A model describing the interaction of geological and biosphere processes is proposed. It is based on the postulate of the gravitational effect of the bodies of the solar system on the lithosphere plates’ movement through magma flows and deep breathing of the Earth. The continuous movement of the plates consist of orogenic cycles. The cycles include a short-term orogenic periods of relatively fast plates’ movement and long-term geosynclinal periods of relatively slow movement. The fast movement is caused by rifting, when magma breaks through the thin shell of the lithosphere and hardens in contact with sea water, forming a new plate. During the orogenic period, oceanic plates collide with the continental margin plate in the subduction zone. The energy of the collisions initiates thermochemical sulfate reduction, in which sedimentary organic matter (OM) is oxidized. The resulting CO2 rises to the surface of the Earth, spreads over the planet, initiating photosynthesis. In the geosynclinal period due to the slow plates’ movement the released collision energy is insufficient to initiate thermochemical sulfate reduction.
### INTRODUCTION
As early as 1926 V.l. Vernadsky (Vernadsky, 1926) developing Biosphere concept, put forward an idea on relationship of geologic and biosphere processes. This idea was put as the basis for the model of the global cycle of biosphere carbon (Ivlev, 2023). To study the processes that occurred in the past, we were guided by the principle of actualism, which claims that the laws of physics and chemistry are invariant in time. This allows applying them to the processes proceeding in the past, but taking into account the conditions that could have been at that time.
## I. A MODEL OF GLOBAL CYCLE OF BIOSPHERE CARBON AND EVOLUTION OF GLOBAL PHOTOSYNTHESIS. PART I
The carbon pathway in the global cycle of biosphere carbon To monitor the carbon pathway in global cycle of biosphere carbon consider Fig.1, in which it is depicted as a closed loop having two branches - reducing and oxidizing ones. The carbon of the natural "carbon dioxide - bicarbonate-carbonate $\left(\mathrm{CO}_{2}-\mathrm{HCO}_{3}^{-}-\mathrm{CO}_{3}^{-}\right)$ " system feeds photosynthesis with a substrate $\mathrm{CO}_{2}$.
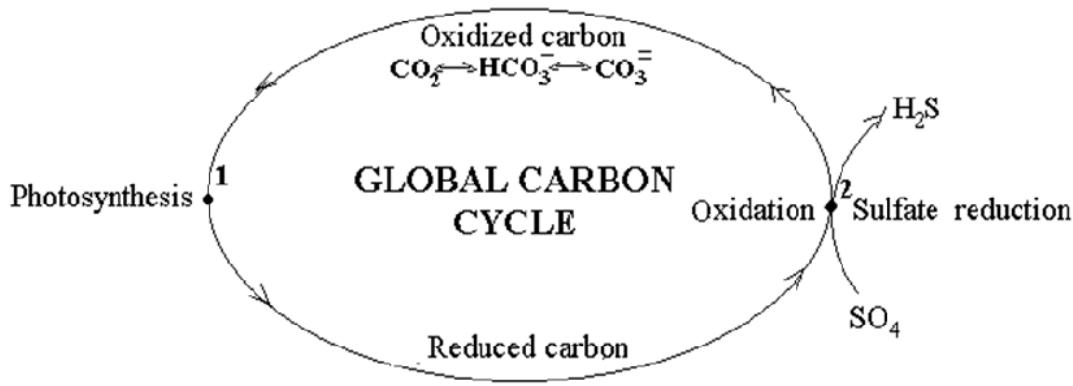 Fig. 1: Putative global carbon cycle in nature: oxidative and reductive branches. The points of carbon transition from the oxidative states to the reduced ones in photosynthesis (point 1) and back in sulfate reduction, proceeding in subduction zone (point 2).
In this universal natural process, photosynthetic biomass is produced. Considering that it is used in trophic chains for the synthesis of heterotrophic biomass, photosynthesis can be regarded as a source of carbon for "living matter" as a whole with carbon atoms with varying degrees of reduction. After mass extinction of the "living" organisms, a sedimentary OM is formed. The residual reduced carbon is preserved in the earth's crust until it is subjected to full final oxidation in the subduction zone, the most submerged part of the crust (point 2). At this point, the global carbon cycle is completed.
From the occurrence of the "living matter", part of the reduced carbon, produced during photosynthesis, is oxidized to its initial state in the processes of respiration. The other part is oxidized in the subsequent processes of the aerobic and anaerobic zones of the earth's crust.
Now it is time to explain, why the cycle was named as biosphere one. In the natural $\text{CO}_2 - \text{HCO}_3^- - \text{CO}_3^{*+}$ system, we excluded from the consideration, carbonate thicknesses since the chemical exchange of carbon atoms in solid state proceeds at an incomparably lower rate than carbon atoms in other components and physical states.
Fig.1 allows monitoring the movement of carbon atoms, but does not make it possible to understand the mechanism of the cycle's work. To study it, let's imagine a cycle in the form of a virtual machine consisting of geological and biosphere parts interacting with each other. Besides, the geological part is the leading one, since it sets the beginning of the work of the biosphere part and the sequence of biosphere events. But first, let's analyze the postulate that underlies all further reasoning.
The key postulate of the model on which subsequent proofs are based. On the probable cause of the uneven movement of lithosphere plates.
Due to the gravitational interaction of the Earth with the bodies of the near-solar system, streams of various liquids arise in the bowels of the Earth. These are flows of molten magma in the asthenosphere, hydrogen fluxes in degassing of the core, and flows of other gases and volatile components of a number of elements. The movement of fluids depends on various factors and has been poorly studied. The manifestation of motion is the continuous movement of lithosphere plates that cover the Earth, forming its solid shell. The lithosphere shell lies over the magma and it is known that the lithosphere plates' movement is uneven (Rutten, 1971; Martin, 1973; Flint, 1977).
There is a short-term phase of rapid plate movement called the orogenic period, which coincides with the tectonically active state of the earth's crust. The state is characterized by intense volcanic and magmatic activity, including mountain formation. The long-term phase of slow motion coincides with the tectonically calm state of the earth's crust. The phase of quiet movement of lithosphere plates is called the geosynclinal period. Both periods form an orogenic cycle that repeats itself in geological time. Some researchers call this sequence of processes the "pulse of the Earth" (Rutten, 1971), others - the "uneven deep breathing of the Earth" (Dobretsov et al., 2001; Letnikov, 2001; Podkletnov). If to stick to the comparison with respiration, then, according to its sense, it can be argued that at the moment of increased respiration in the thinnest part of the lithosphere shell (in the zone of mid-oceanic ridges), magma, together with its accompanying components, breaks the lithosphere shell and comes into contact with oceanic water solidifies, forming a new plate. This pushes the adjacent plates apart, forcing them to move. Thus the intensification of deep breathing of the Earth, including the process of rupture of the lithosphere shell and the subsequent formation of a new plate, is called rifting.
It is natural to assume that the time of acceleration of the lithosphere plates' movement coincides with the time of the orogenic period. At the same time during the long-term geosynclinal period plates' movement slows down as well as tectonic activity. Let's see now, how the described sequence of the processes affects the functioning of the global carbon cycle.
In the deeply submerged zone of the Earth's crust (in the subduction zone), oceanic lithosphere plates collide with the margin of the continental plate. Being smaller in size, oceanic plates sink into magma and dissolve in it. The continental plate rises at the collision site and participates in mountain building.
But the main result of lithosphere plates' collisions in the orogenic period is that the released energy of the accelerated plates collisions initiates thermochemical sulfate reduction, in which gypsum, formed from seawater, oxidizes sedimentary organic matter that has descended from the surface into the subduction zone
$$
SO_{4}^{2-} + 2(CH_{2}O) \rightarrow 2CO_{2} + 2H_{2}O + S^{2-} - Q
$$
Carbon dioxide formed during the orogenic period as a result of the oxidation of OM enters the Earth's surface, filling the atmosphere and hydrosphere of the Earth, then spreads around the planet initiating photosynthesis in different locations in accordance with the local conditions.
In geosynclinal period due to the slow plates' movement the energy, released during collisions, is no longer sufficient to initiate sulfate reduction reaction. That is why, there is no OM oxidation and the main geological processes during this period are weathering, erosion and denudation.
Thus, the main difference between the processes in orogenic and geosynclinal periods is that in orogenic periods active collisions lead to the oxidation of sedimentary OM in the subduction zone and to the initiation of photosynthesis in the biosphere, whereas in the geosynclinal period there is no oxidation of OM in the subduction zone. This makes adjustments to the dynamics of the orogenic cycles, in particular, to the dynamics of biosphere processes.
Let's consider the features of the processes in the biosphere part of the virtual machine during the orogenic period. It is believed that in the orogenic period, when the oxidation of OM occurs, the concentration of $\mathrm{CO}_{2}$ reaches its maximal values. This also determines the maximum values of the photosynthesis rate. Due to the greenhouse properties of $\mathrm{CO}_{2}$, high temperatures prevail on the Earth's surface. Based on the above, it can be concluded that high temperatures and anaerobic conditions, that arise due to the entry of igneous rocks to the surface, characterize the living conditions in the biosphere during the considered period.
During the geosynclinal period, $\mathrm{CO}_{2}$ does not flow from the subduction zone, but photosynthesis goes on the Earth. As a result, the concentration of $\mathrm{CO}_{2}$ in the environment decreases, whereas oxygen content grows. In parallel with the decrease in $\mathrm{CO}_{2}$ concentration, the surface temperature on the Earth decreases and by the end of the geosynclinal period glaciations occur. Such a change in habitat conditions leads to a biotic cycle. The predominance of thermophilic anaerobes at the beginning of the cycle (in the orogenic period) is replaced by the dominance of frost-resistant aerobes by the end of the geosynclinal period of the cycle.
With the change of orogenic cycles, the low-temperature and high-oxygen conditions of the geosynclinal period of the previous cycle abruptly change to the high-temperature anaerobic conditions of the orogenic period of the current cycle (Thunnel et al., 1984). A number of the "living" organisms adapted to other conditions die (Gorican et al., 2013) and a huge amount of biogenic material enters the sediment. It gives rise to formation of sediments rich in OM ("black shales") (Ivlev, 2019; 2023).
In this form without involving additional arguments the model allows explaining biosphere events that have been already known to geologists and paleontologists, such as alternation of periods of warming and glaciations, periodic mass extinctions of living organisms, etc. It evidences for the reasonable basis of the model. We will return later to other arguments justifying the model, but now we shall consider some key items arising from the use of the model.
Global photosynthesis is the key process operating in the biosphere part of the global carbon cycle
The main process in the biosphere part of global carbon cycle model is global photosynthesis, which takes into account the cumulative result of photosynthesis of the entire ensemble of organisms living on the Earth at the time under consideration.
When considering the process, it is important to answer two questions: 1) whether the equation, which is commonly used for description of traditional photosynthesis is applicable to describe global photosynthesis; 2) whether the properties of global photosynthesis differ from those of traditional photosynthesis.
Traditional photosynthesis is understood as a process in the simplest "organism - environment" system, which proceeds in the mode of the substrate depletion and is written as follows (Ivlev, 2020):
$$
\mathrm{C O} _ {2} + \mathrm{H} _ {2} \mathrm{O} \xrightarrow{h v} (\mathrm{C H} _ {2} \mathrm{O}) _ {\text{biomass}} + \mathrm{O} _ {2} \tag{2}
$$
It follows from the analysis of expression (2) that in traditional photosynthesis, the changes in concentration of substrate $(\mathrm{CO}_{2})$ and product $(\mathrm{O}_{2})$ of the reaction should be antiphase, while changes in the concentration reaction products $(\mathrm{CH}_{2} \mathrm{O})$ biomass and $(\mathrm{O}_{2})$ are in-phase.
It was previously shown (lvlev, 2019) that under certain assumptions global photosynthesis can be expressed by exactly the same equation as traditional one
$$
\mathrm{C O} _ {2} + \mathrm{H} _ {2} \mathrm{O} \xrightarrow{h v} (\mathrm{C H} _ {2} \mathrm{O}) _ {\mathrm{O M}} + \mathrm{O} _ {2 \text{atmosphere}} \tag{3}
$$
only in the right part, instead of synthesized biomass, there is a mass of sedimentary OM, and instead of synthesized oxygen, there is oxygen content in the atmosphere at the moment.
It remains to be convinced that expression (3) gives the same connections between substrates and products of global photosynthesis as the expression for traditional photosynthesis. To prove it replace the variables used to describe the theoretical relationship for traditional photosynthesis, the empirical relationship, observed between the rate of the substrate $(\mathrm{CO}_{2})$ loss and the rate of photosynthetic product $(\mathrm{O}_{2})$ accumulation in the Phanerozoic was used (Fig.2) as well as the relationship between the rate of accumulation of photosynthetic products (the rate of $\mathrm{O}_{2}$ production and the rate of OM synthesis). Instead of the OM synthesis rate, in the latter case, a sedimentation rate proportional to it is used.
Figure 2 shows the antiphase connection between the substrate $(\mathrm{CO}_{2})_{\mathrm{atm}}$ and the products $(\mathrm{CH}_{2}\mathrm{O}_{\mathrm{OM}}$ and $\mathrm{O}_2)$ of global photosynthesis for the Phanerozoic, obtained using the Geocarb III model.
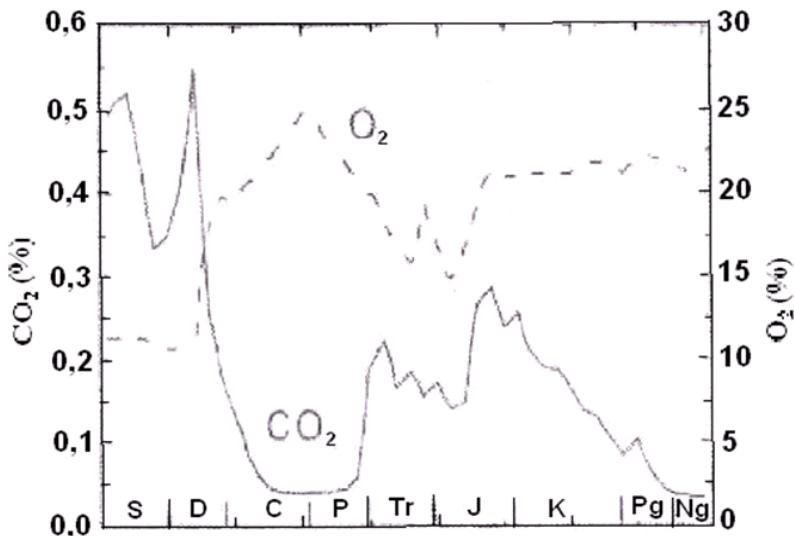
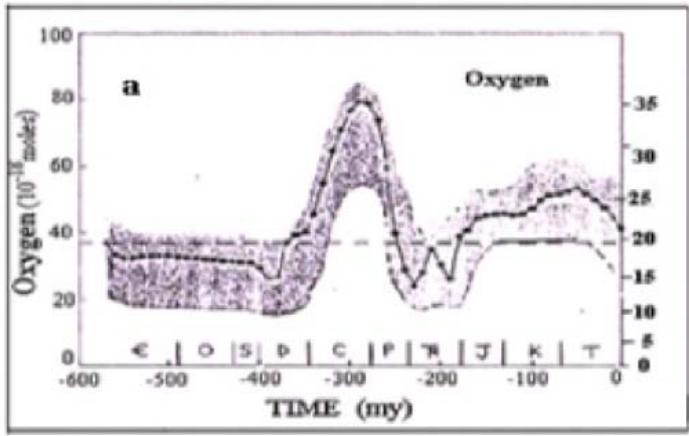 Fig. 2: Antiphase character of changes in the $\mathrm{CO}_{2}$ substrate (solid line) and the $\mathrm{O}_{2}$ product (dashed line) of the photosynthesis reaction in the Phanerozoic according to the calculation results using the Geocarb III model (Igamberdiev, Lea, 2006)
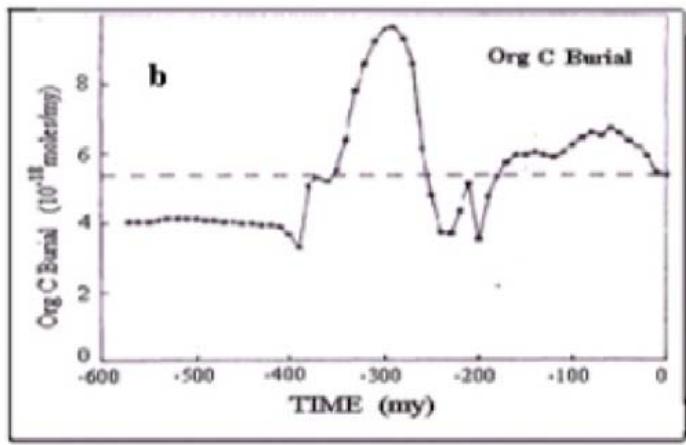 Fig. 3: The in-phase changes of oxygen content in the atmosphere (a) and burial organic matter rates in the sedimentary rocks in Phanerozoic.
Figure 3 shows the in-phase relationship between the products of global photosynthesis (atmospheric oxygen and the sedimentation rate of organic matter, calculated using the model (Berner, Canfield, 1989) for the Phanerozoic.
The fact that the curves describing the consumption of substrates and the yield of products during global photosynthesis in the Phanerozoic, obtained using geological models, are similar to the curves for traditional photosynthesis proves that global photosynthesis really functions in the biosphere part of global carbon cycle.
Properties of global photosynthesis. The evolution of global photosynthesis within the global cycle of biosphere carbon and in geological time (Ivlev, 2023).
It follows from the definition of global photosynthesis that its properties should be close to those of traditional photosynthesis. For the convenience of describing these properties, one should imagine a conditional photosynthesizing organism, the properties of which are obtained by averaging the properties of the ensemble of organisms. Such an organism should have all the properties except the ontogenetic ones related to age.
Global photosynthesis, like traditional one, consists of two oppositely directed reciprocal processes - $\mathrm{CO}_{2}$ assimilation and photorespiration. Taking into account the principle of actualism related to the global carbon cycle, it can be argued that $\mathrm{CO}_{2}$ assimilation is responsible for accumulation of sedimentary OM, while photorespiration reduces it. Moreover, an increase of the $\mathrm{CO}_{2}$ concentration in the environment increases the accumulation of sedimentary OM in the Earth's crust, and an increase in the concentration of $\mathrm{O}_{2}$ in the atmosphere decreases it. According to the proposed model of the global carbon cycle, the rate of sedimentary OM accumulation is maximal in the orogenic period of the cycle and begins to decrease to the minimum in the geosynclinal period, reaching it by the end of the period.
The second important property is the ability to fractionate carbon isotopes. The signs of isotopic effects are opposite: Assimilation of $\mathrm{CO}_{2}$ is accompanied by enrichment of OM in $^{12}\mathrm{C}$, photorespiration is accompanied by enrichment of OM in $^{13}\mathrm{C}$. According to the mechanism of the global carbon cycle, the most light carbon isotope composition is formed in the orogenic period of the cycle. In the geosynclinal period, there is an enrichment of OM carbon in $^{13}\mathrm{C}$, which increases by the end of the period. It should be recalled that the enrichment of OM in $^{13}\mathrm{C}$ in the geosynclinal period occurs not only due to accumulation of oxygen in the atmosphere and corresponding increase of photorespiration, but also due to the effect of $\mathrm{CO}_{2}$ depletion in the "atmosphere-hydrosphere" system because of photosynthesis (Rayleigh depletion isotope effect).
After getting acquainted with the basic properties of global photosynthesis, it is possible to consider its evolution basing on the proposed model and the available factual material. It is easy to see that the nature of the evolution of photosynthesis in a separate orogenic cycle and during geological time differ.
In a separate orogenic cycle, the evolution of photosynthesis has a pulsation character. Indeed, given the short duration of the orogenic period, the $\mathrm{CO}_{2}$ injections coming from the subduction zone can be considered as pulsations. Carbon dioxide that fills the Earth's "atmosphere-hydrosphere" system and achieves maximal values, then gradually decreases during long-term geosynclinal period up to the minimal values thanks to photosynthesis as well as, cooling and glaciation by the end of the geosynclinal period gives one more confirmation in favor of the idea of the pulsation character of evolution of the global photosynthesis within a separate orogenic cycle.
In geological time the evolution of global photosynthesis has a different mode. Each new orogenic cycle begins at a lower $\mathrm{CO}_{2}$ concentration level and ends at a higher oxygen concentration level as compared with the previous orogenic cycle. This allows saying that evolution during geological time occurs stepwise but steadily progressively in the direction of an increase in oxygen content and a decrease in $\mathrm{CO}_{2}$ concentration.
The idea on global photosynthesis evolution allows us considering the question of how the accumulation of sedimentary matter in the Earth's crust and oxygen in the atmosphere occurred.
The idea of global photosynthesis evolution allows considering the question of How did the accumulation of OM in the Earth's crust and oxygen in the atmosphere occur?
From the properties of global photosynthesis described earlier, it clearly follows that $\mathrm{CO}_{2}$ assimilation is responsible for the accumulation of OM in the Earth's crust, and photorespiration is responsible for the decrease of accumulated OM. It is also obvious that photorespiration increases over time, as oxygen accumulates during the evolution of global photosynthesis. This is supported by Table 1, although accuracy of the data provided in it, obtained by different authors, is rather low, the trend is quite obvious. This means that in the course of geological time, the accumulation of OM in the earth's crust decreases.
Table 1: Estimates of the average concentrations of $\mathrm{O}_2$ in the atmosphere during geological time, obtained by different models.
<table><tr><td>Eon/Era
Numerical age
Ma</td><td>Approximate
Value</td><td>References</td></tr><tr><td>Precambrian/ Paleoproterozoic
2200 – 2000</td><td>~ 0,2%</td><td>Holland 1998; Bjerrum, Canfield, 2004</td></tr><tr><td>Precambrian/ Neoproterozoic
1700 – 570</td><td>2 – 3%</td><td>Canfield, Teske, 1996</td></tr><tr><td>Phanerozoic/
Cambrian- Devonian
570 – 350</td><td>< 15 – 17%</td><td>Berner, Canfield, 1989; Berner et al, 2000; Berner, 2003</td></tr><tr><td>Phanerozoic/
Carboniferous- Permian
350 – 230</td><td>25 – 30%</td><td>Lenton, 2001</td></tr><tr><td>Phanerozoic/ Mezozoic
Triassic – Cretaceous
230 - 145</td><td>20%</td><td>Lenton, 2001; Bergman et al., 2004</td></tr><tr><td>Phanerozoic/ Cenozoic/Neogene/Miocene 23</td><td>23%</td><td>Berner, Kothavala, 2003</td></tr></table>
Within the frames of model, the OM accumulation in the Earth's crust may occur only in the orogenic period of the cycle, since namely in this period the CO2, substrate for assimilation, enters the Earth's surface from the subduction zone. In the geosynclinal period CO2 is not enter onto the Earth's surface but photosynthesis goes on, i.e. CO2 is consumed. Therefore, it can be argued that over time (with the repeated orogenic cycles), two trends work in the accumulation of OM in the earth's crust: the accumulation of OM in the crust goes on, but with each new cycle, the accumulation becomes less and less, i.e. the accumulation OM increment strives to 0 because of the increasing photorespiration.
This will go on until the accumulation of OM in the earth's crust stops, while the accumulation increment becomes 0. At the same time the oxygen content in the atmosphere, in accordance with the equation for global photosynthesis, stabilizes.
We called this state of the global carbon cycle by the point of ecological compensation. A similar state in traditional photosynthesis is called the compensation point (Edwards, Walker, 1983). It corresponds to a state where the contribution of photorespiration to plant metabolism becomes comparable to the contribution of $\mathrm{CO}_{2}$ assimilation. Global photosynthesis differs from traditional photosynthesis in that its evolution is always going in one direction, while the oxygen concentration increases all the time, as well as photorespiration. This is confirmed by the observed enrichment of biomass in $^{13}\mathrm{C}$ over time (Garrels, 1975; Hayes et al., 1999; Andrusevich et al., 1998; 2000).
Upon reaching the state, the system goes into a stationary state, in which all characteristics begin to oscillate around some average values (lvlev, 2023).
Table 1 confirms this presentation. As shown from the Table 1, it is obvious that the oxygen content rapidly increases in the atmosphere up to the Carboniferous-Permian epoch, where it gets maximal meaning. Then there appeared significant fluctuations around this value. This may be considered as an approximation to the ECP, where the oxygen content achieves stationary state, and therefore the accumulation of OM in the earth's crust does not occur.
In the ECP state, all the characteristics of the global carbon cycle become oscillatory. Fluctuations occur near some stationary averages. The data presented in Table 1, although empirical and approximate, allow us to see how the oxygen saturation of the atmosphere changes. As can be seen from Table 1, the oxygen concentration increased rapidly until the period. After that, the increase in oxygen content is completed, which indicates the frequency of the proximity of the ECP state.
## II. ARGUMENTS AND FACTS SUBSTANTIATING THE MODEL OF GLOBAL REDOX-CYCLE OF BIOSphere CARBON. PART II
1. Chemical and isotopic data prove the oxidation of sedimentary sediment OM in the subduction zone with the participation of thermochemical sulfate reduction.
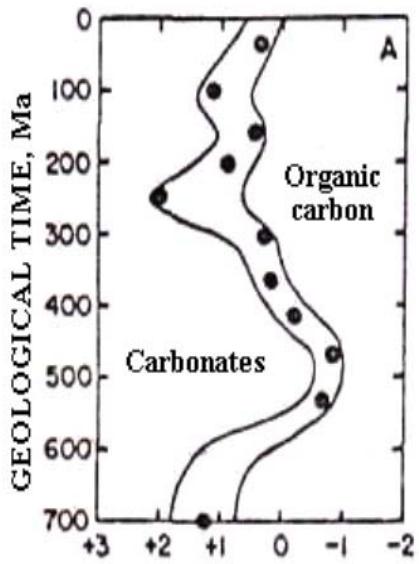
Fig. 4 shows the data of Macktnzie and Piggot (Mackenzie, Piggot, 1981), who studied the secular variations of carbon isotope composition of marine carbonates and sulfur isotope composition of marine sulfates covering Proterozoic and Phanerozoic. The curves display an amazing synchronicity. Given the probable connection of these curves with the oxidation reaction of OM and with the reaction of thermochemical sulfate reduction, this synchronicity indicates, that these reactions are somehow related.
 13 C,%
 $\Delta^{34}\mathbb{S},\%$ Fig. 4: Synchronous variations in the carbon isotope composition of marine carbonates and those sulfur isotope composition of marine sulfates in the Proterozoic and Phanerozoic (Mackenzie, Piggot, 1981).
At the bottom and at the top the curves have two humps oppositely directed (Fig.4). Next to the humps on the curves, the authors made inscriptions indicating the widely spread minerals corresponding to the time intervals of mentioned humps. If examine closely Fig.4, it is easy to find that the lower humps correspond to carbonates and sulfides which are the products of the sedimentary OM oxidation by means of the sulfate reduction. If to address to the upper humps, which the authors link to the prevalence of the sulfates and the rocks, rich in OM, one should conclude they are likely the substrates of the same reactions.
Note, If this assertion is true the opposite directions of humps become clear. This conclusion, based on the analysis of chemical data, are confirmed by the isotopic data based on the analysis of isotope fractionation in the chemical reactions under study.
As shown, the lower hump on the right curve (Fig.4) is directed to the right, what indicates that sulfate reduction occurs, causing their $^{34}\mathrm{S}$ enrichment. At the same time, the hump on the left curve corresponds to the enrichment of carbonates with the isotope $^{12}\mathrm{C}$. This is due to the fact that when sulfate reduction takes place, the oxidation of hydrocarbons proceeds in parallel. The $\mathrm{CO}_{2}$ produced in this case inherits the "light" isotope composition of the OM, since its complete oxidation occurs. Therefore, when $\mathrm{CO}_{2}$, enriched with the isotope $^{12}\mathrm{C}$, enters the marine "carbon dioxide - bicarbonate - carbonate" system, which carbon more enriched in $^{13}\mathrm{C}$, the total carbon of the marine system becomes enriched in $^{12}\mathrm{C}$. The more complete the sulfate reduction is, the more enriched in $^{12}\mathrm{C}$ the carbon of the natural marine system becomes.
One more indirect argument in favor of the above assertion can be given. At the early stages of the evolution when oxygen content in the atmosphere was low, some amount of $\mathsf{H}_2\mathsf{S}$, produced in sulfate reduction, achieved the Earth's surface. It was the reason for the widespread distribution of sulfate-oxidizing bacteria, the prevalence of which was so great that the authors of the work below came to the conclusion that their bacterial biomass was the main supplier (Hayes, 1983) for the OM at that time.
2. Experimental confirmation of the model's conclusion about the regular sequence of biosphere processes.
The suggested mechanism of the redox-cycle of biosphere carbon functioning implies a regular sequence of biosphere events due to the coupling of physico-chemical processes acting in the cycle. It can be traced in the changes of some parameters.
Investigating long-term fluctuations in the carbon isotope composition of marine carbonates in the Proterozoic Derry and colleagues (Derry et al., 1992) have discovered regular variations of the parameter. They used not only their own data, but also those available from the literature. But the cause of regular changes with time, which were clearly visible from the data the authors could not explain.
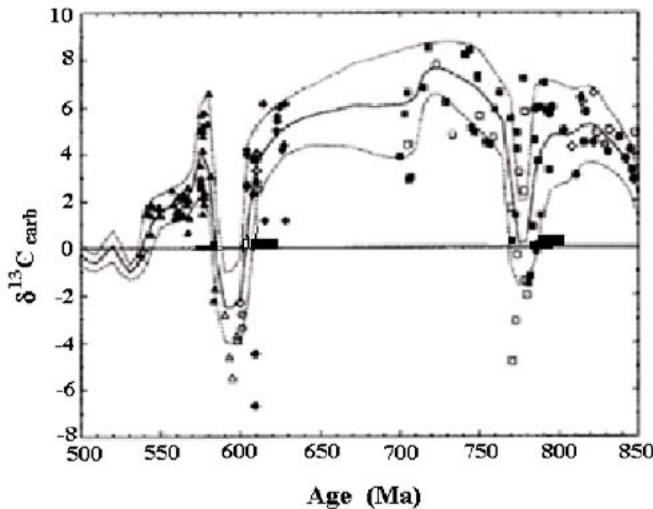 Fig. 5: Long-term variations of $\delta^{13}\mathrm{C}$ marine carbonates in the Late Proterozoic. The samples were taken from various stratigraphic groups (Derry et al., 1992).
However, these isotopic variations are easily explained within the framework of the proposed mechanism of the carbon redox-cycle. Indeed, imagine that the negative excursions of $\delta^{13}\mathrm{C}$ carbonates are the peaks, corresponding to the changes in the parameter during the orogenic periods of two consecutive orogenic cycles. This is supported by their negative values and by the fact that the peaks are narrow, and hence the periods are short. Negative values indicate the association of peaks with the oxidation of OM in sulfate reduction. In addition, negative peaks in orogenic periods are confirmed by the fact (see later) that they are associated with the influx of chemical elements from igneous rocks and with the formation of rocks rich in organic matter ("black" shales) (Kendall et al., 2006; Ivlev, 2023).
The adjacent part of the cycle is wide, what indicates that it corresponds to a long-term geosynclical period. In addition, it corresponds to more positive values of $\delta^{13}\mathrm{C}$, protruding in the opposite direction. But the most convincing argument is the following. As it follows from the mechanism of the cycle, glaciations should occur at the end of the geosynclical period, before the onset of a new cycle. Really, before the beginning of each cycle there are the rectangles, filled with black. They show that before the first cycle there was a Varangian glaciation ( $\sim 780$ Ma), before the second cycle there was a Sturtian glaciation ( $\sim 600$ Ma).
Young (Young, 2015) also discovered the existence of long-term cyclic fluctuations in the isotopic composition of marine carbonates in almost the same time interval. They manifested themselves in the form of negative excursions of $\delta^{13}\mathrm{C}$ carbonates, which were preceded by glaciations. Besides he found that the appearance of negative peaks was accompanied by mass extinction of organisms associated with the increase in the level of radioactivity. However the extinction might be the result of abrupt climatic changes though it might be partly the result of radiation. In other words, the signs described by Young are quite consistent with the regular sequence of events predicted from the model. The high level of radiation is explained by the arrival of radioactive elements from the depths along with igneous rocks in rifting. Thus, the predicted of physically justified regular sequence of biosphere events can be considered as an argument in favor of the model.
3. The stratigraphic unevenness of the distribution of world oil reserves and the number of discovered deposits in the territory of the former USSR proves the existence of orogenic cycles.
Some geologists have revealed a stratigraphic unevenness in the distribution of world oil reserves (Vyshemirsky, Kontorovich, 1997) and an almost synchronous unevenness in the distribution of the number of discovered fields (Korchagin, 2001).
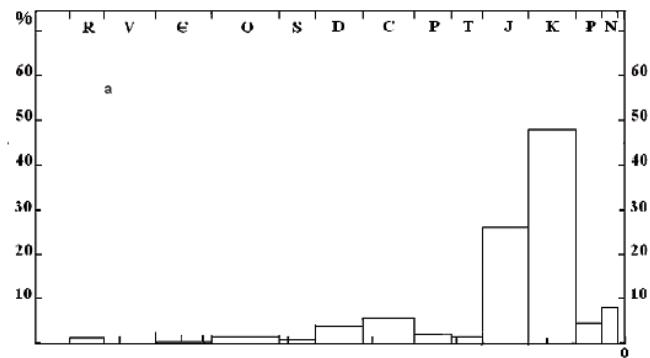
 GEOLOGIC TIME, May years Fig. 6: The comparison of the stratigraphic distribution of the initial explored recoverable oil reserves by the largest oil-bearing basins of the world (Vyshemirsky, Kontorovich, 1997) (a) and the distribution of oil deposits by stratigraphic complexes (b) (% of the number of deposits in the complex from the total number in the territory of the former USSR (Korchagin, 2001)).
The fact that the spikes in the distribution of world oil reserves and in the number of the discovered oil fields occur at approximately the same intervals of geological time indicates that they are caused by the reasons related to the change of orogenic cycles. The first spike dates back to the Riphean, to the period when the first industrial oil fields were discovered. It is notable, it appeared much later than the appearance of photosynthesis (about 2.2 billion years ago). Since, in accordance to the model, in order for industrial accumulation of oil to appear, it takes time for the necessary amount of OM to accumulate, it takes time for its transformation and maturation, the time for the emigration and accumulation of hydrocarbons in traps. In other words, the discovery of oil fields before the Riphean is extremely unlikely. From the other side, taking into account the presence of ECP, one can expect that since Miocene, when ECP occurred, the accumulation of OM in the Earth's crust has ceased and oils as well.
4. The study of the carbon isotope composition of oils is another argument in favor of the existence of orogenic cycles and the evolution of global photosynthesis.
Another parameter that can be used to monitor the functioning of the global carbon cycle and obtain arguments to justify the key points of the model is the isotopic composition of oil carbon.
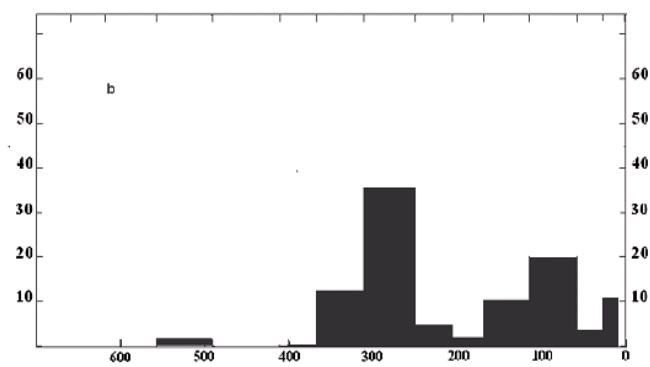
 Fig. 7: The change of the average carbon isotope composition $(\delta^{13}\mathrm{C},\%)$ for saturate fraction C15+ of crude oils. Vertical bars are standard deviations, which increase with decreasing age. Arrows indicate Cambrian - Ordovician, Triassic - Jurassic, and Paleogene-Neogene boundaries where $^{13}\mathrm{C}$ enrichment of oil carbon occurs (Andrusevich, et al., 1998, 2000).
Figure 7 shows a curve illustrating the changes in the carbon isotope composition of oils of different ages and origins. Andrusievich and colleagues (Andsrusevich et al., 1998) have studied a collection of 513 oils for the period from the Proterozoic and Phanerozoic inclusive. The authors found changes in the carbon isotope composition of oil over four time periods. On the arrow of geological time from the Proterozoic to the Neogene, they found four stages of change in the isotopic composition of carbon. Moreover, the direction of the enrichment of carbon in oil with the isotope $^{13}\mathrm{C}$ is clearly traced with each step appearing in time. Oil is increasingly enriched with a heavy isotope, with increasing age, as predicted by the model. The explanation is based on an increase in the photorespiratory activity of global photosynthesis due to an increase in the oxygen content in the atmosphere. The same trend, but using the example of the isotopic composition of carbon OM, was previously noted by Garrels (Garrels, 1974). Following the logic of the model, the observed stages of changes in the carbon isotope composition of oils are related to the sequence of orogenic cycles throughout the evolution of photosynthesis.
One more detail should be noted on Fig. 2. The spread of $\delta^{13}\mathrm{C}$ values has become noticeably larger since the Jurassic. The fact is that by this time photosynthetic Life had finally conquered the land with all its diverse environments. The number of parameters, on which photosynthesis and its isotopic fractionation depend, has increased. This is the reason for the spread of $\delta^{13}\mathrm{C}$ values.
5. The model of global redox-cycle of biosphere carbon is capable to substantiate and explain the origin and conditions of "black shale" formation and the associated traits and geological objects as well.
Most geochemists, having studied the "black shales" origin came to the conclusion that their formation occurred under conditions of oxygen-free phenomena in the ocean (OAE) (Arthur, Schlanger, 1979; Arthur et al., 1987). This is in a good agreement with the model of the global redox-cycle of biosphere carbon considered by us. According to the model, oxygen-free events occurred during short orogenic periods (Twenhofel W. H. (1939) Thunnel et al., 1984) that coincide with rifting, when magma breaks through the lithosphere, turning into a new plate. Along with magma, other endogenous fluids penetrated through the lithosphere, including reduced forms of metals, simultaneously with other components of the Earth's deep breathing.
The reduced forms of endogenous elements, reacting with atmospheric oxygen, led to anoxic conditions. The mass extinction of organisms, caused by abrupt change of climatic conditions, ensured the formation of deposits rich in organic matter, which later turned into "black shales" (Bazhenova, Sokolov, 2002). The model not only reasonably explained the formation of "black shales", but also quite clearly described the traits accompanying their formation (see later).
According to the various estimates, the $\mathrm{CO}_{2}$ content in the periods of the OAE increased 5-6 times, for example, during the OAE in the Cretaceous it reached 2000 ppm (cf. with a modern content of 380 ppm). With the onset of the greenhouse period the temperature of the surface waters of the ocean increased by $7 - 13^{\circ}C$ (Erba, Bottini, 2009). Anoxia and a sharp increase in temperatures caused the mass extinction of organisms. This brought about drastic changes in the abundance and taxonomic diversity of fossil biota (Yudovich, Ketris, 2015).
The fact that the formation of "black shales" occurred when combined with the mineral part of almost any composition (clay, sand-siltstone, carbonate, siliceous, tuff, etc.) with organic part in a variety of polyfacial conditions (lake, delta, littoral, lagoon, shallow, shelf, etc.). This evidences that formation of sediments, rich in organic matter, is due to objective processes accompanying the change of orogenic cycles.
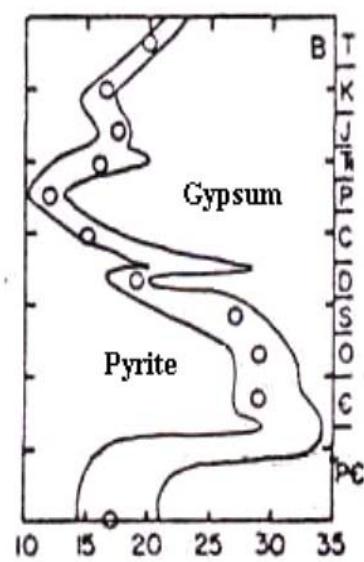
On the cause of high sulfurization of OM and oil of "black shales" rocks
A typical feature of the "black shales", their high sulfur content, has long been noticed by oil geologists. Sometimes it can reach $10\%$. High sulfur content was firstly found in domanic (Baturin, 1978). These rocks are also referred to as "black shales". The high sulfur content was confirmed Parparova (Parparova, 1975) and Kontorovich (Kontorovich, 1976). Moreover, high sulfur content is often combined with the presence of pyrite (FeS2) (Yudovich, Ketris, 2015), which has two sulfur atoms in its composition. The question arose, what was the source of sulfur?
It is still believed that the sulfurization of OM occurs at the interaction with hydrogen sulfide, which is formed during the microbiological reduction of sulfate in the near-surface anaerobic zone of the diagenetic transformation of OM. It is obvious that the oxidizer in this case is seawater sulfate (Volkov, 1984). In order for such a reaction to take place, it is necessary that the conditions for the course of sulfate reduction and the vital activity of bacteria, as well as there should be a certain degree of transformation of OM with multiple carbon bonds and atoms capable of being replaced by sulfur atoms. Although it is very difficult to find such a combination of conditions in nature, but no other alternatives have been proposed. The global biospheric carbon cycle model offers an alternative explanation. Let's consider the abiogenic thermochemical reduction of sulfates, which occurs in the subduction zone and is used for OM oxidation.
In addition to the above data from the work of Mackenzie and Piggott (Mackenzie, Piggott, 1981), which prove the existence of the thermochemical sulfate reduction in subduction zone, there are data, obtained Shaver et al (Shaver et al., 2018), evidencing on activity of sulfate reduction in the depth and thus indirectly confirming the participation of hydrogen sulfide in OM and oil sulfurization. They found that sulfur isotope composition of pyrites is significantly enriched in $^{32}\mathrm{S}$ as compared with sulfur of OM. We explain this interesting fact as follows.
Hydrogen sulfide, formed in the subduction zone in sulfate reduction, along with the produced $\mathrm{CO}_{2}$, passes the zone of "active" (reduced) iron, which binds $\mathrm{H}_{2} \mathrm{~S}$. Only after that, the excess amount of $\mathrm{H}_{2} \mathrm{~S}$ penetrates into the upper layers, where it participates in binding reactions with OM. Remind, the thermochemical sulfate reduction reaction is accompanied by the Rayleigh effect, according to which $\mathrm{H}_{2} \mathrm{~S}$, formed at a deeper degree of sulfate conversion, turns out to be enriched in $^{34} \mathrm{~S}$. Hydrogen sulfide, formed in the subduction zone in sulfate reduction, along with the produced $\mathrm{CO}_{2}$, passes the zone of "active" (reduced) iron, which binds $\mathrm{H}_{2} \mathrm{~S}$. Thus, the combination of temporal isotopic differences in the producing $\mathrm{H}_{2} \mathrm{~S}$ in course of sulfate reduction with the spatial separation of the pyrite formation zone and the zone of interaction of OM with $\mathrm{H}_{2} \mathrm{~S}$ explains the observed isotopic differences.
The nature of abnormal geochemical associations of "black shales" with compounds of rare metals and rare earth elements.
Geologists have long noticed the association of "black shales" with powerful geochemical anomalies represented by compounds of rare earths and metals (Mo, V, Re, Hg, Ni, etc.) (Yudovich, Ketris, 2015; Herazo et al., 2021). The question is, what connects these completely independent biosphere processes: "black shales" formation and geochemical association. The pathway of "black shales" formation was described by the global carbon cycle model. The dominance of volcanic and volcanogenic rocks in the area of the metal association formation supports the idea on volcanic origin of the associations (Twenhofel, 1939). The question remains, what connects these seemingly completely independent processes.
To answer this question, let's see if both processes have a common point of contact. The analysis shows that such a point is rifting, i.e. the moment when the magma flow with the flow of deep fluids, consisting of volcanic gases that include volatile compounds of elements, breaks through lithosphere shell. Further, the destinies of both flows develop independently. Magma flow forms new lithosphere plates, the collisions of which stimulate biosphere processes on the earth's surface. The processes, described above, compose pathway, which can be called "biosphere" pathway.
It can be briefly described as follows. Carbon goes through the stage of photosynthesis, the formation of a "living" matter which dies at orogenic cycles change, forming sediments, rich in OM. "Black shales" are formed during the subsequent transformation of sedimentary OM. The cycle change occurs in the short orogenic period of the cycle when deep fluids along with magma break through the lithosphere shell.
During the orogenic period, volatile compounds of metals, carried by volcanic exhalations, get onto the Earth, form a geochemical association, which is bound
- to "black shales" because both processes occur at the same time (orogenic periods of the cycle) and originate from same source (inner fluids of Earth breathing).
6. Long-term recurring fluctuations in the content of trace elements in seawater are an argument in favor of the global carbon cycle model.
Long-term cyclic variations of the concentrations of bio-essential trace elements (molybdenum, selenium, cadmium, and thallium) and macro-element phosphorous, first was revealed in the Proterozoic by Zerkle and colleagues (Zerkle et al., 2005). Later the fluctuations were described by Large and colleagues (Large et al., 2015). They found that Phanerozoic seawater also demonstrated the same fluctuations of spread from maximum to minimum. The maximum corresponded with a period nutrient rich ocean and vice versa.
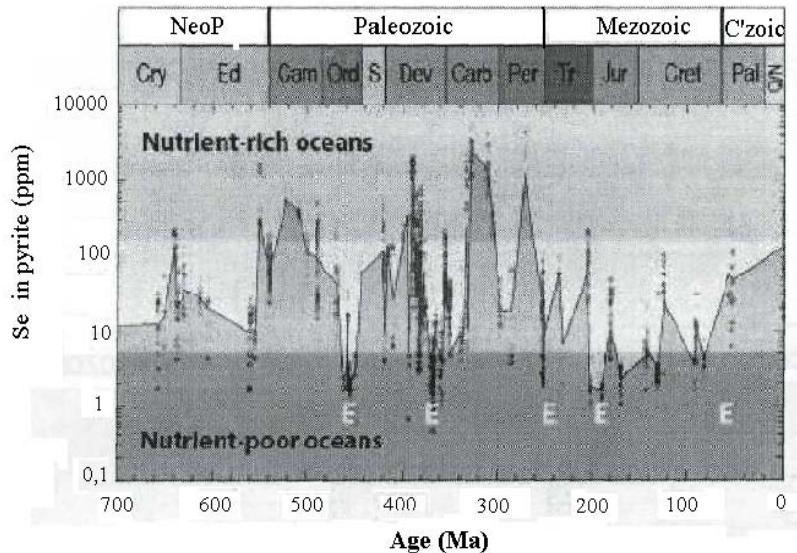 Fig. 8: Temporal pattern of Se in marine pyrite. The gray area, which covers the curve of selenium variations from above, shows an ocean rich in bio-essential microelements. The black bottom area, which covers the curve of selenium variations from below, characterizes the ocean poor in bio-essential microelements. E denotes the fixed times of mass extinctions on a geological scale (Large et al., 2015).
The distinct cyclic variations were confidently confirmed by statistical processing of the material, using Spearman correlation coefficients. The results of the mathematical treatment show the cyclic character of TE variations, which begin with volcanism and end with full depletion of TE and by extinction of organisms The variations of selenium, one of the most indicative elements, is presented on Fig.
8.
A comprehensive study of long-term fluctuations in seawater reveals a direct analogy between the formation of geochemical associations of "black shales" with rare metals and fluctuations of trace elements in seawater. The model of the global redox cycle of biospheric carbon allows displaying this analogy and explaining the nature of this relationship. Two items should be paid attention to.
First, the nature of metals in geochemical association with "black shales" and the composition of trace elements in seawater are unanimously associated with volcanic activity or, taking into account what was said earlier, with deep breathing of the earth.
Secondly, volcanic emissions and volatile compounds of elements, including metals, which together with the magma break through the lithospheric shell, forming a new plate during rifting. These processes coincide with the orogenic period of plate's movement. The further fate of metals making an association with "black shales" and the formation of the "black shales" themselves were discussed above.
Volcanic ash, and igneous rocks brought to the Earth's surface with volcanic exhalations are exposed to the environmental action. The microelements contained in them are leached out. Meteor waters carry solutions of trace elements into the ocean. From there they fall into the sediment. It is obvious that the maximum concentrations of TE are achieved during the orogenic period. During the geosynclinal period, they are used as nutrients by the inhabitants of the oceans and their concentrations drop to the minimum.
It should be noted that the content of trace elements in seawater cannot be measured directly. It was proven that TE content of marine sedimentary pyrite in black shales is a good approximation of characteristics of oceanic water. This can be also explained using the model in question. It follows from the above that the iron participating in the formation of pyrite enters the sedimentary strata, like other trace elements, during the same orogenic period. In the same time the formation of "black shales" occurs. After the absorption of pyrite by the "black shale", trace elements do not participate in the processes in seawater. And therefore, the composition of trace elements preserves the memory of the original composition in seawater.
## III. CONCLUSIONS
The idea of V.I. Vernadsky on the interaction of geological and biospheric processes is considered and a probable mechanism based on the postulate of the gravitational effect of solar system bodies on the movement of lithospheric plates is proposed.
The impact leads to complex magma flows in the asthenosphere and deep breathing of the Earth, in the form of hydrogen stream coming from the core, which is joined with volatile compounds of other elements. The lithosphere shell changing, reflects these processes. When spreading, it expands and become thin. Then rifting comes, in which magma, along with other fluids, breaks through the lithosphere shell and, in contact with oceanic water, solidifies, forming a new plate. The described internal processes manifest themselves in the form of uneven and continuous movement of lithosphere plates moving along the surface of molten magma.
The movement of lithosphere plates is cyclical. Cycles are called orogenic and consist of two periods. A short orogenic period of relatively rapid plate movement coincides with rifting. In parallel with the formation of a new plate, some of the fluids erupt in the form of volcanoes and lava flows. A new plate, pushing apart neighboring plates, causes their accelerated movement. In the subduction zone, oceanic plates collide with plates of the continental margins. Smaller in size oceanic plates sink into magma and dissolve in it. The continental plate rises and participates in mountain formation.
The most important result of collisions in the subduction zone during the orogenic period is that the energy released in them leads to the activation of thermochemical reduction of sulfates, in which the OM is oxidized. The $\mathrm{CO}_{2}$ formed during oxidation rises to the surface, initiating photosynthesis there.
During the long geosynclinal period of the plate movement cycle, the released collision energy turns out to be insufficient to initiate thermochemical sulfate reduction and oxidation of organic matter does not occur. Carbon dioxide does not reach the Earth's surface.
In the biosphere on the Earth's surface, maximum concentrations of $\mathrm{CO}_{2}$ are achieved during the orogenic period, and taking into account the greenhouse properties of this gas, maximum temperatures are achieved as well. The reduced metals of igneous rocks rising with the lava react with atmospheric oxygen, making the environment anaerobic. The rate of photosynthesis is maximal.
In the geosynclinal period, due to the lack of $\mathrm{CO}_{2}$ input from the subduction zone, its concentration begins to fall and becomes minimal by the end of the period. The temperature also drops until glaciations. On contrary, the oxygen concentration, is growing up.
A joint consideration of the changes in environmental parameters during the orogenic cycle allows asserting that the evolution of global photosynthesis within a separate orogenic cycle on the Earth's surface, controlled by the $\mathrm{CO}_{2} / \mathrm{O}_{2}$ ratio, has a pulsatory A joint consideration of the changes in environmental parameters during the orogenic cycle allows asserting that the evolution of global photosynthesis within a separate orogenic cycle on the Earth's surface, controlled by the $\mathrm{CO}_{2} / \mathrm{O}_{2}$ ratio, has a pulsatory character, and evolution during geological time has a stepwise progressive character. As a result, climatic cycles and a biotic turnover arise on the Earth. The evolution of global photosynthesis on the Earth brings the global carbon cycle system to an ecological compensation point, i.e. to a stationary state, when the accumulation of hydrocarbons in the earth's crust is completed, and the oxygen concentration in the atmosphere is stabilized.
The key points of the model are confirmed by natural facts based on chemical and isotopic data and interpretation relying on the principle of actualism and modern physico-chemical laws. character, and evolution during geological time has a stepwise progressive character. As a result, climatic cycles and a biotic turnover arise on the Earth. The evolution of global photosynthesis on the Earth brings the global carbon cycle system to an ecological compensation point, i.e. to a stationary state, when the accumulation of hydrocarbons in the earth's crust is completed, and the oxygen concentration in the atmosphere is stabilized.
Completing the work, it should be noted that, of course, the model that underlies the conclusions obtained, of course, is only an approximation to the real description of the system, but the thing is that it makes possible to describe the processes that lead to facts that can be verified (and such independent facts are quite a lot) suggests that the model is a good approximation and can be used as a working tool for further research.
### ACKNOWLEDGMENTS
The author expresses his sincere gratitude to Prof. V.I. Petersillie and to Prof. N.M. Prjeversky for comprehensive support and useful discussion of the work.
Generating HTML Viewer...
References
51 Cites in Article
Vladimir Andrusevich,Michael Engel,John Zumberge (2000). Effects of paleolatitude on the stable carbon isotope composition of crude oils.
V Andrusevich,M Engel,J Zumberge,L Brothers (1998). Secular, episodic changes in stable carbon isotope composition of crude oils.
M Arthur,S Schlanger,H Jenkyns (1987). The Cenomanian-Turonian Oceanic Anoxic Event, II. Palaeoceanographic controls on organic-matter production and preservation.
M Arthur,S Schlanger (1979). Cretaceous "ocean anoxic events" as causal factors in the development of reef-reservoired giant oil fields.
G Baturin,P Bezrukov (1978). Phosphorites on the sea floor and their origin.
T Boyko,G Butuzova,Yu,D Brown (1986). To the question of the role of volcanic components, sorbed pyroclastics., in feeding the oceans with dissolved material.
R Berner,D Canfield (1989). A new model for atmospheric oxygen over Phanerozoic time.
M Bergman,T Lenton,A Watson (2004). COPSE: a new model of biogeochemical cycling over Phanerozoic time.
Robert Berner (2003). The long-term carbon cycle, fossil fuels and atmospheric composition.
R Berner,D Canfield (1989). A new model for atmospheric oxygen over Phanerozoic time.
R Berner,S Petsch,J Lake,D Beerling,B Popp,R Lane,E Laws,M Westley,N Cassar,F Woodward,W Quick (2000). Isotope fractionation and atmospheric oxygen: implications for Phanerozoic O 2 evolution.
O Bazhenova,B Sokolov (2002). Chapter 8 Origin of oil and natural gas.
R Berner,Z Kothavala (2001). GEOCARB III: a revised model of atmospheric CO 2 over Phanerozoic time.
Christian Bjerrum,Donald Canfield (2004). New insights into the burial history of organic carbon on the early Earth.
D Canfield,A Teske (1996). Late Proterozoic rise in atmospheric oxygen concentration inferred from phylogenetic and sulphur-isotope studies.
L Derry,A Kaufman,S Jacobsen (1992). Sedimentary cycling and environmental change in the Late Proterozoic: Evidence from stable and radiogenic isotopes.
N Dobretsov (2010). Global geodynamic evolution of the Earth and global geodynamic models// Geology and geophysics.
G Edwards,D Walker (1983). C 3 , C 4 : Mechanisms, and cellular and environmental regulation of photosynthesis.
E Erba,C Bottini (2009). The response of Cretaceous calcareous nannoplankton to pCO2 and ocean acidification // Geochim.
R Flint (1973). The Earth and its history.
Š Goričan,E Carter,G O'dogherty,P Wever,D Paulian,R Hori,A Matsuoka,P Whalen (2013). Evolutionary patterns and palaeobiogeography of Pliensbachian and Toarcian (Early Jurassic) Radiolaria.
J Hayes,H Strauss,A Kaufman (1999). The abundence of 13 C in marine organic matter and isotopic fractionation in the global biogeochemical cycle of carbon during the past 800 Ma.
J Hayes (1983). Geochemical evidence bearing on the origin of aerobiosis a speculative hypophesis.
A Herazo,M Reitch,F Barra,I Contreras,R Del (2021). Trace Element Geochemitry of Pyrite From Bitumen-Bering Stratabound Cu-(Ag) Deposits Northern Chile.
H Holland (1965). The history of ocean water and its effect on the chemistry of atmosphere.
A Ivlev (2023). A New View on the Global Redox-Cycle of Biosphere Carbon.
A Ivlev (2023). On the Origin of "Black Shales" in the Framework of the Global Redox Cycle Model of Biosphere Carbon.
A Ivlev (2019). Manifestations of Photosynthesis in the Evolution of the Global Carbon Cycle.
A Ivlev (2019). Manifestations of Photosynthesis in the Evolution of the Global Carbon Cycle.
А Ivlev (2019). Formations of sequences rich in organic matter in the light of new model of global carbon cycle.
A Ivlev,R Pankina,G Galperin (1973). Thermodynamics of oil desalination reaction.
B Kendall,R Cruiser,D Selby (2006). Geochronology of postglacial black shales in Australia: limitations of the Sturtian glaciation.
A Kontorovich (1976). Geochemical methods of quantitative prediction of oil and gas bearing. M. 249.
V Korchagin (2001). General stratigraphic scale and distribution of oil and gas deposits by stratifications of subdivisions of Phanerozoic and Precambrian. Table composed based on acting stratigraphic lowbook.
R Large,J Halpin,E Lounejeva,K Danyushevsky,V Maslennikov,D Gregory,P Sack,P Haines,J Long,Ch Makoundi,S Stepanov (2015). Cycles of nutrient trace elements in the Phanerozoic ocean.
T Lenton (2001). The role of land plants, phosphorous weathering and fire in the rise and regulation of atmospheric oxygen.
F Letnikov (2001). Ultra-deep fluid systems of the Earth and problems of rifting // Geology of ore deposits.
F Mackenzie,J Pigott (1981). Tectonic controls of Phanerozoic sedimentary rock cycling.
A Monin (1977). Istoriya Zemli [History of the Earth.
G Parparova (1975). On the question of the osernenpp of scattered organic matter of rocks.
N Podkletnov (1985). Volcanogenic organic matter.
M Rutten (1971). The origin of life by natural causes Amsterdam.
L Shawar,I Halevy,W Said-Ahmad,Feinstein Sh,V Boyko,Kamyshny Amrani,A (2018). Dynamics of pyrite formation and organic matter sulfurization in organicrich sediments.
Robert Thunell,Douglas Williams,Paul Belyea (1984). Anoxic events in the Mediterranean Sea in relation to the evolution of late Neogene climates.
W Twenhofel (1939). Environments of origin black shales.
I Volkov (1984). Geochemistry of sulfur in ocean sediments.
V Vyshemirskii,A Kontorovich (1997). Cyclic character of oil accumulation in Earth history.
G Young (2015). Environmental upheavals of the Ediacaran period and the Cambrian "explosion" of animal life.
Y Yudovich,M Ketris (2010). ЧЕРНЫЕ СЛАНЦЫ И НАФТОГЕНЕЗ. ОБЗОР.
Yudovich Ya,E Ketris,M (2015). The Geochemistry of Black Shales.
A Zerkle,C House,S Brantley (2005). Biogeochemical signatures through time as inferred from whole microbial genomes.
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.