The article outlines a more general energodynamic theory of biosystems, considering their global and local heterogeneity with the help of added nonequilibrium parameters. The theory returns the concepts of force, speed, and power of real processes to thermodynamics and complements the theory of irreversible processes by considering the useful (reversible) component of real processes. It is shown that relaxation processes in some degrees of freedom of biosystems are accompanied by work “against equilibrium” in other degrees of freedom, which is consistent with the Darwinian concept of the “struggle for existence.” Simpler and more informative non-entropy criteria for the evolution and involution of biosystems are proposed and the unity of energy conversion processes in technical and biological systems is proven.
## I. INTRODUCTION
Despite certain successes in the study of the processes of evolution of living and inanimate nature from the standpoint of the thermodynamics of irreversible processes [1-9], bioenergy [10-15] and synergetics [16-20], there is still an obvious contradiction between equilibrium and nonequilibrium thermodynamics and the nature of biological evolution. The principle of increasing entropy, interpreted as the essence of the second law of thermodynamics, imposes "thermal death" on the Universe as a whole, and on any of its autonomous regions - degradation [21]. The interpretation of evolution as a random process [22] or the result of the spontaneous emergence of "order" from "chaos" [19], and even more so as the absorption of some kind of "negentropy" [11], did not give satisfactory results. Moreover, the very concept of "self-organization" as spontaneous ordering of an isolated system [21], as well as attempts to "explain" antidissipative (thermomechanical, thermochemical, thermoelectric, thermomagnetic, thermogalvanomagnetic) turned out to be in "blatant" contradiction with thermodynamics. threads, etc.) effects as manifestations of some kind of "synergy" [2,3].
A completely different view opens from the position of a more general and non-postulate thermodynamic theory of energy transfer and transformation processes in nonequilibrium systems [23], defended by the author in his doctoral dissertation [24] and then published in the form of a monograph "Thermokinetics" [24], recommended by the Ministry of Science and Education RF as a textbook for technical universities, and after its generalization to any form of energy in the monograph "Energodynamics" [25] - distributed by the Russian Foundation for Basic Research to university libraries. Unlike "pseudothermostatics" by W. Thomson [25] or "quasithermodynamics" by L. Onsager [26], this theory does not exclude from consideration any (irreversible or reversible) part of the phenomena being studied and covers the entire range of real processes - from quasi-static (reversible) to extremely irreversible (dissipative). This is achieved by finding the driving forces of real processes and their generalized rates directly on the basis of the law of conservation of energy in systems far from equilibrium, which opens up new possibilities for applying the method of nonequilibrium potentials to the study of biological systems and eliminates its contradiction with the laws of biological evolution.
## II. METHODOLOGICAL FEATURES OF ENERGODYNAMICS
The fundamental difference between energetodynamics and locally equilibrium thermodynamics of irreversible processes (IRP) and other field theories is the consideration of a nonequilibrium system as a whole, without breaking it up into an infinite number of elementary volumes dV, assumed to be homogeneous. This makes it possible to preserve the so-called system-forming connections that are inherent in the system, but absent in its individual parts. This is what distinguishes a living organism from a simple set of organs as macroscopic and even microscopic "subsystems" formed during such fragmentation. The realization of the hopelessness of attempts to restore the lost system-forming properties by finding "suitable integrals" was, according to A. Poincaré, "the biggest and most profound shock that physics has experienced since the time of I. Newton [28].
Another methodological feature of energodynamics is considering the opposite direction of processes in various parts (regions, phases, components) of a nonequilibrium system. This circumstance makes nonequilibrium processes irreversible even when they occur quasi-statically (infinitely slow). The inconsistency of the concept of "quasi-stativity" of equilibrium thermodynamics of R. Clausius can be seen by presenting any extensive parameter of the system $\Theta_{\mathrm{i}}$ (its mass M, the number of moles of kth substances $\mathsf{N}_{\mathsf{k}}$, entropy S, electric charge $\Theta_{\mathrm{e}}$, impulse P, its moment L, etc.) integral of its local $\rho_{\mathrm{i}} = \mathrm{d}\Theta_{\mathrm{i}} / \mathrm{dV}$ and average $\overline{\rho}_i = \Theta_i / V$ density $\Theta_{\mathrm{i}} = \int \rho_{\mathrm{i}}\mathrm{d}V = \int \overline{\rho}_i\mathrm{d}V$. It at once follows that
$$
\int \left(\rho_ {i} - \bar {\rho} _ {i}\right) d V \equiv 0. \tag {1}
$$
According to this identity, in the elements of the continuum where local equilibrium exists $(\rho_{i} - \overline{\rho}_{i} = 0)$, no internal processes $\mathrm{d}(\rho_{i} - \overline{\rho}_{i}) / \mathrm{d}t \neq 0$ are possible. This provision emphasizes the need to consider spatial heterogeneity (internal disequilibrium) in any element of the system where any processes occur.
From identity (1) it also necessarily follows that in different parts (regions, phases or components), inhomogeneous systems of oppositely directed processes (with different signs of speed $\mathrm{d}(\rho_{i} - \overline{\rho}_{i}) / \mathrm{dt}$ ) arise. This position is called in ergodynamics the "principle of counterdirectional nonequilibrium processes" [24]. It is in tune with the dialectical law of "unity and struggle of opposites" and can serve as its mathematical expression.
Another starting point of energetodynamics is the "principle of certainty of state" of nonequilibrium systems, according to which the number of independent arguments $\Theta_{i}$ of its internal energy $U$ is equal to the number of independent processes occurring in it. This principle, proven in energetodynamics "by contradiction," prevents "underdetermination" or "overdetermination" of a nonequilibrium system, i.e., tries to describe it with a missing or excessive number of parameters. An example of "underdetermination" is the local equilibrium hypothesis of I. Prigogine, according to which the volume elements of a system that is nonequilibrium as a whole are in local equilibrium (despite the occurrence of dissipative processes in them), so that their state can be described by the same number of variables as in equilibrium (despite the appearance of local "thermodynamic forces" $X_{i}$ ), and all equations of classical thermodynamics are applicable to them (despite their inevitable transition into inequalities).
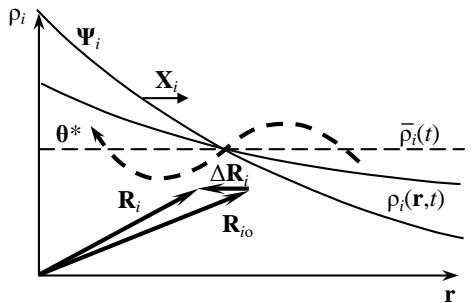
Figure 1: To the Formation of the Distribution Moment
Contrary to this hypothesis, energetodynamics proves the need to introduce added parameters for the spatial heterogeneity of the systems under study. To find them, consider an arbitrary system with a non-uniform density $\rho_{\mathrm{i}}(\mathbf{r}) = \partial \Theta_{\mathrm{i}} / \partial V$ of any extensive parameter $\Theta_{\mathrm{i}}$, considered as a quantitative measure of the i-th energy carrier (Fig. 1). As follows from the figure, when $\rho_{\mathrm{i}}$ deviates from the average value, a certain amount $\Theta_{\mathrm{i}}^*$ of the energy carrier $\Theta_{\mathrm{i}}$ is transferred from one part of the system to another in the direction showed by the dotted arrow. This causes a displacement of the center of its value from the first position $R_{\mathrm{io}} = \Theta_{\mathrm{i}}^{-1} \int \mathrm{rdV} = 0$, to the current $R_{\mathrm{i}} = \Theta^{-1} \int \rho_{\mathrm{i}} \mathrm{rdV}$. In this case, a certain "distribution moment" $Z_{\mathrm{i}}$ arises:
$$
Z_{i} = \Theta_{i} \Delta R_{i} = \int_{V} \left[ \rho_{i}(\boldsymbol{r},t) - \bar{\rho}_{i}(t) \right] r \, dV.
$$
with the shoulder $\Delta R_{\mathrm{i}} = R_{\mathrm{i}} - R_{\mathrm{io}}$ called the "displacement vector" in enerodynamics [25].
Since in a homogeneous state $\Delta R_{i} = 0$, the state of the nonequilibrium system as a whole is characterized by twice the number of state variables $\Theta_{i}$ and $\Delta R_{i}$, i.e. $U = \Sigma_{i} U_{i}(\Theta_{i}, R_{i})$ In this case, its total differential can be represented as an identity [25]:
$$
\mathrm{d}U = \Sigma\mathrm{d}U_{\mathrm{i}} = \Sigma\Psi_{\mathrm{i}}\mathrm{d}\Theta_{\mathrm{i}} + \Sigma F_{\mathrm{i}}\cdot\mathrm{d}R_{\mathrm{i}},
$$
and the total derivative of the energy of the system with respect to time $t$ is in the form
$$
\mathrm{d}U/\mathrm{d}t\equiv\Sigma_{i}\Psi_{i}\mathrm{d}\Theta_{i}/\mathrm{d}t+\Sigma_{i}F_{i}v_{i},
$$
where $\Psi_{i}\equiv (\partial U / \partial \Theta_{i})_{R^{-}}$ averaged value of potential $\psi_{i}$ (absolute temperature T and pressure p, chemical $\mu_{k}$, electrical $\varphi$, gravitational $\psi_{g}$ and other potentials); $F_{i}\equiv -(\partial U / \partial R_{i})_{\Theta^{-}}$ internal forces in their general understanding as the antigradient of the i-th form of energy; $\mathrm{v_i = dR_i / dt - speed}$ of the i-th transfer process.
The specificity of equation (3a) is that its terms no longer decide the heat or work of the $i$ -th process, as would be the case for reversible processes. On the contrary, from the very beginning it is recognized that not only entropy $S$, but also other energy carriers $\Theta_{i}$ can change in the general case both due to external energy exchange and due to internal sources during internal spontaneous processes. In this case, the terms of the first sum in expression (3a) are under conditions of constancy $\Delta R_{i}$, i.e., in the absence of redistribution processes. This means that local potentials $\psi_{i}$ in such processes change to the same extent in all parts of the system (as in homogeneous systems). Due to this, the "global" (nonequilibrium) potentials $\Psi_{i}$ buy the simple meaning of averaged values of local potentials $\psi_{i}$. This kind of change in state is reminiscent of uniform precipitation on an uneven surface. A special case of them are equilibrium (reversible) processes of heat and mass transfer of a system, the work of introducing $k$ -th substances or charge into the system, its all-round compression, etc.
The second sum of identity (3a), on the contrary, owes its origin to the spatial heterogeneity of the systems under study. Its members characterize elementary work of a different kind than in thermodynamics, which is decided by the product of the force of the i-th kind $F_{i}$ and the displacement $dR_{i}$ of the object of its application (energy carrier $\Theta_{i}$ ) caused by it. Since these forces are in conditions of constant parameters $\Theta_{i}$, then $F_{i} = \Theta_{i}X_{i}$, where $X_{i} = -(\partial U / \partial Z_{i})$. The work $dW_{i} = F_{i}\cdot dR_{i} = X_{i}\cdot dZ_{i}$ performed by the forces $F_{i}$ or $X_{i}$ consists of redistributing the energy carrier $\Theta_{i}$ throughout the volume of the system. Thus, energetodynamics introduces into consideration a new class of transfer processes that are of a vector (directional, ordered) nature.
In the particular case of homogeneous systems $(\Delta R_{i} = 0, \Psi_{i} = \psi_{i})$ expression (3) goes into the combined equation of the 1st and 2nd principles of classical thermodynamics of open systems in the form of the generalized Gibbs relation [21], which is given the form in TIP:
$$
\mathrm {d} \bigcup = \Sigma_ {i} \psi_ {i} \mathrm {d} \Theta_ {i}, \tag {4}
$$
where $i = 1,2,n$ - the number of independent forms of energy of the system.
A unique feature of ergodynamics, therefore, is that the main quantities with which TIP operates (thermodynamic forces $\mathrm{Xi}$ and flows $\mathrm{J_i} = \mathrm{dZ_i / dt}$ as generalized rates of transfer processes) are found directly from the thermodynamic identity (3b), thereby avoiding the most labor-intensive procedures for compiling an equation for the "production" of entropy.
## III. FINDING THE DRIVING FORCES AND GENERALIZED RATES OF BIOLOGICAL PROCESSES
In the "quasithermodynamics" of L. Onsager [27], which deals with relaxation processes, the scalar thermodynamic forces $\mathsf{X}\mathsf{i}^{\star}$ and "fluxes" $\mathsf{J}_{\mathsf{i}}^{\star}$ are found from the expression for the rate of entropy increased dS/dt in an adiabatically isolated system:
$$
\mathrm{dS}/\mathrm{d t} = \Sigma_{i} X_{i}^{*} J_{i}^{*},
$$
where $X_{i}^{*} \equiv \partial S / \partial A_{i}$ is the deviation of some i-th parameter of the system $A_{i}$ from its equilibrium value $A_{i0}$; $J_{i}^{*} \equiv dA_{i} / dt -$ "flows," which actually have the meaning of the generalized rate of the i-th relaxation process.
However, in equilibrium thermodynamics the parameters $A_{i}$ are obviously absent. Therefore, Onsager's theory remained a kind of formalism, unrelated to reality, until another future Nobel laureate I. Prigogine proposed moving to the study of so-called "stationary irreversible processes", where the parameters $X_{i}^{*}$ and $J_{i}^{*}$ acquire a vector nature and are maintained unchanged with the help of "external coercion" i.e. performing work $dW_{i}$ "against equilibrium" on the system. In this case, the concept of "flow" Ji acquires a very specific meaning as a parameter of the transfer process and could be found from disciplines operating with the concept of its speed, by isolating from their equations that part of the "production" of entropy $dS/dt$, $d_i S/dt$, which is responsible for dissipation and in such processes is numerically equal to the work done on the system $dW_{i}/dt$. However, this required the compilation of cumbersome equations for the balance of energy, mass, charge, momentum, entropy, etc. in order to then isolate the irreversible part of the process. This most labor-intensive part of the TIP required the user not only to have extraordinary knowledge of the relevant fundamental disciplines, but also to apply a number of hypotheses, since these disciplines were deliberately limited to the consideration of conservative (non-dissipative) systems and did not contain dissipative terms. This was the main reason teaching TIP in higher education turned out to be unrealistic due to students being unprepared for this and going beyond the permissible duration of the course.
The situation is completely different in energetodynamics, where the required forces $X_{i}$ and flows $J_{i}$ are already contained in its main identity (3b). In this case, the concept of force and generalized speed of the process buy a general physical meaning, due to which the inherent arbitrariness in their choice, due to the many ways of dividing the product $X_{i}J_{i}$ into factors, is eliminated. Thus, if the first sum (3a) includes the term TdS, which characterizes reversible heat transfer in classical thermodynamics, then the 2nd sum (3b) will have the term $X_{s}J_{s}$, where $X_{s} = -\nabla T$ is the "thermotive" force, $J_{s} = Sv_{s} -$ entropy flow. Similarly, if the first sum (3a) includes the term pdV, which characterizes the reversible work of expansion, then in the 2nd sum (3) an additional term $X_{\nu}\cdot J_{\nu}$ will appear, characterizing the power of the process of compression of some and expansion of other parts of the system, for example the ventricle and atria $(X_{\nu} = -\nabla p, J_{\nu} = -$ volumetric flow). In the same way, if the first sum (3a) includes the term $M_{k}dN_{k}$, characterizing the diffusion of the kth substance across the boundaries of the system, then in the 2nd sum (3b) the term $X_{k}\cdot J_{k}$ will appear, characterizing the power of the separation process of this substance in the cell membrane or in the dialyzer $(X_{k} = -\nabla \mu_{k}, J_{k} = N_{k}v_{k} -$ flow of the kth substance). In an analogous way, driving forces can be found in so-called "polyvariant" systems that perform other types of work in addition to expansion work. Thus, if in the 1st sum (3a) we include the term $\varphi d\Theta_{e}$, which characterizes the reversible work of introducing an electric charge $\Theta X_{k} = -$ $\nabla \mu_{k}, J_{k} = N_{k}v_{k}$ into a region with an average electric potential $\varphi$, then in the second sum (3) the term $X_{e}\cdot J_{e}$ will appear, characterizing the power of the process polarization (charge separation) in a cell membrane or in a galvanic cell $(X_{e}\equiv E = -\nabla \varphi -$ electric field strength, $J_{e} -$ electric current).
It is easy to notice that the terms of the second sum (3a) can have different signs depending on whether the system does work, or work is done on the system. This is the fundamental difference between energetodynamics and TIP, in which the terms $X_{i} \cdot J_{i}$ is always positive (as is the "entropy production" $d_{i}S/dt$ ). Meanwhile, in biosystems that consume free energy from the environment, $X_{i} \cdot J_{i} < 0$, which contradicts (5). This approach allows energetodynamics to study real processes without excluding from consideration any of their components (reversible or irreversible).
A significant advantage of energetodynamics over TIP is that it eliminates the need to draw up cumbersome equations for the balance of energy, mass, charge, momentum, entropy, etc. It is also important that energetodynamicscan reflect, at a quantitative and qualitative level, the emergence of new (acquired) properties. Indeed, the parameters $Z_{i}$ are absent in equilibrium systems $(\Delta R_{i} = 0)$ and their elements and arise only when they deviate from a homogeneous state. Thus, energetodynamicsis distinguished by considering not only the local nonequilibrium of the systems under study $(\nabla \psi_{j}\neq 0)$, but also the ability to reflect the evolution of biosystems, which consists in the appearance of new properties (degrees of freedom) in them. It will be shown below that this also makes it possible to substantiate the dialectical unity of the processes of evolution and involution [30].
## IV. CORRECTION OF THE EQUATIONS OF "PASSIVE TRANSPORT" IN BIOSYSTEMS
In "quasi-thermodynamics" by L. Onsager [27], it is postulated that each of the flows Ji linearly depends on all thermodynamic forces $\mathbf{X}_{\mathrm{j}}$ ( $j = 1,2,n$ ) acting in the system. The corresponding equations are called "Onsager's phenomenological laws":
$$
J_{i} = \sum_{j} L_{ij} X_{j}.
$$
Here $L_{ij}$ are constant (independent of forces $X_j$ ) kinetic coefficients, called phenomenological and subject to the so-called "reciprocity relations" of Onsager $L_{ij} = L_{ji}$. These relationships reflect, in his opinion, the interconnection of $Ji$ flows, which is the cause of the above-mentioned "side" effects of their "overlay."
The terms $\mathsf{J}_{\mathrm{ij}} = \mathsf{L}_{\mathrm{ij}}\mathbf{X}_{\mathrm{j}}$ of the flow $\mathsf{J}_{\mathrm{i}} = \Sigma_{\mathrm{j}}\mathsf{J}_{\mathrm{ij}}$ according to (6) have the same sign. This is natural for the case of purely dissipative processes when the total rate of approach of the system to equilibrium is the sum of the rates of individual relaxation processes. Meanwhile, a class of so-called "conjugate" processes is known, when some of them go ahead in the direction of equilibrium, while others, on the contrary, move it away from it. These are, for example, cyclic Belousov-Zhabotinsky reactions, called "chemical clocks" [31], "active transport" in biosystems (transfer of substances to the region of increased reaction affinity) [32], "upward diffusion" in metals and alloys [33], processes of concentration of matter in the Universe [30], etc. This means that at least part of the forces $\mathbf{X}_{\mathrm{j}}$ and flows $\mathbf{J}_{\mathrm{i}}$ have the opposite sign, i.e., when any i-th relaxation process occurs, the system moves away from equilibrium along other, j-th degrees of freedom. In other words, along with dissipative phenomena, processes of the opposite direction are seen in such systems. These include the phenomena of "self-organization" in biosystems, as well as the processes of structure formation in solutions and melts. Since it is possible to remove a system from a state of equilibrium only by performing work on it of an antidissipative nature, we must admit that in biosystems, along with external energy exchange, internal work is performed that is not related to the production of entropy. Equations (6) do not take this specificity of systems performing useful work into account at all. This makes consumer goods inapplicable to biological systems.
In TIP, the interpretation of the effects of superposition of heterogeneous processes also turns out to be mistaken. Indeed, if the flows $J_{i}$ in the TIP are found as derivatives with respect to time $t$ from the independent parameters $A_{i}$, then they are also independent of each other and therefore cannot interact (they nevertheless overlap). This is especially obvious for stationary states, when some of the "overlapping" flows simply disappear, and yet the effects of "overlapping" take on a maximum value. This means that the explanation of various (thermomechanical, thermoelectric, thermochemical, electromagnetic, etc. effects) in TIP because of the "superposition" or "entrainment" of heterogeneous flows, and not the summation of forces $F_{i}$ and $F_{j}$, does not correspond to the essence affairs.
The transport equations appear in a completely different light from the perspective of energetodynamics. It attracts equations of state or transfer from the outside as a kind of uniqueness conditions but does not build them into the foundations of the theory, as classical thermodynamics and thermodynamics do. This makes the mathematical apparatus of energetodynamics independent of the form of these equations and cuts the contradiction between the TIP and mechanics. In it, for each independent process one can find a single (resulting) force $\mathsf{F}_{\mathrm{i}}$ that generates the given process and disappears with its cessation [25]. The components of this force $\mathsf{F}_{\mathrm{ij}}$ differ in their physical nature, but unlike $\mathbf{X}_{\mathrm{i}}$ they have the same dimension [H]. If such (resulting) force is found, $\mathsf{L}_{\mathrm{ji}}\mathbf{X}_{\mathrm{i}} = 0$ and laws (6) take on the so-called "diagonal" form, like the equations of thermal conductivity, electrical conductivity, diffusion, etc., i.e., not having cross terms with $\mathrm{i} \neq \mathrm{j}$ [34]:
$$
J_{i} = L_{ii}F_{i} = L_{ii}\Sigma_{j}F_{ij}.
$$
In this case, the Onsager reciprocity relations are fulfilled trivially $(\mathsf{L}_{\mathrm{ij}} = \mathsf{L}_{\mathrm{ji}} = 0)$ and become redundant, and with them the requirement of linearity of laws (6), necessary for the fulfillment of these relations, disappears. This means that the phenomenological coefficients $L_{ii}$ in (7) can be arbitrary functions of the variables $\Theta_{i}$ and forces $F_{\mathrm{iji}}$ and the laws (6) in the general case are nonlinear. As shown in [24], the laws of passive transport of k-substances take the form:
$$
J _ {k} = L _ {k} X _ {k} = - L _ {k} \nabla \mu_ {k}, \tag {8}
$$
where $\mathsf{L}_{\mathsf{k}}$ are the coefficients of osmotic diffusion of the k-th substance, depending on the fields of temperature, pressure, and concentration of all independent components of the system; $\nabla \mu_{\mathrm{k}}$ is the gradient of chemical potential in the membrane. Meanwhile, in the laws of diffusion proposed by Onsager himself, the sum $\nabla \mu_{\mathrm{k}}$ appears, as in (5) [27]. With the diagonal form of laws (7.8), their nonlinearity, due to the variability of the coefficients $\mathsf{L}_{\mathsf{k}}$, no longer prevents the detection of superposition effects. Let us demonstrate this using the example of a biological membrane that is permeable to the kth substance. Expanding the expression for the total differential of the chemical potential $\mu_{\mathrm{k}}$ as a function of temperature, pressure, and concentrations of all j - th independent components of the system $(j = 2.3, K)$, we have:
$$
\mathrm{d}\mu_{\mathrm{k}} = \left(\partial\mu_{\mathrm{k}}/\partial T\right)\mathrm{d}T + \left(\partial\mu_{\mathrm{k}}/\partial p\right)\mathrm{d}p + \Sigma_{\mathrm{j}} \left(\partial\mu_{\mathrm{k}}/\\partial c_{\mathrm{j}}\right)\mathrm{d}c_{\mathrm{j}}. \tag{9}
$$
This means that the transport equation (6) for discontinuous media in their integral form has the form:
$$
J _ {k} = - L _ {k} \left[ \left(\partial \mu_ {k} / \partial T\right) \Delta T + \left(\partial \mu_ {k} / \partial p\right) \Delta p + \Sigma_ {j} \left(\partial \mu_ {k} / \partial c _ {j}\right) \Delta c _ {j} \right], \tag {10}
$$
where $\Delta T$, $\Delta p$, $\Delta c_{j}$ are differences in temperature, pressure, and concentration of j-th substances on the membrane. The terms of this expression represent the components of the resultant force $X_{k} = -\Delta \mu_{k}$, the first of which is responsible for the phenomenon of thermal diffusion (substance transfer due to temperature difference), the second - for the phenomenon of barodiffusion (substance transfer due to pressure difference), and the third - for the phenomenon of ordinary (concentration) diffusion. The mutual compensation of these components of the resulting force $(X_{k} = 0)$ is the reason for the onset of a stationary state, which would be more correctly called the state of "partial equilibrium". At the same time, the stationary effects themselves such as $\Delta T / \Delta p$, $\Delta T / \Delta c_{j}$ and $\Delta p / \Delta c_{j}$ are obtained in energetodynamics because of the "superposition" of heterogeneous forces and not flows.
From (10) at $J_{k} = 0$, regardless of the value of $L_{k}$, the well-known expression for the stationary effect of the appearance of the so-called osmotic pressure $\Delta p$ in a binary isothermal system (the first part is the solvent) directly follows:
$$
\left(\Delta p / \Delta c _ {2}\right) _ {\text{set}} = - \left(\partial \mu_ {1} / \partial c _ {2}\right) / \left(\partial \mu_ {1} / \partial p\right), \tag{11}
$$
where $\Delta c_{2}$ is the stationary difference in the concentration of the dissolved substance on both sides of the biological membrane.
This made it possible to propose a method for studying superposition effects in nonlinear systems [23], which opens the possibility of studying the kinetics of processes in biological systems that are far from equilibrium.
## V. SUBSTANTIATION OF THE RELATIONSHIP BETWEEN CHEMICAL REACTIONS AND METABOLIC PROCESSES
When applying TIP to chemical reactions, another contradiction of this theory was discovered, this time with the Curie principle, which sets up the conditions for the conservation of bonds under various transformations of the coordinate system in crystallography. About transfer and relaxation processes, this principle states that the generalized rate of any i-th process $J_{i}$ in Onsager's laws (6) can depend only on thermodynamic forces $X_{j}$ of the same (or even) tensor rank [1,2]. This means that chemical reactions described in TIP by scalar terms of the type $\Sigma_{r}A_{r}d\xi_{r}$ (where $A_{r}$ is the standard chemical affinity of the r-th chemical reaction, $\xi_{r}$ is the degree of its completeness) cannot interact with metabolic processes that have a vector nature. Meanwhile, it is known that metabolism plays a decisive role in the vital processes of biosystems. To resolve this contradiction, I. Prigogine put forward the theory of "stationary coupling", in which the fact of the presence of active transport of substances through biological membranes due to the occurrence of chemical reactions on them was explained by the specificity of stationary processes with their inherent relationships between the costs of individual reagents. However, this did not solve the problem since the mentioned relationship between chemical reactions and metabolic processes was preserved in non-stationary processes. The solution to this problem is given by the basic identity of energetodynamics in the form (3a) and (3b). If in the first sum (3a) the term $\Sigma_{\mathrm{r}}\mathrm{A}_{\mathrm{r}}\mathrm{d}\xi_{\mathrm{r}}$ appears, describing the r-th scalar chemical reactions in homogeneous media, then in the second sum (3b) additional terms of a vector nature $\Sigma_{\mathrm{r}}\mathrm{X}_{\mathrm{r}}\cdot \mathrm{J}_{\mathrm{r}}$ will appear, describing the same reactions, but carried out in a flow (in flow reactors, Van't Hoff boxes, cell membranes, etc.), where $\mathbf{X}_{\mathrm{r}} = -\nabla (\mathbf{A}_{\mathrm{r}}\xi_{\mathrm{r}})$, where $\mathbf{A}_{\mathrm{r}}\xi_{\mathrm{r}}$ is the current (local) value of the chemical affinity of the r-th chemical reaction in a given section of the flow reactor; $\mathbf{J}_{\mathrm{r}}$ is the flow of reagents participating in it. Indeed, for steady-state reactions, the term $\Sigma_{\mathrm{r}}\mathrm{A}_{\mathrm{r}}\mathrm{d}\xi_{\mathrm{r}}$ can be represented as $\Sigma_{\mathrm{r}}[\partial (\mathbf{A}_{\mathrm{r}}\xi_{\mathrm{r}}) / \partial \mathbf{R}_{\mathrm{m}}]$ dRm = - $\Sigma_{\mathrm{r}}F_{\mathrm{r}}\cdot dR_{\mathrm{m}}$ where $F_{\mathrm{r}} = -\nabla (A_{\mathrm{r}}\xi_{\mathrm{r}})$ is the local value of the driving force of the r-th flow chemical reaction; $\mathsf{R}_{\mathfrak{m}}$ is the coordinate of the "reaction front" in the flow reactor. In this case, the laws of active transport of substances in membranes take the form:
$$
J_{m} = - L_{m} \sum_{r} \nabla (A_{r} \xi_{r}),
$$
where $J_{m} = \Sigma_{k}N_{k}dR_{m} / dt$ is the flow of chemically reacting substances through the biological membrane. Thus, under conditions of spatial separation of reagents (as in a Van't Hoff box), chemical reactions buy a directional (vector) character, which decides their interaction with metabolic processes in full accordance with the Curie principle. In the absence of transfer of reagents in the field of intermolecular forces, chemical reactions inevitably buy a dissipative character, which is considered in the TIP by assigning the term $\Sigma_{i}A_{i}d\xi_{i}$ to heat sources. This removes one of the main contradictions between consumer goods and bioenergy [24].
## VI. NON-ENTROPIC CRITERIA FOR THE EVOLUTION AND INVOLUTION OF BIOLOGICAL SYSTEMS
In classical thermodynamics and in thermodynamics there are no parameters or state functions that could be sufficiently general and strict criteria for the development (ontogenesis) and evolution (phylogeny) of biosystems. Entropy S is inapplicable for this purpose, since in biosystems exchanging energy and matter with the environment, it can change due to heat or mass transfer in the absence of chemical reactions, or, on the contrary, remain unchanged if the system moves away from equilibrium or approaches it due to useful (reversible) work. In particular, the entropy of a biosystem can increase when more organized biomass is added to it, although in this case such an "extended" system only moves away from equilibrium.
As for the known "global" thermodynamic potentials such as the Helmholtz free energy $F$ or Gibbs free energy $G$, they are defined only for closed homogeneous systems and are not applicable in the boundary conditions specified by matter flows [27]. Nor can its exergy (technical performance) [35] serve as a measure of the orderliness of a biosystem, since in extended systems it depends on environmental parameters, as well as on energy coming from outside in the process of performing work, and therefore is not a function of the state of such systems.
The so-called "production" of entropy $\mathrm{d}S/\mathrm{d}t$ (the rate of its increase due to irreversibility) also does not meet these requirements, since this indicator has a minimum only for stationary states of linear systems, and then only near equilibrium [36].
Energodynamics again suggests a way out of the situation. The inhomogeneity parameters $Z_{i} = \Theta_{i}\Delta R_{i}$ and $X_{i} = -\partial U / \partial Z_{i}$ introduced in it, expressed by potential gradients $-\nabla \psi_{i}$ averaged over the volume of the system $V$, are already such criteria. The advantage of such (non-entropy) criteria is that they are able to reflect the evolution of the system for each of its inherent degrees of freedom (mechanical, thermal, pressure, chemical, electrical, etc.), and are able to reflect not only its approach to equilibrium, but also removal from it:
$$
dR_{i}, dZ_{i}, |dX_{ ext{i}}| > 0 (\text{эволюция}),
$$
$$
dR_{i}, dZ_{i}, |dX_{i}| < 0 (ИНВОЛЮЦИЯ),
$$
These criteria for the evolution and involution (degradation) of systems are suitable not only for biological, but also for any other systems. They are simpler, more visual, and informative, since they allow us to monitor the behavior of each degree of freedom of systems separately, which entropy criteria cannot do. This also applies to evolution in various parts (regions, phases, and components) of the system, which makes it possible to detect the opposite direction of processes in them. Such criteria make it possible to distinguish the stationary states of nonequilibrium systems from the state of partial equilibrium, in which not only their differentials, but also these nonequilibrium parameters themselves vanish:
$$
dR_i, dZ_i, d|X_i| = 0 (стационарный процесс),
$$
$$
R_i, Z_i, |X_i| = 0 (частичное равновесие)
$$
The ability to distinguish partial (incomplete) equilibrium from complete (characterized by the cessation of all processes) allows one to distinguish the relaxation time of different subsystems of the body, which entropy criteria also cannot do. Finally, since in isolated systems all processes are spontaneous, the presence of non-entropy criteria makes the concept of self-organization meaningful, if we mean not the system, but its individual degrees of freedom. All this gives researchers a simple and extremely informative tool for analyzing evolutionary problems.
## VII. UNITY OF THE LAWS OF ENERGY TRANSFORMATION IN TECHNICAL AND BIOLOGICAL SYSTEMS
One of the most important achievements of energetodynamics is the establishment of the unity of the laws of energy conversion in thermal and non-thermal, cyclic, and non-cyclic, direct, and reverse machines. Let us now show that this unity extends to biological systems, including muscular movers. To this end, consider the expression $\Sigma_{\mathrm{i}}\mathbf{X}_{\mathrm{i}}\mathbf{J}_{\mathrm{i}} = 0$ which follows from the energetodynamic identity 3b in the steady process (dU/dt =0, dΘi/dt =0) of energy conversion of the i-th form of energy into the j-th. It implies numerical equality and the opposite sign of the power of the process of transforming the i-th form of energy into the j-th:
$$
\mathrm{X}_i\cdot\mathrm{J}_j = -\mathrm{X}_j\cdot\mathrm{J}_j
$$
This means that previously independent flows and forces in energy conversion have a relationship $\mathrm{J_i / X_j} = -\mathrm{J_j / X_i}$, independent of their direction in space. At the same time, the laws of thermal conductivity, electrical conductivity, diffusion, etc. give way to more complex equations reminiscent of L. Onsager's laws of relaxation.
$$
J_{i} = \Sigma_{i} L_{i j} X_{j},
$$
but differing from them in the antisymmetric nature of the matrix of its phenomenological coefficients $L_{ij}$. For the considered case of a two-stream system, they have the form:
$$
J_{i} = L_{ij} X_{i} - L_{ij} X_{j}.
$$
$$
J_{j} = L_{ji} X_{i} - L_{ji} X_{j}.
$$
This nature of the phenomenological laws of the energy transformation process becomes more understandable when considering, for example, a welding transformer, which converts the electrical energy of one voltage $X_{i}$ into a lower one $X_{j}$ while simultaneously increasing the current strength $J_{i}$ to $J_{j}$. As is known, it clearly distinguishes between the "no-load" modes, in which the current in the primary circuit $J_{j} \rightarrow 0$, and the voltage in the secondary circuit $X_{j}$ reaches its maximum value $X_{j0}$ (open circuit voltage), and the "short circuit" mode, when $X_{j} \rightarrow 0$, and the current $J_{j}$ is maximum and equal to the "short circuit current" $J_{jk}$. Moreover, as the overcome forces $X_{j}$ increase (approaching the "no-load" mode), the current in both the primary $J_{i}$ and the secondary circuit $J_{j}$ decreases. This is reflected by laws (19) and (20).
An analogue of such an energy converter in biological systems is a muscle element - a fibril, which can contract when a chemical reaction is started, i.e., converting chemical energy into mechanical energy. In it, the analogue of the primary energy carrier flow $J_{i}$ is the flow of reagents $J_{r}$ of a given chemical reaction, and the secondary flow $J_{j}$ is the impulse of the contracted muscle. Each of these flows, in accordance with (19) and (20), depends on both forces, the first of which $X_{i}$ in this case is the current value of the affinity of the chemical reaction $X_{i} = A_{i} \xi_{r}$, equal to the product of the standard affinity of this reaction $A_{r}$ and the degree of its completeness $\xi_{r}$, and as $X_{j}$ - the force of fibril contraction. In this case, the analogue of "idling" ( $J_{j} = 0$ ) is the so-called "unloaded muscle contraction" mode, and the "short circuit" mode ( $X_{j} = 0$ ) is the so-called "isometric muscle contraction" mode.
Expressing $\mathbf{X}_{\mathrm{jo}}$ and $\mathbf{J}_{\mathrm{jk}}$ through phenomenological coefficients $L_{\mathrm{ij}}$ (assuming their constancy), under conditions of constant forces $\mathbf{X}_{\mathrm{i}}$, laws (19) and (20) can be represented in a dimensionless form that does not contain phenomenological coefficients:
$$
X_{j}/X_{j o} + J_{j}/J_{j k} = 1.
$$
The applicability of these equations to biological systems is confirmed by the experimentally found Hill kinetic equations, which quite accurately describe the characteristics of muscles taken from various animal species [5]. The load characteristics of muscular movers constructed on their basis are shown in Figure 2.
Curves 1, 2, 3, 4 in this figure correspond to varying degrees of nonlinearity of Hill's kinetic equations. In linear systems, the dependence of $\mathbf{X}_j / \mathbf{X}_{j_0}$ on $\mathbf{J}_j / \mathbf{J}_{jk}$ is expressed by a straight line intersecting the ordinate axes at $\mathrm{J}_j / \mathrm{J}_{jk} = 1$ and $\mathbf{X}_j / \mathbf{X}_j = 1$ (curve 1). It is easy to notice that precisely this character follows from the kinetic laws (21). Thus, the work of the fibril as an element of any muscle is subject to the same laws as technical energy converters.
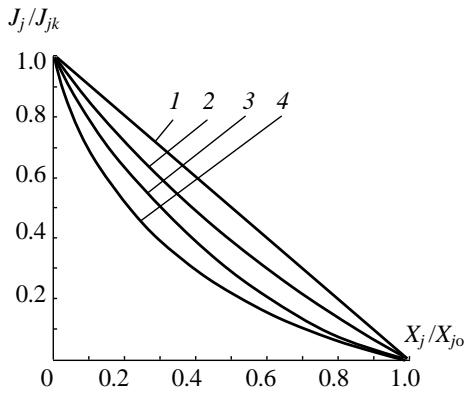
Figure 2: Load Characteristic of the Muscular Propeller
This unity makes it possible to use the theory of similarity of energy installations developed within the framework of energetodynamics [37] to analyze biological energy converters. Based on equation (21), this theory introduces a number of dimensionless criteria for the similarity of linear energy-converting systems. One of them, the "constructiveness criterion" $\Phi = L_{\mathrm{ii}}L_{\mathrm{jj}} / L_{\mathrm{ij}}L_{\mathrm{ji}}$, is similar to the ratio of reactive and active resistances, known in radio engineering as the quality factor of the circuit, and, up to the temperature multiplier, coincides with the so-called "quality factor" $\Phi$, introduced by A. Ioffe when analysis of thermoelectric generators (TEG) as their general characteristics. Its value ranges from zero to infinity ( $0 < \Phi < \square$ ), increasing with an increase in "reactive" resistances $R_{\mathrm{ji}} = L_{\mathrm{ii}}^{-1}$ (from the payload side) and a decrease in "active" resistances (from the side of dissipation forces) $R_{\mathrm{ii}}$ and $R_{\mathrm{jj}}$. Like thermal resistances in the theory of heat transfer, these resistances depend on the properties of muscle tissue and their cross-section, composition, etc.
Another dimensionless criterion $\mathsf{B} = \mathsf{J}_{\mathrm{j}} / \mathsf{J}_{\mathrm{jk}} = 1 - \mathsf{X}_{\mathrm{j}} / \mathsf{X}_{\mathrm{jo}}$, called "relative load", is composed of boundary conditions specified by the value of forces $\mathsf{X}_{\mathrm{j}}, \mathsf{X}_{\mathrm{jo}}$ or flows $\mathsf{J}_{\mathrm{j}}, \mathsf{J}_{\mathrm{jk}}$. It changes from zero in the "idle" mode ("unloaded muscle contraction") to one in the "short circuit" mode ( $\mathsf{X}_{\mathrm{j}} = 0$ ). As a determined criterion, this theory uses the "power" efficiency $\eta_{N} = N_{j} / N_{i}$, expressed by the ratio of the output power $N_{j}$ and the input power $N_{i}$. This efficiency is numerically equal to the so-called "exergetic efficiency" and complements this concept by considering the kinetics of the energy conversion process through the transition to exergy flows. It considers all types of losses and therefore most fully reflects the perfection of the converter, i.e., the degree to which it realizes the capabilities that nature provides. Using these criteria, expression (21) can be given the form of a criterion equation for the energy conversion process:
$$
\eta_{\mathrm{N}} = (1 - \mathrm{B}) / (1 + 1 / \mathrm{B} \Phi).
$$
Criterion equation (22) allows us to construct a universal load characteristic that expresses the dependence of this efficiency $\eta_{N}$ on the power Nj and load B of the energy converter at varying degrees of its design perfection F (Figure 3) [37]. The solid lines on it reflect the dependence of the efficiency $\eta_{N}$ on the load criterion B for various values of the quality factor factor $\Phi$, and the dash-dotted line shows the dependence of the output power of the installation Nj on it. These characteristics reveal a circumstance unknown to classical thermodynamics - the efficiency $\eta_{N}$ goes to zero twice: at idle ( $B = 0$ ) and in the "short circuit" mode ( $B = 1$ ). Another, also previously unknown, application is to demonstrate that the loads corresponding to maximum efficiency and maximum power diverge more noticeably the more advanced the energy converter is. In the absence of any energy losses (from friction, heat generation, all kinds of "leaks" of reagents, losses during no-load operation of the installation, etc., i.e., at $\Phi = \infty$ ), the power efficiency of the installation increases linearly with decreasing load, and at $B \to 0$ reaches, as one would expect, one. This case corresponds to an ideal Carnot machine, which has the highest thermodynamic efficiency, but has negligible power. This shows the existence of a best operating mode for each type of energy converter, including a muscular propulsion device. Universal characteristics facilitate the search for such a regime and allow the results of research on one of them to be transferred to others. Thus, a significant step is taken towards bringing the results of theoretical analysis of energy converters closer to reality.
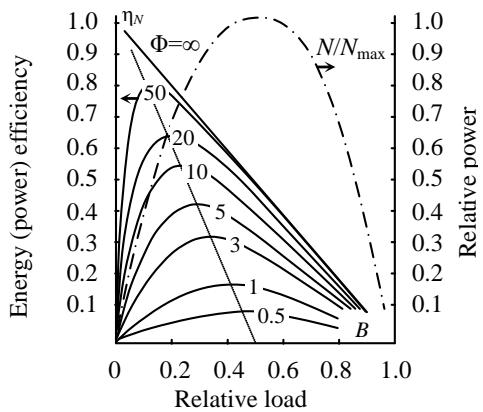
Figure 3: Universal Load Characteristics of Linear Energy Converters Systems
## VIII. SURVIVAL AS THE BASIC LAW OF EVOLUTION OF BIOSYSTEMS
Among the numerous nontrivial consequences of energetodynamics [38], associated with its introduction of nonequilibrium parameters $X_{i}$ and $R_{i}$, is the possibility of studying the kinetics of relaxation processes in systems that simultaneously perform useful work. This allows us to raise the question of the life expectancy of living organisms and their reproductive period, which the "quasi-thermodynamics" of L. Onsager and TIP could not do. Meanwhile, this is precisely what decides the life expectancy and its reproductive period of bioorganisms. According to identity (3b), after isolating a nonequilibrium system $(\dot{U} \equiv \mathrm{d}U / \mathrm{d}t = 0)$, processes of interconversion of energy occur in it until complete equilibrium occurs (all $\mathrm{Ji} = 0$ ), subject to the condition
$$
\Sigma_{i} X_{i} \cdot J_{i} = 0.
$$
Let us now consider that a change in any parameter $Z_{i}$ can be caused not only by the relaxation of a given degree of freedom, but also by the external $dW_{i}^{e} = F_{i} \cdot dr_{i}$ and internal $dW_{i}^{u} = X_{i} \cdot dZ_{i}$ work by the forces $F_{i}$ and $X_{i}$. This circumstance can be expressed by equations like the entropy balance equations $dS = d_{e}S + d_{u}S$ proposed by I. Prigogine:
$$
dZ_{i} = d_{e}Z_{i} + d_{u}Z_{i} + d_{r}Z_{i},
$$
where $d_{e}Z_{i} = dW_{i}^{e} / X_{i}$; $d_{u}Z_{i} = dW_{i}^{u} / X_{i}$; $d_{i}Z_{i} = dW_{i}^{r} / X_{i} < 0$ components of the total change in the parameter $Z_{i}$ caused by external Wie, internal Wiu and dissipative work $W_{i}^{r}$, respectively.
So, the flow $J_{i} = \frac{dZ_{i}}{dt}$ as the generalized rate of any i-th transfer process includes external $J_{i}^{e}$, reversible internal $J_{i}^{u}$ and dissipative internal $J_{i}^{r}$ components:
$$
J_{i} = \frac{d}{dt}Z_{i} + \frac{d}{dt}Z_{i} + \frac{d}{dt}Z_{i} = J_{i}^{e} + J_{i}^{u} + J_{i}^{r}
$$
In this case, condition (24) takes the form in isolated systems $(J_{i}^{e} = 0)$:
$$
\Sigma_ {i} X _ {i} \cdot \left(J _ {i} ^ {u} + J _ {i} ^ {r}\right) = 0. \tag {26}
$$
It follows that if relaxation processes $(\mathsf{J}_i^{\mathsf{r}}\neq 0)$ occur in the system, oppositely directed (antidissipative) processes of performing internal work "against equilibrium" will also occur in it. In energodynamics, this position is called the "principle of counterdirectivity" of nonequilibrium processes, which can be considered as a mathematical expression of the law of "unity and struggle of opposites" in dialectics [39].
In this case, this principle means that the approach to equilibrium of some (i-th) degrees of freedom of the system $(X_{i} \cdot J_{i} > 0)$ is accompanied by the removal of other, j-th degrees of freedom $(X_{i} \cdot J_{i} < 0)$ from it. This implies the unity of the processes of evolution and involution (degradation) of nonequilibrium systems. Unlike classical thermodynamics, which cannot say anything about the rate of approach of a biosystem to equilibrium, ergodynamics allows us to raise the question of the time of approach of a system to a state of equilibrium, depending on the distance of the biosystem from the state of equilibrium. According to (3b), under comparable conditions, the rate of relaxation of the system prescribed by the second law of thermodynamics depends on the rate of reversible processes $J_{i}$. In this case, one can compare the rate of approach to equilibrium of two arbitrary biological systems of varying complexity (with different numbers of degrees of freedom). If in an arbitrary system there are no reversible processes associated with the performance of internal work "against equilibrium" $(J_{i}^{u} = 0)$, then the rate of its approach to equilibrium will be equal to
$$
\dot{U}_{r} = - \Sigma_{i} X_{i} \cdot J_{i}^{r}.
$$
In the presence of reversible work $W_{i}^{u}$, this speed is decided by expression (26). Comparing (23) and (26), considering the work of $W_{i}^{u}$, we find that their ratio is decided by the expression:
$$
\dot {U} / \dot {U} _ {r} = 1 + \Sigma_ {i} X _ {j} \cdot J _ {j} ^ {u} / \Sigma_ {i} X _ {i} \cdot J _ {i} ^ {r}. \tag {28}
$$
This ratio can be either greater or less than one depending on the sign of the sum $\Sigma_{i}X_{i}\cdot J_{i}^{u}$, since $\Sigma_{i}X_{i}\cdot J_{i}^{r}$ is always positive. If $\Sigma_{i}X_{i}\cdot J_{i}^{u} < 0$, i.e., work is done in the system "against equilibrium," then the rate of approach of such a system to equilibrium decreases in comparison with a system where such processes are absent:
$$
\dot{U}/\dot{U}_{r} = 1 - \Sigma_{i} X_{i} \cdot J_{i}^{\mathrm{u}} / \Sigma_{i} X_{i} \cdot J_{i},
$$
Among the macroprocesses in which this kind of work is carried out is the so-called "active transport" of substances, which leads to the accumulation of reagents with a high Gibbs energy in the corresponding organs. These are the above-mentioned processes of "upward diffusion" in alloys, as well as the so-called "conjugate chemical reactions." Some of them, like the cyclic reactions of Belousov-Zhabotinsky ("chemical clock") or the process of circulation of matter in the Universe, can continue indefinitely. All processes of this kind arise only at a certain stage of their evolution. The lifespan of biosystems, as well as their reproductive period, depends on their intensity, which affects the evolution of the entire subsequent population of this type of bioorganism. When the work "against equilibrium" done by external forces $\mathrm{dW_j^e / dt} = X_j\cdot J_j^e$ becomes equal to the work of a dissipative nature, the so-called "stationary state" of the nonequilibrium system will occur.
Here lies the key to understanding the general direction of the evolution of a biological system, understood as a transition from simple to complex. This direction of evolution is not imposed by a "higher mind" - it is a consequence of purely physical reasons, reflected in the energodynamic principle of "counterdirectivity" of nonequilibrium processes. These are any processes leading to the ordering of the system, its acquisition of new properties (increasing the number of degrees of freedom), complication of the structure, etc.
Since it is precisely this environment (including the field form of matter) that is responsible for the emergence of the biosystem and its further evolution, the advantage in extending the reproductive period is given to those systems in which mutations of hereditary characteristics lead to the complication of the system and the consolidation of new degrees of freedom and forms of interaction of the biosystem with this environment. This position can be briefly formulated in the form of the "principle of survival": "The more complex the biosystem and the more intense the evolutionary processes in it, the longer their life expectancy." This position is so general that it can be considered the basic law of biological evolution. It is easy to show that this law corresponds to the well-known "triad" of Darwin's doctrine of evolution - adaptability, variability, and heredity [40]. He also removes the "blatant contradiction between thermodynamics and biological evolution" emphasized by I. Prigogine. At the same time, it also debunks the myth of "the emergence of order from chaos" [20], which contradicts classical thermodynamics, since maintaining "order" requires the expenditure of a certain external work, due to the absence of which the ordered energy of the system decreases. In other words, "order" in the system arises not from chaos, but due to a higher order in its environment. This fundamentally changes our worldview.
## IX. CONCLUSION
1. The introduction of the missing parameters of spatial heterogeneity into thermodynamics reveals the occurrence in nonequilibrium systems of reversible processes of redistribution of energy carriers throughout the volume of the system and their opposite direction in different regions or degrees of freedom of the nonequilibrium system. This "opposite direction of processes" reflects at the physical level one of the main laws of dialectics, according to which the processes of evolution in some parts of a nonequilibrium system are invariably accompanied by processes of involution in others.
2. Description of the state of nonequilibrium systems requires the introduction of intensive Xi and extensive $Z_{i}$ parameters of spatial heterogeneity, characterizing their deviation from equilibrium. This cuts the limitations of the thermodynamics of irreversible processes, which excluded their reversible part from consideration, which is why it also turned out to be unable to cut the contradiction between thermodynamics and evolution.
3. The introduction of nonequilibrium parameters $X_{i}$ and $Z_{i}$ allows us to return the concepts of force, speed, and power of real processes to thermodynamics, and obtain an energodynamic identity that remains valid for irreversible processes. It follows from this that the simultaneous occurrence in various parts (areas, phases, and components) of a non-equilibrium system of counter-directed processes of relaxation and the performance of work "against equilibrium" in the system, which reflects the dialectical unity and "struggle" of opposites.
4. The use of nonequilibrium parameters $X_{i}$ and $Z_{i}$ as criteria for the evolution and involution of biological systems makes it possible to reflect their approach to equilibrium and their distance from it both as a whole and for each inherent degree of freedom separately. Considering their vanishing in a state of equilibrium, this makes the mentioned criteria simpler and more informative than entropy.
5. Energodynamic identity allows us to develop a theory of similarity of energy machines, which allows
us to consider the dependence of the efficiency of technical and biological propulsion on the conditions and mode of their operation. The theory implies the unity of the laws of transformation of thermal and non-thermal forms of energy, including biological (muscular) and technical propulsion, which makes it possible to transfer the experience of studying one of them to others.
6. The occurrence of oppositely directed processes in biological systems slows down their approach to equilibrium and allows us to formulate the basic law of biological evolution as "the principle of their survival," which is based on the increase in the reproductive period of life and as they self-organize."
7. The reason for the "flagrant contradiction" of equilibrium thermodynamics to the laws of biological evolution is the absence of time as a physical parameter in its equations, which deprives it of the ability to consider the kinetics of real processes. It is precisely this that holds the key to understanding the relationship between reversible and irreversible processes occurring in them and deciding the duration of the reproductive period of biosystems.
8. The energodynamic theory of evolution confirms the main provisions of Darwin's theory of evolution, explaining them, however, by natural causes. This once again confirms the validity of the laws of materialist dialectics.
Generating HTML Viewer...
References
44 Cites in Article
Hans Driesch (1893). Die Biologie als selbstandige grundwissenschaft.
E Schrödinger (1951). Was is Leben? München.
Ervin Bauer (1935). Theoretical biology.
Lev Blumenfeld (1977). Nonequilibrium Thermodynamics and Biological Physics.
S Shnol (1979). Physicochemical factors of biological evolution.
V Antonov Unknown Title.
M Media (2010). Unknown Title.
G Nicolis,I Prigogine (1979). Self-organization in nonequilibrium systems: From dissipative structures to ordering through fluctuations.
G Haken,M Synergetics,Mir (1980). Dynamics of Synergetic Systems.
P Berger,I Pomo,K Vidal (1991). Order in chaos.
Y Danilov,B Kadomtsev (1983). What is synergetics. // Nonlinear waves. Self-organization-nation.
I Prigogine (1984). Order and Haos, Man's new dialog with Nature.
Bazarovip (1991). Thermodynamics. Edn 4. M., Vysshayashkola.
L Boltzmann (1984). Selected Editions of Smith’s Works.
V Etkin (1998). Bulletin of the South-Russian state technical University (NPI) Series Socio-economic Sciences.
V Etkin,Thermokinetics (2010). Synthesis of Heat Engineering Theoretical Grounds).
Valeriy Etkin (1999). PLANK'S RADIATION LAW AS A CONSEQUENCE OF NONEQUILIBRIUM THERMODYNAMICS.
V Etkin,Energodynamics (2011). Solving the riddles of multiverse from the position of energodynamics.
V Etkin (2008). Energodynamics (synthesis of theories of energy transfer and transformation.
W Tomson (1882). 1882: Extracts from Reports of the Committee on Solar Physics.
Lars Onsager (1931). Reciprocal Relations in Irreversible Processes. II..
H Poincare (1908). Unknown Title.
Gyarmatii (1970). Non-EquilibriumThermodynamics. Field Theory and Variation Principles.
V Etkin (2020). On the Dialectic Unity of Evolution and Involution.
A Katchalsky,Peter Curran (1967). Nonequilibrium Thermodynamics in Biophysics.
S Keplen,E Essig (1968). Bioenergetics, and linear thermodynamics of irreversible processes.
I Prigogine,Pierre Rysselberghe (1955). Introduction to Thermodynamics of Irreversible Processes.
A Rudenko (1969). The theory of self-development of open catalytic systems.
A Rubin (1984). Thermodynamics of biological processes.
G Gladyshev (1997). Thermodynamics Theory of the Evolution of Living Beings.
Y Demirel (2014). Nonequilibrium Thermodynamics. Transport and Rate Processes in Physical, Chemical and Biological Systems.
A Zhabotinsky (1974). Concentration fluctuations.
I Prigogine,R Defey (1966). Ilya Prigogine 1917–2003.
S Oshkukov,A Baikin,A Galkin,P Glazkova,D Shavyrin,V Voloshin,K Shevyrev,T Biryukova,I Dementiev,E Petritskaya,A Kolmakov,E Nasakina,S Konushkin,M Kaplan,K Sergienko,M Sevostyanov (1966). Prospects of low-modulus alloys of the TI-(15-20)NB-(5-10)TA system for engineering implants used in traumatology and orthopedics. A preclinical study.
V Etkin (1989). 80 Years to the Zhurnal Fizicheskoi Khimii (Russian Journal of Physical Chemistry).
V Etkin (1989). 80 Years to the Zhurnal Fizicheskoi Khimii (Russian Journal of Physical Chemistry).
V Etkin (2018). New Сriteria of Evolution and Involution of the Isolated Systems.
V Etkin (2019). Similarity Theory of Energy Conversion Processes.
V Etkin (2014). Solving the riddles of multiverse from the position of energodynamics.
V Etkin (2020). Non-trivial consequences of energodynamics.
V Etkin (2016). Principle of non-equilibrium processes counter directivity.
Darwin Ch (1859). On the Origin of Spices by Means of Natural Selection, or the Preservation of Favoured Races in the Struggle for Life.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Etkin Valeri. 2026. \u201cEnergodynamic Theory of Biological Evolution\u201d. Global Journal of Science Frontier Research - C: Biological Science GJSFR-C Volume 25 (GJSFR Volume 25 Issue C1): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
The article outlines a more general energodynamic theory of biosystems, considering their global and local heterogeneity with the help of added nonequilibrium parameters. The theory returns the concepts of force, speed, and power of real processes to thermodynamics and complements the theory of irreversible processes by considering the useful (reversible) component of real processes. It is shown that relaxation processes in some degrees of freedom of biosystems are accompanied by work “against equilibrium” in other degrees of freedom, which is consistent with the Darwinian concept of the “struggle for existence.” Simpler and more informative non-entropy criteria for the evolution and involution of biosystems are proposed and the unity of energy conversion processes in technical and biological systems is proven.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.