Degenerative mitral valve disease (DMVD) consists of progressive degeneration of the mitral valve that can occur either in isolation or in association with the tricuspid valve, leading to incomplete coaptation of its leaflets and subsequent valve regurgitation. It’s the most common cardiac condition in small animals, accounting for about 75% of cases of heart disease in dogs, mainly affecting elderly small-breed dogs. Breeds with a genetic predisposition, such as Cavalier King Charles Spaniels and Bull Terriers, tend to develop the disease early. It can be classified into four stages: A, B, C, and D, with B having subdivisions (B1 and B2). Animals in stages A, B1, and B2 are asymptomatic, showing clinical signs only from stage C onwards. The report details the case of a 13-year-old Fox Terrier diagnosed at stage B2 of the disease, progressing to stage C within two years-a cardiologist conducted annual monitoring.
Simple Summary- This work aims to report the case of an animal referred to a cardiologist in 2021, being diagnosed with Myxomatous Mitral Valve Disease in stage B2, and demonstrating the evolution of the disease to date.
## I. INTRODUCTION
D egenerative myxomatous mitral valve disease (DMVM) is the most common heart disease in small animals, accounting for around $75\%$ of cases of heart disease in dogs [9]. The prevalence of MVD increases markedly with age in small dogs, with up to $85\%$ showing evidence of valve damage by 13 years of age [3].
According to Petrus Gimenes and Mantovani (2019), the proper functioning of the mitral valve is based on the structural and functional performance of six components: the posterior wall of the left atrium, the valve ring, the valve leaflets or cusps, the tendon chordae, the papillary muscles of the left ventricle and the left ventricular wall, each of these components plays an independent and synergistic role, contributing to complex functions that maintain valve competence. Any structural changes in the elements of the mitral apparatus affect valve mechanics, compromising its efficiency.
The disease involves a gradual myxomatous degeneration over time with the disorganization of collagen bundles and a reduction in their content, excess production of glycosaminoglycan that results in a change in the valve structure leading to poor coaptation of the leaflets. Poor coaptation of the leaflets allows regurgitation of the mitral valve, resulting in the murmur characteristic of the disease [7].
According to the latest consensus published by ACVIM in 2019, an echocardiogram (ECHO) and chest x-ray in the absence of ECHO are recommended to diagnose the disease. However, caution should be taken due to the marked variation in thoracic conformation and racial differences in the vertebral cardiac scales. The disease can also be recognized during a screening or routine examination by auscultating a typical heart murmur when there is regurgitation of the mitral valve.
The main symptoms that the animal presents when it reaches the advanced stage of the disease are Exercise intolerance, dry cough, decreased appetite, difficulty breathing, and syncope, which are findings associated with a worse prognosis in affected dogs [6].
According to the classification system for treating dogs with DMVM, published by Atkins et al. 2019, described below (Table 1) [9], there are four primary stages of the disease.
<table><tr><td>Stages</td><td>Description</td></tr><tr><td>Stage A</td><td>Identifies patients at high risk of developing heart disease but who do not yet have identifiable structural changes in the heart.</td></tr><tr><td>Stage B</td><td>Identifies patients with structural heart disease but who have never developed clinical manifestations of heart failure. This stage is divided into: B1: Asymptomatic patients who do not present radiographic or echocardiographic evidence of cardiac remodeling in response to valve disease.</td></tr><tr><td></td><td>B2: Asymptomatic patients who present hemodynamically significant mitral valve regurgitation, evidenced by radiographic and echocardiographic findings of left heart enlargement.</td></tr><tr><td>Stage C</td><td>Patients with previous or current clinical symptoms of heart failure associated with structural changes in the heart.</td></tr><tr><td>Stage D</td><td>Patients with end-stage heart disease with signs of heart failure who are refractory to mainstay therapy require particular or advanced treatment strategies to remain comfortable regardless of the presence of the disease.</td></tr></table>
<table><tr><td>Stages</td><td>Description</td></tr><tr><td>Stage A</td><td>Identifies patients at high risk of developing heart disease but who do not yet have identifiable structural changes in the heart.</td></tr><tr><td>Stage B</td><td>Identifies patients with structural heart disease but who have never developed clinical manifestations of heart failure. This stage is divided into: B1: Asymptomatic patients who do not present radiographic or echocardiographic evidence of cardiac remodeling in response to valve disease.</td></tr><tr><td></td><td>B2: Asymptomatic patients who present hemodynamically significant mitral valve regurgitation, evidenced by radiographic and echocardiographic findings of left heart enlargement.</td></tr><tr><td>Stage C</td><td>Patients with previous or current clinical symptoms of heart failure associated with structural changes in the heart.</td></tr><tr><td>Stage D</td><td>Patients with end-stage heart disease with signs of heart failure who are refractory to mainstay therapy require particular or advanced treatment strategies to remain comfortable regardless of the presence of the disease.</td></tr></table>
This work aims to report the case of an animal referred to a cardiologist in 2021, diagnosed with Myxomatous Mitral Valve Disease in stage B2, and demonstrate the evolution of the disease to date.
## II. CASE REPORT
A Fox Terrier dog male, 13 years old, neutered, weighing $12\mathrm{kg}$, was treated at the Veterinary Clinic Animal Medical Center (AMC) in Pouso Alegre - MG, Brazil, and was sent for cardiological evaluation. He consulted a general practitioner due to complaints of intense tremors, where a chest x-ray was requested. During this examination, an increase in the cardiac silhouette was observed in a topography corresponding to the left atrium, resulting in the patient being referred to a specialist.
In the anamnesis, the owners reported that the animal never had coughs, syncope, or convulsions, just tremors since it was a puppy. On physical examination, a grade 3/6 murmur was heard in the mitral focus, normal heart, and respiratory rates, systemic arterial hypertension (180mmHg), and normal-colored mucous membranes.
An echocardiogram (ECHO) (Figure 1) showed a thickened mitral valve and enlarged heart chambers. At first was prescribed Pimobendan PO at a dose of $0.25\mathrm{mg/kg}$ BID, continuous use, and requested to return in 30 days.
Upon return, the owners reported that the animal was more active and was not quickly tired. His pressure had already decreased from $180 \mathrm{mmHg}$ to $160 \mathrm{mmHg}$, and he continued using pimobendan.
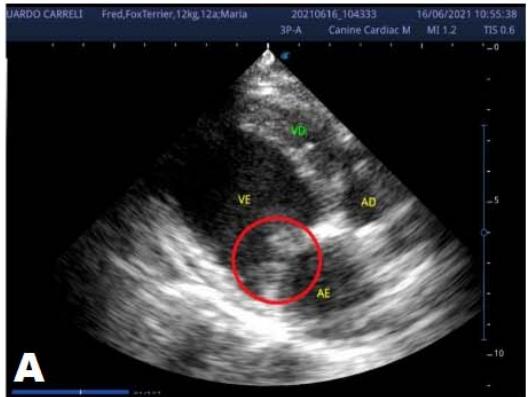
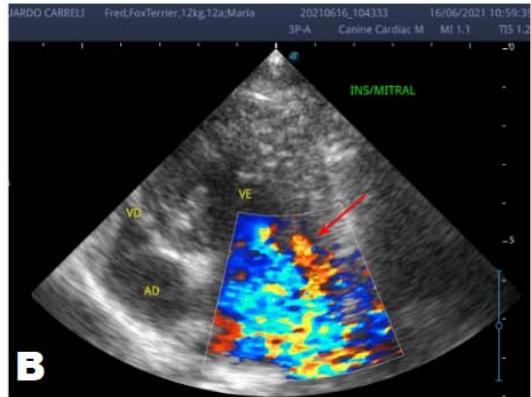
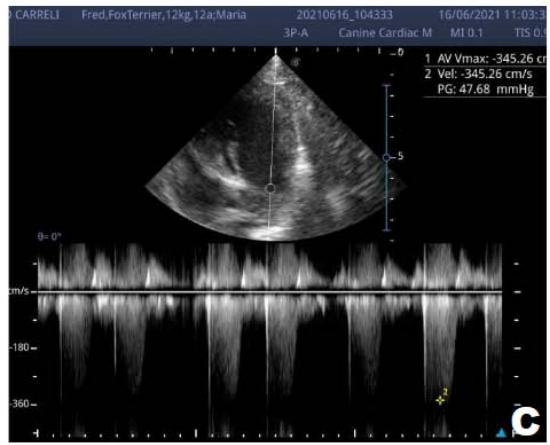 Figure 1: Echocardiogram 2021: Thickened/degenerated mitral valve (A); Observed in Doppler mode, significant mitral valve insufficiency (B); Hemodynamic assessment - Mitral regurgitation gradient: $3.45 \mathrm{~m} / \mathrm{s} / 47.68 \mathrm{mmHg}$; enlargement of left cardiac chambers; left ventricular diastolic dimension above normal limits was observed
One year later, the animal returned for a new evaluation, repeating the ECHO (Figure 2), which showed the progression of the increase in the left atrium. The electrocardiogram (ECG) (Figure 3) shows baseline sinus arrhythmia with the presence of a premature ventricular complex (Figure 4). Owners report drowsiness, hyporexia, and syncope. However, they did not present tiredness or cough. Blood pressure was 80 mmHg.
Due to the arrhythmia, Sotalol was prescribed at a dose of $25\mathrm{mg/kg}$ PO, BID for continuous use, and Omega 3 PO at a dose of $600\mathrm{mg/kg}$ SID became necessary, and pimobendan continued.
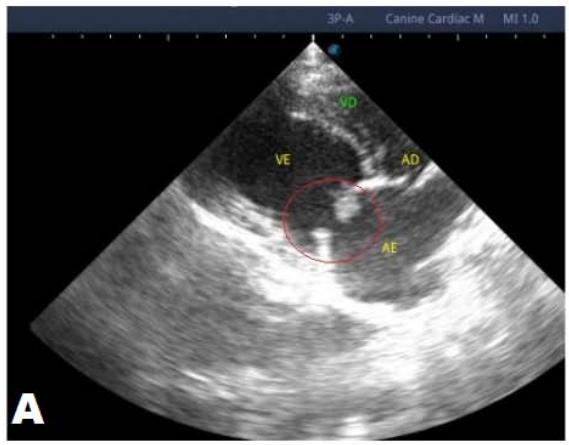
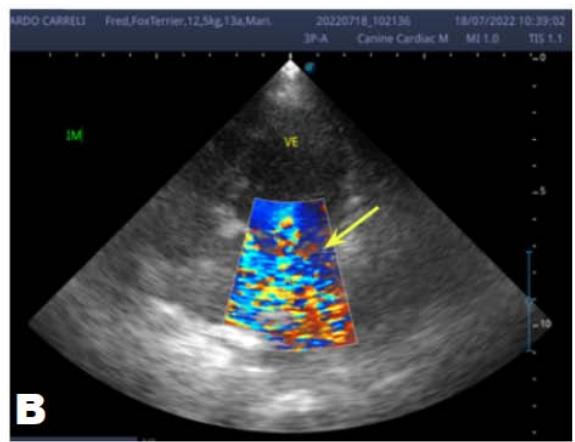
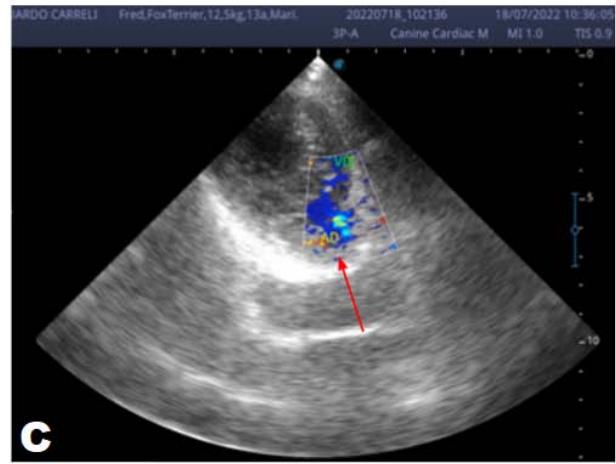 Figure 2: Echocardiogram (2022): observed increase in the left atrium and ventricle; thickened/degenerated mitral valve (A); observed in a Doppler study, turbulent systolic flow within the left atrium, characterizing significant mitral valve insufficiency (B); systolic turbulent flow within the right atrium, representing mild tricuspid valve insufficiency (C); hemodynamic assessment - maximum velocity gradient mitral regurgitation: $3.51\mathrm{m / s} / 49.22\mathrm{mmHg}$; observed left ventricular diastolic dimension above normal limits with normal systolic function parameters, characterizing systolic dysfunction; preserved diastolic function
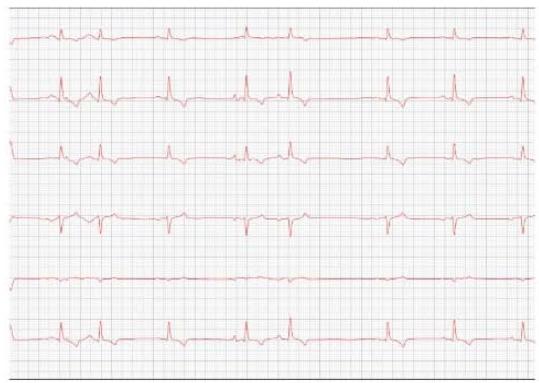
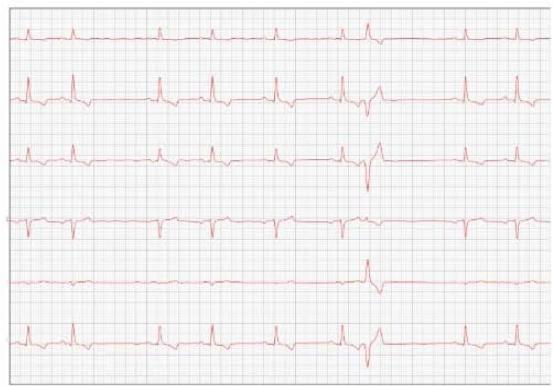
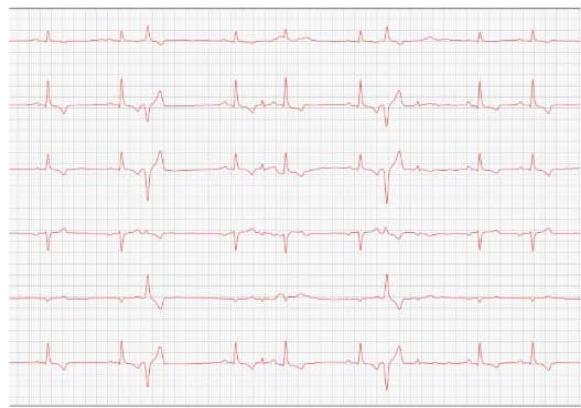 Figure 3: Electrocardiogram (2022)
Table 2: Electrocardiographic report (2022)
<table><tr><td>Observed parameters</td><td>Observed parameters</td><td>Observed parameters</td></tr><tr><td>QRS axis: 67.07°</td><td>QT Interval: 210 ms</td><td>Duration of T: 54 ms</td></tr><tr><td>P axis: 58.01°</td><td>PR Interval: 108 ms</td><td>QRS duration: 72 ms</td></tr><tr><td>Amplitude of S: -0.06 mV</td><td>R amplitude: 2.26 mV</td><td>Minimum HR: 49 bpm</td></tr><tr><td>PR Segment: 52 ms</td><td>P amplitude: 0.22 mV</td><td>Average HR: 95 bpm</td></tr><tr><td>ST segment: 84 ms</td><td>T amplitude: -0.74 mV</td><td>Maximum HR: 297 bpm</td></tr><tr><td>Duration of P: 56 ms</td><td></td><td></td></tr></table>
Comments
Baseline sinus arrhythmia.
The QRS axis is within normal limits for the species. Episodes of the premature ventricular complex were observed.
An increase in the P wave and QRS complex duration was observed, suggesting atrial and left ventricular overload.
Ventricular repolarization disorder was observed due to the rise in the amplitude of the T wave ( $>25\%$ of the R wave), which is compatible with electrolyte changes and myocardial hypoxia.
Conclusions: Baseline sinus arrhythmia with the presence of premature ventricular complex
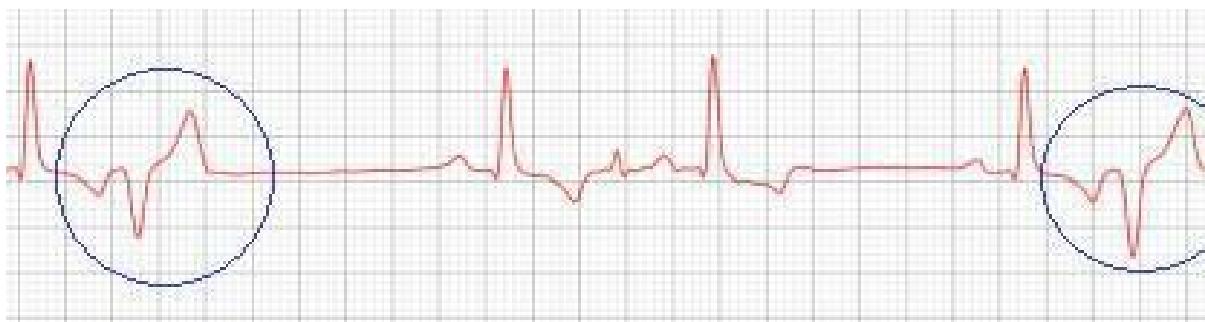 Figure 4: Electrocardiographic tracing (2022): showing episodes of premature ventricular complex (circles)
At the beginning of 2023, the patient returned to repeat the cardiological evaluation. During the anamnesis, the Guardian reported that the patient did not present episodes of syncope and remained stable during this period, continuing the previously adopted treatment.
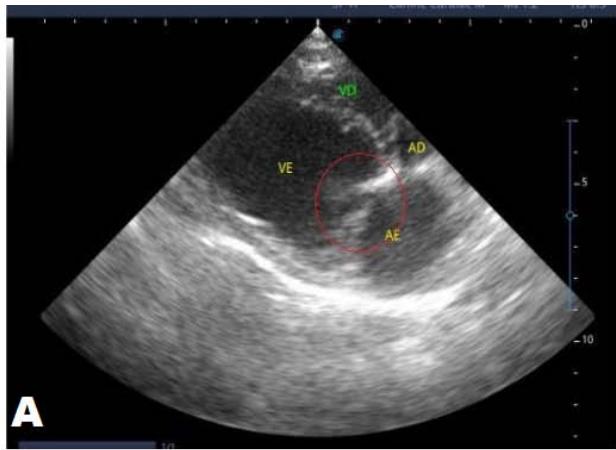
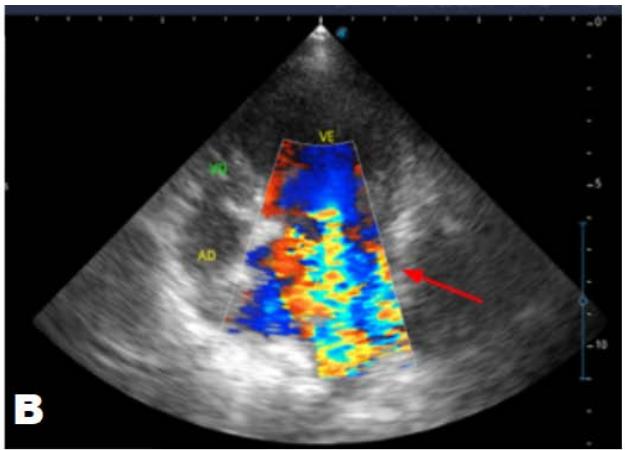
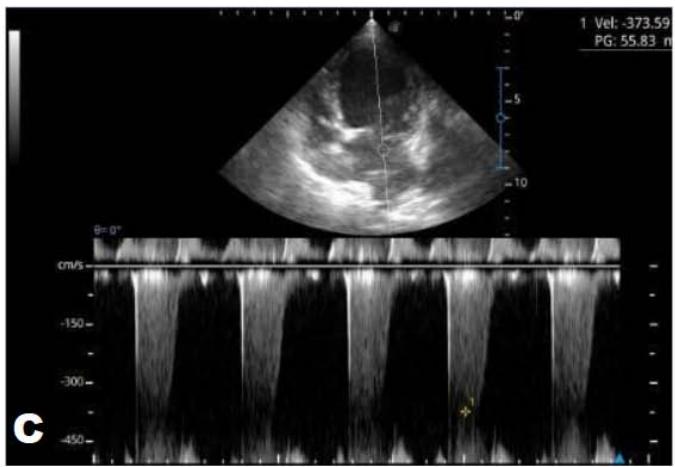 Figure 5: Echocardiogram (January/2023): observed increase in the left atrium and ventricle; thickened/degenerated/prolapsed mitral valve (A); observed in Doppler study, systolic turbulent flow within the left atrium characterizing severe mitral valve insufficiency (B). Hemodynamic assessment - Maximum velocity gradient mitral regurgitation: $3.74 \, \text{m/s} / 55.83 \, \text{mmHg}$: Observed left ventricular diastolic dimension above standard limits, with normal systolic function parameters, characterizing systolic dysfunction; preserved diastolic function
After eight months, the patient returned with a worsening condition, repeated ECHO (Figure 6) and ECG (Figure 7), where diastolic dysfunction and worsening of the arrhythmia were observed. Amiodarone $8.7\mathrm{mg/kg}$ orally was prescribed, replacing Sotalol, one tablet BID, and Furosemide (Lasix) 1.7 mg/kg orally was prescribed every 12 hours.
Pimobendan was continued. After a week, a reassessment was carried out, and the ECG demonstrated stability in the arrhythmia (Figure 8).
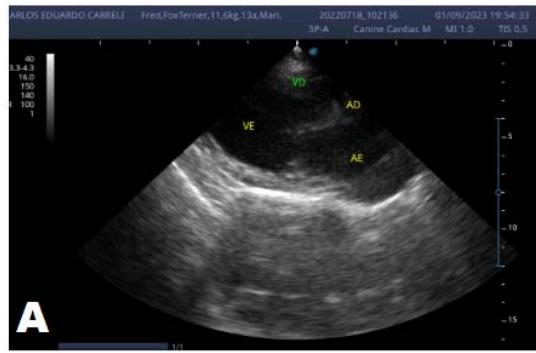
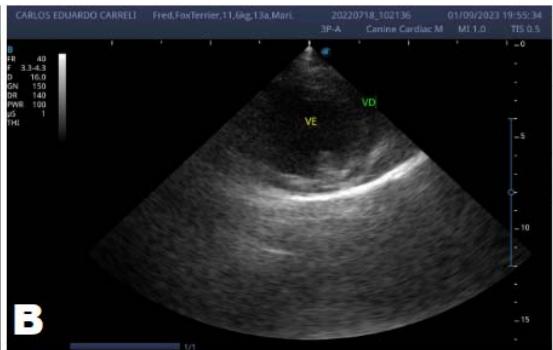
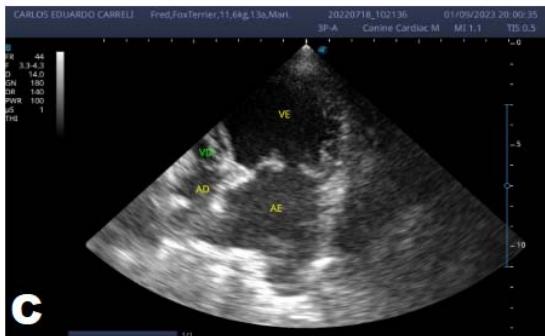
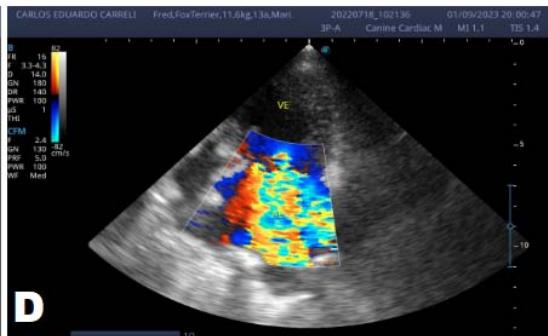
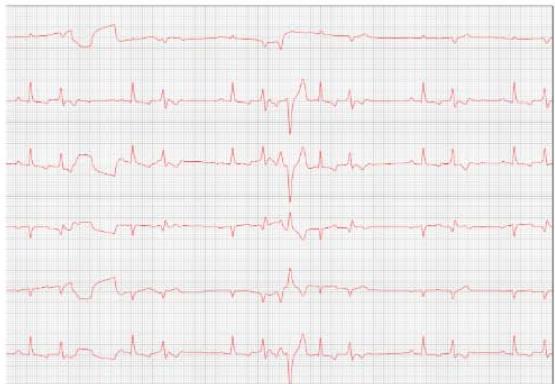 Figure 6: Echocardiogram (September/2023): significant enlargement of the left heart chambers observed; observed in a Doppler study, systolic turbulent flow within the left atrium characterizing significant mitral valve insufficiency; Observed left ventricular diastolic dimension above standard limits, with normal systolic function parameters, indicating systolic dysfunction; A left ventricular filling pattern of the $\mathrm{E} > \mathrm{A}$ wave type and an increased E/A ratio were observed, characterizing diastolic dysfunction (restrictive or pseudo-restrictive pattern)
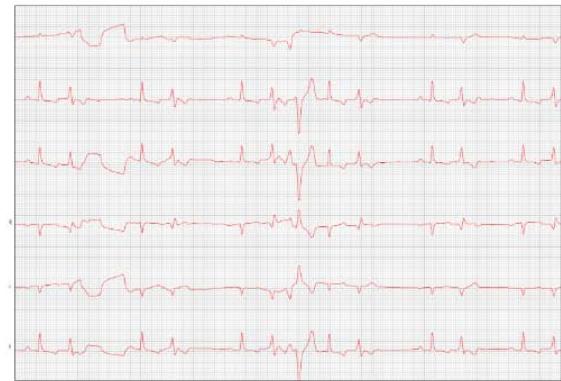 Figure 7: Electrocardiogram (September/2023)
Table 3: Electrocardiographic report (September/2023)
<table><tr><td>Observed parameters</td><td>Observed parameters</td><td>Observed parameters</td></tr><tr><td>Minimum HR: 71 bpm</td><td>T duration: 68 ms</td><td>S amplitude:: -0.01 mV</td></tr><tr><td>Average HR: 125 bpm</td><td>QT interval: 196 ms</td><td>T amplitude: -0.32 mV</td></tr><tr><td>Maximum HR: 400 bpm</td><td>ST segment: 56 ms</td><td>P axis: 83.89 °</td></tr><tr><td>P duration:: 48 ms</td><td>P amplitude: 0.29 mV</td><td>QRS axis: 81.32 °</td></tr><tr><td>QRS duration:: 72 ms</td><td>R amplitude: 1.69 mV</td><td>PR Interval: 104 ms</td></tr></table>
Comments:
Sustained ventricular bigeminy with clusters of ventricular trigeminy and sinus arrhythmia.
Duration and amplitude of the P wave and QRS complex with values within normal limits for the patient's species, size, and age.
Normal electric axis.
Normal T wave.
Presence of polymorphic premature ventricular extrasystoles, isolated in pairs, in triplets, and at times organized into ventricular bigeminy and trigeminy.
Nothing else is worth noting during the 4 minutes and 41 seconds of monitoring.
Conclusions: Sustained ventricular bigeminy with clusters of ventricular trigeminy and sinus arrhythmia.
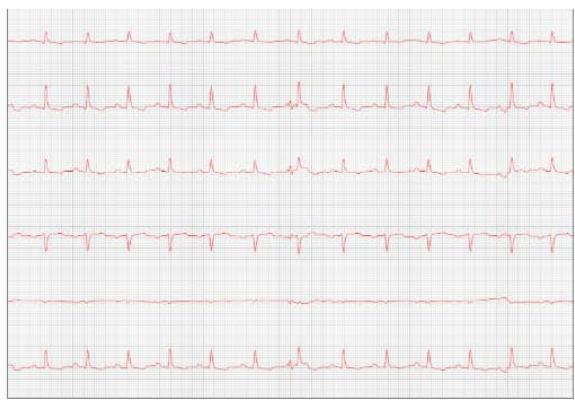
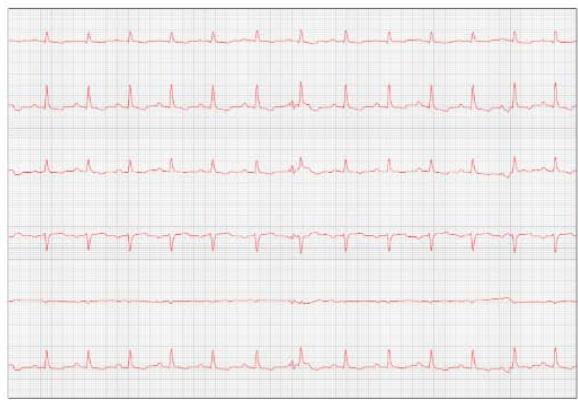 Figure 8: Reevaluation of the electrocardiogram (September/2023)
## III. DISCUSSION
In the reported, he was asymptomatic, and the echocardiographic examination showed enlargement of the left heart chambers, thickened and degenerated mitral valve (Figure 1), left ventricular diastolic dimension above normal limits. with normal systolic function parameters, characterizing systolic dysfunction. The thickening and degeneration of the mitral valve indicates its insufficiency, and the other changes represent diastolic dysfunction.
Diagnosed with degenerative myxomatous mitral valve disease, treatment with pimobendan began. According to Boswood et. al. (2016), its mechanism of action includes a positive inotropic combination and balanced vasodilation caused by calcium sensitization and phosphodiesterase inhibition, as a result, the effects of pimobendan may consist of increased cardiac output (CO), myocardial contractility and decreased preload and afterload.
In 2022, the animal returned for annual exams one year after the diagnosis. The ECHO showed an increase in the left atrium and ventricle, thickened/degenerated mitral valve (Figure 2), preserved diastolic function, and low probability of pulmonary hypertension; the ECG detected an increase in the duration of the P wave and the QRS complex (Figure 3), suggestive of atrial and left ventricular overload, baseline sinus arrhythmia with the presence of a premature ventricular complex.
In most cases, cardiac arrhythmias are not clinically relevant unless they are associated with heart disease or cause an extreme change in heart rate, being very slow, fast, or irregular. [10]. When choosing a medication for treatment, one must consider some things, such as clinical status, associated cardiac or systemic comorbidities, and drug association [13]. Class III antiarrhythmics block potassium channels, preventing a new action potential from occurring before complete repolarization, causing a prolongation of this potential and the refractory period [5].
Sotalol was prescribed to control the arrhythmia presented by the animal with DMVM. According to Treseder et al. (2019), Sotalol is an antiarrhythmic agent commonly used in human and veterinary medicine to control ventricular and supraventricular arrhythmias. Its antiarrhythmic properties are well established and are attributed mainly to the blockade of potassium channels with concomitant non-selective $\beta$ -adrenergic blockade.
In 2023, the animal returned and repeated only the ECHO (Figure 5), where no significant changes were observed during this period, except for a structural change in the mitral valve, identifying a prolapse and increase in the left ventricular diastolic dimension, with systolic function parameters, characterizing systolic dysfunction; diastolic function remained preserved.
Petrus, Gimenes, and Mantovani (2019, p. 156) report that mitral valve prolapse is a common complication that generally occurs with the progression of MVD. As MVD progresses, mitral valve regurgitant flow increases, promoting volume overload in the left atrium, with an increase in filling pressures in the left heart chambers. There is also progressive cardiac remodeling, characterized by eccentric hypertrophy, initially in the left atrium and, later, in the left ventricle. Systolic dysfunction occurs in the advanced stages of MVD, which culminates in hemodynamic changes and progressive diastolic dysfunction of the left ventricle, affecting general cardiac performance and determining the appearance of congestive heart failure syndrome.
In the same year, eight months later, the patient returned with a worsening condition, presenting pulmonary edema. The animal's condition progressed to stage C with these changes, requiring repeat echocardiographic and electrocardiographic examinations.
The ECHO (Figure 6) showed an increase in the cardiac chambers and diastolic dysfunction compared to the previous exam. The ECG (Figure 7) revealed polymorphic premature ventricular extrasystoles. Due to this change, Sotalol was replaced by amiodarone.
Amiodarone is also a class III antiarrhythmic, being the most indicated in these cases as it is considered broad spectrum and can be used in supraventricular and ventricular arrhythmias [5].
However, its use should be reserved for cases where the animal no longer responds to sotalol treatment due to its various side effects [10]. Its most common adverse effects are liver changes, causing an increase in the concentration of gastrointestinal and enzymes, such as vomiting, diarrhea, and anorexia [11]. Usually, these effects are dose-dependent and reversible if their supply is interrupted at the beginning of clinical manifestations [10].
For pulmonary edema, furosemide (Lasix) was prescribed. Furosemide is a loop diuretic widely used in veterinary clinics. The Food and Drug Administration (FDA) has approved furosemide to treat conditions with volume overload and edema secondary to exacerbation of congestive heart failure, liver failure, or renal failure, including nephrotic syndrome [4]. Its mechanism of action is to inhibit the tubular reabsorption of sodium and chloride in the proximal and distal tubules and in the thick ascending loop of Henle, inhibiting the sodium chloride cotransport system, resulting in excessive excretion of water along with sodium, chloride, magnesium, and calcium [4].
One week later, the patient returned for a reevaluation of the ECG (Figure 8), demonstrating stability in the arrhythmia.
In 2023, there were some complications regarding the patient's treatment. The owner reported that he was no longer able to administer the medications as recommended, providing pimobendan every 24 hours or at a longer interval. Regarding other medications, it was said that the animal accepted it more quickly but could not administer it at the correct time, which may justify the worsening of the condition in a short period. The animal is currently stable and continues to receive monitoring.
## IV. CONCLUSION
Myxomatous mitral valve degeneration is a progressive disease responsible for the most significant number of heart diseases in small animals. We observed that the time for the disease to progress from stage B2 to C was two years, with correct monitoring and treatment reducing its progression over a while. Medications were essential in this case to keep the animal's condition stable.
Generating HTML Viewer...
References
13 Cites in Article
A Al-Mosawi (2023). Pharmacological Treatment of Chronic Symptomatic Premature Ventricular Contractions: An Educational Article and Expert Opinion.
A Boswood,J Häggström,S Gordon,G Wess,R Stepien,M Oyama,B Keene,J Bonagura,K Macdonald,M Patteson,S Smith,P Fox,K Sanderson,R Woolley,V Szatmári,P Menaut,W Church,M O'sullivan,J-P Jaudon,J-G Kresken,J Rush,K Barrett,S Rosenthal,A Saunders,I Ljungvall,M Deinert,E Bomassi,A Estrada,M Fernandez Del Palacio,N Moise,J Abbott,Y Fujii,A Spier,M Luethy,R Santilli,M Uechi,A Tidholm,P Watson (2016). Effect of Pimobendan in Dogs with Preclinical Myxomatous Mitral Valve Disease and Cardiomegaly: The EPIC Study—A Randomized Clinical Trial.
Bruce Keene,Clarke Atkins,John Bonagura,Philip Fox,Jens Häggström,Virginia Fuentes,Mark Oyama,John Rush,Rebecca Stepien,Masami Uechi (2019). ACVIM consensus guidelines for the diagnosis and treatment of myxomatous mitral valve disease in dogs.
Tahir; Khan,Roshan; Patel,Abdul Siddiqui (2018). Furosemide. Study Guide from StatPearls Publishing.
Maria Larsson,Matiko Helena,Akao (2023). Medicamentos antiarrítmicos.
Giulio Menciotti,Michele Borgarelli (2017). Review of Diagnostic and Therapeutic Approach to Canine Myxomatous Mitral Valve Disease.
Andrew Mcnair,Greg Markby,Qiyu Tang,Vicky Macrae,Brendan Corcoran (2023). TGF-β phospho antibody array identifies altered SMAD2, PI3K/AKT/SMAD, and RAC signaling contribute to the pathogenesis of myxomatous mitral valve disease.
A Pinkos,C Stauthammer (2021). Degenerative Valve Disease: Classification, Diagnosis, and Treatment of Mitral Regurgitation.
Jim Riviere,Mark Papich,Booth (2021). Farmacologia e Terapêutica Veterinária.
Kátia Tárraga,Mitsube (2017). Anais do(a) Anais da escola de Nanotecnologia Aplicada à Farmacologia Veterinária & Simpósio Internacional de Farmacologia e Terapêutica Veterinária.
J Treseder,N Leblanc,K Scollan (2019). Inotropic and chronotropic effects of sotalol in healthy dogs.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Keren Souza. 2026. \u201cEvolution of Degenerative Myxomatous Mitral Valve Disease in a Canine Fox Terrier: A Case Report\u201d. Global Journal of Medical Research - G: Veterinary Science & Medicine GJMR-G Volume 24 (GJMR Volume 24 Issue G1): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Degenerative mitral valve disease (DMVD) consists of progressive degeneration of the mitral valve that can occur either in isolation or in association with the tricuspid valve, leading to incomplete coaptation of its leaflets and subsequent valve regurgitation. It’s the most common cardiac condition in small animals, accounting for about 75% of cases of heart disease in dogs, mainly affecting elderly small-breed dogs. Breeds with a genetic predisposition, such as Cavalier King Charles Spaniels and Bull Terriers, tend to develop the disease early. It can be classified into four stages: A, B, C, and D, with B having subdivisions (B1 and B2). Animals in stages A, B1, and B2 are asymptomatic, showing clinical signs only from stage C onwards. The report details the case of a 13-year-old Fox Terrier diagnosed at stage B2 of the disease, progressing to stage C within two years-a cardiologist conducted annual monitoring.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.