Parkinson’s Disease (PD) is a progressive neurodegenerative disorder marked by motor symptoms due to dopaminergic degeneration and non-motor symptoms such as depression, anxiety, and cognitive impairment, which significantly affect patients’ quality of life. Traditional dopaminergic therapies address motor symptoms but offer limited efficacy for neuropsychiatric manifestations. Psilocybin, a serotonergic compound with strong affinity for the 5-HT2A receptor, has emerged as a promising candidate for addressing the complex symptomatology of PD, including its neuropsychiatric components. This review examines the pharmacological effects of psilocybin, particularly its ability to modulate serotonergic and dopaminergic systems, enhance neuroplasticity, and reduce neuroinflammation, offering a potential therapeutic approach for PD. While clinical research in PD remains limited, evidence from related conditions such as Major Depressive Disorder (MDD) and Substance Use Disorder (SUD) supports the notion that psilocybin could modulate both motor and non-motor symptoms in PD.
## I. INTRODUCTION
Parkinson's disease (PD) is a progressive neurodegenerative disorder primarily characterized by both motor and debilitating nonmotor symptoms, including depression, anxiety, and cognitive decline. These symptoms often appear before the onset of motor deficits and contribute significantly to reduced quality of life. While dopaminergic therapies such as levodopa remain the cornerstone of motor symptom management, they offer limited benefit for neuropsychiatric symptoms and may lead to complications with prolonged use. As the limitations of conventional therapies become more evident, interest has grown in exploring alternative approaches such as psychedelic-assisted therapy. This narrative review examines the current evidence surrounding psilocybin's neurobiological mechanisms and therapeutic potential in the context of Parkinson's disease. Psilocybin, a serotonergic compound derived from certain species of mushrooms, has shown promising effects in treating mood disorders and substance use disorders by acting on the 5-HT2A receptor. PD, MDD, and SUD all involve underlying dopaminergic dysfunction, suggesting a potential mechanistic overlap. Psilocybin's ability to modulate serotonergic activity and indirectly influence dopaminergic pathways may offer a novel strategy for addressing the complex symptom profile of PD.
## II. PSILOCYBIN: PHARMACOLOGICAL OVERVIEW
Psilocybin is a naturally occurring psychedelic compound found in several species of mushrooms, colloquially referred to as "magic mushrooms." After ingestion, psilocybin is rapidly converted in the body to its active form, psilocin, which acts primarily as a serotonin receptor agonist, with high affinity for the 5-HT2A receptor\*. Activation of this receptor is thought to underlie the drug's characteristic effects on perception, mood, and cognition. Psilocybin's influence on neural plasticity, including increased dendritic growth and synaptogenesis, has drawn significant attention for its potential in treating psychiatric and neurodegenerative disorders. These structural changes are believed to be mediated in part through downstream signaling cascades involving brain-derived neurotrophic factor (BDNF) and the mTOR pathway, both of which play crucial roles in synaptic remodeling and neuronal resilience [2].
Pharmacokinetically, psilocin typically reaches peak plasma concentration within 1 to 2 hours after oral administration, with a terminal half-life of approximately 2 to 3 hours. Despite its relatively short half-life, the subjective effects can last 4 to 6 hours, aligning well with the temporal needs of a therapeutic session. This brief but intense window of altered consciousness allows clinicians to structure therapy sessions with predictable onset and offset, reducing risks associated with prolonged intoxication. The short duration of action, combined with the low potential for physiological dependence and a favorable safety profile in controlled settings, supports the use of psilocybin in supervised clinical environments [3].
In a double-blind study on the effects of psilocybin, participants found their experience "[had] substantial personal meaning and spiritual significance and attributed to the experience sustained positive changes in attitudes and behavior consistent with changes rated by community observers" [4]. Its ability to induce meaningful psychological experiences, when administered with appropriate therapeutic support, has made it a leading candidate in the emerging field of psychedelic-assisted therapy.
## III. NEUROBIOLOGICAL BASIS
### a) Pathophysiology of Parkinson's Disease
Parkinson's Disease is characterized by the progressive degeneration of dopaminergic neurons in the substantia nigra, leading to a significant reduction in dopamine levels in the striatum. This dopaminergic cell loss is the hallmark of PD, which disrupts the basal ganglia's motor control pathways, resulting in motor symptoms such as bradykinesia, tremors, and rigidity. PD, however, "not only affects dopamine systems but also includes alterations in serotonergic, cholinergic, and other neurotransmitter systems, exacerbating symptoms such as depression and cognitive dysfunction," [5]. This broader involvement of neurotransmitter systems contributes to a diverse range of both motor and non-motor symptoms, including mood disorders, sleep disturbances, and cognitive decline, all of which complicate the management of PD. Moreover, the progressive nature of the disease is compounded by mechanisms like neuroinflammation, mitochondrial dysfunction, and oxidative stress, [6]. Additionally, the aggregation of, "misfolded alpha-synuclein proteins plays a central role in the progression of Parkinson's disease, leading to neuronal death and dysfunction," [7]. These multifaceted molecular processes: dopaminergic degeneration, altered neurotransmitter systems, neuroinflammation, mitochondrial dysfunction, and alpha-synuclein aggregation, collectively contribute to the complex pathophysiology of Parkinson's disease, making it a challenging disorder to treat and manage effectively.
### b) 5-HT2A Receptor in Psilocybin: Mechanism of Action
Parkinson's disease involves not only dopaminergic neuron degeneration in the substantia nigra but also early and significant changes in the serotonergic system. Degeneration of serotonergic neurons in the raphe nuclei, a primary source of brain serotonin, occurs early in the disease and contributes to widespread serotonin deficits. As noted, "serotonergic dysfunction is a prominent feature of Parkinson's disease, contributing significantly to both motor and nonmotor symptoms" [6]. This dysfunction underlies symptoms such as depression, anxiety, and cognitive decline. Altered serotonin receptor density and signaling, including changes in 5-HT2A receptor expression, have been observed in cortical and subcortical regions affected by PD, highlighting the critical role of serotonergic-dopaminergic interactions in the disease's pathophysiology and therapeutic approaches.
The 5-HT2A Receptor, commonly referred to as the serotonin-2A receptor, is a G-protein-coupled-receptor (GPCR) activated by the neurotransmitter serotonin (5-hydroxytryptamine). Psilocybin, a serotonergic psychedelic, exerts its primary pharmacological effects through partial agonism at the 5-HT2A receptor, densely expressed in cortical and subcortical brain regions. Activation of this receptor initiates a cascade of intracellular events, including phospholipase C stimulation and subsequent increases in intracellular calcium levels, which influence gene transcription and synaptic plasticity [7]. These signaling pathways are implicated in enhanced cortical connectivity and neuroplasticity, mechanisms increasingly viewed as therapeutically valuable in neurodegenerative diseases. Psilocybin-induced 5-HT2A receptor activation has also been shown to indirectly affect dopaminergic systems, particularly within the mesocorticolimbic and nigrostriatal pathways, suggesting a potential mechanism by which psilocybin could modulate motor and affective symptoms in Parkinson's disease, [7]. In this context, the serotonergic-dopaminergic interplay is especially relevant, as serotonin receptors may help normalize dysregulated dopamine transmission: a hallmark of PD pathophysiology. 5-HT2A activation can promote, "[mediate] Psychedelic-induced hyperconnectivity in the brain...[this] may facilitate neural rewiring, which could offer therapeutic benefits for Parkinson's disease," [9]. Collectively, these findings underscore the 5-HT2A receptor as a key mediator of psilocybin's neurophysiological effects and a potential target in the development of novel interventions for Parkinson's disease.
## IV. PSILOCYBIN IN THE TREATMENT OF NEUROLOGICAL AND PSYCHIATRIC DISORDERS
Historically, Psilocybin has been actively used in treating various medical and mental health conditions. Specifically focused on Substance Abuse Disorder (SUD) or addiction issues, psilocybin (specifically the properties of 5-HT2A) has been successfully proven to help mitigate symptoms of withdrawal or craving, as well as improve neuroplasticity after the effects of substance abuse damages the brain. The "proposed working mechanisms of psychedelics, including psilocybin, for improving psychiatric symptoms such as depression and SUD are both biological (e.g., by inducing brain neuroplasticity through elevating Brain-Derived Neurotrophic Factor (BDNF) levels, which are diminished in psychiatric conditions) and psychological...." [10]. Neuroplasticity is seen in patients of addiction and neurological damage significantly after the use of Psychedelic Administration Therapy (PAP) due to psychoactive properties. Psilocybin's psychoactive properties have demonstrated clinically meaningful antidepressant effects in controlled studies. Psilocybin's favorable safety profile, particularly in controlled therapeutic settings, supports its
investigational use for patients with MDD (Major Depressive Disorder). In a randomized psilocybin trial study conducted at Braxia Health in Ontario, Canada, patients (not participating in pharmacological treatment) were administered "one, two or three psilocybin sessions with a fixed dose of $25\mathrm{mg}$ synthetic psilocybin powder dissolved in water....accompanied by one preparatory therapy session (1-2 h), a supportive dosing session (6-8 h) and two integration therapy sessions (1-2 h each)...," [12]. After observing longer term effects of overall mental health, the following results can be viewed below:
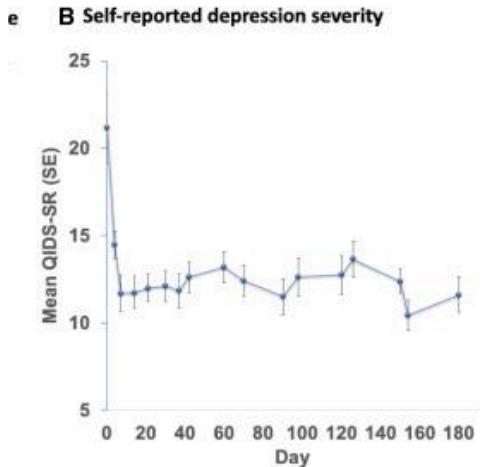
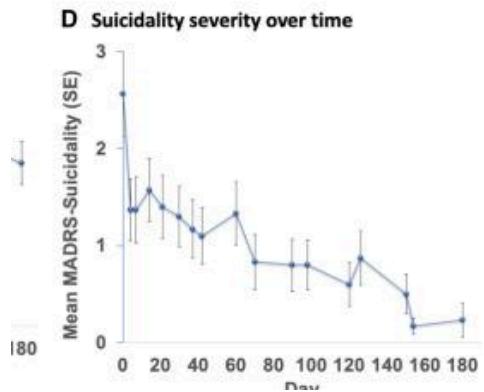
Based on the mean standard errors for both depression severity and suicidality, the trial demonstrated significant reductions from baseline levels, highlighting the potential of Psilocybin therapy as a promising intervention for psychological well-being.
## V. CLINICAL PARALLELS: WHY SUCCESS IN SUD AND MDD MATTERS FOR PD
Though there has not been extensive direct research on the specific effects of psilocybin in Parkinson's disease (PD), neurobiological, mechanistic, and clinical parallels provide a biologically plausible rationale for its therapeutic exploration. Psilocybin promotes neuroplasticity by increasing dendritic spine growth and enhancing synaptic connectivity, primarily through activation of serotonin receptors such as 5-
[11] Psilocybin with psychological support for treatment-resistant depression: An open-label feasibility study.
HT2A, which leads to elevated brain-derived neurotrophic factor (BDNF) levels. These neuroplastic changes have demonstrated benefits in conditions marked by impaired plasticity and neurodegeneration, including major depressive disorder (MDD) and PD [14].
Importantly, affective symptoms such as apathy and anhedonia are shared across MDD, substance use disorder (SUD), and PD, although their clinical presentations and underlying neurocircuitry may differ. In PD, apathy often correlates with dopaminergic degeneration in nigrostriatal pathways, while in MDD and SUD, disruptions in frontostriatal and limbic circuits contribute to these symptoms. Despite these differences, all three conditions involve dysregulated reward processing and motivation pathways modulated by serotonergic and dopaminergic signaling. As reviewed, "apathy and anhedonia manifest across these disorders through overlapping but distinct neural mechanisms, highlighting transdiagnostic relevance," [13].
Regarding BDNF, its function diverges between neurodegenerative and mood disorders. In mood disorders like MDD, reduced BDNF expression is linked primarily to impaired synaptic plasticity and neuronal resilience, contributing to mood dysregulation. In contrast, in PD, BDNF deficiency additionally exacerbates dopaminergic neuron loss and neurodegeneration. Psilocybin's ability to elevate BDNF may thus serve a dual role: restoring synaptic plasticity in mood disorders while potentially promoting neuronal survival in neurodegenerative disease [13].
Furthermore, psychedelics modulate critical intracellular signaling pathways—including nuclear factor kappa-light-chain-enhancer of activated B cells (NF- $\kappa$ B), phosphatidylinositol-3-kinase/protein kinase B (PI3K/Akt), and mechanistic target of rapamycin (mTOR)—that are implicated in neuroplasticity and inflammation. These pathways are disrupted across MDD, SUD, and PD, highlighting a shared therapeutic target space where psilocybin's anti-inflammatory and neurorestorative effects could have broad applicability [12].
Specific mechanism depiction can be seen below:
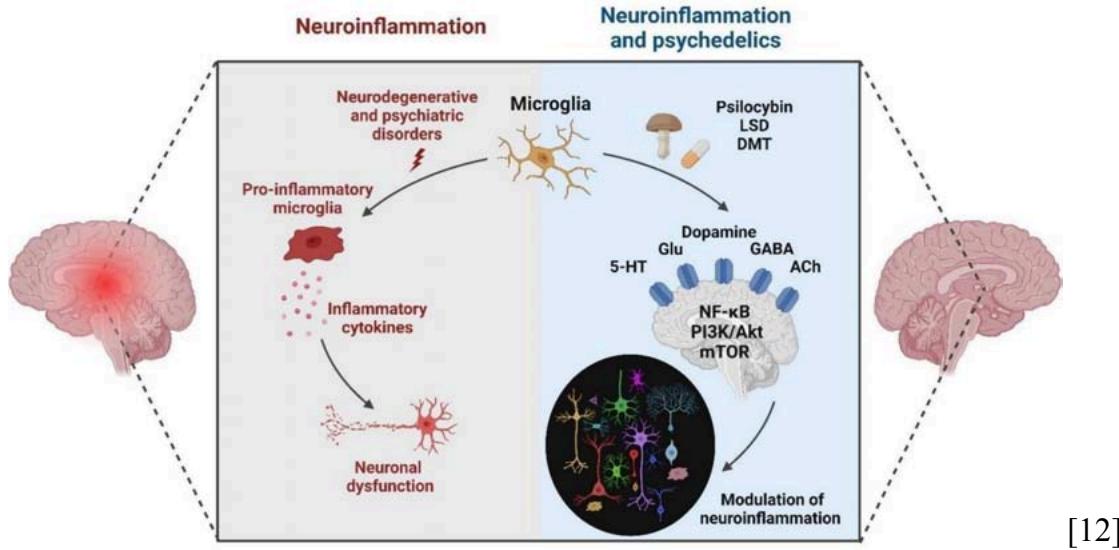 [12] Augmentation in depression: Esketamine, a new standard?
On a psychological level, a multicenter trial led by Dr. James Rucker of King's College London, found that a $25\mathrm{mg}$ dose of psilocybin on human participants, alongside psychological support, significantly impacted depression symptoms in participants with treatment-resistant depression. The study observed greater reductions in depression scores three weeks after treatment in the high-dose group compared to the placebo group [16].
## VI. CURRENT PAP AND PD CONDUCTED
### RESEARCH
While direct research on psilocybin's effects in Parkinson's Disease remains limited, the existing neurobiological, mechanistic, and clinical evidence suggests promising potential for its application in this population. The observed ability of psilocybin to enhance neuroplasticity, modulate inflammatory pathways, and alleviate depressive symptoms in related conditions like MDD and SUD highlights its potential as a multifaceted therapeutic approach for the non-motor symptoms of Parkinson's. A pilot study at UC San
Francisco, led by Ellen R. Bradley, investigated the effects of psilocybin on mood, cognition, and motor function in individuals with Parkinson's disease. Participants experienced clinically significant improvements in mood and motor symptoms that lasted for weeks after treatment. The study suggests that psilocybin may help repair brain function and alleviate non-motor symptoms associated with PD. After administering psilocybin to a moderately sized group of participants with PD and observing results, "non-motor (MDS-UPDRS Part I: -13.8 ± 1.3, p < 0.001, Hedges' g = 3.0) and motor symptoms (Part II: -7.5 ± 0.9, p < 0.001, g = 1.2; Part III: -4.6 ± 1.3, p = 0.001; g = 0.3) as well as performance in select cognitive domains (Paired Associates Learning [-0.44 ± 0.14, p =.003, g = 0.4], Spatial Working Memory [-0.52 ± 0.17, p = 0.003, g = 0.7], and Probabilistic Reversal Learning [2.9 ± 0.9, p = 0.003, g = 1.3]) improved post-treatment," supporting the plausibility of extending findings from related conditions to PD [8]. Additionally, on a preliminary case-report done on a middle-aged woman (PD patient) with a lack of depressive-symptoms, positive effects of PAP were seen. Post-treatment, the patient reflected on a more positive outlook on the severity of her disorder, worrying less about mortality or her future, "promoting profound decentration from habitual thoughts and emotions, improving mood and PD acceptance," [8]. Though these observed effects could be attributed to potential placebo, the existence of general outlook improvement is not one to overlook, and is often a pivotal tool for the wellbeing of those with chronic neurodegenerative disorders like PD.
## VII. NEUROIMAGING AND MECHANISTIC EVIDENCE
Psilocybin has been shown to enhance brain network connectivity, particularly within the default mode network (DMN), as demonstrated by fMRI studies. These studies reveal that psilocybin induces hyperconnectivity across multiple brain regions, including the prefrontal cortex, which is associated with subjective alterations in cognition and perception, [8]. This heightened connectivity may be particularly beneficial for PD, where reduced brain connectivity contributes to both motor and non-motor symptoms, [15]. These findings support the idea that psilocybin's neuroplastic effects may provide relief from symptoms often associated with neurodegenerative conditions.
## VIII. DOPAMINE-SEROTONIN SYSTEM INTERACTIONS
Psilocybin's effects are mediated primarily through serotonin 5-HT2A receptor activation, which in turn influences dopamine release, particularly in regions involved in motor control. This interaction is crucial for Parkinson's Disease, where dopamine dysregulation plays a central role in both motor and non-motor symptoms. It's highlighted that psilocybin, "primarily activates serotonin 5-HT2A receptors, which modulate dopamine release, potentially alleviating symptoms related to PD" [12]. The regulation of both serotonin and dopamine systems could improve symptoms in PD patients, especially those who do not respond to conventional dopamine-based therapies, with "serotonin-dopamine interactions in Parkinson's Disease [being critical] for understanding treatment efficacy, with psilocybin potentially [modulates] both neurotransmitter systems to improve motor and non-motor symptoms" [14].
## IX. SAFETY, ETHICS, AND THERAPEUTIC CONSIDERATIONS
As interest in psilocybin as a therapeutic tool for neurodegenerative disorders like PD grows, safety and ethical considerations must be carefully addressed. Psilocybin, like other psychedelics, carries potential risks, particularly when used outside of controlled environments or by individuals with certain health conditions. Common side effects include transient anxiety, confusion, and disorientation, which may be particularly concerning for individuals with cognitive impairments, such as those with PD. It's not to be forgotten that psilocybin possesses psychoactive properties, with potential for hallucinogenic and psychedelic symptoms if taken at a high or non-controlled dose. However, studies have generally found that psilocybin is well-tolerated when administered in a supervised clinical setting, with careful screening to ensure patient suitability.
Additionally, the issue of cognitive decline, neural plasticity damage, and deficit as symptoms of PD become present in the ethical issue of consent by participants. Depending on the stage/onset of a participant's PD, it can potentially be a violation of conscious consent to administer a substance like psilocybin considering the patient may not be at the same level of consciousness as pre-diagnosis. In terms of decision-making, based on the pathophysiology of the disorder, "a progressive loss of cells in the midbrain producing the neurotransmitter dopamine...the controlled release of dopamine into synapses connecting the cerebral cortex and the basal ganglia plays a central role during the selection and reinforcement of actions," [10]. Due to potential decision-making impairment, the clinician in charge of administering a patient with said psychedelic must be perceptive on the patient's level of cognitive decline and ability to make a conscious decision in order to avoid ethical or HIPAA violations. Once these factors are taken into account, however, PAP appears to be an effective and low risk strategy towards cognitive and emotional recovery/stability.
## X. LIMITATIONS IN CURRENT RESEARCH
Psilocybin has emerged as a leading candidate in contemporary psychedelic research. Considering this, there are still major limitations on clinical exploration/trials for neurodegenerative disorders like PD. As mentioned above, the cognitive decline and ambiguous or impaired decision-making capacity in cognitively affected individuals may raise ethical concerns regarding informed consent. Due to the severity of PD's neurodegenerative impacts, sample sizes and lengths of trials are significantly shorter than desired, therefore altering the professionalism and accuracy of results. Short trial durations and limited sample sizes restrict statistical power, increase variability, and limit the generalizability of findings. Additionally, the long-term safety profile of repeated or high-dose psilocybin administration remains insufficiently characterized, particularly for populations already vulnerable to cognitive decline or psychosis. Regulatory constraints and the classification of psilocybin as a Schedule I substance also pose significant barriers to large-scale, multi-site studies. Until these limitations are addressed through more rigorous, targeted, and longitudinal trials (including those specific to PD), clinical recommendations for psilocybin use in neurodegenerative contexts must remain cautious and exploratory.
## XI. FUTURE DIRECTIONS AND RESEARCH PRIORITY
To fully assess psilocybin's therapeutic potential for Parkinson's Disease, future research must prioritize disease-specific clinical trials that evaluate both motor and non-motor outcomes. These studies should include larger, more diverse patient populations, with extended follow-up periods to better understand long term efficacy and safety. There is also a pressing need for trials that examine optimal dosing regimens, frequency of administration, and the role of psychological support during psychedelic-assisted therapy. Particularly, the effects of repeated dosing on serotonergic neurotoxicity, emotional lability, or precipitating latent psychosis in predisposed individuals remain poorly understood. Mechanistic studies using advanced neuroimaging and biomarker analysis, particularly those exploring serotonin-dopamine interactions and network level brain changes, will be critical in determining how psilocybin affects PD specific neurobiology. In addition, comparative trials that evaluate psilocybin against existing pharmacological and behavioral therapies could help clarify its position within the broader treatment landscape. As regulatory frameworks continue to evolve, ethical and equitable access to psychedelic therapy must remain a central concern, ensuring that any future therapeutic model is both scientifically grounded and socially responsible.
## XII. CONCLUSION
Psilocybin emerges as a promising candidate with systematically supported potential for treating neuropsychiatric symptoms in PD. Its mechanism of action, centered on 5-HT2A receptor activation, initiates a cascade of neuroplastic and neuromodulatory processes that extend beyond mood regulation to potentially influence dopaminergic function, cortical network connectivity, and inflammatory pathways. While direct research on psilocybin in PD remains limited, strong clinical and mechanistic parallels from studies in depression and substance use disorders offer a valuable foundation for future inquiry. Although limited, early pilot data suggests that psilocybin may positively affect both motor and non motor symptoms, warranting further investigation. However, ethical, regulatory, and methodological challenges must be addressed through rigorous, disease-specific clinical trials. In particular, the use of psilocybin in vulnerable populations such as those with PD necessitates robust consent frameworks, clearly defined inclusion criteria, and stringent monitoring protocols to ensure patient safety and uphold ethical standards. Further research should prioritize Phase I safety studies in early-stage PD, with a focus on affective symptoms, quality of life metrics, and neurobiological markers of change. With appropriate safeguards and continued interdisciplinary research, psilocybin could help redefine the therapeutic landscape for Parkinson's Disease, offering a novel avenue for enhancing quality of life in a population long underserved by conventional treatments.
Friederike Holze,Anna Becker,Karolina Kolaczynska,Urs Duthaler,Matthias Liechti (2023). Pharmacokinetics and Pharmacodynamics of Oral Psilocybin Administration in Healthy Participants.
R Griffiths,W Richards,U Mccann,R Jesse (2006). Psilocybin can occasion mystical-type experiences having substantial and sustained personal meaning and spiritual significance.
A Mastinu,M Premoli,S Bonini (2023). Psilocybin for treating psychiatric disorders: Pharmacological profile and clinical perspectives.
M Politis,K Wu,C Loane (2010). Serotonergic dysfunction in Parkinson's disease and its relevance to nonmotor symptoms.
P Fleury,D Goff,R Murray (2025). Psychedelic compounds as treatment strategies for neuropsychiatric symptoms in Parkinson's disease: Rationale and emerging evidence.
P Bradley,J Holtman,S Haroutounian (2025). Psychedelic-assisted therapy for neurodegenerative diseases: Challenges and promises.
D Brengel,L Keller,H Möller (2024). Psychedelic drug-induced hyperconnectivity in the brain helps clarify altered subjective experiences.
Robin Carhart-Harris,Mark Bolstridge,James Rucker,Camilla Day,David Erritzoe,Mendel Kaelen,Michael Bloomfield,James Rickard,Ben Forbes,Amanda Feilding,David Taylor,Steve Pilling,Valerie Curran,David Nutt (2016). Psilocybin with psychological support for treatment-resistant depression: an open-label feasibility study.
Joshua Rosenblat,Roger Mcintyre (2023). Augmentation in depression: Esketamine, a new standard?.
Masud Husain,Jonathan Roiser (2018). Neuroscience of apathy and anhedonia: a transdiagnostic approach.
S Fereydouni,C Andrade,E Osuch (2025). Serotonergic psychedelics and dopamine: An interaction with therapeutic implications ons.
De Deus,J Polari,A Maia,F (2025). Neuroinflammatory and plasticity pathways affected by psychedelics: Potential relevance to Parkinson's disease.
D Herz,S Eickhoff,A Løkkegaard,H Siebner (2017). Functional neuroimaging of motor control in Parkinson's disease: A meta-analysis.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Dr. Nayiri Barton. 2026. \u201cEvaluating Psilocybin as a Treatment for Neuropsychiatric Symptoms in Parkinson’s Disease\u201d. Global Journal of Medical Research - A: Neurology & Nervous System GJMR-A Volume 25 (GJMR Volume 25 Issue A1): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Parkinson’s Disease (PD) is a progressive neurodegenerative disorder marked by motor symptoms due to dopaminergic degeneration and non-motor symptoms such as depression, anxiety, and cognitive impairment, which significantly affect patients’ quality of life. Traditional dopaminergic therapies address motor symptoms but offer limited efficacy for neuropsychiatric manifestations. Psilocybin, a serotonergic compound with strong affinity for the 5-HT2A receptor, has emerged as a promising candidate for addressing the complex symptomatology of PD, including its neuropsychiatric components. This review examines the pharmacological effects of psilocybin, particularly its ability to modulate serotonergic and dopaminergic systems, enhance neuroplasticity, and reduce neuroinflammation, offering a potential therapeutic approach for PD. While clinical research in PD remains limited, evidence from related conditions such as Major Depressive Disorder (MDD) and Substance Use Disorder (SUD) supports the notion that psilocybin could modulate both motor and non-motor symptoms in PD.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.