Lactobacilli play a fundamental role in maintaining health and preventing infections of the female urogenital tract. Complementary use of vegetal extracts selected for their ethnopharmacological characteristics and conventional uses for the different conditions in the human body as an adequate alternative therapy for the restoration of the vaginal microbiome has emerged. Compatibility of phytoextracts with lactobacilli for the design of phytobiotic formulas was determined previously. Safety of selected combinations by the intravaginal (i.va.) administration in a murine model was evaluated to determine if some type of adverse effect was produced in the host. Lactobacillus gasseri CRL1320, Limosilactobacillus reuteri CRL1324, Ligilactobacillus salivarius CRL1328 and Lacticaseibacillus rhamnosus CRL1332 combined with Hamamelis-virginiana, Amaranthus-muricatus and Smilax-áspera, were inoculated individually or combined in mice vagina (7 daily doses).
## I. INTRODUCTION
Lactobacilli play a fundamental role in the urogenital tract by maintaining health or preventing infections through different mechanisms of action, demonstrated by a wide diversity of publications (Lazarenko et al., 2012; Karlsson et al., 2012; Wagner and Johnson et al., 2012; De Gregorio et al., 2014, 2015, 2016, 2019; Nader and Juarez-Tomas 2015;
Nader Macías et al., 2021; Mashatan et al., 2023; Szczerbiec et al., 2024; Gupta et al., 2024). Plants extracts are applied to treat different pathologies in human and animals, which is one of the reasons to support their selection, supported by their ethnopharmacological characteristics to be used in urogenital tract infections (UGTI) prevention or treatment (Argentine Pharmacopoeia; Palmeira-de-Oliveira et al., 2015; Flower et al., 2016; Montorsi et al., 2016; Aziz y col., 2017; Moreno et al., 2018; Marchesi et al., 2020). Different products were applied for UGTI treatment, most of them derived from natural products, given the requirement of new therapies to prevent and treat chronic infections (Palmeira-de-Oliveira et al., 2013). In recent years, the frequent use of antimicrobials (such as antibiotics, antimicotics, and antivirals) is constantly questioned due to the appearance of resistant strains (Falagas et al., 2006; Flores-Mireles et al., 2015; Karam et al., 2019) which support the search for alternatives therapies or strategies to prevent or treat female urogenital infections, and to restore the microbiota of vaginal tract.
"Probiotics" are defined as "live microorganisms, when administered in adequate amounts, evidence a beneficial physiological effect on the consumer" (Hill et al., 2014). "Pharmabiotics" are "living or dead microorganisms and their microbial constituents and metabolites that can beneficially interact with the host" (Shanahan et al., 2009). "Phytobiotic" formulas refer to the "combination of plant extracts with probiotic microorganisms to maintain or prevent health" (NaderMacias and Juarez-Tomás et al., 2015). "Phytoextracts" or "Vegetal extracts" are the substances obtained by maceration process of plants in $40\%$ alcohol for medicinal use, approved by pharmacopoeias (Argentine Pharmacopoeia Method). Despite the fact that lactobacilli are generally recognized as safe (Generally Regarded as Safe, GRAS) by international organizations (FAO/WHO, 2012), and plant extracts which different applications in humans are described in pharmacopoeias (Argentine and European Pharmacopoeias) there is an imperative requirement to determine their safety and innocuity. It is of high importance to demonstrate that there is not production of adverse effects in animal models, before advancing in the evaluation of i.va. formulations in human clinical trials (Falagas et al., 2007; Nader-Macias et al., 2008;
2021; Alfaro et al., 2013; Silva et al., 2023). Selection of strains and extracts was also performed to further design phytobiotic formulas aimed to treat, prevent or maintain the human vaginal health (Mishra et al., 2018).
Safety of vaginal lactobacilli was determined previously, supported by requirements established for the design of probiotic formulas for human beings. Intraurethral and i.va. administration of vaginal lactobacilli was applied in a murine experimental model evidencing their persistence in the urogenital tract, absence of adverse effects, protection against Staphylococcus aureus, Streptococcus agalactiae, Escherichia coli and Candida albicans challenges, and immune system modulation (Silva-Ruiz et al., 2004; Zárate et al., 2007; De Gregorio et al., 2012; 2015; 2016; 2019; Leccese-Terraf et al., 2015). Experimental animal model in BALB/c mice was used for its small size, easy manipulation and short reproductive cycle. Safety of lactobacilli was also demonstrated in phase I trial in healthy women (De Gregorio et al., 2020). Our research group demonstrated that the i.va. administration of functional nanofibers was safe in murine models, producing a viable lactobacilli increase and promoting their permanence in the murine vaginal tract (Silva et al., 2023).
The physiological characteristics of the murine vaginal tract are different from those of women, mainly in the neutral vaginal pH, the low numbers of lactobacilli in the autochthonous microbiota and the length and characteristics of the sexual cycle (Patras et al., 2013). McLean et al. (2012) described a simple and noninvasive protocol to determine the estrous cycle stage of female mouse without altering its reproductive cycle, similar to the one used in this work. Murine models success described by numerous scientists to evaluate the vaginal tract is useful to predict the expected behaviour in human beings (Silva-Ruiz et al., 2001; Zárate et al., 2009; Muench et al., 2009; Spurbeck and Arvidson 2011; Joo et al., 2011; Joo et al., 2012; Patras et al., 2013; De Gregorio et al., 2014; 2015; 2016; 2019).
Phytobiotics were formulated previously in our research group with 30 different beneficial vaginal lactobacilli (BVL) strains combined with phytoextracts (selected by their application and characteristics related with vaginal health improvement) to produce a synergic or complementary pharmacological effect (Marchesi et al., 2020). However, no safety assays were published referred to formulas designed with vegetal extracts and probiotic bacteria combined, administered by the vaginal way. Thus, the aim of this work was to evaluate the safety of phytobiotic formulas designed with beneficial lactobacilli and vegetal extracts combined, in order to define the permanence of the strains, and if some type of adverse reactions was evidenced in the murine experimental model.
## II. RESULTS AND DISCUSSION
### a) Quantification of Viable Lactobacilli in Vaginal Washing
The results obtained when evaluating the number of viable lactobacilli from vaginal washing (v.w.) of mice inoculated with the different phytobiotics did not show significant differences $(p > 0.05)$ referred to mice inoculated only with BVL (control), indicating that a stimulatory or inhibitory effect by the phytoextracts on the colonization capability of lactobacilli was not produced (Fig.1). The comparison of the i.va. inoculation of the four BVL strains, showed higher viable cell numbers of L. gasseri CRL1320 $(10^{6}\mathrm{CFU / ml}$ v.w.), while L. reuteri CRL1324 and L. salivarius CRL1328 were in a lower value $(10^{2 - 3}\mathrm{CFU / ml})$. In control mice, viable lactobacilli were not detected (in MRS-pH-5.5, selective medium) suggesting that the vaginal isolated lactobacilli came from the exogenous administration. Mice receiving phytobiotics-L. rhamnosus CRL1332showed similar values than L. gasseri CRL1320. L. salivarius CRL1328+phytocompounds mice produced a higher number of viable lactobacilli, but not significant compared with the strain without vegetal extracts. At the end, L. salivarius and L. rhamnosus demonstrated a higher colonization in mouse vagina when administered in phytobiotic formulas. These results were different to those obtained from in vitro assays (Marchesi et al., 2020) where some vegetal-extracts showed a stimulatory or inhibitory effect on BVL. Differences between in vitro and in vivo assays were also observed in BVL interaction with St. agalactiae (De Gregorio et al., 2014). These differences support the requirement to apply different criteria for probiotic selection, including both in vitro an in vivo safety assays (Nader-Macias and Juarez, 2015; Nader-Macias et al., 2021). Viable lactobacilli quantification in v.w. were similar to those obtained previously when the optimal i.v.a. dose of L. reuteri CRL1324 was determined (De Gregorio et al., 2015).
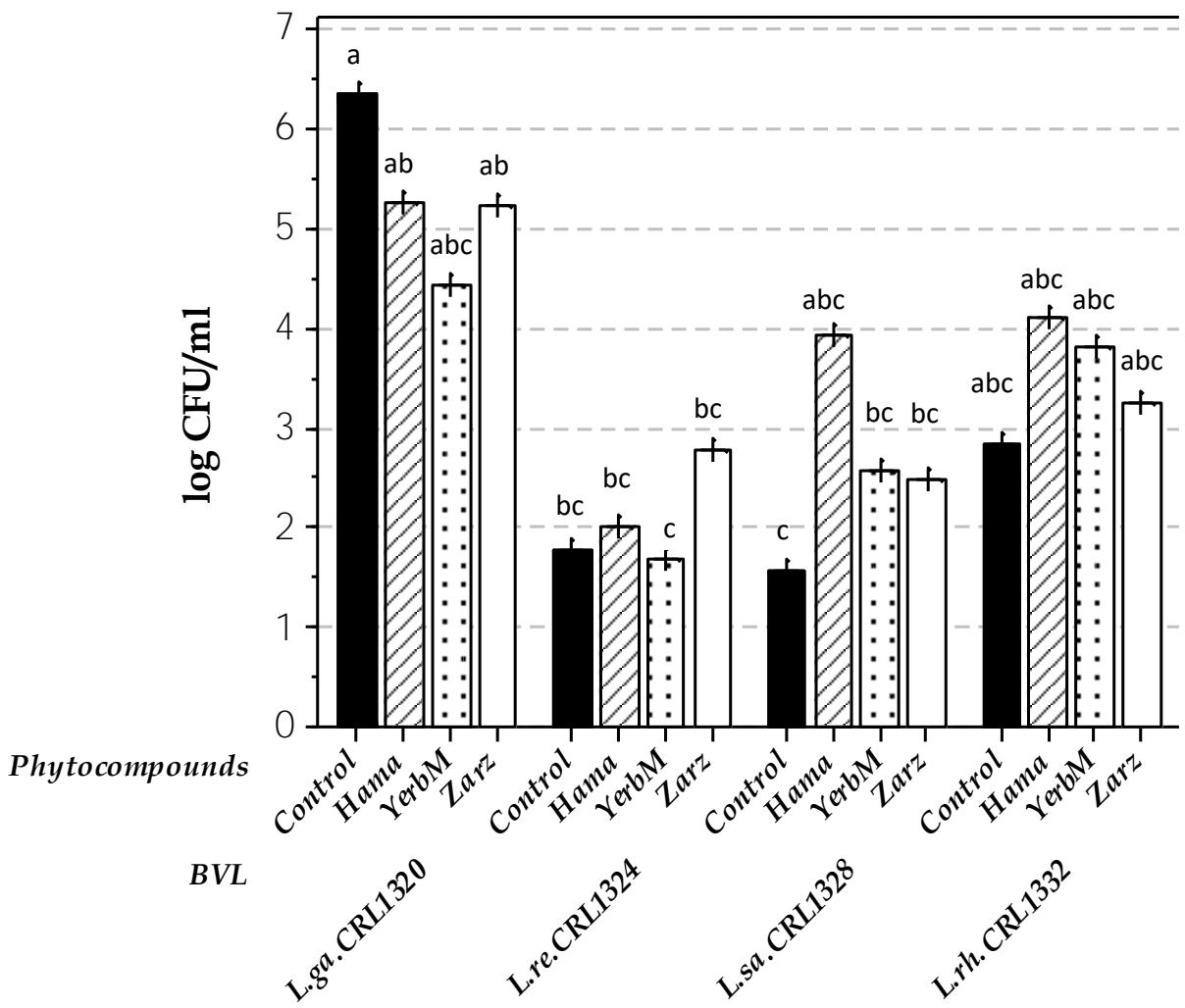
Fig.1: Quantification of viable BVL in murine v.w. of mice inoculated with lactobacillivegetal extracts: Control (blackbar:Hama:Hamamelis (oblique-bar:N), YerbM:Yerba meona (dot-bar:N) and Zarz:Zarzaparilla (whitebar:N). The results represent the log CFU/ml mean values of L. gasseri CRL1320, L. reuteri CRL1324, L. salivarius CRL1328, and L. rhamnosus CRL1332 ± standard error. Significant differences in the number of each BVL strain and their combination with extracts are indicated by different letters $(p < 0.05)$.
### b) Vaginal Cytology
Intravaginal administration of phytobiotic formulas did not produce adverse effect or inflammatory response in the murine experimental model. May Grünwald-Giemsa vaginal smears from phytobiotics, BVL or vegetal extracts inoculated mice obtained, by optical microscopy are included in Fig.2.A. Cytological evaluation evidenced absence of adverse effect at this level. All samples showed similar patterns to control mice, indicating pseudo-estrous state induced by hormonal inoculation, characterized by the presence of keratinized epithelial cells (irregular shape of scales) and absence of nucleated epithelial cells and leukocytes (which are indicative of an inflammatory or adverse response). Safety of BVL in the urogenital tract was also demonstrated previously. Silva de Ruiz et al. (2003) administered intraurethrally L. fermentum CRL1508 in a murine model with no adverse effect or significant changes in the organs (kidney, ureter, bladder or
urethra) at the structural and ultra-structural level, indicating its safety. De Gregorio et al. (2012) evidenced that the administration of five different human BVL to BALB/c-mice for 4 163 days did not produce adverse effects in cytological and immunological assays. Zarate et 164 al. (2009) also showed the protection against S. aureus challenge by the i.va. 165 administration of human BVL strains with no adverse effects. Recently, Silva et al. (2023) demonstrated the absence inflammatory response at the cytological level after the i.va. administration of nanofibers with L. rhamnosus CRL1332 immobilized in murine model.
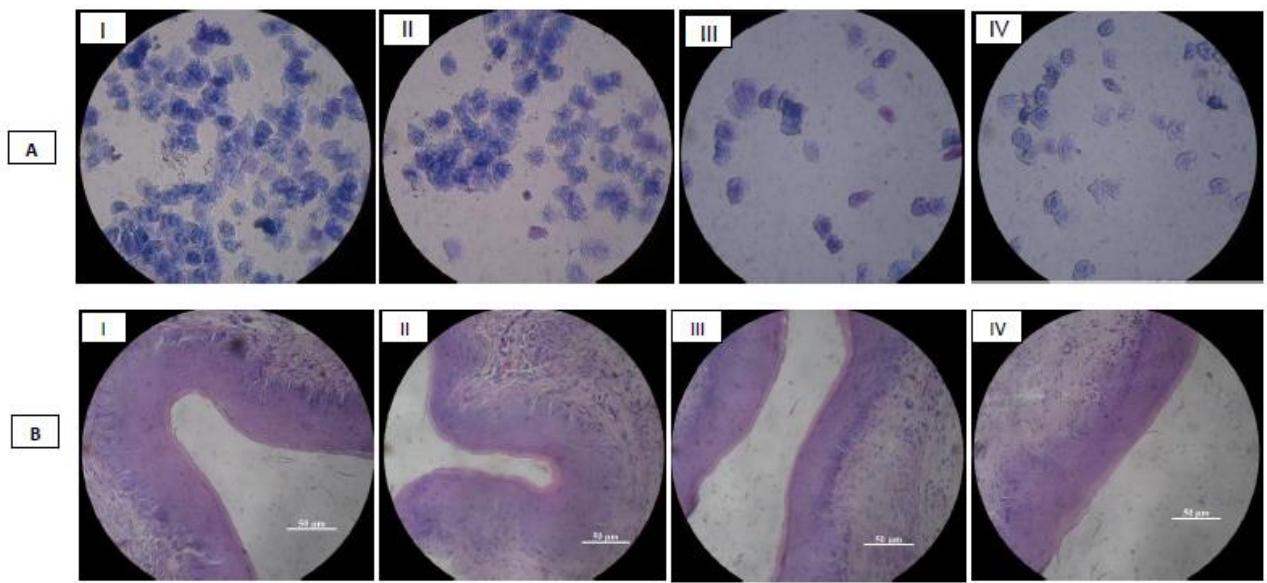
Fig. 2: Vaginal Cytology (A) and Histology (B) images of BALB/c mice after 171 intravaginal administration with different phytobiotics. A-I: L. rhamnosus 172 CRL1332+Hamamelis; A-II: Hamamelis; A-III: L. reuteri CRL1324+Hamamelis; A173 IV: Control. B-I: L. gasseri CRL1320+Hamamelis; B-II: Hamamelis; B-III: L. gasseri 174 CRL1320; B-IV: Control.
### c) Vaginal Histology
Microscopy evaluation is useful to facilitate diagnosis, as some authors claim. Donders 178 et al. (2019) recommend the use of microscopy for an exact observation of vaginal 179 epithelium condition, and helps to define different therapeutic alternatives. Histological 180 structure of vaginal tract of mice i.va. inoculated with phytobiotics, phytocompounds or 181 lactobacilli showed normal lamina propia characteristics, multilayer epithelium, 182 keratinized epithelial cells, indicating the pseudo-estrous state (Fig. 2. B). The observed 183 pattern was similar to control mice (Fig. 2. B-IV), with absence of inflammatory response 184 in murine tissue.
Zarzaparilla has antifungal, antiseptic and diuretic uses; Hamamelis has antiviral, antiseptic and anti-inflammatory activities, while Yerba meona evidenced diuretic, antitumor, drastic purging, warts, herpes, depurative, and other applications, supporting their selections to evaluate the beneficial properties when used in combination with BVL (Argentine-Pharmacopoeia; European-Pharmacopoeia; Theisen et al., 2014; Qi et al., 2017; Marchesi et al., 2020). Different scientists published the effect of plant derivatives administered orally or intravaginally in women suggesting a variety of effects and mechanisms of action. Moraes et al. (2012) demonstrated the efficacy and safety of Mentha crispa as a suitable alternative in the therapy of Trichomonas vaginalis infections. Satthakarn et al. (2015) administered Houttuynia cordata aqueous-extract in women urogenital tract determining an increase of cells participating in vaginal immune response. Espino et al. (2019) showed the complementary antifungal effect when administering L.
plantarum cream+two extracts in candidiasis treatment in in vitro protocols. No publications were detected referred to protocols of i.va. administration of phytobiotics in a murine experimental models. Recently, Miranda and Nader-Macias (2024) showed that the i.va. administration of probiotic and phytobiotic formulations to pregnant female cows at pre and postpartum increased significantly the number of lactic acid bacteria with no adverse local and systemic effects in cows.
### d) Ultrastructure of Murine Vagina
Ultrastructure of murine vagina inoculated with phytobiotics was characterized by a keratinized epithelium with anucleated polystratified cells, supported on the lamina propia with anucleated cells. Microphotographs of the vaginal tract of lactobacilli and phytocompounds i.va. inoculated mice did not evidence modifications, being similar in experimental group (L. salivarius CRL1328+Hamamelis) (Fig.3.A,B) and control mice (Fig. 3. G,H). Absence of inflammatory response, ultrastructure maintenance, and no other cells participating in inflammation indicate no adverse effect. Normal polystratified murine vaginal epithelium was evidenced. Mice inoculated with phytobiotics and lactobacilli showed bacteria or bacilli close or in contact with the epithelial cells surface (black arrows). These bacteria were not detected in mice i.va. inoculated only with phytoextracts or control animals. Therefore, it could be suggested that the bacteria evidenced in vagina of mice are those i.va. inoculated for 7-days, with no adverse effects. As control mice did not show bacteria, lactobacilli permanence is supported by their exogenous administration to mice.

Fig. 3: Ultrastructure of mouse vagina i.va. inoculated with phytobiotics, lactobacilli or phytocompounds analysed by transmission electron microscopy. A:L. salivarius CRL1328+Hamamelis (2500X); B: L. salivarius CRL1328+Hamamelis (4000X); C: Hamamelis (1200X); D: Hamamelis (4000X); E: L. salivarius CRL1328 (1200X); F: L. salivarius CRL1328 (4000X); G: Peptone-water Control Mice Group (800X); H: peptone-water Control Mice Group (4000X). Bacteria or bacilli close or in contact with the epithelial vaginal cells are indicated with black arrows.
## III. MATERIALS AND METHODS
Microorganisms: Four BVL strains previously characterized and genetically identified were used in this work:Lactobacillus gasseri CRL1320, L. reuteri CRL1324, L. salivarius CRL1328 and L. rhamnosus CRL1332 (Marchesi et al., 2020) (Table 1). The strains were freezed-stored in milk yeast-extract (10% skim-milk, 0.5% yeast-extract, 1% glucose), and they were inoculated, and subcultured 3 times in MRS broth (DeManRogosa and Sharpe) (Biokar-Diagnostics-Beauvais-France) at $37^{\circ}\mathrm{C}$ for 13-14 h before their use. For the i.va. administration in mice, the strains were centrifuged at 3000g for 10 min (Presvac-Argentina), and resuspended
in $50 \mu l$ agarose-peptone [1% meatpeptone, 1.5% agar] or combined with the vegetal extracts selected (Table 2).
Table 1: Beneficial Vaginal Lactobacilli (BVL) Properties
<table><tr><td>BVL Strains</td><td>Beneficial Properties</td></tr><tr><td>L. gasseri CRL1320</td><td>H2O2and lactic acid production, high hydrophobicity, pathogens inhibition, biofilm formation</td></tr><tr><td>L. reuteri CRL1324</td><td>H2O2and lactic acid production, high hydrophobicity, biofilm formation, pathogens inhibition, colonization, of BALB/c vaginal tract mice, adhesion to fibrinogen and mucin</td></tr><tr><td>L. salivarius CRL1328</td><td>bacteriocin production, pathogens inhibition</td></tr><tr><td>L. rhamnosus CRL1332</td><td>H2O2production, high hydrophobicity, biofilm, formation, high resistance to lyophilization, pathogens inhibition, colonization of BALB/c mice vaginal tract, adhesion to fibrinogen and mucin</td></tr></table>
Table 2: Vegetal Extracts and uses
<table><tr><td>Scientific name</td><td>Popular name</td><td>Uses</td><td>Pharmacopoeia</td></tr><tr><td>Hamamelis viriginiana</td><td>Hamamelis</td><td>Astringent, antiseptic, antiinflammatory, antiviral, venotonic</td><td>Argentine 8thedition</td></tr><tr><td>Amaranthus muricatus</td><td>Yerba-meona</td><td>Diuretic, antitumor, drastic purging, warts, herpes, depurative</td><td>Argentine 6thedition</td></tr><tr><td>Smilax aspera</td><td>Zarzaparrilla</td><td>Diuretic renal depurative, antibacterial, antifungal, antiseptic</td><td>European</td></tr></table>
Phytocompounds: "Hamamelis-virginiana" (Hamamelis), "Amaranthus-muricatus" (Yerba meona) and "Smilax-áspera" (Zarzaparilla) were selected for their ethnopharmacological properties and compatibility against different BVL, summarized in Table 2 (Marchesi et al., 2020). From dry extracts of Hamamelis (leaves), Yerbameona (leaves-stem), and Zarzaparilla (bark+branches+fruits), previously obtained by maceration (according to the Argentine Pharmacopoeia) and dried, 1 mg of extract was weighed with 1 ml of alcohol 40%, taking only 20 μl of each plant extract (1mg/ml in 40%-alcohol) were mixed with 30μl agarose-peptone, 50μl i.va. administered to mice.
Microorganisms+Phytocompounds: the pellet from $3^{\text{rd}}$ BVL subculture was mixed with $20\mu \text{l}$ vegetal-extract $+30\mu \text{l}$ agarose-peptone for mouse i.va. administration. The protocol of administration in the murine model is indicated in Figure 4.
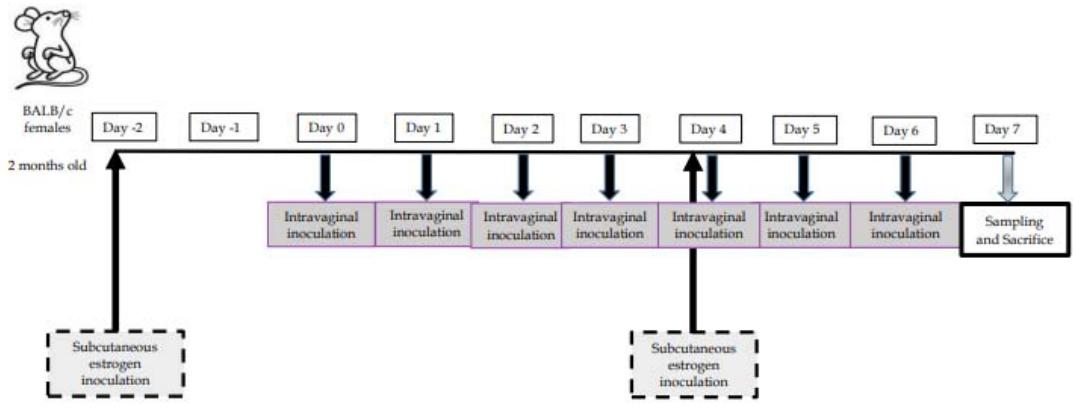
Fig. 4: Protocol of Administration of Phytobiotics, Lactobacilli and Phytocompounds by I.Va. Route to Estrogenized-Adult BALB/C Female Mice
### a) Female BALB/C Mice as Experimental Model
Female BALB/c mice, 45-days-old, 20-25 average-weight, were provided and maintained in CERELA nursery, keeping constant environmental conditions, fed ad libitum with conventional balanced diet. Pseudostrous status was induced with $\beta$ -estradiol-valerate subcutaneous administered on days “-2” and “3”, according to previously set-up protocols, to avoid the variations from the estrous cycle state, and to promote lactobacilli permanence (De Gregorio et al., 2012). Hormone was prepared from a stock solution (2 mg/ml, Sigma-Life-Sciences, Switzerland) resuspended and diluted in sesame-oil (Sigma-Life-Sciences, Mexico) at $0.2\mathrm{mg / ml}$. Then, $0.1\mathrm{ml}$ was injected subcutaneously $(0.02\mathrm{mg})$. The experimental protocol, hormone administration and sampling days are shown in Figure 4. 100 mice were used, divided in 20 groups, assigning 5 mice randomly to each group, as follows:
1) Phytobiotic Groups (12): The following combinations of probiotic BVL+phyto extracts were i.va administered for 7 days: L. gasseri CRL1320+ Hamamelis, L. gasseri CRL1320+ Zarzaparrilla, L. gasseri CRL1320+Yerba-meona, L. reuteri CRL1324+Hamamelis,L. reuteri CRL1324+ Zarzaparrilla, L. reuteri CRL1324+ Yerbameona, L. salivarius CRL1328+Hamamelis, L. salivarius CRL1328+Zarzaparrilla, L. salivarius CRL1328+ Yerba-meona, L. rhamnosus CRL1332+ Hamamelis, L. rhamnosus CRL1332+Zarzaparrilla, and L. rhamnosus CRL1332+Yerba-meona.
2) Vegetal Extracts Groups (3): mice were i.va. administered for 7 days with: Hamamelis-virginiana (Hamamelis), Amaranthus-muricatus (Yerba-meona) and Smilaxáspera (Zarzaparrilla).
3) BVL Strains Groups (4):Lactobacillus gasseri CRL1320, Limosilactobacillus reuteri CRL1324, Ligilactobacillus salivarius CRL1328 or Lacticaseibacillus rhamnosus CRL1332 were individually i.va. administered during 7 days, at $10^{7}-$ $10^{8}$ CFU each dose:
4) Control Group (1): $20 \mu l$ saline $+30 \mu l$ $1 \%$ -agarose-peptone.
Two independent assays were carried out. CERELA Institutional Committee for the Care and Use of Laboratory Animals approved the experimental protocol CRL-BIOT-LMP2010/1A.
### b) Mice Sampling and Analytical Procedures
Murine vagina was washed in sterile conditions with $50 \mu l$ phosphate-buffered-saline (PBS: $8.1 \mathrm{mM} \mathrm{Na}_{2} \mathrm{HPO}_{4}$, $1.5 \mathrm{mM} \mathrm{KH}_{2} \mathrm{PO}_{4}$, $140 \mathrm{mM} \mathrm{NaCl}$, pH 7.2) 7 times and v.w. of each animal pooled. Vaginal washes were used for different assays:
1) Quantification of microorganisms: serial dilutions were prepared and inoculated in selective MRS agar (pH 5.5) to quantify viable BVL after $48\mathrm{h}$ incubation at $37^{\circ}\mathrm{C}$. Number of microorganisms was expressed as log CFU/ml v.w.
2) Cytological studies: $10\mu l$ aliquots were spread on slides, fixed and stained with MayGrünwald-Giemsa technique to assess whether inoculation with phytobiotics produced any type of adverse effect at cytological level. Preparations were observed under light microscope (40x, Axio-Scope-A1, Carl-Zeiss) (McLean et al., 2012).
3) Histological studies: mice were sacrificed by cervical dislocation at the $7^{\text{th}}$ day and dissected to extract vagina, which was transferred to the appropriate solvents for subsequent process for histological and electronic microscope observation, as follows:
i. Histological evaluation (light microscopy): vaginal tissues were fixed in $4\%$ (v/v) formaldehyde at $4^{\circ}\mathrm{C}$, embedded in paraffin by applying routine laboratory methods. Organs were processed according to Silva de Ruiz et al. (2003) using Carl Zeiss Microscope (40x).
ii. Ultrastructural evaluation (transmission electron microscopy): mouse vagina samples were placed in Karnovsky's fixative (2.66% paraformaldehyde,
### c) Statistics Analysis
Analysis of variance (ANOVA) using a general linear model was applied to define the main effects of experimental groups on the number of viable lactobacilli. Significant differences $(p$ -value $< 0.05)$ between mean values were determined by Tukey's test, using MINITAB statistical software (version-16 for Windows).
#### ACKNOWLEDGEMENTS
This work was supported by CONICET and ANPCYT grants.
Generating HTML Viewer...
References
55 Cites in Article
M Alfaro,S Manrique-Rodríguez,C Fernández-Llamazares (2013). Empleo clínico de los probióticos y aspectos prácticos de su empleo.
(2018). º-8° and 9.
Muhammad Aziz,Amir Khan,Muhammad Adnan,Izatullah Izatullah (2017). Traditional uses of medicinal plants reported by the indigenous communities and local herbal practitioners of Bajaur Agency, Federally Administrated Tribal Areas, Pakistan.
M Daniele,L Pascual,L Barberis (2014). Curative effect of the probiotic strain Lactobacillus fermentum L23 in a murine model of vaginal infection by Gardnerella vaginalis.
Priscilla De Gregorio,María Juárez Tomás,Viviana Santos,María Nader-Macías (2012). Beneficial lactobacilli: effects on the vaginal tract in a murine experimental model.
Priscilla De Gregorio,María Tomás,María Terraf,María Nader-Macías (2014). In vitro and in vivo effects of beneficial vaginal lactobacilli on pathogens responsible for urogenital tract infections.
P De Gregorio,M Juárez-Tomás,M Leccese-Terraf,M Nader Macías (2015). Preventive effect of Lactobacillus reuteri CRL1324 on group-B Streptococcus vaginal colonization in an experimental mouse model.
Priscilla De Gregorio,María Juárez Tomás,María Nader‐macías (2016). Immunomodulation of <i>Lactobacillus reuteri</i> CRL1324 on Group B <i>Streptococcus</i> Vaginal Colonization in a Murine Experimental Model.
P De Gregorio,N Maldonado,E Pingitore,M Leccese-Terraf,M Juareztomás,C Silvia-De-Ruiz,V Santos,B Wiese,E Bru,M Paiz,M Reina,D Schujman,M Nader-Macías (2020). Intravaginal administration of gelatine capsules containing freeze-dried autochthonous lactobacilli: a double-blind, randomised clinical trial of safety.
Priscilla De Gregorio,Jessica Silva,Antonella Marchesi,María Nader-Macías (2019). Anti-<i>Candida</i>activity of beneficial vaginal lactobacilli in<i>in vitro</i>assays and in a murine experimental model.
Gilbert Donders,Kateryna Ruban,Gert Bellen,Svitrigaile Grinceviciene (2019). Pharmacotherapy for the treatment of vaginal atrophy.
Magdalena Espino,Manuel Solari,María Fernández,Joana Boiteux,María Gómez,María Silva (2019). NADES-mediated folk plant extracts as novel antifungal agents against Candida albicans.
Robert Uerpmann-Wittzack (2018). European Directorate for the Quality of Medicines and Healthcare (EDQM).
M Falagas,G Betsi,S Athanasiou (2007). Probiotics for the treatment of women with bacterial vaginosis.
Matthew Falagas,Gregoria Betsi,Theodoros Tokas,Stavros Athanasiou (2006). Probiotics for Prevention of Recurrent Urinary Tract Infections in Women.
U Behnke (2012). Joint FAO/WHO Expert Committee on Milk Hygiene. Third Report, Genf, 22.–28. April 1969, 82 Seiten. World Health Organization Technical Report Series No. 453, FAO Agricultural Studies No. 83. Herausgegeben von der World Health Organization, Genf 1970. Preis 8 s; 1,25 $; 4,— Sw.fr.
Ana Flores-Mireles,Jennifer Walker,Michael Caparon,Scott Hultgren (2015). Urinary tract infections: epidemiology, mechanisms of infection and treatment options.
Andrew Flower,Kim Harman,George Lewith,Michael Moore,Felicity Bishop,Beth Stuart,Nicholas Lampert (2016). Standardised Chinese herbal treatment delivered by GPs compared with individualised treatment administered by practitioners of Chinese herbal medicine for women with recurrent urinary tract infections (RUTI): study protocol for a randomised controlled trial.
Varsha Gupta,Paola Mastromarino,Ritu Garg (2024). Effectiveness of Prophylactic Oral and/or Vaginal Probiotic Supplementation in the Prevention of Recurrent Urinary Tract Infections: A Randomized, Double-Blind, Placebo-Controlled Trial.
C Hill,F Guarner,G Reid,G Gibson,D Merenstein,B Pot,L Morelli,R Canani,H Flint,S Salminen,P Calder,M Sanders (2014). The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic.
Hyun-Min Joo,Yang-Jin Hyun,Kil-Sun Myoung,Young-Tae Ahn,Jung-Hee Lee,Chul-Sung Huh,Myung Han,Dong-Hyun Kim (2011). Lactobacillus johnsonii HY7042 ameliorates Gardnerella vaginalis-induced vaginosis by killing Gardnerella vaginalis and inhibiting NF-κB activation.
H Joo,K Kim,K Myoung,Y Ahn,J Lee,C Huh,M Han,D Kim (2012). Lactobacillus helveticusHY7801 ameliorates vulvovaginal candidiasis in mice by inhibiting fungal growth and NF-κB activation.
M Karam,M Habibi,S Bouzari (2019). Urinary tract infection: Pathogenicity, antibiotic resistance and development of effective vaccines against Uropathogenic Escherichia coli.
Mattias Karlsson,Nikolai Scherbak,Gregor Reid,Jana Jass (2012). Lactobacillus rhamnosus GR-1 enhances NF-kappaB activation in Escherichia coli-stimulated urinary bladder cells through TLR4.
Liudmyla Lazarenko,Lidiia Babenko,Liubov Sichel,Valentyn Pidgorskyi,Viktoriia Mokrozub,Olga Voronkova,Mykola Spivak (2012). Antagonistic Action of Lactobacilli and Bifidobacteria in Relation to Staphylococcus aureus and Their Influence on the Immune Response in Cases of Intravaginal Staphylococcosis in Mice.
M Leccese-Terraf,M Juárez-Tomás,M Nader Macías,C Silva (2012). Screening of biofilm formation by beneficial vaginal lactobacilli and influence of culture media components.
María Leccese Terraf,María Juarez Tomás,Lucie Rault,Yves Le Loir,Sergine Even,María Nader-Macías (2017). In vitro effect of vaginal lactobacilli on the growth and adhesion abilities of uropathogenic Escherichia coli.
A Marchesi,J Silva,C Ficoseco,B Wiese,M Nader-Macias (2020). Effect of phytoderivatives on the growth of homologous beneficial vaginal lactobacilli (BVL) strains and their compatibility for the design of phytobiotics for the vaginal tract health.
María Miranda,María Nader-Macías (2024). Pharmabiotic/phytobiotic formulas approach and their intravaginal effect on different parameters.
Nripendra Mishra,Ajay Kesharwani,Aakanksha Agarwal,Suja Polachira,Reshmi Nair,Satish Gupta (2018). Herbal Gel Formulation Developed for Anti-Human Immunodeficiency Virus (HIV)-1 Activity Also Inhibits In Vitro HSV-2 Infection.
F Montorsi,G Gandaglia,A Salonia,A Briganti,V Mirone (2016). Effectiveness of a Combination of Cranberries, Lactobacillus rhamnosus, and Vitamin C for the Management of Recurrent Urinary Tract Infections in Women: Results of a Pilot Study.
Maria Moraes,Gilmara Cunha,Mirna Bezerra,Francisco Fechine,Andréa Pontes,Wanda Andrade,Fernando Frota Bezerra,Manoel Moraes,Pacífica Cavalcanti (2012). Efficacy of the Mentha crispa in the treatment of women with Trichomonas vaginalis infection.
María Moreno,Laura Gómez-Mascaraque,Myriam Arias,Iris Zampini,Jorge Sayago,Liudis Ramos,Guillermo Schmeda-Hirschmann,Amparo López-Rubio,María Isla (2018). Electrosprayed chitosan microcapsules as delivery vehicles for vaginal phytoformulations.
María Moreno,Laura Gómez-Mascaraque,Myriam Arias,Iris Zampini,Jorge Sayago,Liudis Ramos,Guillermo Schmeda-Hirschmann,Amparo López-Rubio,María Isla (2018). Electrosprayed chitosan microcapsules as delivery vehicles for vaginal phytoformulations.
D Muench,D Kuch,H Wu,A Begum,S Veit,M Pelletier,Á Soler García,A Jerse (2009). Hydrogen Peroxide-Producing Lactobacilli Inhibit Gonococci in-vitro but Not during Experimental Genital Tract Infection.
María Nader‐macías,Priscilla De Gregorio,Jessica Silva (2021). Probiotic lactobacilli in formulas and hygiene products for the health of the urogenital tract.
María Nader-Macías,María Juárez Tomás (2015). Profiles and technological requirements of urogenital probiotics.
A Palmeira-De-Oliveira,B Silva,R Palmeira-De-Oliveira,J Martinez-De-Oliveira,L Salgueiro (2013). Are Plant Extracts a Potential Therapeutic Approach for Genital Infections?.
R Palmeira-De-Oliveira,A Palmeira-De-Oliveira,J Martinez-De-Oliveira (2015). New strategies for local treatment of vaginal infections.
Zhe‐chen Qi,Chao Shen,Yu‐wei Han,Wei Shen,Man Yang,Jinliang Liu,Zong‐suo Liang,Pan Li,Cheng‐xin Fu (2017). Development of microsatellite loci in Mediterranean sarsaparilla (<i>Smilax aspera</i>; Smilacaceae) using transcriptome data.
Surada Satthakarn,Florian Hladik,Aornrutai Promsong,Wipawee Nittayananta (2015). Vaginal innate immune mediators are modulated by a water extract of Houttuynia cordata Thunb.
C Silva De Ruiz,M Del R. Rey,M Nader‐macías (2003). Structural and ultrastructural studies of the urinary tract of mice inoculated with <i>Lactobacillus fermentum</i>.
C Silva-De-Ruiz,M Rey,A Pesce-De-Ruiz Holgado,M Nader-Macías (2001). Experimental administration of estradiol on the colonization of Lactobacillus fermentum and Escherichia coli in the urogenital tract of mice.
Jessica Silva,Priscilla De Gregorio,María Nader-Macías (2023). Safety and Effects of Intravaginal Administration of Lacticaseibacillus rhamnosus CRL1332 Immobilized on Nanofibers in a Murine Experimental Model.
Rachel Spurbeck,Cindy Arvidson (2011). Lactobacilli at the Front Line of Defense Against Vaginally Acquired Infections.
Dominika Szczerbiec,Mirosława Słaba,Agnieszka Torzewska (2023). Substances Secreted by Lactobacillus spp. from the Urinary Tract Microbiota Play a Protective Role against Proteus mirabilis Infections and Their Complications.
L Theisen,C Erdelmeier,G Spoden,F Boukhallouk,A Sausy,L Florin,C Muller (2014). Tannins from Hamamelis-virginiana bark extract: characterization and improvement of the antiviral efficacy against influenza A virus and human papillomavirus.
R Wagner,S Johnson (2012). Probiotic Lactobacillus and estrogen effects on vaginal epithelial gene expression responses to Candida albicans.
Renato Zampini,Ximena Castro-González,Luciana Sari,Alfredo Martin,Ana Diaz,Martin Argañaraz,Silvana Apichela (2020). Effect of Cooling and Freezing on Llama (Lama glama) Sperm Ultrastructure.
Gabriela Zárate,Viviana Santos,María Nader-Macias (2007). Protective Effect of Vaginal<i>Lactobacillus paracasei</i>CRL 1289 against Urogenital Infection Produced by<i>Staphylococcus aureus</i>in a Mouse Animal Model.
G Zárate,V Santos,M Nader-Macias (2009). Protective effect of vaginal Lactobacillus paracasei CRL1289 against urogenital infection produced by Staphylococcus aureus in a mouse animal model.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Antonella Marchesi. 2026. \u201cEffect of the Intravaginal Inoculation of Phytobiotics in a Murine Experimental Model\u201d. Global Journal of Medical Research - G: Veterinary Science & Medicine GJMR-G Volume 24 (GJMR Volume 24 Issue G1): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Lactobacilli play a fundamental role in maintaining health and preventing infections of the female urogenital tract. Complementary use of vegetal extracts selected for their ethnopharmacological characteristics and conventional uses for the different conditions in the human body as an adequate alternative therapy for the restoration of the vaginal microbiome has emerged. Compatibility of phytoextracts with lactobacilli for the design of phytobiotic formulas was determined previously. Safety of selected combinations by the intravaginal (i.va.) administration in a murine model was evaluated to determine if some type of adverse effect was produced in the host. Lactobacillus gasseri CRL1320, Limosilactobacillus reuteri CRL1324, Ligilactobacillus salivarius CRL1328 and Lacticaseibacillus rhamnosus CRL1332 combined with Hamamelis-virginiana, Amaranthus-muricatus and Smilax-áspera, were inoculated individually or combined in mice vagina (7 daily doses).
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.