The classification of elements serves as a rich source of knowledge in chemistry. This article explores the atomic model based on wave mechanics, represented by a graph characterizing the atom, to elucidate both periodic and ordered classifications of elements. Three types of classifications emerge from this graph: periodic, ordered, and hybrid classifications, with the latter resembling periodic tables but incorporating elements of the ordered form. The periodic table stands out as one of the most profound and unifying concepts in modern science. New illustration methods, such as condensed tables with orders and periods, are introduced. The results underscore the conclusiveness of the findings, revealing that classifications extend beyond periodicity, encompassing ordered types of tables as well. This research sheds light on the diverse approaches to classifying elements and opens avenues for further exploration in the field of chemistry.
## I. INTRODUCTION
In any chemistry class or laboratory, there is almost always a periodic table hanging on the wall. This table, which includes all the known elements, provides a lot of information on each. As one progresses in the study of chemistry, the usefulness of the periodic table becomes more and more obvious[1].
Atoms were first suggested by the Greek philosophers Democritus and Leucippus around 400 BC, the concept was primarily based on a hunch. In fact, for several centuries thereafter, no convincing experimental evidence was available to support the existence of atoms. The first real scientific data were collected by Lavoisier and others from quantitative measurements of chemical reactions. Thus around 1790 the scientist was already able to identify 33 chemical elements. The results of these stoichiometric experiments allowed John Dalton to propose the first systematic atomic theory. This one, although crude, has stood the test of time extremely well.Once atoms were admitted, logically a number of questions arose such as: What is the nature of an atom? How is it composed? What are the constituent parts, their properties etc.?
The most striking phenomenon in chemistry was very early the discovery of an analogy and the periodic repetition of the properties of the elements with each other. Several groups of elements with great similarities in their chemical behavior were identified. This is how the need to classify the elements arose. During the first 30 years of the 20th century, the appearance of a new theory, quantum mechanics, made it possible to explain the behavior of atoms under the effect of light. This so-called modern theory made it possible to develop the electronic structure of atoms. This confirmed the periodic behavior of chemical elements in terms of electronic configuration and then the properties of atoms.
In 1817 Döbereiner succeeded in relating the atomic mass of certain elements to their properties. He noticed the existence of similarities between elements grouped in threes which he called "triads". He highlighted the fact that the mass of one of the three elements of the triad was the intermediate (the average) of the other two. In 1850, we could count more than twenty triads to arrive at a first coherent classification.
In 1862 Chancourtois, a French geologist, highlighted a certain periodicity between the properties of the elements of the table. Later the English chemist Newlands announced that "the eighth element which follows a given element resembles the first as the eighth note of the octave resembles the first". But this law could not apply to elements beyond calcium: "The notions of transition elements were unknown". This classification therefore remained insufficient, but the periodic table was beginning to take shape.
In 1869 and 1870 respectively, Dmitri Mendeleev and Lothar Meyer claimed that the properties of elements could be represented as periodic functions of their atomic weight and presented their ideas in the form of a periodic table. As new elements have been discovered, the original form of the periodic table has been significantly modified and it is now recognized that periodicity is a consequence of variation in ground state electronic configurations. A modern periodic table emphasizes blocks of 2, 6, 10, and 14 elements that result from filling the s, p, d, and f atomic orbitals, respectively. An exception is He, which for chemical reasons is placed in the rare group [2].
The most famous of the periodic classifications was that of MENDELEIEV in 1869. It is to him that the merit goes to having presented the first, a well-structured and coherent classification of all the elements known at his time. He then realized the periodic trend in the properties of these placed in increasing order of their atomic masses [3]. This table had 6 columns with short periods with empty boxes according to his manuscript. This intuition of the scientist was confirmed by the later discovery of these new tenants corresponding to his prediction. More than a century after the death of the main founder of the periodic system, different types of periodic tables have emerged. It is time to revisit the origins, the precursors and even the status of this classification which has had a profound influence on the development of chemistry and modern physics. Scientists today are still debating the best possible presentation of the periodic table [4].
The American chemist Glenn T. Seaborg, Lawrence Berkeley National Laboratory, Berkely, California, United States of America whose only address is composed of five chemical elements (Sg, Lr, Bk, Cf, and Am respectively seaborgium, lawrencium, berkelium, californium and americium) had the merit of completing MENDELEF's periodic table of elements with the addition of Actinides in 1945 [5]. From Lavoisier in 1790 to Glenn T. Seaborg, nearly two centuries of scientific exchange and research have led to several types of classifications, all periodic. It only differs in the mode of presentation.
In 1990 FERNANDO Dufour proposed a three-dimensional classification which he called "element tree" or "tree of elements" also called "periodic tree".
In 1995 PIERRE Demers of the University of Montreal proposed a classification system called "Québécium", a pyramid with 4 faces and 4 levels; the upper level made up only of 2 elements of the "s" block (1st period); the next is composed of the elements of the "s and p" blocks including 4 of "s" and 12 of "p" (the 2nd and 3rd periods), the penultimate level includes the elements of the "s, p and d" blocks $\gg$ including respectively 4, 12 and 20 elements (periods 4 and 5) and the last level the base containing the elements of the four blocks "s, p, d and f" respectively 4, 12, 20 and 28 components of periods 6 and 7. The atomic number values Z provide an undeniable if not sufficient ordering principle. A classification called modern, somewhat analogous to the Quebec system but more explicit, is also presented as a pyramid of 7 levels, each corresponding to a period. From the top "the 1st period" to the base "the 7th period" of the pyramid, lines evolve from top to bottom materializing the identity of the valence electrons, that is to say the families (columns) of the periodic table of elements [6].
Eric SCERRI, for the sake of greater regularity, proposes a table in which the hydrogen and helium of the 1st period are brought back to the 2nd period respectively at the level of halogens and rare gases with the columns $\mathsf{np}^5$ and $\mathsf{np}^6$ of block p cut and moved to the right, this leads to a disrupted periodic table of elements, with a regularity of the type 8 8 18 18 32 32 2 as opposed to 2 8 8 18 18 32 32 [7].
All these classifications present a certain discontinuity from one period to the next according to certain researchers, this seems to mark a disagreement with the logical sequence of the usual ordination of natural numbers in which the atomic numbers $Z$ of the atoms would be found (Z ranging from 1.........118).
This reason indicated above (mentioned) aroused concern and caught the attention of other researchers and served as a reference for them to promote new types of classification. Thus were born the spiral classifications. Professors PHILIP Steaward and JAN Scholten each proposed a spiral classification model, periodic spiral systems or "periodic tables" which according to them respect the natural ordination of atomic numbers. Théodor BENFEY's snail system from the 1960s and others are also of the spiral model. It should be noted that the difference between these presentation models is the simple notion of parallel and spiral linear chains. Periodicity is the only criterion for all these classifications. The period is an instantaneous phenomenon, it is the time that a phenomenon takes to complete the phases of its duration, it is the time interval at the end of which a phenomenon reproduces under the same conditions, such as the properties of the elements chemical; In chemistry periodicity is examined in terms of the number of elements with which identical properties recur from one group to another. It is this phenomenon that the English chemist Newlands wanted to explain with his famous quote: "the eighth element which follows a given element resembles the first as the eighth note of the octave resembles the first".
In 1989 Ouahès, R. made a proposal for a new periodic table of elements. This other table is still called periodic of 8 periods: He explains his table in these terms: "This means, in terms of atomic structure, that each period corresponds to the filling of shells and subshells according to $n + l = k$ ( $k$: constant) where $n$ and $l$ are the principal and azimuthal quantum numbers. The constant $k$ is the rank of the period. The results are given in table 1. The number of periods is 8 instead of 7."
This article will answer questions such as:
- Can we explain the periodic classification using a more practical vision than that, entirely empirical, of Mendeleev?
- How does the distribution of electrons in an atom help explain periodic properties?
- Is this other table periodic?
- Is the number of periods greater than 7?
In general, the electronic structure gives all the information about an element, mainly its coordinates in the different tables. Its physical and chemical properties also depend on it. Valence electrons provide the main concepts of chemical language. They open the way to forms of classification of elements, hence the different methods of illustrating the classification [8].Obtaining specific graphs from the characteristic graph of the atom was explained in the methodology section. The types of classification of chemical elements were illustrated and discussed and a conclusion was drawn.
## II. METHODOLOGY
The specific graphs describing the orders and periods concretely explain the principles of stability,
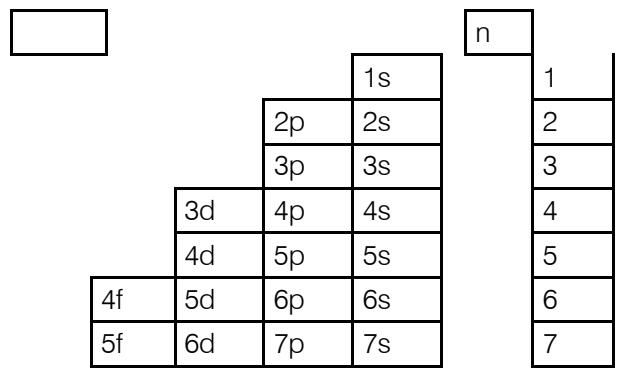
Electronic structure according to order (y-axis) aufbau, and in general the Kletchkovsky rule [9]. The distribution of the electrons of the different atoms along the axes of these graphs (abscissa and ordinate) by the periods and the orders respectively make it possible to explain each classification model in the literature and even to propose new methods of illustrating the classifications of the elements. A new criterion for classification of chemical elements becomes the order of the energy level " $\mathrm{O_E}$ ", absolutely different from the period " $n$ ", has even emerged, it leads to a new classification called the ordered classification of the elements [10].
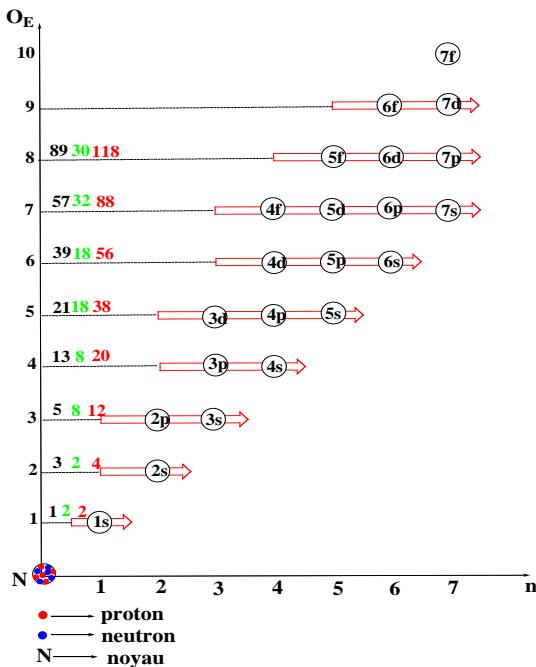 **Graphene 1:** Electronic structure of elements according to order
- Electronic structure along the period axis
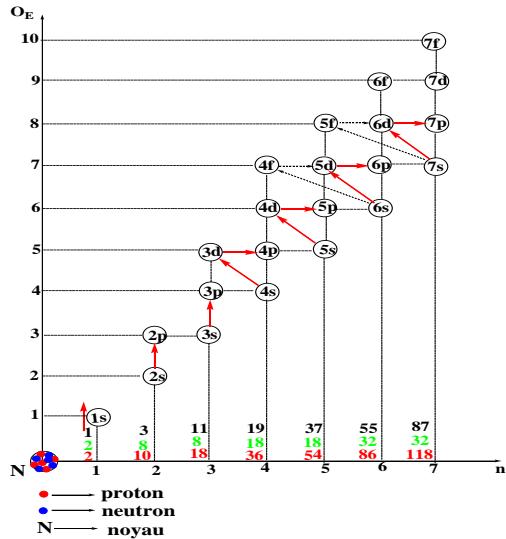 Graphe 2: Electronic structure of elements according to period
The ordered tables are obtained according to graph 1. The classification is made according to the ordinate axis " $O_E$ " which becomes the new classification criterion leading to tables of a new type called ordered tables, the set of ordered tables is summarized by a so-called condensed order table. This goes without saying for all periodic classifications, the only classic classification criterion of which is the period "n" according to graph 2 and are all also summarized by a condensed period table. These two condensed tables are analogous but different, the energy order $\mathrm{O_E}$ is different from the period "n". See below the respective condensed forms which are effectively new methods of illustrating the classifications.
Table 1: Condensed table to order
<table><tr><td>Order</td><td>1</td><td>2</td><td>3</td><td>4</td><td>5</td><td>6</td><td>7</td><td>8</td></tr><tr><td>Underlays</td><td>1s</td><td>2s</td><td>2p3s</td><td>3p4s</td><td>3d4p5s</td><td>4d5p6s</td><td>4f5d6p7s</td><td>5f6d7p...</td></tr><tr><td>Z</td><td>1- 2</td><td>3- 4</td><td>5- 12</td><td>13- 20</td><td>21- 38</td><td>39- 56</td><td>57- 88</td><td>89-118...</td></tr><tr><td>Type of tables</td><td>2</td><td>2</td><td>8</td><td>8</td><td>18</td><td>18</td><td>32</td><td>30......</td></tr></table>
Table 2: Condensed period table
<table><tr><td>Periods</td><td>1</td><td>2</td><td>3</td><td>4</td><td>5</td><td>6</td><td>7</td></tr><tr><td>Underlays</td><td>1s</td><td>2s2p</td><td>3s3p</td><td>4s3d4p</td><td>5s4d5p</td><td>6s4f5d6p</td><td>7s5f6d7p...</td></tr><tr><td>Z</td><td>1- 2</td><td>3- 10</td><td>11- 18</td><td>19- 36</td><td>37- 54</td><td>55- 86</td><td>87-118...</td></tr><tr><td>Type of tables</td><td>2</td><td>8</td><td>8</td><td>18</td><td>18</td><td>32</td><td>32...</td></tr></table>
## III. RESULTS
To make the classification, whether periodic or ordered, it is enough to arrange vertically the stacks of the different sub-layers in each case. Give each block the number of columns corresponding to its electronic capacity: 2 for block "s", 6 for "p", 10 for "d" and 14 for "f". The classifications are either short, medium and long depending on the stacks chosen as a basis. The main stacks are "sp/ps" for tables with 8 columns "sdp/dps" for those with 18 columns and finally "sfdp/fdps" for long classifications with 32 columns.
The vertical arrangements of the sublayers by stacking the periods and/or order offer the appearance of tables: the periodic tables are fairly well-known emblematic figures which adorn quite a few scientific circles today. Ordered tables constitute a completely new classification model and appear in the form of stairs with regular steps. The longest ordered arrays are made up of four steps corresponding to the four blocks of elements. These stairs start from block "f" via those of "d, p" and end with that of "s", from bottom to top respectively.
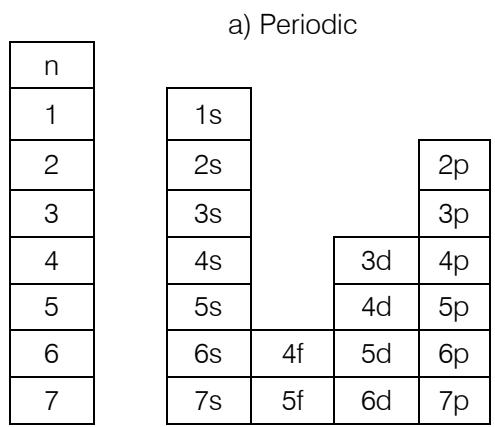
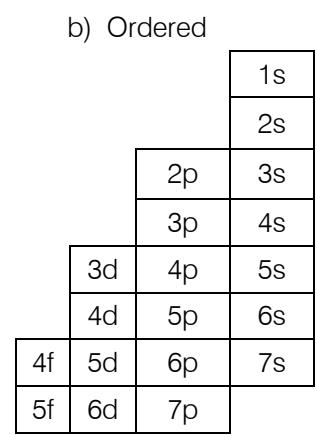

Comparing these two classifications, it appears that the order is obtained by the transfer of the "s" block from the left of Mendeleev's periodic table to its right with a shift of one level upwards; which allows you to go from 7 periods to 8 orders. Orders always end with "s" while periods begin with "s" and are composed respectively as follows: $(n - 3)f(n - 2)d(n - 1)pns$ and $ns(n - 2)f(n - 1)dnp$.
The ordered classification puts the transitional elements, blocks f and d, of group "B" before the nontransitional elements, blocks s and p, of group "A". This leads to a more regular, coherent presentation, compared to the periodic system which inserts transition elements between normal and non-normal ones. In the
periodic system the elements "d" start with IIIB progress to VIIIIB then follow groups IB and IIB marking the end of the "d" block. The ordered system puts the normal elements "s and p" of the large group "A" in the same arrangement as those of the transition. The "p" elements start with IIIA progress to VIIIIA and follow the IA and IIA of the "s" block.
In an ordered array the transitional elements are at the beginning and the non-transitional ones at the end. This can justify the reason which divides the four blocks of elements into two large groups A (normal elements) and B (transition elements). By the amplitude of the stacks or the types of tables of orders and/or periods, the methods of illustrating the classifications are summarized by series with values corresponding to the number of elements by order " $O_E$ " and/or by period « n »:
- Order series $\langle \mathrm{O}_{\mathrm{E}}\rangle$: 2 8 18 1832 30
- Series with period $\ll n\gg$: 2 8 8 18 18 32 32
## IV. DISCUSSION
Knowing that for the block "s" the order is identical to the period and for the three other blocks "p, d, f" the period is less than the order of one unit, it is possible to go from one classification ordered to a periodic system. This is possible by tilting the "s" block of the ordered system one level down to obtain a periodic system like the ordered one, thus we go from 8 orders to 7 periods corresponding to the periodic and ordered system. This hybrid classification conveys the characteristics of both types of classifications.
 a) Ordered
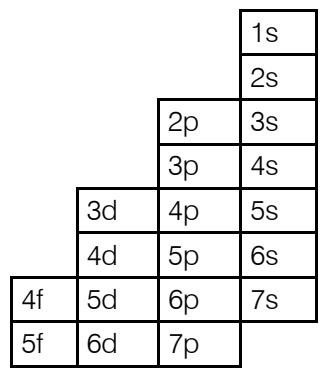
 b) Periodic like the ordered one
The transformation of the ordered system into the periodic classification in its image is a stepped but periodic system. It has all the form of the ordered table but it is also periodic. It also comes in 8, 18, and 32 columns as in the case of Mendeleev's periodic table.
This is how its 18-column classification would look like:
<table><tr><td>1</td><td>2</td><td>3</td><td>4</td><td>5</td><td>6</td><td>7</td><td>8</td><td>9</td><td>10</td><td>11</td><td>12</td><td>13</td><td>14</td><td>15</td><td>16</td><td>17</td><td>18</td></tr><tr><td colspan="10">GROUPE B</td><td colspan="8">GROUPE A</td></tr><tr><td>III</td><td>IV</td><td>V</td><td>VI</td><td>VII</td><td colspan="3">VIII</td><td>I</td><td>II</td><td>III</td><td>IV</td><td>V</td><td>VI</td><td>VII</td><td>VIII</td><td>I</td><td>II</td></tr><tr><td>3</td><td>4</td><td>5</td><td>6</td><td>7</td><td>8</td><td>9</td><td>10</td><td>11</td><td>12</td><td>13</td><td>14</td><td>15</td><td>16</td><td>17</td><td>18</td><td>1</td><td>2</td></tr><tr><td rowspan="3" colspan="10">(BlocKs "f & d)</td><td colspan="4">(BlocKs "s & p")</td><td>2</td><td>1</td><td>2</td><td></td></tr><tr><td>5</td><td>6</td><td>7</td><td>8</td><td>9</td><td>10</td><td>3</td><td>4</td></tr><tr><td>13</td><td>14</td><td>15</td><td>16</td><td>17</td><td>18</td><td>11</td><td>12</td></tr><tr><td>21</td><td>22</td><td>23</td><td>24</td><td>25</td><td>26</td><td>27</td><td>28</td><td>29</td><td>30</td><td>31</td><td>32</td><td>33</td><td>34</td><td>35</td><td>36</td><td>19</td><td>20</td></tr><tr><td>39</td><td>40</td><td>41</td><td>42</td><td>43</td><td>44</td><td>45</td><td>46</td><td>47</td><td>48</td><td>49</td><td>50</td><td>51</td><td>52</td><td>53</td><td>54</td><td>37</td><td>38</td></tr><tr><td>57*</td><td>72</td><td>73</td><td>74</td><td>75</td><td>76</td><td>77</td><td>78</td><td>79</td><td>80</td><td>81</td><td>82</td><td>83</td><td>84</td><td>85</td><td>86</td><td>55</td><td>56</td></tr><tr><td>89*</td><td>104</td><td>105</td><td>106</td><td>107</td><td>108</td><td>109</td><td>110</td><td>111</td><td>112</td><td>113</td><td>114</td><td>115</td><td>116</td><td>117</td><td>118</td><td>87</td><td>88</td></tr></table>
\*
<table><tr><td>58</td><td>59</td><td>60</td><td>61</td><td>62</td><td>63</td><td>64</td><td>65</td><td>66</td><td>67</td><td>68</td><td>69</td><td>70</td><td>71</td></tr><tr><td>90</td><td>91</td><td>92</td><td>93</td><td>94</td><td>95</td><td>96</td><td>97</td><td>98</td><td>99</td><td>100</td><td>101</td><td>102</td><td>103</td></tr></table>
Graphs 1 and 2 depict the composition of the electron cloud elements in order and period respectively. The arrangement of the sublayers from 1s to 7p is practically done according to the increasing order of the energy level either by the ordinate " $\mathsf{O}_{\mathsf{E}}$ " or by the abscissa " $n$ " in the usual ordination of natural numbers.
The structure of an order according to the accepted values is $(n - 3)f(n - 2)d(n - 1)p$ ns and that of the period is $ns$ $(n - 2)f(n - 1)dnp$, it is remarkable that the order ends with the sublayer "ns" while it can start with any sublayer even with ns if the order is short as for the cases: $n = 1$ and 2. Practically the orders go from 1 to 8. For the periods, they start with "ns" and end with "np", the coefficients of the intermediate sublayers are counted from "n" to the limit values. The order is then different from the period, the periods go from 1 to 7. The coefficients of the sublayers in each case are linked to $O_{E}$ and/or n, knowing that for the block "s" $O_{E} = n$. Thus, it is enough to be able to count from 1 to 7 and/or from 1 to 8 to develop the rule of stability.
Table 3: Group of sublayers according to the order of energy (Graph 1)
<table><tr><td>Orders « O_E »</td><td>1</td><td>2</td><td>3</td><td>4</td><td>5</td><td>6</td><td>7</td><td>8</td></tr><tr><td>Underlays</td><td>1s</td><td>2s</td><td>2p 3s</td><td>3p 4s</td><td>3d 4p 5s</td><td>4d 5p 6s</td><td>4f 5d 6p 7s</td><td>5f 6d 7p...</td></tr></table>
Table 4: Group of sub-layers according to the period (Graph 2)
<table><tr><td>Periods « n »</td><td>1</td><td>2</td><td>3</td><td>4</td><td>5</td><td>6</td><td>7</td></tr><tr><td>Underlays</td><td>1s</td><td>2s 2p</td><td>3s 3p</td><td>4s 3d 4p</td><td>5s 4d 5p</td><td>6s 4f 5d 6p</td><td>7s 5f 6d 7p</td></tr></table>
These two tables sufficiently express the difference between an " $O_E$ " order and a " $n$ " period, but these two criteria are well linked.
Table 5: Relationship between period and order (Graphs 1 & 2)
<table><tr><td>Periods « n »</td><td>1</td><td colspan="2">2</td><td colspan="2">3</td><td colspan="2">4</td><td colspan="2">5</td><td colspan="2">6</td><td colspan="2">7</td></tr><tr><td>Underlays</td><td>1s</td><td>2s</td><td>2p</td><td>3s</td><td>3p</td><td>4s</td><td>3d4p</td><td>5s</td><td>4d5p</td><td>6s</td><td>4f5d6p</td><td>7s</td><td>5f6d7p...</td></tr><tr><td>Orders « O_E »</td><td>1</td><td>2</td><td colspan="2">3</td><td colspan="2">4</td><td colspan="2">5</td><td colspan="2">6</td><td colspan="2">7</td><td>8</td></tr></table>
These two very similar tables are different. For block "s", $O_E = n$, the order is identical to the period and for the others the orders are greater than the period by one unit. The condensed period form confirms all current periodic classifications of the elements. It is the summary of all the classic period processes developed according to the abscissa axis "n" whatever the two or three dimensional model, spiral or not.
The condensed ordered table is also another classification process leading to entirely new forms of tables which are far from being periodic. They are ordered or ordered tables of the elements, obtained along the " $O_E$ " ordinate axis. These resulting paintings are presented in the form of a staircase also with 8, 18 and 32 columns. If for the periodic system the classification criterion is the period "n", the ordered tables are carried out around a new classification criterion, namely the order " $O_E$ ". Except that they are not periodic. The word "period" or its adjective "periodic" was not accidentally used by the founding father of the classification system. This choice expressed the behavior of the properties of the groups of elements which were born, evolved then diminished and canceled after a certain number of elements which make up said period. An order is a combination of the elements of two successive periods.
## V. CONCLUSION
This new classification conforms to the electronic structures of the elements according to order and period. It can have historical and educational interest, because it is based on simple to understand concepts.
This work demonstrated that all classifications in the literature are not only periodic. The characteristic graph of the atom made it possible to confirm the existence of the two classification criteria leading to fairly classic periodic tables built around the period "n going from 1 to $7^{\prime \prime}$ and completely new ordered or ordered tables which carry out around the order of the increasing level of energy order of the sublayers $O_{E}$ starting from 1 to $8^{\prime \prime}$ following the ordinate of the graph. The ordered table corresponds to the transfer of the "s" block from the left of the periodic table of elements to its right with the tilting of the latter one level upwards from 7 stacks to 8 others.A third classification intermediate to the other two was envisaged by the reduction of the block "s" of the ordered form by one level downwards from 8 stacks to 7 others, hence a periodic classification in the image of the ordered form. A new illustration of the classification of elements.
We hope to soon provide a rigorous proof of atomistic terminology using the equation of the line, such as the deduction of valence electrons, the electronic transition and the relationship between the nucleus and the electron cloud of an atom.
Generating HTML Viewer...
References
11 Cites in Article
Steven Zumdahl (1988). traduit de l'anglais par Serge GéribaJdi, 1997Susan E. Thomas, , De Boeck Université, N.-Y. (1992), p. 92 6 chapitres, 2-7445-0030-5..
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Ousmane BARRY. 2026. \u201cCharting Atomic Characteristics: A Pathway to Element Classification\u201d. Global Journal of Science Frontier Research - B: Chemistry GJSFR-B Volume 24 (GJSFR Volume 24 Issue B1): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
The classification of elements serves as a rich source of knowledge in chemistry. This article explores the atomic model based on wave mechanics, represented by a graph characterizing the atom, to elucidate both periodic and ordered classifications of elements. Three types of classifications emerge from this graph: periodic, ordered, and hybrid classifications, with the latter resembling periodic tables but incorporating elements of the ordered form. The periodic table stands out as one of the most profound and unifying concepts in modern science. New illustration methods, such as condensed tables with orders and periods, are introduced. The results underscore the conclusiveness of the findings, revealing that classifications extend beyond periodicity, encompassing ordered types of tables as well. This research sheds light on the diverse approaches to classifying elements and opens avenues for further exploration in the field of chemistry.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.