This work aims to evaluate the effects of rapid thermoresistance on the morphofunctional aspects of membranes using flow cytometry, as well as the quality of sperm chromatin using the Toluidine Blue technique, both before and after the thermoresistance test. Six sets of frozen semen from ten bulls were used. The samples were thawed in a water bath at 36ºC and separated into two aliquots. The first aliquot, control group (C), was kept in a test tube previously heated to 36ºC with subsequent evaluationsusingmicroscopy, cytometry, and chromatin analyses. The second aliquot, experimental group (E), underwent the rapid thermoresistance test (RTT) in a water bath preheated at 46ºC for 30 minutes with subsequent evaluations usingmicroscopy, cytometry, and chromatin analyses. For statistical analysis, the paired T test, significance (P
## I. INTRODUCTION
Artificial insemination with the use of genetically superior animals has been gaining prominence due to the excellent results in the commercialization of cryopreserved bovine semen. Thus, the concern with bull fertility is recurrent among rural producers, as well as scholars who dedicate
Author p: Universidade Federal do Triangulo Mineiro (UFTM), Uberaba, MG, Brazil.
Author 0: Faculdade Associadas de Uberaba (FAZU), Uberaba, MG, Brazil.
Author ¥§: Universidade Federal de Uberlândia (UFU), Uberlândia, MG, Brazil.
themselves to sperm analysis to detect changes in sperm cells (Alquezar-Baeta et al 2019).
The fertility potential of male cattle is determined through semen analysis in a set of evaluations. However, for this analysis to be more efficient, it is also necessary to evaluate the internal structure of the sperm in conjunction with the rapid thermoresistance test (RTT) (Alves et al 2015). This can be used to evaluate the fertility of frozen semen, simulating a biological process. One of the possibilities to mimic this condition is to promote thermal stress, under controlled conditions, in a water bath, a situation close to the conditions to which it is exposed in the female genital tract (Cunha et al 2012).
The potential study of RTT is to promote thermal stress in sperm, and in conjunction with flow cytometry and combined with the use of fluorophores, it allows evaluating the integrity of the plasma and acrosomal membrane, as well as the integrity of the DNA, after the induction of stress, providing pre- and post-heating parameters of post-thawed bull semen (Arruda et al 2007). Also adding to the analysis, the Toluidine Blue (TB) technique has been widely used in the study of chromatin changes (Mello 1982).
Therefore, this research aims to evaluate the effects of rapid thermoresistance on the morphofunctional aspects of plasma and acrosomal membranes, as well as on the chromatin quality of post-thawed bull semen, both before and after the rapid thermoresistance test.
## II. MATERIALS AND METHODS
### a) Obtaining Bovine Semen Samples
Six batches of frozen semen from ten bulls were used, obtained from Sexing Technologies Repro I.C.M.G.A. Ltda. (Brindes) Use Authorization Protocol 135190833813983 08/11/2019 - CNPJ 23.694.902/001-70 - Indaiatuba - SP.Only samples containing at least $30\%$ sperm with progressive motility were used in the study (CBRA Manual, 2013).
### b) Sperm Preparation
To carry out the experiment, the samples were thawed in a water bath at $36.8^{\circ}\mathrm{C}$ (CBRA Manual, 2013) and separated into two aliquots. The first aliquot, control group (C), was kept in a test tube previously heated to $36.8^{\circ}\mathrm{C}$ and subsequently evaluated by microscopy, cytometry and chromatin analysis. The second aliquot, experimental group (E), was kept in a water bath at $46^{\circ}\mathrm{C}$ for 30 minutes to perform the rapid thermoresistance test (RTT) (Arruda et al 2007, Cunha et al., 2012), followed by microscopy, cytometry and chromatin analyses. The experiment was carried out in triplicate and the evaluations were carried out by a single evaluator.
### c) Sperm Evaluations
For the microscopic evaluations, a bright field microscope (Nikon eclipse e200) was used. $10\mu \mathrm{L}$ of the sample were added between the slide and coverslip, both before and after the thermoresistance test, and sperm motility and vigor were evaluated at $10x$ magnification, according to Brazilian College of Animal Reproduction (Manual CBRA, 2013).
In the cytometric evaluations, a flow cytometer (FACSC aliburTM) was used to detect the integrity parameters of the plasma (propidium iodide /1.5 mM) and acrosomal (Fitc-PNA/1.125 g/mL) membranes. A 560 nm short pass dichroic mirror, green fluorescence (FL1) was collected using a 515 meter545 nm bandpass filter. Red fluorescence (FL3) was collected through a 650 nm long-pass filter, after 640 nm filtering. long pass filter. The sheath/sample was placed on "low" and adjusted to a flow rate of 100 cells per second when analyzing a sample with a concentration of 1.25 x105 sperm/mL. Data acquisition from 15,000 cells was collected in list mode using BD Cell Quest Pro software version 4.0 (Becton Dickinson®, San Jose, CA, USA) as described by (Vasconcelos et al 2017).
For chromatin analysis, the protocol as reported by Beletti and collaborators (2005) was followed. For this, the smear technique was performed on the samples taken at the two moments (before and after the thermoresistance test). They were fixed with absolute alcohol: acetic acid: 3:1, for one minute and afterwards immersed in $70\%$ alcohol for three minutes. With this, four slides of each sample were made, which were later kept in 4N hydrochloric acid for 25 minutes, followed by washing in distilled water. After this procedure, the slides were kept at room temperature (dry) followed by the addition of three to four drops of Toluidine blue and covered with a coverslip. For the analysis, a microscope (Leica DM500) coupled to an image capture system (Leica ICC50) with a 100X oil-immersion objective in an optical microscope coupled to the camera was used, from which 30 to 50 images were obtained of each prepared slide.
The images obtained were light blue in color when they presented normal chromatin condensation, a variation from light blue to dark blue when the chromatin was moderately decondensed and a variation from dark blue to violet when the chromatin was highly decondensed. In view of this, to avoid the subjectivity of the chromatin alteration evaluations, sperm head segmentation was used, developed in MATLAB and executed with Octave software. The system performs the reading of the sperm according to the color present (light blue to magenta), making it possible to define the chromatin compaction index (Beletti et al 2005). Statistics
For statistical analysis, the program GraphPad prism $6.0^{5}$ was used. Fisher's and Student-Newman-Keuls tests were used to analyze progressive sperm motility, sperm with plasma and acrosomal membranes integrity and analyze chromatin expressed as means and standard deviation (SD) (three repetitions), test at $5\%$ significance $(P < 0.05)$.
## III. RESULTS
The semen samples before the rapid thermoresistance test showed mean values of $37.5\%$ for motility and 2.5 for vigor. These values were within the range established by the Brazilian College of Animal Reproduction, with references of $30\%$ and 3 for motility and vigor, respectively. However, after the rapid thermoresistance test, a significant drop $(P < 0.05)$ in motility $(16.5\%)$ and vigor (1.6) was observed, with a reduction of approximately $56\%$ and $36\%$ for motility and vigor, respectively (Figure 1). According to Cunha et al. (2012), bovine semen after being subjected to rapid thermoresistance test (RTT) should show a decrease in straight, progressive sperm motility and sperm vigor.
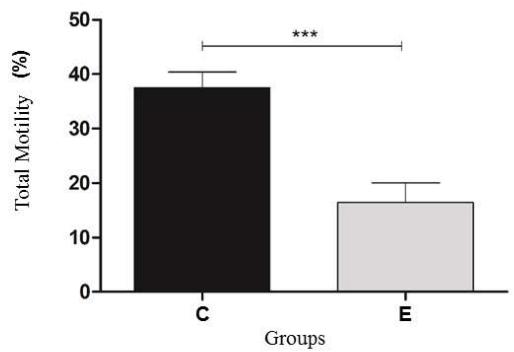
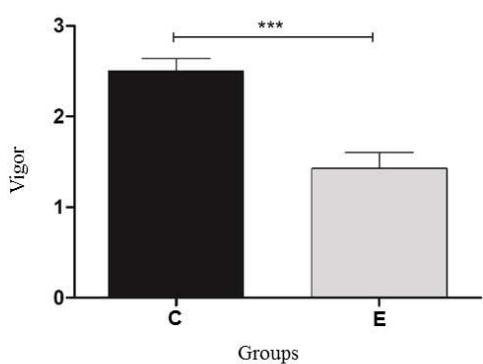 Figure 1: Result of the mean and standard deviation of the total motility and vigor of bovine sperm samples, after thawing $(36^{\circ}\mathrm{C})$ Control Group (C) and after the rapid thermoresistance test $(46^{\circ}\mathrm{C} / 30\mathrm{min})$, Experimental Group (E) Statistical significance $(P < 0.05)$
For the flow cytometry analysis, the correspondence of red, IP (FL3 fotodetector), green and Fitc-PNA (FL1 Fotodetector) fluorescence was observed. The sperm that fluoresced in red were classified as having non-intact plasma membranes and those that fluoresced in green had a reacted outer acrosomal membrane.
The results of thermoresistance test the Control (C) and experimental (E) Groups. Q1 (IP positive) percentage of sperm with reactive plasma membrane; Q2 (IP positive /PNA positive) percentage of sperm with reactive plasma and acrosomal membranes; Q3 (PNA positive) percentage of sperm with intact plasma and acrosomal membranes; Q4 (IP negative/PNA negative). The program Cell Quest was used for this analysis (Figure 2).
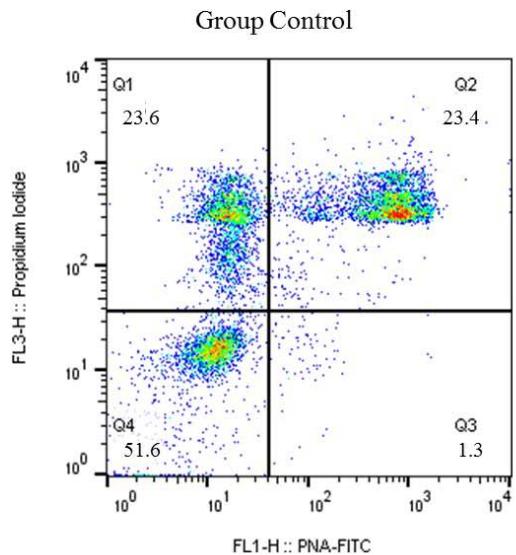
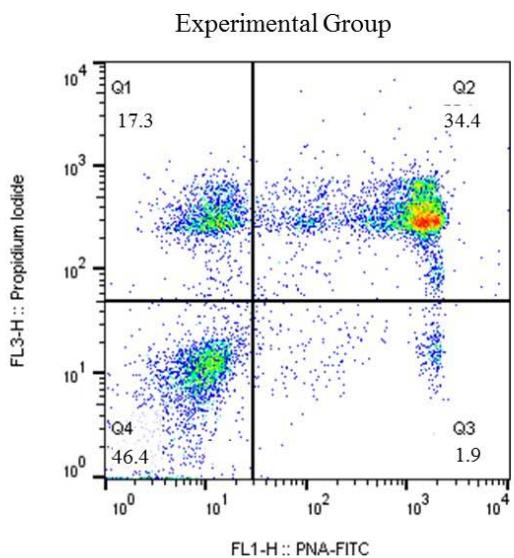 Figure 2: Graphic representation of spot distribution of bovine sperm marking, post-thawing, from Control and Experimental groups, marked with: Q1 - Propidium iodide (PI); Q2 - double marking of propidium iodide (IP) and Fic – PNA; Q3 – Fitc-PNA; Q4 without marking (viable sperm)
Before the thermoresistance test (Control Group), it was observed that the sperm were less reactive to the markings. Because of this condition, the values of the control group (pre-test) were higher than the cells from the experimental group (post-test) in regard to the plasma membrane Q1 $(23.6\%$ vs $17.3\%)$ and the number of viable Q4 cells $(51.6\%$ vs $46.4\%)$, respectively. For the Q2 double marking evaluation, higher percentages were observed for the experimental group (34.4%) when compared to the control group (23.4%); a result also observed in the Q3 group evaluations (1.4% vs 1.9%). In the flow cytometry analysis, a significant difference $(P < 0.05)$ was observed for all points analyzed for both the plasma membrane and the acrosomal membrane. (Figure 3).
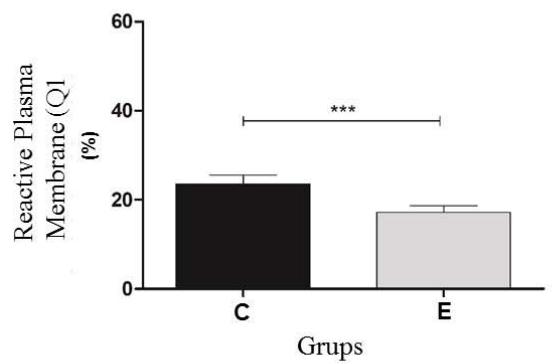
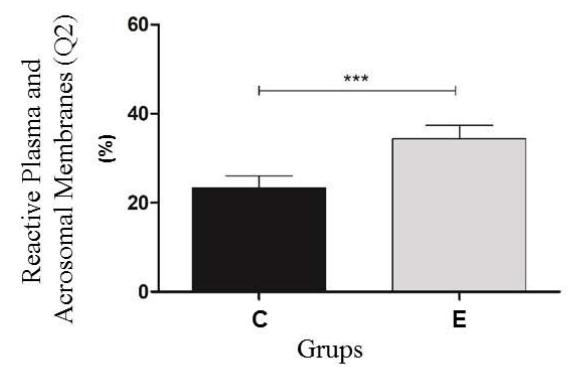
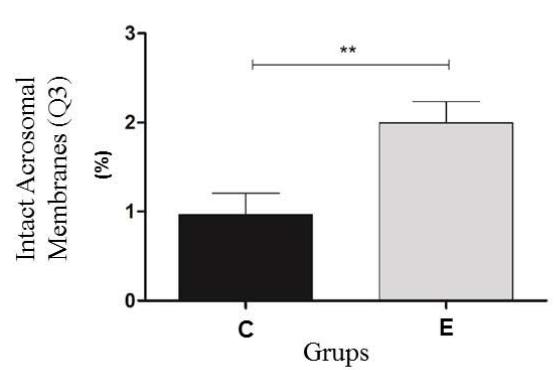
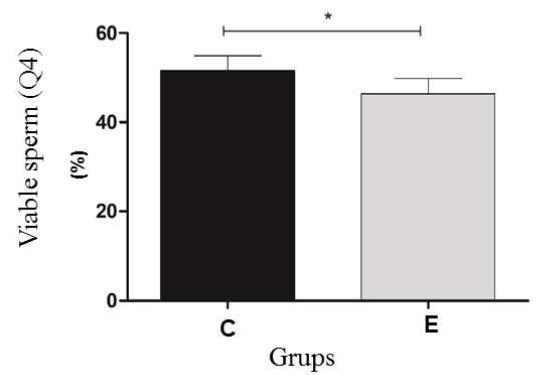 Figure 3: Experimental analysis of the flow cytometry tests of the Control (C) and experimental (E) Groups. Q1 (IP positive) percentage of sperm with reactive plasma membrane; Q2 (IP positive /PNA positive) percentage of sperm with reactive plasma and acrosomal membranes; Q3 (PNA positive) percentage of sperm with intact plasma and acrosomal membranes; Q4 (IP negative/PNA negative) percentage of sperm with reactive acrosomal membrane. Statistical significance $(\mathsf{P} < 0.05)$
Sperm membranes perform numerous functions that are related to cellular metabolism and the maintenance of motility and training in sperm. Therefore, the loss of membrane integrity, as seen in figure 2 and figure 3, indicates a change in the maintenance of cellular homeostasis, directly observed in the decrease in sperm motility (figure 1) From the decomposition and heterogeneity analyses, all sperm were averaged, both before and after the thermoresistance test. As shown in Figure 4, it was observed that there was no significant difference $(P > 0.05)$ regarding the two evaluated points.
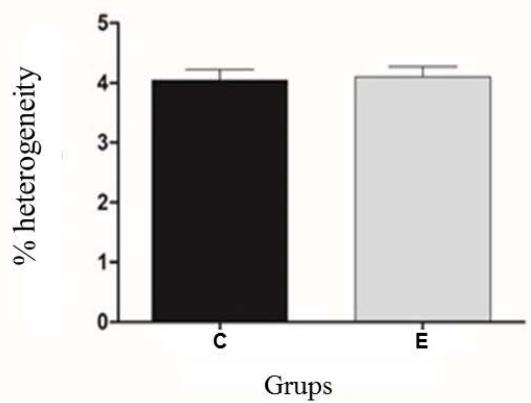
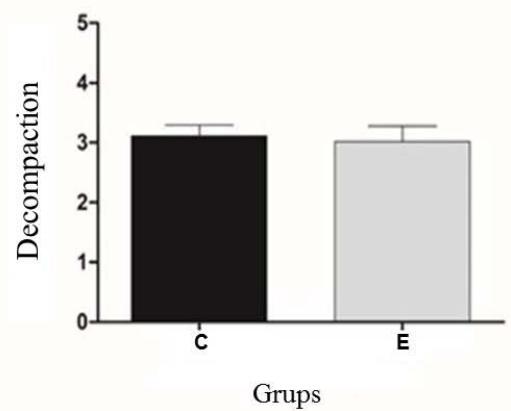 Figure 4: Experimental analysis of chromatin heterogeneity and decompaction of Control (C) and Experimental (E) Groups. No statistical difference $(P > 0.05)$
The result may be related to the physiological aspect of nuclear differentiation that occurs during spermatogenesis, as in the condensation of sperm chromatin, somatic DNA histones are replaced by protamines (P1), compacted structures (toroid or donut).
## IV. DISCUSSION
During fertilization, sperm capacitation, acrosomal reaction and fusion with the ovum are events that require functional and intact plasma and acrosomal membranes. The cryogenics process injures the sperm, decreasing their viability. Aware of the extreme need for sperm to maintain good viability in order to obtain high fertility in a herd, the use of techniques in the field of reproduction biotechnology has intensified, being identified as extremely important tools for genetic improvement programs and efficiency in the production of animal products (Coutinho et al 2010).
The importance of applying additional techniques to evaluate semen batches before commercialization is evident, as it contributes to the quality control of the ejaculate, thus resulting in a better fertility rate and herd productivity (Celeghini et al 2017).
In this study, the effects of the rapid thermoresistance on the morphofunctional aspects of plasma and acrosomal membranes were evaluated through flow cytometry, as well as through chromatin quality, using the Toluidine Blue technique.
Semen samples taken before the rapid thermoresistance test presented values with greater significance in relation to those taken after the rapid thermoresistance test (RTT), possibly due to the time and temperature at which the sperm were kept. This may have led to a drop in sperm viability with changes in membrane structures, mainly the acrosomal structure. This aspect was observed in the flow cytometry analysis since the sperm were more reactive to the plasma and acrosomal membrane markings after the RTT.
The hypothesis for this result is established since, with the temperature increase, there is a structural change in the membrane's biochemical components, in addition to the promotion of an increase in the sperm's metabolic rate. It is known that the membrane is flexible, composed of lipids and proteins, and that it possesses self-sealing characteristics and ion channels defined by transmembrane proteins, which selectively act on solutes through active and passive transport (Flesh & Gadella 2000). Thus, the temperature increase can somehow act on ATP-dependent ion channels, such as the sodium/potassium ATPase pump and calcium ion channels, promoting instability in both membranes, and rather, greater reactivity to markers, Q2 quadrant. (PI Positive/PNA Positive).
Another point that corroborates this discussion is that in the experimental group (post-thermoresistance test), quadrant Q3 (PNA positive) showed an increase in the number of reactive sperm, which indicated a possible spontaneous capacitation induced by temperature.
Sperm capacitation is a physiological process that can be induced by several factors, such as variation in hydrogen potential and in intracellular ionic concentration. In addition, it is possible, given the results of the present study, that this sperm capacitation could also be related to the temperature used in the RTT. This is because the temperature can change the fluidity of the plasma membrane, which would promote an increase in metabolism and consequently greater motility, though for a very short period due to the availability of nutrients and oxygen in the medium (Yanagimachi1994; Flesh F.M. & Gadella 2000).
However, according to the results obtained with the Toluidine Blue technique, it can be observed that RTT does not interfere with the chromatin structure. This is probably due to the DNA structure of mammalian sperm, which have a basic unit of protamine toroid. Thus, after the interaction of these protamines with the sperm chromatin, it becomes an inert and highly stable structure, mainly due to the interaction of the protamine's amine residues with the phosphate groups of the DNA strands (Hamilton et al 2016), which could promote greater DNA stability regarding temperature changes.
## V. CONCLUSION
It can be concluded that the rapid thermoresistance test (RTT) alters the structures, mainly the acrosomal structure, and does not interfere with the structure of chromatin compaction, and should be considered, in association with other evaluations, as a complementary parameter in evaluating bovine semen quality.
## ACKNOWLEDGEMENTS
Universidade Federal de Uberlândia (UFU) for the support given during the research and the Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG) for granting the financial resources.
Declaration of Interest: The authors report no conflicts of interest. The authors alone are responsible for the content of the paper.
Generating HTML Viewer...
References
13 Cites in Article
Carlos Alquézar-Baeta,Silvia Gimeno-Martos,Sara Miguel-Jiménez,Pilar Santolaria,Jesús Yániz,Inmaculada Palacín,Adriana Casao,José Cebrián-Pérez,Teresa Muiño-Blanco,Rosaura Pérez-Pé (2019). OpenCASA: A new open-source and scalable tool for sperm quality analysis.
M Alves,M Oliveira,Lançoni R,S Florez-Rodrigues,E Celeghini,P Arruda R,A Andrade (2015). Investigando a compactação e a fragmentação não induzida do DNA espermático: refinamento da avaliação espermática -parte 1.
P Arruda R,A Andrade,K Peres,C Raphael,J Nascimento,E Celeghini (2007). Biotécnicas aplicadas à avaliação do potencial de fertilidade do semen equino.
M Beletti,L Costa,M Guardieiro (2005). Morphometric features and chromatin condensation abnormalities evaluated by toluidine blue staining in bull spermatozoa.
E Celeghini,P Arruda R,S Rodriguez,F Santos,M Alves,B Oliveira (2017). Impacto da qualidade do sêmensobre a fertilidade a campo embovinos.
Manual Para Exame Andrológico Eavaliação De Sêmen Animal (2013). Colégio Brasileiro de Reprodução Animal.
E Cunha,C Silva,C Martins (2012). Estudo comparativo dos testes de termo-resistência rápido, lento e fisiológico em sêmen criopreservado bovino importado.
Thais Hamilton,Letícia Castro,Juliana Delgado,Patrícia De Assis,Adriano Siqueira,Camilla Mendes,Marcelo Goissis,Teresa Muiño-Blanco,José Cebrián-Pérez,Marcílio Nichi,José Visintin,Mayra D'ávila Assumpção (2016). Induced lipid peroxidation in ram sperm: semen profile, DNA fragmentation and antioxidant status.
Frits Flesch,Barend Gadella (2000). Dynamics of the mammalian sperm plasma membrane in the process of fertilization.
M Mello (1982). Induced metachromasia in bull spermatozoa.
A Vasconcelos,J Zandonaide,A Fernandes Sobrinho,B Silva,P Quintal (2017). A comparative study of three different dyes evaluating the physical integrity of the plasma membrane of cryopreserved bovine spermatozoa.
Yanagimachi R (1994). Stability of the mammalian sperm nucleus.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Ana Clara Ferreira Batista. 2026. \u201cEvaluating the Impact of Rapid Thermoresistance (RTT) on Membrane and Chromatin Features in Post-Thawed Bovine Semen\u201d. Global Journal of Medical Research - G: Veterinary Science & Medicine GJMR-G Volume 24 (GJMR Volume 24 Issue G1): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
This work aims to evaluate the effects of rapid thermoresistance on the morphofunctional aspects of membranes using flow cytometry, as well as the quality of sperm chromatin using the Toluidine Blue technique, both before and after the thermoresistance test. Six sets of frozen semen from ten bulls were used. The samples were thawed in a water bath at 36ºC and separated into two aliquots. The first aliquot, control group (C), was kept in a test tube previously heated to 36ºC with subsequent evaluationsusingmicroscopy, cytometry, and chromatin analyses. The second aliquot, experimental group (E), underwent the rapid thermoresistance test (RTT) in a water bath preheated at 46ºC for 30 minutes with subsequent evaluations usingmicroscopy, cytometry, and chromatin analyses. For statistical analysis, the paired T test, significance (P
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.