## I. INTRODUCTION
Wound is the disruption of cellular and anatomic continuity of living tissue produced by physical, chemical, electrical or microbial insults to the tissue. Wound healing is the dynamic process of regeneration or repair of broken tissue [1]. Chronic wounds are rapidly growing problem worldwide, due to increasing health care costs, an ageing population, and a sharp rise in the incidence of diseases such as diabetes and obesity [2]. The skin is under constant stress from the sun, smog, friction, tension, temperature, and other external factors. Therefore, under sufficient stress that causes injury, it results in wounds. Wounds may be classified as; open and closed, acute and chronic, avulsion and degloving, clean and contaminated, infected and colonized, laceration, incision and abrasion, puncture, penetration, and gunshot wounds. Nonetheless, they exist in various forms comprising crush injuries, ulcers, skin tears, bruises, and post-operative, which directly or indirectly affect human health conditions. If it is not treated correctly, it may ultimately lead to death. Wounds can be caused by various microorganisms such as bacteria, fungi, parasites, and viruses. Some of the commonly associated bacteria organisms include Staphylococcus aureus, Pseudomonas aeruginosa, Escherichia coli, Klebsiellaspp., Acinetobacterspp [3]. Studies conducted in 2014 and 2021 reported a wound infection prevalence of $10\%$ and $5.95\%$ respectively [4, 5]. Medicinal plants and synthetic drugs have been the most valuable sources of molecules with therapeutic potential throughout the history of mankind. Folk medicine of each civilization is based on natural products and, nowadays, medicinal plants still represent an important pool for the identification of novel drug leads [6]. African panaxia being a synthesized herbal medicine is made from various herbal plants such as ginseng root, Optimum gratisimum, Panaxquifolium, Aloe-tera and water with active ingredients ginsenosides, methyl eugenol, saponins and salicylic acid respectively [7].
### a) Rationale
The advent of the resistance that pathogenic microorganisms have developed against antibiotics has necessitated much attention to be paid on plant extracts and biologically active compounds isolated from natural plants used in herbal medicine [8]. Despite the use of various synthesized herbal plants in the treatment of bacteria wound infection, there is limited information regarding the use of African panaxia in the treatment of wound infections. Furthermore, no study has been carried out in this study area. Hence there is need to investigate on the in-vitro activity of African panaxia on bacteria wound infections.
b) Goal of study
- The goal of this study was to provide base line data on the effect of African panaxia on bacteria wound infection.
c) Hypothesis
- There is no significant effect of African panaxia on bacterial wound infections.
d) Objectives of study
## i. General objective
- The general objective of this study was to determine the effect of African panaxia on bacteria wound infection. ii. Specific objectives To determine the efficacy of distilled water extract of African panaxia on Staphylococcus aureus and Klebsiella species bacteria from wound infection.
To determine the efficacy of Luke warm water extract of African panaxia on Staphylococcus aureusand Klebsiella species bacteria from wound infection.
To determine the efficacy of ethanoic extract of African panaxia on Staphylococcus aureus and Klebsiella species bacteria from wound infection.
## II. MATERIALS AND METHODS
a) Study area and setting
- This study was carried out in TikO, in Maflekumen Medical Teaching and Research Laboratory situated in TikO is a subdivision of Fako Division in the South West Region of Cameroon with a [81]. The life style and occupation of inhabitants of TikO including the dusty, windy and hot nature, farming, bike riding and much more favours the acquisition of wounds b humans thus making the area suitable for this study.
b) Study design and duration
- This was a Laboratory based experimental study designed that was conducted from November 2022, to June 2023.
c) Specimens and sampling
- This study made use of bacteria isolates from people with bacteria wound infection in Tiko community.
d) Ethical consideration
- An introductory letter was obtained from MAFLEKUMEN Higher Institute of Health Sciences TIKO (APPENDIX A) and was taken to regional delegation in Buea for the approval of the project. An administrative authorization was obtained from the regional delegation (APPENDIX B) and was presented to the administration of MAFLEKUMEN. An authorisation was gotten from the MAFLEKUMEN administration to carry out the research.
e) Data collection and techniques
## i. Sample collection
Bacterial isolates were obtained from Maflekumen diagnostic laboratory. Preparation of
MacConkey agar, blood agar and EMB was done by weighing the powder using an electronic balance and dissolved in distilled water following the manufacturer's instructions and was cooked to obtain the gel using a Bunsen burner and allowed to cool to $40^{\circ}\mathrm{C}$. The agar was poured into petri dishes and allowed to solidify. The samples were inoculated in the plate and read after 24hrs. Presumptive identification of bacteria was done based on colony characteristics, gram reactions were recorded. Confirmatory biochemical tests were done to confirm the bacteria. For staphylococcus aureus, and Klebsiella species respectively.
## ii. Catalase test
A drop of Hydrogen peroxide was placed on a slide and a colony of isolated bacteria picked and emulsified on the slide containing the hydrogen peroxide. The appearance of air bubbles indicate catalase positive.
## iii. Coagulase test
A drop of normal saline was placed at both ends of the same slide, one labeled test and the other control. A colony of the isolated bacteria was emulsified on each drop of the normal saline. Serum was placed on the test path and emulsified and nothing was added on the control. The presence of coagulation indicates coagulase.
## iv. Indole test
Test organism was inoculated in a bijou bottle containing $3\mathrm{ml}$ of sterile tryptone water. Incubate at $35-37^{\circ}\mathrm{C}$ for up to $48\mathrm{h}$. $0.5\mathrm{ml}$ of Kovac's reagent was added and shake gently, examination for a red color in the surface layer within 10 minutes macroscopically.
v. Extraction of African panaxia (Alcohol, distilled water and luke warm water)
African panaxia was bought from the Moghamo express in Mutengene and transported to the laboratory for sensitivity testing on the bacteria isolates. One gram of African panaxia was weighted on an electric scale balance and put in a 250 ml flask, followed by adding 100 ml of solvent (95% ethanol). The flask was then left at room temperature for two days preceding filtration funnel and Wattman No. 1 filter paper. The filtrate was concentrated under decreased pressure with an evaporator at $40^{\circ}\mathrm{C}$. This crude extract was saved at $4^{\circ}\mathrm{C}$ until use, this extract of African panaxia was considered as the $100\%$ concentration for ethanol extract, different stock solution for distilled water were made equally and also for look warm water respectively. Then the concentrations (100%, 75%, 50%, and 25%) were made by diluting the concentrated extract of African panaxia with appropriate volumes of sterile distilled respectively for luke warm water. Serial dilutions were made to determine the minimum inhibitory, and bactericidal concentration respectively.
i. Different stock solutions of African panaxia were made (Absolute alcohol, luke warm water and distilled water, in which different volumes were used 100, 75, 50 and 25 in which the isolated species of bacteria were used to test for the minimum inhibitory concentration and minimum bactericidal concentration using dilution technique and absorbance was measured using a spectrophotometry machine at a wavelength of 660nm.
ii. A solution of the isolated bacteria was prepared and standardized by matching to the 0.5 McFarland turbidity standards using sterile saline to produce approximately $1.5 \times 10^{8}$ colony forming units per ml. iii. Serial dilutions were made on the different stock solutions of African panaxia using four sterile dry tubes per isolate and per stock solution respectively. iv. Two (2ml) of nutrient broth was placed in each sterile test tube, followed by adding 2ml of each stock solution in the first tube, mix well and transfer 2ml to the next tube continuously and to the fourth to remove 2ml and discard respectively 1drop of the bacterial suspension was place in each test tube respectively.
v. They labeled test tubes were sealed and incubated at $37^{\circ} \mathrm{C}$ for 18 to 24 hours in which the and minimum inhibitory concentration and minimum bactericidal concentration recorded by checking the turbidity of each tube and absorbance was measured using a spectrophotometer at $660 \mathrm{~nm}$ following the control of the absorbance of 0.5 McFarland standard and Azithromycin.
### f) Data analysis
Data was analyzed using Microsoft excel and the results was presented in tables and figures
## III. RESULTS AND DISCUSSION
### a) Results
This chapter presents the results obtained from the effect of African panaxia on Staphylococcus and Klebsiella isolated from wounds. Based on extract with distilled water the concentration with $75\%$ and $50\%$ stocks were effective in inhibiting the growth of Staphylococcus and Klebsiella respectively. Also using a stock of $50\%$ and $25\%$, it exhibited bactericidal properties on Staphylococcus and Klebsiella respectively as presented on figure 1 below.
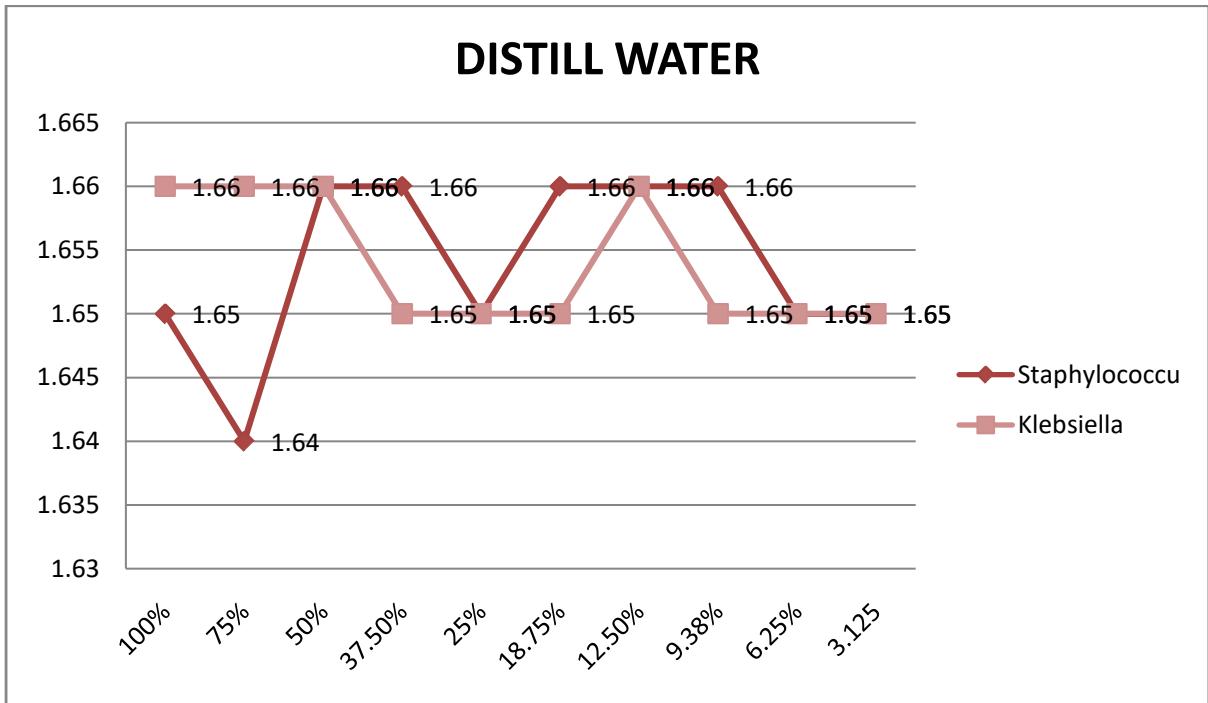 Figure 1: Effects of African panaxia on Staphylococcus aureus and Klebsiella pneumoniae
Based on extract with luke-warm water the concentration with stocks of $100\%$, $75\%$ were effective in inhibiting the growth of Staphylococcus without Klebsiella and with stocks of $50\%$ and $25\%$ having bactericidal activity against Staphylococcus with stock of $25\%$ having inhibitory properties as presented on figure 2 below.
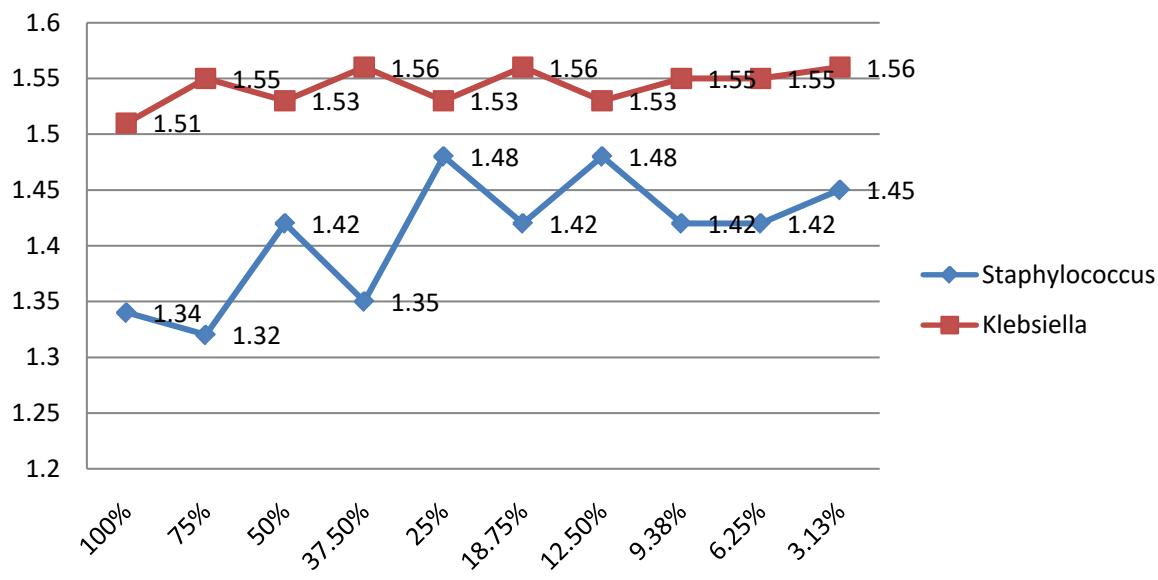 LUKE WARM WATER Figure 2: Effects of African panaxia on Staphylococcus and Klebsiella pneumoniae
Based on extract with absolute alcohol the concentration with $95\%$ stocks were bactericidal on Staphylococcus and Klebsiella and with stocks of $75\%$
was bactericidal and inhibitory on the growth of Staphylococcus and Klebsiella respectively as presented on figure 3 below.
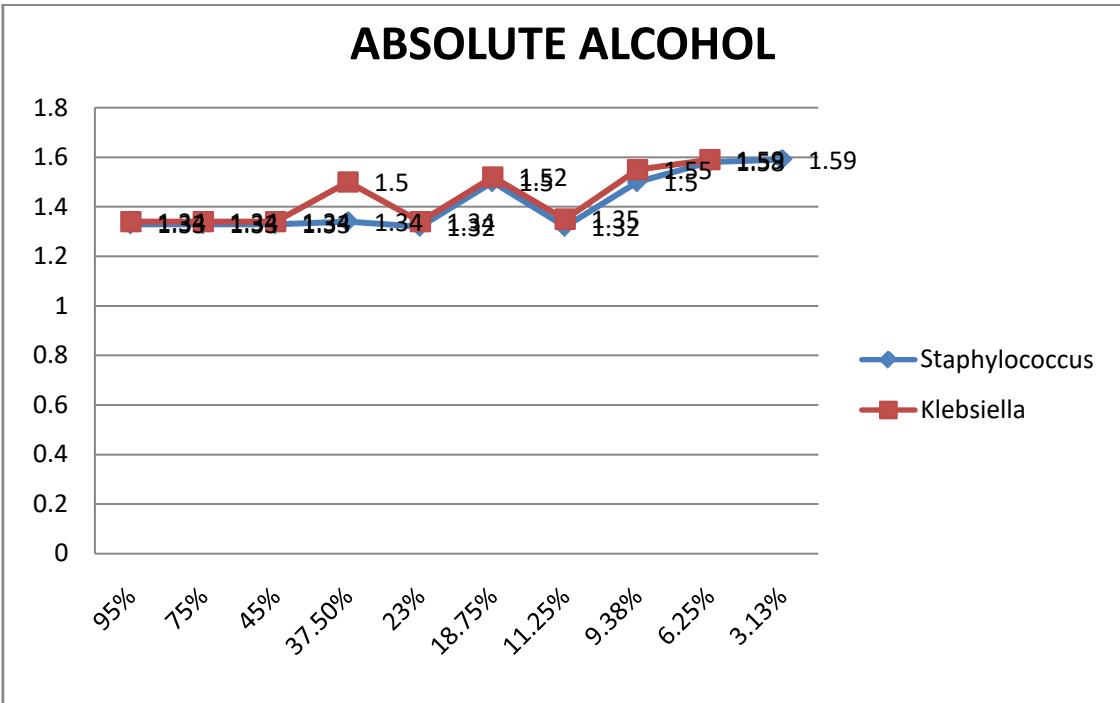
Based on the inhibitory and bactericidal property of Africa panaxia on staphylococcus and Klebsiella, of all the different concentrations made with distilled water, at $25\%$ stock concentration, the extract of African panaxia was both inhibitory and bactericidal on Staphylococcus and Klebsiella. African panaxia extract with look warm water revealed that the extract was bactericidal at $50\%$ stock and $25\%$ stock concentration on Staphylococcus and bacteriostatic at $25\%$ stock concentration on Klebsiella. Finally with alcoholic extract of the African panaxia, the plant extract was bactericidal at $95\%$ stock concentration on Staphylococcus and Klebsiella and bactericidal and bacteriostatic at $75\%$ stock concentration on Staphylococcus and bacteriostatic on Klebsiella as presented on table 1 below.
Table 1: Effects of African panaxia extract on Staphylococcus and Klebsiella pneumoniae
<table><tr><td rowspan="2">DILUENT</td><td rowspan="2">Stock CONC.</td><td colspan="2">Staphylococcus</td><td colspan="2">Klebsiella</td></tr><tr><td>Turbidity</td><td>No turbidity</td><td>turbidity</td><td>No turbidity</td></tr><tr><td rowspan="4">DISTILL WATER</td><td>100%</td><td>4</td><td>0</td><td>4</td><td>0</td></tr><tr><td>75%</td><td>3</td><td>1</td><td>4</td><td>0</td></tr><tr><td>50%</td><td>3</td><td>1</td><td>3</td><td>1</td></tr><tr><td>25%</td><td>2</td><td>2</td><td>2</td><td>2</td></tr><tr><td rowspan="3">LUKE WARM WATER</td><td>100%</td><td>3</td><td>1</td><td>4</td><td>0</td></tr><tr><td>75%</td><td>2</td><td>2</td><td>4</td><td>0</td></tr><tr><td>50%</td><td>1</td><td>3</td><td>4</td><td>0</td></tr><tr><td rowspan="4">ALCOHOL</td><td>25%</td><td>1</td><td>3</td><td>3</td><td>1</td></tr><tr><td>95%</td><td>0</td><td>4</td><td>0</td><td>4</td></tr><tr><td>75%</td><td>2</td><td>2</td><td>3</td><td>1</td></tr><tr><td>50%</td><td>4</td><td>0</td><td>4</td><td>0</td></tr></table>
### b) Discussions
Isolation and identification of Staphylococcus aureus and Klebsiella pneumoniae from wound infections
Infection of wounds comes from so many sources and the most common bacteria which might infect and complicate wounds include Pseudomonas aeruginosa, Klebsiella pneumonia, Staphylococcus aureus, Enterococcus faecalis and Acinetobacter baumannii. Based on the results obtained from these findings, it was revealed that the most common bacteria isolated from wounds were Staphylococcus aureus, and Klebsiella pneumonia. These findings are similar to the results obtained by Mohamed Salahet al., in 2022, who isolated Staphylococcus sp., Klebsiella sp., Pseudomonas sp., Bacillus sp., E.coli diabetic wound infections [83]. Also, Mohammed et al., in 2019, also revealed that the most common bacteria isolated from wounds were Staphylococcus aureus, and Klebsiella pneumonia [84]. Also, these findings agree with other findings by Obi et al., in 2015 who reported that common bacteria isolates from the different types of wounds were Pseudomonas aeruginosa, Klebsiella pneumonia, Staphylococcus aureus, Enterococcus faecalis and Acinetobacter baumannii [85].
To determine the minimum inhibitory concentration of African panaxiaon Staphylococcus aureus and Klebsiella pneumoniae from wound infections
The minimum inhibitory concentrations were determined using extract from different concentrations such as distilled water, luke warm water and ethanoic extracts of the African panaxia. From the findings it was revealed that African panxia extracts of distilled water and luke warm water were more inhibitory at $75\%$, $50\%$ stock respectively than Bactericidal. These findings are similar to results of Korukluoglu et al. in 2010 who reported that extraction of aqueous solvent resulted in a product with greater overall antimicrobial activity than extraction with water, as aqueous extracts of all the olive oil displayed little or no antimicrobial activity against any of the bacteria tested [86]. Similarly Weerakkody et al.,
(2010) [5] observed that water extracts of oregano and rosemary had little or no antimicrobial activity compared to ethanol or hexane extracts. Again, Sofia et al., (2007) [6] reported that water extracts of mustard, cinnamon, garlic and clove had good inhibitory activities against E. coli and S. aureus, To determine the minimum bactericidal concentration of African panaxia on Staphylococcus aureus and Klebsiella pneumoniae from wound infections
Comparing results found in this study with those of the literature, we notice in a previous work on antimicrobial activity of some medicinal plants from Tunisia, that methanolic extracts of C. monspeliensis leaves have shown an interesting activity against P. aeruginosa, S. aureus, E. faecalis with inhibition zones diameters of 18.0, 20.0 and $15.0\mathrm{mm}$, respectively.26
Whereas, water-methanol extracts of fruit peels of pomegranate (P. granatum) have demonstrated a moderate activity when they were tested on S. aureus, P. aeruginosa and K. pneumoniae (13.0, 18.0 and 16.0 mm, respectively)[27]. This activity of pomegranate peels could be attributed to tannins, for which antimicrobial activity has been demonstrated.[4] On the other hand, the results found in the study concerning the activity of R. tripartita aerial parts extracts are in agreement with other previous works which found significant antibacterial activity of leaves alcoholic extracts against methicillin-resistant S. aureus, 16 and no activity against E. coli and P. aeruginosa. 29 For W. frutescens, El Bouzidi et al. have reported different antibacterial activities of leaves methanolic extracts against S. aureus(11.5 mm), K. pneumoniae (18.0 mm), P. fluorescens (14.5 mm) and no activity against E. coli.30
## IV. CONCLUSION
Based on the results obtained from the study, it could be concluded that the most common bacteria isolates obtained from this study were Staphylococcus aureus, Pseudomonas aeruginosa and Klebsiella pneumonae. African panaxia extract from distilled water was more inhibitory than bacteriocidal on Staphylococcus aureus and Klebsiella pneumonia and the lastly the African panaxia extract from ethanol was totally bactericidal on both Staphylococcus aureus and Klebsiella pneumoniae.
## V. RECOMMENDATIONS
From the results obtained from this study, the following recommendations can be made Ethanoic extract of African panaxia should be used on wounds infected with Klebsiella pneumoniae and Staphylococcus aureus to obtain maximum success. Also other natural herbs should be used to determine their inhibitory and bactericidal properties on Klebsiella pneumoniae and Staphylococcus aureus.
Generating HTML Viewer...
References
86 Cites in Article
T Dons,S Soosairaj (2018). Evaluation of wound healing effect of herbal lotion in albino rats and its antibacterial activities.
A Frankova,L Vistejnova B,T Merinas-Amo C,Z Leheckova B,I Doskocil D,J Wong Soon E,T Kudera F,F Laupua E,A Alonso-Moraga C,L Kokoska F (2021). In vitro antibacterial activity of extracts from Samoan medicinal plants and their on proliferation and migration of human fibroblasts.
O Olawale,Ebenezer (2021). Antibacterial activity of Jatrophatanjorensis leaf extracts against bacteria associated with wound infections from the clinical setting.
Avalere Health,Llc (2018). Worker health chartbook, 2004..
S Kyungmi W, Jiyoun,A Vivtoria (2022). Exploring prevalence of wound infections and related patients characteristics in home care using natural language processing.
S Bittner Fialová,K Rendeková,P Muˇcaji,M Nagy,L Slobodníková (2021). Antibacterial Activity of Medicinal Plants and Their Constituents in the Context of Skin and Wound Infections, Considering European Legislation and Folk Medicine-A Review.
Josephine Ezekwesili-Ofili,Okaka Ozioma,Nwamaka Antoinette,Chinwe (2019). Herbal Medicines in African Traditional Medicine.
D Mohapatra,V Thakur,S Brar (2011). Antibacterial Efficacy of Raw and Processed Honey.
D Phua,A Zosel,K Heard (2009). Dietary supplements and herbal medicine toxicities—when to anticipate them and how to manage them.
Jzrs Tabuti (2006). Herbal medicines used in the treatment of malaria in Budiope county, Uganda.
M Wilcox,G Bodeker (2004). Traditional herbal medicine for malaria.
World Health Organization (2002). WHO/CONRAD Technical Consultation on Nonoxynol-9, World Health Organization, Geneva, 9–10 October 2001: Summary Report.
E Sofowora (1993). Medicinal Plants and Traditional Medicine in Africa.
P Mhame,K Busia,Mjk Kasilo (2010). Clinical practice of African traditional medicine.
(2005). WHO traditional medicine global summit 2023 - meeting recommendations.
D Tasha (2012). Traditional African Healing.
G Otitodun,A Ala,S Nwaubani,M Omobowale,S Ajao,M Ogundare,A Olenloa,G Busari,G Abel,J Braimah,O Kolayemi,J Ogwumike,G Opit,K Ileleji,S Mcneill (1992). Assessing efficacies of insect pest management methods to preserve nutritional composition of bagged maize in storehouses located in markets in Nigeria.
Jacob Olupona (2004). Owner of the Day and Regulator of the Universe:.
D Cumes,Divination_Bones (2014). Author response image 3..
Peter Omonzejele (2008). African Concepts of Health, Disease, and Treatment: An Ethical Inquiry.
S Thorpe (1993). African Traditional Religion.
Marcia Angell,Jerome Kassirer (1998). Alternative Medicine — The Risks of Untested and Unregulated Remedies.
Bernard Kamsu-Foguem,Clovis Foguem (2014). Adverse drug reactions in some African herbal medicine: literature review and stakeholders’ interview.
World Health Organization (2002). WHO/CONRAD Technical Consultation on Nonoxynol-9, World Health Organization, Geneva, 9–10 October 2001: Summary Report.
J Ezekwesili-Ofili,N Onyemelukwe,P Asogwa,I Orji (2014). The bioload and aflatoxin content of herbal mediines from seleted states in Nigeria.
O Hamman (1991). The Joint IUCN-WWF Plants Conservation Programme and its Interest in Medicinal Plants.
Asamoah-Gyadu Jk (2014). Therapeutic strategies in African religion: Health, herbal medicinal and indigenous Christian spirituality.
E-M Co,Iii Keen,Ef,W Aldous (2011). Prevalence of methicillin-resistant Staphylococcus aureus in a combat support hospital in Iraq.
B Hartman,A Tomasz (1981). Altered penicillin-binding proteins in methicillin-resistant strains of Staphylococcus aureus.
Aula Abbara,Timothy Rawson,Nabil Karah,Wael El-Amin,James Hatcher,Bachir Tajaldin,Osman Dar,Omar Dewachi,Ghassan Abu Sitta,Bernt Uhlin,Annie Sparrow (2018). A summary and appraisal of existing evidence of antimicrobial resistance in the Syrian conflict.
I Sabri,K Adwan,T Essawi,M Farraj (2013). Molecular characterization of methicillin resistant Staphylococcus aureus isolates in three different Arab world countries.
Y Tabana,S Dahham,B Al-Hindi,A Al-Akkad,Mbk Ahamed (2015). Prevalence of Methicillin-Resistant Staphylococcus aureus (MRSA) among Medical Staff in Three Syrian Provinces: Damascus, Daraa and Al-Swayda.
Aula Abbara,Timothy Rawson,Nabil Karah,Wael El-Amin,James Hatcher,Bachir Tajaldin,Osman Dar,Omar Dewachi,Ghassan Abu Sitta,Bernt Uhlin,Annie Sparrow (2018). Antimicrobial resistance in the context of the Syrian conflict: Drivers before and after the onset of conflict and key recommendations.
H Ruud,O Alewijn,H Dunja,J Sofia,R John,Alexander (2017). High prevalence of Panton -Valentine leukocidin among methicillin-resistant Staphylococcus aureus from refugees in the Netherlands, 2014 -2015. MRSA and MSSA: epidemiology and resistance.
U Heudorf,S Albert-Braun,K Hunfeld,F Birne,J Schulze,K Strobel (2016). The Impact of Screening Patients for Multidrug Resistant Organisms within the Health-Care Setting.
Claudia Reinheimer,Volkhard Kempf,Stephan Göttig,Michael Hogardt,Thomas Wichelhaus,Fiona O’rourke,Christian Brandt (2015). Multidrug-resistant organisms detected in refugee patients admitted to a University Hospital, Germany June‒December 2015.
M Oelmeier,B Glatz,S Willems,A Kossow,M Strobel,B Stuhmer (2017). Prevalence of Multidrug Resistant Bacteria in Refugees: A Prospective Case Control Study in an Obstetric Cohort.
Rein Piso,Roman Käch,Roxana Pop,Daniela Zillig,Urs Schibli,Stefano Bassetti,Dominik Meinel,Adrian Egli (2017). A Cross-Sectional Study of Colonization Rates with Methicillin-Resistant Staphylococcus aureus (MRSA) and Extended-Spectrum Beta-Lactamase (ESBL) and Carbapenemase-Producing Enterobacteriaceae in Four Swiss Refugee Centres.
J Pieters (2015). Hospitalized asylum seekers to be quarantined for MRSA.
Tommi Kärki,Christian Napoli,Flavia Riccardo,Massimo Fabiani,Maria Dente,Manuel Carballo,Teymur Noori,Silvia Declich (2014). Screening for Infectious Diseases among Newly Arrived Migrants in EU/EEA Countries—Varying Practices but Consensus on the Utility of Screening.
K Hiramatsu,L Cui,M Kuroda,T Ito (2001). The emergence and evolution of methicillinresistant Staphylococcus aureus.
M David,R Daum (2010). Community-associated methicillin-resistant Staphylococcus aureus: epidemiology and clinical consequences of an emerging epidemic.
Tobias Tenenbaum,Klaus-Peter Becker,Bettina Lange,Anka Martin,Peter Schäfer,Stefan Weichert,Horst Schroten (2016). Prevalence of Multidrug-Resistant Organisms in Hospitalized Pediatric Refugees in an University Children’s Hospital in Germany 2015–2016.
Cdc (1999). Four Pediatric Deaths From Community-Acquired Methicillin-Resistant<i>Staphylococcus aureus</i>—Minnesota and North Dakota, 1997-1999.
B Fomda,M Thokar,A Khan,J Bhat,D Zahoor,G Bashir,A Majid,P Ray (2014). Nasal carriage of Methicillin-resistant Staphylococcus aureus among healthy population of Kashmir, India.
J Hageman,J Patel,P Franklin,K Miscavish,L Mcdougal,D Lonsway (2008). Occurrence of a USA300 vancomycin-intermediate Staphylococcus aureus.
Madeleine Sowash,Anne-Catrin Uhlemann (2014). Community-Associated Methicillin-Resistant Staphylococcus aureus Case Studies.
José Mediavilla,Liang Chen,Barun Mathema,Barry Kreiswirth (2012). Global epidemiology of community-associated methicillin resistant Staphylococcus aureus (CA-MRSA).
N Hussein,A Alyas,M Majeed,M Assafi (2015). Prevalence Rate and Prevalent Genotypes of CA-MRSA in Kurdistan Region: First Report from Iraq.
N Hussein,P Butaye,Z Basharat,A Muhammed,Al-Dabbagh Sa (2015). Comparative Evaluation of MRSA Nasal Colonization Epidemiology in the Urban and Rural Secondary School Community of Kurdistan, Iraq.
Nawfal Hussein,Zarrin Basharat,Ary Muhammed,Samim Al-Dabbagh (2014). Comparative Evaluation of MRSA Nasal Colonization Epidemiology in the Urban and Rural Secondary School Community of Kurdistan, Iraq.
M Assafi,R Mohammed,N Hussein (2015). Nasal Carriage Rates of Staphylococcus aureus and CA-Methicillin Resistant Staphylococcus aureus among University Students.
Nawfal Hussein,Reving Salih,Narin Rasheed (2019). Prevalence of Methicillin-Resistant Staphylococcus aureus in Hospitals and Community in Duhok, Kurdistan Region of Iraq.
Narin Rasheed,Nawfal Hussein (2019). Methicillin-resistant Staphylococcus aureus carriage rate and molecular characterization of the staphylococcal cassette chromosome mec among Syrian refugees in Iraq.
Masood Abdulrahman,Arazoo Taher (2018). Prevalence of Methicillin Resistant Staphylococcus Aureus Among Food Handlers in Duhok City.
X Xie,Y Bao,N Ouyang,X Dai,K Pan,B Chen (2016). Molecular epidemiology and characteristic of virulence gene of community-acquired and hospitalacquired methicillinresistant Staphylococcus aureus isolates in Sun Yat-sen Memorial hospital, Guangzhou, Southern China.
S Deresinski (2005). Methicillin-Resistant Staphylococcus aureus: An Evolutionary, Epidemiologic, and Therapeutic Odyssey.
Yu-Yu Chuang,Yhu-Chering Huang (2013). Molecular epidemiology of community-associated meticillin-resistant Staphylococcus aureus in Asia.
S Tokajian (2014). New epidemiology of Staphylococcus aureus infections in the Middle East.
M Otto (2010). Basis of virulence in community-associated methicillin-resistant Staphylococcus aureus.
A Bartlett,K Hulten (2010). Staphylococcus aureus pathogenesis: secretion systems, adhesins, and invasins.
Laura Shallcross,Ellen Fragaszy,Anne Johnson,Andrew Hayward (2013). The role of the Panton-Valentine leucocidin toxin in staphylococcal disease: a systematic review and meta-analysis.
A Woodin (1960). Purification of the two components of leucocidin from <i>Staphylococcus aureus</i>.
Pauline Yoong,Victor Torres (2013). The effects of Staphylococcus aureus leukotoxins on the host: cell lysis and beyond.
M Paczosa,J Mecsas (2016). Klebsiella pneumoniae: going on the offense with a strong defense.
K Holt,H Wertheim,R Zadoks (2015). Genomic analysis of diversity, population structure, virulence, and antimicrobial resistance in Klebsiella pneumoniae, an urgent threat to public health.
Kelly Wyres,Margaret Lam,Kathryn Holt (2020). Population genomics of Klebsiella pneumoniae.
Liang Chen,Barry Kreiswirth (2018). Convergence of carbapenem-resistance and hypervirulence in Klebsiella pneumoniae.
Danxia Gu,Ning Dong,Zhiwei Zheng,Di Lin,Man Huang,Lihua Wang,Edward Wai-Chi Chan,Lingbin Shu,Jiang Yu,Rong Zhang,Sheng Chen (2018). A fatal outbreak of ST11 carbapenem-resistant hypervirulent Klebsiella pneumoniae in a Chinese hospital: a molecular epidemiological study.
Thomas Russo,Candace Marr (2019). Hypervirulent Klebsiella pneumoniae.
N Dong,X Yang,R Zhang,Ew-C Chan,Chen (2018). Tracking microevolution events among ST11 carbapenemase-producing hypervirulent Klebsiella pneumoniae outbreak strains.
Miran Tang,Xin Kong,Jingchen Hao,Jinbo Liu (2020). Epidemiological Characteristics and Formation Mechanisms of Multidrug-Resistant Hypervirulent Klebsiella pneumoniae.
Candace Marr,Thomas Russo (2019). Hypervirulent <i>Klebsiella pneumoniae</i>: a new public health threat.
Sylvain Brisse,Francine Grimont,Patrick Grimont (2006). The Genus Klebsiella.
Margaret Rrw,Watts Mclam,C Stephen,Cerdeira Louise,T,Wyres Kelly,L,Kathryn (2021). Holt A genomic surveillance framework and genotyping tool for Klebsiella pneumoniae and its related spcies complex.
Nadia Rodríguez-Medina,Humberto Barrios-Camacho,Josefina Duran-Bedolla,Ulises Garza-Ramos (2019). <i>Klebsiella variicola</i> : an emerging pathogen in humans.
Carla Rodrigues,Virginie Passet,Andriniaina Rakotondrasoa,Sylvain Brisse (2018). Identification of Klebsiella pneumoniae, Klebsiella quasipneumoniae, Klebsiella variicola and Related Phylogroups by MALDI-TOF Mass Spectrometry.
Edward Feil,Bao Li,David Aanensen,William Hanage,Brian Spratt (2004). eBURST: Inferring Patterns of Evolutionary Descent among Clusters of Related Bacterial Genotypes from Multilocus Sequence Typing Data.
Rebecca Martınez,Jonathan Plucker (2004). Identifying Gifted Students: How Far Has Gifted Education Progressed and Where Is it Heading?.
S Navon-Venezia,K Kondratyeva,A Carattoli (2017). Klebsiella pneumoniae: a major worldwide source and shuttle for antibiotic resistance.
S Brisse,C Fevre,V Passet (2009). Virulent clones of Klebsiella pneumoniae: identification and evolutionary scenario based on genomic and phenotypic characterization.
Lavan Singh,M Cariappa,Mandeep Kaur (2016). Klebsiella oxytoca: An emerging pathogen?.
Ngum Ntonifor,Irene Sumbele,Tabot Ebot (2017). Soil-Transmitted Helminth Infections and Associated Risk Factors in a Neglected Region in the Upper Nkongho-mbo Area, South-west Region, Cameroon.
Mohamed Salah,Gamal Badr,Helal Hetta,Walaa Khalifa,Ahmed Shoreit (2022). Isolation and identification of pathogenic biofilm-forming bacteria invading diabetic wounds.
Abas Mohammed,B Atif (2020). Chemical profile, antiproliferative, antioxidant and enzyme inhibition activities of Ocimum basilicum L. and Pulicaria undulata (L.) CA Mey. grown in Sudan.
C Obi (2015). Isolation and sensitivity pattern of bacterial isolates of wound infections from patients of Federal Medical Centre, Umuahia, Abia State.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Augustine Eyong Bate. 2026. \u201cEffects of African Panaxia Extracts on Staphylococcus Aureus and Klebsiella Pneumoniae from Bacteria Wound Infection in Tiko\u201d. Global Journal of Medical Research - B: Pharma, Drug Discovery, Toxicology & Medicine GJMR-B Volume 23 (GJMR Volume 23 Issue B2): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.