Between 2017 and 7 March 2025, Ebola virus disease occasioned eight outbreaks killing 2,471 people out of 3,892 cases in sub-Saharan Africa. Given the pandemic capacities of the largest Ebola epidemics that occurred in West Africa from 2014-2016 and the socioeconomic catastrophe wrought by the COVID-19 pandemic, all hands must be on deck to mitigate recurrent Ebola outbreaks. Hence, this piece explores the historical trajectory of Ebola virus disease and its epidemiological attributes in the contemporary era. We deployed a narrative review of relevant articles on the Ebola virus disease from its inception in 1976 to 2025. Between 2001 and 2025, 26 outbreaks of EVD occurred in sub-Saharan Africa, with an average case fatality rate of 43 percent. We discovered that 46 percent of the epidemics occurred in DRC Congo, and Zaire Ebola strains were responsible for 73 percent of outbreaks.
## I. INTRODUCTION
There is a recurrence of high-risk pathogenout breaks in the contemporary era. Filoviral diseases including Ebola virus diseases (EVD) have occasioned different epidemics with catastrophic mortalities in $21^{\mathrm{st}}$ century sub-Saharan Africa. Given the devastating economic and social consequences of the COVID-19 pandemic, efforts should be geared towards mitigating another cataclysmic pandemic. In 2022, the World Health Organization marshaled an action plan against high-risk pathogens capable of triggering the next pandemic. These contagious organisms included viral hemorrhagic viruses including Ebola virus and Marburg virus. $^{1}$ Ebola virus disease (EVD) and Marburg virus disease (MVD) have extremely high fatality rates ranging up to 90 percent in past and recent epidemics. They belong to the family, filoviridae composed of three genera Cueavirus, Marburgvirus, and Ebolavirus. Six species have been identified in the genus Ebolavirus, they are Zaire, Bundibugyo, Sudan, Tai Forest, Reston, and Bombali.2 The latter two strains have not been attributed to any Ebola infections.
The repeated outbreaks of EVD in contemporary sub-Saharan Africa and the likelihood of escalation to worldwide pandemics spurred our interest in this review. For example, Uganda has witnessed five outbreaks of Sudan virus disease (SVD), a family of EVD since 2000, with the largest outbreak in 2022 claiming 55 lives out of 164 cases.3 As of 20 February 2025, one death has been recorded out of nine cases of SVD in Uganda.3 The largest epidemic of EVD specifically ravaged Liberia, Guinea, and Sierra Leone between 2014 and 2016, claiming 11, 308 lives out of 28, 600 cases.4 Between 2017 and 2022, sub-Saharan Africa has witnessed seven outbreaks of EVD with five of them occurring in Democratic Republic of Congo and one occurring in Guinea and another one in Uganda. Within this period, there were 2412 reported deaths out of 3714 Ebola sufferers.4 Given the increasing rate of road interconnectivity, transportation, and urbanization across sub-Saharan Africa, the risk of escalation of filoviral epidemics to pandemics has never been greater, hence the continuing advocacy for efficient disease preparedness, community engagement and continuing vaccine research against filoviruses.
The Ebola virus genome is a negative-sense single-stranded RNA. It is composed of a viral envelope, matrix, and nucleocapsid components. It encrypts seven structural proteins: nucleoprotein (NP), polymerase cofactor (VP35, VP40, GP), transcription activator (VP30, VP24), and RNA-dependent RNA polymerase. $^{5}$ Ebola virus belongs to the family of filoviridae and comprises of six identified species. They are Zaire, Bundibugyo, Sudan, Reston and Tai Forest, and Bombali. Zaire, Bundibugyo, and Sudan ebolaviruses have been linked to huge epidemics in sub-Saharan Africa. The virus occasioning the catastrophic 2014 West African epidemics belongs to the Zaire species. $^{6}$ Fruit bats of the family, Pteropodidae are the documented vector and reservoir of the virus. The first cases of EVD were reported in 1976 in Nzara, Sudan, and in Yambuku in Zaire, now Democratic Republic of Congo. The latter occurred around Ebola River where the contagion derived its appellation $^{6}$. Due to its high fatalityrates, Ebola virus is listed as an organism included in WHO
Risk Group 4 Pathogen requiring biosafety level 4-equivalent containment.
There is no paucity of research on the transmission of the Ebola virus as well as the clinical manifestations, diagnosis, and management of EVD. Most scholars document initial zoonotic transmission from Ebola virus reservoirs via contact with them and their droppings or through their consumption as foods. Such reservoirs include fruits bats, chimpanzees, monkeys, and antelopes. Subsequently, after a spillover, human transmission assumes the dominant dispersal mechanism via close contact with sufferers, blood, sweats, clothing, and even seminal fluids. Moreover, contact with infected bodies could transmit EVD. Therefore, hygienic and safe burial practices are recommended for Ebola dead bodies to mitigate the dispersal of the high-risk pathogen. Regarding its clinical features, the hallmark of EVD is hemorrhagic fever. The incubation period is between two to 21 days.
It is characterized by an initial phase of flu-like manifestations like fever, chills and rigors, myalgia, and weakness.6 After a few days, patients usually present with gastrointestinal features such as vomiting, diarrhea, and abdominal pain. The most common symptoms are fever, extreme weakness, vomiting, and maculopapular rash around the face, neck, and arms occurring between five and 7 days of the onset of symptoms.8 After the flu-phase, hemorrhagic manifestations occur in EVD leading to conjunctival hemorrhage, petechiae, and ecchymosis. Other non-common features include cough, dyspnea, and localized chest, abdominal, muscle, and joint pains.11 Some patients may recover at this stage while others progress into severe EVD. Massive bleeding usually occurs in severe disease, particularly in the gastrointestinal tract.12 Such patients are prone to massive fluid loss, electrolyte imbalance, shock, and multi-organ failure leading to a higher risk of mortalities.13
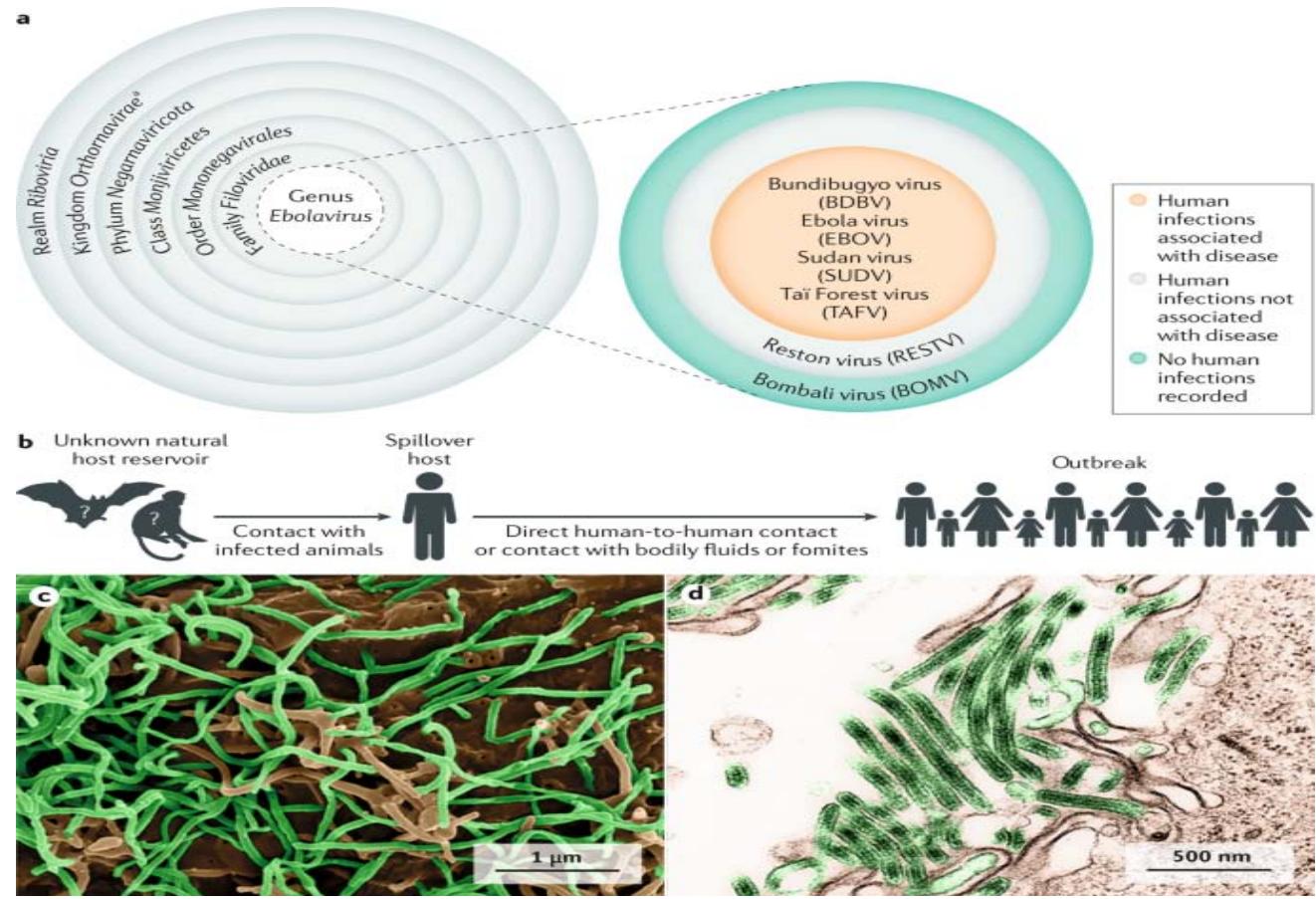 Figure 1: Epidemiology of Ebola Virus Disease https://www.nature.com/articles/s41572-020-0147-3/figures/
Taxonomy of the genus Ebola virus. Thus far, five ebolaviruses have been associated with human infections, and four of them have been identified as pathogens.
b) The natural reservoir host(s) of Ebola virus (EBOV) has (have) yet to be identified. Multiple data indicate a direct or indirect role of bats in EBOV ecology, but to date, EBOV has not been isolated.
c) Scanning electron microscopic (SEM) image of EBOV particles (green) budding from grivet cells.
d) Transmission electron microscopic (TEM) image of EBOV particles (green) budding from grivet cells.
Some scholars dwell on the need for prompt diagnosis of EVD to mitigate the wider spread of the outbreaks, especially in some sub-Saharan settings with weakened health systems. Given that the initial phase of EVD is indistinguishable from other prevailing ailments like malaria, typhoid, yellow fever, and dengue. Therefore, adequate case definition and a high index of clinical suspicion, especially in high-risk areas remain an effective modality of initial empirical diagnosis and outbreak containment.[14] In sub-Saharan Africa and elsewhere, laboratory confirmation of EVD is essential amidst myriads of high-risk pathogens, which could cause hemorrhagic fever. To confirm infection, Antigen-capture enzyme-linked immunosorbent assay (ELISA) testing, IgM ELISA, polymerase chain reaction (PCR), and virus isolation tests are deployed as diagnostic tests.[8,14] Due to its highly virulent nature, EVD's laboratory diagnosis should be performed in a well-equipped laboratory with up to bio safety level 4 biocontaminant facilities for viral culturing. Prompt case identification, early fluid care, and hospitalization improve the chances of survival in EVD patients. Regarding the prevention of EVD, there are two recommended vaccines. ERVEBO was licensed by the European Medicines Agency and the Food and Drug Administration in 2019 and is indicated for use in persons above12 months. It has a shelf life of three years. ERVEBO, rVSV-ZEBOV was developed against Zaire type Ebola virus. It made massive ring vaccinations of contacts and health workers possible during the 2016 epidemic in Guinea, and the 2018-19 outbreaks in the Democratic Republic of Congo.[14, 15] The vaccine has been approved in Burundi, Central African Republic, Côte d'lvoire, Democratic Republic of the Congo (DRC), Ghana, Guinea, Republic of the Congo, Rwanda, Sierra Leone, Uganda, and Zambia.[16] The second vaccine, the chimpanzee adenovirus serotype 3 vectored vaccine (ChAd3-EBO-Z), boosted with the MVA vaccines, Zabdeno/Mvabea is recommended for preventive vaccination in areas at lower risk for Ebola or areas neighboring an epidemic because the full regimen requires two doses administered 56 days apart.[16] However, there are concerns regarding the long-term side effects and immunogenicity of the vaccines. Besides, the vaccine is not effective against other strains, hence community engagement and surveillance systems, early case identification and management, and prompt disease preparedness remain the effective modalities of mitigating EVD outbreaks in sub-Saharan Africa.[17-20]
Despite the ravaging impacts of recurrent EVD epidemics in sub-Saharan Africa, there is dearth of a research highlighting the current epidemiological features of EVD. Some researchers have focused on the epidemiological attributes of EVD from global perspectives without much emphasis on contemporary sub-Saharan situations. $^{8,9\&14}$ Besides, only a handful of studies have attempted to calculate a case fatality ratio specifically for contemporary sub-Saharan Africa. For instance, a recent review deploying global data computed a grand case fatality ratio of EVD without a detailed recourse to contemporary sub-Saharan African realities. $^{21}$ A focus of this caliber is significant. It would help to reinforce the enormity of EVD in the region to continually advocate for community engagement, disease preparedness, and strengthening of health systems to redress recurrent high-risk pathogen outbreaks. Given the frequency of EVD outbreaks in sub-Saharan Africa, the importance of delineating the contemporary epidemiological attributes of EVD could not be overemphasized in the context of preventing the next pandemic. Hence, we set out to highlight the epidemiological characteristics of recent EVD outbreaks including the computation of the case fatality ratio of this highly virulent pathogen in sub-Saharan Africa.
## II. METHODOLOGY
We deployed mainly an narrative review and a measure of rapid systematic review for this study. We synthesize and analyze relevant existing studies on Ebola through a descriptive approach to arrive at laudable research conclusions. We searched PubMed, Google Scholar, and Web of Science for publications with the terms "Ebola", "Ebola virus", "Ebola transmission", "Ebola epidemiology", "Ebola virus disease", "Ebola pathogenesis", "Ebola diagnosis", and "Ebola treatment" in various combinations without any language limitations. The search covered the period from 1976, the year of the discovery of EVD, until January 2025. Review articles were cited when appropriate and additional sources were derived from selected journals via snowballing method. Research published in Google Scholar published since the 2014-16 West African outbreak was given particular attention. Research focusing extensively on other hemorrhagic fever was excluded from this review article. We excluded research articles on Reston and Bombali Ebolaivirus because they do not cause human infections. We provide an overview of Ebola by focusing on updating the reader on recent advances and controversies. Information from sources other than peer-reviewed journals has been evaluated but was not considered to add indispensable new details, so is not covered in this review article.
### a) Historical Epidemiology of Ebola Virus Disease
Filoviruses have probably existed for over 10 million years, however Marburg virus and Ebola virus departed from a common progenitor over 10,000 years ago.[22,23] In August 1967, the first reported cases of Marburg virus disease occurred in Marburg when laboratory workers were exposed to African green monkeys (Cercopithecus a ethiops) imported from Uganda.[24] Laboratories in Belgrade and Frankfurt that also received a consignment of green African monkeys reported sick staff after their exposures to the mammals. Consequently, nine deaths were recorded out of 37 cases.[25] The first cases of EVD were reported nine years later in the Democratic Republic of Sudan. On 27 June 1976, a man residing in a rural area but working in a factory in a township, Nzara, developed fever, headache, and chest pain.[26] Due to the severity and persistence of his symptoms after four days, he was admitted for acute care in a hospital, after which he developed bleeding from the nose and mouth, and bloody diarrhea on the second day at the hospital. He subsequently died on July 6 after infecting his coworkers via close contacts. The heightened human-human dispersal led to a devastating outbreak of EVD that killed 151 people out of 284 victims.[25]
The Democratic Republic of Congo (formerly Zaire) encountered the next outbreak of EVD between September and October 1976. The clinical manifestations of the index patient revealed other epidemiological features of EVD different from the previous cases in Sudan. The patient presented with malarial symptoms and was duly treated with intramuscular chloroquine. There was an initial resolution of his clinical signs and symptoms. Nevertheless, he developed a set of new clinical features within five days along with other patients who were admitted with him at Yambuku Mission Hospital (YMC) around the Ebola River.[27] They developed a severe sore throat, macula-papular rash, and varied gastrointestinal manifestations such as nausea, dysphagia, abdominal pain, and gastrointestinal hemorrhage. Despite the limited diagnostic protocols at the time, some patients developed non-icteric hepatitis, acute pancreatitis, and disseminated intravascular coagulopathy (DIC).[27] Hence, they were incorporated into the usual clinical manifestations of EVD. By the end of the epidemic in late October 1976,280 patients have died out of 380 identified cases.[25,27] Among the dead included women who attended ante-natal clinics and took injections at YMC. This gives credence to the theory that YMC staff used non-sterilized needles and syringes among patients, thereby facilitating the exchange of body fluids which promoted the rapid dispersal of EVD, and hence fatalities.
The Sudan ebolavirus virus and Zaire ebolavirus were isolated and ascribed correspondingly to the EBV epidemics in Sudan and Congo DRC. However, in 1989, the third strain, Restonebolavirus was isolated in cynomologus monkeys (Macaca fascicularis) imported from the Philippine Islands to Reston, Virginia, where they were intended for research purposes.[28] The virus has not been linked to any human infections and the swine has been recognized as its natural host.[29] In 1994, Tai forest ebolavirus, another species of ebolavirus was isolated in West Africa. An ethnologist conducting an autopsy on a dead chimpanzee at Parc National de parc in Tai, Cote D' Ivoire contracted EVD. She survived after spending two weeks in the hospital. In 2007, in Uganda, the fifth species, Bundibugyo ebolavirus triggered an outbreak killing 30 persons out of 116 infected patients. Between 1976 and 2013, there were 24 outbreaks of EVD, with most epidemics linked to Zaire ebolavirus and Sudan ebolavirus.[25] From the largest outbreak between 2014-2016 in West Africa to 2025, there have been eight outbreaks till 7 March 2025.
### b) Epidemiology of Ebola Virus Disease
This study concentrates on the epidemiological features of EVD in sub-Saharan Africa. We focus on the Ebola virus outbreaks from 2001 to 2025 for our contemporary era's analysis. However, before we delve into this review, we will show a detailed table of the epidemiology of EVD in the past era, from 1976-2000. It would ensure appropriate comparison with the epidemiological attributes of EVD in the $21^{\text{st}}$ century. Table 1 shows the epidemiological features of EVD from 1976 to 2000.
Table 1: Epidemiological Features of EVD between 1976 and 2000 in Sub-Saharan Africa
<table><tr><td>Year of Outbreak</td><td>Country of Affection</td><td>Ebola virus Species</td><td>Cases</td><td>Mortality</td><td>Case Fatality Ratio (CFR)</td></tr><tr><td>1976</td><td>Sudan</td><td>Sudan virus</td><td>284</td><td>151</td><td>53.2</td></tr><tr><td>1976</td><td>Congo DRC</td><td>Zaire virus</td><td>318</td><td>280</td><td>88.1</td></tr><tr><td>1977</td><td>Congo DRC</td><td>Zaire virus</td><td>1</td><td>1</td><td>100</td></tr><tr><td>1979</td><td>Sudan</td><td>Sudan virus</td><td>34</td><td>22</td><td>64.7</td></tr><tr><td>1994</td><td>Gabon</td><td>Zaire virus</td><td>51</td><td>31</td><td>60.8</td></tr><tr><td>1994</td><td>Cote D' Ivoire</td><td>Tai Forest virus</td><td>1</td><td>0</td><td>0.0</td></tr><tr><td>1995</td><td>Congo DRC</td><td>Zaire virus</td><td>315</td><td>254</td><td>80.6</td></tr><tr><td>1996</td><td>Republic of South Africa</td><td>Zaire virus</td><td>2</td><td>1</td><td>50.0</td></tr><tr><td>1996</td><td>Gabon</td><td>Zaire virus</td><td>91</td><td>66</td><td>72.5</td></tr><tr><td>2000</td><td>Uganda</td><td>Sudan virus</td><td>425</td><td>224</td><td>52.7</td></tr><tr><td>Total</td><td></td><td></td><td>1522</td><td>1030</td><td>67.8</td></tr></table>
Adapted and Modified from Izudi J, Bajunirwe F. Case fatality rate for Ebola disease, 1976-2022: A meta-analysis of global data. Elservier Journal of Infection and Public Health. 2024; 17: 25-34.
Table (1) displays the main epidemiological features of EVD outbreaks in sub-Saharan Africa. We could decipher that EVD outbreaks occur predominantly in Congo DRC, Sudan, and Gabon. The western and south African region were outliers regarding the prevalence of EVD epidemics. The Sudan and Zaire ebolavirus were responsible for most epidemics of EVD between 1976 and 2000. In all, there were ten outbreaks of EVD from 1976 to 2000, which resulted in 1030 fatalities out of 1522 patients. The case fatality ratio(CFR)spanned from 50 to 100, and the average CFR was 67.8. The Zaire ebolavirus had the highest case fatality ratio ranging from 50 to 100 percent compared to the Sudan ebolavirus with a CFR of 52.7 to 64.7 percent. Congo DRC had the highest CFR of 100 during the 1977 epidemic while the Republic of South Africa witnessed the lowest CFR in the 1996 outbreak. All the afflicted countries had a zoonotic spread of Ebola virus before human-human contacts predominated as the main transmission route. Table (2) displays the current EVD's epidemiological attributes between 2001 and 2025.
Table 2: Epidemiological Features of EVD between 2001 and 2025 in Sub-Saharan Africa
<table><tr><td>Year of Outbreak</td><td>Country of Affection</td><td>Ebola virus Species</td><td>Cases</td><td>Mortality</td><td>Case Fatality Ratio (CFR)</td></tr><tr><td>2001</td><td>Gabon</td><td>Zaire virus</td><td>65</td><td>53</td><td>81.5</td></tr><tr><td>2001</td><td>Congo DRC</td><td>Zaire virus</td><td>59</td><td>44</td><td>74.6</td></tr><tr><td>2003</td><td>Congo DRC</td><td>Zaire virus</td><td>178</td><td>157</td><td>88.2</td></tr><tr><td>2004</td><td>Sudan</td><td>Sudan virus</td><td>17</td><td>7</td><td>41.2</td></tr><tr><td>2005</td><td>Congo DRC</td><td>Zaire virus</td><td>12</td><td>10</td><td>83.3</td></tr><tr><td>2007</td><td>Congo DRC</td><td>Zaire virus</td><td>264</td><td>187</td><td>70.8</td></tr><tr><td>2007</td><td>Uganda</td><td>Bundibugyo virus</td><td>131</td><td>42</td><td>32.1</td></tr><tr><td>2008</td><td>Congo DRC</td><td>Zaire virus</td><td>32</td><td>15</td><td>46.9</td></tr><tr><td>2011</td><td>Uganda</td><td>Sudan virus</td><td>1</td><td>1</td><td>100</td></tr><tr><td>2012</td><td>Uganda</td><td>Sudan virus</td><td>17</td><td>7</td><td>41.2</td></tr><tr><td>2012</td><td>Congo DRC</td><td>Bundibugyo virus</td><td>36</td><td>13</td><td>36.1</td></tr><tr><td>2014</td><td>Congo DRC</td><td>Zaire virus</td><td>69</td><td>49</td><td>71.0</td></tr><tr><td>2014</td><td>Senegal</td><td>Zaire virus</td><td>1</td><td>0</td><td>0.0</td></tr><tr><td>2014</td><td>Guinea</td><td>Zaire virus</td><td>3814</td><td>2544</td><td>66.7</td></tr><tr><td>2014</td><td>Liberia</td><td>Zaire virus</td><td>10678</td><td>4810</td><td>45.0</td></tr><tr><td>2014</td><td>Sierría Leone</td><td>Zaire virus</td><td>14124</td><td>3956</td><td>28.0</td></tr><tr><td>2014</td><td>Mali</td><td>Zaire virus</td><td>8</td><td>6</td><td>75.0</td></tr><tr><td>2014</td><td>Nigeria</td><td>Zaire virus</td><td>20</td><td>8</td><td>40.0</td></tr><tr><td>2017</td><td>Congo DRC</td><td>Zaire virus</td><td>8</td><td>4</td><td>50.0</td></tr><tr><td>2018</td><td>Congo DRC</td><td>Zaire virus</td><td>3524</td><td>2320</td><td>65.8</td></tr><tr><td>2020</td><td>Congo DRC</td><td>Zaire virus</td><td>130</td><td>55</td><td>42.3</td></tr><tr><td>2021</td><td>Guinea</td><td>Zaire virus</td><td>23</td><td>12</td><td>52.5</td></tr><tr><td>2021</td><td>Congo DRC</td><td>Zaire virus</td><td>23</td><td>15</td><td>65.2</td></tr><tr><td>2022</td><td>Congo DRC</td><td>Zaire virus</td><td>6</td><td>6</td><td>100</td></tr><tr><td>2022</td><td>Uganda</td><td>Sudan virus</td><td>164</td><td>55</td><td>33.5</td></tr><tr><td>2025</td><td>Uganda</td><td>Sudan virus</td><td>14</td><td>4</td><td>28.6 (As of 7 March 2025)</td></tr><tr><td>Total</td><td></td><td></td><td>33, 418</td><td>14,380</td><td>43.0</td></tr></table>
Table (2) highlights the epidemiological features of EVD in the contemporary era, 2001-2025 in sub
Saharan African context. Likewise in the previous EVD era between 1976 and2000, DRC Congo, Uganda and Gabon witnessed the lion's share of EVD epidemics in sub-Saharan Africa.DRC Congo particularly witnessed EVD epidemics, 12 times out of 26 between 2001 and 2025 (46 percent). Apart from the 2014-2016 large EVD epidemics in West African clime, central and southern Africa were particularly afflicted with recurrent EVD outbreaks. Zaire ebolavirus and Sudan ebolavirus are the predominant causes of EVD epidemics in sub-Saharan Africa. Zaire virus was responsible for EVD on
19 occasions out of 26 epidemics encountered between 2001 and 2025 (73 percent). Contrastingly to the previous era, Zaire ebola virus had a casefatality ratio of 28 to 100 percent, which compares with the case fatality ratio of the Sudan ebolavirus ranging from 28.6 to 100 percent.
As shown in Table (1), EVD outbreaks particularly occurred in the central African belt of DRC Congo and Uganda. West African countries such as Guinea, Liberia, Mali, Sierra Leone, Senegal, and Nigeria encountered the main incursion of Ebola epidemics only from 2014 to 2016, and they remained the largest outbreaks of EVD claiming over 11,300 lives out of over 28,600 cases.[21] The CFR during the West African outbreaks ranged from 28 to 75 percent while the overall case fatality ratio between 2001 and 2025 spanned from 28 to 100 percent. From 2017 to 7 March 2025, Table (2) shows that sub-Saharan Africa witnessed 3,892 cases including 2,471 fatalities with a CFR of 64 percent. Table (2) highlights 26 epidemics of EVD resulting in a total of 14,380 deaths out of 33,418 cases with an average CFR of 43.0 between 2001 and 2025. It is in sharp contrast to ten EVD epidemics occasioning 1030 mortalities out of 1522 sufferers and an average CFR of 67.8 from 1967 and 2000.
## III. DISCUSSIONS
Congo DRC has witnessed the lion's share of EVD outbreaks since its first epidemics in 1976. From our analysis, 46 percent of EVD epidemics occurred in DRC Congo between 2001 and 2025. The reasons are not far-fetched. Congo DRC formerly Zaire, especially the eastern area has been bedeviled with a long history of wars, political instabilities, and concomitant social displacements.[31] It has the highest rate of internally Displaced Persons (IDPs) globally.[32] This has continuously occasioned populations' encroachment into the sanctuaries of wild games and bats. Consequently, they are exposed to known reservoirs of ebolavirus. The weakened health system, frequent migration of people from troubled areas, and poor health infrastructures have predisposed Congo DRC to frequent outbreaks of filoviral diseases including MVD and EVD. On the regional front, the risk of another EVD outbreak is even greater soon because of the present socio-political unrest. As of 26 February 2025, the M23 faction has captured Goma and Bukavu in eastern Congo DRC with the possibilities of heightened hostilities and its attendant humanitarian crisis spreading around central Africa.[33]
Regarding other surrounding countries, Uganda and Sudan as well as most sub-Saharan states are blessed with lush and thick forests that harbor fruit bats, which are known as the major dispersal of fruits and enhancer of forestation through their extensive migration.[33] It is noteworthy that the principally hot and humid climate of most sub-Saharan states favors the breeding and migration of fruit bats. $^{34}$ Their migratory impacts to nearby countries with weakened health systems and poor surveillance, especially in predominantly rural settings enhance recurrent EVD epidemics. Concerning the 2014-16 Ebola epidemics in West Africa, the consumption of wild animals, cultural practices regarding dead bodies and mistrust of Western medicine, and poor surveillance and disease preparedness facilitated the swift dispersal of EVD causing huge mortalities in Liberia, Guinea, and Sierra Leone. $^{35}$
Regarding the causative strains of EBV, Zaire virus and Sudan virus are the predominant Ebola virus species causing over 90 percent of outbreaks between 2001 and 2025. Zaire ebola virus is regarded as the primary cause of EBV. It has the highest mortality rates among the six strains of ebolavirus due to its high virulence and transmissibility rate among humans after initial zoonotic transmission.[36] However, this piece reveals comparable case fatalities of Sudan's ebola virus in the contemporary era. This might be due to increasing urbanization and exposure of humans to other virulent strains of ebolavirus from wild games and bats.
In this review, we discovered a case fatality rate of 28 to 100 percent and an average of 43.0 percent CFR between 2001 and 2025. This high fatality rate is consistent with W.H.O data that stipulates a CFR range of 25 to 90 percent, and an average CFR of 50 percent from past outbreaks depending on response and peculiarities. Our average value of 43.0 CFR is more consistent with W.H.O data and a similar reviews that calculated an average CFR of 43.8 percent for all EVD outbreaks since 1976. This is because we collated data and computed the CFR from past EVD epidemics like W.H.O and previous reviews on EVD, hence the correlation of epidemiological data. However, the average CFR of EVD between 2001 and 2025, 43.0 percent, is comparatively lower than 67.8 percent obtained from 1976 to 2000. It might be due to marginal improvement in disease preparedness, clinical suspicion, laboratory testing, and community awareness and engagement. For instance, in the Ebola outbreak in Uganda, in 2022, 55 mortalities were recorded out of 164 and a lower CFR rate of 38.6 percent was reported. The index cases were promptly identified, contacts were traced, and the victims had hygienic burial to mitigate escalation to wider epidemics. The 2025 Ebola outbreak in Uganda was even better managed and effectively contained so far. Within days of announcement of Sudan ebola virus outbreak, the Ugandan Ministry of Health had launched a trial of candidate Ebola vaccine and vaccinated up to 264 contacts. The distribution of about 2000 doses of antiviral drugs effective in stemming the viral loads in EVD patients is ongoing in Uganda.
## IV. CONCLUSIONS AND RECOMMENDATIONS
Given the high virulent and transmissibility rate of EVD, its capability of transcending regional boundaries to escalate to a devastating pandemic like COVID-19 remains plausible. The devastating mortalities occasioned by the Zaire ebolavirus in West Africa from 2014-16 is a testament to this reality. Between 2017 and 7 March 2025, there were eight ebola epidemics in sub-Saharan Africa. They killed 2,471 people out of 3,892 cases with a CFR of 64 percent. The recurrent rate of EVD outbreaks is a grave public health issue in the postmodern era. Hence the world needs to mitigate the pandemics occasioned by high-risk pathogens such as ebolavirus. In this light, this work chronicled the historical trajectory of EVD, computed and compared key epidemiological values in sub-Saharan Africa between 1976 and 2000 to and 2001-2025. We discovered there were ten EVD epidemics from 1976 to 2000, in contrast to 26 EVD outbreaks encountered between 2001 and 2025 in sub-Saharan Africa. DRC Congo witnessed the lion's share of EVD outbreaks since 1976 due to its recurrent political turmoil, massive socio-cultural displacements, weakened healthcare in infrastructures. There was a preponderance of Zaire ebolavirus in the causation of EVD from 1976 to 2025. However, its dominance as a cause of EVD became more pronounced in the present era. It accounted for 73 percent of outbreaks between 2001 and 2025. We found an average CFR of 43.0 percent for EVD outbreaks between 2001 and 2005 as opposed to a higher CFR of 67.8 percent from 1976 to 2000. The reasons alluded for this difference are marginal improvement in disease preparedness, surveillance, case testing and vaccinations as well as community education and promotion in contemporary settings.
To mitigate recurrent EVD epidemics in sub-Saharan Africa, there should be heightened community education about common reservoirs, early symptoms and general management of EVD. This is imperative given the indistinguishable nature of early EBV from common tropical diseases such as malaria, typhoid, gastroenteritis, and yellow fever. Some evidence proves transmission via care giving and burial preparations in cultural contexts of some sub-Saharan African countries.[39] Hence, there is a continuing need for community education regarding hygienic burials and prevention of undue exposure to Ebola dead bodies to prevent escalations of infections. Local populations and tourists should be abreast of transmission reservoirs such as fruit bats and wild animals, and efforts should be made to minimize contact with them and their discharges. Miners and tourists should be equipped with hand gloves, face masks, and protective clothing when they visit mines and caves.
Even though fruit bats have been recognized as the putative reservoirs of the Ebola virus, it is astounding to highlight that the virus has never been isolated in them. $^{40,41}$ Therefore, we do not completely understand how fruits bats transmit it to other animals and humans. To comprehend these dynamics, we must intensify our studies on fruit bats, especially of the order Chiroptera that serve as the intermediate hosts of ebolavirus. Bat surveillance should be strengthened to understand the migration, breeding, and transmission mechanism of Chiroptera among themselves and between chimpanzees, monkeys, wild games and humans. This would further enhance our knowledge concerning the epidemiology of EVD to curtail its dispersal to humans and prevent its recurrent outbreaks in contemporary sub-Saharan Africa.
The predominantly rural settings of most sub-Saharan countries are characterized by overwhelming poverty and ignorance of dwellers, and poor access to health care. Besides, there is a level of mistrust in government health institutions due to inadequate manpower and laboratory capacities. To adequately manage the contagion, a quick and correct laboratory diagnosis is imperative. The early infections are detected through serological assays detecting immunoglobulin M via enzyme-linked immunosorbent assay (ELISA). The confirmation of diagnosis of EVD is mainly premised on detecting viral RNA in blood samples using reverse transcription-polymerase chain reaction (RT-PCR).[42] Many sub-Saharan climates, especially the rural and remote areas lack these laboratory capacities in their healthcare system. Therefore, the need for improvement of health funding and strengthening of disease surveillance mechanisms in most economies could not be overstated. It behooves African Union as well as the United Nations to find lasting solutions to recurrent wars to curb social and health crises in the DRC Congo and the surrounding countries. This would go a long way in stemming recurrent EVD outbreaks occasioned by social dislocations of people with concomitant exposure to reservoirs of ebola virus.
There is no definitive treatment for EBV except supportive management. However, the WHO has approved the deployment of two monoclonal antibodies for treating EBV, namely mAb114 (Ansuvimab; Ebanga) and REGN-EB3 (Inmazeb).2 They have been shown to reduce the production of anti-Ebola virus antibodies in survivors of the disease, which could predispose to reinfection or reactivation.43 Regarding the prevention of EVD and reduction of frequent outbreaks, ERVEBO and Zabdeno/Mvabea are two recognized vaccines against Zaire ebola virus. However, they are only effective against Zaire ebola virus limiting their use for EVD caused by other strains. Other research aimed at discovering vaccines for other strains is ongoing. However, there are fears regarding the suitability and safety of the existing Ebola vaccines on the long term. In the future, we foresee the discovery of vaccines for othere bolavirus species and their deployment in vulnerable areas in sub-Saharan Africa. Apart from vaccines, community education, early diagnosis, and commencement of prompt management of EVD cases are effective in reducing its fatalities and curbing the swift dispersal of ebolavirus. Then, early hospitalization, early fluid and electrolyte replacement, oxygen care, blood transfusion, and other supportive treatment remain the mainstay of management in the present era and the nearest future. They remain viable and effective modalities of reducing mortalities due to EVD and mitigating its escalation to a cataclysmic pandemic.
A Nanbo,S Watanabe,P Halfmann,Y Kawaoka (2013). The spatio-temporal distribution dynamics of Ebola virus proteins and RNA in infected cells.
A Kadanali,G Karagoz (2015). An Overview of Ebola Virus Disease.
Suresh Rewar,Dashrath Mirdha (2014). Transmission of Ebola Virus Disease: An Overview.
M Broadhurst,Tim Brooks,Nira Pollock (2016). Diagnosis of Ebola Virus Disease: Past, Present, and Future.
B Zawilińska,M Kosz-Vnenchak (2014). General introduction into the Ebola virus biology and disease.
Pierre Nouvellet,Tini Garske,Harriet Mills,Gemma Nedjati-Gilani,Wes Hinsley,Isobel Blake,Maria Van Kerkhove,Anne Cori,Ilaria Dorigatti,Thibaut Jombart,Steven Riley,Christophe Fraser,Christl Donnelly,Neil Ferguson (2015). The role of rapid diagnostics in managing Ebola epidemics.
A Leligdowicz,Fischer Wa 2nd,T Uyeki (2016). Ebola virus disease and critical illness.
T West,Von Saint André-Von Arnim,A (2014). Clinical presentation and management of severe Ebola virus disease.
Luke Hunt,Ankur Gupta-Wright,Victoria Simms,Fayia Tamba,Victoria Knott,Kongoneh Tamba,Saidu Heisenberg-Mansaray,Emmanuel Tamba,Alpha Sheriff,Sulaiman Conteh,Tom Smith,Shelagh Tobin,Tim Brooks,Catherine Houlihan,Rachael Cummings,Tom Fletcher (2015). Clinical presentation, biochemical, and haematological parameters and their association with outcome in patients with Ebola virus disease: an observational cohort study.
Denis Malvy,Anita Mcelroy,Hilde De Clerck,Stephan Günther,Johan Van Griensven (2019). Ebola virus disease.
Pierre-Stéphane Gsell,Anton Camacho,Adam Kucharski,Conall Watson,Aminata Bagayoko,Séverine Nadlaou,Natalie Dean,Abdourahamane Diallo,Abdourahmane Diallo,Djidonou Honora,Moussa Doumbia,Godwin Enwere,Elizabeth Higgs,Thomas Mauget,Diakite Mory,Ximena Riveros,Fofana Oumar,Mosoka Fallah,Alhassane Toure,Andrea Vicari,Ira Longini,W Edmunds,Ana Henao-Restrepo,Marie Kieny,Sakoba Kéïta (2017). Ring vaccination with rVSV-ZEBOV under expanded access in response to an outbreak of Ebola virus disease in Guinea, 2016: an operational and vaccine safety report.
(2023). World Health Organization expedites trials for potential Ebola vaccines.
S Okware,F Omaswa,A Talisuna,J Amandua,J Amone,P Onek,A Opio,J Wamala,J Lubwama,L Luswa,P Kagwa,T Tylleskar (2015). Managing Ebola from rural to urban slum settings: experiences from Uganda.
G Caleo,J Duncombe,F Jephcott (2018). The factors affecting household transmission dynamics and community compliance with Ebola control measures: a mixed-methods study in a rural village in Sierra Leone.
T Shoemaker,S Balinandi,A Tumusiime (2018). Impact of enhanced viral haemorrhagic fever surveillance on outbreak detection and response in Uganda.
S Funk,I Ciglenecki,Tiffany (2017). The impact of control strategies and behavioural changes on the elimination of Ebola from Lofa County, Liberia.
J Izudi,F Bajunirwe (2024). Case fatality rate for Ebola disease, 1976-2022: A meta-analysis of global data.
Derek Taylor,Robert Leach,Jeremy Bruenn (2010). Filoviruses are ancient and integrated into mammalian genomes.
Y Suzuki,T Gojobori (1997). The origin and evolution of Ebola and Marburg viruses.
M Balter (2000). Emerging diseases. On the trail of Ebola and Marburg viruses.
Michael Murray (2015). Ebola Virus Disease.
(1978). Tough challenges for testing Ebola therapeutics.
T Muyembe,M Kipasa (1978). Ebola haemorrhagic fever in Kikwit, Zaire.
P Jahrling,T Geisbert,E Johnson,C Peters,D Dalgard,W Hall (1990). Preliminary report: isolation of Ebola virus from monkeys imported to USA.
R Barrette,S Metwally,J Rowland,L Xu,S Zaki,Nichol St (2009). Discovery of swine as a host for the Reston ebolavirus.
L Schnirring (2025). Second Cluster recorded in Uganda's Ebola Sudan Outbreak.
O Kavulikirwa,F Sikakulya (2022). Recurrent Ebola outbreaks in the eastern Democratic Republic of the Congo: A wake-up call to scale up the integrated disease surveillance and response strategy.
Augustine Avwunudiogba,Elisha Dung (2021). The Spatial Distribution of Internally Displaced Persons (IDPs) in Africa.
Prince Muzuva,Aaram Gwiza,Vain Db Jarbandhan (2025). “Kagametizing” the DRC conflict: Dissecting historical connotations and influence of the invisible hands.
S Languon,O Quaye (2019). Filovirus Disease Outbreaks: A Chronological Overview.
Hui-Ju Han,Hong-Ling Wen,Chuan-Min Zhou,Fang-Fang Chen,Li-Mei Luo,Jian-Wei Liu,Xue-Jie Yu (2015). Bats as reservoirs of severe emerging infectious diseases.
T Kamorudeen,K Adedokun,A Olarinmoye (2020). Ebola outbreak in West Africa, 2014 -2016: Epidemic timeline, differential diagnoses, determining factors, and lessons for future response.
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Between 2017 and 7 March 2025, Ebola virus disease occasioned eight outbreaks killing 2,471 people out of 3,892 cases in sub-Saharan Africa. Given the pandemic capacities of the largest Ebola epidemics that occurred in West Africa from 2014-2016 and the socioeconomic catastrophe wrought by the COVID-19 pandemic, all hands must be on deck to mitigate recurrent Ebola outbreaks. Hence, this piece explores the historical trajectory of Ebola virus disease and its epidemiological attributes in the contemporary era. We deployed a narrative review of relevant articles on the Ebola virus disease from its inception in 1976 to 2025. Between 2001 and 2025, 26 outbreaks of EVD occurred in sub-Saharan Africa, with an average case fatality rate of 43 percent. We discovered that 46 percent of the epidemics occurred in DRC Congo, and Zaire Ebola strains were responsible for 73 percent of outbreaks.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.