Introduction-There is very little literature on the floral biology, synchronization of maturity of male and female flowers, flowering phenology and controlled pollination of Dioscorea dumetorum (D. dumetorum). However, the genetic improvement of this yam species, which is widely consumed in Cameroon and has exceptional nutritional qualities, cannot be achieved without knowledge and understanding of these biological attributes or phenomena. The aim of this study was to (1) -examine the sexual reproductive biology of D. dumetorum; (2) -study the phonology of flowering in this plant species; and (3) -develop a pollination method in order to obtain hybrid seedlings.
## I. INTRODUCTION
Dioscoreaceae (yam) of the order Dioscoreales and genus Dioscorea, constitute a family of monocotyledonous flowering plants with about 750 known species in 9 genera (Xu and Chang, 2017) distributed in 59 sections (Govaerts and al; Coursey, 1967). True seeds are seldom used probably due to variation of flowering in yam. Several yam species are grown for their edible tuberous roots, which constitute a staple food or traditional medicine for most communities. For the same amount of raw materials consumed, the energy value of yam is generally lower than that of other roots, tubers or starchy fruit, but the coverage of protein requirements, minerals and vitamins is more complete (Trèche, 1998). Like other root or tuber crops (cassava, irish potato, sweet potato), yams are generally vegetatively propagated from seed tubers or sections of tubers, rhizomes and bulbils. However, yams can also reproduce by seed. This is the most successful way to improve the species.
Eight yam species are commonly grown and consumed in West and Central Africa. They include D. dumetorum, D. esculenta, D. bulbifera, D. schimperiana, D. semperflorens, D. alata and D. burkilliana. Among them, D. dumetorum (bitter yam) is the most nutritious (Sefa-Dedeh and Afoakwa, 2002). Bitter yam belongs to the section Lasiophyton which contains 51 edible species originating from tropical Africa. It occurs in both wild and cultivated forms (Degras, 1993), resulting in a huge unexplored genetic diversity that could benefit the genetic improvement of this orphan species.
A major constraint to the international and even local development of this yam species with high nutritional potential is caused by its storage ability. Postharvest hardening of the tubers of this yam species begins within 24 h after harvest and renders them unsuitable for human consumption (Sefa-Dedeh and Afoakwa, 2002; Siadjeu and al, 2021). Hardening is manifested by the loss of culinary quality due to a combination of factors resulting from normal but inadvertently deleterious reactions leading to textural changes (Medoua, 2005). Therefore, D. dumetorum is consumed exclusively during its limited harvest period. Thus, only freshly harvested tubers are cooked and sold to consumers. Ground work has been done in our previous endeavors towards the genetic improvement of D. dumetorum. This includes studies on the genetic diversity using morphological markers (Siadjeu and al, 2015); genetic diversity and population structure (Siadjeu and al, 2018); the influence of some agronomic traits on yield indicating the optimal seedling weight at 300 gr, (Mahbou and al, 2015), or the influence of the cultivar on postharvest hardening of trifoliate yam (Siadjeu and al, 2016) and more. recently, a candidate gene controlling the post-harvest phenomenon has been detected (Siadjeu and al, 2021). A second major constraint for the amelioration of Dioscorea spp. could be due to erratic flowering and poor seed production and fruiting (Mondo and al, 2020) as noted on related species Dioscorea alata and Dioscorea cayenensis-rotundata complex. A third constraint that may need to be addressed in breeding is the asynchronous flowering of male and female plants as noticed in Dioscorea cayenensis-rotundata complex (Zoundjihékpon and al, 1997). True seeds are being used successfully in breeding programs (Girma and al, 2014), such as the case of D. alata and D. cayenensis-rotundata (Lebot, 2009) in addition to tuber sets. Hybridization of yam has become feasible due to a better understanding of the reproductive biology of the cultivated species (Asiedu and al, 1998). However, unlike other yams, little information is available concerning the complete description of the flowers and flowering pattern of $D$. dumetorum.
The improvement of a species by conventional means requires (1) - mastery of its floral biology; (2) - an understanding of factors that could influence anthesis, i.e. the blossoming of flowers; (3) knowledge of phenology of biological phenomena leading to flowering; (4) - the development of a viable pollination method leading to viable seeds for the production of seedlings. The aim of this study is to give answers to these four concerns by gathering information, which could be useful for the genetic improvement of this orphan crop species.
### a) Plant materials, methods and study area
The plant material used in this study consists of 47 cultivars of $D$. dumetorum yam collected in three regions of Cameroon (Table 1). In order to study floral biology, a total of 20 plants of each cultivar were planted in two repetitions of 10 plants/block. The experimental site is located at the Boukue School Farm (FEBO) in the West Region of Cameroon, Hauts- Plateaux-Department, Baham District. Location, climate and soil characteristics are indicated in previous studies (Mahbou and al, 2021).
### b) Field planting
The yam tubers ware planted in the field at 1 m spacing in each row, and 1 m between rows. The tubers were arranged in a randomized complete block with a plot of two replicates of ten tubers per accession. Each plant was supported by an individual stake of Phyllostachys to induce good canopy development. Tuber sets were planted in April 2018, 2019 and 2020. Given the acidity of the soil, each plant received 0.5kg of poultry manure and 100 gr of Tropicote (21% N + 25% CaO) and 50 gr of NPK (20/10/10) each year.
# c) Biology of
Observations were made twice from mid-June to November in 2018 and 2019. Five plants of each cultivar were randomly selected to observe inflorescence and flower behaviour. Observations included Inflorescence Number (InfN), Inflorescence Length (InfLg), Number of Flowers (NumF), Flower Length (FlwLg), Flower Diameter (FlwD) and Flower Density calculated as a ratio of number of flowers per centimeter of inflorescence. The description of the floral structure was done with the help of a magnifying glass (x8, x10, x16) and camera. The analysis of variance (ANOVA), followed by the comparison of means was carried out using Duncan's method at the $5\%$ threshold. Pearson correlation coefficient was calculated to determine the relationship among traits.
### d) Phenology of biological phenomena leading to flowering
## i. Flowering pattern within a year and anthesis
In order to study flowering pattern, data was collected including number of Days to Emergence (DE), Days to First Bud Emergence (DFB), Days to First Flower Opening (DFFO), Days to First Bud emergence to First Flower Opening (DTB-DFFO), and Number of Flowered Plants (NFP). For anthesis, observations included those made on a weekly basis within a day (number of newly opened flowers per hour in one day, and number of newly opened flowers per week in one month) on the one hand and those made annually on a three-year period on the other hand. The dimensions (length and diameter) of the reproductive system were measured using electronic calipers and ruler. These parameters were recorded until senescence. Data represented the mean of five inflorescences per plant.
Quantitative data recorded in this section (InfN, InfLg, NumF, FlwLg, FlwD, DE, DFB, DFFO, DFB-DFFO, and NFP) were subject to exploratory statistics to test the normality of the distribution using the Kolmogorov test. In case of normality of data, ANOVA analysis were done and means separated using Duncan Multiple Range Test at $5\%$ threshold. In case of abnormality of the distribution, comparison was done with the Mann Whitney test according to Monte Carlo method at $5\%$ significance level. Statistical analyses were performed using IBM/SPSS statistics 20 software.
### e) Flowering rate of different D. dumetorum accessions as a function of time
Data on the flowering status of 47 cultivars were collected during three years. Analysis of data was realized by the binomial logistic regression analysis with Monte Carlo permutation in order to establish the relation between flowering, year and sex and lastly at $95\%$ confidence interval and Kendall correlation coefficients at $5\%$ threshold to evaluate possible relations among year, sex and flowering.
### f) Synchronism of maturation of male and female flowers
In order to evaluate the synchrony of maturation of the male and female flowers of the different accessions with respect to the possibilities of crossbreeding, we use time to maturity (TM). TM is defined as the time (measure in days) to maturity in flowering plant, considered to be the period from emergence to the maximum opening of male and female flowers. This was recorded over a three-year period during the years 2018, 2019 and 2020. Data were analyzed using a T test at $5\%$ significance level for time to maturity (TM). For flowering response as a function of year and sex of the plant, Binomial logistic regression was performed.
### g) Evaluation of the best pollination method
To evaluate the possibilities of controlled sexual hybridization, three controlled pollination methods were tested. Bamboo splinter pollination, wire brush pollination and syringe/pollen solution pollination as illustrated on figure 3. Two controls (positive control and negative control) allowed us to evaluate the performance of these pollination techniques with respect to fruiting. The analysis of variance (ANOVA), followed by the comparison of means were carried out using LSD method at the $5\%$ threshold based on fructification performance.
## II. RESULTS
### a) Biology of sexual reproduction of D. dumetorum
In general, significant variations were observed on all the morphological parameters between male and female accessions, showing that there was a significant source of variation that could be used in the improvement programs of $D$ dumetorum based on the selection of objectives and to mitigate the problems observed relative to flowering in yams.
### b) Structure of the female flowers
The female inflorescences are spikes (Figure 1A). They are formed in the leaf axils at the level of the flowering axes. Each flowering node has 1 or 2 inflorescences. The inflorescences are oriented downwards in relation to the branches. The perianth is of a single bell-shaped leaflet forming a large three-angled capsule, corresponding to three lobes. The corolla is absent, the calyx consists of three sepals and the pistil consists of a trilocular inferior ovary, three simple styles and a central trifurcate or trigonal stigma (divided into three branches) (Figure 1B). Each lobe of the ovary contains two apically placentating ovules (Figure 1C). The flowers are piliferous and purple or violet in colour and have two lanceolate bracts.
### c) Structure of the male flowers
The male inflorescences are panicles (formed by clusters of spikelets) (Figure 2A). Morphologically, the immature inflorescences are very different from the mature ones. They appear to be single spiked at first but after full development, panicles are observed. The flowers are sessile, spherical and protected by bracts. The flowers consist of a calyx with 3 sepals and a corolla with 3 petals, arranged regularly and almost similar in size and appearance (Figure 2B). The sepals are white and the petals greenish. The stamens are 6 in number and each consists of a very short, capillary filament and a single anther. The anthers at maturity take on different colours depending on the accessions. They are white (Muyuka 3, Mabondji sweet white 1), yellowish (Ibo sweet 2) and purple (Bayangam 1). This result confirms that D. dumetorum belong to the narrow $10\%$ of flowering plants that are said to be doiecious, a prevalent mating system in Cucurbitacea family (Ainsworth C, 2000) (Figure 1 & 2), more precisely $D$ dumetorum is gynodioecious.
### d) Morphology of female flowers
The number of inflorescences (InfN) varied from $4.60 \pm 2.60$ (Bamendjou 2) to $33.7 \pm 21.6$. (Country yam 1) with a mean of $14.6 \pm 8.2$. The length of the inflorescences (InfLg) varies from $40 \pm 17 \mathrm{~mm}$ (Bambui 1) to $215 \pm 69$ (Ibo sweet 1) mm with a mean of $92 \pm 46$. The number of flowers (FlwN) per inflorescence varied from $11.3 \pm 2.1$ (Bambui 1) to $18.1 \pm 2.8$ (Bamendjou 2) with a mean of $15.1 \pm 2.4$. The length of the flower (FlwLg) varies from $2.20 \pm 0.4$ (Baigon 1) to $13.6 \pm 1.8 \mathrm{~mm}$ (Ibo sweet 1) with a mean of $10.7 \pm 3.0$. The diameter of the flower (FlwD) varies from $1.4 \pm 0.2 \mathrm{~mm}$ (Baigon 1) to $5.7 \pm 0.6 \mathrm{~mm}$ (Mankon 1) with a mean of $(4.2 \pm 1.2)$. Density varies from $0.7 \pm 0.3$ (Ibo Sweet 1) to $3.4 \pm 1.9$ (Bambui 1) with a mean of $2.1 \pm 0.7$. This is illustrated in figure 3. The results obtained show that the length of the female flowers is greater than the diameter. In general, the diameter of a flower is proportional to its length. Cultivars with the longest flowers are those with the widest flowers.
### e) Morphology of male flowers
The number of inflorescences (InfN) varies from $3.0 \pm 2.0$ (Lysoka sweet 1) to $30.5 \pm 21.6$ (Mabondji sweet white 1) with a mean of $17.7 \pm 9.9$. The length of the inflorescences (InfLg) varies from $12 \pm 4$ mm (Lysoka sweet 1) to $78 \pm 8$ (Mabondji sweet white 1) mm with a mean of $34 \pm 20$. The number of flowers (FlwN) per inflorescence varies from $20.2 \pm 6.4$ (Lysoka sweet 1) to $158.9 \pm 61.7$ (Mabondji sweet white 1) with a mean of $95.7 \pm 43.3$. The length of the flower (FlwLg) varies from $0.95 \pm 0.12$ (Buea sweet 1) to $1.25 \pm 0.04$ mm (Mabondji sweet white 1) with a mean of $11.0 \pm 1$ mm. The diameter of the flower (FlwD) varies from $1.07 \pm 0.05$ mm (Bayangam 1) to $1.48 \pm 0.06$ mm (Mabondji sweet white 1) with a mean of $1.3 \pm 0.1$. For density, it varies from $20.1 \pm 9.6$ (Lysoka sweet 1) to $49.4 \pm 8.3$ (Bayangam 1) with a mean of $31.5 \pm 11.3$. Density varies from $20.1 \pm 9.60$ (Lysoko Sweet 1) to $49.4 \pm 8.3$ (Bayangam 1) with a mean of $31.1 \pm 11$. Statistical analysis showed that there is no significant difference ( $p = 0.518$ ) between the inflorescence number (InfN) of male and female cultivars. Sex strongly influenced (P < 0.000) InfLg, NumF, FlwLg, FlwD and Density independently of cultivar. This is illustrated in figure 3 where it can be observed that the male inflorescences are 15 times denser as compared to the female inflorescences. The female flower is approximately 10 times longer, 3 times wider and its inflorescences 3 times longer than the male (Figure 3). Indeed, in dioecious clonal plants, the reproductive effort required to set seeds is responsible for the larger investment in sexual reproduction by females (Mizuki and al, 2005).
This result can be explained under the principle of Bateman as mentioned in (Willson, 1994) and (Queller, 1994) suggesting that males invest more in blooming to increase the chances of success of pollination while females invest more in seed production.
The relatively large size of the female flower (106.3 mm) is probably an evolutionary adaptation of this species to produce a high number of seeds for each pollination event. This theory is shared by (Bawa and al, 2019) who suggested that single or few seeded fruits may be correlated with small flowers. In contrast, those with multiple ovaries or many seeded fruits (like $D$. dumetorum) may be associated with large flowers with specialised pollination mechanisms. Furthermore, both combinations of traits, small flowers and one or few seeds per fruit, and large flowers and many seeded fruits, have persisted in monocots longer than other combinations of traits.
### f) Flowering patterns of female and male flowers within the year
Female plants emerge 5 days earlier (DE, $p = 0.027$ ), than male plants. Similar work by (Zoundjiékpon and al, 1997) found instead a 25-day earliness of emergence of male plants over females with cultivars of the Dioscorea cayenensis-rotundata species complex. These authors worked with a sample of 13 plants. Despite the delay in emergence, the flower bud appears 15 days (DFB, $p = 0.017$ ) earlier in male plants. The time from the appearance of the first flower bud to the opening of the first female flower is on average 20 days before for female flowers (DFB-DFFO, $p < 0.0001$ ). There is no effect of sex on DFFO ( $p = 0.356$ ) and NFP ( $p = 0.118$ ) (Table 3). All of this is illustrated on figure 4.
We also note a strong positive correlation (0.74; $p = 2.19 \times 10^{-04}$ ) between flower length and inflorescence length. The longer the inflorescence, the longer the flower and the fewer flowers it will have (-0.75; $p = 1.33 \times 10^{-04}$ ). The longer the inflorescence, the lower the flower density (-0.63; $p = 3.03 \times 10^{-03}$ ). The longer the flower, the lower the flower density (-0.83; $p = 5.61 \times 10^{-06}$ ) (Table 4).
The opening of the female flowers in $D$. dumetorum occurs gradually during maturation. They become receptive after a certain point in the maturation process. Once opened, they do not close again until they turn brown. These flowers do not have a characteristic strong smell. The receptive female flower has prominent, colourful and spotless stigmas, is well open, the stigmas are clearly visible, separate and distinct. It remains in this morphological state for 5 to 7 days after which it turns brown and fades if not pollinated. A weekly count for more than a month of new receptive open female flowers in each variety shows that their number varies according to the variety; the number of open flowers is very irregular over time (Figure 5). The total number of opened flowers per hour between 9 a. m. and 5 p.m. was 4-6 days for male flower. For all accessions considered, maximum opening was observed between 11 a.m. and 2 p.m. Flowers opened in the morning (9 a.m.) and started to close by 5 p.m. in the evening (Figure 6). Zoundjihékpon and al 1997 (up cit) obtained similar result in their work with Dioscorea Cayenensis-rotundata. They reported a cultivar on which flowering last 8 days, with peak of flowering occurring between the fourth and eighth day.
### g) Flowering pattern over the three-year evaluation
Flowering monitoring over three years demonstrates the erratic nature of flowering in $D$. dumetorum. Of the 47 accessions used in this study, 30 are flowering (i.e. $63.83\%$ ), including 9 males ( $19.15\%$ ) and 21 females ( $44.68\%$ ). The remaining 17 ( $36.17\%$ ) accessions did not flower during the three years evaluation. they are said to be non-flowering. Of the $63.83\%$ of accessions that flowered, $19.15\%$ flowered continuously over the three years. The cultivars Muyuka 3, Bayangam 2 and Bamendjou 2 flowered in the first two years and did not flower in the third year. Conversely, the cultivars Country yam 2, Muyuka 3, Bana 1 and Bangang 1 did not flower in the first two years of the evaluation and did flower in the third (Table 5). The flowering rate of cultivars is influenced by the year ( $p = 0.03$ ). An increasing number of cultivars flowered from year to year (Figure 7). A cultivar planted in the second year of trial has 3.67 more chances to flower than one planted a year before (Table 6). It can also be stated, but with little certainty ( $p = 0.09$ ), that sex influences flowering. Its erratic character seems to be more pronounced in male plants than in females. However, this hypothesis remains to be consolidated. Positive Kendall correlation coefficient was noted between year and flowering ( $0.22$; $p = 13 \times 10^{-03}$ ) and between sex and flowering ( $0.18$; $p = 46 \times 10^{-03}$ ) (Table 7). The presence of non-flowering cultivars in the yam collection may limit sexual breeding in them. Similar results showing the absence of flowering in some yam accessions was highlighted by (Yolou and al, 2015). In the collection, the rate of female clones exceeding that of males excludes the problem of scarcity of female clones which is a limiting factor for sexual breeding of yam (Segnou and al, 1992; Asiedu and al, 1992). The temporary flowering discontinuity observed in some accessions in the collection was also highlighted by other workers (Zoundjiékpon, 1993) on the $D$. cayenenses-rotundata complex. This is one of the characteristics of yams that according to Segnou et al (1992) hinders their improvement by sexual means. The presence of some continuous flowering accessions in the collection reduces this problem of lack of flowering continuity over the years. The absence and discontinuity of flowering is thought to be due to the disturbance caused during the domestication process. Indeed, all accessions of $D$. dumetorum would be potentially flowering like other angiosperms. However, the variation in the environment during domestication probably repressed the expression of the genes responsible for flowering. This repression was probably slight in the case of accessions with discontinuous temporary flowering, or accentuated in the case of non-flowering accessions during the three years of the study. This result is in agreement with the ideas of some workers (Asiedu and al, 1992) who believe that the time lag between the flowering of male and female plants limits fertilisation in yam.
### h) Maturation synchrony of male and female flowers
Data of maturation time are illustrated in figure 8. Using a Student's t test, we found that the time to maturity of the male flowers is on average 16 days shorter than that of the female flowers $(p = 0.001)$ over a three-year period. This consistency does not change over the years. For pollination to take place, not only must the flowering period of the male and female flowers be synchronised, but also the opening period. Our results show that the male flowers open 1 week after the female flowers (figure 4). As a consequence, there is no synchronism of either flowering initiation or flower opening in yams. Indeed, this situation, which leads to a much-reduced availability of both viable pollen and receptive female flowers, has been reported in D. rotundata (Zoundjihékpon et al., 1997). Pollination requires not only synchronism in male and female flowering periods but it also requires that flowers open at the same time during the day. Our results showed that female flowers (Figure 5) opened gradually during their maturation and were completely open at full maturity (anthesis). It could not have a flower opening peak hour during the day as reported on other yam species (Zoundjihékpon et al., 1997; (Li and al, 2014). Male flowers started opening during the morning and closed in the evening (Figure 6). Flower opening peaked from the $4^{\text{th}}$ to $6^{\text{th}}$ day between 11 a.m. and 2 p.m. According to (Hamon and Koechlin, 1991), pollination due to insects is higher between 10 a.m. and 12 p.m. Moreover Zoundjihékpon et al. (1997) reported that in yams, the availability of pollen was highest in the morning when the natural pollinators (thrips, Larothrips dentipes) are present and active. Thus, an overlap of flower opening was considerable between male and female accessions.
### i) Pollination method trial
Results are indicated on figure 9. Significant effects were obtained while comparing different pollination technique (p < 0.05) with respect to fructification. No significant difference between the fruiting rates of the positive control (1%) and the negative control (0%) flowers was noted. The best result was obtained with the pollen solution (64%). which is highly superior to wire brush (9%) and the bamboo splinter (9%) methods. This confirms that the fruits formed on the artificially pollinated plants were indeed controlled and not subject to parthenogenesis. The plants in the positive control had a fruiting rate of $1.11\%$ and did not differ significantly from those in the negative control. They provide information on the efficiency of natural pollination, which in this case is low. This failure of natural pollination could be due to the absence of pollinating agents in our experimental site. These are insect pollinators such as Larothrips dentipes, Chirothrips sp., Haplothrips sp. and Xylocopas brasilianorum. Their absence could be due to environmental conditions that do not favour their development. The presence of insecticides sprayed in the experimental field possibly prevented the development and even the presence of these pollinating insects. The superiority of the pollination method involving the pollen solution over the pollination methods with the bamboo splinter and the metal brush could be due to the insufficient dehiscence of the anthers in the latter two methods. The crushing of the anthers during the preparation of the pollen solution must have caused the rupture of the anther and the release of the pollen into the solvent (rainwater). This pollen was certainly deposited directly on the stigma of the female flowers, using the syringe, and must have relatively favoured fertilisation and therefore fruiting. However, pollination with the bamboo splinter and the metal brush allowed the deposition of anthers on the stigma of female flowers; these anthers probably had not undergone sufficient dehiscence after their deposition in order to release the pollen. This lack of dehiscence in these two pollination methods possibly limited fertilisation and therefore fruiting, which was significantly lower than that observed in the case of pollination with the pollen solution. This fruiting rate of $64\%$ is much higher than those obtained in Nigeria on D. rotundata $(41.3\%)$ (Akoroda, 1985); on D. alata $(19.7\%)$ in Cote d'Ivoire (Ehounou and al, 2019) and are comparable to that obtained on D. alata $(74\%)$ (Abraham and al, 1990) in India.
### j) Fructification and germination of D. dumetorum seeds
Artificially pollinated flowers developed into fruits (Figure 10). The germination kinetics of these seeds as a function of time is shown in Figure 11. During germination in $D$ dumetorum, the cotyledons remain buried in the soil, so germination is said to be hypogeal (Figure 12). The germination curve of $D$ dumetorum seeds as a function of time has a sigmoidal shape. $D$ dumetorum seeds start germinating after 23 days; this is the germination delay. After 23 days, the germination rate increases exponentially and most seeds germinate after 40 days. At 45 days after germination, the seeds do not germinate anymore; this is reflected in the horizontal shape of the curve, which marks the end of germination. The fruiting rate of the negative control flowers is almost zero because of the lack of pollination of their flowers before covering them with cloth.
The seeds of $D$. dumetorum germinated because they each contained an embryo resulting from the fusion of the antherozoite and the oosphere. The germination of $D$. dumetorum seeds is hypogea! becaucse this species is a Monocotyledon. Their germination time is longer than that of $D$. cayenenses-rotundata which germinates from day 13 (Zoundjihekpon, 1993). The same is true for the average germination time of $D$. cayenenses-rotundata which is relatively shorter (20 days). These differences in germination time are thought to be due to genetic variability between these two yam species $(D$. cayenenses-rotundata and $D$. dumetorum).
## III. CONCLUSION
This work consisted in evaluating the possibilities of artificial hybridization of sweet yellow yam (D. dumertorum) through controlled pollination in the field. The studies were conducted in West Cameroon, more precisely at the Boukué School Farm in Baham. The effect of temporary flowering discontinuity and the lack of synchronisation in the flowering of male and female flowers on the possibilities of controlled hybridisation of 47 accessions of D. dumetorum was evaluated. On average, female flowers (21 days) bloom quickly compared to male flowers (26 days). In addition, the male flowers bloom 2 weeks before the female flowers. However, the females open 1 week before the males. This shows that there is no synchronisation between the flowering of male and female flowers, however this problem could be solved by staggered sowing. Thus, the slow flowering individuals will be sown before the fast flowering ones by respecting the flowering time lag during sowing. Female flowers of D. dumetorum open gradually during maturation and are fully open at full maturity. Male flowers, in contrast open only when they reach maturity. They open in the morning and close at the end of the day. The opening peaks between 11 am and 2 pm correspond to the favourable pollination period in D. dumetorum. From this study on floral biology it appears that D. dumetorum can be improved by sexual reproduction (Akoroda, 1985). Of the sweet yellow yam pollination techniques studied (bamboo splinter, wire brush and pollen solution injection techniques), pollen solution injection technique performed best with respect to fruiting rate. The fruits formed contained seeds which germinated after 24 days to produce hybrids. For breeding purposes, accessions with continuous temporary flowering may be preferred as progenitors. The pollination method with pollen solution is recommended for controlled crosses. Based on our results, the two main potential problems (temporary flowering discontinuity and the lack of synchronisation) in yam breeding in general can be overcome by sexual reproduction through breeding of D. dumetorum.
Our work contributes to a better control of hybridisation by sexual reproduction in sweet yellow yam. It aims at the genetic improvement of the $D$. dumetorum species through field crosses. For a more efficient and reasonable implementation of these results, it will be necessary to (1)- Develop a method of pollen conservation; (2)- develop an approach to stimulate non-flowering accessions with growth hormones for instance; (3) and characterize hybrids obtained using the best controlled pollination technique with a proper cross-breeding plan in order to enhance the breeding programme of $D$. dumetorum Cameroon.
### ACKNOWLEDGEMENTS
We thank the Appropriate Development for Africa Foundation (ADAF), especially the President, Dr. Paul Kammogne Fokam for financial support.
Table 2: Mann-Whitney statistics table based on Monte Carlo Permutation method and $99\%$ confidence interval indicating the sex influence $(p < 0.001)$ on Inflorescence number (InfNum); Inflorescence length (InfLg); Flower number (FlwNum); Flower length (FlwLg); Flower diameter (FlwD) and Flower density (Density).
<table><tr><td></td><td>InfN</td><td>InfLg</td><td>FlwN</td><td>FlwLg</td><td>FlwD</td><td>Density</td></tr><tr><td>Mann-Whitney U</td><td>44</td><td>5</td><td>0</td><td>0</td><td>2</td><td>0</td></tr><tr><td>Wilcoxon W</td><td>122</td><td>49,5</td><td>78</td><td>45</td><td>47</td><td>78</td></tr><tr><td>Z</td><td>-0.711</td><td>-3.521</td><td>-3.839</td><td>-3.838</td><td>-3.695</td><td>-3.840</td></tr><tr><td>Monte Carlo Sig. (2-tailed)</td><td>0.518</td><td>0.000</td><td>0.000</td><td>0.000</td><td>0.000</td><td>0.000</td></tr></table>
Table 3: Mann-Whitney statistics table based on Monte Carlo Permutation method and
$99\%$ confidence interval indicating the sex influence on DE (Days to Emergence; $\mathsf{p} = 0.027$ ), DFB (Days to Emergence of the First Bud; $\mathsf{p} = 0.017$ ) and DTB-DFFO (Days to first bud emergence to First Flower Opening; $\mathsf{p} < 0.027$ )
<table><tr><td>.</td><td>DE</td><td>DFB</td><td>DFFO</td><td>DFB-DFFO</td><td>NFP</td></tr><tr><td>Mann-Whitney U</td><td>14</td><td>12</td><td>29</td><td>0</td><td>21</td></tr><tr><td>Wilcoxon W</td><td>80</td><td>33</td><td>95</td><td>66</td><td>87</td></tr><tr><td>Z</td><td>-1.94</td><td>-2.11</td><td>-0.40</td><td>-3.33</td><td>-1.24</td></tr><tr><td>Monte Carlo Sig. (1-tailed)</td><td>0.027</td><td>0.017</td><td>0.356</td><td>0.000</td><td>0.118</td></tr></table>
Table 4: Pearson correlation coefficients between morphologic traits using Pearson
<table><tr><td></td><td>InfN</td><td>InfLg</td><td>NumF</td><td>FlwLg</td><td>FlwD</td></tr><tr><td>InfN</td><td>1</td><td></td><td></td><td></td><td></td></tr><tr><td>InfLg</td><td>0.18</td><td>1</td><td></td><td></td><td></td></tr><tr><td></td><td>4.38E-01</td><td></td><td></td><td></td><td></td></tr><tr><td>NumF</td><td>0.33</td><td>-0.41</td><td>1</td><td></td><td></td></tr><tr><td></td><td>1.50E-01</td><td>7.16E-02</td><td></td><td></td><td></td></tr><tr><td>FlwLg</td><td>0.00</td><td>0.74***</td><td>-0.75***</td><td>1</td><td></td></tr><tr><td></td><td>9.97E-01</td><td>2.19E-04</td><td>1.33E-04</td><td></td><td></td></tr><tr><td>FlwD</td><td>-0.05</td><td>0.63**</td><td>-0.71***</td><td>0.95***</td><td>1</td></tr><tr><td></td><td>8.21E-01</td><td>2.81E-03</td><td>5.02E-04</td><td>1.52E-10</td><td></td></tr><tr><td>Density</td><td>0.05</td><td>-0.63***</td><td>0.84***</td><td>-0.83***</td><td>-0.78***</td></tr><tr><td></td><td>8.30E-01</td><td>3.03E-03</td><td>3.53E-06</td><td>5.61E-06</td><td>4.48E-05</td></tr></table>
Table 5: Different cultivars having flowered (F) or not (NF) during the three-years evaluation
<table><tr><td></td><td>Year 1</td><td>Year 2</td><td>Year 3</td></tr><tr><td>Male cultivars</td><td></td><td></td><td></td></tr><tr><td>Muyuka 3</td><td>F</td><td>F</td><td>F</td></tr><tr><td>Mabondjisweet white 1</td><td>F</td><td>F</td><td>F</td></tr><tr><td>Banga bakundu sweet 1</td><td>F</td><td>F</td><td>F</td></tr><tr><td>Ibo sweet 2</td><td>F</td><td>F</td><td>F</td></tr><tr><td>Bayangam 1</td><td>F</td><td>F</td><td>F</td></tr><tr><td>Buea sweet 1</td><td>NF</td><td>F</td><td>F</td></tr><tr><td>Lysoka sweet 1</td><td>NF</td><td>F</td><td>F</td></tr><tr><td>Ibo sweet 3</td><td>NF</td><td>F</td><td>F</td></tr><tr><td>Country yam 2</td><td>NF</td><td>NF</td><td>F</td></tr><tr><td>Female cultivars</td><td></td><td></td><td></td></tr></table>
Table 6: Binomial logistic regression based on Wald method, indicating a significant effect of the year on the flowering pattern
$(p = 0.03)$. A cultivar planted in year 2019 has 3.67 times more chances to flower than one planted in 2018
<table><tr><td>Dschang 1</td><td>NF</td><td>F</td><td>F</td></tr><tr><td>Bamendjou 1</td><td>F</td><td>F</td><td>NF</td></tr><tr><td>Muyuka 3</td><td>NF</td><td>NF</td><td>F</td></tr><tr><td>Bamendjou 2</td><td>F</td><td>F</td><td>NF</td></tr><tr><td>Bangang 2</td><td>F</td><td>F</td><td>F</td></tr><tr><td>Bayangam 2</td><td>F</td><td>F</td><td>NF</td></tr><tr><td>Ibo sweet 1</td><td>F</td><td>F</td><td>F</td></tr><tr><td>Bamokonbou1</td><td>F</td><td>F</td><td>F</td></tr><tr><td>Bangou 1</td><td>F</td><td>F</td><td>F</td></tr><tr><td>Fonkouankem 1</td><td>NF</td><td>F</td><td>F</td></tr><tr><td>Bana1</td><td>NF</td><td>NF</td><td>F</td></tr><tr><td>Guzang 1</td><td>NF</td><td>F</td><td>F</td></tr><tr><td>Mankon 1</td><td>NF</td><td>F</td><td>F</td></tr><tr><td>Bambui 1</td><td>NF</td><td>F</td><td>NF</td></tr><tr><td>Bangang1</td><td>NF</td><td>NF</td><td>F</td></tr><tr><td>Country yam 1</td><td>NF</td><td>F</td><td>F</td></tr><tr><td>Penda-boko 1</td><td>F</td><td>NF</td><td>F</td></tr><tr><td>Bekora 2</td><td>F</td><td>NF</td><td>F</td></tr><tr><td>Baigon 1</td><td>NF</td><td>F</td><td>F</td></tr><tr><td>Fongo-tongo 1</td><td>F</td><td>NF</td><td>NF</td></tr><tr><td>Mabondji sweet yellow 1</td><td>F</td><td>NF</td><td>NF</td></tr></table>
<table><tr><td rowspan="2" colspan="2"></td><td rowspan="2">B</td><td rowspan="2">S.E.</td><td rowspan="2">Wald</td><td rowspan="2">df</td><td rowspan="2">Sig.</td><td rowspan="2">Exp(B)</td><td colspan="2">95,0% C.I.for EXP(B)</td></tr><tr><td>Lower</td><td>Upper</td></tr><tr><td rowspan="5">Step 1a</td><td>Year</td><td></td><td></td><td>5,335</td><td>2</td><td>0,069</td><td></td><td></td><td></td></tr><tr><td>Year(1)</td><td>1.3</td><td>0.60</td><td>4.74</td><td>1</td><td>0.03</td><td>3.67</td><td>1.14</td><td>11.81</td></tr><tr><td>Year(2)</td><td>0.39</td><td>0.62</td><td>0.38</td><td>1</td><td>0.54</td><td>1.47</td><td>0.43</td><td>4.99</td></tr><tr><td>Sex(1)</td><td>-0.99</td><td>0.58</td><td>2.92</td><td>1</td><td>0.09</td><td>0.37</td><td>0.12</td><td>1.16</td></tr><tr><td>Constant</td><td>-1.15</td><td>0.47</td><td>5.9</td><td>1</td><td>0.02</td><td>0.32</td><td></td><td></td></tr></table>
Table 7: Kendall correlation coefficient indicating a strong relation between flowering and sex (p = 0,047) and flowering and year (p = 0,013)
<table><tr><td></td><td></td><td>Flower</td><td>Sex</td><td>Year</td></tr><tr><td rowspan="2">Flower</td><td>Correlation Coefficient</td><td>1</td><td></td><td></td></tr><tr><td>Sig, (1-tailed)</td><td></td><td></td><td></td></tr><tr><td>Sex</td><td>Correlation Coefficient</td><td>0.18</td><td>1</td><td></td></tr><tr><td rowspan="3">Year</td><td>Sig, (1-tailed)</td><td>0.046</td><td></td><td></td></tr><tr><td>Correlation Coefficient</td><td>0.22</td><td>0.000</td><td>1</td></tr><tr><td>Sig, (1-tailed)</td><td>0.013</td><td>0.500</td><td></td></tr></table>


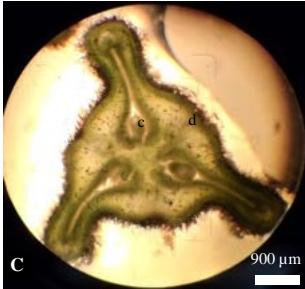
 Figure 1: Floral traits: A: Female inflorescence, B: Opened female flower; C: sectionofthe female flower a: sepal; b: stigma; Cross-section, c: ovule; d: ovary
 Figure 2: Floral trait, A: male inflorescence, B: opened male flower; a: sepal; b: petal; c: stamen
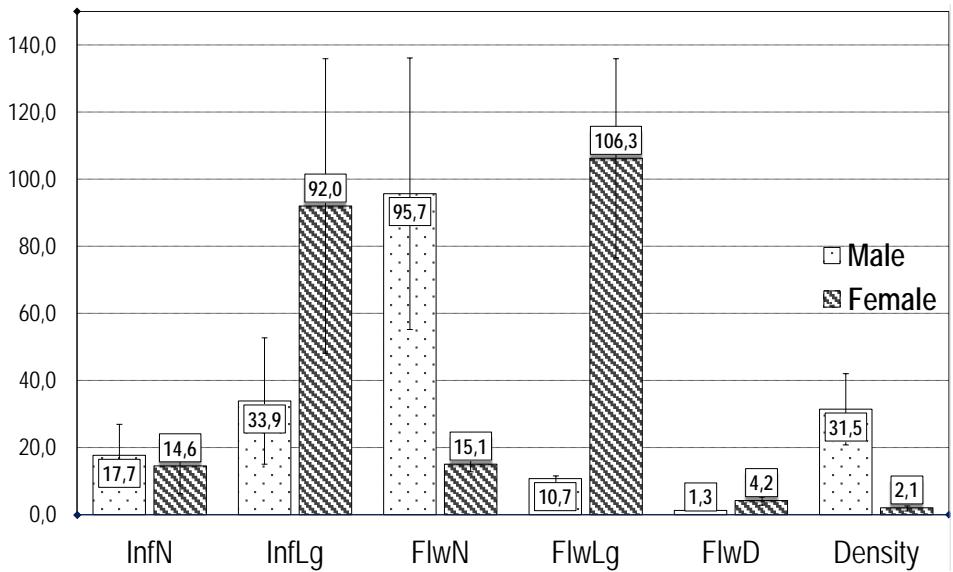 Figure 3: Male and female floral morphologic traits indicating the mean value of inflorescence number (InfN), Inflorescence length (InfLg), Flower number (FlwN), Flower length (Flwlg), Flower diameter (FlwD) and Density. Lengths are in milimeters. Bars are standard deviation. ANOVA indicates significant effect of sex on InLg, FlwN
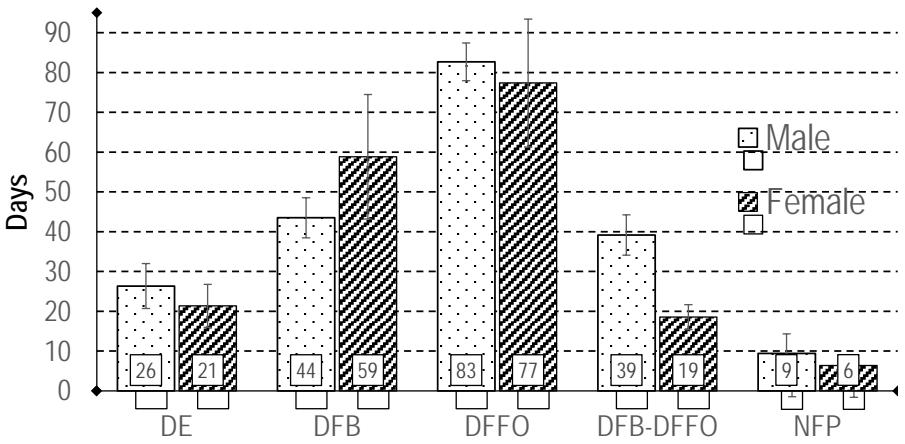
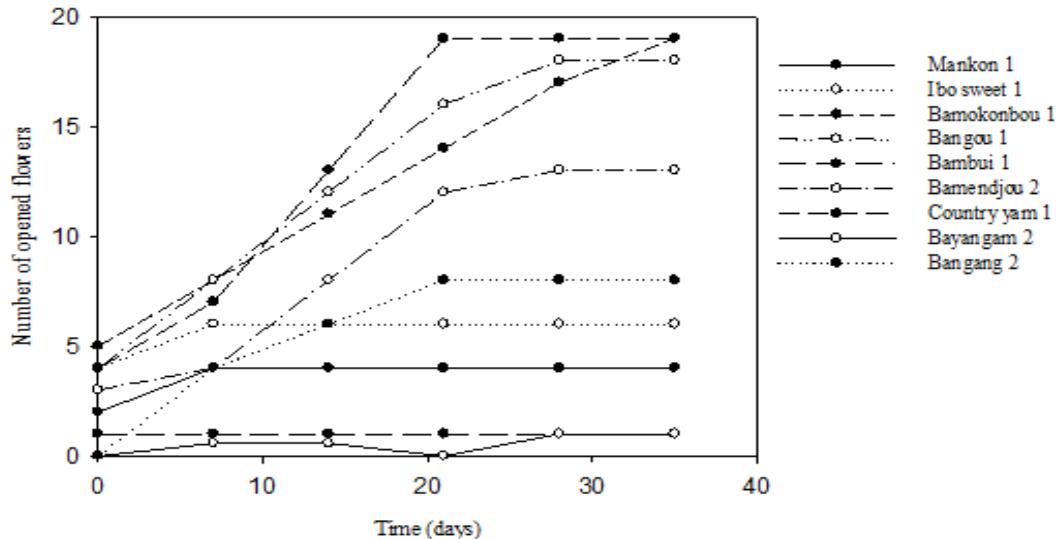 Figure 4: Flowering pattern of female and male flowers: Days to Emergence (DE), Days to first Bud Emergence (DBE), Days to First Flower Opening (DFFO), Days to emergence - First Bud emergence to First Flower Opening (DFB-DFFO) and Number of Flowering Plants (NFP). Bars indicate standard deviations.
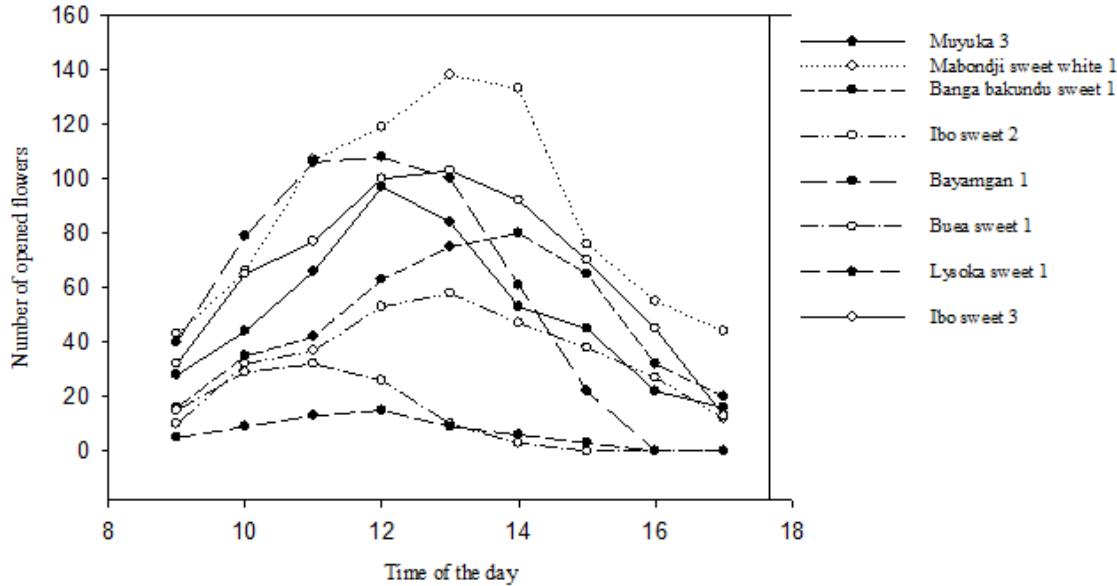 Figure 6: Number of newly opened flowers of male cultivars per hour in one day
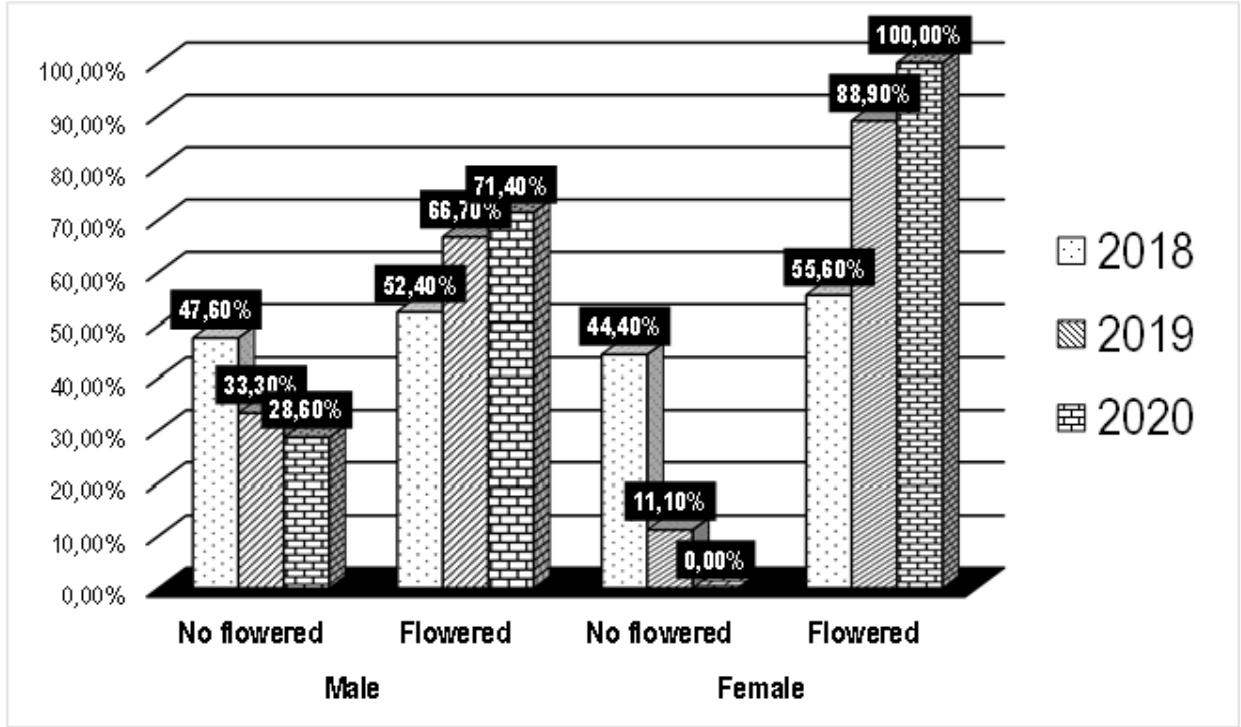 Figure 5: Number of newly opened flowers of female cultivars per week in one month.
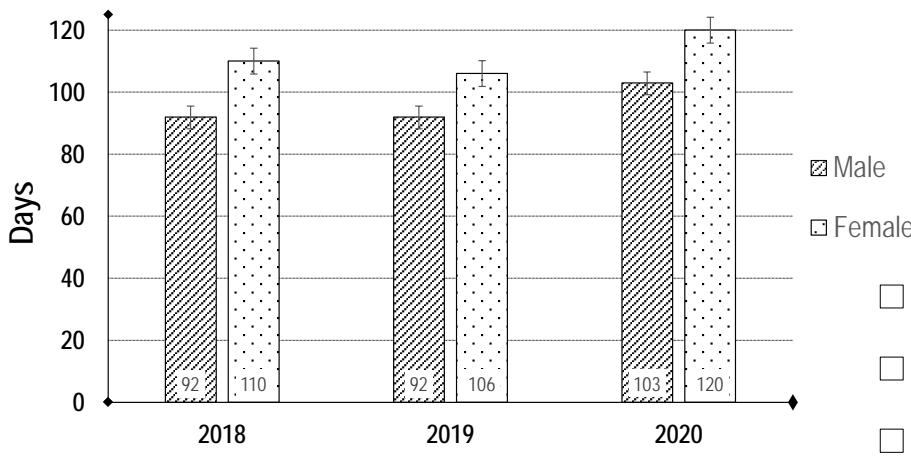 Figure 7: Flowering rate of $D$. dumetorum seed over time
 Figure 9: Illustration of pollination methods. Pollen collected on the anthers (a) and transferred to the stigma (b) during $D$. dumetorum pollination processes. This is done with the pollen solution (f), the bamboo splinter (e) or the metal brush (d). The pollination was followed by covering the pollinated female flowers with a white cloth (g).
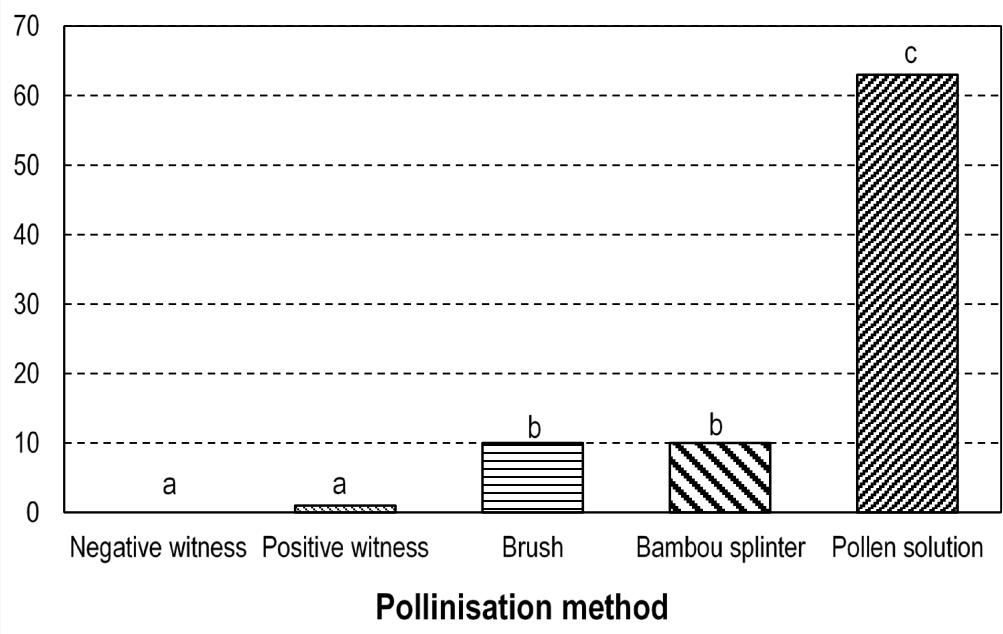 Fructification (%) Figure 10: Fructification rate obtained using three pollination methods
 Figure 8: Maturation time of male and female flowers of $D$. dumetorum during the three years evaluation

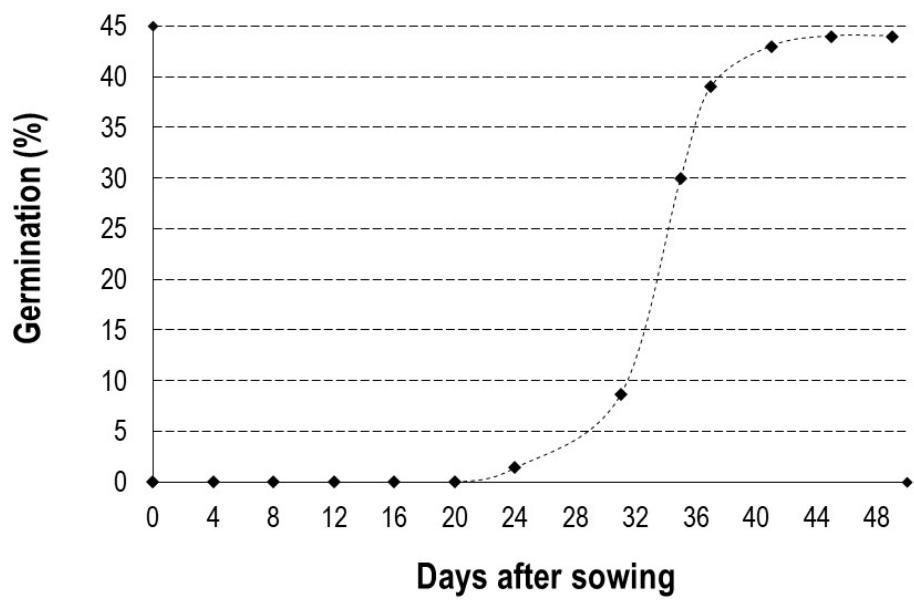 Figure 11: Fruit of D. dumetorum. A, Fruit; B, Seeds. Figure 12: Germination rate curve of $D$. dumetorum seeds versus time
 Figure 13: A- Mature seeds of D. dumetorumand B -hybrid seedlings at three stages of growth
Generating HTML Viewer...
References
40 Cites in Article
L K Abraham,Gopinathan Nair (1990). Floral biology and artificial pollination in Dioscorea alata.
C Ainswortth (2000). Boys and Girls Come Out to Play: The Molecular Biology of Dioecious Plants.
M Akoroda (1983). Floral biology in relation to hand pollination of white yam.
M Akoroda (1985). Pollination management for controlled hybridization of white yam.
R Asiedu,K Bai,R Terauchi,Ago Dixon,S Hahn (1992). Status of wide crosses in cassava and yam.
R Asiedu,N Wanyera,Syc Ng,N Ng (1998). Yams.
Kamaljit Bawa,Tenzing Ingty,Liam Revell,K Shivaprakash (2019). Correlated evolution of flower size and seed number in flowering plants (monocotyledons).
D Coursey (1967). Yams, an account of the nature, origins, cultivation and utilisation of the useful members of the Dioscoreaceae.
L Degras (1993). The Yam, a Tropical Root Crop.
A Ehounou,A Kouakou,N,C Dibi,K B E Bakayoko,Y Essis,B,N 'zue,B Arnau,G Maledon,E Asfaw,A (2019). Production of hybrid seeds by intraspecific crossing in yam Dioscorea alata L.
Fabien Cormier,Guillaume Martin,Hélène Vignes,Laurie Lachman,Denis Cornet,Yoana Faure,Erick Maledon,Pierre Mournet,Gemma Arnau,Hâna Chaïr (2021). Genetic control of flowering in greater yam (Dioscorea alata L.).
Gezahegn Girma,Katie Hyma,Robert Asiedu,Sharon Mitchell,Melaku Gedil,Charles Spillane (2014). Next-generation sequencing based genotyping, cytometry and phenotyping for understanding diversity and evolution of guinea yams.
R Govaerts,P Wilkin,R Saunders,K (2007). World Checklist of Dioscoreales, Yams and their Allies.
Serge Hamon,Jean Koechlin (1991). The reproductive biology of okra. 2. Self-fertilization kinetics in the cultivated okra (Abelmoschus esculentus), and consequences for breeding.
M-A Hartmann (2009). The way the dioecious plant Actinidia deliciosa attracts bees: critical role of volatile terpenes released from kiwifruit flowers of both genotypes.
V Lebot (2009). Aroids: developmental physiology..
M Li,Q Yan,X Sun,Y Zhao,Y Zhou,Y Hang (2014). A preliminary study on pollination biology of three species in Dioscorea (Dioscoreaceae).
Mahbou Somo Toukam,G Siadjeu,C Bell,J M Nkwate,S Bomda,J (2015). Influence de quelques caractères agronomiques sur le rendement de l'igname sucrée (Dioscorea dumetorum Kunth Pax) au Cameroun.
Mahbou Somo,Toukam Gabriel,Ntsomboh-Ntsefong Godswill,Tiokeng,Marie Noel Ateko,Nono Benoit,Youmbi Emmanuel (2021). Cultivar and Soil Fertilizer TreatmentAffect Seed Production of Sweet Yellow Yam (Dioscorea dumetorum) on Highly Acidic Soilsof the Western.
Mbome Lape,I (1991). Utilisation de l'igname Dioscorea dumetorum comme base des bouillies de sevrage Brazzaville, Congo.
Medoua Nama,G (2005). Cadre multipartite pour intervenir dans les systèmes semenciers de racines, de tubercules et de bananes - cadre multipartite en faveur des interventions dans les systèmes semenciers des racines, tubercules et bananes.
Medoua Nama,G Mbome Lape,I Agbor-Egbe,T Mbofung,Cmf (2008). Influence of fermentation on some quality characteristics of trifoliate yam (Dioscorea dumetorum) hardened tubers.
Inoue Mizuki,Kiyoshi Ishida,Kihachiro Kikuzawa (2005). Sexual and vegetative reproduction in the aboveground part of a dioecious clonal plant, <i>Dioscorea japonica</i> (Dioscoreaceae).
Jean Mondo,Paterne Agre,Alex Edemodu,Patrick Adebola,Robert Asiedu,Malachy Akoroda,Asrat Asfaw (2020). Floral Biology and Pollination Efficiency in Yam (Dioscorea spp.).
(2018). MORPHOLOGICAL AND BIOCHEMICAL EVALUATION OF SOME EGYPTIAN WHEAT GENOTYPES UNDER SALINITY STRESS CONDITIONS.
Prince Emmanuel Norman,Asrat Asfaw,Pangirayi Bernard Tongoona,Agyema Danqua,Eric Danquah,David Koeyer,Robert Asiedu (2018). Pollination Success in Some White Yam Genotypes Under Polycross and Nested Mating Designs.
David Queller (1994). Male-Female Conflict and Parent-Offspring Conflict.
S Sefa-Dedeh,E Afoakwa (2002). Biochemical and textural changes in trifoliate yam Dioscorea dumetorum tubers after harvest.
Segnou,C Fatokun,M Akoroda,S Hahn (1992). Studies on the reproductive biology of white yam (Dioscorea rotundata Poir.).
C Siadjeu,Mahbou Somo Toukam,G Bell,J M Nkwate,S (2015). Genetic diversity of sweet am "Dioscorea dumetorum "(Kunth) Pax revealed by morphological traits in two agro-ecological zones of Cameroon.
Christian Siadjeu,Eike Mayland-Quellhorst,Dirk Albach (2018). Genetic diversity and population structure of trifoliate yam (Dioscorea dumetorum Kunth) in Cameroon revealed by genotyping-by-sequencing (GBS).
C Siadjeu,E Mayland-Quellhorst,S Pande,S Laubinge,D Albach (2021). Plants.
Christian Siadjeu,Emmanuel Akdowa Panyoo,Gabriel Mahbou Somo Toukam,Joseph Bell,Benoit Nono,Gabriel Medoua (2016). Influence of Cultivar on the Postharvest Hardening of Trifoliate Yam (<i>Dioscorea dumetorum</i>) Tubers.
S Trèche (1998). Valeur nutritionnelle de l'igname.
S Trèche,Mbome Lape,I Agbor-Egbe,T (1984). Variation de la valeur nutritionnelle au cours de la préparation de produits séchés à partir d'ignames cultivées au Cameroun (Dioscorea dumetorum et D. rotundata).
Mary Willson (1994). Sexual Selection in Plants: Perspective and Overview.
Zhenghao Xu,Le Chang (2017). Dioscoreaceae.
M Yolou,I Anizehou,R Dossou-Yovo,A Akoegninnou,J Zongo,J Zoundjihekpon (2015). Etat des lieux de la reproduction sexuée des ignames africaines <i>Dioscorea cayenensis</i> – <i>Dioscorea rotundata</i> cultivées au Bénin.
A Yaogo,A Bangoua,D Aboubacar,M Diallo,M Sarifou,Y Oumarou,Kco Kouassi,A Diallo,R Ponan,R Gnininfoun,A Bouaffo,S Adjeka,D Doffou,H Anzouan-Kacou,C Assi,A Attia (1993). Prevalence of peptic ulcers in cirrhotics at the Cocody and Angré University Hospitals in Côte d'Ivoire.
J Zoundjihekpon,P Hamon,M Noirot,B Tio-Toure,S Hamon (1997). Flowering synchronisation between male and female West African cultivated yams (Dioscorea cayenensis-rotundata complex).
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Gabriel Mahbou Somo Toukam. 2026. \u201cFloral biology, Field Controlled Pollination and Hybrid Obtention of Dioscorea Dumetorum (Kunth) Pax) in Cameroon\u201d. Global Journal of Science Frontier Research - C: Biological Science GJSFR-C Volume 22 (GJSFR Volume 22 Issue C1).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.