The characteristic graph of the atom makes it possible to explain the various concepts of atomics in a general way. Each concept is interpreted by specific graphics derived from the first. About the electronic structure, the particular graphs illustrating the order and the period of the subshells which obey the stability classify them in stacks according to the abscissas and the ordinates. The distribution of the electrons of the heaviest “118th” atom leads to new methods of illustrating classification called “condensed tables” with or without period and order subshells. The distribution of electrons according to the order of the energy level of the subshells denoted “OE,” is based on a new criterion for the classification of chemical elements. This stems from arrays called condensed arrays to order. These condensed tables are summarized by numerical series whose informed reading makes it possible to explain the secrets of atomistic. The electronic structure can be worked out even by mental calculation. The counting from 1 to 7 or from 1 to 8 with the corresponding value series solves the development of the electronic structure. All the value series: 2, 8, 8, 18, 18, 32, 32, and 2, 2, 8, 8, 18, 18, 32, 30 made it possible to achieve this objective.
## I. PRESENTATION
Man has always tried to find an explanation for the complexity of the matter that surrounds him. It was first thought that the elements of matter consisted of water, earth, fire, and air. We realized over time, thanks to the improvement of experimental, chemical, and physical techniques, that matter was in reality more complex than it seemed. Nineteenth-century chemists then felt the need to order the new known elements. Mendeleev argued that in addition to periodic properties due to atomic weight, each element has its proper personality [1]. Several classifications were proposed before arriving at the current periodic table.
The periodic table of elements, introduced by Mendeleev in 1869, was based on the following statement: "the chemical behavior of the elements is a periodic function of their atomic weight. "Currently, the periodic table is constructed as a result of the electronic structure of the elements [2]. The periodicity of electronic configurations finds its origin in the periodic values of the quantum numbers $n$, $l$, and $m$. Each new period starts with a new main layer [3].
To arrive at this elaborate version of the classification of the chemical elements showing the periodicity of the chemical properties, according to the atomic weight (mass), as planned by Mendeleiev, the scientists gradually penetrated the secrets of matter. By way of illustration, Joseph John Thomson will experimentally prove the existence of the electron in 1897; Ernest Rutherford discovered the atomic nucleus in 1911[3]. The discovery of the electron will prove the chemist's intuition right.
It is easy to see that the periodicity indeed corresponds to a "period" of filling of the electronic shells one after the other, conferring chemical properties more or less similar to the elements. This periodicity has made it possible to gradually and coherently build the painting, which now bears the name of its inventor. In 2010, it had 109 elements. In 2016, the International Union of Pure and Applied Chemistry (IUPAC) validated new "candidates," bringing to 118 the number of elements in the Mendeleev Table, which has become essential for anticipating the chemical properties and advancing knowledge [4].
It is important to remember that the electronic structure is the distribution of the electrons of an atom in the different sub-shells arranged in increasing order of their energy, respecting the electronic capacity of each sub-shell and especially the number of electrons (Z atomic number). This structure is done either by tables or in general order. This electronic structure poses three (3) simultaneous problems:
- The arrangement of the subshells in ascending order of their energy level;
- The electronic capacity of the sub-layers;
- The respect for the number of electrons to leave, gives rise to repetitive additions.
The purpose of this work is to demonstrate how analytical geometry applied to the atomic model based on wave mechanics makes it possible to explain and construct the electronic structure of atoms.
In a previous article [3], it was recalled that the organization of the electronic cloud in energy levels (order) is the same for all atoms. The electronic cloud includes electronic shells subdivided into sub-shells having atomic orbitals (electronic cells). Each layer can be defined as the set of states (or electrons) characterized by the exact value of $n$; a subshell as the subset of states (or electrons) corresponding to the same value of $n$ and the exact value of $l$. An atomic orbital is defined by the quantum triplet $(n, l, m)$. However, the state of an electron is wholly determined by all four quantum numbers $(n, l, m, m_s)$ [5]. Thus the distribution of electrons in the electronic cloud of a polyelectronic atom is how its Z electrons are distributed between shells, subshells, and atomic orbitals. The knowledge of Z gives few indications on the chemical behavior of an atom, but that of its electronic configuration allows, on the contrary, to explain or to predict this behavior to a large extent. It is an essential basis for understanding reactivity and the periodic table of elements.
The electronic configuration (or electronic structure) in polyelectronic atoms makes it possible to specify the distribution of electrons in the various layers and sub-layers of these atoms in the ground state according to the increasing order of the energy level. It obeys a certain number of principles which are: the principle of stability, the principle of Aufbau, the rule of Klechkowski, the principle of exclusion of Pauli, and the rule of Hund [6].
Thus the electronic structure makes it possible to divide the electrons of an atom into two groups: the "core electrons" and the "valence electrons". These represent the identity of the element. Indeed, a good reading of the valence electrons makes it possible to list all chemical elements and to develop the different concepts of chemical language, thusdescribing their periodic properties.
The characteristic graph of the atom [7], which correlates with all the literary diagrams, explains the composition of the electronic cloud. Among the various specific graphs of the electronic cloud, two are in agreement with the rule of stability; these are those which are specific to orders and periods. These classify the different subshells either in period or in order. In these charts, the sub-layers are arranged as a stack from one order to another or from one period to the next. Within a stack, the different sub-layers are classified according to the increasing order of "n" the period; this is the Aufbau principle. The different stacks follow each other as in the usual ordination of natural numbers; they are easily and logically memorized in each case.; thus expressing the highlights of atomistic. The exploiting of the characteristic graph of the atom and the Order and Period Tables presented in the previous works will allow easy assimilation of the atomistic concepts and their use for the electronic configuration of the atoms.
The objective of this work is to propose innovations simplified and concrete for the development of the electronic structure of atoms in the ground state. Its niche is to develop creative imaginations that will make it possible to work out this structure even mentally in the place of the rather abstract classical methods [8].
## II. METHODOLOGY
### a) Description of the Electronic Structure
The electronic structure is the distribution of electrons on the shells and, or sub-shells of an atom under the principle of stability. The electrons saturate the sub-shells of the lowest energy levels and gradually move towards the sub-shells of the higher energy levels until the exhaustion of Z (atomic number). It is known that the process of saturation of the sub-layers is carried out under the rule of Klechkowski, that is to say, following the increasing order of $O_E$ ( $n + l$ ) and within the same order following "n" increasing. The different sub-layers of an order form a stack. The order of a stack is $O_E$, whatever the sub-layers compose it (Aufbau principle). Like the order, the period corresponds to a stack.
The layout of the subshells is described by graphs illustrating orders and periods [5]. From graphs 1 and 2, the distribution of the 118 electrons of the heaviest atom is carried out along the $O_{E}$ axis (the order) and the n axis (the period).
The known atoms are in a logical sequence of natural numbers ranging from 1 to 118. The distribution of the Z electrons of the element containing 118 protons makes it possible to fly over the electronic structure of all the others. The electrons of a polyelectronic atom are distributed in different populations by their energy levels whereas the nucleons exist at the same energy level. Thus from graph 1, it suffices to simply saturate the various sub-layers of each "OE" and "n" stack progressively, starting from 1 to 8 and 1 to 7, respectively. The distribution of the electrons is carried out, in this case, following a progression with variable steps at every of two stage. We get graphs 1 and 2.
### b) Electronic Structure and Order and Period Charts
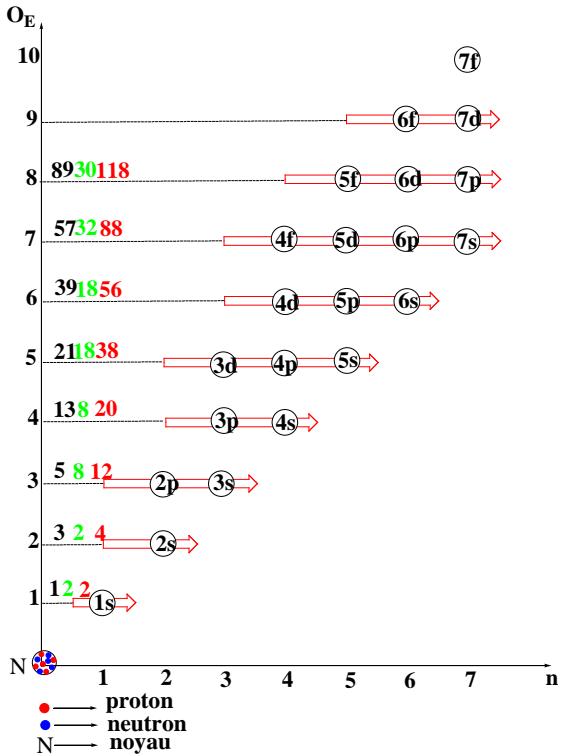 Graph 1: Electronic Structure of The Elements According to The Order
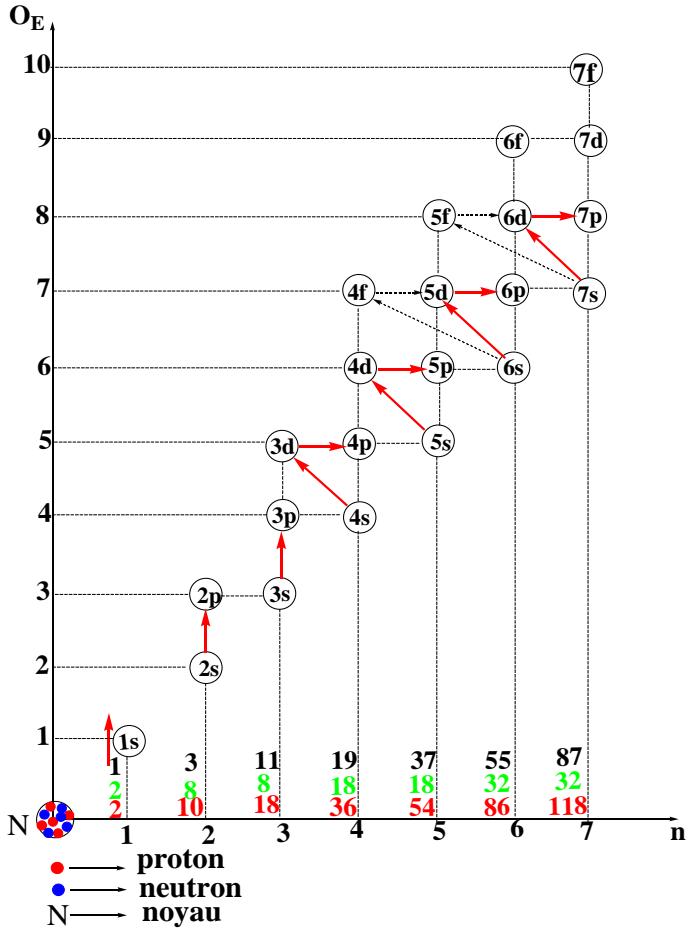 Graph 2: Electronic Structure of The Elements According to the Period
According to graphs 1 and 2, the electrons are divided into populations characterized by their order of energy or their period. The number of electrons per energy level is a specific function of the electronic capacitance of the subshells that make up the indicated level (stacking); It also recalls the different types of known arrays, and it is then identical to the number of elements saturating each of the levels. Knowledge of these values (number of populations of electrons per level) is an inexhaustible source of the secrets of atomistic, of chemical language.
## III. RESULTS
### a) Reading the two Graphs
Graphs 1 and 2 convey a set of parameters: the level orders, the periods and their subshells, the number of electrons, and the interval of the elements saturating each level. All these atomistic parameters are interdependent. From the coordinates $(O_E$ and, orn) to the last sub-layer of the stack via the limits of each interval and their amplitudes (number of elements per stack), there is a perfect correlation. This is recorded in tables called condensed tables with order and period.
### b) Condensed Arrays with Order and Period
The periods and, or (the orders) are formed by 1, 2, 3, or 4 sub-layers repeating 2 to 2 at each step change. They are: s(s) sp (ps) sdp (dps) sfdp (fdps). They are the different stacks. They are 2 to 2 at each the stepvariation of except the first in the case of periods. The electronic capacities per level or stack are: 2 8 18
32. These are also the various populations of electrons: 2(s), 8(sp), 18(sd), 32(sf) satisfying the formula of the number of electrons per level.
In practice, all the subshells from 1s to 7p are divided into seven (7) periods and, or eight (8) orders. Each of its levels has some well-defined elements per interval. This number of chemical elements defines the electronic capacity of the level. All subshells are arranged according to the principle of stability, regardless of order or period. The electronic capacitance (amplitude) per period and/or order is given by the following formula: $N_{\mathrm{e / level}} = \Sigma 2(2I + 1)$; $I$: is the secondary quantum number at each of the sub-shells of the level (stack).
For example, if $O_E = 5$ and $n = 5$, What are the numbers of elements per level and their amplitudes?
For $O_E = 5$ the subshells are 3d 4p 5s, and the amplitude $\mathsf{N}\acute{\mathsf{e}} = \Sigma 2(2\mathsf{l} + 1)\colon 10 + 6 + 2 = 18\acute{\mathsf{e}}.$
For $n = 5$ the subshells are 5s 4d 5p and the amplitude $N_{\mathrm{e}} = \Sigma 2(2l + 1): 2 + 10 + 6 = 18\mathrm{e}$.
Please note that orders are not always similar to periods and vice versa.
For example, if $O_E = 4$, we obtain "3p 4s" and the fourth period is "4s 3d 4p".
Condensed tables are very captivating and even fascinating educational tools:
- Condensed order table (along the ordinate axis)
According to the order chart, the main information is summarized in this table:
Table 1: Condensed Order Table
<table><tr><td>Order</td><td>1</td><td>2</td><td>3</td><td>4</td><td>5</td><td>6</td><td>7</td><td>8</td></tr><tr><td>Subshells</td><td>1s</td><td>2s</td><td>2p3s</td><td>3p4s</td><td>3d4p5s</td><td>4d5p6s</td><td>4f5d6p7s</td><td>5f6d7p...</td></tr><tr><td>Z</td><td>1- 2</td><td>3- 4</td><td>5- 12</td><td>13- 20</td><td>21- 38</td><td>39- 56</td><td>57- 88</td><td>89-118...</td></tr><tr><td>Type tables</td><td>2</td><td>2</td><td>8</td><td>8</td><td>18</td><td>18</td><td>32</td><td>30......</td></tr></table>
- Condensed period table (along the abscissa axis)
According to the distribution of the electrons by the period, a table is obtained analogously to that of the order:
Table 2: Condensed Period Table
<table><tr><td>Periods</td><td>1</td><td>2</td><td>3</td><td>4</td><td>5</td><td>6</td><td>7</td></tr><tr><td>Subshells</td><td>1s</td><td>2s2p</td><td>3s3p</td><td>4s3d4p</td><td>5s4d5p</td><td>6s4f5d6p</td><td>7s5f6d7p...</td></tr><tr><td>Z</td><td>1- 2</td><td>3- 10</td><td>11- 18</td><td>19- 36</td><td>37- 54</td><td>55- 86</td><td>87-118...</td></tr><tr><td>Type tables</td><td>2</td><td>8</td><td>8</td><td>18</td><td>18</td><td>32</td><td>32...</td></tr></table>
- The first line for each of the tables corresponds to the usual ordination of the natural numbers " $O_E$ " and " $n$," knowing that each number is linked to the coefficients of the different corresponding sub-shells (n, OE) are the coordinates of the sub-shells;
- The second line indicates the sequenced arrangement of the sub-shells according to the Aufbau principle and, or stability (KLECHKOWSKI
rule); it gives the electronic structure to each of the elements;
- The third line gives the sequenced ordination of all the atomic numbers of the elements by interval in each level (this is a classification of the chemical elements).
- The last line of the tables has enough meanings, including among others the types of tables, the electronic capacity by stacking, the different
populations of electrons by level, the amplitudes,thoseare to say the size (magnitude),etc...
These two very similar but different tables convey all the secrets of classification and electronic structure, which is why they are called condensed forms. They illustrate graphs 1 and 2.
Order and period condensed tables are new methods of illustrating the classification of elements. The condensed period form confirms all current periodic classifications of the chemical elements. The condensed period table summarizes all the classical period processes along the abscissa axis.
The condensed order table is also another classification process leading to entirely new forms of tables: Order tables of the elements obtained along the ordinate axis. The resulting tables are in the form of a staircase: they are also 8, 18, and 32 columns. They are carried out around a new classification criterion, namely the order " $O_E$ " instead of " $n$ " the period. Order tables or ordered tables of the elements already exist in the literature, but they continue to bear the name periodic [9]. They are different from periodic tables. The specific criteria for their formation are other from the order and from the period [2].
These new processes for illustrating classifications along the different $O_{E}$ and $n$ axes are summarized by series with period and order values, the main ones of which are, respectively:
- Series to order $\langle O_E\rangle$: 2 2 8 8 18 18 32 30
- Series to period « n »: 2 8 8 18 18 32 32
## IV. DISCUSSIONS
By an informed reading, these two series with values (amplitudes) make it possible to interpret the various secrets of atomistic. It is possible to reconstitute each of the tables by their series of values or by the coordinates of their axes. These two very simple series, as simple as telephone numbers, are logically memorized. It is possible to work out the electronic structure of each and, or all the elements mentally. Knowing that the sub-shells of each level or stack are linked to the order numbers and, or period and to the amplitudes, i.e., the different populations of electrons, it is possible or even enjoyable to mask the sub-shells. Thus these condensed tables with order and period are summarized in numeral tables, that is to say, formed only of series with values.
Table 3: Condensed Order Table
<table><tr><td>Order</td><td>1</td><td>2</td><td>3</td><td>4</td><td>5</td><td>6</td><td>7</td><td>8</td></tr><tr><td>Z</td><td>2</td><td>4</td><td>12</td><td>20</td><td>38</td><td>56</td><td>88</td><td>118...</td></tr><tr><td>Type tables</td><td>2</td><td>2</td><td>8</td><td>8</td><td>18</td><td>18</td><td>32</td><td>30......</td></tr></table>
Condensed period table (along the abscissa axis)
According to the distribution of the electrons by the period, a table analogous to that of the order is obtained:
Table 4: Condensed Period Table
<table><tr><td>Period</td><td>1</td><td>2</td><td>3</td><td>4</td><td>5</td><td>6</td><td>7</td></tr><tr><td>Z</td><td>2</td><td>10</td><td>18</td><td>36</td><td>54</td><td>86</td><td>118</td></tr><tr><td>Type tables</td><td>2</td><td>8</td><td>8</td><td>18</td><td>18</td><td>32</td><td>32...</td></tr></table>
Interpreting the series with values resulting from these condensed tables makes it possible to bring out various secrets of atomics. These three series of each of the tables are strongly interdependent.
How to make the electronic structure of the elements?
These condensed tables of order and period numerals are also new ways of illustrating the classification of elements. The electronic structure of a chemical element is as follows:
$S_{\text{elect}} = E_{\text{core}} + E_{\text{val}}$, where $E_{\text{core}}$ core electrons and $E_{\text{val}}$ valence or exchangeable electrons.
In Tables 3 and 4, the lines of Z give the abbreviated numerical form of the electronic structure by the noble gases according to the period and by the alkaline earth metals according to the order. From one stack to another, the core electrons are the value of the previous one for the next; to note,for the first stack,the core electrons are zero "0". The case is specific for the periodic classification; for the ordered classifications one, hypothetically admits as core electrons the value of the preceding for the following for the need of the analogous calculation.
First Example
Table 3, either works out the electronic structure of $Z = 47$. The chemical element $Z = 47$ is of the 5th period, whose subshells are: "5s 4d 5p". The electronic structure is then: Ecoeur = 36 (previous gas); Eval = 47-36=11é (11 electrons to leave for 5s 4d and 5p). Thus we get:36//5s24d9 (5s14d10).
The same calculation is possible with the order table except that the hypothetical core electrons contain the two (2) electrons of the "s" sub-shell of the corresponding period. The electrons of hearts, if necessary, are redistributed for the periods preceding that of the elements to obtain the whole electronic structure (complete): $1s^{2}$; $2s^{2} 2p^{6}$; $3s^{2} 3p^{6}$; $4s^{2} 3d^{10}$; $4p^{6} // 5s^{2} 4d^{9}$ ( $5s^{1} 4d^{10}$ ).
### Second Example
By this method, making the electronic structure is carried out either by masking the different sub-layers or by making them appear:
### a) Without highlighting the under layers (they are hidden)
$$
For example Z=64
$$
$$
64:2-8-8-18-18+10\left\{ \begin{array}{l} 2 \\ 7 \\ 1 \end{array} \right.
$$
### b) With subshells
$$
\begin{array}{c} Z = 6 4: 1 s ^ {2} / 2 s ^ {2} 2 p ^ {6} / 3 s ^ {2} 3 p ^ {6} / 4 s ^ {2} 3 d ^ {1 0} 4 p ^ {6} / 5 s ^ {2} 4 d ^ {1 0} 5 p ^ {6} \\\hline / 6 s ^ {2} 4 f ^ {7} 5 d ^ {1}. \end{array}
$$
Note that in the literature tables, the sublayers are placed by layer and by period. All forms of tables are interpreted by these small tables with values. Thus the arrangement of the subshells in a graph made it possible to simplify the electronic structure of the chemical elements by using either the period or the order.
## V. CONCLUSION
This work made it possible to sequence the different sub-layers of the electronic cloud either in seven parts (periodically) or in eight, according to the order. These other parts are stacks whose characteristics (coefficients and amplitudes) are linked to the abscissas and, or ordinates of the graph. At each value of these, logically emerges the nature and the number of constituent sub-layers, as well as the amplitude of the corresponding stacking. This has led to new processes for illustrating the classification of elements, which are "condensed tables with period and order". It is by clinging to basic concepts and striving to reason logically that we will gradually realize that atomistic is only a matter of common sense.
A new classification criterion has emerged. All the classical classifications took place around period "n." The new classification criterion is, this time, the order of the energy level $\mathrm{O_E}$, which leads to a new classification; this is called the ordered classification by analogy to the periodic one. The interdependence of the quantities in these condensed tables even makes it possible to mask the different sub-layers of the electronic cloud, thus leading to condensednumerical tables. A careful reading of these describes all the secrets of atomistic and classification. Through these tables, it is now possible to mentally work out even the most formidable of atomistic, the electronic structure of one or more element(s) in one line.
In this paper, results of the simplification of the establishment of the electronic structure of the elements confirming the use of an affine equation have been provided. We hope to bring soon a rigorous proof of the terminology of atomistic using the equation of the straight line, such as the methods of illustrations of the different classifications of the chemical elements at order and period, the deduction of electrons valence, and electronic jump.
Generating HTML Viewer...
References
9 Cites in Article
Scerri Eric (2020). The periodic table _ its story and its significance 2nd edition.
S Baldé (2017). Atomistic Study Described by an Affine Equation: Hypothesis of a New Table.
Ousmane Barry,Mamadou Balde,Souleymane Balde,Aboubacar Sylla (2022). Interpretation of Some Atomistic Concepts from the Characteristic Graph of the Atom.
Camille Gevaudran (2016). Sciences Les nouveaux éléments s'appelleront Nihonium, Moscovium, Tennessine et Organesson.
Aude Ganier,Claire Abou,Michel Szlazac (2016). Les défis du CEA Octobre 2016 N°211 (infographie).
Steven Zumdahl (1988). traduit de l'anglais par Serge GéribaJdi, 1997Susan E. Thomas, , De Boeck Université, N.-Y. (1992), p. 92 6 chapitres, 2-7445-0030-5..
Souleymane Balde,Ousmane Barry,Aboubacar Sylla (2022). New Overview of the Energy Classification of Underlays.
Faustino Menegus (2019). Atom’s Nuclear Structure and the Periodic Table of the Chemical Elements.
R Ouahès (1989). A Proposal for a New Periodic Table of the Elements.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Ousmane BARRY. 2026. \u201cElectronic Structure of Chemical Elements Described by the Characteristic Graph of the Atom\u201d. Global Journal of Science Frontier Research - B: Chemistry GJSFR-B Volume 23 (GJSFR Volume 23 Issue B1): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
The characteristic graph of the atom makes it possible to explain the various concepts of atomics in a general way. Each concept is interpreted by specific graphics derived from the first. About the electronic structure, the particular graphs illustrating the order and the period of the subshells which obey the stability classify them in stacks according to the abscissas and the ordinates. The distribution of the electrons of the heaviest “118th” atom leads to new methods of illustrating classification called “condensed tables” with or without period and order subshells. The distribution of electrons according to the order of the energy level of the subshells denoted “OE,” is based on a new criterion for the classification of chemical elements. This stems from arrays called condensed arrays to order. These condensed tables are summarized by numerical series whose informed reading makes it possible to explain the secrets of atomistic. The electronic structure can be worked out even by mental calculation. The counting from 1 to 7 or from 1 to 8 with the corresponding value series solves the development of the electronic structure. All the value series: 2, 8, 8, 18, 18, 32, 32, and 2, 2, 8, 8, 18, 18, 32, 30 made it possible to achieve this objective.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.