## I. INTRODUCTION
Enterococci are Gram-positive cocci that occur in pairs or short chains, nonspore forming, catalase and oxidase-negative, and facultative anaerobic and are ubiquitous microorganisms that could be present in different environments such as soil, water, sewage, and plants [11]. Enterococcus is a part of the normal flora of the intestine of a wide variety of hosts—humans and other mammals, birds, reptiles, and insects—but for reasons that are not entirely clear, they emerged in the 1970s and 1980s as a global significant public health concern and now established as the leading cause of multidrug-resistant nosocomial infections of the bloodstream, urinary tract, surgical wounds, and other sites [1,2, 4, 7,10, 12]. E. faecalis is responsible for 80-90 percent and E. faecium 5-10 percent of the human Enterococcal infections[2, 3, 4, 8, 9, 12].
It is noteworthy that the Enterococcus species (E. faecalis, E. faecium) that were recognized before 1950, are capable of causing human infection. Presently E. faecalis the most pathogenic species, but E. faeciumis considered to be the more resistant to antimicrobials in humans [1].Since then, the proportion of antibiotic resistance particularly Vancomycin and Penicillin, has increased in E. faecium isolates [2, 10, 11]. A high mortality rate of Enterococcal infections is due to increasing Multidrug resistance (MDR) of the many antimicrobials, which include the $\beta$ lactam antibiotics, the aminoglycosides, and most importantly, glycopeptides like Vancomycin warrant continued surveillance and early detection of VRE along with Minimum Inhibitory Concentrations (MIC) [12].
Other species, including Enterococcus avium, E.gallinarum, E.casseliflavus, and E.hirae, also have been isolated from human infection; such, cases are rare and considered opportunistic [1,16] Enterococcus gallinarum, and Enterococcus casseliflavus infections are of particular interest because of their intrinsic Vancomycin resistant, an antibiotic used to treat the aminoglycoside-resistant enterococcal infections that became problematic in the mid-1980s [14,16].
Therefore, the study was done to know the emergence of Multidrug resistance of Enterococcus spp. to determine the diversity of species in various clinical isolates and identify suitable drugs for improving empirical therapy, focusing on Vancomycin susceptibility.
## II. MATERIALS AND METHODS
The present study was conducted in the Department of Microbiology, Dr Lal Path labs, Delhi, India, from January 2020 to April 2022. A total of 96,526 clinical samples such as urine, pus, blood, sputum, vaginal swab, and body fluids were collected and analyzed for routine culture at the Department of Microbiology. Of these samples, 5475 isolates of Enterococci were identified, with the majority being isolated from urine samples(4373).
The samples were inoculated on various agar plates, such as MacConkey's agar, blood agar, and CHROMagar, was used for the semi-quantitative urine culture and incubated overnight at $37^{\circ}\mathrm{C}$. Identification of the Enterococcal isolates was done through colony characteristics, MALDI-TOF system (Bruker Daltonics), and VITEK-2 automated system (bioMerieux, India) using micro dilution method for antimicrobial susceptibility testing. Penicillin, Ciprofloxacin, Levofloxacin, Daptomycin, Linezolid, Vancomycin, High-level Gentamicin, Tetracycline, Teicoplanin, Tigecycline, and Nitrofurantoin were tested on Vitek XLusing the GP card P628 along with a control strain of ATCC E.faecalis 29212 and the antibiotic susceptibility pattern was interpreted as per the Clinical and Laboratory Standards Institute (CLSI) guidelines, 2022.
## III. STATISTICAL ANALYSIS
The analysis done using the statistical software package Myla (Biomerieux) to compare age, antibiotic susceptibility pattern with MIC were included as variables in this study between various clinical isolates among Enterococcus species.
## IV. RESULTS
From various clinical samples, 5475 Enterococcus species were isolated in 2 years and four months, and the infection rate was estimated to be $5.7\%$. Among these, E. faecalis was the commonest 5272 $(96.3\%)$ species isolated, followed by E. faecium171 $(3.1\%)$, and 32 $(0.5\%)$ were other Enterococcus spp. (E.casseliflavus (5), E. gallinarum (13), E. hirae (3), and E. avium (11) [Table 1].
The maximum number of isolates were from Urine 4373(79.9%), followed by the isolates from Pus 366(6.7%), fluid (4.5%), genital vaginal (4.7%), blood (3.5%), and respiratory (0.6%) [Table 1]. In this study, Urinary tract infections are mainly caused by E. faecalis and E. faecium. At the same time, there have only been 32 cases of other Enterococcus spp. (i.e., E. hirae, E. avium, E. casseliflavus and E. gallinarum) can cause clinical infections in the blood, pus, and fluid, such cases are rare and considered opportunistic. Out of 171 isolates of Enterococcus faecium accounted for most of these isolates were isolated from blood, followed by pus, urine, and fluid [Table 1].
The maximum number of Enterococcus isolates, 1832 (33.5%), were obtained from the young adults age group (13-35), followed by elderly adults (>65), 1428 (26.1%) [Fig.1] and 3359 (61.3%), of the Enterococcus spp. were isolated from the males, and 2116 (38.7%) were isolated from female patients.
Table 1: Distribution of Enterococcus species from various clinical specimens.
<table><tr><td>Enterococcus species</td><td>Blood</td><td>Pus</td><td>Fluid</td><td>Respiratory</td><td>High vaginal</td><td>Urine</td></tr><tr><td>E. faecalis (n=5272)</td><td>135</td><td>314</td><td>212</td><td>28</td><td>245</td><td>4338</td></tr><tr><td>E. faecium (n=171)</td><td>50</td><td>37</td><td>34</td><td>6</td><td>9</td><td>35</td></tr><tr><td>E. casseliflavus (n=5)</td><td>4</td><td>0</td><td>1</td><td>0</td><td>0</td><td>0</td></tr><tr><td>E. gallinarum (n=13)</td><td>7</td><td>3</td><td>2</td><td>0</td><td>1</td><td>0</td></tr><tr><td>E. hirae (n=3)</td><td>1</td><td>1</td><td>1</td><td>0</td><td>0</td><td>0</td></tr><tr><td>E. avium (n=11)</td><td>0</td><td>11</td><td>0</td><td>0</td><td>0</td><td>0</td></tr><tr><td>TOTAL (n=5475)%</td><td>197 (3.5%)</td><td>366 (6.7%)</td><td>250 (4.5%)</td><td>34 (0.6%)</td><td>255 (4.7%)</td><td>4373 (79.9%)</td></tr></table>
## Age wise prevalence of Enterococcus species
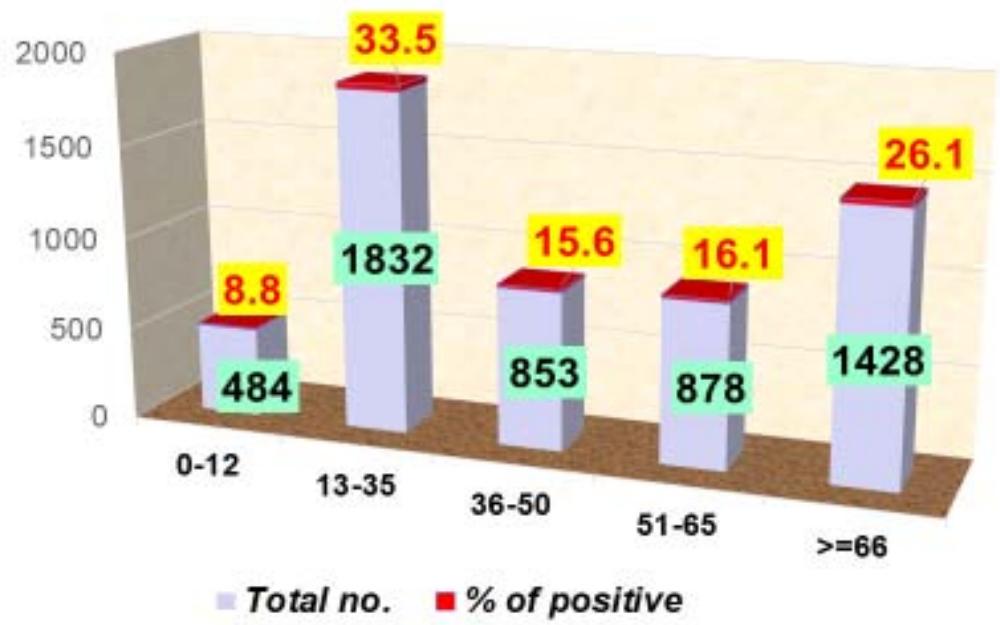 Figure 1: Age-specific distribution of Enterococcus Species
Cumulative interpretation of antimicrobial susceptibility pattern of Enterococcus species in diverse samples during Jan 2020- April 2022
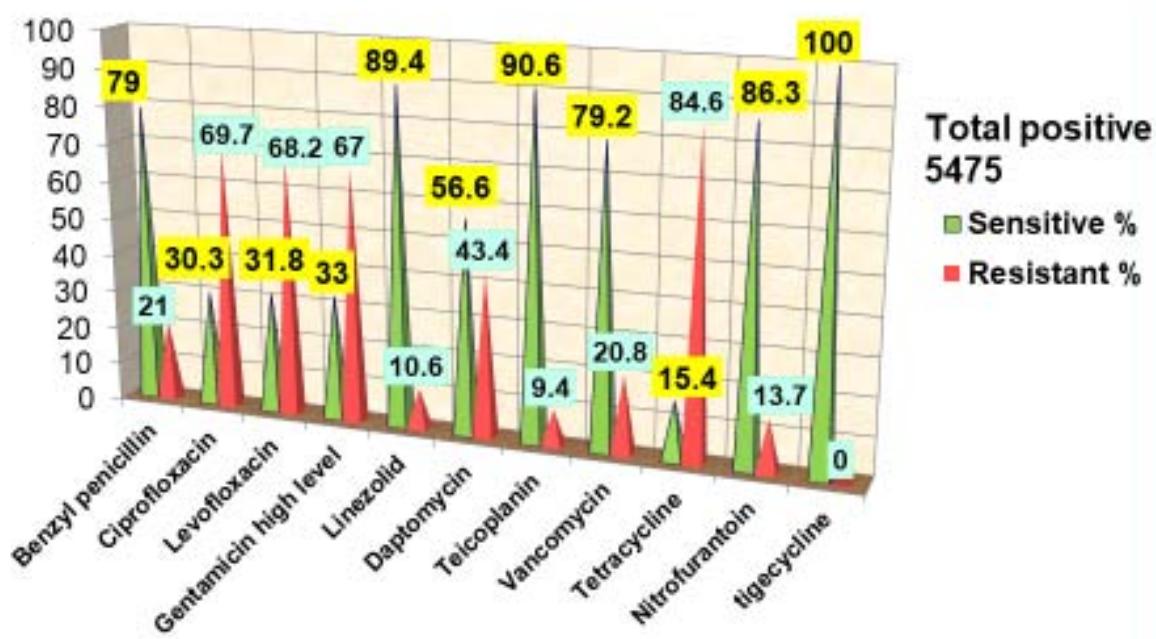 Figure 2: Cumulative interpretation of antimicrobial susceptibility pattern of Enterococcus species in various samples from January 2020 to April 2022
Antibiotic susceptibility was performed on all 5475 isolates, which were isolated over 2 years four months and the infection rate was estimated to be $5.7\%$.
In our study, we have reported $21\%$ resistance to Penicillin, and a total of 1141 $(20.8\%)$ isolates were found to be Vancomycin- resistant and had a MIC $>8$
$\mu \mathrm{g} / \mathrm{ml}$ and $< 16 \mu \mathrm{g} / \mathrm{ml}$ and $32 \mu \mathrm{g} / \mathrm{ml}$, which can be considered as Vancomycin intermediately resistant and resistant respectively. The resistance pattern of these Enterococcus isolates showed predominantly resistance to antibiotics like Ciprofloxacin (69.7%), Levofloxacin (68.2%), and Daptomycin (43.4%).
According to this data, reduced susceptibility among Enterococcus isolates was observed for High
Gentamicin Level (67%) and the highest antibiotic resistance rate (84.6%) was shown against Tetracycline. In contrast Enterococcus isolates showed a low frequency of antimicrobial resistance to Teicoplanin (9.4%), Linezolid (10.6%), and Nitrofurantoin (13.7%). However, all isolates were susceptible to Tigecycline [Figure 2].
Table 2: Difference in Antibiotic susceptibility (%) among Enterococcus species with Cumulative MIC (50/90) from various clinical samples
<table><tr><td rowspan="2">Antibiotics</td><td rowspan="2">Range</td><td colspan="2">E. faecalisn=5272</td><td colspan="2">E. faeciumn=171</td><td colspan="2">E. casseliflavusn=5</td><td colspan="2">E. avium n=11</td><td colspan="2">E. gallinarumn=13</td></tr><tr><td>S%</td><td>MIC50/90</td><td>S%</td><td>MIC50/90</td><td>S%</td><td>MIC50/900</td><td>S%</td><td>MIC50/90</td><td>S%</td><td>MIC50/90</td></tr><tr><td>Penicillin</td><td>≤ 8 - ≥16</td><td>82</td><td>2/32</td><td>18</td><td>64/64</td><td>80</td><td>2/32</td><td>50</td><td>2/64</td><td>92</td><td>2/8</td></tr><tr><td>Ciprofloxacin</td><td>≤ 0.5 - ≥4</td><td>30</td><td>8/8</td><td>16</td><td>8/8</td><td>100</td><td>0.5/1</td><td>38</td><td>8/8</td><td>42</td><td>8/8</td></tr><tr><td>Levofloxacin</td><td>≤ 1 - ≥4</td><td>32</td><td>8/8</td><td>17</td><td>8/8</td><td>100</td><td>0.5/1</td><td>25</td><td>8/8</td><td>42</td><td>8/8</td></tr><tr><td>Gentamicin high level</td><td>***</td><td>52</td><td>***</td><td>34</td><td>***</td><td>**</td><td>**</td><td>**</td><td>**</td><td>**</td><td>**</td></tr><tr><td>Linezolid</td><td>≤ 4- ≥8</td><td>90</td><td>2/4</td><td>77</td><td>2/8</td><td>100</td><td><=1/2</td><td>50</td><td><=2/8</td><td>100</td><td><=1 /2</td></tr><tr><td>Daptomycin</td><td>≤ 1- ≥8</td><td>58</td><td>2/8</td><td>**</td><td>**</td><td>**</td><td>**</td><td>**</td><td>**</td><td>**</td><td>**</td></tr><tr><td>Teicoplanin</td><td>≤ 8- ≥32</td><td>92</td><td><=0.5/4</td><td>72</td><td><=0.5/32</td><td>100</td><td>0.5/0.5</td><td>50</td><td><=0.5/ 32</td><td>100</td><td>0.5/0.5</td></tr><tr><td>Vancomycin</td><td>≤ 2- ≥16</td><td>81</td><td><=0.5/16</td><td>68</td><td>0.5/32</td><td>20</td><td>4/32</td><td>50</td><td>2/32</td><td>0</td><td>8/32</td></tr><tr><td>Tetracycline</td><td>≤ 4- ≥16</td><td>15</td><td>16/16</td><td>25</td><td>16/16</td><td>80</td><td>4/8</td><td>38</td><td>16/16</td><td>42</td><td>16/16</td></tr><tr><td>Nitrofurantoin (for urine isolates only)</td><td>≤ 16 - ≥64</td><td>89</td><td><=16/64</td><td>33</td><td>64/128</td><td>**</td><td>**</td><td>**</td><td>**</td><td>**</td><td>**</td></tr><tr><td>Tigecycline</td><td>≤ 0.5 - ≥2</td><td>100</td><td><=0.12/<=0.12</td><td>100</td><td><=0.12/<=0.12</td><td>100</td><td><=0.12<<=0.12</td><td>100</td><td><=0.12/<=0.12</td><td>100</td><td><=0.12/<=0.12</td></tr></table>
The difference in antibiotic susceptibility profile of the different Enterococcal species isolates in this study is shown in Table 2. According to this data, Infections due to Enterococcus faecalis tend to be more virulent than infections due to Enterococcus faecium, and clinical isolates of $E.$ faecalis tend to be considerably more susceptible to Penicillin than clinical isolates of $E.$ faecium (minimum inhibitory concentration for 50 and 90 percent of isolates $\left[\mathrm{MIC}_{50 / 90}\right] 2 / 32 \mu \mathrm{g} / \mathrm{mL}$ versus $\left[\mathrm{MIC}_{50 / 90}\right] 64 / 64 \mu \mathrm{g} / \mathrm{mL}$ respectively). In this study, Enterococcus species carry a more resistance to Quinolones and Tetracycline, which were isolated from various samples. Quinolones (MIC50/90 8/8) and Tetracycline activity (MIC50/90 16/16) recorded a high resistance rate against Enterococcus spp. demonstrated that $50\%$ and $90\%$ of isolates were within $8 \mu \mathrm{g} / \mathrm{ml}$ and $16 \mu \mathrm{g} / \mathrm{ml}$, respectively. Penicillin, Linezolid, Teicoplanin, and Nitrofurantoin were sensitive to more than $80\%$ of $E.$ faecalis. Another susceptibility trait that helps distinguish these two species is that isolates of $E.$ faecalis are usually susceptible to Nitrofurantoin; in contrast $E.$ faecium isolates are resistant to Nitrofurantoin had a MIC50/90 (64/128μg/ml) demonstrated that 50% and 90% of isolates were within 64μg/ml and 128μg/ml respectively [Table 2].
Vancomycin was sensitive to $81\%$ of the $E.$ faecalis and (MIC50/90 $<= 0.5 / 16$ ) demonstrated that $50\%$ and $90\%$ of isolates were within $<= 0.5\mu \mathrm{g} / \mathrm{ml}$ and $16\mu \mathrm{g} / \mathrm{ml}$ respectively whereas $(68\%)$ isolates of $E.$ faecium were found to be Vancomycin sensitive had an MIC50/90 $(0.5 / 32\mu \mathrm{g} / \mathrm{ml})$ demonstrated that $50\%$ and $90\%$ of isolates were within $0.5\mu \mathrm{g} / \mathrm{ml}$ and $32\mu \mathrm{g} / \mathrm{ml}$ respectively. Furthermore, High-level Gentamicin resistance was higher in $E.$ faecium $(66\%)$ than $E.$ faecalis $(48\%)$.
90% of E. faecalis isolates were susceptible to Linezolid and Teicoplanin in comparison to E. faecium 77% and 72% had an MIC50/90(2/8μg/ml) and
$(<=0.5 / 32 \mu \mathrm{g} / \mathrm{ml})$, respectively demonstrating that $2 \mu \mathrm{g} / \mathrm{ml}$ and $<=0.5 \mu \mathrm{g} / \mathrm{ml}$ within $50\%$ and $90\%$ of isolates were within eight and $32 \mu \mathrm{g} / \mathrm{ml}$ respectively. In addition, all Enterococcus species were found to be less than $50\%$ susceptible to Ciprofloxacin, Norfloxacin, and
Tetracycline except E.casseliflavus. Surprisingly, other Enterococcus species were sensitive to most antibiotics except Vancomycin, and Tetracycline while E. avium were resistant to all antibiotics except Tigecycline [Table 2].
Table 3: Percentage of Vancomycin -Resistant Enterococci (VRE) isolated out of 5475 Enterococcus isolates from various samples
<table><tr><td>Specimen</td><td>VR% in E.faecium + other Enterococcus spp</td><td>VR% in E.faecalis</td><td>Total VRE</td></tr><tr><td>Blood</td><td>30</td><td>44</td><td>74 (1.3%)</td></tr><tr><td>Urine</td><td>21</td><td>892</td><td>913 (16.6%)</td></tr><tr><td>Pus</td><td>20</td><td>41</td><td>61(1.1%)</td></tr><tr><td>Fluid</td><td>16</td><td>40</td><td>56 (1.0%)</td></tr><tr><td>Genital vaginal</td><td>1</td><td>29</td><td>30 (0.5%)</td></tr><tr><td>Respiratory</td><td>3</td><td>4</td><td>7 (0.1%)</td></tr><tr><td>Total</td><td>91 (1.6%)</td><td>1050 (19.2%)</td><td>1141 (20.8%)</td></tr></table>
A total of 1141 VRE included 1050 E. faecalis, 67 E. faecium, 5 E. casseliflavus isolates, 6 E. avium, and 13 E. gallinarum isolates. Among the VRE, 1050 E. faecalis (892 from urine, 44 from blood, 40 from fluid, 41 from pus, and four from respiratory). And of the total, 91 E. Faecium and other Enterococcus species isolates (30 from blood and 21 from urine, 16 from fluid, 20 from pus, and three from respiratory). Vancomycin resistance was seen in 1141(20.8%) of Enterococcus spp., Most of the VRE isolates were Enterococcus faecalis (19.2%), the rest were Enterococcus faecium with other species of Enterococcus. And in present study, the highest numbers of VRE were isolated from the urine samples (16.6%), followed by blood, pus, and fluid, respectively (as shown in Table 3).
## V. DISCUSSION
Enterococcus is one of the significant pathogens affecting all age groups. According to our knowledge, this is the first study to determine the emergence of Multidrug resistant Enterococci in Delhi, North India. In the present study, the isolation rate of $E$ faecalis was more than that of $E$ faecium. Similar results have been reported from studies done elsewhere [2, 3, 8, 9]. However, studies carried out abroad and in India has shown $E$ faecium to be responsible for a more significant of Enterococcal infections than $E$ Faecalis [1, 5].
The rate of isolation of Enterococcus spp. was higher from urine (79.9%) and pus (6.7%), followed by body fluids (4.5%) and blood (3.5%) as shown in Table1 similar to studies done in India [3, 12] and other report showed different rates of isolation of Enterococcus spp. from clinical samples, which ranged from 10 to 80% from urine, from 16 to 45% from pus, and from 3 to 36% from blood. Thus, our report and other studies indicate that variation in isolation rate depends on the geographical area and the clinical samples chosen in the study [2, 8, 9, 12, 13].
The recent emergence of $E$. Faecium and $E$ faecalis as MDR nosocomial pathogens. Hence, speciation and antibiotic susceptibility testing are necessary to detect the emergence and changing drug resistance pattern. Multidrug resistance among enterococci appears to be driven by the ubiquitous nature and other factors, such as the plasticity of their genomes, and it has the ability to translocate from the gastrointestinal tract to various tissues and organs as well as their virulence and antibiotic resistance are risk factors, and the acquisition of pathogenic microbial features [7, 16].
In our study, we have reported 21 percent of the Enterococcal isolates was resistant to Penicillin which were in concordance with other studies by the East and North part of India and abroad, who reported Penicillin resistance as 100 percent [1, 3, 5]. However, other studies have reported higher rates of Penicillin resistance in 30-80 percent [1, 2, 8, 13, 14]. Results of the present study E.faecium reported $80\%$ resistance to Penicillin which was in concordance with another study in South India [9].
According to our data, the highest resistance was seen against Tetracycline, Ciprofloxacin, Levofloxacin, and Gentamicin high Level (HLG), which is in agreement with other studies carried out in India and abroad [1,2,3,5,6,13]. In this study, $48\%$ and $66\%$ of $E.$ faecalis and $E.$ faecium isolated from diverse samples were resistant to HLG, respectively, similar to the study done in South India [12]. HLGR in our study was observed to be higher than that reported from other Indian studies [4, 14], thus reflecting more significant usage of Gentamicin in this region. Our study was concordance with Jain and Shridhar et al., [5, 12], who have reported more resistance to HLG among $E.$ faecium as compared to $E.$ faecalis isolates. Studies conducted in other regions have reported HLGR prevalence to be as high as 70 -100 percent [2, 9].
Overall, results of the present study were in agreement with previous studies, with the higher resistance to tetracycline appears in major Enterococcus spp. among $E$ faecalis, $E$ faecium likely reflecting the widespread use of these antibiotics in Delhi [2, 5, 7, 16].
Most of the studies are in correlate with present findings that Enterococci identified as the most frequent uropathogenic [3, 14]. And majority of the urine infections caused by E. faecalis isolates, $86.3\%$ of Enterococcus isolates, were found to be sensitive to Nitrofurantoin which is in agreement with the previous study [9]. Nitrofurantoin is an excellent drug against enterococcal urinary tract infection and has been used for many years. It is both bacteriostatic and bactericidal [3, 6, 14].
Vancomycin-resistant Enterococcus is a significant cause of concern it can be expected to be a major problem in the coming years as this might share its resistance gene with other bacterial strains, causing a potential risk of Vancomycin resistant gene transfer from Enterococcus to Staphylococcus aureus [3, 12]. Meanwhile the rate of resistance of Enterococcus was found to be $20.3\%$ to Vancomycin (Table 3). In present study, the highest numbers of VRE were isolated from the urine samples (as shown in Table 3). These data are supported by the study carried out by another study [6]. In a similar earlier study on Enterococcus isolates from India, and abroad, the prevalence of VRE has been reported to be between 0-30 percent [1, 2, 3, 6, 9, 13]. In our study, 1141 $(20.8\%)$ isolates were found resistant to Vancomycin, with E.faecalis $(19.2\%)$ showing higher resistance than E. faecium with other Enterococcus spp. $(1.9\%)$ as shown in Table 3. Our study was similar to Wada et al.,[15], who had also reported more resistance among E. faecalis isolates though Arabi et al.,[2]found vancomycin resistance to be greater among E. faecium isolates.
Worryingly, resistance to antibiotics are used to treat VRE, like Linezolid, Daptomycin, and Teicoplanin has left us with very few therapeutic options for Enterococcal infection. Teicoplanin resistance was found in 9.3 percent of the isolates in our study (Figure 2), with E. faecium $(28\%)$ showing more resistance as compared to E. faecalis $(8\%)$, as shown in Table 2. Teicoplanin and Linezolid resistance in our study was observed to be higher than that reported from other Indian studies [9, 4, 5, 12, 13, 14] thus reflecting more significant usage of Teicoplanin in this region.
Enterococci are considered reservoirs of antimicrobial resistance genes, which can be transferred to humans via the food chain. Other species, including Enterococcus avium, E.gallinarum, E. casseliflavus, and E.hirae, also have been isolated from human infection and were similar to other studies [3,14,16] whereas
Enterococcus gallinarum and Enterococcus casseliflavus infections of particular interest because of their intrinsic Vancomycin-resistant [14], but susceptible to other drugs but E.avium have shown sensitive against only Tigecycline.
Additionally, with increasing rates of Vancomycin resistance among Enterococcus isolates, good stewardship combined with aggressive treatment with targeted antibiotics is necessary to treat these frequently encountered infection. Our study concordance with another study that nitrofurantoin are the preferred agent for VRE in UTIs [3]. Linezolid has been shown to be an effective first-line medication for endocarditis, although it is a bacteriostatic drug, and Tigecycline may be used to treat invasive severe infections specifically considered a preferred agent for polymicrobial intraabdominal infections caused by Vancomycin-resistant Enterococci, still, should not be used for VRE bacteremia as it distributes primarily to tissues and achieves low serum concentrations [10,16].
## VI. CONCLUSION
In conclusion, Penicillin in E.faecium and High-level aminoglycoside resistance made this ineffective a treatment option for Enterococcal/infection in Delhi. Multidrug and Vancomycin-resistant enterococci which can be expected to be a significant persisting clinical problem in the coming years because of the use of Vancomycin, Linezolid, Daptomycin and Teicoplanin can increase the selective pressure of these antibiotics hence in near future it is necessary to know the antibiogram of the Enterococcal isolates in an area to formulate antibiotic policy which may help in all health care settings to contain the dissemination of the resistant. As an alternative, Nitrofurantoin can be a better treatment option for the urinary infections caused by only E. faecalis. In addition, Tigecycline has been shown to be effective as the drug of choice for Multidrug and Vancomycin-resistant Enterococcal infection in intra-abdominal and soft tissue infections in Delhi, North India.
Ethical Approval: It is not applicable.
Conflicts of Interest: There are no conflicts of interest.
Acknowledgements: We are thankful to Dr Reena Nakra, Principal Lab Director, Operations, National Reference Laboratory, Dr Lal Path Labs, Delhi for providing us operational support and Team Microbiology for technical assistance in this study.
Generating HTML Viewer...
References
19 Cites in Article
A Abamecha,B Wondafrash,A Abdissa (2015). Antimicrobial resistance profile of Enterococcus species isolated from intestinal tracts of hospitalized patients in Jimma, Ethiopia.
L Arbabi,M Boustanshenas,M Rahbar,P Owlia,M Adabi,S Koohi,M Afshar,S Fathizadeh,Ali Majidpour,A Talebi-Tahe,M (2016). Antibiotic Susceptibility Pattern and Virulence Genes in Enterococcus spp. isolated From Clinical Samples of Milad Hospital of Tehran, Iran.
J Barman,R Nath,S Lahari (2016). Drug resistance in Enterococcus species in a tertiary level hospital in Assam, India.
S Fernandes,B Dhanashree (2013). Drug resistance & virulence determinants in clinical isolates of Enterococcus species.
S Jain,A Kumar,B Kashyap,I Kaur (2011). Clinicoepidemiological profile and high level aminoglycoside resistance in enterococcal septicemia from a tertiary care hospital in east Delhi.
S Kaarthiga,J Bhavana,Mahantesh,S Yadukul (2020). Study on prevalence of Vancomycin Resistant Enterococcus and high level Gentamicin resistance among Enterococcus isolates in a Pediatric tertiary care hospital.
Beata Krawczyk,Paweł Wityk,Mirosława Gałęcka,Michał Michalik (1900). The Many Faces of Enterococcus spp.—Commensal, Probiotic and Opportunistic Pathogen.
D Mendiratta,H Kaur,V Deotale,D Thamke,R Narang,P Narang (2008). Status of high level aminoglycoside resistant<i>Enterococcus faecium</i>and<i>Enterococcus faecalis</i>in a rural hospital of central India.
Prathvi Nayak,Dhanashree Biranthabail,Shalini Shenoy,Shashidhar Kotian (2018). Antibiogram and genetic relatedness of clinical isolates of Enterococcus spp. in Mangalore, India.
Amg Prieto,W Schaik (1. 1). Unknown Title.
F Baquero,Corander,R Willems (2016). Global emergence and dissemination of enterococci as nosocomial pathogens: Attack of the clones? Front.
Sónia Ramos,Vanessa Silva,Maria Dapkevicius,Gilberto Igrejas,Patrícia Poeta (2020). Enterococci, from Harmless Bacteria to a Pathogen.
S Shridhar,B Dhanashree (2019). Antibiotic Susceptibility Pattern and Biofilm Formation in Clinical Isolates of Enterococcus spp.
S Sreeja,Sreenivasa Babu,P Prathab,A (2012). The Prevalence and the Characterization of the Enterococcus Species from Various Clinical Samples in a Tertiary Care Hospital.
B Tuhina,S Anupurba,T Karuna (2016). Emergence of antimicrobial resistance and virulence factors among the unusual species of enterococci, from North India.
Y Wada,A Harun,C Yean,A Zaidah (2020). Vancomycin-Resistant Enterococci (VRE) in Nigeria: The First Systematic Review and Meta-Analysis.
Rahat Zaheer,Shaun Cook,Ruth Barbieri,Noriko Goji,Andrew Cameron,Aaron Petkau,Rodrigo Polo,Lisa Tymensen,Courtney Stamm,Jiming Song,Sherry Hannon,Tineke Jones,Deirdre Church,Calvin Booker,Kingsley Amoako,Gary Van Domselaar,Ron Read,Tim Mcallister (2020). Surveillance of Enterococcus spp. reveals distinct species and antimicrobial resistance diversity across a One-Health continuum.
(2001). M100-S11, Performance standards for antimicrobial susceptibility testing.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Puneeta Singh. 2026. \u201cEmergence of Multidrug Resistant Enterococcus Species Isolated from Clinical Samples in North India\u201d. Global Journal of Medical Research - C: Microbiology & Pathology GJMR-C Volume 23 (GJMR Volume 23 Issue C1).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.