Colorectal cancer continues to be one of the leading causes of mortality among patients with neoplasia worldwide, just behind to breast and prostate cancer. The aim of this study was to evaluate the efficacy of pembrolizumab in advanced colorectal neoplasms, to offer patients greater survival. A search for scientific papers was carried out on the Pubmed and Lilacs platforms, using the following descriptors “colorectal cancer”, “pembrolizumab” and “efficacy”, both in Portuguese and in English, both being connected by the Boolean descriptor “AND”. Thus, a total of 33 articles were included after inclusion and exclusion criteria. Despite the progress of studies, these lines of treatment are limited to patients due to the high cost of medications.
## I. INTRODUCTION
C olorectal cancer (CRC), an important public health problem1, is the third most common type of neoplasm and the fourth most common cause of death worldwide 2. The majority of cases are found in Western countries 3 and, to develop this disease, factors such as age, genetics, history of inflammatory bowel disease and the patient's lifestyle habits are considered3.
The main tool for good patient evolution still remains early detection4, 5 and in newly diagnosed patients, the first line of treatment is cytoreductive surgery followed by chemotherapy and/or radiotherapy 3, 5, 6. However, there are numerous difficulties encountered in this scenario, as mutation rates result in ineffectiveness in many affected patients or many patients are diagnosed at an advanced stage (stage 4 tumor).
To ensure greater chances of survival for these patients, increasingly advanced studies are launched annually using immunotherapies7, especially this study, with the drug pembrolizumab. Studies show that many patients benefited from the use of this medication, presenting lasting responses8, with highly modified tumors, chemotherapy proved to be inferior9 compared to pembrolizumab.
This medicine is considered a monoclonal antibody 12, 13 and is capable of causing changes in defense T cells, guaranteeing an anti-tumor effect 14. Small doses are used as neoadjuvant therapy and, 3 weeks after insertion of the medication, cytoreductive surgery is performed 14. Patients who are diagnosed in the metastatic phase benefited from the use of pembrolizumab15.
The present study aimed to analyze the effectiveness of using pembrolizumab in advanced CRC, in order to guarantee greater survival for affected patients.
## II. MATERIAL METHOD
This is an integrative literature review, which consists of a research method that allows the synthesis and criticism of a given topic for study investigation. This review was prepared following the six steps of the process for carrying out an integrative review. To survey existing scientific productions on the topic, the first stage was developed to define the guiding question: "Efficacy of pembrolizumab in relation to advanced colorectal tumors".
The second stage consisted of defining the inclusion and exclusion criteria. Complete articles were included in the study, in Portuguese and English, year of publication between 2016 and 2024, original articles, randomized and controlled studies and articles related to the guiding question and the descriptors. The exclusion criteria were scientific articles that did not have the full text available, were duplicates and strayed from the topic.
The third stage consisted of researching scientific articles, for this, data collection took place in the months of March and April 2024, an electronic search was carried out in the U.S. National Library of Medicine and the National Institutes Health (PubMed) databases. and through the Virtual Health Library (VHL), in the following information base: Latin American and Caribbean Literature in Health Sciences (LILACS), using the combination of descriptors belonging to the Health Sciences Descriptors (DeCS): "colorectal cancer", "pembrolizumab" and "efficacy", which were connected by the Boolean descriptor "AND".
Based on the results found by searching the articles in the databases with the descriptors, following the inclusion and exclusion criteria presented, the fourth stage of the article began, which consisted of a thorough reading of the titles and abstracts of each work in order to verify compatibility with the guiding question of the present study. Then, the fifth stage of the work began, which consisted of qualitative and descriptive analysis of the collected data and for this, the selected articles were organized in the form of a table (table 1) with the following items: author, year, main results and/or conclusions and sample of children to facilitate understanding when analyzing the data found.
Finally, the sixth stage comprises the discussion and synthesis of results, which was carried out in a comparative way of the data interpreted in the analysis of scientific articles in order to contribute information for the preparation of the work.
## III. RESULTS
99 articles were found in Pubmed, using only descriptors, when applying the inclusion criteria, 66 articles remained, 33 of which were excluded and, after being part of the exclusion criteria, the final selection resulted in 33 articles selected. In the VHL-LILACS, 1 article was found and, after applying the exclusion criteria, no article was selected.
Therefore, 66 articles remained for complete reading and, after that, 33 were excluded, leaving 33 articles for the final analysis, as shown in figure 1. After evaluating the findings obtained in this research, it was possible to observe that of the 33 articles, 27 were in favor of the use of pembrolizumab in advanced RCC. One article stated that the main line of treatment for this type of tumor is surgery followed by chemotherapy/ radiotherapy. Furthermore, 1 article stated that pembrolizumab did not show sufficient efficacy for colorectal cancer, being much more used in breast cancer or small cell lung cancer.
Quadro 1: Relação dos artigos incluídos no estudo segundo as variáveis estudos, por ordem de seleção dos artigos, ano e principais resultados encontrados
<table><tr><td>Authors</td><td>Year</td><td>Main Results</td></tr><tr><td>André T, Shiu KK, et all</td><td>2020</td><td>This phase 3 randomized clinical trial showed that front-line pembrolizumab was superior to chemotherapy with regard to progression-free survival in patients with MSI-H-dMMR metastatic colorectal cancer.</td></tr><tr><td>Ganesh, K., et all</td><td>2019</td><td>Immune checkpoint inhibitor (ICI) treatment, specifically with monoclonal antibodies targeting programmed cell death 1 (PD1) and cytotoxic T lymphocyte antigen 4 (CTLA4), results in improved survival in dMMR-MSI-H metastatic RCC</td></tr><tr><td>Fan A, et all</td><td>2021</td><td>The high efficacy of neoadjuvant immunotherapy in early-stage RCC has been proven.</td></tr><tr><td>Zhang X, et all</td><td>2022</td><td>For patients who have advanced unresectable or mCRC and are suitable for high-intensity chemotherapy, the NCCN v1.2021 guidelines recommend nivolumab ± ipilimumab (O ± Y) or pembrolizumab as first-line treatment for patients with MSI-H/dMMR status.</td></tr><tr><td>Picard E, et all</td><td>2020</td><td>Initial approaches using anti-PD-1 mAbs in CRC were disappointing as only little, if any, clinical benefit was obtained.</td></tr><tr><td>Weng J, et all</td><td>2022</td><td>Immune checkpoint blockade (ICB) therapy has made significant progress in the treatment of advanced malignancies, and patients who benefit from this therapy can achieve a durable response.</td></tr><tr><td>Rosati G, et all</td><td>2022</td><td>In the metastatic setting, a phase II study demonstrated that tumors with a high mutation burden benefit most from the use of pembrolizumab. Pembrolizumab was superior to chemotherapy in terms of PFS.</td></tr><tr><td>Zhu J, et all</td><td>2023</td><td>Surgery combined with chemotherapy and radiotherapy remains the standard component of curative multimodal treatment approaches for LACRC.</td></tr><tr><td>Roth MT, et all</td><td>2021</td><td>Despite the antitumor activity of pembrolizumab in patients with MSI-H RCC, 30% to 35% of patients fail to obtain any benefit.</td></tr><tr><td>Manz SM, et all</td><td>2021</td><td>Despite the antitumor activity of pembrolizumab in patients with MSI-H RCC, 30% to 35% of patients fail to obtain any benefit. Keynote-177, the first randomized trial specifically recruiting MSI-H/dMMR mCRCs, showed superior PFS for patients treated with first-line pembrolizumab compared with standard chemotherapy, establishing first-line immunotherapy as the standard of care for MSI-H/mCRC. dMMR</td></tr><tr><td>André T, et all</td><td>2023</td><td>Results from the phase II KEYNOTE-164 study showed that pembrolizumab monotherapy had clinical activity in patients with mCRC who had been treated with ≥2 prior lines of standard therapy and in patients who had received ≥1 prior line of therapy (rate of objective response to pembrolizumab monotherapy led to progression-free survival</td></tr><tr><td>Justesen TF,</td><td>2023</td><td>ICIs, such as anti-PD-1 pembrolizumab, revitalize dysfunctional immune cells to enhance the anticancer immune response. For the majority of patients with CRC, ICIs have limited efficacy, however, for the subgroup of patients with dMMR tumors, promising results were found.</td></tr><tr><td>Maiorano BA, et all</td><td>2022</td><td>Early studies of ICIs in mCRC included pretreated patients with MSI. Both KEYNOTE-016 and KEYNOTE-164 demonstrated the efficacy of pembrolizumab as monotherapy in chemo-resistant patients, with an overall response rate.</td></tr><tr><td>Rawla P, et all</td><td>2019</td><td>Checkpoint inhibitors, a new form of immunotherapy, have been shown to be effective for mCRC patients with MMR deficiency and high MSI, while other forms of immunotherapy have not yet shown significant promise..</td></tr><tr><td>Cervantes B, et all</td><td>2024</td><td>Since 2020 and the results of the phase III KEYNOTE 177 clinical trial, pembrolizumab [anti-programmed cell death protein 1 (PD1)] is the new standard of care in first-line MSI/dMMR mCRC</td></tr><tr><td>Kuang C, et all</td><td>2022</td><td>The combination of pembrolizumab and azacitidine is safe and tolerable with modest clinical activity in the treatment of chemotherapy-refractory mCRC.</td></tr><tr><td>Ronnekleiv-Kelly SM, et all</td><td>2016</td><td>A unique histologic feature of certain tumors in patients with microsatellite instability is infiltration by lymphocytes at the tumor-stromal interface. This feature highlights the biology of the tumor in its microenvironment and underlies the efficacy of the programmed death inhibitor pembrolizumab in patients with microsatellite unstable metastatic colorectal cancer.</td></tr><tr><td>Miyamoto Y, et all</td><td>2022</td><td>The results confirm that pembrolizumab should be the standard of care for first-line treatment of patients with MSI-H/dMMR mCRC</td></tr><tr><td>Chen X, et all</td><td>2024</td><td>Monoclonal antibodies (mAbs), such as nivolumab and pembrolizumab, have been used to improve the effectiveness of CRC treatments.</td></tr><tr><td>Wang C, et all</td><td>2020</td><td>Although no responses were recorded in our cohort, disease control in five patients, four without liver metastases, suggests its clinical activity when combined with nivolumab in a subgroup of MSS metastatic colorectal cancer. Furthermore, prolonged MS of 7 months or more in two patients with prior progression on pembrolizumab and atezolizumab provides preliminary evidence suggesting potential synergy between regorafenib and PD-1 inhibitors</td></tr><tr><td>Cancanelli L, et all</td><td>2021</td><td>In this study, after a median follow-up of 32.4 months, pembrolizumab was superior to chemotherapy in terms of progression-free survival (median, 16.5 vs. 8.2 months; hazard ratio, 0.60; 95%CI, 0.45-0.80; P=0.0002), but data on OS still remain blinded until final analysis</td></tr><tr><td>Zhang X, et all</td><td>2022</td><td>Cinco inibidos de PD-1/PD-L1 aprovados pela FDA são usados na terapêutica do cancer: nivolumabe, pembrolizumabe, atezolizumabe, durvalumabe e avelumab</td></tr><tr><td>Jung G, et all</td><td>2020</td><td>The clinical trial design was of high quality and, given the compelling clinical benefit, pembrolizumab is highly recommended as a first-line treatment option for MMRd mCRC.</td></tr><tr><td>Fountzilas C, et all</td><td>2021</td><td>Pembrolizumab has evolved as an active treatment option in patients with advanced MSI-H/dMMR colorectal cancer, regardless of SARS or BRAF status.</td></tr><tr><td>Morse MA, et all</td><td>2020</td><td>Recently, immune checkpoint inhibitors have demonstrated impressive activity in patients with CRC and other solid tumors that are deficient in mismatch repair (dMMR).</td></tr><tr><td>Razak AR, et all</td><td>2020</td><td>Patients who received 1100 mg AMG 820 plus pembrolizumab had a better irPR response, including 2 of 41 patients with metastatic RCC.</td></tr><tr><td>Kim DW, et all</td><td>2021</td><td>Ibrutinib 560 mg daily plus pembrolizumab 200 mg every 3 weeks appears to be well tolerated with limited anticancer activity in metastatic RCC.</td></tr><tr><td>Elez E, et all</td><td>2024</td><td>In a subgroup analysis of this study, patients with mCRC carrying the BRAF V600E mutation benefited from treatment with pembrolizumab compared with SOC chemotherapy.</td></tr><tr><td>Wang C, et all</td><td>2019</td><td>Surveillance CT showed a significant and persistent reduction in tumor burden throughout therapy, with complete remission in January 2017.</td></tr><tr><td>Rahma OE, et all</td><td>2022</td><td>The combination of ziv-aflibercept and pembrolizumab demonstrated an acceptable safety profile with antitumor activity in solid tumors.</td></tr><tr><td>Água doce T, et all</td><td>2017</td><td>The development of immunotherapy with immune checkpoint inhibitors (ICIs) has advanced treatment strategies for various types of cancer.</td></tr><tr><td>Smith HG, et all</td><td>2023</td><td>The advent of immunotherapy in the form of immune checkpoint blockade (ICB) has led to dramatic improvements in the treatment of several cancers with historically poor prognoses.</td></tr><tr><td>Pereira LD, et all</td><td>2018</td><td>PD-L1 testing as a predictive biomarker is only recommended for the use of Pembrolizumab in lung cancer, and a predictive role for PD-L1 expression in colorectal and urothelial cancer has not been demonstrated.</td></tr></table>
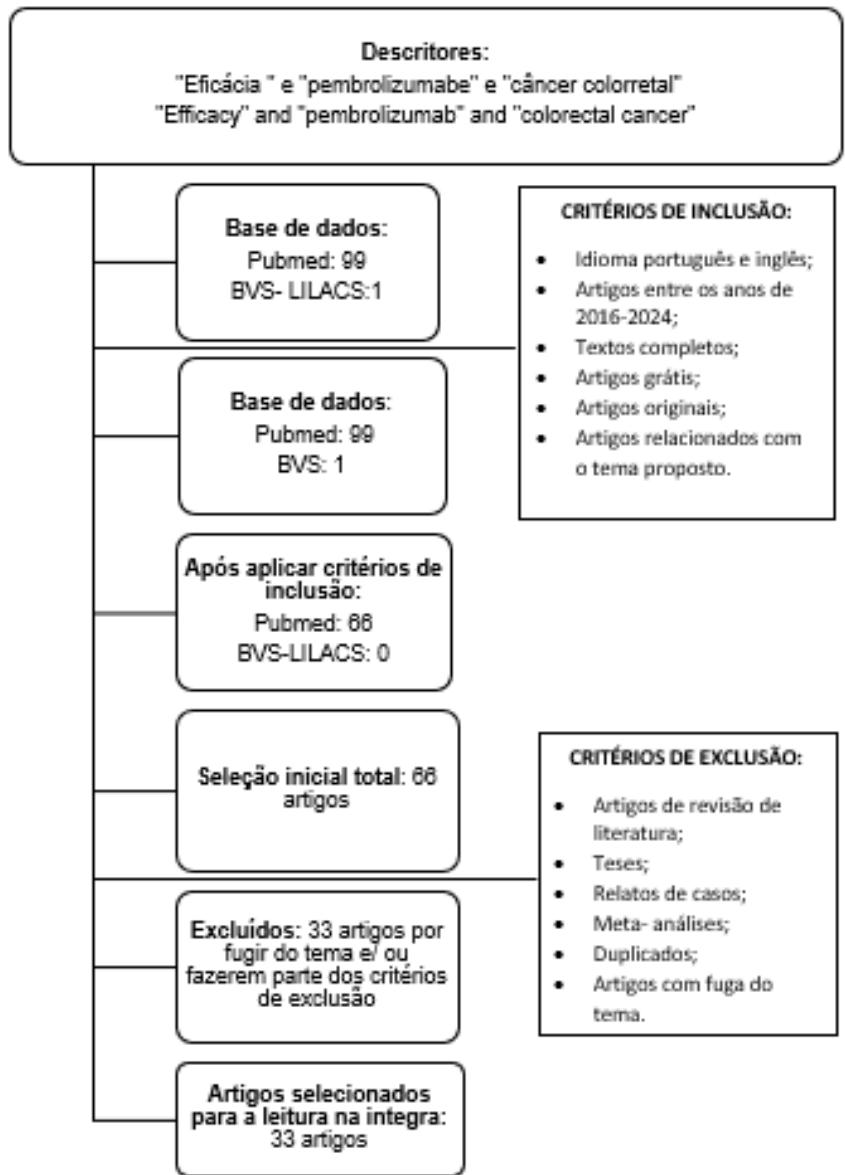 Figura 1: Fluxograma de seleção de artigos inclíidos no estudo, 2024
## IV. DISCUSSION
The present study indicates that pembrolizumab monotherapy was well tolerated in patients with heavily pretreated PD-L1-positive advanced RCC. KEYNOTE-177 was the first randomized phase 3 study comparing pembrolizumab, a PD-1 antibody, with standard chemotherapy in a first-line setting. Initial data analysis demonstrated a higher overall response rate with pembrolizumab, with a notable duration of response, suggesting a plateau in the Kaplan-Meier analysis beyond 3 years of follow-up. The toxicity profile and quality of life outcomes also clearly favored pembrolizumab over chemotherapy.
It was observed in the studies, that in the pembrolizumab line, the ORR was reported between $41\% - 43.8\%$ compared to $32\% - 33.1\%$ following standard chemotherapy. The median PFS for pembrolizumab overall was 16.5 months versus 8.3 months in standard line chemotherapy and the 24-month PFS rates were $48.3\%$ versus $18.6\%$, respectively. In summary, in the overall population, first-line treatment with pembrolizumab provided significantly longer PFS, higher ORR, and prolonged duration of response compared to chemotherapy. OS was also longer with pembrolizumab versus chemotherapy within a worldwide range.
The benefit of pembrolizumab persisted in most groups, however, those with a KRAS or NRAS mutation did not appear to benefit from pembrolizumab compared with standard chemotherapy. Furthermore, MSI-H CRC tumors also appear to have higher PD-L1 expression than their MSS counterparts, possibly suggesting that PD-1 checkpoint blockade may be particularly beneficial in the treatment of MSI-H CRC.
Pembrolizumab was also well tolerated in KEYNOTE-177, with $22\%$ of patients experiencing a grade 3 or higher adverse event based on the National
Cancer Institute Common Terminology Criteria for Adverse Events, relative to $66\%$ of patients in the standard chemotherapy who experienced a grade 3 or higher adverse event. In the analysis of health-related quality of life, pembrolizumab was also superior to standard chemotherapy. Time to deterioration was prolonged for global health status/quality of life, physical functioning, social functioning, and fatigue for patients who received pembrolizumab compared to those who received standard chemotherapy.
The dose of pembrolizumab administered in the latest studies (10 mg/kg Q2W) is higher than that used in the early trials (2 mg/kg every 3 weeks [Q3W], 10 mg/kg Q3W, or 200 mg Q3W) and is superior at currently approved doses (2 mg/kg Q3W and 200 mg Q3W). However, the tolerability of pembrolizumab at all three doses was similar in cohorts of patients with various advanced solid tumors. Therefore, the current study does not support any potential benefit in increasing the dose of pembrolizumab as a way to improve therapeutic response.
## V. CONCLUSION
The results found in this present study suggest that the monoclonal antibody pembrolizumab is of paramount importance in the treatment of advanced colorectal cancer, whether neoadjuvantly, adjuvantly or in association with cytoreductive surgeries. Such a medicine is capable of connecting to tumor cells, causing the effect of antitumor cells on defense T cells.
This effect led to an improvement in survival rates in patients with advanced colorectal cancer, compared to patients who underwent palliative treatments with surgery associated with FOLFOX (5-fluorouracil, oxaliplatin and folinic acid) or FOLFIRI (5-fluorouracil and irinotecan).
Therefore, given that its cost in Brazil is high, it would be of great value if pembrolizumab were available to the entire population affected by CRC, guaranteeing greater survival and quality of life.
Generating HTML Viewer...
References
35 Cites in Article
Alice Shin,Filippo Giancotti,Anil Rustgi (2023). Metastatic colorectal cancer: mechanisms and emerging therapeutics.
Inés Mármol,Cristina Sánchez-De-Diego,Alberto Pradilla Dieste,Elena Cerrada,María Rodriguez Yoldi (2017). Colorectal Carcinoma: A General Overview and Future Perspectives in Colorectal Cancer.
Thierry André,Kai-Keen Shiu,Tae Kim,Benny Jensen,Lars Jensen,Cornelis Punt,Denis Smith,Rocio Garcia-Carbonero,Manuel Benavides,Peter Gibbs,Christelle De La Fouchardiere,Fernando Rivera,Elena Elez,Johanna Bendell,Dung Le,Takayuki Yoshino,Eric Van Cutsem,Ping Yang,Mohammed Farooqui,Patricia Marinello,Luis Diaz (2020). Pembrolizumab in Microsatellite-Instability–High Advanced Colorectal Cancer.
Karuna Ganesh,Zsofia Stadler,Andrea Cercek,Robin Mendelsohn,Jinru Shia,Neil Segal,Luis Diaz (2019). Immunotherapy in colorectal cancer: rationale, challenges and potential.
Ahui Fan,Boda Wang,Xin Wang,Yongzhan Nie,Daiming Fan,Xiaodi Zhao,Yuanyuan Lu (2021). Immunotherapy in colorectal cancer: current achievements and future perspective.
Xuan Zhang,Tao Wu,Xinyi Cai,Jianhua Dong,Cuifeng Xia,Yongchun Zhou,Rong Ding,Renfang Yang,Jing Tan,Lijuan Zhang,Ya Zhang,Yuqin Wang,Chao Dong,Yunfeng Li (2022). Neoadjuvant Immunotherapy for MSI-H/dMMR Locally Advanced Colorectal Cancer: New Strategies and Unveiled Opportunities.
Emilie Picard,Chris Verschoor,Grace Ma,Graham Pawelec (2020). Relationships Between Immune Landscapes, Genetic Subtypes and Responses to Immunotherapy in Colorectal Cancer.
Junyong Weng,Shanbao Li,Zhonglin Zhu,Qi Liu,Ruoxin Zhang,Yufei Yang,Xinxiang Li (2022). Exploring immunotherapy in colorectal cancer.
Gerardo Rosati,Giuseppe Aprile,Alfredo Colombo,Stefano Cordio,Marianna Giampaglia,Alessandro Cappetta,Concetta Porretto,Alfonso De Stefano,Domenico Bilancia,Antonio Avallone (2022). Colorectal Cancer Heterogeneity and the Impact on Precision Medicine and Therapy Efficacy.
Jiahao Zhu,Jie Lian,Benjie Xu,Xiangyi Pang,Shengjun Ji,Yutian Zhao,Haibo Lu (2023). Neoadjuvant immunotherapy for colorectal cancer: Right regimens, right patients, right directions?.
Marc Roth,Satya Das (2021). Pembrolizumab in unresectable or metastatic MSI-high colorectal cancer: safety and efficacy.
Salomon Manz,Marco Losa,Ralph Fritsch,Michael Scharl (2021). Efficacy and side effects of immune checkpoint inhibitors in the treatment of colorectal cancer.
Thierry André,Filippo Pietrantonio,Antonio Avallone,Mahmut Gumus,Lucjan Wyrwicz,Jong Kim,Suayib Yalcin,Mariusz Kwiatkowski,Sara Lonardi,Jakub Zolnierek,Amos Odeleye-Ajakaye,Pierre Leconte,David Fogelman,Tae Kim (2023). KEYSTEP-008: Phase II Trial of Pembrolizumab-Based Combination in MSI-H/dMMR Metastatic Colorectal Cancer.
Tobias Justesen,Ismail Gögenur,Line Tarpgaard,Per Pfeiffer,Camilla Qvortrup (2023). Evaluating the efficacy and safety of neoadjuvant pembrolizumab in patients with stage I–III MMR-deficient colon cancer: a national, multicentre, prospective, single-arm, phase II study protocol.
Brigida Maiorano,Alessandro Parisi,Evaristo Maiello,Davide Ciardiello (2022). The Interplay between Anti-Angiogenics and Immunotherapy in Colorectal Cancer.
Prashanth Rawla,Adam Barsouk,Andreas Hadjinicolaou,Alexander Barsouk (2019). Immunotherapies and Targeted Therapies in the Treatment of Metastatic Colorectal Cancer.
Chaoyuan Kuang,Yongseok Park,Ryan Augustin,Yan Lin,Douglas Hartman,Lindsey Seigh,Reetesh Pai,Weijing Sun,Nathan Bahary,James Ohr,John Rhee,Stanley Marks,H Beasley,Yongli Shuai,James Herman,Hassane Zarour,Edward Chu,James Lee,Anuradha Krishnamurthy (2022). Pembrolizumab plus azacitidine in patients with chemotherapy refractory metastatic colorectal cancer: a single-arm phase 2 trial and correlative biomarker analysis.
Sean Ronnekleiv-Kelly,Richard Burkhart,Timothy Pawlik (2016). Molecular markers of prognosis and therapeutic targets in metastatic colorectal cancer.
Yuji Miyamoto,Katsuhiro Ogawa,Mayuko Ohuchi,Ryuma Tokunaga,Hideo Baba (2022). Emerging evidence of immunotherapy for colorectal cancer.
Xiang Chen,Ling-Juan Chen,Xiao-Fei Peng,Ling Deng,Yan Wang,Jiu-Jiang Li,Dong-Li Guo,Xiao-Hua Niu (2024). Anti-PD-1/PD-L1 therapy for colorectal cancer: Clinical implications and future considerations.
Chongkai Wang,Dawnyel Chevalier,Janelle Saluja,Jaideep Sandhu,Cecilia Lau,Marwan Fakih (2020). Regorafenib and Nivolumab or Pembrolizumab Combination and Circulating Tumor DNA Response Assessment in Refractory Microsatellite Stable Colorectal Cancer.
Luca Cancanelli,Melania Rivano,Lorenzo Di Spazio,Marco Chiumente,Daniele Mengato,Andrea Messori (2021). Efficacy of Immune Checkpoint Inhibitors in Patients with Mismatch Repair-Deficient or Microsatellite Instability-High Metastatic Colorectal Cancer: Analysis of Three Phase-II Trials.
Xiao Zhang,Zhengyang Yang,Yongbo An,Yishan Liu,Qi Wei,Fengming Xu,Hongwei Yao,Zhongtao Zhang (2022). Clinical benefits of PD-1/PD-L1 inhibitors in patients with metastatic colorectal cancer: a systematic review and meta-analysis.
Gerhard Jung,Daniel Benítez-Ribas,Ariadna Sánchez,Francesc Balaguer (2020). Current Treatments of Metastatic Colorectal Cancer with Immune Checkpoint Inhibitors—2020 Update.
Christos Fountzilas,David Bajor,Sarbajit Mukherjee,Joel Saltzman,Agnieszka Witkiewicz,Orla Maguire,Hans Minderman,Ram Nambiar,Hanna Rosenheck,Erik Knudsen,Jason Muhitch,Scott Abrams,Chong Wang,Alan Hutson,Kristopher Attwood,Karen Hicks,Jennifer Jurcevic,Pawel Kalinski,Renuka Iyer,Patrick Boland (2021). Phase Ib/II Study of Cetuximab plus Pembrolizumab in Patients with Advanced RAS Wild-Type Colorectal Cancer.
M Morse,H Hochster,A Benson (2020). Perspectivas no tratamento do câncer colorretal metastático com terapia inibidora de checkpoint imune.
Albiruni Razak,James Cleary,Victor Moreno,Michael Boyer,Emiliano Calvo Aller,William Edenfield,Jeanne Tie,R Harvey,Annemie Rutten,Manish Shah,Anthony Olszanski,Dirk Jäger,Nehal Lakhani,David Ryan,Erik Rasmussen,Gloria Juan,Hansen Wong,Neelesh Soman,Marie-Anne Damiette Smit,Dirk Nagorsen,Kyriakos Papadopoulos (2020). Safety and efficacy of AMG 820, an anti-colony-stimulating factor 1 receptor antibody, in combination with pembrolizumab in adults with advanced solid tumors.
Dae Kim,Elaine Tan,Jun-Min Zhou,Michael Schell,Maria Martinez,James Yu,Estrella Carballido,Rutika Mehta,Jonathan Strosberg,Iman Imanirad,Richard Kim (2021). A phase 1/2 trial of ibrutinib in combination with pembrolizumab in patients with mismatch repair proficient metastatic colorectal cancer.
Elena Elez,Scott Kopetz,Josep Tabernero,Tanios Bekaii-Saab,Julien Taieb,Takayuki Yoshino,Gulam Manji,Kathrine Fernandez,Antonello Abbattista,Xiaosong Zhang,Van Morris (2023). SEAMARK: phase II study of first-line encorafenib and cetuximab plus pembrolizumab for MSI-H/dMMR <i>BRAF</i> V600E-mutant mCRC.
Chongkai Wang,Jaideep Sandhu,Marwan Fakih (2019). Complete response to pembrolizumab in a patient with metastatic colon cancer with microsatellite instability and a history of Guillain-Barre syndrome.
Osama Rahma,Kevin Tyan,Anita Giobbie-Hurder,Andrew Brohl,Philippe Bedard,Daniel Renouf,Elad Sharon,Howard Streicher,Emma Hathaway,Rachel Cunningham,Michael Manos,Mariano Severgnini,Scott Rodig,F Stephen Hodi (2022). Phase IB study of ziv-aflibercept plus pembrolizumab in patients with advanced solid tumors.
T Água Doce,A Kondic,M Ahamadi,C Li,R De Greef,D De Alwis,J Pedra (2017). Avaliação da estratégia de dosagem de pembrolizumabe para indicações oncológicas.
Henry Smith,Anne Bodilsen,Lisbeth Rose,Rahim Altaf,Lene Iversen,Line Walker (2023). Challenges presented by complete response to immune checkpoint blockade in patients with dMMR colorectal cancer: A case report.
L Pereira,F Nunes,P Santos (2018). Biomarcadores preditivos em imuno-oncologia. Oncologia: da prevenção ao tratamento.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Ariane Luiza de Siqueira Braga. 2026. \u201cEfficacy of Pembrolizumab in the Treatment of Advanced Colorectal Cancer\u201d. Global Journal of Medical Research - B: Pharma, Drug Discovery, Toxicology & Medicine GJMR-B Volume 24 (GJMR Volume 24 Issue B2): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Colorectal cancer continues to be one of the leading causes of mortality among patients with neoplasia worldwide, just behind to breast and prostate cancer. The aim of this study was to evaluate the efficacy of pembrolizumab in advanced colorectal neoplasms, to offer patients greater survival. A search for scientific papers was carried out on the Pubmed and Lilacs platforms, using the following descriptors “colorectal cancer”, “pembrolizumab” and “efficacy”, both in Portuguese and in English, both being connected by the Boolean descriptor “AND”. Thus, a total of 33 articles were included after inclusion and exclusion criteria. Despite the progress of studies, these lines of treatment are limited to patients due to the high cost of medications.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.