh (2015) [36], state its primary role for the treatment of large macro-cystic malformations, some others like Burrows (2008) declare an even higher efficacy than OK-432 in case of microcystic lesions [11]. Shields (2009) agreed with the former and employed it also in cases of post-surgical recurrences [40]. Shergill (2012) put together the theories from the previous authors claiming that doxycycline could be used for nearly all types (macrocystic, micro-cystic or mixed) of lymphangiomas [41].
Despite the few quotations, sodium tetradactyl-sulphate or sotradecol $3\%$ (table 4) is used in many paediatric centres in the UK as second like after doxycycline. It has shown to determine an average response of $80 - 90\%$ (complete in $40\%$ ) and complication rate of $17\%$ only, including swelling, oedema, mild allergic reaction, chronic facial pain, infections, cutaneous necrosis and nerve injury. Dose varies between 3 to $6\mathrm{mL}$ and its main indication remains orbital LM [25]. Farnoosh (2015) achieved similar results, with an even better outcome if combined with doxycycline [36].
Alcohol $98\%$ (table 4), used for many arteriovenous malformations, causes rapid cellular dehydration and protoplasm precipitation. On one hand, it shows good therapeutic response (from 64 to $96\%$ ), large availability and low cost. On the other hand, it can determine unexpected damage within the surrounding tissues and, at high doses, severe systemic effects such as hypotension, respiratory depression, arrhythmias, seizures, hypoglycaemia and exitus. For this reason, low doses are employed $(0,5 - 1\mathrm{mL} / \mathrm{kg})$, mostly associated with injuries of skin, mucosae and peripheral nerves
(7,5-27,95%) or thrombotic phenomena. Recurrence rate is around $30\%$ [6]. Impellizzeri et al. (2010) reported their experience with CT-guided instillation of 5-15 mL of alcoholic solution with complete disappearance of the lesion in 7 patients, with only one needing a second injection. Only one patient experienced self-limiting erythema and tenderness and no recurrence was observed at 2 years [1]. Puig et al. affirmed that the use of ethanol for lymphatic malformations could cause the extravasation as a major risk. To avoid it, he proposed a double-needle procedure to limit the total volume of ethanol injected in order to reduce intra-lesional pressure and thus extravasation [42]. Ethanolamine oleate is obtained from a combination of an organic base with oleic acid and shows alcohol-like effect, although it has lower toxicity [12].
Although few experiences about the use of acetic acid at $30 - 50\%$ (table 4) demonstrate a more rapid effect compared with the other SA and the frequent need for a single treatment session only, the extravasation into the nearby tissues represents a serious issue. As an example by Won et al, a patient with a cervico-mediastinal lymphangiomas experienced infiltration in the lung parenchyma surrounding the lesion [43].
Alcoholic solution of Zein (Ethibloc, table 4) contains Zein (a prolamine), diatrizoate sodium (radio-opaque marker), poppyseed oil and $96\%$ ethanol. It is biodegradable, effective and safe, but the risk of scars, salivary fistulas, infections and the poor aesthetic results (30 days of trans-cutaneous elimination) have limited its use. The recommended dose is between 1 and $7.5 \mathrm{~mL}$. M. A. Emran et al. reported satisfactory to excellent results in $84\%$ of macro-cystic/mixed and in $77\%$ of micro-cystic lesions and considered Ethibloc an effective alternative to surgery for macro-cystic lymphangiomas or post-surgical recurrences. Failure and subsequent recurrence rate seems around $23\%$ [44].
Fibrin glue (Tissucol, table 4) is an expensive haemostatic agent making the cysts collapse, the dose being 10 to $15\%$ of the suctioned volume; Castanon et al obtained complete remission of 8 of 9 monocystic neck lymphangiomas [45]. Polidocanol (table 4), a local anaesthetic, is administered at the dose of $1\mathrm{mL}$ for each cm of lesion, does not need further anaesthesia and causes erythema with induration of the skin only; Jain et al achieved a volume reduction of $96\%$ to $100\%$ in 3 patients [46].
To the knowledge, some substances are administered orally in addition to local treatments and overseen by a physician in normal circumstances. Sirolimus or rapamycin is a mTOR-PI3K pathway inhibitor increasingly employed when surgery fails or is not feasible. It has been used for orbital lymphatic malformations and (later) conjunctival or superficial periocular lesions [12, 47]. Other systemic treatments include Cyclophosphamide (alkylating antineoplastic agent), interferon, steroids (promoting inflammation and cicatrisation respectively), isotretinoin, Bevacizumab, Thalidomide (anti-angiogenic), Alpelisib and TIE2 (targeted therapy against PIK3CA and tunica intima endothelial kinase 2), propranolol and sildenafil (relaxing smooth muscle thus causing cystic decompression and opening of secondary lymphatic spaces). The last one, a phosphodiesterase type 5 inhibitor used for erectile dysfunction and pulmonary hypertension, proved effective in reducing the number and severity of bleeding episodes, especially with macro-cystic and mixed lesions [12]. Newest target therapies, appearing more effective with macro-cystic lesions, include PI3K inhibitors (LY294002, BYL719, wortmannin), AKT inhibitors (ARQ092, MK-2206), MAPK inhibitors, (U0126), multiple kinase inhibitors (sorafenib, trametinib), VEGF-A inhibitors (bevacizumab), BMP and Wnt modulators (dorsomorphin, LDN-193189 and calyculin A), JAK inhibitors (ruxolitinib), calcium channel blockers (amlodipine), KATP activators (minoxidil), zoledronic acid, interferon a 2b, prednisolone, sunitinib. However, large-scale studies are required in order to confirm efficacy and potential side-effects for each substance [10].
A recent review by Liu et al. (2021) confirms how, although surgery remains the first-line strategy for lymphangiomas (especially large lesions for possible complete removal), risk of scars, incomplete resection/recurrences and injury to blood vessels or cranial nerves (especially the submandibular branch of the facial nerve) is high. Thus, despite above-mentioned side effects like soft-tissue oedema and skin necrosis as well as variable recurrence rates, sclerotherapy may often represent the elective treatment; moreover, microcystic lesions appear more and more susceptible to SA like OK-432. However, since "single treatments" often achieve insufficient results, ongoing strategies tend to administer multiple therapies. For example, surgery may obtain debulking of large lesions, with remaining tissue being treated with sclerotherapy (e.g. bleomycin) and systemic drugs administered to alleviate the symptoms of pain and bleeding. Analogous strategies might be employed with superficial micro-cystic mucosal lesions, where RFA or laser-therapy plays the main role. Moreover, oral medications themselves may be used prior to surgery and/or sclerotherapy. However, further studies are necessary in order to promote univocal guidelines; thus, to date, as already stated by Liu et al., "individual therapies" (also including target-therapy) represent the most appropriate strategy in the treatment of lymphangiomas, with no exception for cervical lesions [10].
## V. CONCLUSION
Treatment of cervical lymphangiomas represents a rare but challenging issue. Despite the wide range of treatments discussed, cervical lymphangiomas remains a relatively infrequent finding. Thus, a limited number of studies is reported in literature and small cohorts of patients are considered, with subsequent difficulties in performing a reliable meta-analysis about the efficacy of any single treatment.
Despite the lack of worldwide-accepted guidelines, our research highlights the role of sclerotherapy as the first-line non-surgical strategy because of the lower rates of aesthetic sequelae, recurrence, complications (e.g. injury to blood vessels or cranial nerves) and mortality and the better course, so that it might represent a valid or equivalent alternative to surgical therapy. Moreover, the two treatments can be used in combination (e.g. when remaining lymphangiomatous or fibrous tissue is present) or sclerotherapy can prove more efficient (e.g. micro-cystic lesions when using OK-432). Sclerosing agents should usually be employed with macro-cystic lesions, and no substantial difference is reported in terms of efficacy from a SA to another, all of them ranging from about 60 to $90\%$ in different studies. Although no longer used in several countries (including the UK), OK-432 would be optimal for its minimal extravasation (and thus perilesional fibrosis and aesthetic sequelae), the lowest rate of both local and systemic complications and recurrences (around $10\%$ ) and, eventually, the feasibility with either micro-cystic lesions. Bleomycin can represent a valid option as long as low doses are administered, due to the well-known risk of pulmonary fibrosis; the issue is partially solved when employing pingyangmycin which is still, however, indicated mostly for peri-ocular lesions. Doxycycline is more available, cheap and apparently even more efficient than OK-432 in cases of micro-cystic lymphangiomas; as an alternative, we suggest tetradactyl-sulphate. Despite its wide availability and low cost, the higher risks of neurovascular/visceral injuries (about 10 to $30\%$ ), systemic side effects and recurrences (around $30\%$ ) make ethanol a definitely second-line treatment.
However, several considerations must be made. Firstly, multicentric prospective studies are necessary in order to better evaluate this technique and define the best sclerosing agent to be used. Secondly, as already mentioned, ongoing strategies tend to combine the above-mentioned treatments (including oral medications and the newest "target-therapies") according to each patient's characteristics and lesions (as well as psychological aspects), giving rise to a multi-faceted approach.
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
The authors have no relevant financial or non-financial interests to disclose.
All authors contributed to the study conception and design, read and approved the final manuscript.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent was obtained from all individual participants included in the study.
Consent for publication was obtained for every individual person's data included in the study.
The authors affirm that human research participants provided informed consent for publication of the images.
1. Impellizzeri P, Romeo C, Borruto FA et al. Sclerotherapy for cervical cystic lymphatic malformations in children. Our experience with computed tomography-guided $98\%$ sterile ethanol insertion and a review of the literature. J Pediatr Surg. 2010; 45(12): 2473-8.
2. Cheng J. Doxycycline sclerotherapy in children with head and neck lymphatic malformations. J Pediatr Surg. 2015; 50(12): 2143-2146.
3. Joshi PS, Hongal B, Sanadi A. Cystic lymphangioma: A differential diagnosis. J Oral Maxillofac Pathol. 2015; 19(3): 393-395.
4. Monsignore LM, Nakiri GS, Santos D, Abud TG, Abud DG. Imaging findings and therapeutic alternatives for peripheral vascular malformations. Radiol Bras. 2010; 43(3): 185-194.
5. Verma R, Verma RR, Verma RR Sardana NK. Isolated Lymphangiomatous Polyp Nasopharynx in an Adult First Case Report in English Literature. Indian J Otolaryngol Head Neck Surg. 2014; 66(4): 460-463.
6. Manzini M, Schweiger C, Manica D, Kuhl G. Response to OK-432 sclerotherapy in the treatment of cervical lymphangioma with submucosal extension to the airway. Braz J Otorhinolaryngol. 2020; 86(1): 127-129.
7. Olímpio HO, Bustirff-Silva J, Oliveira Filho AG, Araujo KC. Cross sectional study comparing different therapeutic modalities for cystic lymphangiomas in children. Clinics (Sao Paulo). 2014; 69(8): 505-508.
8. Baraldini V, Dalmonte P, El Hachem M et al. Linee guida anomalie vascolari. SISAV - Società Italiana per lo Studio delle Anomalie Vascolari.
2014.
9. Nammour S, Vanheusden A, Namour A, Zeinoun T. Evaluation of a New Method for the Treatment of Invasive, Diffuse, and Unexcisable Lymphangiomas of the Oral Cavity with Defocus CO2 Laser Beam: A
- 20-Year Follow-Up. Photomed Laser Surg. 2016; 34(2): 1-6.
10. Xiaowei Liu, Cheng Cheng, Kai Chen, Yeming Wu, Zhixiang Wu. Recent Progress in Lymphangioma. Front Pediatr. 2021 Dec 15; 9:735832.
11. Wiegand S, Eivazi B, Zimmermann AP, Sesterhenn AM, Werner JA. Sclerotherapy of lymphangiomas of the head and neck. Sclerotherapy of Lymphangiomas. Head Neck. 2011; 33(11): 1649-55.
12. Lam SC, Yuen HKL. Medical and sclerosing agents in the treatment of orbital lymphatic malformations: what's new? Curr Opin Ophthalmol. 2019; 30(5): 380-385.
13. Ogita S, Tsuto T, Tokiwa K, Takahashi T. Treatment of Cystic Hygroma in Children with Special Reference to OK-432 Therapy. Z Kinderchir. 1987; 42(5): 279-281.
14. Gao Q, Wang C, Wen Y. A study of effects of pingyangmycin injection on treatment of lymphangiomas in oral, maxillofacial and cervical regions. Hua Xi Kou Qiang Yi Xue Za Zhi. 2002; 20(3): 184-6.
15. Efe N, Altas E, Mazlumoglu MR et al. Excellent Result with the Use of Single-Dose OK-432 in Cervical Macrocystic Lymphangioma. J Craniofac Surg. 2016; 27(7): 1802-1803.
16. Hazim M, Moses SS, Tang IP. Intralesional Injection of OK-432 in Cystic Hygroma. Med J Malaysia. 2016; 71(6): 328-330.
17. Luzzatto C, Midrio P, Tchaprassian Z, Guglielmi M. Sclerosing treatment of lymphangiomas with OK-432. Arch Dis Child. 2000; 82(4): 316-318.
18. Sichel JY, Udassin R, Gozal D, Koplewitz BZ, Dano I, Eliashar R. OK-432 Therapy for Cervical Lymphangioma. Laryngoscope. 2004; 114(10): 1805-1809.
19. Knipping S, Bau V; Lymphatische Malformationen im Kopf-Hals-Bereich. HNO. 2011; 59(7):683-688.
20. Yoo JC, Ahn Y, Lim YS et al. OK-432 sclerotherapy in head and neck lymphangiomas: Long-term follow-up result. Otolaryngol Head Neck Surg. 2009; 140(1): 120-123.
21. Rebuffini E, Zuccarino L, Grecchi E, Carinci F, Merulla VE. Picibanil (OK-432) in the treatment of head and neck lymphangiomas in children; Dent Res J. (Isfahan) 2012; 9(2): S192-S196.
22. Rozman Z, Thambidorai CR, Zaleha AM, Zakaria Z, Zulfiqar MA. Lymphangioma: Is intralesional bleomycin sclerotherapy effective? Biomed Imaging Interv J 2011; 7(3):e18.
23. Ruiz Jr E, Valera ET, Veríssimo F, Tone LG. OK-432 therapy for lymphangioma in children. J Pediatr. (Rio J) 2004; 80(2): 154-8.
24. Zhou G, Zheng JW, Mai HM et al. Treatment guidelines of lymphatic malformations of the head and neck. Oral Oncol. 2011; 47(12): 1105-1109.
25. Tanigawa N, Shimomatsuya T, Takahashi L et al. Treatment of cystic hygroma and lymphangioma with the use of bleomycin fat emulsion, Cancer 1987; 60(4): 741-749.
26. Sung MW, Chang SO, Choi JH, Kim JY. Bleomycin sclerotherapy in patients with congenital and lymphatic malformation in the head and neck. Am J Otolaryngol. 1995; 16(4): 236-241.
27. Karavelioğlu A, Temucin CM, Tanyel FC, Ciftci AO, Şenocak ME, Karnak I. Sclerotherapy with bleomycin does not adversely affect facial nerve function in children with cervicofacial cystic lymphatic malformation. J Pediatr Surg. 2010; 45(8): 1627-1632.
28. Zulfiqar MA, Zaleha AM, Zakaria Z, Amin T. The Treatment of Neck Lymphangioma with Intralesional injection of Bleomycin. Med J Malaysia. 1999; 54(4): 478-81.
29. Sandlas G, Kothari P, Karkera P, Gupta A. Bleomycin: A worthy alternative. Indian J Plast Surg. 2011; 44(1): 50-53.
30. Kumar V, Kumar P, Pandey A, Gupta DK, Shukla RC, Sharma SP, Gangopadhyay AN. Intralesional Bleomycin in Lymphangioma: An Effective and Safe Non-Operative Modality of Treatment. J Cutan Aesthet Surg. 2012; 5(2): 133-136.
31. Jain V, Mohta A, Sengar M, Bansal K, Val K. Use of intralesional bleomycin as primary therapy in macrocystic lymphangiomas. Indian J Dermatol Venereol Leprol. 2013; 79(4): 524-5.
32. Porwal PK, Dubey KP, Morey A, Singh H, Pooja S, Bose A. Bleomycin Sclerotherapy in Lymphangiomas of Head and Neck: Prospective Study of 8 Cases. Indian J Otolaryngol Head Neck Surg. 2018; 70(1): 145-148.
33. Hashmi AMKS, Hussain M, Ahmad S, Gillani K. Efficacy of intralesional bleomycin injection sclerotherapy in macrocystic lymphangioma in paediatric patients. J Ayub Med Coll Abbottabad. 2020; 32(1): 42-45.
34. Orford J, Barker A, Thonell S, King P, Murphy J. Bleomycin Therapy for Cystic Hygroma. J Pediatr Surg. 1995; 30(9): 1282-1287.
35. Mathur NN, Rana I, Bothra R, Dhawan R, Kathuria G, Pradhan T. Bleomycin sclerotherapy in congenital lymphatic and vascular malformations of head and neck. Int J Pediatr Otorhinolaryngol. 2005; 69(1): 75-80.
36. Farnoosh S, Don D, Koempel J, Panossian A, Anselmo D, Stanley P. Efficacy of doxycycline and sodium tetradecyl sulfate sclerotherapy in pediatric head and neck lymphatic malformations. Int J Pediatr Otorhinolaryngol. 2015; 79(6): 883-7.
37. Cordes BM, Seidel FG, Sulek M, Giannoni CM, Friedman EM. Doxycycline sclerotherapy as the primary treatment for head and neck lymphatic
- malformations. Otolaryngol Head Neck Surg. 2007; 137(6): 962-964.
38. Nehra D, Jacobson L, Barnes P, Mallory B, Albanese CT, Sylvester KG. Doxycycline sclerotherapy as primary treatment of head and neck lymphatic malformations in children. J Pediatr Surg. 2008; 43(3): 451-460.
39. Cahill AM, Nijs E, Ballah D et al. Percutaneous sclerotherapy in neonatal and infant head and neck lymphatic malformations: a single center experience. J Pediatr Surg. 2011; 46(11): 2083-2095.
40. Shiels WE II, Kang DR, Murakami JW, Hogan vMJ, Wiet GJ. Percutaneous treatment of lymphatic malformations. Otolaryngol Head Neck Surg. 2009; 141(2): 219-224.
41. Shergill A, John P, Amaral JG. Doxycycline sclerotherapy in children with lymphatic malformations: outcomes, complications and clinical efficacy. Pediatr Radiol. 2012; 42 (9): 1080-1088.
42. Puig S, Aref H, Brunelle F. Double-needle sclerotherapy of lymphangiomas and venous angiomas in children: a simple technique to prevent complications. Am J Roentgenol 2003; 180(5): 1399-401.
43. Je Hwan Won, Byung Moon Kim, Chul-Ho Kim, Sang Woo Park, Man Deuk Kim. Percutaneous Sclerotherapy of Lymphangiomas with Acetic Acid. J Vasc Interv Radiol 2004; 15: 595-600.
44. Emrana MA, Duboisb J, Labergea L, Al-Jazaeria A, Bu" tetra A, Yazbecka S. Alcoholic solution of zein (Ethibloc) sclerotherapy for treatment of lymphangiomas in children. J Pediatr Surg. 2006; 41(5): 975-9.
45. Castanon M, Margarit J, Carrasco R, Vancells M, Albert A, Morales L. Long-Term Follow-Up of Nineteen Cystic Lymphangiomas Treated With Fibrin Sealant. J Pediatr Surg. 1999; 34: 7276-1279.
46. Jain R, Bandhu S, Sawhney S, Mittal R. Sonographically guided percutaneous sclerosis using $1\%$ polidocanol in the treatment of vascular malformations. J Clin Ultrasound. 2002; 30: 416-423.
47. Amodeo I, Colnaghi M, Raffaeli G et al. The use of sirolimus in the treatment of giant cystic lymphangioma; Four case reports and update of medical therapy. Medicine (Baltimore). 2017; 96(51): e8871.
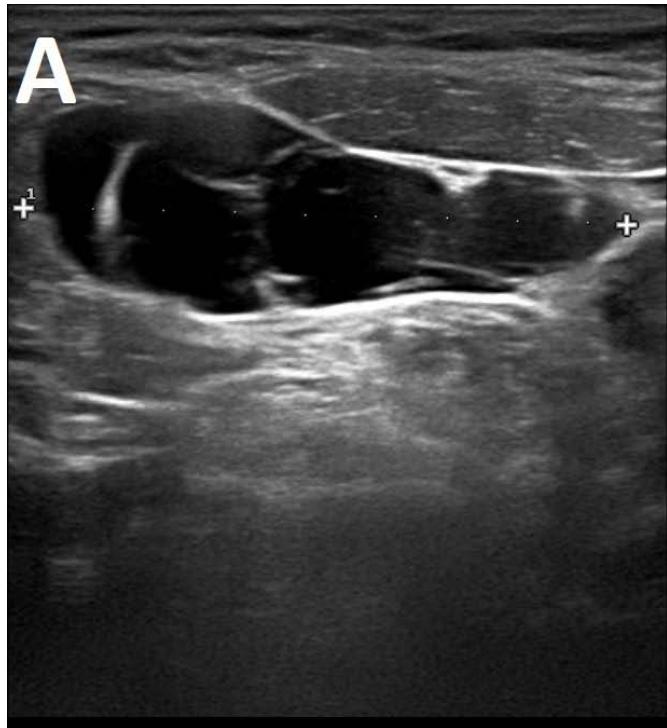
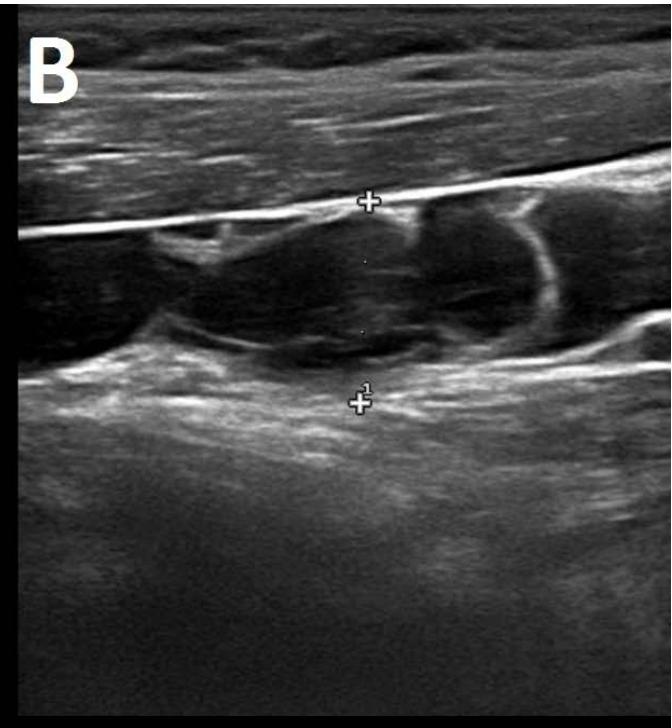
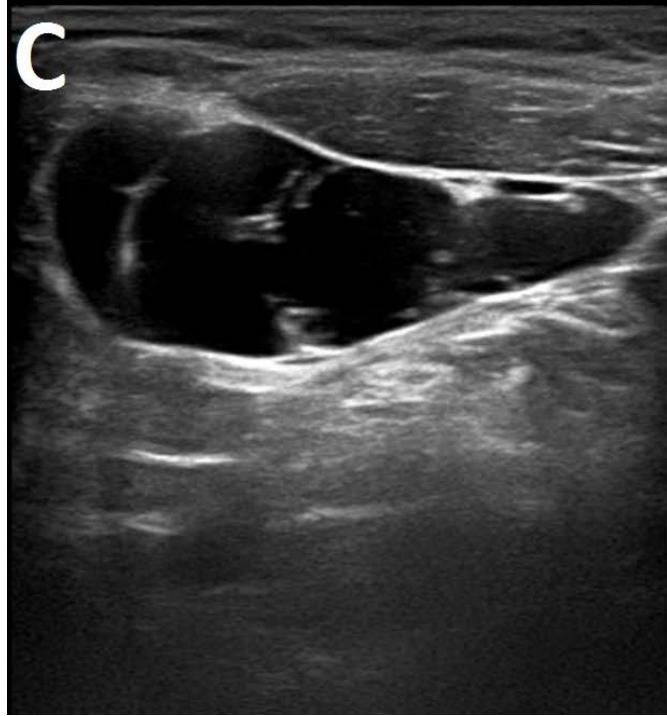
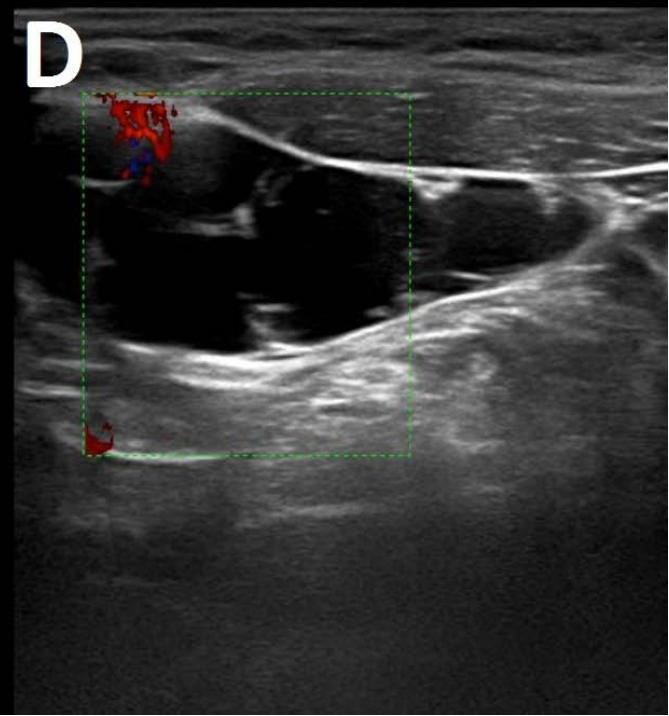
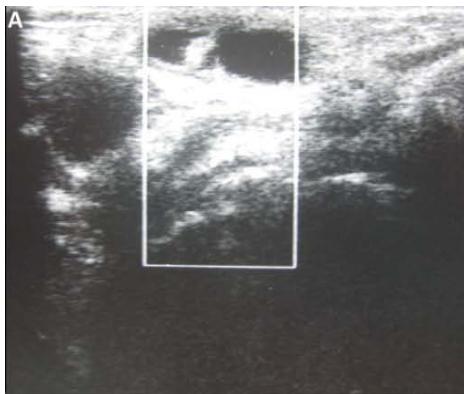
Figure/Patient 1
A 7 year-old girl presenting with a tumefaction at the level of the right latero-cervical region. US scans show a $43 \times 14$ mm subcutaneous fluid formation with septa extending from the root of the neck to the submandibular region (A-B). Doppler-mode demonstrates lack of blood flow within the lesion or the septa (C-D). Diagnosis of cystic lymphangioma is done.

Figure/Patient 2
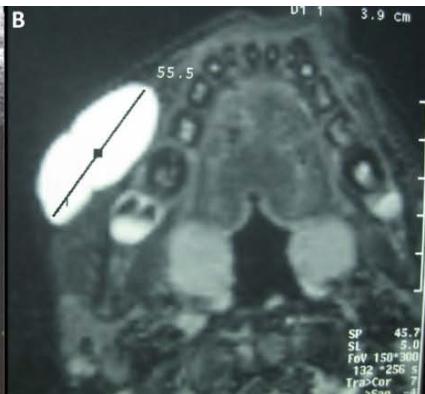
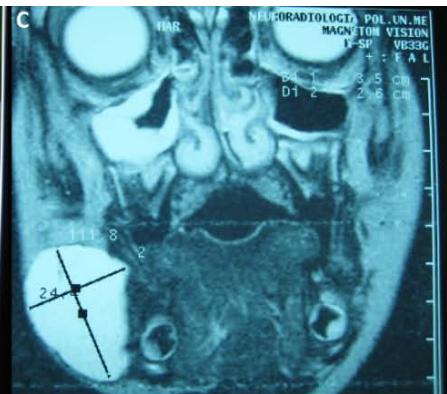
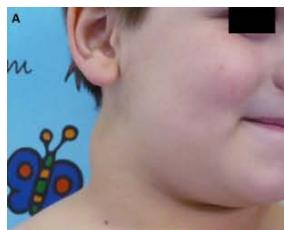
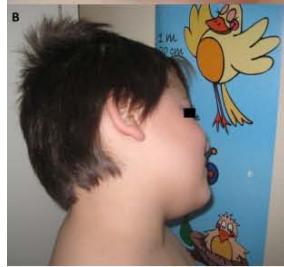
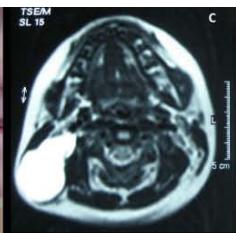
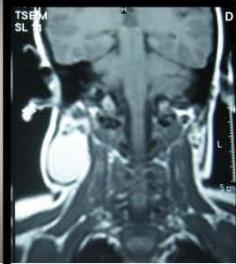
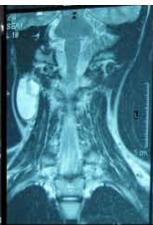
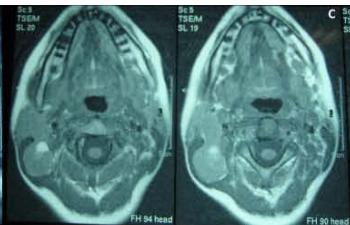
A case of right mandibular lymphangioma in a 10 year-old boy, hypoechoic with some septations and no blood vessels (A) and hyper-intense on T2 axial (B) and coronal (C) MR scans.
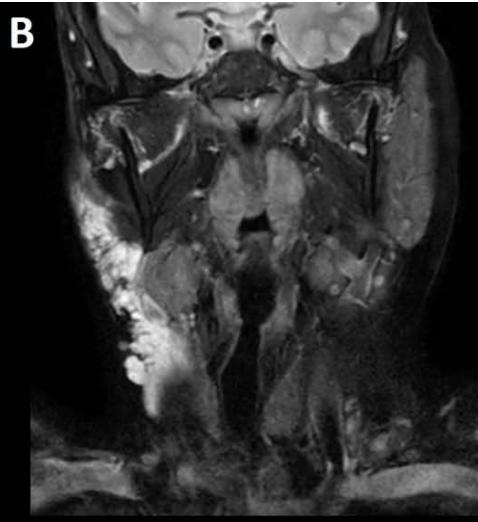
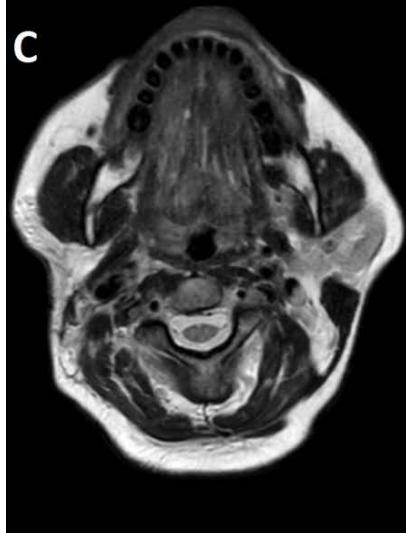
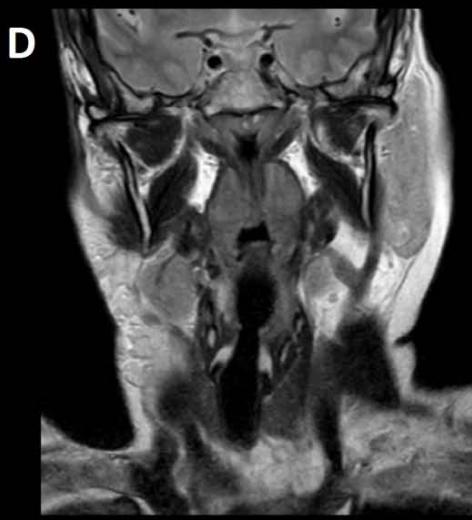
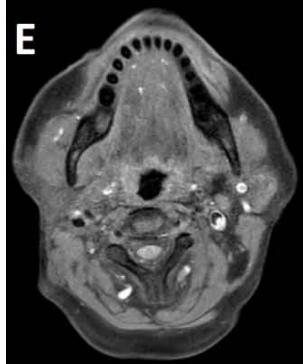
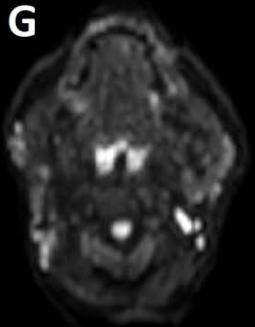
Figure/Patient 3
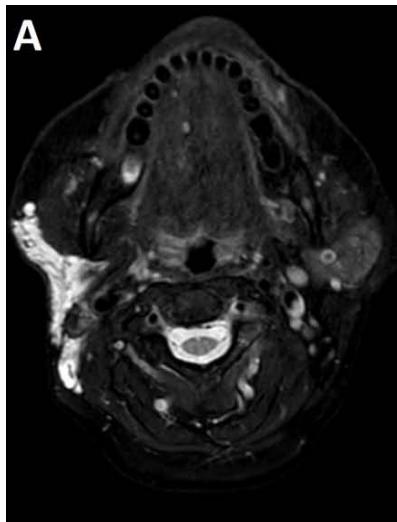
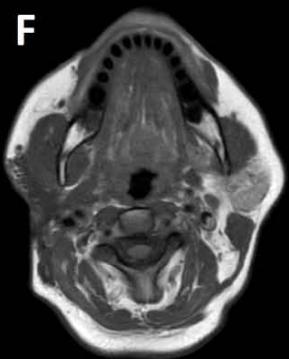
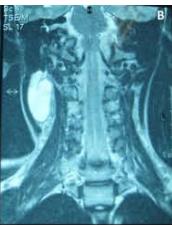
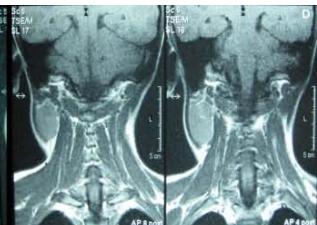
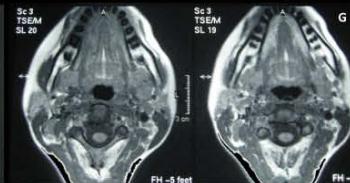
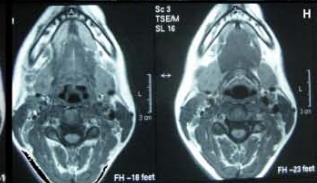
An 8 year-old girl with prior surgical excision of a right latero-cervical lymphangioma presenting with a new-onset tumefaction within the same region. MRI scans show a 9-10 cm subcutaneous multilocular lesion, extending between two virtual planes crossing the retro-molar trigone and the thyroid respectively, with deep invasion of parapharyngeal spaces cranially and the space between the submandibular gland and the vascular peduncle of the neck caudally. The lesion appears hyper-intense on T2 weighted scans with (A-B) and without (C-D) fat suppression, hypo-intense on T1 weighted scans with (E) and without (F) fat suppression and hyper-intense on DWI scans with high B values (G). Diagnosis of loco-regional recurrence of cystic lymphangioma is done.
Figure/Patient 4
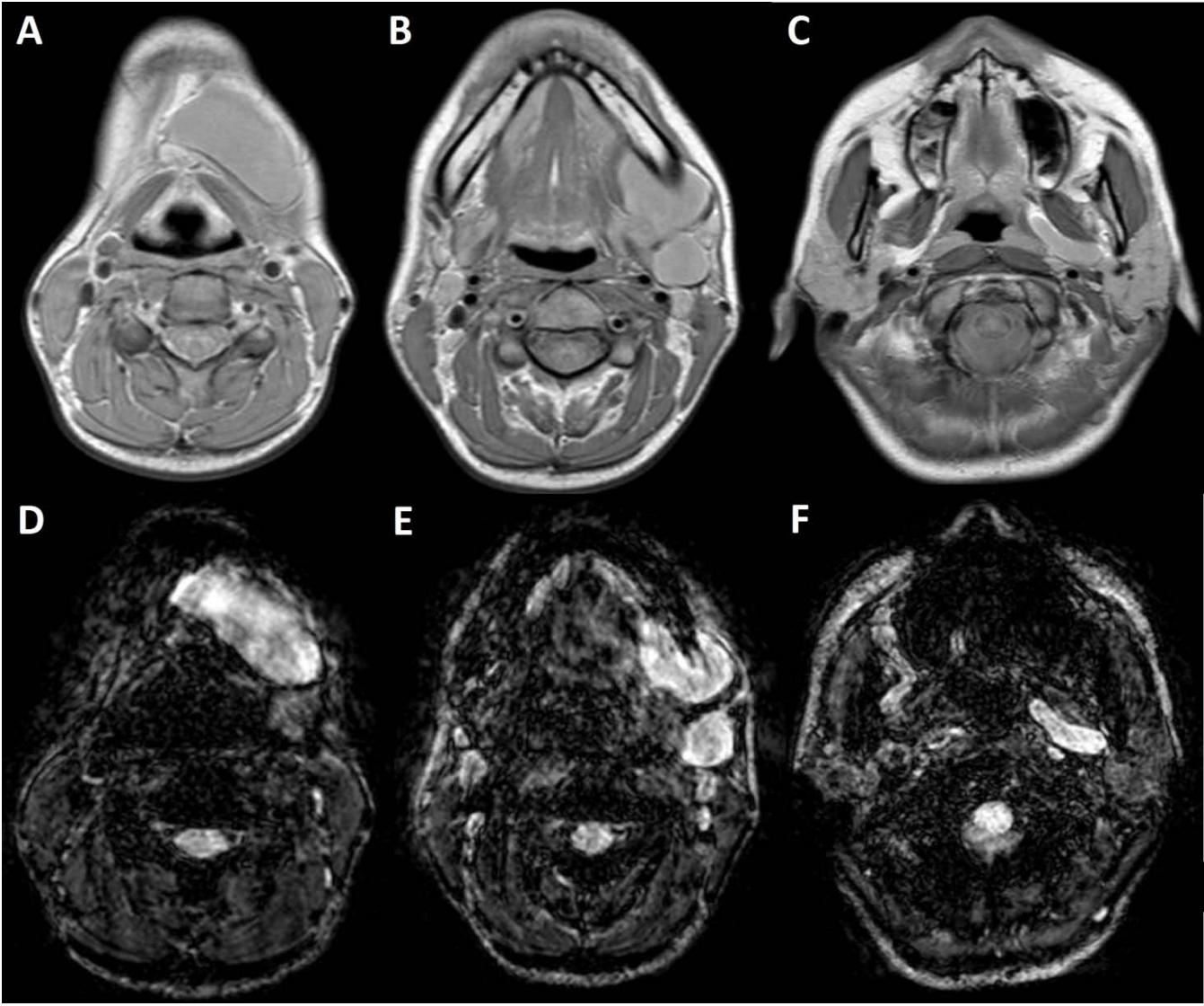
1. A 7 year-old girl presenting with a tumefaction at the level of the left latero-cervical region. MRI scans show a subcutaneous lesion occupying the left submandibular, parotidal, para-pharyngeal and part of the masticatory spaces. The lesion appears slightly hyper-intense on T1 (A-C) and markedly hyper-intense on T2 (D-F) scans. Diagnosis of cystic lymphangioma is done.
2. The same patient undergoing two different sessions (A-D and E-G, respectively) of sclerotherapy with absolute ethanol. An 18-G needle is introduced into the lesion and about 10 cc of citrine-turbid fluid are aspirated (B and F). Then, 5 cc of ethanol are injected and the patient is mobilised in order to distribute the SA throughout the lesion (D and G). The patient is dismissed and a one-week antibiotic therapy is prescribed.
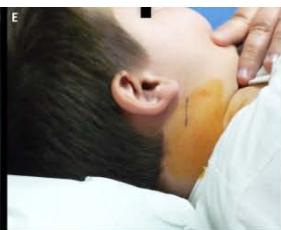
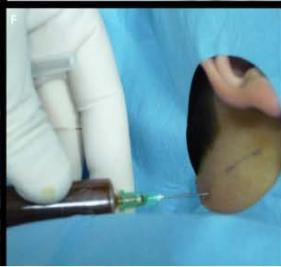
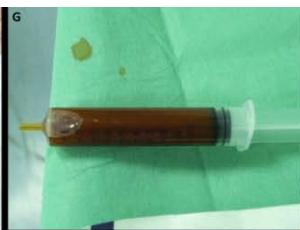
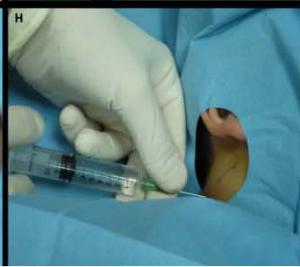
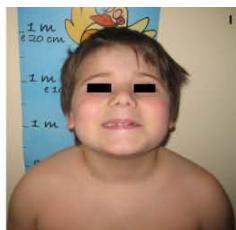
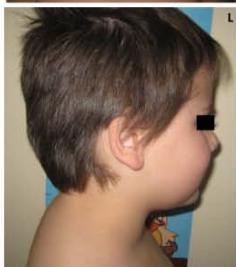
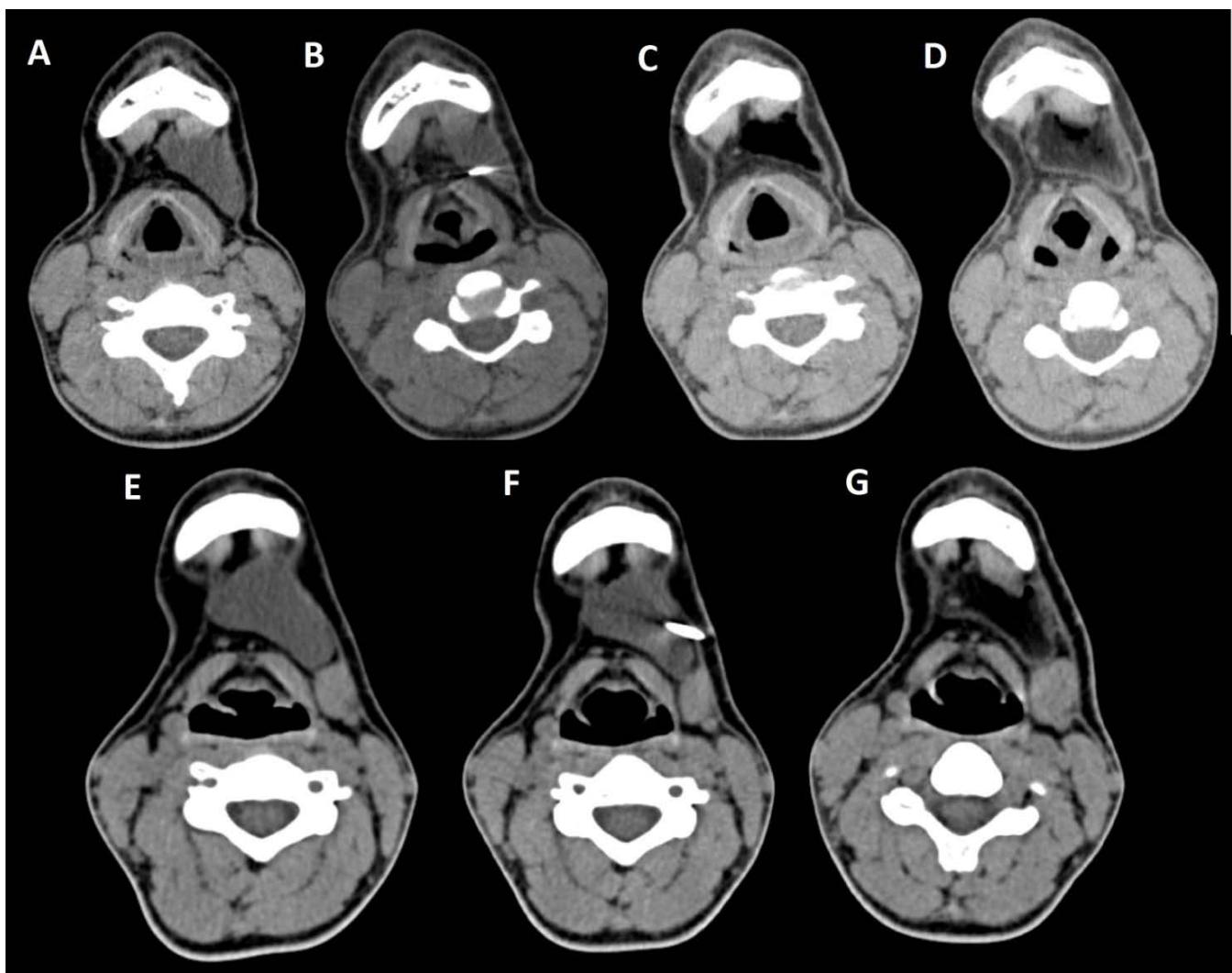
Figure/Patient 5
A 6 year-old boy presenting with a right latero-cervical tumefaction (A-B): axial (C) and coronal (D) MR scans show an expansive subcutaneous formation with high T2-signal, consistent with lymphangioma. The patient is demanded for sclerotherapy with ethanol: after disinfection and sterilisation of the skin (E), a fine needle is introduced into the lesion (F) and a yellowish viscous fluid is aspirated (G); then, alcohol is injected into the lesion (H). No recurrence is visible at two months-follow up (I-L).
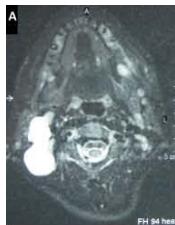
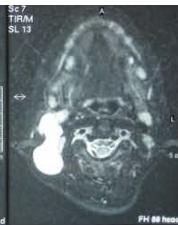
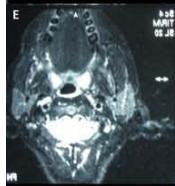
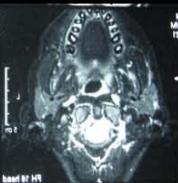
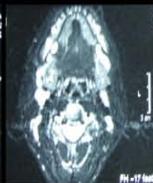
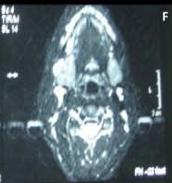
Figure/Patient 6
Another case of a right latero-cervical lymphangioma in a 8 year-old boy, markedly hyper-intense on T2 axial (A) and coronal scans (B) and slightly hyper-intense on T1 axial (C) and coronal (D) MR scans. The patient is demanded for sclerotherapy with ethanol: no recurrence is visible at the follow-up MR T2 (E-F) and T1 scans (G-H).
Table 1: Data from literature about OK-432
<table><tr><td></td><td>Advantages</td><td>Success rate</td><td>Recurrence rate</td><td>Complications</td><td>Dose</td></tr><tr><td>Ogita S. et al (1987) [13]</td><td>Low toxicity, immuno-stimulating effect</td><td>89% (total: 9 patients)</td><td>11%</td><td>Local inflammation, fever</td><td>As much as aspired by mixing each 0.1 mg with 10 cc distilled water, not exceeding 0.2 mg on total; next injection at 3-4 week</td></tr><tr><td>Luzzatto G. et al (2000) [17]</td><td>effective for primitive, residual and recurrent lesions, low invasivity and scarring</td><td>Excellent results in macrocystic, good and poor in 25% and 75% of mixed type (total: 11 and patients respectively</td><td>9.1%</td><td>Fever, signs of local inflammation, cervical swelling needing a feeding tube</td><td>0.1-0.2 mg/dose (one to 16, average 3)</td></tr><tr><td>Ruiz E. et al (2004) [23]</td><td>significant volume reduction also in microcystic lesions, feasible when there is risk of airway obstruction</td><td>complete remission in 63% (prevalently macrocystic), partial response (50 to 70%) in 27% (prevalently mixed); (total: 19 patients)</td><td>None</td><td>Fever, erythema, oedema, infection</td><td>(see Ogita) One to eight administrations</td></tr><tr><td>Sichel et al (2004) [18]</td><td>No perilesional fibrosis</td><td>complete or sub complete resolution in 73%, no response in 27% (total: 11 patients)</td><td>9.1%</td><td>Local inflammation, fever, odynophagia</td><td>0.01 mg/mL of the lesion's fluid, with a maximum of 0.2 mg in the first injection and 0.3 mg in the second or third injections</td></tr><tr><td>Yoo et al (2009) [20]</td><td>only minor complications, high long-term efficacy</td><td>Short term (83.5%) and long-term (76.3%), varying from macrocystic (92%) to microcystic (62%), from unilocular (94%) to multilocular (64%), from below (77.8%) to above mylohyoid muscle (21.7%) or mixed (20.3%); (total: 55 patients)</td><td>42% after first administration, lower in the next</td><td>Neck discomfort, swallowing difficulty, disfigurement of face, dyspnoea</td><td>(see Ogita) One to five administrations</td></tr><tr><td>Rebuffini E. et al (2012) [21]</td><td>No perilesional fibrosis, good response to additional treatments</td><td>Complete remission in 53.33%, marked reduction in 33.33%, moderate response in 13.33% (total: 15 patients)</td><td>12.5%</td><td>pain, redness, induration, fever, anaemia, transitory increase of platelets' concentration</td><td>0.2 mg per session (one to three), each separated by 6 months</td></tr><tr><td>Efe N. et al (2016) [15]</td><td>Minimal extravasation and damage of nearby tissues, does not preclude further surgery</td><td>66% for microcystic lesions, almost 100% for macrocystic lesions</td><td>12%</td><td>Local pain, heat, oedema, anaemia and rare penicillin allergies</td><td>(see Ogita)</td></tr><tr><td>Hazim et al (2016) [16]</td><td>Minimally invasive, little scar, does not interfere with subsequent surgery</td><td>Complete remission in 16.66%, marked reduction in 50%, mild reduction in 16.66% and no response in 16.66% (total: 6 patients)</td><td>None within responding lesions</td><td>Fever</td><td>0.01 mg/m in one to eight administrations</td></tr></table>
Table 2: Data from literature about Bleomycin
<table><tr><td></td><td>Advantages</td><td>Success rate</td><td>Recurrence Rate</td><td>Complications</td><td>Dose</td></tr><tr><td>Tanigawa et al (1987) [25]</td><td>Less recurrences when compared with surgery, feasible after surgery failure</td><td>excellent results in 53%, good results in 47% (total: 15 patients)</td><td>none</td><td>Swelling, fever, vomiting, diarrhoea, local infection, dyspnoea</td><td>0.3 to 0.5 ml, once every 4 to 6 weeks, microsphere in-oil emulsion</td></tr><tr><td>Sung et al (1995) [26]</td><td>feasible with unresectable lesions (debulking)</td><td>complete or nearly complete shrinkage 40%, marked reduction in size in 30%, partial or minimal reduction in 30% (total: 10 patients)</td><td>None within responding lesions</td><td>local swelling and inflammation, pneumonia of uncertain origin</td><td>1 mg/mL, from 3 to 7 sessions</td></tr><tr><td>Orford et al (1995) [34]</td><td>low risk of neurovascular damage, no surgical scar</td><td>excellent results in 45%, good in 36%, no response in 18% (total: 11 patients)</td><td>9%</td><td>fever, vomiting, cellulitis, skin discoloration</td><td>0.3 to 0.6 mg/kg</td></tr><tr><td>Zulfiqar et al (1999) [28]</td><td>Safe and effective, especially in macrocystic lesions</td><td>excellent results in 36%, good in 45%, no response in 18% (total: 11 patients)</td><td>None in short-term</td><td>none</td><td>0.5 mg/kg from 1 to 4 sessions</td></tr><tr><td>Mathur et al (2005) [35]</td><td>no major complications, no mortality</td><td>complete or near complete response in 30%, good response in 70% (total: 10 patients)</td><td>30% (70% residual disease)</td><td>local swelling and inflammation</td><td>1 mg/kg with a maximum of 6 mg/kg, every 2 weeks, in a solution of 1 mg/ml in large lesions and 2 mg/ml in smaller lesions.</td></tr><tr><td>Rozman et at (2010) [22]</td><td>Safe and effective, no major complication when using normal doses</td><td>Complete resolution in 63%, good response in 21% and poor response in 16% (total: 22 patients)</td><td>8%</td><td>Swelling, stridor, difficulty in breathing or swallowing, pain, tenderness, erythema</td><td>0.5 mg/kg with a maximum of 5 mg/kg, from one to six sessions</td></tr><tr><td>Sandlas et al (2011) [29]</td><td>Safe and effective with unilocular lymphatic malformations</td><td>significant response in 53.33%, good response in 33.33% poor response in 13.33% (total: 15 patients)</td><td>None within responding lesions</td><td>fever, swelling, skin discoloration</td><td>0.6 to 0.8 mg/kg</td></tr><tr><td>Kumar et al (2012) [30]</td><td>Safe and effective</td><td>excellent response in 20%, good in 74.29%, poor in 5.71% (total: 35 patients)</td><td>None within responding lesions</td><td>fever, transient increase in size of swelling, local infection, intraluminal bleed, skin discoloration</td><td>0.5 mg/kg body weight, no more than 10 units at a time</td></tr><tr><td>Jain et al (2013) [31]</td><td>Safe and effective with macrocystic</td><td>Complete response in 100% (total: 8 patients)</td><td>None in short-term</td><td>Fever, erythema</td><td>0.6 mg/kg, from one to three sessions</td></tr><tr><td>Porwal et al (2018) [32]</td><td>Safe and effective with macrocystic lesions, no major complications</td><td>Complete resolution in 62.5%, good response in 25%, poor response in 12.5% (total: 8 patients)</td><td>None within responding lesions</td><td>Fever, erythema, pain, restricted movement of the neck, abscess</td><td>0.5 mg/kg, from one to three sessions</td></tr><tr><td>Hashmi S et at (2020) [33]</td><td>Safe and effective, especially with macrocystic lesions</td><td>Excellent results in 20%, good results in 72.5%, poor results in 7.5% (total: 31 patients)</td><td>None within responding lesions</td><td>Post-procedural swelling, fever, local tenderness, skin changes</td><td>0.3 IU/kg, from one to three sessions</td></tr></table>

Table 3: Data from literature about doxyclicline
<table><tr><td></td><td>Advantages</td><td>Success rate</td><td>Recurrence Rate</td><td>Complications</td><td>Dose</td></tr><tr><td>Cordes et al (2007) [37]</td><td>readily available, inexpensive</td><td>Resolved or marked reduction in all patients (total: 12 patients)</td><td>None within responding lesions</td><td>Swelling, haemorrhage into the cystic cavity</td><td>10 mg/mL</td></tr><tr><td>Nehra et al (2008) [38]</td><td>readily available, inexpensive</td><td>Complete response in macrocystic lesions, partial response in mixed lesions (total: 11 patients)</td><td>18%</td><td>None</td><td>10-mg/mL up to a maximum of 200 mg</td></tr><tr><td>Burrows et al (2008) [11]</td><td>Effective, safe, few complications</td><td>Excellent response in macrocystic (90%), good in combined (80%) and in microcystic (60%) lesions (total: 41 patients)</td><td>None within responding lesions</td><td>pain and swelling, skin blisters, hair loss, Horner's syndrome</td><td>10 mg/mg, with a maximum dose of 1000 mg or 20 mg/kg, from 1 to 6 administrations</td></tr><tr><td>Shiels et al (2009) [40]</td><td>Feasible for primary lesions (even microcystic) or post-surgical recurrences</td><td>Excellent response in 90% (total: 17 patients)</td><td>13%</td><td>Cellulitis, haemorrhage into cysts</td><td>20 mg/mL, one to four sessions</td></tr><tr><td>Cahill et al (2011) [39]</td><td>Feasible for large macrocystic head and neck lymphatic malformations</td><td>Excellent response in 65%, good in 23%, poor in 12% (total: 17 patients)</td><td>None within responding lesions</td><td>emolytic anaemia in 2 infants, hypoglycaemic and metabolic acidosis, transient hypotension, skin excoriation, delayed neural complications, Horner's syndrome, transient left lip weakness, right facial nerve palsy and left hemidiaphragm paralysis</td><td>10 mg/mL with a dose range of 50 to 500 mg</td></tr><tr><td>Shergill et al (2012) [41]</td><td>Safe and effective with all types of lesions</td><td>Excellent or satisfactory response in 90% (total: 14 patients)</td><td>None within responding lesions at short-term</td><td>respiratory distress, pain, swelling, cosmetic deformity, leakage from the skin, Horner's syndrome</td><td>10mg/mL with a maximum of 300 mg in patients under 12 and 1200 mg in patients over 12</td></tr><tr><td>Cheng (2015) [2]</td><td>inexpensive, widely available, minimal side effects</td><td>84.2% with 60.5% after the first session (total: 38 patients)</td><td>Not available (patients lost to follow-up)</td><td>Tooth discoloration, allergic reactions, haemorrhage into cysts, cellulitis, pain, increased swelling, scarring, skin excoriation, Horner's syndrome</td><td>10mg/mL with a maximum of 20mg/mL</td></tr><tr><td>Farnoosh et al (2015) [36]</td><td>Safe and effective, especially with macrocystic and mixed lesions</td><td>complete resolution in 50%, satisfactory improvement in 25%, poor response in 25% (total: 29 patients)</td><td>None within responding lesions</td><td>infection, exacerbation of the lesion, jaw pain, facial swelling</td><td>10 mg/ml, ranging from 20 to 150</td></tr></table>
Table 4: Data from literature about other sclerosing agents
<table><tr><td></td><td>Advantages</td><td>Success rate</td><td>Recurrence rate</td><td>Complications</td><td>Dose</td></tr><tr><td>Pingyangmicin (Jia et al, 2014) [12]</td><td>feasible for orbital or periorbital malformations</td><td>marked improvement in 76%, moderate in 18% (total: 33 patients)</td><td>none within responding patients</td><td>swelling of the conjunctiva, localized subcutaneous atrophy</td><td>1 mL/cm2, be lower than 8 mg per single injection and 40 mg in total</td></tr><tr><td>Pingyangmicin (Gao et al, 2002) [14]</td><td>could be the primary therapy for oral, maxillofacial and cervical lesions</td><td>curative in 100% of cystic, 46.36% of capillary, 16.16% of cavernous and 19.05% of mixed capillary and cavernous lesions (total: 195 patients)</td><td>none within responding patients</td><td>mild and local</td><td>5 mg to 70 mg and 5 to 58 times, 1 time per 2-4 weeks</td></tr><tr><td>Alcohol 98% (Impellizzeri et al, 2010) [1]</td><td>large availability and low cost</td><td>100% (total: 8 patients)</td><td>none at 2 years</td><td>erythema and tenderness (if high doses are employed: hypotension, respiratory depression, arrhythmias, seizures, hypoglycaemia, exitus)</td><td>5-15 mL of solution, each but one needing one session only</td></tr><tr><td>Sodium tetradecyl-sulphate (Farnoosh et al, 2015) [36]</td><td>feasible for orbital lesions</td><td>complete resolution in 60%, satisfactory to poor response in 40% (total: 29 patients)</td><td>none within responding patients</td><td>tongue swelling, bleeding, difficulty chewing, oedema, mild allergic reaction, chronic facial pain, infections, cutaneous necrosis, nerve injury</td><td>3 to 6 mL</td></tr><tr><td>Acetic Acid (Won et al, 2004) [43]</td><td>Effective, no serious complications (if no systemic absorption occurs)</td><td>Complete resolution in 60%, good resolution in 40% (total: 5 patients)</td><td>none</td><td>pain</td><td>2 to 5 mL</td></tr><tr><td>Ethibloc (Emran et al, 2006) [44]</td><td>Safe, effective and biodegradable</td><td>satisfactory to excellent results in 84% of macrocystic/mixed and in 77% of microcystic lesions (total: 35 patients)</td><td>none within responding patients</td><td>scars, salivary fistulas, infections, aesthetic sequelae</td><td>1 to 7,5 mL</td></tr><tr><td>Tissucol (Castanon et al, 1999) [45]</td><td>Excellent long-term results and few complications</td><td>Remission in 89% (total: 9 patients)</td><td>11%</td><td>none</td><td>10% to 15% of the suctioned volume</td></tr><tr><td>Polidocanol (Jain et al, 2002) [46]</td><td>Safe, painless, effective</td><td>Remission in 100% (total: 3 patients)</td><td>none</td><td>superficial erythema, induration of the skin</td><td>1 to 6 mL, in 1 to 20 injections</td></tr></table>
Generating HTML Viewer...
References
47 Cites in Article
Pietro Impellizzeri,Carmelo Romeo,Francesca Borruto,Francesca Granata,Gianfranco Scalfari,Francesco Saverio De Ponte,Marcello Longo (2010). Sclerotherapy for cervical cystic lymphatic malformations in children. Our experience with computed tomography-guided 98% sterile ethanol insertion and a review of the literature.
Jeffrey Cheng (2015). Doxycycline sclerotherapy in children with head and neck lymphatic malformations.
Priyashirish Joshi,Bhagyalaxmi Hongal,Appasaheb Sanadi (2015). Cystic lymphangioma: A differential diagnosis.
L Monsignore,G Nakiri,D Santos,T Abud,D Abud (2010). Imaging findings and therapeutic alternatives for peripheral vascular malformations.
Ravinder Verma,Ravneet Verma,Rohan Verma,N Sardana (2014). Isolated Lymphangiomatous Polyp Nasopharynx in an Adult First Case Report in English Literature.
Michelle Manzini,Cláudia Schweiger,Denise Manica,Gabriel Kuhl (2020). Response to OK-432 sclerotherapy in the treatment of cervical lymphangioma with submucosal extension to the airway.
H Olímpio,J Bustirff-Silva,Oliveira Filho,A Araujo,K (2014). Cross sectional study comparing different therapeutic modalities for cystic lymphangiomas in children.
V Baraldini,P Dalmonte,El Hachem,M (2014). Linee guida anomalie vascolari.
Samir Nammour,Alain Vanheusden,Amaury Namour,Toni Zeinoun (2016). Evaluation of a New Method for the Treatment of Invasive, Diffuse, and Unexcisable Lymphangiomas of the Oral Cavity with Defocus CO <sub>2</sub> Laser Beam: A 20-Year Follow-Up.
S Wiegand,B Eivazi,A Zimmermann,A Sesterhenn,J Werner (2011). Sclerotherapy of lymphangiomas of the head and neck. Sclerotherapy of Lymphangiomas.
S Lam,Hkl Yuen (2019). Medical and sclerosing agents in the treatment of orbital lymphatic malformations: what's new?.
S Ogita,T Tsuto,K Tokiwa,T Takahashi (1987). Treatment of Cystic Hygroma in Children with Special Reference to OK-432 Therapy.
Q Gao,C Wang,Y Wen (2002). A study of effects of pingyangmycin injection on treatment of lymphangiomas in oral, maxillofacial and cervical regions.
Nihal Efe,Enver Altas,Muhammet Mazlumoglu,Bulent Aktan,Harun Ucuncu,Suat Eren,Fatih Oner (2016). Excellent Result With the Use of Single-Dose OK-432 in Cervical Macrocystic Lymphangioma.
M Hazim,S Moses,I Tang (2016). Intralesional Injection of OK-432 in Cystic Hygroma.
C Luzzatto,P Midrio,Z Tchaprassian,M Guglielmi (2000). Sclerosing treatment of lymphangiomas with OK-432.
S Knipping,V Bau (2011). Lymphatische Malformationen im Kopf-Hals-Bereich.
Jae Yoo,Youngjin Ahn,Yune Lim,J Hun Hah,Tack‐kyun Kwon,Myung‐whun Sung,Kwang Kim (2009). OK‐432 Sclerotherapy in Head and Neck Lymphangiomas: Long‐term follow‐up Result.
E Rebuffini,L Zuccarino,E Grecchi,F Carinci,V Merulla (2012). Picibanil (OK-432) in the treatment of head and neck lymphangiomas in children.
Dr. Fiaz (2011). Effectiveness of Intralesional Bleomycin Sclerotherapy for Lymphangioma in Children.
E Ruiz,E Valera,F Veríssimo,L Tone (2004). OK-432 therapy for lymphangioma in children.
G Zhou,J Zheng,H Mai (2011). Treatment guidelines of lymphatic malformations of the head and neck.
Nobuhiko Tanigawa,Takumi Shimomatsuya,Kohji Takahashi,Yukihiro Inomata,Koichi Tanaka,Kisaku Satomura,Yorinori Hikasa,Mitsuru Hashida,Shozo Muranishi,Hitoshi Sezaki (1987). Treatment of cystic hygroma and lymphangioma with the use of bleomycin fat emulsion.
Myung-Whun Sung,Sun Chang,Joong Choi,Jin Kim (1995). Bleomycin sclerotherapy in patients with congenital lymphatic malformation in the head and neck.
Afra Karavelioğlu,Çağrı Temuçin,F Tanyel,Arbay Ciftci,Mehmet Şenocak,İbrahim Karnak (2010). Sclerotherapy with bleomycin does not adversely affect facial nerve function in children with cervicofacial cystic lymphatic malformation.
M Zulfiqar,A Zaleha,Z Zakaria,T Amin (1999). The Treatment of Neck Lymphangioma with Intralesional injection of Bleomycin.
Gursev Sandlas,Paras Kothari,Parag Karkera,Abhaya Gupta (2011). Bleomycin: A worthy alternative.
V Kumar,P Kumar,A Pandey,D Gupta,R Shukla,S Sharma,A Gangopadhyay (2012). Intralesional Bleomycin in Lymphangioma: An Effective and Safe Non-Operative Modality of Treatment.
V Jain,A Mohta,M Sengar,K Bansal,K Val (2013). Use of intralesional bleomycin as primary therapy in macrocystic lymphangiomas.
Prateek Porwal,K Dubey,Amit Morey,Harshita Singh,S Pooja,Alokendu Bose (2018). Bleomycin Sclerotherapy in Lymphangiomas of Head and Neck: Prospective Study of 8 Cases.
Dr. Fiaz (2020). Effectiveness of Intralesional Bleomycin Sclerotherapy for Lymphangioma in Children.
Neeraj Mathur,Indu Rana,Rajendra Bothra,Rajiv Dhawan,Geeta Kathuria,Tapaswini Pradhan (2005). Bleomycin sclerotherapy in congenital lymphatic and vascular malformations of head and neck.
Soroush Farnoosh,Debra Don,Jeffery Koempel,Andre Panossian,Dean Anselmo,Philip Stanley (2015). Efficacy of doxycycline and sodium tetradecyl sulfate sclerotherapy in pediatric head and neck lymphatic malformations.
Brett Cordes,F Seidel,Marcelle Sulek,Carla Giannoni,Ellen Friedman (2007). Doxycycline sclerotherapy as the primary treatment for head and neck lymphatic malformations.
Deepika Nehra,Lara Jacobson,Pat Barnes,Baird Mallory,Craig Albanese,Karl Sylvester (2008). Doxycycline sclerotherapy as primary treatment of head and neck lymphatic malformations in children.
A Cahill,E Nijs,D Ballah (2011). Percutaneous sclerotherapy in neonatal and infant head and neck lymphatic malformations: a single center experience.
William Shiels,D Kang,James Murakami,Mark Hogan,Gregory Wiet (2009). Percutaneous Treatment of Lymphatic Malformations.
Arvind Shergill,Philip John,Joao Amaral (2012). Doxycycline sclerotherapy in children with lymphatic malformations: outcomes, complications and clinical efficacy.
Stefan Puig,Hussein Aref,Francis Brunelle (2003). Double-Needle Sclerotherapy of Lymphangiomas and Venous Angiomas in Children: A Simple Technique to Prevent Complications.
Je Hwan Won,Moon Byung,Chul-Ho Kim,Sang Kim,Man Park,Kim (2004). Percutaneous Sclerotherapy of Lymphangiomas with Acetic Acid.
Mohammad Emran,Josée Dubois,Louise Laberge,Ayman Al-Jazaeri,Andreana Bütter,Salam Yazbeck (2006). Alcoholic solution of zein (Ethibloc) sclerotherapy for treatment of lymphangiomas in children.
M Castanon,J Margarit,R Carrasco,M Vancells,A Albert,L Morales (1999). Long-Term Follow-Up of Nineteen Cystic Lymphangiomas Treated With Fibrin Sealant.
Rajeev Jain,Suman Bandhu,Sukhpal Sawhney,Ravi Mittal (2002). Sonographically guided percutaneous sclerosis using 1% polidocanol in the treatment of vascular malformations.
Ilaria Amodeo,Mariarosa Colnaghi,Genny Raffaeli,Giacomo Cavallaro,Fabrizio Ciralli,Silvana Gangi,Ernesto Leva,Lorenzo Pignataro,Irene Borzani,Lorenza Pugni,Fabio Mosca (2017). The use of sirolimus in the treatment of giant cystic lymphangioma.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Pietro Pitrone. 2026. \u201cCervical Lymphangiomas in Children: Non-Surgical Treatment with Focus on Sclerotherapy. Literature Review\u201d. Global Journal of Medical Research - D: Radiology, Diagnostic GJMR-D Volume 22 (GJMR Volume 22 Issue D1).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Subject: Global Journal of Medical Research - D: Radiology, Diagnostic
Authors:
Pietro Pitrone, Donatella Di Fabrizio, Antonino Cattafi, Alessandra Coglitore, Maria Ludovica Carerj, Italo Giuseppe Bellone, Enrico Monsù, Giuseppe Nirta, Dora Bonanno, Carmelo Romeo, Pietro Impellizzeri (PhD/Dr. count: 0)
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.