## I. INTRODUCTION
Elastofibroma is a rare slowly growing benign soft tissue tumor of mesenchymal origin, characterized by typical localization in the subscapular region. It consists of proliferating fibrous and adipose tissue.
The first elastofibroma was diagnosed in 1958 [1]. The first clinical case called "Elastofibroma of the Back" was presented at the XII Scandinavian Congress of Pathologists by doctors O. Jarvi and A.E. Saxen. However, this case was published only in 1961 [2, 3].
In 1982, a clinical and pathological study of 170 cases of elastofibromas was conducted in Okinawa Prefecture, Japan. As a result, a large number of conclusions were made that still remain relevant today [4]. Elastofibroma has a rather typical localization: it is located in soft tissues in the area of the scapula angle deeply in the latissimus dorsi muscle, rhomboid muscle and serratus anterior muscle above the ribs \[4-8\](Fig. 1).
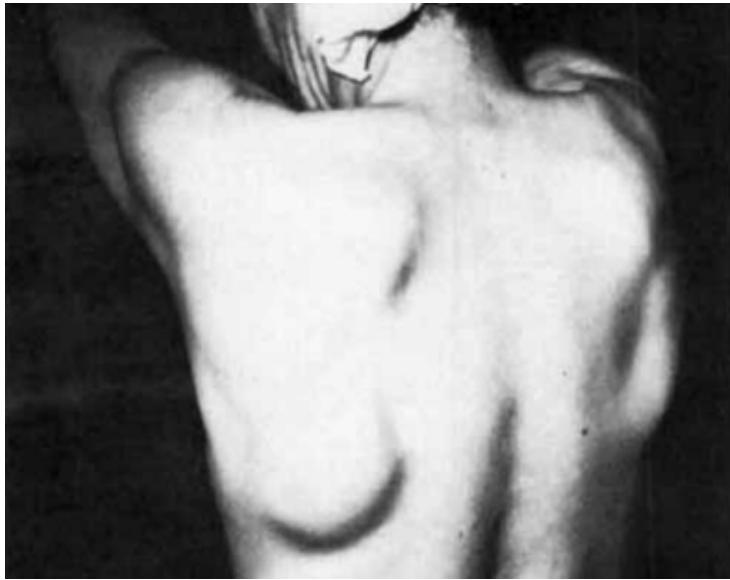 Figure 1: Bilateral subscapular elastofibroma in a 59-year-old female patient.
There are also other options for the localization of elastofibroma: the area of the olecranon, the area of the ischial tuberosity, as well as in the chest wall [4, 9]. The connection between the occurrence of elastofibroma and heredity was also identified for the first time, thanks to a study in Okinawa Prefecture [4]. However, there are other etiological theories: for example, O. Jarvi and A.E. Saxen proposed a concept, according to which elastofibroma is not a real tumor, but a consequence of a degenerative-regenerative process during the movement of the scapula relative to the thoracic fascia, which is also confirmed by researchers of the 21st century [1, 10, 11]. Recent studies revealed that elastofibroma is a monoclonal, neoplastic process with genomic karyotypic instability, which is an important link in the structural modification that affects almost all chromosomes [12,13]. However, M. Hisaoka and H. Hashimoto concluded that the development of elastofibroma is affected by a combination of factors: activation of mesenchymal $\mathrm{CD34+}$ cells and prolonged mechanical impact [14].
According to modern data, elastofibroma is more common, as a rule, in women aged from 65 till 70 [4, 6, 15-18]. It is described in the literature that the incidence rate is 0.23 per 100,000 and is only $1 - 2\%$ of all primary tumors of the chest wall [6, 19]. In 2013, only 400 cases of elastofibroma have been described [20].
Clinically, elastofibroma manifests itself only in $50\%$ of cases [21]. Discomfort, crunching in the area of the scapula, combined with pain on movement, are characteristic clinical signs [22].
Various imaging modalities such as ultrasound, CT, and MRI play an important role in establishing diagnosis. Ultrasound examination of elastofibroma shows an alternation of linear sections that have a different echo signal and form a layered structure \[23\](Fig. 2).
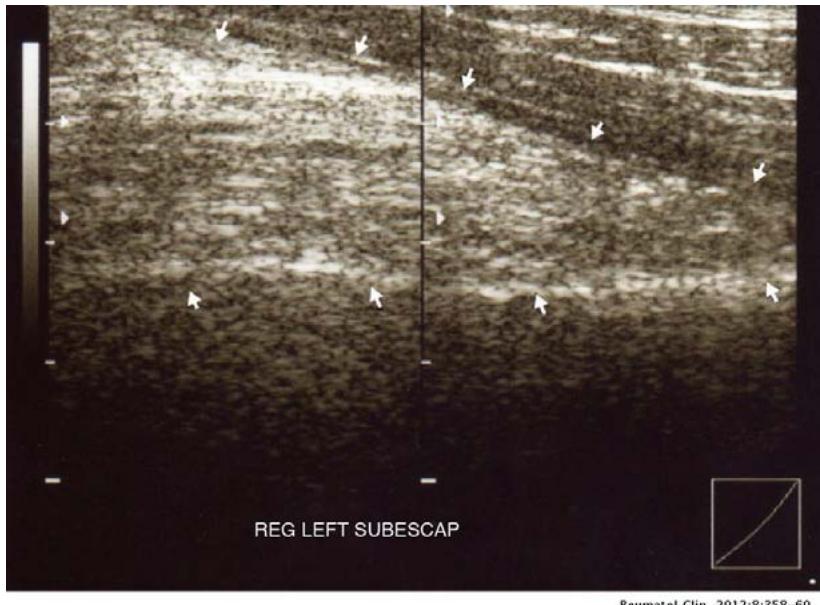 Figure 2: Ultrasound image showing a solid mass with alternating hypo- and hyperechoic areas below the serratus anterior (arrows).
CT reveals an isodense structure with hypodense stripes \[21, 23\](Fig. 3).
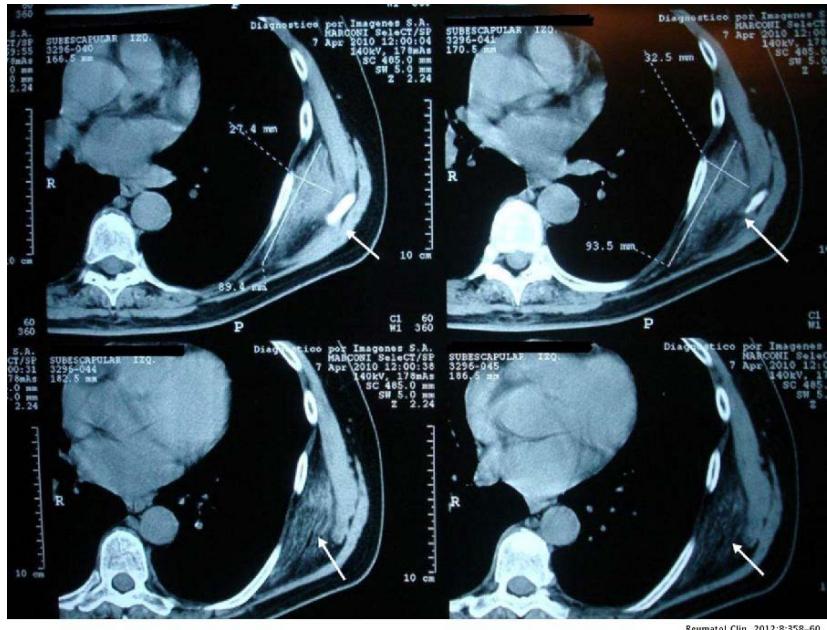 Figure 3: An unenhanced CT scan shows a left subscapular mass that is in contact with intercostal muscles of similar density to skeletal muscle, with areas of weakness similar to subcutaneous fat (arrows).
MRI is the most informative method for diagnosing elastofibroma, which is visualized as a heterogeneous soft tissue component with a well-defined border [24]. PET-CT is performed for another pathology, and elastofibroma is an incidental finding. The study notes a low or moderate metabolism of F18-deoxyglucose, but cases of hypermetabolism are also described [25].
Many authors believe that morphological confirmation of the diagnosis is inappropriate if ancomprehensive radiological conclusion has been obtained [26, 27]. However, in differential diagnosis with lipoma, liposarcoma, hemangioma, hematoma, fibromatosis, desmoidtumor and malignant histiocytoma, morphological examination is necessary [28].
Thus, already by 1969, elastofibroma was described in some detail on the basis of not only a histological study, but also a biochemical one. Chemical analysis showed that about $8\%$ of the total dry mass of the tumor is elastin, and $75 - 80\%$ is collagen, while normal subcutaneous connective tissue contains only $2\%$ of elastin \[1\](Fig. 4).
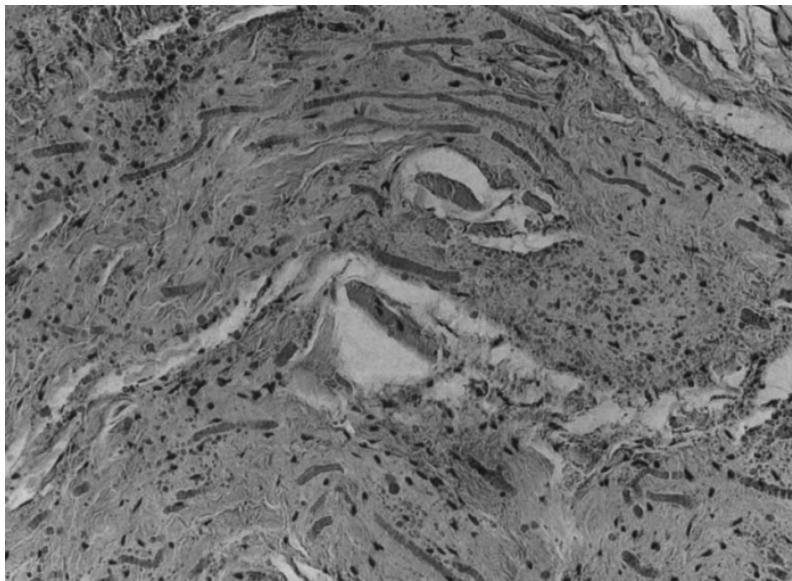 Figure 4: Microslide. Bands of connective tissue with swollen and partially segmented elastic fibers. Both elastic fibers and collagen stain red and differ mainly in refraction. The bands are separated by loose connective tissue and fat.
The histological diagnosis is established by the presence of elastic fibers, often fragmented into a collagen matrix. In electronic microscopy: eosinophilic material often contains mature elastic fibers \[23, 28\](Fig. 5).
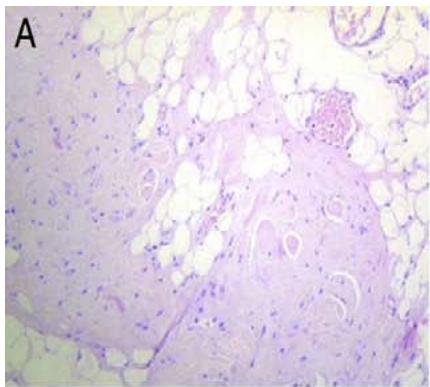
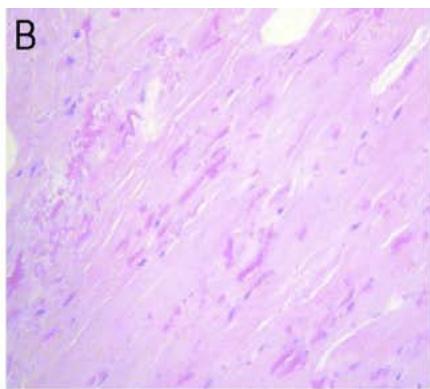
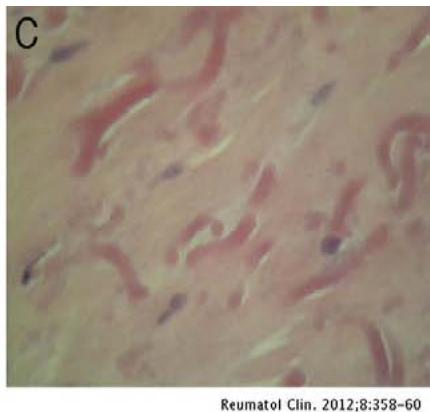 Figure 5: A - Growth of dense small-celled collagen tissue and fat. B - Small nuclei, elongated and fragmented elastic fibers and rounded contours are visualized. C - Details of abnormal elastic fibers.
Complete surgical excision of elastofibroma is the most rational and effective method of treatment. However, observation is indicated in asymptomatic cases [1, 29].
## II. CLINICAL CASE
Patient A., 70 years old, complained of a painless tumor-like subcutaneous mass in the region of the lower angle of the right scapula, applied to the surgeon at the Clinical and Diagnostic Center of ANO CCH MP. She considers herself ill for 5 years, when she first noted a volumetric formation on her back on the right, which progressed and increased in size.
When viewed, a volumetric tumor-like ball-shaped formation measuring $7.5 \times 6.0 \times 2.0$ cm of soft-elastic consistency (Fig. 6) is determined on the back along the lower edge of the right shoulder blade. On palpation the formation is painless, not soldered to the skin. The skin over the formation is not changed. Preliminary diagnosis: Lipoma of the back.
During additional examination, an ultrasound scan (Fig. 7), chest MSCT (Fig. 8) were performed, which showed no evidence for a lipoma, but there was an elastofibroma of the back detected. For differential diagnosis, a biopsy material was taken followed by a histological examination, the obtained results confirmed the diagnosis of "elastofibromadorsi" (Fig. 9).
Thus, the pathoanatomical diagnosis does not contradict the clinical one.
Based on the results of clinical work, it was decided to adhere to observational tactics.
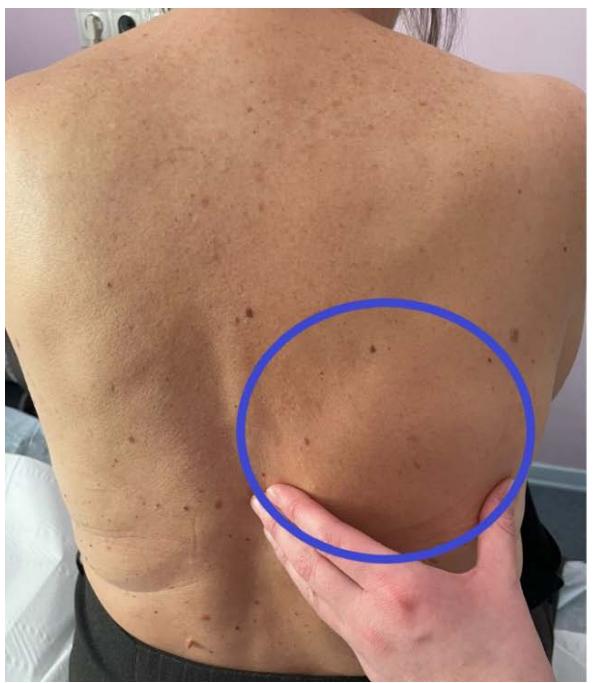
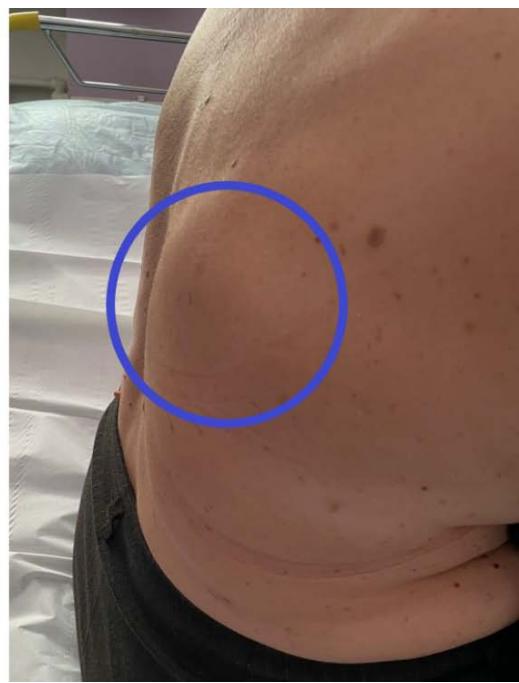
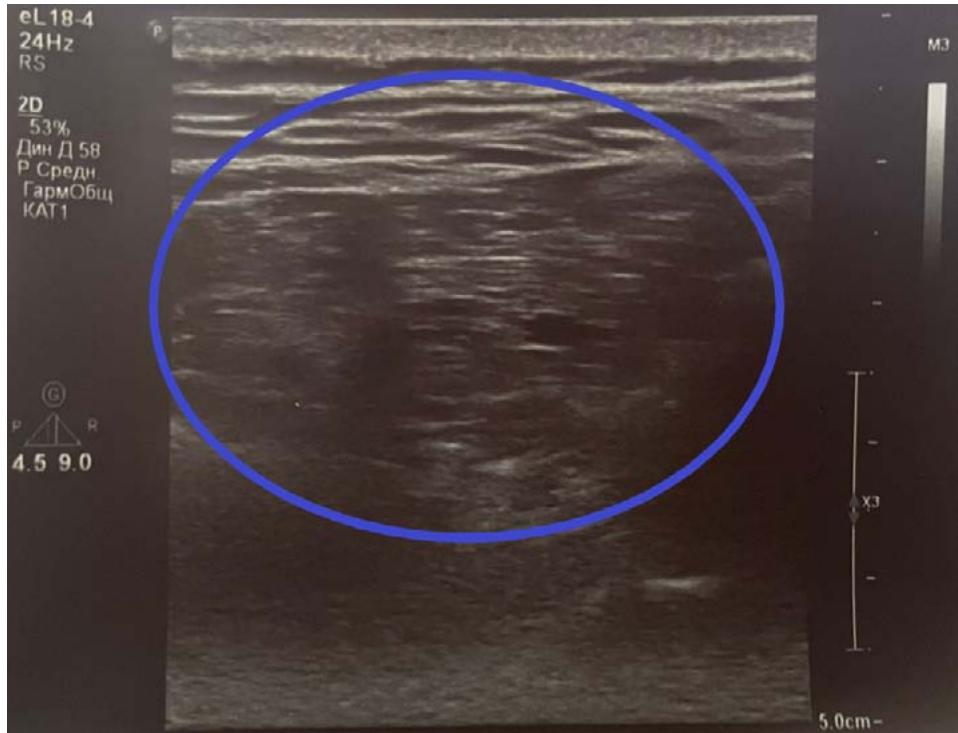 Figure 6: On examination: a voluminous tumor-like formation along the lower edge of the right scapula. Figure 7: Ultrasound, transverse scan: posterior chest wall: a volumetric mass with an inhomogeneous echostructure is determined.
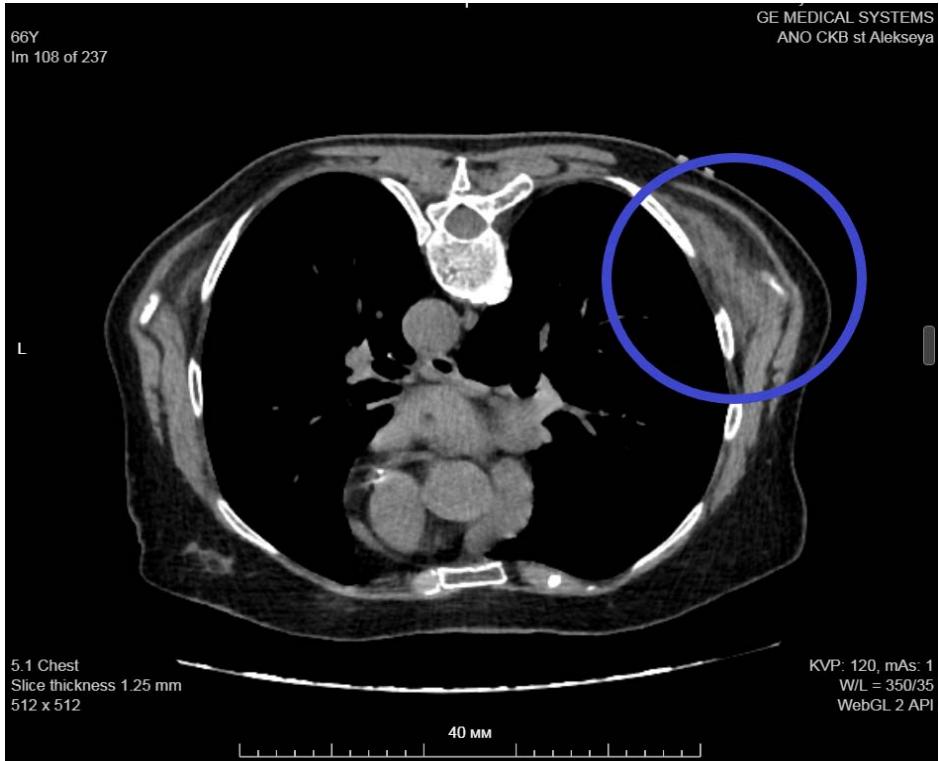 Figure 8: Chest MSCT. Right elastofibromadorsi: isodense structure with hypodense streaks.
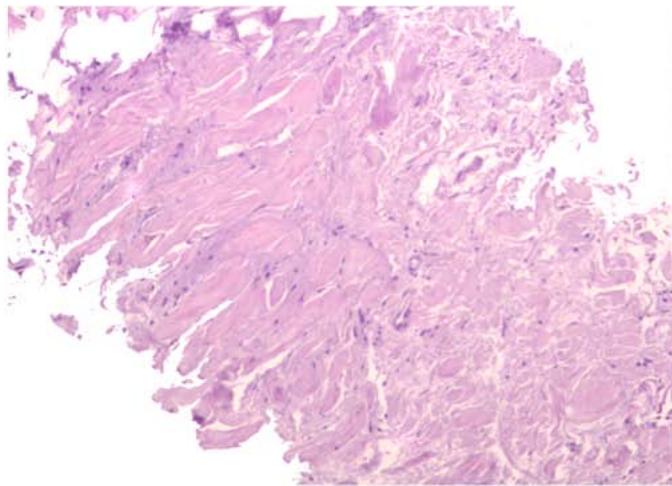 Panel label: a.
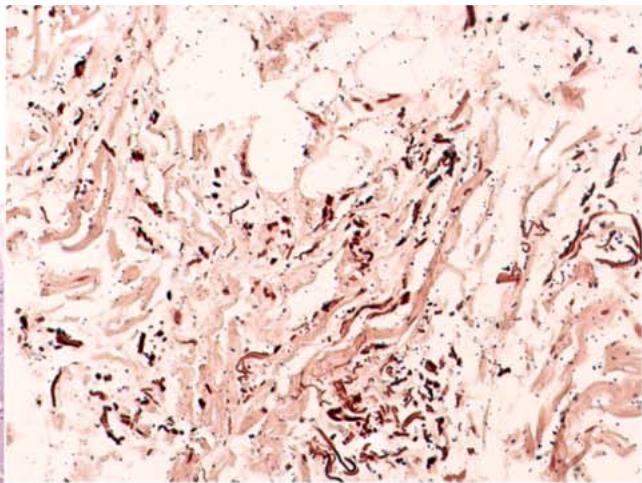 B Figure 9: Microslide of elastofibromadorsi. a - Fibro-adipose tissue with collagen and elastic fibers. b - Orcein staining. Elastic fibers are stained brown.
## III. CONCLUSION
The presented data on clinical cases indicate that elastofibroma is a rare tumor of the soft tissues of the chest wall, which often does not manifest itself clinically, and its diagnosis is random. Imaging methods currently allow an accurate diagnosis, however, a histological examination is necessary for differential diagnosis. Out own clinical case proves that the presence of elastofibroma may not be accompanied by pain, and in this case, observation is advisable.
Generating HTML Viewer...
References
28 Cites in Article
O Järvi,A Saxén,V Hopsu‐havu,J Wartiovaara,V Vaissalo (1969). Elastofibroma—a degenerative pseudotumor.
O Jarvi,E Saxen (1961). Elastofibromadorse // ActaPathol.
M Kransdorf,J Meis,E Montgomery (1992). Elastofibroma: MR and CT appearance with radiologic-pathologic correlation.
C Fletcher,K Unni,F Mertens (2002). Pathology and genetics of tumors of soft tissue and bone; elastofiroma.
J Freixinet,P Rodriguez,M Hussein,B Sanroman,J Herrero,R Gil (2008). Elastofibroma of the thoracic wall.
D Yildirim,M Eroglu,Y Sanli,M Tamam (2012). Нypermetabolic bilateral infrascapular lesions in pet-ct imaging: elastofibromadorsi // Yeditepe.
P Kapff,D Hocken,R Simpson (1987). Elastofibroma of the hand.
M Kastner,M Salai,S Fichman (2009). The Israel Medical Association journal.
M Parratt,J Donaldson,A Flanagan,A Saifuddin,R Pollock,J Skinner,S Cannon,T Briggs (2010). Elastofibroma dorsi: management, outcome and review of the literature.
Juan Fibla,Laureano Molins,Vicente Marco,Javier Pérez,Gonzalo Vidal (2007). Élastofibrome dorsal bilatéral.
Keith Mortman,Gary Hochheiser,Erica Giblin,Yorell Manon-Matos,Kenneth Frankel (2007). Elastofibroma Dorsi: Clinicopathologic Review of 6 Cases.
E Brandser,J Goree,G El-Khoury (1998). Elastofibroma dorsi: prevalence in an elderly patient population as revealed by CT..
O Jarvi,P Lansimies (1975). Subclinical elastofibromas in the scapular region at autopsy series // ActaPathol.
M Naylor,A Nascimento,A Sherrick,R Mcleod (1996). Elastofibroma dorsi: radiologic findings in 12 patients..
Y Blumenkrantz,G Bruno,C González,M Namías,A Osorio,P Parma (2011). Characterization of Elastofibroma Dorsi with 18FDG PET/CT: a retrospective study.
I Mariño,P Solis,A Lara,J Malo,Luz Vazquez,M Tamimi,F (2013). Sensitivity and positive predictive value of magnetic resonance imaging in the diagnosis of elastofibromadorsi: review of fourteen cases.
Emna Braham,Sara Zairi,Mouna Mlika,Aida Ayadi-Kaddour,Olfa Ismail,Faouzi El Mezni (2013). Malignant glomus tumor of trachea: a case report with literature review.
S Oueslati,W Douira,R Dhieb,L Charada,N Mlika,L Rezgui,K Zaouia,M Jamoussi,N Mnif,M Chaabene (2006). IRM de l'arthrite tuberculeuse du genou.
M Hammoumi,A Qtaibi,A Arsalane (2014). Elastofibromadorsi: clinicopathological analysis of 76 cases.
Javier Cavallasca,Debora Sohn,Ariel Borgia,María Maliandi,Jorge Musuruana (2012). Elastofibroma Dorsi: Review of 4 Cases.
Jennifer Ochsner,Sarah Sewall,Gregory Brooks,Rashmi Agni (2006). Elastofibroma Dorsi.
E Falidas,D Arvanitis,G Anyfantakis (2013). Painful elastofibromadorsi: a report of a case and a brief review of the literature.
J Kourda,A Ayadi-Kaddour,S Merai,S Hantous,K Miled,F Mezni (2009). Bilateral elastofibroma dorsi. A case report and review of the literature.
Jerome Pierce,Robert Henderson (2004). Hypermetabolism of Elastofibroma Dorsi on PET–CT.
O Hocar,V Buffard,N Ortonne,M Bagot (2012). Élastofibrome dorsal bilatéral.
Youssraouhamou Maouniiliass,M Meryemgridda (2023). El Absi Dorsal Elastofibroma: About Two Cases.
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.