The pathogenesis of human disease is commonly considered to be unique to each organ or system. This has led to specialization in medical practice leaving little space for global pathogenesis concepts. In this review we have departed from a biochemical concept of coenzyme Q 10 (CoQ 10 ) deficiency and its relation to the initiation of hypoxia response in a systemic way. Several conditions of secondary CoQ 10 deficiency are known in the literature by which the organs involved could be affected by hypoxia and switch to glycolytic metabolism. The most salient biomarkers of this situation are the low T3 syndrome and the elevation of IL-6. These parameters together with CoQ 10 deficiency delineate a condition of acquired mitochondrial dysfunction. Additional related biochemical deficiency conditions affect magnesium, selenium, and iron levels. Visualization of glycolysis can be clearly achieved by diagnostic imaging methods based on the use of 18 F-fluoro-deoxyglucose ( 18 F-FDG). We present several examples of diagnostic imaging with 18 F-FDG to demonstrate our model of acquired mitochondrial dysfunction and disease.
## I. INTRODUCTION
Through a series of observational studies on the pathogenesis of benign thyroid disease we were able to deduce in 2021 that the central pathogenetic mechanism was a condition of glycolytic metabolism as evidenced through combined diagnostic imaging with 3D-power Doppler sonography and positron emission tomography (PET) using $^{18}\mathrm{F}$ -fluorodeoxyglucose ( $^{18}\mathrm{F}$ -FDG) (Figure 6 in (1)). In the $^{18}\mathrm{F}$ -FDG study both the thyroid and the heart displayed an intense tracer uptake (Figure 1), an association never commented before in the literature (2). Since the innate capability of desoxyglucose is to indicate glycolysis (3), we interpreted the image as an in-vivo depiction of this metabolic process which correlated with increased vascularity. Given that patients with benign thyroid disease frequently have low levels of coenzyme $\mathsf{Q}_{10}$ ( $\mathrm{CoQ}_{10}$ ) and that experimental $\mathrm{CoQ}_{10}$ deficiency is linked to hypoxia as shown by Liparulo et al., in 2021 (4), our current interpretation of the diagnostic image shown below is that it represents glycolysis and is related to conditions of hypoxia. In Liparulo's experiment with the human T67 glioma cell line, $\mathrm{CoQ}_{10}$ deficiency was produced by inhibiting its biosynthesis using the competitive inhibitor 4-nitrobenzoate leading finally to low intracellular levels of oxygen, i.e., hypoxia (4).
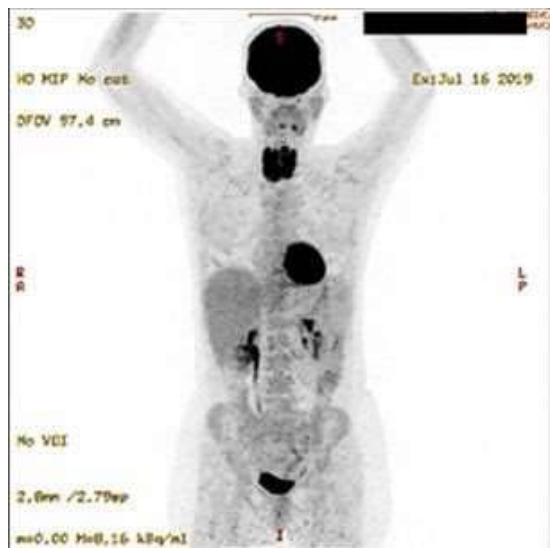
Figure 1: $^{18}\mathrm{F}$ -FDG PET Study of a Patient with Recurrent Active Thyroiditis. Intense Tracer Uptake is Seen in the Thyroid and the Heart. The Brain Shows a Physiological Uptake. The renal pelvis, the ureters, and the urinary bladder depict the Physiological Excretion Path. Panel B Shows the Simple Pathogenetic Mechanism Leading to Hypoxia.
Our previous experience with $\mathrm{CoQ}_{10}$ deficiency had shown that this condition is reversible when $\mathrm{CoQ}_{10}$ supplementation at a dose of $0.9\mathrm{mg / kg}$ body weight combined with magnesium citrate $2400\mathrm{mg / d}$, and selenium $200\mu \mathrm{g / d}$ was given. This reversibility can be demonstrated by the restitution of normal perfusion and morphology of the thyroid (5). The clinical investigation of our patients was based on a conceptual model of coordinated interactions between magnesium, selenium, iron, and $\mathrm{CoQ}_{10}$ as they relate to mitochondrial function (Figure 2). The clinical impact of these processes has been discussed elsewhere (1, 5-11).
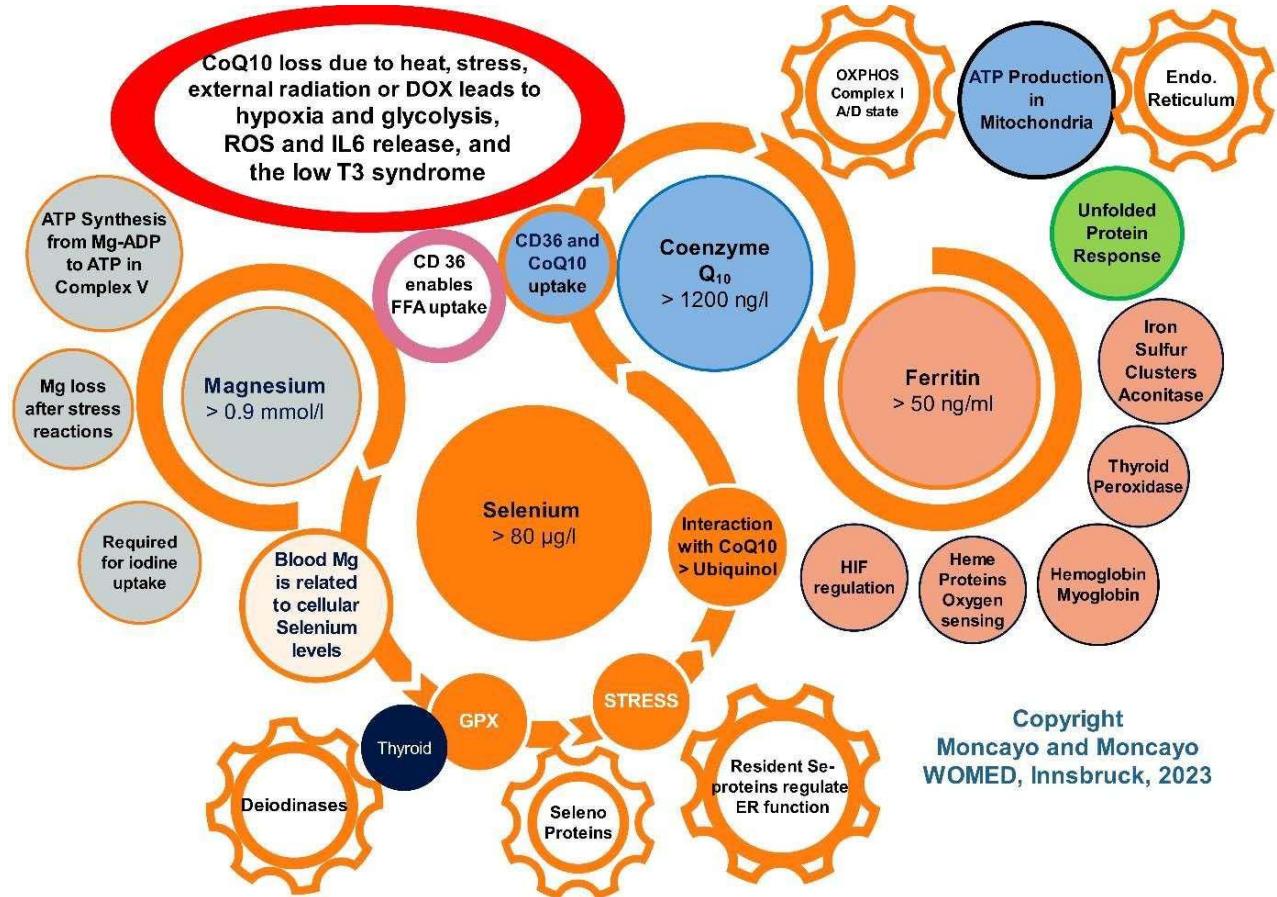 Figure 2: The WOMED model of the interactions between magnesium, selenium,
$\mathrm{CoQ}_{10}$, and ferritin in relation to mitochondrial function. Magnesium is required ATP synthesis, for iodine uptake, and selenium uptake. Selenium is the basic element of selenoproteins. Selenoproteins are related to the thyroid hormone deiodinases, to anti-oxidant functions, and to the endoplasmic reticulum as a resident element. Selenium is a key factor for maintaining $\mathrm{CoQ}_{10}$ levels. $\mathrm{CoQ}_{10}$ is the central element in Complex I of the OXPHOS chain and is maintained by CD36. Anti-oxidative actions of $\mathrm{CoQ}_{10}$ arise from the conversion of $\mathrm{CoQ}_{10}$ to ubiquinol, a process that requires selenium. Iron is essential for the synthesis of iron sulfur clusters which are constituents of many proteins of the OXPHOS chain. Furthermore, iron stabilizes the prolyl hydroxylases inhibiting the activation of HIF1. Desirable blood levels of the parameters are shown in the circles. The red circle signalizes the crucial situation of $\mathrm{CoQ}_{10}$ in the context of hypoxia.
The main messages from Figure 2 include: 1) daily life and physical stressors can lead to magnesium deficiency (12, 13) which will affect iodine and selenium uptake and thyroid hormone formation (14-16). 2) Selenium deficiency (7) will affect the anti-oxidative protection of the body by altering selenoprotein synthesis (17, 18) and altered activation of $\mathrm{CoQ}_{10}$ to ubiquinol through thioredoxin reductase (19), potentiating oxidative damage. One fact that is often overlooked is that $\mathrm{CoQ}_{10}$ has a positive correlation to selenium levels (20). 3) A decrease of magnesium availability will affect primarily the production of ATP from Mg-ADP (21) as well as the uptake of selenium. 4)
The lack of ATP will alter the function of the unfolded protein response of the endoplasmic reticulum (22). Iron deficiency affects thyroid peroxidase (23), mitochondrial iron sulfur clusters (24, 25), and regulation of HIF through prolyl hydroxylases.
To demonstrate the concept of $\mathrm{CoQ}_{10}$ deficiency we will first review basic concepts on the substance and add an introduction on diagnostic imaging procedures based primarily on the use of $^{18}\mathrm{F}$ -FDG.
Nuclear Medicine diagnostic imaging based on the use of $^{18}\mathrm{F}$ -FDG PET has been primarily considered as a diagnostic method for oncological work (26). This rather restrictive notion emerged during the early years of clinical use of $^{18}\mathrm{F}$ -FDG when tracer availability and imaging capacity at specialized institutions were limited (26). Disregarding these conceptual and practical limitations, in 2003 we applied the methodology for the diagnosis of arteritis (27). Visualization of fatty acid oxidation can be achieved using the specific tracer $\left[{ }^{123} \mathrm{I}\right]$ - $\beta$ -Methyl iodophenyl-pentadecanoic acid (BMIPP) (28,
29). The beta oxidation of free fatty acids constitutes the primary source of energy and ATP production in the heart (30). Once the oxidation of free fatty acids is diminished, glycolysis takes over in the failing heart (31). This is accompanied by increased glycolytic metabolism. Figure 3 shows summarily mechanisms of $\mathrm{CoQ}_{10}$ deficiency and their relation to metabolism and Nuclear Medicine diagnostic imaging.
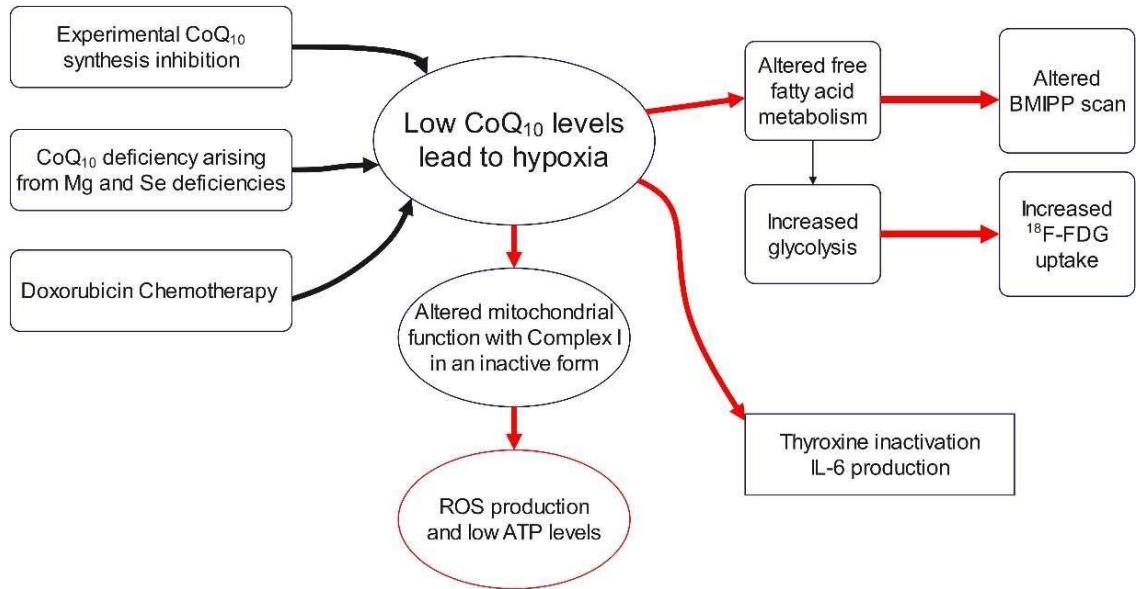 Figure 3: General changes of mitochondrial metabolism and systemic parameters under hypoxia.
### a) Basic notes on coenzyme $Q_{10}$
The first description of coenzyme $Q_{10}$ ( $CoQ_{10}$ ) was done by Festenstein in 1955 who was working on the general topic of quinones (32). $CoQ_{10}$ is a small lipophilic molecule located widely in cell membranes. Due to its distribution, it was named ubiquitous quinone. Beginning in 1957 Crane and coworkers published a series of studies on the characterization of $CoQ_{10}$. An early publication dealt with the isolation of a quinone from the mitochondria of beef heart. The authors described the capability of the compound to undergo oxidation and reduction in a reversible way. The denomination Q-275 for $CoQ_{10}$ was derived from the absorption spectrum of the substance (33). Different compounds having quinone structure have been described in different species (34). In 1959 $CoQ_{10}$ and the succinoxidase activity of the electron transport system were studied by Crane (35) as well as by Hendlin and Cook in 1960 (36). The structure of $CoQ_{10}$ characterized as a 2,3-dimethoxy benzoquinone structure was published by Wolf et al., in 1958 (37).
The biochemical characteristics of quinones were summarized by Hoffmann-Ostenhof in 1947 (38). In 1948 Friedmann, Marrian, and Simonreuss described the antimitotic effects of quinones in relation to their structure (39). Further information on the biochemistry of quinones was published by Barnes in 1963 (Section 3 in (40)).
Gale et al., evaluated the mean content of $\mathrm{CoQ}_{10}$ in different human organs in 1961 (41) showing that the brain and thyroid have the lowest concentrations (Figure 2).
Due to frequent incidence of thyroid disease in the general population, we hypothesize that organs with higher levels of $\mathrm{CoQ}_{10}$ can be more resistant to alteration and retain function. The thyroid would be at the end of the scale.
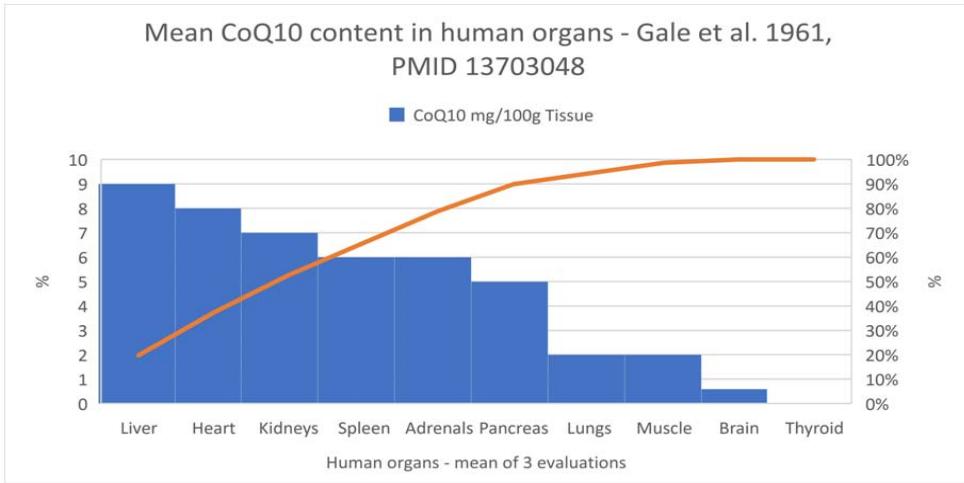 Figure 4: Mean
$\mathrm{CoQ}_{10}$ tissue levels in selected organs. Adapted from Gale et al. (41)
The relations between the organ content of $\mathrm{CoQ}_{10}$ or ubiquinone and age have been studied by Kalén, Appelkvist, and Dallner in 1989 (42). An abridged summary of their data is shown in Table 1. The highest levels are found in the age group 19-21 years while older subjects have lower levels.
Table 1: CoQ<sub>10</sub> levels in selected organs according to age (extracted from (42)).
<table><tr><td rowspan="2"></td><td colspan="2">Ubiquinone Age</td><td colspan="3">μg/d ww</td></tr><tr><td>1-3 days</td><td>0.7 - 2 years</td><td>19 - 21 years</td><td>39 - 43 years</td><td>77 - 81 years</td></tr><tr><td>Heart</td><td>36.7</td><td>78.5</td><td>110.0</td><td>75.0</td><td>47.2</td></tr><tr><td>Kidney</td><td>17.4</td><td>53.4</td><td>98.0</td><td>71.1</td><td>64.0</td></tr><tr><td>Liver</td><td>12.9</td><td>45.1</td><td>61.2</td><td>58.3</td><td>50.8</td></tr></table>
In 1996 Ravaglia et al., evaluated the correlation of $\mathrm{CoQ}_{10}$ levels with body composition and found that levels decreased in a parallel form as did the fat-free body mass and speculated that this diminution would affect muscle performance (43).
Basic information on the pharmacokinetics of $\mathrm{CoQ}_{10}$ can be found in the study by Tomono et al., who used a deuterium labeled molecule (44). A first peak in blood was seen at 6 hours, and the half-life was found to be 33 hours. Data on PET tracer biodistribution studies using $^{11}\mathrm{C}$ -labeled $\mathrm{CoQ}_{10}$ and $^{11}\mathrm{C}$ -ubiquinol were reported by Watanabe et al., in 2019 (45). We have interpolated and summarized their results in Table 2.
Table 2: Biodistribution of ${}^{11}\mathrm{C}$ -labeled ${\mathrm{{CoQ}}}_{10}$ and ubiquinol (45).
<table><tr><td colspan="2">CoQ10distribution</td><td colspan="3">% ID/g</td></tr><tr><td></td><td>Ubiquinol2 0min</td><td>90min</td><td>Ubiquinone2 0min</td><td>90min</td></tr><tr><td>Heart</td><td>0.5</td><td>0.6</td><td>1.4</td><td>0.7</td></tr><tr><td>Kidney</td><td>0.4</td><td>0.28</td><td>0.3</td><td>0.2</td></tr><tr><td>Liver</td><td>3.8</td><td>5.7</td><td>3.7</td><td>5.8</td></tr><tr><td>Spleen</td><td>2.0</td><td>2.2</td><td>3.5</td><td>6.0</td></tr><tr><td>Blood</td><td>1.9</td><td>0.8</td><td>1.4</td><td>0.6</td></tr><tr><td>Cerebrum</td><td>0.038</td><td></td><td>0.026</td><td></td></tr><tr><td>Muscle</td><td>0.04</td><td></td><td>0.03</td><td></td></tr></table>
Bentinger et al. published a study on the content of $\mathrm{CoQ}_{10}$ in endocrine organs (46). Table 1 of this publication shows the high uptake of dietary $\mathrm{CoQ}_{10}$
in steroid producing cells such as adrenal cells and ovaries while the thyroid had a much lower uptake.
### b) Oxidative processes, selenium, and CoQ<sub>10</sub>
Several investigators have evaluated the effect of external radiation and oxygen toxicity on biological systems since the 1940s. The biological extent of life has been proposed to be associated with concepts of cellular damage due to oxidative processes that produced reaction intermediates originating from unpaired electrons (47). In 1954 Gerschman et al., studied the topic of oxygen poisoning as well as x-irradiation proposing a common mechanism which was related to the oxidizing free radicals (48). In 1955 Harman published a theory of aging by describing deleterious effects arising from injuries due to free radicals (49). These investigations as well as those of Ritossa on the changes caused by heat shock (50), had in common an external component which was acting as a pathogenic factor affecting living cells ending with oxidative changes.
Early investigations on selenium addressed its role in liver disease. In 1957 Schwarz and Foltz (51) described the beneficial effect of selenium in the context of dietary necrotic liver degeneration. They identified selenium in a fraction originally called Factor 3. Modern research has described the complex array of selenoproteins with antioxidant functions including glutathione peroxidase (52-54).
We have chosen to consider selenium and $\mathrm{CoQ}_{10}$ together based on observations made by Green et al., in 1961 (55) and by Hidiroglou in 1967 (56) who found a close functional relation between both substances such that selenium administration improved the tissue levels of $\mathrm{CoQ}_{10}$ in experimental animals. Vadhanavikit and Ganther published similar observations in 1993 and 1994 (57, 58). The authors speculated that lowering of $\mathrm{CoQ}_{10}$ levels depended on the depressed activity of GSH-Px which was the consequence of selenium deficiency (58). This finding implies that hypoxia can develop starting from selenium deficiency which then affects $\mathrm{CoQ}_{10}$ levels. Unfortunately, this important physiological connection has not been considered in many studies. Putting the focus only on one of these elements in the context of supplementation have remained inconclusive (59-61). On the other hand, specific single mechanisms can be indeed characterized.
Ernster and Forsmark-Andree described the anti-oxidant actions of ubiquinol, i.e. the reduced form of $\mathrm{CoQ}_{10}$, in aerobic organisms (62). Sugiyama studied the anti- oxidative effect of $\mathrm{CoQ}_{10}$ administering a dose of $2\mathrm{mg / k}$ which is equivalent to $\sim 140$ mg for an adult with $70\mathrm{k}$ body weight. Both the in-vivo experiments as well as the in-vitro studies demonstrated that under $\mathrm{CoQ}_{10}$ lipo-peroxides were significantly lower as compared to the controls (Table 1 in (63)). Degli Esposti et al. characterized the functional role of $\mathrm{CoQ}_{10}$ in Complex I of the OXPHOS chain emphasizing its energetic function (64). In 2017 Shimizu et al. described an association between low $\mathrm{CoQ}_{10}$ levels and hospital mortality in patients with cardiac disease (65). Heart arrest patients also share this feature of having very low levels of $\mathrm{CoQ}_{10}$ (66).
Under experimental conditions the administration of selenium compounds has resulted in inhibition of angiogenesis as well as of VEGF expression (67, 68). In our studies we have observed a similar action for $\mathrm{CoQ}_{10}$ (5). We propose that these actions add a positive effect to counteract changes due to hypoxia.
### c) Warburg and the development of 2-deoxy-D-glucose
The theory behind the development of 2-desoxy-D-glucose can be traced back indirectly to the work of Otto Warburg. In 1911 he carried out experiments with the sea urchin (Arbacia pustulosa) and with nucleated avian erythrocytes (69). The blood cells had been exposed to low temperatures between $-15^{\circ}$ and $-20^{\circ}$ C to freeze them. The frozen material was then exposed to $38^{\circ}$ C to break the cells. This handling produced a temperature increase of approximately $60^{\circ}$ C. The resultant experimental acellular material was a dark liquid. Oxygen consumption in the material won from younger blood cells was higher than in intact red blood cells. A closer analysis of the 1923 publication by Warburg and Minami clearly showed the influence of raised temperature (70). These experiments were unknowingly ahead of time to those on the heat shock response done in 1962 by Ritossa where heat shock was produced by setting the temperature to $30^{\circ}$ C for thirty minutes (50). Warburg's experiments have never been interpreted in the context of the heat shock (71-74) nor of endoplasmic reticulum stress (75). Endoplasmic reticulum stress is a logical consequence since heat shock will produce protein aggregation (76). Experimental heat shock can induce glycolysis to compensate ATP balance (77) and activate the endoplasmic reticulum stress response mechanisms (78).
In 1928 Warburg published "The metabolism of tumors in the body" (79) where he described the metabolic characteristics of 2 tumor models, Flexner-Jobling's rat carcinoma (80), and Jensen's rat sarcoma (81). In their experiment they measured the glucose content in in-going and out-going tumor vessels. The average values in the experiments with the Jensen sarcoma substrate revealed a mean glucose concentration of $124\mathrm{mg}\%$ in the arterial vessels, and 54 mg% in the venous vessel (Table II in (79)). On the other hand lactic acid in the tumor cavity was higher than in the aorta by $69\mathrm{mg}\%$ (Table V in (79)).
In 1922 Bergmann, Schotte, and Lechinsky published their work on the development of deoxyglucose as an antagonist of glycolysis and added experimental data aimed at elucidating the reactivity of glucosides after introducing structural changes at the
C2 position of the glucose molecule (82). Further experimental work on deoxy- glucose as an antagonist of glycolysis was published by Francis B. Cramer and Gladys Woodward in 1952 (3, 83-85). They showed a reduction of approximately $60\%$ of glucose metabolism under anaerobic conditions working with slices of Walker 256 rat carcinoma samples when an equimolar quantity of 2-deoxy-D-glucose was added. The Walker 256 rat carcinoma originated in 1928 from a mammary gland tumor of a pregnant rat (page 261 in (86)). Confirmation of the avidity of Walker-256 tumor cells for ${}^{18}\mathrm{F}$ FDG was provided in 2019 by Li et al. (Figure 2 in (87)).
Woodward and Cramer (3) included only two literature references in their publication: their previous work and a 1930 translation of Warburg's previous work (88). The word "relation" was used to connect the content of both publications.
In 1957 Wick et al. described the primary metabolic block on glucose that 2-deoxyglucose produces (89). In 1961 Barban described this mechanism as follows: "... inhibits glycolysis and growth of cultured human cells" (page 1890 in (90)). These results clearly associated 2-deoxyglucose with glycolysis. With the development of $^{18}\mathrm{F}$ -labelled desoxyglucose by Som in 1980, a diagnostic tool for glycolysis imaging became available (91).
### d) Narrative description of hypoxia and cardiac metabolism
Hypoxia stands for a biochemical state where oxygen availability is limited. According to Richalet in "The Invention of Hypoxia", the term appeared in the literature about 1909 in a publication originating from Germany (92). At this time Joseph Barcroft published two studies dealing with the consequences of insufficient oxygen supply to the body. In 1908 he presented a review of the scientific evidence known up to that time (93). In 1920 he discussed the consequences of insufficient oxygen supply as follows: "The statement has been made that "anoxaemia not only stops the machine, but wrecks the machinery" (94). In modern times we can interpret this statement as suggesting multi-organ failure (95).
In 1964 Nunn and Freeman discussed that tissue hypoxia can arise from hemorrhagic shock 1964 (96). In 1969 Henderson (97) and McDowall (98) presented summaries of the biochemical concepts of hypoxia which appeared in the British Journal of Anesthesiology. In 1977 Alberti discussed the consequences of hypoxia on biochemical terms. Equation 11 of this publication identified the role of $\mathrm{CoQ}_{10}$ in the synthesis of ATP (99).
In 1987 Bihari discussed the relation of hypoxia to septic shock (100). Some biochemical characteristics of hypoxia were described by Webster in 1987 who reported the relation between hypoxia and activation of glycolysis (101).
In 1988 Goldberg, Dunning, and Bunn characterized the oxygen sensing function as being associated with heme protein (102). In 1992 Loike et al. showed that when endothelial cells were exposed to hypoxia an upregulation of glucose transport could be observed (103). At a cellular level, the metabolic consequences of hypoxia include diminution of fatty acid oxidation and activation of glycolysis (Figure 1 in (104)) as well as an increased expression of interleukin 6 (105).
The transcriptional adaptation to experimental hypoxia was characterized in 1993 by Wang and Semenza defining a role for the hypoxia inducible factor 1 (HIF-1 $\alpha$ ) using hepatoma tumor cells (Hep3B) (106). In 1999 Ouddir et al. demonstrated that the expression of the glucose transporter in alveolar epithelial cells could be upregulated by hypoxia. In their experiments this was reflected by increased uptake of deoxyglucose (Figure 2 in (107)) which is connected to increased glycolysis.
In 2010 Solaini et al. described major changes in mitochondria found in hypoxia. Figure 1 depicted a connection between hypoxia and diminished electron transport and increased ROS production. The authors conceived these changes as stabilizing factors of HIF (108). Lee, Chandelier, and Simon discussed mechanisms of cellular changes following hypoxia adding the involvement of endoplasmic reticulum stress and of the unfolded protein response (Figure 2 in (109)).
Recent reviews on hypoxia research have been published by Thompson in 2016 (110) as well as by Luo et al. in 2022. Luo's publication included an informative pictorial summary of relevant landmarks (111). Kierans and Taylor idealized that increased glycolysis was the event that goes directly from hypoxia to HIF-1 $\alpha$ expression ending with the expression of enzymes involved in glycolysis (112). Della Rocca and coworkers summarized clinical data on diseases related to hypoxia in 2022 (113).
The beta oxidation of free fatty acids constitutes the primary source of energy and ATP production in the heart (30). Tateno and coworkers have demonstrated that myocardial infarction patients show a diminished utilization of free fatty acids which did not correlate with the findings of myocardial perfusion (114). Changes in cardiac fatty acid utilization show a dynamic course as reported by Zen et al., (115). Biswas et al., demonstrated lower BMIPP uptake following acute infarction of the myocardium (116) as well as a negative correlation between left ventricular ejection fraction and tracer wash out rate. They also described a positive correlation between washout rate of BMIPP and blood level of NT-proBNP (Figures 4 and 5, respectively in (117)).
The graphical appearance of decreased BMIPP uptake as seen in planar images was shown in Figure 6 by Biswas. Increasing levels of NT-proBNP, a biomarker of heart failure (118), correlated positively with an increased wash-out rate of BMIPP (Figure 5 in (119)) as well as a higher incidence of cardiac events when cardiac tracer uptake was low (Figure 6 in (119)).
### e) Clinical situations associated with CoQ $_{10}$ deficiency
Low levels of $\mathrm{CoQ}_{10}$ can be found in a series of situations. A clinical review by Boreková found that situations of poor nutrition, infection, and a variety of stressors can diminish the availability of $\mathrm{CoQ}_{10}$ (120). The publication included data from a previous study by Anna Gvozdjáková which had shown such conditions.
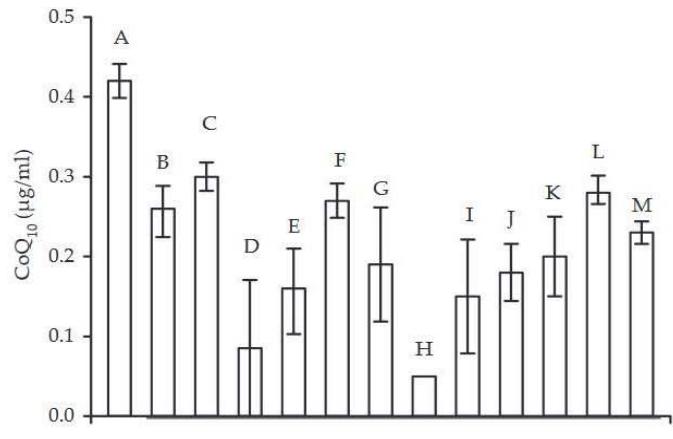
Figure 4. Coenzyme $Q_{10}$ in human blood (A: healthy people, B-E: stressed people, F-M: patients with some diseases) (by GVOZDIKAOVÁ et al. 2000)
Reprinted with the permission of the authors 2007
- A - healthy people
- B - stressed people (managers)
- C - 2 Slovak astronauts made ready for start to Cosmos
- D-30 Slovak fighter pilots
- E - people working with ionising radiation
- F - patients with insulin dependent diabetes mellitus
- G - patients with non insulin dependent diabetes mellitus patients
- H - patients with nephropathy
- I - patients with myopathy
- J - patients with osteoporosis
- K - patients with chronic heart disease
- L - patients with cardiomyopathy of unknown ethiology
- M - patients after heart transplantation
Low levels of $\mathrm{CoQ}_{10}$ have been found in children who presented food allergies (121). The same applied to acute influenza (122). A similar situation can be found in patients after cardiac arrest as well as septic patients (66, 123) and in critical illness (124). Studies on postoperative follow-up have shown indirectly that surgery induces a decrease of $\mathrm{CoQ}_{10}$ levels. This drop can be prevented by supplementation with $\mathrm{CoQ}_{10}$ (125).
Experimental studies done by Fischer et al., have shown alterations in $\mathrm{CoQ}_{10}$ levels after dietary restriction (126). Experimental burns can lead to mitochondrial damage and dysfunction and can be corrected by $\mathrm{CoQ}_{10}$ administration (127). Similar observations have been published by Xu et al., relating $\mathrm{CoQ}_{10}$ administration to protection against myocardial cell changes induced by heat stress (128, 129).
Experimental hypoxia in skeletal muscle of rabbits is accompanied by a reduction of the levels of ubiquinone (130). A decrease of the $\mathrm{CoQ}_{10}$ levels goes along with low ATP levels in isolated rabbit ventricles under hypoxia (Table 2 in (131)).
The administration of antineoplastic antibiotics such as adriamycin can inhibit the synthesis of $\mathrm{CoQ}_{10}$ (132). This interference could arise from the similar quinone structure of both $\mathrm{CoQ}_{10}$ and adriamycin (133-138). A further effect of this inhibition is the decrease of free fatty acid oxidation. In 2000 and 2003, Saito et al. demonstrated that cardiac BMIPP uptake was decreased after doxorubicin (139) and taxan (140) administration. On the other hand, it has been shown that doxorubicin cardiotoxicity can be prevented by administration of $\mathrm{CoQ}_{10}$ (141). Bradamante et al. considered hypoxia to be a risk factor for doxorubicin-induced cardiotoxicity (142). Muhammed, Ramasarma, and Kurup described in 1983 that adriamycin inhibited oxidative phosphorylation in freshly prepared mitochondria (135).
In 2013 Umezawa et al., demonstrated that external radiation therapy affected BMIPP uptake. The irradiated area of the heart (red arrows in Figure 4) showed no tracer uptake (143). Although the $\mathrm{CoQ}_{10}$ levels were not evaluated, the image pattern is highly suggestive of induced $\mathrm{CoQ}_{10}$ deficiency. In a subsequent publication the authors described a relation between the radiation dose applied and the degree of BMIPP alteration (144).
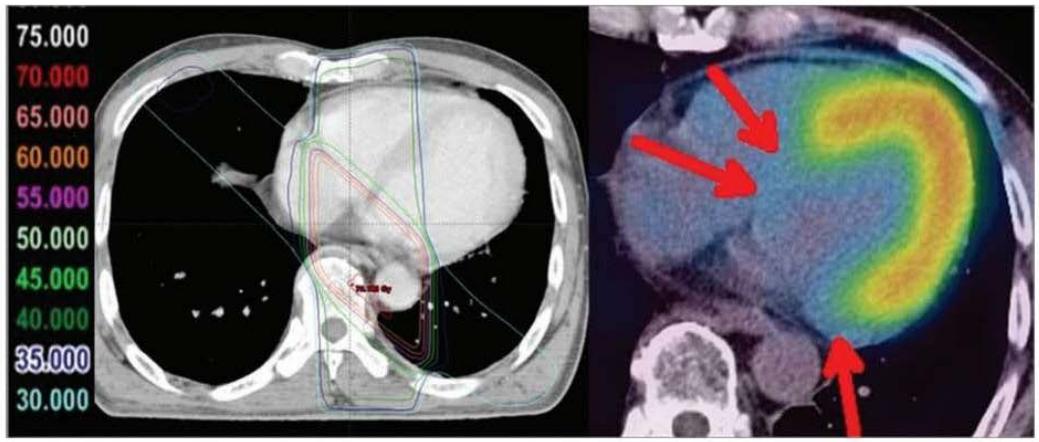
Fig. 3. Images of a remarkable case (62-year-old male) at 29 months after radiotherapy for esophageal cancer. Reduced uptake corresponding to radiation fields was distinct in this case.
Following the concept of metabolic switch from free fatty oxidation to glycolysis, Yan et al., have demonstrated that radiation-induced changes in myocardial metabolism are accompanied by a high uptake of $^{18}\mathrm{F}$ -FDG (Figures 2 and 3 in (145)), thus reflecting glycolysis.
A series of studies have demonstrated that altered free fatty acid oxidation can be restituted by administrating the ACE-inhibitor enalapril (146-150) (Figure 5).
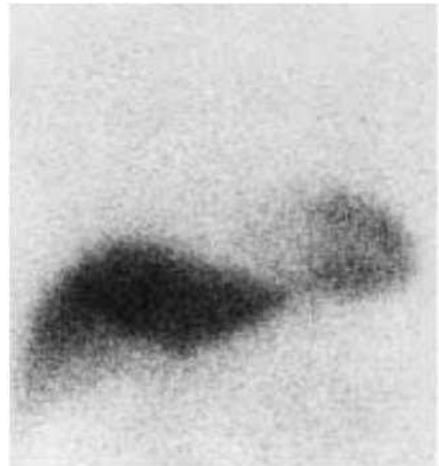
Fig. 2. Scintigraphic findings. Significantly more cardiac uptake of $^{123}\mathrm{I}$ BMIPP (H/M) was observed 3 months after treatment (right) than before treatment (left). H/M=1.99
### f) Clinical situations associated with diminished BMIPP uptake
The uptake mechanism of free fatty acids requires a transport system that involves the CD36 molecule. When there is a deficiency of this structure, $^{18}\mathrm{F}$ -FDG uptake has been shown to be increased. This alteration can be corrected by administrating carvedilol (151) (Figure 6).
Figure 4: Taken from Umezawa (143). The Figure is used on the basis of Creative Common CC-BY-NC. Umezawa R, Takase K, Jingu K, Takanami K, Ota H, Kaneta T, et al. Evaluation of Radiation-Induced Myocardial Damage Using Iodine-123 B-Methyl-Iodophenyl Pentadecanoic Acid Scintigraphy. J Radiat Res (2013) 54(5): 880-9. Epub 2013/02/16. doi: https://doi.org/10.1093/jrr/rrt011
Fig 3. FDG-PET in the fasting state before and after 3 months of carvedilol therapy. (a) FDG uptake was enhanced throughout the left ventricle and (b) alleviated by carvedilol therapy. Figure 6: Taken from Hirooka et al. (151). Hirooka K, Yasumura Y, Ishida Y, Komamura K, Hanatani A, Nakatani S, et al. Improvement in Cardiac Function and Free Fatty Acid Metabolism in a Case of Dilated Cardiomyopathy with Cd36 Deficiency. Jpn Circ J (2000) 64(9): 731-5. doi: https://doi.org/10.1253/jcj.64.731. License granted
The complementary situation, i.e., diminished free fatty oxidation, can be found in patients with CD36 deficiency. Nishimura demonstrated that subjects with CD36 deficiency and cardiomyopathy had low BMIPP uptake. This defect could be corrected by the administration of $\mathrm{CoQ}_{10}$ using a dose of $30\mathrm{mg/d}$ for 35 days (152) (Figure 7).
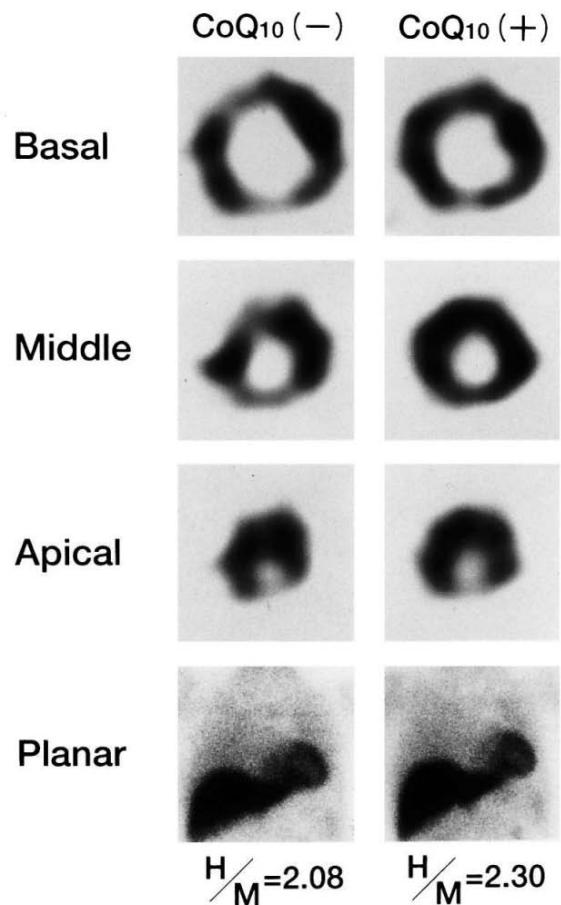
Fig. 2. $^{123}\mathrm{I}$ BMIPP myocardial SPET images before and after Q10 treatment in a representative patient (case 3) with idiopathic dilated cardiomyopathy Figure 7: Taken from Kim et al.(152). Therapeutic Effect of Co-Enzyme Q10 on Idiopathic Dilated Cardiomyopathy: Assessment by Iodine-123 Labelled 15-(P-Iodophenyl)-3(R,S)- Methylpentadecanoic Acid Myocardial Single-Photon Emission Tomography. European journal of nuclear medicine (1997) 24(6): 629-34. Epub 1997/06/01. License CCBY. https://doi.org/10.1007/bf00841400.
The images presented above demonstrate the ability of Nuclear Medicine to reveal the metabolic situation of the heart. Besides the therapeutic use of $\mathrm{CoQ}_{10}$, enalapril administration has also led to normalization of BMIPP defects. This supports the clinical value of enalapril in the treatment of patients with heart failure with reduced cardiac function (153). The data suggest that there is a restitution of normal oxidative phosphorylation (154-156).
Alba Timón-Gómez, and Antoni Barrientos have summarized data on changes of the functional status of mitochondrial Complex I from active to dormant under hypoxia (157). In addition to this the microRNA molecule miR-210 has been found to be involved in these changes.
Inhibition of NDUFS, NDUFA4, and NDUFA4L2 affect also Complex I and Complex IV (Figure 1 in (157)). NDUFS is the mitochondrial NADH-ubiquinone oxidoreductase 75 kDa subunit and NDUFA corresponds to NADH:ubiquinone oxidoreductase subunit A. Alterations of these molecules affect the function of Complex I.
The scientific literature contains scarce information about $\mathrm{CoQ}_{10}$ and hypoxia in human disease. Hypoxemia is a recognized problem associated with congenital heart disease (158), and with viral infections (Figure 2 in (159), Figure 2 in (160)). The role of hypoxia in chronic kidney disease has been discussed by Wang et al., (161). Chen et al., depicted the progression of kidney disease linking hypoxia and fibrosis (Figure 4 in (162)). Hypoxia in the context of COVID-19 has been recently summarized by Østergaard in 2022 setting a focus on capillary changes (163).
The condition of chronic hypoxemia is associated with diminished cardiac function and low levels of ATP (164). Morita et al. described the beneficial effect of $\mathrm{CoQ}_{10}$ in an experimental model of hypoxemia-reoxygenation injury done with heart tissue homogenates (164).
Recent studies on the rare disease Moyamoya have revealed some single possible similarities to the changes we have found in patients with thyroid disease. These findings include excessive collateral vessels and elevated VEGF levels (165), expression of HIF-1 and TGF-beta in the endothelium (166) as well as specific vascular changes (167). In 2022 Ma et al., described that the blood levels of $\mathrm{CoQ}_{10}$ in this rare disease were significantly lower as compared to controls (168).
The selective evolution of mitochondrial diversity has been described as a central element for cell typespecific expression (Figure 1 in (169)). A recent statistical meta- analysis has added information on the polymorphisms of the hypoxia-inducible factor 1-alpha gene finding specific associations with different entities such as cardiovascular disease, skin disease, COPD, and complications of diabetes (170). Following stabilization of HIF there is a downstream signaling activation in several organs (171). This finding tells us that hypoxia has different phenotypes.
### g) Clinical conditions and biochemical features associated with hypoxia
Figure 8 shows our theoretical abstraction of biochemical processes arising from hypoxia where we place $\mathrm{CoQ}_{10}$ deficiency as a key central element. In the following sections we will describe the biochemical changes associated with hypoxia such as the low T3 syndrome and elevation of interleukin-6 (IL-6) levels.
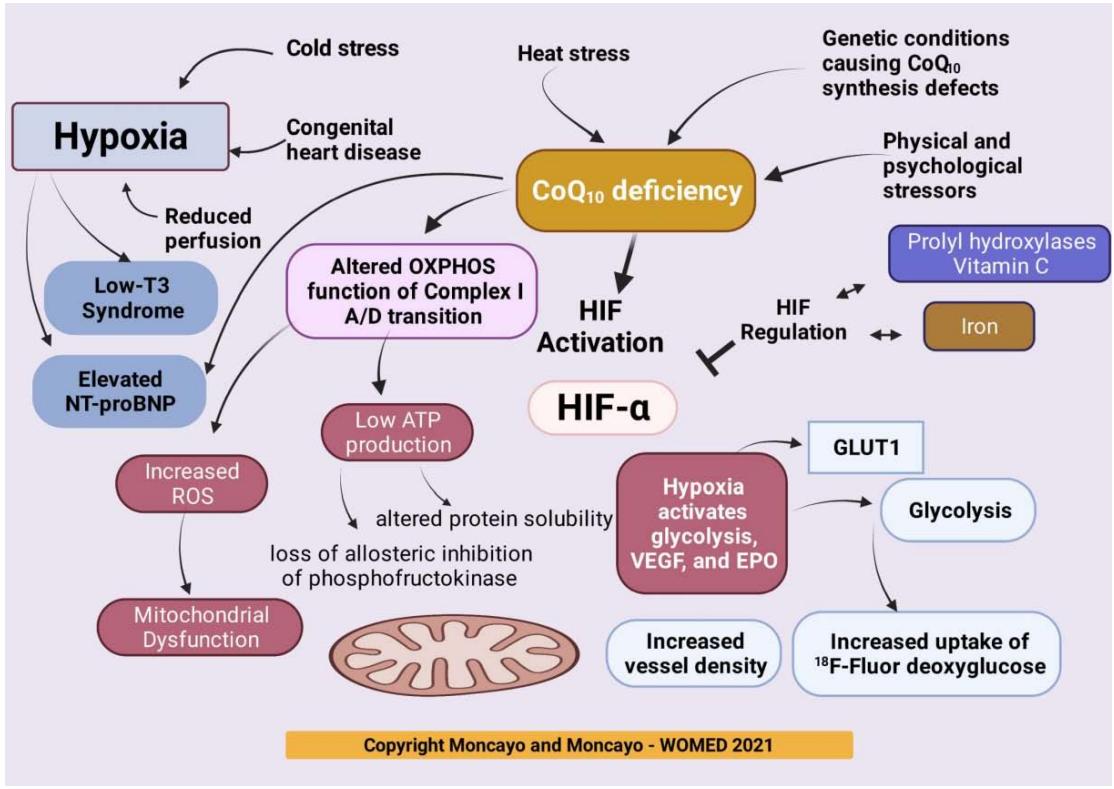 Figure 8: Biochemical pathways involved in hypoxia. Key elements include the initiation of glycolysis arising from diminished levels of
$\mathrm{CoQ}_{10}$ together increased ROS production, low-T3 syndrome, and diminished ATP production. Glycolysis can be detected by Nuclear Medicine diagnostic imaging using $^{18}\mathrm{F}$ -Fluor deoxyglucose. An equivalent image of glycolysis in the thyroid can be achieved by 3D power Doppler sonography. This method shows a condition of increased vascularity.
h) Hypoxia and the low T3 syndrome - a causal path?
The low T3 syndrome or euthyroid sick syndrome has been described by several authors as being associated with many different conditions. In 1974 decreased levels of total triiodothyronine were observed in patients with chronic liver disease, chronic obstructive lung disease, chronic renal failure, and intensive care situations. The authors attributed these changes to lower production of TRH following stress or undernutrition (172). A review on the euthyroid sick syndrome by Wartofsky and Burman published 1982 mentioned possible endocrine mechanisms behind this condition. In Table 2 they listed fasting, anorexia nervosa, and protein-calorie malnutrition as conditions that presented the low T3 situation. Many other factors related to the low T3 syndrome can be found in this review. One interesting postulate was that this condition is related to energy conservation by reducing catabolism (173).
In 1983 Vitek, Shatney, Lang, and Cowley studied the time relation between induction of hemorrhagic shock in dogs and changes in the level of thyroid hormones. They described lower levels of T3 and T4 after 20 minutes of shock. A similar situation was described in 3 patients with trauma or shock. The authors interpreted the changes as belonging to the euthyroid sick syndrome (174). In the late 1990s, several investigators had observed changes of thyroid function parameters in relation to severe illness (172), systemic illness (173), and aging (175). This last study by Schroffner considered the low T3 syndrome to be quite frequent in different clinical conditions (175). The mechanisms leading to this condition were described as reduced activation of T4 (176), and as increased degradation of thyroid hormone in septic shock patients (177). In 1980, Moshang et al. reported that hypoxia affected thyroid function parameters (178). Also in 1980, Becker et al., described low levels of triiodothyronine in patients who had suffered thermal injuries (179). In 1990 Sawhney, and Malhotra had demonstrated that hypoxia was related to low levels of thyroid hormones (Figure 1 in (180)).
In 2001 Ross and Petros reviewed the euthyroid sick syndrome in pediatric cardiac patients including those treated at the intensive care unit (181). The authors discussed three potential settings in this situation: adaptive reduction of the metabolic rate, contributor to the disease, of simply due to the severity of illness (181). Table 2 included potential triggers, however hypoxia was not included.
In 2003 Iervasi et al. found that the low T3 syndrome had a prognostic relevance predicting fatal outcome in cardiac patients. The authors evaluated the data from 1058 patients seen at a National Council
Research Institute of Clinical Physiology in Pisa in 1999 (182). The authors expressed doubt as to whether changed thyroid metabolism had contributed to impairment of heart function.
In 2007 Adler and Wartofsky reviewed the euthyroid sick syndrome and considered it to represent an adaptation to several clinical conditions (183).
An important mechanistic finding relating thyroid hormone metabolism to hypoxia was described in 2008 by Simonides et al.. In their study HIF interacted with thyroid hormone deiodinases leading to the low T3 syndrome (184). Their study was based on a previous observation that transforming growth factor beta, a target for HIF, could promote thyroid hormone inactivation (185). This process resulted in lower levels of T3, which are the key feature of the low-T3 syndrome. Considering that deiodinases are selenoproteins (186, 187) and that our model of acquired mitochondrial dysfunction includes selenium deficiency (10, 11), we must add that selenium deficiency must be quite severe to affect the deiodinase system (188). The study by Fraczek-Jucha et al., demonstrated that selenium deficiency does not correlate with the low T3 condition. Their study found a significant association between low T3 and elevated NT-proBNP levels (Table 2 in (189)). Elevated levels of NT-proBNP correlate with diminished dysfunction of the left ventricle in cardiac disease patients (190).
The low T3 syndrome has been found to show an inverse correlation with coronary artery disease (191) as well as with elevated blood levels of NT-proBNP (192, 193).
Bolen et al., described an association of low T3 syndrome with elevated levels of IL-6 (194). In a clinical study with patients that had presented an acute coronary syndrome, Brozaitiene et al., found an association between mortality and low T3 levels as well as with elevated values of NT-proBNP (195). Minai analyzed the data from 625 heart failure patients in 2021 and found that low T3 and NT-proBNP showed an inverse correlation (196).
Many publications hypothesize that changes leading to the low T3 syndrome arise from the thyroid (Figure 1 in (197)) or from the heart in patients with heart disease (Figure 5 in (198)). The incidence of the low T3 syndrome in patients with congestive heart failure has been called "unexpectedly high" (199).
Biegelmeyer et al., reported that low T3 levels are prevalent among non-critically ill patients and that they are predictive of 30-day hospital mortality (200). Cerillo added an observation as to the relation between low T3 and decreased cardiac output in patients scheduled for coronary bypass surgery (201). Abdu et al. reported a higher incidence of cardiac events in patients with MINOCA (Figure 1 in) (202).
A review published by Langouche, Jacobs, and Van den Berghe in 2019 described the appearance of the nonthyroidal illness syndrome across all ages. The authors added a comment as to the still unknown causality of the disease (203).
### i) Sepsis and critical illness
Several Investigations on the metabolism of sepsis by L'Her, and Sebert have described an environment of altered mitochondrial function where oxygen utilization and Complex I function are affected (204, 205) stating: "Energy- metabolism disturbances during sepsis are characterized by enhanced glycolytic fluxes and reduced mitochondrial respiration. However, it is not known whether these abnormalities are the result of a specific mitochondrial alteration, decreased pyruvate dehydrogenase (PDH) complex activity, depletion of ubiquinone $\left(\mathrm{CoQ}_{10}$; electron donor for the mitochondrial complex III), or all $3^\prime\prime$.",
Low levels of $\mathrm{CoQ}_{10}$ have been described in patients with sepsis (123). He et al., conducted a study on pediatric sepsis evaluating levels of $\mathrm{CoQ}_{10}$ as well as the activity of Complex I and III. They found that low levels of these parameters were associated with mortality in children affected by sepsis (206).
Vassiliou et al., reported that septic patients in a critical condition had low levels of $\mathrm{CoQ}_{10}$ (207). Coppadoro et al., described that low levels of $\mathrm{CoQ}_{10}$ are found in a broad range of critically ill patients (124).
Abitagaoglu et al., reported carried out an experimental study on the effect of $\mathrm{CoQ}_{10}$ administration in sepsis. They found that multi-organ involvement of the heart, kidney, and spleen, was decreased under this treatment (208). Soltani et al., carried out a clinical study on the effect of $\mathrm{CoQ}_{10}$ in sepsis and found a positive effect on clinical parameters and mitochondrial function (209). In 2001 Fink advanced the notion of cytopathic hypoxia as a mechanism of disease (210). In 2002 Brealey et al., discussed the association between outcome of septic shock and mitochondrial dysfunction (211). Cornu et al., evaluated the prognostic value of low T3 syndrome in patients with septic shock and found an association with high mortality (212).
### j) Energetics and physical exercise: nutritional theories vs. $^{18}F$ -FDG imaging
Several publications dealing with nutritional theories and energetics have a common element, i.e., the so-called reference man. The original publication on the reference man from 1974 (ICRP 23) corresponds to a description of the chemical composition of the body, the mass of the organs, as well as to data on physiology (213). This publication originated from the need to respond to concerns about the medical effects of ionizing radiation. The International Commission on Radiological Protection was founded in 1928 at the second International Congress of Radiology (ICRP). It is obvious, that energy intake analysis was not the primary worry of the ICRP. Section B of the publication deals with Energy Expenditure stating that food and oxygen are used according to energy requirements. Oxygen was described together with the process of combustion. Section B7 described energy requirement arising during pregnancy and lactation showing that these conditions require a higher level of energy. The ICRP 23 publication was followed by ICRP 89 in 2002 which was again dedicated to workers dealing with radiation protection (214).
The concept of energetics in the field of ecology does not include biochemical parameters in relation to function (Figure 1 in (215)). In the field of life history research, the economics model discussed by Pontzer and McGrosky mentions metabolic energy as being involved, however the publication contains no evidence as to how energy can be generated in the mitochondria (216). One important aspect included in Life History Theory is that of trade-off between survival and reproduction (217) as well as resource allocation (218). An example of this philosophy can be taken from Jasienka, who described suppressed ovarian function in relation to physical work (219). This concept is closer to general physiology since it considers at least 2 bodily functions while conventional medical physiology picks out single players, e.g., the heart, the thyroid, or human reproduction. It should be noted that classical research on resilience also lacks the inclusion of basic biochemical processes (220, 221).
Energetics changes occurring during lactation have been described since decades. Bauman and Currie discussed a process of nutrient partitioning in 1980 (222). Mowry et al. described metabolic changes aimed at increasing mitochondrial efficiency in relation to reproduction (223). Energetics and fatigue in sports have been described as being related to brain and body functions (224). The theory launched by St. Clair Gibson, Swart, and Tucker has little connection to biochemistry. Their model idealizes a competitive homeostasis system that involves psychological and physiological elements without mentioning mitochondrial function.
Lavin et al., discussed the spectrum of adaptations to exercise at a molecular level putting the item physiological resilience on top (Figure 3 in (225)). Their definition or resilience states "resilience (i.e., ability to tolerate and recover from stressors)" (Page 11 in (225)).
In a previous publication we launched a concept of shared resources as being the central event related to health and disease. When the level of resources is sufficient, survival is assured resulting in resilience. Unbalanced levels of resources are connected to disease (Figure 12 in (1)). Elements of organ function as well as the so-called psychosomatic components of life can be traced down to specific biochemical parameters where magnesium deficiency plays a central role (9).
Mitochondrial function and the generation of ATP via oxidative phosphorylation constitute a central biochemical element in physiology. This function was summarily described by Brand, Orr, Perevoshchikova, and Quinlan in 2013 (226) stating that ATP generation results from the energy release after nutrients have been oxidated (Box 1).
The classic role of mitochondria is oxidative phosphorylation, which generates ATP by utilizing the energy released during the oxidation of the food we eat. ATP is used in turn as the primary energy source for most biochemical and physiological processes, such as growth, movement and homeostasis.
#### Box 1: Role of oxidative phosphorylation in relation to food. Adapted from (226)
The topic of mitochondrial adaptation to nutrient availability was addressed by Liesa and Shirihai in 2013 (227). In 2022 Kyriazis et al., described processes of mitochondrial adaptation to changes in diet (Figure 5 in (228)). The simplistic idea that dietary evaluations and simple calorie counting reflect the level of energetics in the body contradict this physiological principle. Anorexia nervosa represents a condition of severe nutritional alteration. It has been included in the list of diseases associated with low-T3 syndrome (173). Niklowitz et al., described signs of $\mathrm{CoQ}_{10}$ depletion in patients with anorexia nervosa (229).
A similar situation can be found in the setting of physical exercise. Loucks and Callister published a study that combined energy cost and exercise intensity in 1993 (230). They showed that the low T3 condition occurred in amenorrhoeic women under energy limitation. They concluded that exercise could have compromised energy availability. Low energy availability is a term used frequently in relation with physical exercise. Slater, Brown, McLay-Cooke, and Black evaluated this aspect in exercising subjects (231). Loucks discussed the role of energy availability in relation to reproductive disruption in 2003 (232).
In the field of sports physiology, the term "Relative Energy Deficiency in Sport (RED- S)" is being used currently. A recent review on this topic published by Cabre et al. in 2022 insisted on the concept of energy availability in terms of kcal/kg (Figure 1 in (233)). The publication does not include any biochemical data as to how the body can use this apparent energy supply (Table 2 in (233)). This simplified model contrasts sharply with the biochemical adaptation of mitochondrial respiration responding to energetic needs and nutrient availability as described by Bennett, Latorre-Muro, and Puigserver (Figure 2 in (234)). We have taken the following statement from Box 3 to reinforce our hypothesis:
Mitochondrial respiratory chain activity is required for normal function in tissues with high energy demand such as brain, heart and skeletal muscle. Diseases caused by underlying mitochondrial dysfunction often affect these organ systems.
Following our line of thought on hypoxia we would like to add data related to exercise. Exercising subjects can develop hypoxemia (235, 236) and physical activity can lead to HIF activation in skeletal muscle (237). Experimental hypoxia in skeletal muscle of rabbits is accompanied by a reduction of the levels of ubiquinone (130). A decrease of the $\mathrm{CoQ}_{10}$ levels is seen together with diminished ATP levels in isolated rabbit ventricles under hypoxia (Table 2 in (131)). Metabolic changes related to exercise can be seen with diagnostic $^{18}$ F-FDG imaging. Nakase et al., demonstrated focal uptake in football players (238) (Figure 9).
Whole Body Muscle Activity during the FIFA 11+
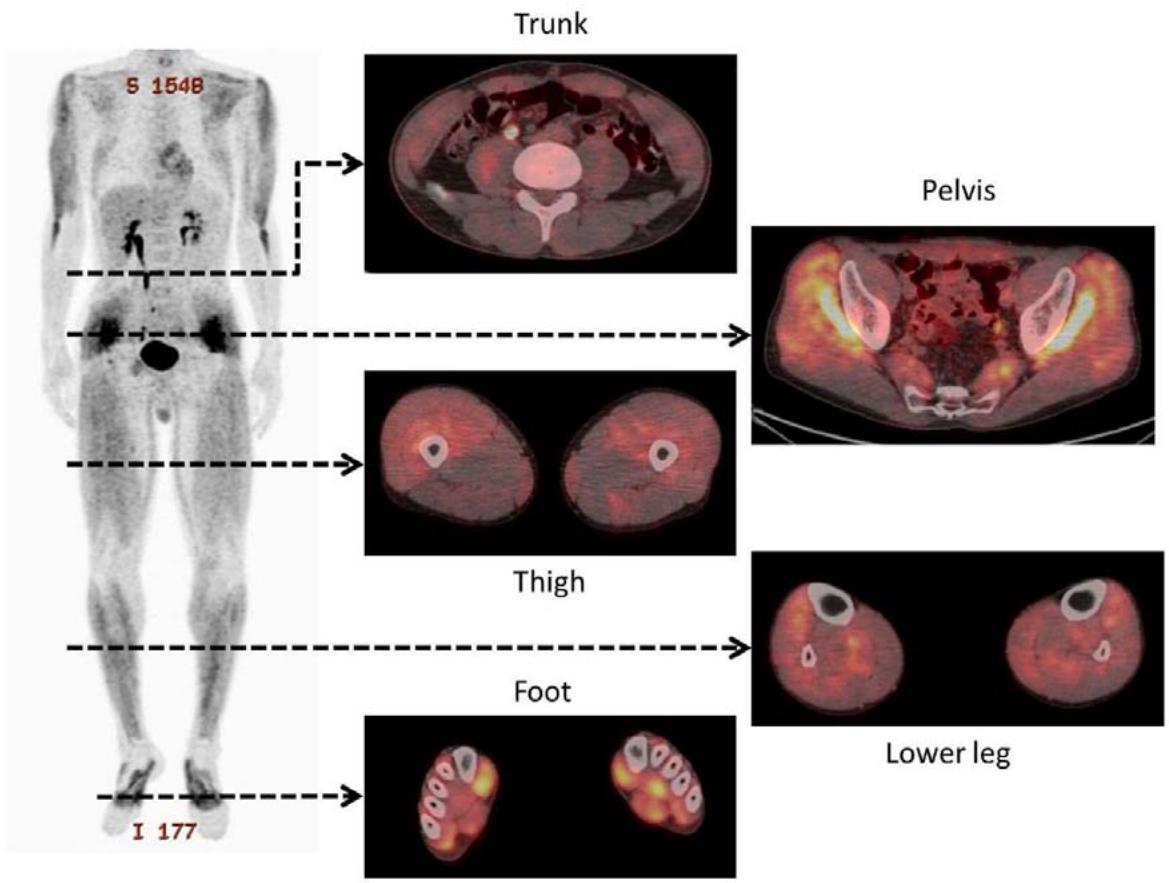
Figure 9: Taken from Nakase J, Inaki A, Mochizuki T, Toratani T, Kosaka M, Ohashi Y, et al. Whole Body Muscle Activity During the Fifa 11+ Program Evaluated by Positron Emission Tomography. PLoS One (2013) 8(9):e73898. Epub 2013/09/26. doi: https://doi.org/10.1371/journal.pone.0073898. License: Creative Commons Attribution License
doi:10.1371/journal.pone.0073898.g002
Figure 2. Representative whole-body positron emission tomography images after performance of the $11+$ by patients in the exercise group.
Tracer uptake can be more intense according to the intensity of exercise such as weightlifting as shown by Yoshimizu et al., in 2022 (239). Not only the muscle involved in weightlifting but also the heart showed intense tracer uptake (figure 10) revealing the systemic dimension of exercise.
 Figure 10: Taken from Yoshimizu R. Whole Body Muscle Activity During Weightlifting Exercise Evaluated by Positron Emission Tomography (2022). Available from: https://www.researchsquare.com/article/rs-612816/v2. License: CC BY 4.0 Figure 2
#### Representative whole-body PET images after performance of HPC
Representative whole-body PET images of the HPC group are shown. SUVs were significantly increased in 29 of 71 skeletal muscles in the whole body. PET; positron emission tomography, HPC; hang power clean
k) COVID-19: the low T3 syndrome, IL-6, and $^{18}$ F-FDG imaging
Zou et al., evaluated the data from 149 COVID-19 patients in 2020 looking for the characteristics of the low T3 syndrome, which was indeed found in forty-one cases. Disease severity and parameters of inflammation were significantly associated with the changes of thyroid parameters (240). The low T3 syndrome is an indicator of poor prognosis and mortality in COVID-19 patients (241). The low T3 syndrome together with elevated levels of NT-proBNP are indicators of poor outcome (242).
Leyfman et al. discussed an interaction of IL-6 and hypoxia in COVID-19 patients. They postulated an interaction between viruses and different organs which led to IL-6 elevation (Figure 1 in (243)). In this depiction, hypoxia appears as an isolated element. This idea of disease can be found in many publications, e.g., (the graphical abstract in (112)), (Figure 1 in (244)).
COVID infection is related to glycolysis (245), therefore diagnostic imaging with $^{18}\mathrm{F}$ -FDG should demonstrate organs affected by hypoxia. The characterization of persisting symptoms of long-COVID has been recently demonstrated by Kiatkittikul et al., (246). The image panel shows a variety of $^{18}\mathrm{F}$ -FDG uptake patterns (Figure 10) including skeletal muscles, heart, vessels, and the lung. We interpret these changes as being related to hypoxia and consequently highly suggestive of $\mathrm{CoQ}_{10}$ deficiency and altered mitochondrial function.
Fig.1 Maximal intensity projection F-18 FDG PET images of patients with post-acute COVID-19. (a) Case 1: diffuse hypermetabolism of the skeletal muscle throughout the body (arrow). (b) Case 7: multiple hypermetabolism at respiratory muscle (arrow) and quadratus lumbrum muscle (dot arrow) with hypermetabolic node at right hilar region (arrow head). (c) Case 2: diffuse hypermetabolism along the vascular wall throughout the body. (d) Case 4 with bacterial pneumonia: focal hypermetabolism along the vascular walls of bilateral popliteal vessels (arrow) with focal hypermetabolism in the right lung (arrowhead) and diffuse bone marrow and splenic uptake. (e) Case 13 with psoriasis exacerbation: multiple hypermetabolic skin lesions (arrow). Abbreviation: FDG fluorodeoxyglucose, PET positron emission tomography, COVID-19 coronavirus disease 2019
A recent case report on hypoxia in COVID-19 infection was delivered by Serbanescu-Kele et al., in 2022 (247). The publication presented the decreasing course of oxygen saturation during hospital treatment. Quite unfortunately the authors utilized the term "happy hypoxaemia" ignoring the fatal outcome for the patient. Figure 1B of this publication showed the macroscopic appearance of a dark arterial blood sample which to our clinical understanding did not look happy at all (247).
The term happy hypoxia in relation to COVID-19 disease was used by Jennifer Couzin-Frankel in 2020 referring to the clinical experiences of Reuben Jesse Strayer, an emergency physician working at the Maimonides Medical Center, New York (248). It is quite important to know that the Jennifer Couzin-Frankel has a major in history of science, but no medical education and works as a staff writer for Science. Her article was not peer-reviewed and contained no scientific references. The original publication by Strayer on hypoxia and COVID-19 patients (249) was not cited. The original authors acknowledged that by 2020 little was known about the pathophysiology of COVID-19 disease. It is our personal feeling that this misleading and erroneous term should be banned from the literature.
A recent study of the UK Biobank, published 19 January 2023, found that cardiovascular disease and mortality were associated with long term COVID-19 disease. The authors could not identify conclusively any mechanism involved and simply provided a rather general and disappointing recommendation for ongoing clinical monitoring (250). Affected patients need indeed monitoring, however treatment implementation should be added since it has a higher ethical priority.
In 2022 Hansen et al., published an industry-backed study (Pharma Nord) that evaluated a high dose therapy with $\mathrm{CoQ}_{10}$ in patients presenting a post COVID condition. A sound and scientific rationale for choosing the dose of $500\mathrm{mg}$ $\mathrm{CoQ}_{10}$ was not given (251). Initial and final $\mathrm{CoQ}_{10}$ levels in the patients were not evaluated. The duration of $\mathrm{CoQ}_{10}$ administration was quite short (6 weeks). In our experience, the combined supplementation with magnesium, selenium, and $\mathrm{CoQ}_{10}$ starts to show recovery results after 9-12 months. Full recovery of the thyroid requires two to three years. Our recommended dose for $\mathrm{CoQ}_{10}$ supplementation is $0.9\mathrm{mg/kg}$, i.e., app. $60\mathrm{mg/d}$.
### i) Heat stress and cardiac disease - a pathogenesis proposal
Recently two publications have looked at the relationship between cardiac disease and heat stress. Ranek et al., described changes of heat shock proteins in heart failure, pointing towards the increased expression of hsp70, hsp90 and BAG-3 (252). In 2012 Iguchi et al., looked at cardiovascular and hormonal changes following heat stress produced by sitting in a heat stress chamber for 30 minutes at $73^{\circ}\mathrm{C}$ (253). They demonstrated that this stimulus had an elevation of hsp72 and prolactin. The increase in prolactin levels seemed related to the endurance capacity in response to exercise under heat conditions (254). Heat exposure and muscular activity can also affect blood magnesium levels inducing a severe decrease to sub-optimal levels (255).
Given these concepts, we can state that Ritossa observed the effects of an external heat source which led to the heat shock and the unfolded protein reactions. A modern view of heat shock response includes more toxic elements besides heat exposure. Trautinger provided the following description: "All organisms respond to sudden environmental changes with the increased transcription of genes belonging to the family of heat shock proteins (hsps). Hsp-inducing stress factors include elevated temperatures, alcohol, heavy metals, oxidants, and agents leading to protein denaturation. The induction of heat shock proteins is followed by a transient state of increased resistance to further stress, and the heat shock response is generally thought to represent an evolutionary conserved adaptive mechanism to cope with hostile environmental conditions" (256). Current knowledge of the complex processes involved in the hsp response describes essential interactions with ATP in the case of ATP-dependent hsp chaperones (257, 258). Alteration in protein structure will also involve the repair mechanisms of the unfolded protein response (259).
Heat stress has been investigated under clinical conditions. Bouchama, from Saudia Arabia, has published a series of studies dealing with heatstroke in humans. In 1995 a historical review on heatstroke included a discussion on the potential role of endotoxins in the disease (260). Recently the effects of acute, supra-physiological temperature stress have been examined at the transcriptome level. The test subjects were exposed to a mean temperature of $75^{\circ}\mathrm{C}$ in a sauna. The analysis of blood mononuclear gene expression revealed several patterns of change (Figure 2 in (261)). One key finding referred to altered mitochondrial function involving several complexes of the OXPHOS chain, including Complex I. Both electron transfer, as well as ATP production, were repressed. Bouchama et al., also identified an alteration of cytochrome C oxidase, corresponding to Complex IV (262). Both Complex I and Complex IV depend on iron-sulfur clusters for their function. Situations of physical exertion in hot environments with temperatures up to $41.5^{\circ}\mathrm{C}$ can be found in sports activities (263). Elevated temperature as a source of stress is not only limited to sports. It can also affect everyday activities (264). Furthermore, these changes are like those seen in heart failure, where the function of the OXPHOS chain is altered due to changes in Complex I and V (265). As a whole, ATP production can be disturbed (266). It must be kept in mind that the heat stress response requires sufficient ATP since some hsp are ATP-dependent (258). Experimental repeated exposure to heat stress can negatively affect the heat shock response of the heart (267). The increase of hsp70 levels after heat stress appears to decline with time, thus increasing the susceptibility of the heart to damage (268). This propensity to a myocardial injury can be corrected by $\mathrm{CoQ}_{10}$, resulting in higher hsp70 levels (128). In a similar study, the use of the selenium analog ebselen, resulted in improved expression of hsp70, which was associated with reduced myocardial infarct size (269).
Experimental work by Liedtke and Hughes in 1976 showed the detrimental effect of hyperthermia on cardiac specimens producing functional impairment together with increased glycolysis and lower ATP levels. The authors described the results as an additional energy drain to the heart (270). In 1989 Huang and Liedtke described the metabolic situation in re-perfused myocardium as being unable to synthesize ATP due to an inefficient electron transport and OXPHOS (271).
In a recent epidemiological analysis, Wang et al. described an association between previous heat stress situations and heart and kidney disease (272). Mechanisms leading to these changes were not discussed; however, these observations imply that heat exposure had left an altered biochemical system behind. Nzvere et al., have reached similar conclusions in evaluating the long-term consequences of heatstroke on cardiac disease (273).
Heat stress is also a serious condition in human medicine (274). Heat stress per-se stimulates the production of mitochondrial superoxide (275). The administration of dietary $\mathrm{CoQ}_{10}$ can attenuate oxidative changes produced by heat shock in chickens (276). A similar protection can be observed through administration of $300\mathrm{mg}\mathrm{CoQ}_{10}$ prior to heat exposure in elite swimmers (277). The production of mitochondrial small heat-shock protein (lmw Hsp) protects electron transport from Cl to CIV (278). The authors declared that ubiquinone is the most thermo-labile protein in Complex I.
We have included this description keeping in mind the condition of postpartum cardiomyopathy (279) as studied by Cénac 30 years ago where the central deficiency was low selenium (280). Due to birth customs in Nigeria these women are exposed to extreme heat stress in the postpartum period. Experimental data on heat stress effects on the chicken heart has shown that these deleterious effects can be modulated by $\mathrm{CoQ}_{10}$ (281). An indirect hint on prolonged cardiac dysfunction following heatstroke was published by Zahger, Moses and Weiss in 1989 (282). A description of the characteristics of heat stroke and cardiac dysfunction published by Marchand and Gin were centered on thermoregulation and heat dissipation (Figure 1 in (283)). Other authors have found a relation between biomarkers and mortality after exertional heat stress where fatal cases had higher levels of NT-proBNP (284). A current view on heatstroke which does not consider the role of $\mathrm{CoQ}_{10}$ was published by Bouchama (285).
## II. DISCUSSION AND OUTLOOK
Our clinically oriented literature review has disclosed that benign thyroid disease and several other entities including notable heart disease and COVID-19 share common features such as the symptom fatigue, and the events of hypoxia and glycolysis, elevated IL-6 levels, and the low T3 syndrome. We propose that $\mathrm{CoQ}_{10}$ deficiency plays a fundamental role in these diseases affecting the functionality of Complex I of the mitochondrial respiratory chain. The following Table contains an approximation to this process.
1. Initiation of glycolysis resulting from stabilization of HIF- $1\alpha$ (4). Interactions with $\mathrm{CoQ}_{10}$ and iron.
2. Increased production of reactive oxygen species follows functional alteration of respiratory Complex I (286)
3. Altered OXPHOS lead to reduced ATP production (287)
4. In situations of lack of oxygen - hypoxia - Complex I can be modified to a deactive state (288)
5. Experimental function loss of Complex I goes along glycolysis and with $^{18}\mathrm{F}$ -FDG uptake (289)
We have briefly reviewed the role of diagnostic imaging keeping our eyes on glycolysis as demonstrated by $^{18}\mathrm{F}$ -FDG scans, and on fatty acid uptake based on the use of iodine-labeled methyl iodophenyl-pentadecanoic acid (BMIPP) (28). The clinical rationale for imaging these metabolic paths has been summarized by Yoshinaga et al. in 2007 (290). Contrasting with this structured description we must refer to a study by Dilsizian et al. where ideas about the interpretation of BMIPP scans were given. The conclusions offered by the authors had no profound biochemical foundation (291). What is useful in this publication is Figure 2 where a comparison between a heart perfusion and heart BMIPP study is shown. This image clearly shows that perfusion does not reveal metabolic changes (Figure 11).
An interesting observation on altered mitochondrial function can be taken from studies dealing with cardiac disease and the influence of cardiac surgery in restoring fatty acid metabolism. Morishima et al. found persistent alteration of fatty acid metabolism in patients with atherosclerotic changes even though the surgical intervention had resolved the ischemic condition (292). Our interpretation of persistent glucose utilization is that such patients present persistent, unrecognized, $\mathrm{CoQ}_{10}$ deficiency.
In 2014 Yoshinaga referring to the mystical-magical term of ischemic "memory brought" stated the following (293):
Metabolic imaging has been used to detect the footprint left by previous ischaemic episodes evident due to delayed recovery of myocardial metabolism (persistent dominant glucose utilization with suppression of fatty acid oxidation).
The following graphical allegory describes the medical setting relating hypoxia and cardiac disease. Superficial research has not considered biochemical changes sufficiently. Adding the biochemical background one can see that there are many more components to be explored. It is evident that keeping an eye only on the flow or on the uptake of $^{18}\mathrm{F}$ -FDG will not open a perception for fatty acid uptake. The following graphical allegory about the swimmer and the diver illustrates this situation (Figure 12).

A
Superficial view of the situation as seen by a skin diver, i.e., looking only at myocardial perfusion studies.

Figure 13 illustrates our proposal on the essential biochemistry of resilience that maintains health.
Deep view of the situation as seen by a scuba diver. A profound look will find hypoxia activation after $\mathrm{CoQ}_{10}$ deficiency and hypoxia-related changes such as elevated IL-6, low T3 syndrome.
### B
Figure 12: A. Graphical allegory - Myocardial perfusion studies correspond to the left image of the superficial snorkeling diver who sees nothing but flow. B. The lower panel shows the option to go deeper into the process by using additional gear signalized by the self-contained underwater breathing apparatus (SCUBA). This second image represents biochemical elements as described throughout the text. The images were taken from WikiCommons.
Additional data on the information delivered by BMIPP as compared to FDG can be found in publications authored by Yoshinaga (294). Yamagishi et al., summarized the impact of BMIPP and FDG diagnostic imaging going well beyond perfusion (Table 29 in (295)).
Looking beyond organ-related disease, the transmission of intergenerational trauma is a process that is also related to hypoxia and mitochondrial dysfunction (296).
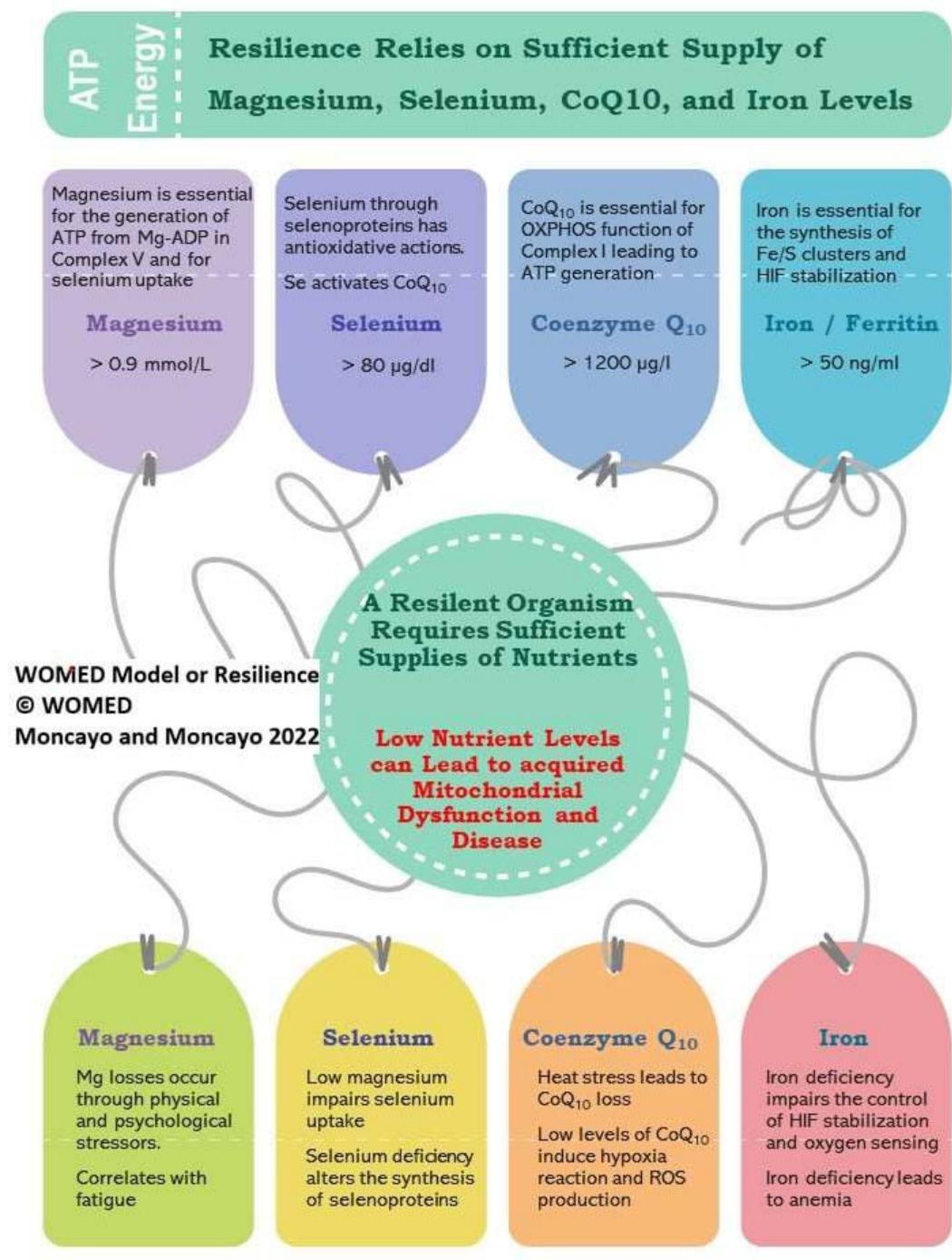 Figure 13: The 2022 WOMED concept of resilience and health based on essential biochemical parameters.
Figure 14 illustrates our model of cardiac disease based on the information reviewed here. The model can be applied to other clinical conditions related to hypoxia.
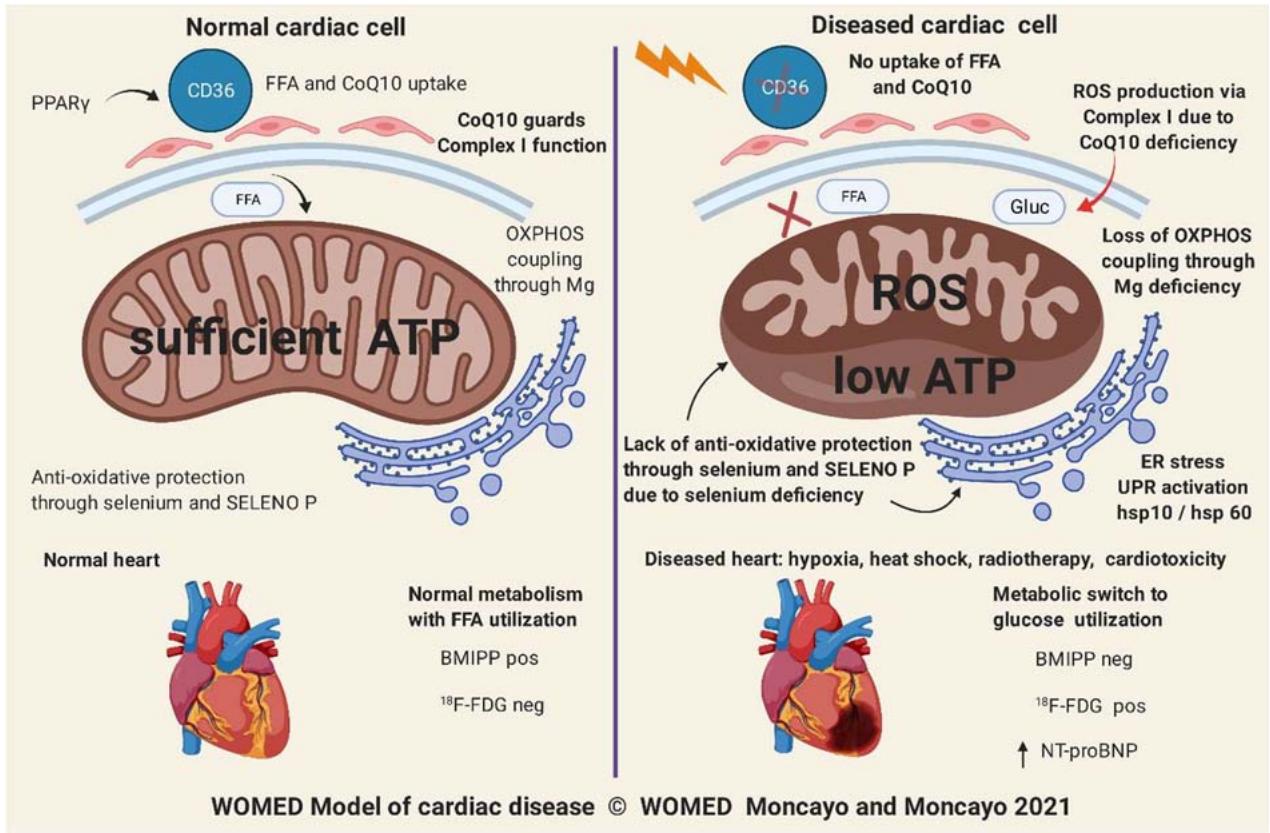 Figure 14: The 2021 WOMED model of cardiac disease
#### Supplemental information
A simplified clinical description of the symptoms of hypoxia has been published by the Cleveland Clinic.
Hypoxia: Causes, Symptoms, Tests, Diagnosis & Treatment (clevelandclinic.org)
An overview of causes and diseases related to hypoxia authored by Rhiannon Brett can be found in the Calgary Guide to Understanding Disease.
Hypoxemia: Pathogenesis and clinical findings | Calgary Guide (ucalgary.ca)
Generating HTML Viewer...
References
287 Cites in Article
(2015). Unknown Title.
G Woodward,F Cramer (1952). 2-Desoxyl-D-Glucose as an Inhibitor of Anaerobic Glycolysis in Tumor Tissue.
R Moncayo,H Moncayo (2015). Proof of Concept of the Womed Model of Benign Thyroid Disease: Restitution of Thyroid Morphology after Correction of Physical and Psychological Stressors and Magnesium Supplementation.
Roy Moncayo,Helga Moncayo (2007). A musculoskeletal model of low grade connective tissue inflammation in patients with thyroid associated ophthalmopathy (TAO): the WOMED concept of lateral tension and its general implications in disease.
Roy Moncayo,Alexander Kroiss,Manfred Oberwinkler,Fatih Karakolcu,Matthias Starzinger,Klaus Kapelari,Heribert Talasz,Helga Moncayo (2008). The role of selenium, vitamin C, and zinc in benign thyroid diseases and of selenium in malignant thyroid diseases: Low selenium levels are found in subacute and silent thyroiditis and in papillary and follicular carcinoma.
Roy Moncayo,Helga Moncayo (2012). Thyroid Sonography in 3D with Emphasis on Perfusion.
Roy Moncayo,Helga Moncayo (2014). Exploring the aspect of psychosomatics in hypothyroidism: The WOMED model of body–mind interactions based on musculoskeletal changes, psychological stressors, and low levels of magnesium.
Roy Moncayo,Helga Moncayo (2015). The WOMED model of benign thyroid disease: Acquired magnesium deficiency due to physical and psychological stressors relates to dysfunction of oxidative phosphorylation.
Roy Moncayo,Helga Moncayo (2017). Applying a systems approach to thyroid physiology: Looking at the whole with a mitochondrial perspective instead of judging single TSH values or why we should know more about mitochondria to understand metabolism.
Ibolja Cernak,Veljko Savic,Jelena Kotur,Vera Prokic,Milic Veljovic,Dragan Grbovic (2000). Characterization of Plasma Magnesium Concentration and Oxidative Stress Following Graded Traumatic Brain Injury in Humans.
G Grases,J Pérez-Castelló,P Sanchis,A Casero,J Perelló,B Isern (2006). Anxiety and Stress among Science Students. Study of Calcium and Magnesium Alterations.
H Humphray,F Heaton (1972). RELATIONSHIP BETWEEN THE THYROID HORMONE AND MINERAL METABOLISM IN THE RAT.
Yukifumi Nagai,Toichiro Hosoya (1977). Properties of the Na+,K+-stimulated Adenosine Triphosphatase System Associated with the Plasma Membrane of Pig Thyroid Glands1.
A Chandra,H Goswami,P Sengupta (2014). Effects of Magnesium on Cytomorphology and Enzyme Activities in Thyroid of Rats.
A Shreenath,M Ameer,J Dooley (2022). Selenium Deficiency. Statpearls. Treasure Island (FL).
Dietrich Behne,Horst Hilmert,Stefan Scheid,Hildegard Gessner,Walter Elger (1988). Evidence for specific selenium target tissues and new biologically important selenoproteins.
H Pedersen,S Mortensen,M Rohde,Y Deguchi,G Mulvad,P Bjerregaard,J Hansen (1999). High serum coenzyme Q<sub>10</sub>, positively correlated with age, selenium and cholesterol, in Inuit of Greenland. A pilot study.
J Vitale,M Nakamura,D Hegsted (1957). THE EFFECT OF MAGNESIUM DEFICIENCY ON OXIDATIVE PHOSPHORYLATION.
Maria Depaoli,Jesse Hay,Wolfgang Graier,Roland Malli (2018). The enigmatic ATP supply of the endoplasmic reticulum.
Sonja Hess,Michael Zimmermann,Richard Hurrell,Myrtha Arnold,Wolfgang Langhans (2002). Iron Deficiency Anemia Reduces Thyroid Peroxidase Activity in Rats.
Roland Lill,Kerstin Diekert,Anita Kaut,Heike Lange,Winfried Pelzer,Corinna Prohl,Gyula Kispal (1999). The Essential Role of Mitochondria in the Biogenesis of Cellular Iron-Sulfur Proteins.
John Maguire,Kelvin Davies,Peter Dallman,Lester Packer (1982). Effects of dietary iron deficiency on iron-sulfur proteins and bioenergetic functions of skeletal muscle mitochondria.
Martin Wenger,Rudolf Gasser,Eveline Donnemiller,Hermann Erler,Hartmut Glossmann,Josef Patsch,Roy Moncayo,Michael Schirmer (2003). Generalized Large Vessel Arteritis Visualized by <sup>18</sup> Fluorodeoxyglucose-Positron Emission Tomography.
Arvind Chopra,Liang Shan,W Eckelman,Kam Leung,Martin Latterner,Stephen Bryant,Anne Menkens (2004). Molecular Imaging and Contrast Agent Database (MICAD): Evolution and Progress.
F Knapp,J Kropp (1999). BMIPP-design and development.
Gary Lopaschuk (2017). Metabolic Modulators in Heart Disease: Past, Present, and Future.
William Stanley,Fabio Recchia,Gary Lopaschuk (2005). Myocardial Substrate Metabolism in the Normal and Failing Heart.
G Festenstein,F Heaton,J Lowe,R Morton (1955). A constituent of the unsaponifiable portion of animal tissue lipids (λmax. 272mμ.).
F Crane,Y Hatefi,R Lester,Carl Widmer (1957). Isolation of a quinone from beef heart mitochondria.
Frederick Crane (1959). Isolation of Two Quinones with Coenzyme Q Activity from Alfalfa..
F Crane,C Widmer,R Lester,Y Hatefi,Wanda Fechner (1959). Studies on the electron transport system.
David Hendlin,Thomas Cook (1960). The Activity of Coenzyme Q10 and Its Analogues in the Succinoxidase System of Electron Transport Particles.
Donald Wolf,Carl Hoffman,Nelson Trenner,Byron Arison,Clifford Shunk,Bruce Linn,James Mcpherson,Karl Folkers (1958). COENZYME Q. I. STRUCTURE STUDIES ON THE COENZYME Q GROUP.
Otto Hoffmann-Ostenhof (1947). Die Biochemie der Chinone.
E Friedmann,D Marrian,I Simon‐reuss (1948). ANALYSIS OF ANTIMITOTIC ACTION OF CERTAIN QUINONES.
M Barnes,R Morton (1962). Ubiquinone, Plastoquinone and Related Substances.
Paul Gale,Frank Koniuszy,Atwood Page,Karl Folkers,Henry Siegel (1961). Coenzyme Q. XXIV. On the significance of coenzyme Q 10 in human tissues.
Anders Kalén,Eeva‐liisa Appelkvist,Gustav Dallner (1989). Age‐related changes in the lipid compositions of rat and human tissues.
G Ravaglia,P Forti,F Maioli,R Scali,F Boschi,A Cicognani,P Morini,A Bargossi,G Gasbarrini (1996). Coenzyme Q10 plasma levels and body composition in elderly males.
Y Tomono,J Hasegawa,T Seki,K Motegi,N Morishita (1986). Pharmacokinetic Study of Deuterium-Labelled Coenzyme Q10 in Man.
Kyosuke Watanabe,Satoshi Nozaki,Miki Goto,Ken-Ichi Kaneko,Emi Hayashinaka,Satsuki Irie,Akira Nishiyama,Kazuaki Kasai,Kenji Fujii,Yasuhiro Wada,Kei Mizuno,Kenji Mizuseki,Hisashi Doi,Yasuyoshi Watanabe (2019). PET imaging of 11C-labeled coenzyme Q10: Comparison of biodistribution between [11C]ubiquinol-10 and [11C]ubiquinone-10.
C (2019). Ubiquinone-10.
Magnus Bentinger,Michael Tekle,Kerstin Brismar,Tadeusz Chojnacki,Ewa Swiezewska,Gustav Dallner (2008). Stimulation of coenzyme Q synthesis.
Barry Commoner,Jonathan Townsend,George Pake (1954). Free Radicals in Biological Materials.
Rebeca Gerschman,Daniel Gilbert,Sylvanus Nye,Peter Dwyer,Wallace Fenn (1954). Oxygen Poisoning and X-irradiation: A Mechanism in Common.
D Harman (1956). Aging: A Theory Based on Free Radical and Radiation Chemistry.
F Ritossa (1962). A new puffing pattern induced by temperature shock and DNP in drosophila.
Klaus Schwarz,Calvin Foltz (1957). SELENIUM AS AN INTEGRAL PART OF FACTOR 3 AGAINST DIETARY NECROTIC LIVER DEGENERATION.
H Tapiero,D Townsend,K Tew (2003). The antioxidant role of selenium and seleno-compounds.
Ujang Tinggi (2008). Selenium: its role as antioxidant in human health.
J Barciela,C Latorre,S García-Martín,R Peña (2008). A Brief Study of the Role of Selenium as Antioxidant.
J Green,E Edwin,A Diplock,J Bunyan (1961). Role of Selenium in Relation to Ubiquinone in the Rat.
M Hidiroglou,K Jenkins,R Carson,G Brossard (1967). SELENIUM AND COENZYME Q<sub>10</sub> LEVELS IN THE TISSUES OF DYSTROPHIC AND HEALTHY CALVES.
S Vadhanavikit,H Ganther (1993). Decreased Ubiquinone Levels in Tissues of Rats Deficient in Selenium.
S Vadhanavikit,H Ganther (1994). Selenium deficiency and decreased Coenzyme Q levels.
Esther Van Zuuren,Amira Albusta,Zbys Fedorowicz,Ben Carter,Hanno Pijl (2013). Selenium supplementation for Hashimoto's thyroiditis.
Nadine Flowers,Louise Hartley,Daniel Todkill,Saverio Stranges,Karen Rees (2014). Co-enzyme Q10 supplementation for the primary prevention of cardiovascular disease.
Mohammed Madmani,Ahmad Yusuf Solaiman,Khalil Tamr Agha,Yasser Madmani,Yasser Shahrour,Adib Essali,Waleed Kadro (2013). Coenzyme Q10 for heart failure.
L Ernster,P Forsmark-Andr�e (1993). Ubiquinol: an endogenous antioxidant in aerobic organisms.
S Sugiyama,M Kitazawa,T Ozawa,K Suzuki,Y Izawa (1980). Anti-oxidative effect of coenzyme Q10.
Degli Esposti,M Ngo,A Mcmullen,G Ghelli,A Sparla,F Benelli,B (1996). The Specificity of Mitochondrial Complex I for Ubiquinones.
Megumi Shimizu,Tetsuro Miyazaki,Atsutoshi Takagi,Yurina Sugita,Shoichiro Yatsu,Azusa Murata,Takao Kato,Shoko Suda,Shohei Ouchi,Tatsuro Aikawa,Masaru Hiki,Shuhei Takahashi,Makoto Hiki,Hidemori Hayashi,Takatoshi Kasai,Kazunori Shimada,Katsumi Miyauchi,Hiroyuki Daida (2016). Low circulating coenzyme Q10 during acute phase is associated with inflammation, malnutrition, and in-hospital mortality in patients admitted to the coronary care unit.
Michael Cocchi,Brandon Giberson,Katherine Berg,Justin Salciccioli,Ali Naini,Catherine Buettner,Praveen Akuthota,Shiva Gautam,Michael Donnino (2012). Coenzyme Q10 levels are low and associated with increased mortality in post-cardiac arrest patients.
C Jiang,W Jiang,C Ip,H Ganther,J Lü (1999). Selenium-Induced Inhibition of Angiogenesis in Mammary Cancer at Chemopreventive Levels of Intake.
C Jiang,H Ganther,J Lü (2000). Monomethyl Selenium--Specific Inhibition of Mmp-2 and Vegf Expression: Implications for Angiogenic Switch Regulation.
Otto Warburg (1911). Über Beeinflussung der Sauerstoffatmung..
O Warburg,S Minami (1923). Versuche an Überlebendem Carcinom-Gewebe.
V Gabai,V Mosina,K Budagova,A Kabakov (1995). Spontaneous Overexpression of Heat-Shock Proteins in Ehrlich Ascites Carcinoma Cells During in Vivo Growth.
Susanna Airaksinen,Christina Råbergh,Lea Sistonen,Mikko Nikinmaa (1998). Effects of heat shock and hypoxia on protein synthesis in rainbow trout (<i>Oncorhynchus mykiss</i>) cells.
J Lepock,H Frey,K Ritchie (1993). Protein denaturation in intact hepatocytes and isolated cellular organelles during heat shock.
Michael Freeman,Michael Borrelli,Michael Meredith,James Lepock (1999). On the path to the heat shock response: destabilization and formation of partially folded protein intermediates, a consequence of protein thiol modification.
V Nguyen,O Bensaude (1994). Increased Thermal Aggregation of Proteins in Atp-Depleted Mammalian Cells.
Liangli Wang,Uwe Schumann,Yuefei Liu,Olga Prokopchuk,Jürgen Steinacker (1985). Heat shock protein 70 (Hsp70) inhibits oxidative phosphorylation and compensates ATP balance through enhanced glycolytic activity.
Xu Xu,Sounak Gupta,Wenli Hu,Barbara Mcgrath,Douglas Cavener (2011). Hyperthermia Induces the ER Stress Pathway.
O Warburg,F Wind,E Negelein (1927). The Metabolism of Tumors in the Body.
S Flexner,J Jobling (1907). Metaplasia and metastasis of a rat tumor.
C Jensen (1909). Uebertragbare Rattensarkome.
M Bergmann,H Schotte,W Lechinsky (1922). Über Die Ungesättigten Reduktionsprodukte Der Zuckerarten Und Ihre Umwandlungen, Iii.: Über 2-Desoxyglucose (Gluco-Desose).
E Mcdonald (1952). Note from the Biochemical Research Foundation.
Francis Cramer,Gladys Woodward (1952). 2-Desoxy-D-glucose as an antagonist of glucose in yeast fermentation.
G Woodward,M Hudson (1954). Abstract of the effect of 2-desoxy-D-glucose on glycolysis and respiration of tumor and normal tissues.
H Stewart Transplantable and Transmissible Tumors of Animals.
D Washington (1959). Armed Forces Institute of Pathology.
Panli Li,Qiufang Liu,Xiuying Wang,Gang Huang,Shaoli Song (2019). 18F-Deoxyglucose (18F-FDG) Positron Emission Tomography/Computed Tomography (PET/CT) Monitoring of Dynamic Growth Characteristics of Walker-256 Tumor Models in 3 Different Locations in Rats.
Otto Warburg,K Posener,E Negelein (1924). �ber den Stoffwechsel der Carcinomzelle.
A Wick,D Drury,H Nakada,J Wolfe (1957). Localization of the Primary Metabolic Block Produced by 2-Deoxyglucose.
Stanley Barban,Henry Schulze (1961). The Effects of 2-Deoxyglucose on the Growth and Metabolism of Cultured Human Cells.
P Trayhurn (2019). Oxygen-a Critical, but Overlooked, Nutrient.
S Yan,I Tritto,D Pinsky,H Liao,J Huang,G Fuller (1995). Induction of Interleukin 6 (Il-6) by Hypoxia in Vascular Cells. Central Role of the Binding Site for Nuclear Factor-Il-6.
G Wang,G Semenza (1993). General Involvement of Hypoxia-Inducible Factor 1 in Transcriptional Response to Hypoxia.
A Ouiddir,C Planès,I Fernandes,A Vanhesse,C Clerici (1999). Hypoxia Upregulates Activity and Expression of the Glucose Transporter Glut1 in Alveolar Epithelial Cells.
G Solaini,A Baracca,G Lenaz,G Sgarbi (2010). Hypoxia and Mitochondrial Oxidative Metabolism.
P Lee,N Chandel,M Simon (2016). Cellular Adaptation to Hypoxia through Hypoxia Inducible Factors and Beyond.
Z Luo,M Tian,G Yang,Q Tan,Y Chen,G Li (2022). Hypoxia Signaling in Human Health and Diseases: Implications and Prospects for Therapeutics.
S Kierans,C Taylor (2020). Regulation of Glycolysis by the Hypoxia-Inducible Factor (Hif): Implications for Cellular Physiology.
Della Rocca,Y Fonticoli,L Rajan,T Trubiani,O Caputi,S Diomede,F (2022). Hypoxia: Molecular Pathophysiological Mechanisms in Human Diseases.
M Tateno,N Tamaki,M Yukihiro,T Kudoh,N Hattori,E Tadamura (1996). Assessment of Fatty Acid Uptake in Ischemic Heart Disease without Myocardial Infarction.
K Zen,K Ito,T Hikosaka,Y Adachi,S Yoneyama,S Katoh (2000). Uncommon and Dynamic Changes Detected by 123i-15-(P-Iodophenyl)-3-R,S-Methylpentadecanoic Acid Myocardial Single Photon Emission Computed Tomography in a Stunned Myocardium Induced by Coronary Microvascular Spasm.
S Biswas,M Sarai,H Toyama,H Hishida,Y Ozaki (2012). Discrepancy between Myocardial Perfusion and Fatty Acid Metabolism Following Acute Myocardial Infarction for Evaluating the Dysfunctional Viable Myocardium.
Shankar Biswas,Masayoshi Sarai,Hiroshi Toyama,Akira Yamada,Sadako Motoyama,Hiroto Harigaya,Tomonori Hara,Hiroyuki Naruse,Hitoshi Hishida,Yukio Ozaki (2012). P-116 Impact of Defect Score and Washout Rate of 123I-BMIPP on Left Ventricular Functional Recovery Following Acute Myocardial Infarction.
A Hammerer-Lercher,E Neubauer,S Müller,O Pachinger,B Puschendorf,J Mair (2001). Head-to-Head Comparison of N-Terminal Pro-Brain Natriuretic Peptide, Brain Natriuretic Peptide and N-Terminal Pro-Atrial Natriuretic Peptide in Diagnosing Left Ventricular Dysfunction.
I Nakae,S Matsuo,T Koh,K Mitsunami,M Horie (2006). Iodine-123 Bmipp Scintigraphy in the Evaluation of Patients with Heart Failure.
M Boreková,J Hojerová,V Koprda,K Bauerová (2008). Nourishing and Health Benefits of Coenzyme Q10.
M Miles,P Putnam,L Miles,P Tang,A Degrauw,B Wong (2011). Acquired Coenzyme Q10 Deficiency in Children with Recurrent Food Intolerance and Allergies.
M Chase,M Cocchi,X Liu,L Andersen,M Holmberg,M Donnino (2018). Coenzyme Q10 in Acute Influenza.
M Donnino,M Cocchi,J Salciccioli,D Kim,A Naini,C Buettner (2011). Coenzyme Q10 Levels Are Low and May Be Associated with the Inflammatory Cascade in Septic Shock.
A Coppadoro,L Berra,A Kumar,R Pinciroli,M Yamada,U Schmidt (2013). Critical Illness Is Associated with Decreased Plasma Levels of Coenzyme Q10: A Cross-Sectional Study.
A Khan,D Johnson,S Carlson,L Hocum-Stone,R Kelly,A Gravely (2019). Nt-Pro Bnp Predicts Myocardial Injury Post-Vascular Surgery and Is Reduced with Coq 10 : A Randomized Double-Blind Trial.
A Fischer,M Klapper,S Onur,T Menke,P Niklowitz,F Döring (2015). Dietary Restriction Decreases Coenzyme Q and Ubiquinol Potentially Via Changes in Gene Expression in the Model Organism C. Elegans.
H Nakazawa,K Ikeda,S Shinozaki,S Yasuhara,Y Yu,Jaj Martyn (2019). Coenzyme Q10 Protects against Burn-Induced Mitochondrial Dysfunction and Impaired Insulin Signaling in Mouse Skeletal Muscle.
J Xu,S Tang,E Song,B Yin,D Wu,Bao (2017). Hsp70 Expression Induced by Co-Enzyme Q10 Protected Chicken Myocardial Cells from Damage and Apoptosis under in Vitro Heat Stress.
J Xu,S Tang,B Yin,J Sun,Bao (2018). Co-Enzyme Q10 Upregulates Hsp70 and Protects Chicken Primary Myocardial Cells under in Vitro Heat Stress Via Pkc/Mapk.
Z Demel,I Szilágyi,J Szabó,L Józsa (1978). Effect of Experimental Hypoxia on the Ubiquinone Level of Skeletal Muscle in Rabbits.
T Furuta,I Kodama,N Kondo,J Toyama,K Yamada (1982). A Protective Effect of Coenzyme Q10 on Isolated Rabbit Ventricular Muscle under Hypoxic Condition.
K Folkers,M Liu,T Watanabe,T Porter (1977). Inhibition by Adriamycin of the Mitochondrial Biosynthesis of Coenzyme Q10 and Implication for the Cardiotoxicity of Adriamycin in Cancer Patients.
Y Iwamoto,I Hansen,T Porter,K Folkers (1974). Inhibition of Coenzyme Q10-Enzymes, Succinoxidase and Nadh-Oxidase, by Adriamycin and Other Quinones Having Antitumor Activity.
J Doroshow,K Davies (1986). Redox cycling of anthracyclines by cardiac mitochondria. II. Formation of superoxide anion, hydrogen peroxide, and hydroxyl radical..
H Muhammed,T Ramasarma,C Kurup (1983). Inhibition of Mitochondrial Oxidative Phosphorylation by Adriamycin.
K Suzuki,Y Mizuno,M Yoshida (1988). Inhibition of Mitochondrial Nadh-Ubiquinone Oxidoreductase Activity and Atp Synthesis by Tetrahydroisoquinoline.
K Davies,J Doroshow (1986). Redox cycling of anthracyclines by cardiac mitochondria. I. Anthracycline radical formation by NADH dehydrogenase..
O Marcillat,Y Zhang,K Davies (1989). Oxidative and Non-Oxidative Mechanisms in the Inactivation of Cardiac Mitochondrial Electron Transport Chain Components by Doxorubicin.
K Saito,K Takeda,S Okamoto,R Okamoto,K Makino,Y Tameda (2000). Detection of Doxorubicin Cardiotoxicity by Using Iodine-123 Bmipp Early Dynamic Spect: Quantitative Evaluation of Early Abnormality of Fatty Acid Metabolism with the Rutland Method.
K Saito,K Takeda,K Imanaka-Yoshida,H Imai,T Sekine,Y Kamikura (2003). Assessment of Fatty Acid Metabolism in Taxan-Induced Myocardial Damage with Iodine-123 Bmipp Spect: Comparative Study with Myocardial Perfusion, Left Ventricular Function, and Histopathological Findings.
N Domae,H Sawada,E Matsuyama,T Konishi,H Uchino (1981). Cardiomyopathy and Other Chronic Toxic Effects Induced in Rabbits by Doxorubicin and Possible Prevention by Coenzyme Q10.
S Bradamante,E Monti,L Paracchini,G Perletti (1989). Hypoxia as a Risk Factor for Doxorubicin-Induced Cardiotoxicity: A Nmr Evaluation.
R Umezawa,K Takase,K Jingu,K Takanami,H Ota,T Kaneta (2013). Evaluation of Radiation-Induced Myocardial Damage Using Iodine-123 Β-Methyl-Iodophenyl Pentadecanoic Acid Scintigraphy.
R Umezawa,K Takanami,N Kadoya,Y Nakajima,M Saito,H Ota (2015). Assessment of Myocardial Metabolic Disorder Associated with Mediastinal Radiotherapy for Esophageal Cancer -a Pilot Study.
R Yan,J Song,Z Wu,M Guo,J Liu,J Li (2015). Detection of Myocardial Metabolic Abnormalities by 18f-Fdg Pet/Ct and Corresponding Pathological Changes in Beagles with Local Heart Irradiation.
T Ono,T Kohya,E Tsukamoto,T Mochizuki,K Itoh,Y Itoh (2000). Improvement in Fatty Acid Utilization in Relation to a Change in Left Ventricular Hypertrophy in Spontaneously Hypertensive Rats.
K Watanabe,M Sekiya,T Tsuruoka,J Funada,M Miyagawa (2001). Usefulness of 123 i-Bmipp with Myocardial Imaging for Evaluation of the Efficacy of Drug Therapy in Patients with Chronic Heart Failure.
S Yamauchi,Y Takeishi,O Minamihaba,T Arimoto,O Hirono,H Takahashi (2003). Dynamic 123 i-Bmipp Single-Photon Emission Computed Tomography in Patients with Congestive Heart Failure: Effect of Angiotensin Ii Type-1 Receptor Blockade.
K Hirooka,Y Yasumura,Y Ishida,K Komamura,A Hanatani,S Nakatani (2000). Improvement in Cardiac Function and Free Fatty Acid Metabolism in a Case of Dilated Cardiomyopathy with Cd36 Deficiency.
Y Kim,Y Sawada,G Fujiwara,H Chiba,T Nishimura (1997). Therapeutic Effect of Co-Enzyme Q10 on Idiopathic Dilated Cardiomyopathy: Assessment by Iodine-123 Labelled 15-(P-Iodophenyl)-3(R,S)-Methylpentadecanoic Acid Myocardial Single-Photon Emission Tomography.
S Yusuf,B Pitt,C Davis,W Hood,Jr Cohn,J (1992). Effect of Enalapril on Mortality and the Development of Heart Failure in Asymptomatic Patients with Reduced Left Ventricular Ejection Fractions.
V Pelouch,F Kolár,B Ost,M Milerová,R Cihák,J Widimský (1997). Regression of Chronic Hypoxia-Induced Pulmonary Hypertension, Right Ventricular Hypertrophy, and Fibrosis: Effect of Enalapril.
E De Cavanagh,F Inserra,L Ferder,C Fraga (2000). Enalapril and Captopril Enhance Glutathione-Dependent Antioxidant Defenses in Mouse Tissues.
Elena De Cavanagh,Barbara Piotrkowski,Nidia Basso,Ines Stella,Felipe Inserra,Leon Ferder,Cesar Fraga (2003). Enalapril and losartan attenuate mitochondrial dysfunction in aged rats.
G Humphreys (1947). The Diagnosis and Treatment of Congenital Cyanotic Heart Disease.
A Reyes,N Corrales,Nms Gálvez,S Bueno,A Kalergis,P González (2020). Contribution of Hypoxia Inducible Factor-1 During Viral Infections.
R Huang,M Huestis,E Gan,E Ooi,M Ohh (2021). Hypoxia and Viral Infectious Diseases.
B Wang,Z-L Li,Y-L Zhang,Y Wen,Y-M Gao,B-C Liu (2022). Hypoxia and Chronic Kidney Disease.
P Chen,W Chiu,P Hsu,S Lin,I Peng,C Wang (2020). Pathophysiological Implications of Hypoxia in Human Diseases.
L Østergaard (2021). Sars Cov-2 Related Microvascular Damage and Symptoms During and after Covid-19: Consequences of Capillary Transit-Time Changes, Tissue Hypoxia and Inflammation.
N Silverman,J Kohler,S Levitsky,D Pavel,R Fang,H Feinberg (1984). Chronic Hypoxemia Depresses Global Ventricular Function and Predisposes to the Depletion of High-Energy Phosphates During Cardioplegic Arrest: Implications for Surgical Repair of Cyanotic Congenital Heart Defects.
G Bedini,K Blecharz,S Nava,P Vajkoczy,G Alessandri,M Ranieri (2016). Vasculogenic and Angiogenic Pathways in Moyamoya Disease.
Y Takagi,K Kikuta,K Nozaki,M Fujimoto,J Hayashi,H Imamura (2007). Expression of Hypoxia-Inducing Factor-1 Alpha and Endoglin in Intimal Hyperplasia of the Middle Cerebral Artery of Patients with Moyamoya Disease.
B Fox,K Dorschel,M Lawton,J Wanebo (2021). Pathophysiology of Vascular Stenosis and Remodeling in Moyamoya Disease.
J Ma,X Fu,S Zhou,E Meng,Z Yang,H Zhang (2022). Study on the Serum Level of Coq 10 b in Patients with Moyamoya Disease and Its Mechanism of Affecting Disease Progression.
Hilda Delgado De La Herran,Yiming Cheng,Fabiana Perocchi (2021). Towards a systems-level understanding of mitochondrial biology.
J Lee,J Ko,C Ju,H Eltzschig (2019). Hypoxia Signaling in Human Diseases and Therapeutic Targets.
J Carter,C Eastman,J Corcoran,Lazarus (1974). Effect of Severe, Chronic Illness on Thyroid Function.
L Wartofsky,K Burman (1982). Alterations in Thyroid Function in Patients with Systemic Illness: The "Euthyroid Sick Syndrome.
V Vitek,C Shatney,Dorothy Lang,R Cowley (1983). Relationship of Thyroid Hormone Patterns to Survival in Canine Hemorrhagic Shock.
W Schroffner (1987). Comparative Aging, Geriatric Functional Assessment, Aging and Disease.
R Peeters,P Wouters,E Kaptein,H Van Toor,T Visser,G Van Den Berghe (2003). Reduced Activation and Increased Inactivation of Thyroid Hormone in Tissues of Critically Ill Patients.
A Rodriguez-Perez,F Palos-Paz,E Kaptein,T Visser,L Dominguez-Gerpe,J Alvarez-Escudero (2007). Identification of Molecular Mechanisms Related to Nonthyroidal Illness Syndrome in Skeletal Muscle and Adipose Tissue from Patients with Septic Shock.
T Moshang,K Chance,M Kaplan,R Utiger,O Takahashi (1980). Effects of Hypoxia on Thyroid Function Tests.
R Becker,D Wilmore,C Goodwin,Jr Zitzka,C Wartofsky,L Burman,K (1980). Free T4, Free T3, and Reverse T3 in Critically Ill, Thermally Injured Patients.
R Sawhney,A Malhotra (1990). Thyroid Function During Intermittent Exposure to Hypobaric Hypoxia.
O Ross,A Petros (2001). The Sick Euthyroid Syndrome in Paediatric Cardiac Surgery Patients.
G Iervasi,A Pingitore,P Landi,M Raciti,A Ripoli,M Scarlattini (2003). Low-T3 Syndrome: A Strong Prognostic Predictor of Death in Patients with Heart Disease.
S Adler,L Wartofsky (2007). The Nonthyroidal Illness Syndrome.
W Simonides,M Mulcahey,E Redout,A Muller,M Zuidwijk,T Visser (2008). Hypoxia-Inducible Factor Induces Local Thyroid Hormone Inactivation During Hypoxic-Ischemic Disease in Rats.
S Huang,M Mulcahey,A Crescenzi,M Chung,B Kim,C Barnes (2005). Transforming Growth Factor-Beta Promotes Inactivation of Extracellular Thyroid Hormones Via Transcriptional Stimulation of Type 3 Iodothyronine Deiodinase.
I Callebaut,C Curcio-Morelli,J Mornon,B Gereben,C Buettner,S Huang (2003). The Iodothyronine Selenodeiodinases Are Thioredoxin-Fold Family Proteins Containing a Glycoside Hydrolase Clan Gh-a-Like Structure.
J Bates,V Spate,J Morris,St Germain,D Galton,V (2000). Effects of Selenium Deficiency on Tissue Selenium Content, Deiodinase Activity, and Thyroid Hormone Economy in the Rat During Development.
M Fraczek-Jucha,K Zbierska-Rubinkiewicz,M Kabat,K Plens,R Rychlak,J Nessler (2019). Low Triiodothyronine Syndrome and Selenium Deficiency -Undervalued Players in Advanced Heart Failure? A Single Center Pilot Study.
S Dong,L De Las Fuentes,A Brown,A Waggoner,G Ewald,Dávila-Román,Vg (2006). N-Terminal Pro B-Type Natriuretic Peptide Levels: Correlation with Echocardiographically Determined Left Ventricular Diastolic Function in an Ambulatory Cohort.
M Coceani,G Iervasi,A Pingitore,C Carpeggiani,L' Abbate,A (2009). Thyroid Hormone and Coronary Artery Disease: From Clinical Correlations to Prognostic Implications.
C Passino,A Pingitore,P Landi,M Fontana,L Zyw,A Clerico (2009). Prognostic Value of Combined Measurement of Brain Natriuretic Peptide and Triiodothyronine in Heart Failure.
R Pfister,N Strack,K Wielckens,G Malchau,E Erdmann,C Schneider (2010). The Relationship and Prognostic Impact of Low-T3 Syndrome and Nt-Pro-Bnp in Cardiovascular Patients.
A Boelen,Platvoet-Ter Schiphorst,M Wiersinga,W (1993). Association between Serum Interleukin-6 and Serum 3,5,3'-Triiodothyronine in Nonthyroidal Illness.
J Brozaitiene,N Mickuviene,A Podlipskyte,J Burkauskas,R Bunevicius (2016). Relationship and Prognostic Importance of Thyroid Hormone and N-Terminal Pro-B-Type Natriuretic Peptide for Patients after Acute Coronary Syndromes: A Longitudinal Observational Study.
H Takahashi,Y Kashiwagi,T Nagoshi,Y Tanaka,Y Oi,H Kimura (2021). Low Triiodothyronine Levels Correlate with High B-Type Natriuretic Peptide Levels in Patients with Heart Failure.
Abdel-Moneim A Gaber,A Gouda,S Osama,A Othman,S Allam,G (2020). Relationship of Thyroid Dysfunction with Cardiovascular Diseases: Updated Review on Heart Failure Progression.
Sara Danzi,Irwin Klein (2002). Thyroid Abnormalities in Heart Failure.
E Biegelmeyer,I Scanagata,L Alves,M Reveilleau,F Schwengber,S Wajner (2021). T3 as Predictor of Mortality in Any Cause Non-Critically Ill Patients.
A Cerillo,S Storti,E Kallushi,D Haxhiademi,A Miceli,M Murzi The Low Triiodothyronine Syndrome: A Strong Predictor of Low Cardiac Output and Death in Patients Undergoing.
F Abdu,A Mohammed,L Liu,W Zhang,G Yin,B Xu (2021). Low Free Triiodothyronine as a Predictor of Poor Prognosis in Patients with Myocardial Infarction with Non-Obstructive Coronary Arteries.
L Langouche,A Jacobs,G Van Den Berghe (2019). Nonthyroidal Illness Syndrome across the Ages.
L' Her,E Sebert,P (2001). A Global Approach to Energy Metabolism in an Experimental Model of Sepsis.
L' Her,E Sebert,P (2004). Effects of Dichloroacetate and Ubiquinone Infusions on Glycolysis Activity and Thermal Sensitivity During Sepsis.
D He,N Li,X Lu,W Li,Y Chen,Z Sun (2022). Association of Mitochondrial Respiratory Chain Enzymes with the Risk and Mortality of Sepsis among Chinese Children.
A Vassiliou,Z Mastora,E Jahaj,C Keskinidou,M Pratikaki,E Kampisiouli (2020). Serum Coenzyme Q10 Levels Are Decreased in Critically-Ill Septic Patients: Results from a Preliminary Study.
S Abitagaoglu,S Akinci,F Saricaoglu,M Akinci,N Zeybek,S Muftuoglu (2015). Effect of Coenzyme Q10 on Organ Damage in Sepsis.
R Soltani,B Alikiaie,F Shafiee,H Amiri,S ; Mousavi,M Fink (2001). Cytopathic Hypoxia. Mitochondrial Dysfunction as Mechanism Contributing to Organ Dysfunction in Sepsis.
D Brealey,M Brand,I Hargreaves,S Heales,J Land,R Smolenski (2002). Association between Mitochondrial Dysfunction and Severity and Outcome of Septic Shock.
M Cornu,Aln Martinuzzi,P Roel,L Sanhueza,M Sepúlveda,M Orozco (2020). Incidence of Low-Triiodothyronine Syndrome in Patients with Septic Shock.
W Snyder,M Cook,E Nasset,L Karhausen,Parry Howells,G Tipton,I (1979). Report of the Task Group on Reference Man.
J Valentin (2002). Basic anatomical and physiological data for use in radiological protection: reference values.
S Tomlinson,S Arnall,A Munn,S Bradshaw,S Maloney,K Dixon (2014). Applications and Implications of Ecological Energetics.
H Pontzer,A Mcgrosky (2022). Balancing Growth, Reproduction, Maintenance, And activity in Evolved Energy Economies.
E Bolund (2020). The Challenge of Measuring Trade-Offs in Human Life History Research.
A Van Noordwijk,Jong De (1986). Acquisition and Allocation of Resources: Their Influence on Variation in Life History Tactics.
G Jasieńska,P Ellison (1998). Physical Work Causes Suppression of Ovarian Function in Women.
R Rahe,M Meyer,M Smith,G Kjaer,T Holmes (1964). Social Stress and Illness Onset.
T Holmes,R Rahe (1967). The Social Readjustment Rating Scale.
D Bauman,W Currie (1980). Partitioning of Nutrients During Pregnancy and Lactation: A Review of Mechanisms Involving Homeostasis and Homeorhesis.
A Mowry,Z Donoviel,A Kavazis,W Hood (2017). Mitochondrial Function and Bioenergetic Trade-Offs During Lactation in the House Mouse (Mus Musculus).
Clair St,A Gibson,J Swart,R Tucker (2018). The Interaction of Psychological and Physiological Homeostatic Drives and Role of General Control Principles in the Regulation of Physiological Systems, Exercise and the Fatigue Process -the Integrative Governor Theory.
K Lavin,P Coen,L Baptista,M Bell,D Drummer,S Harper (2022). State of Knowledge on Molecular Adaptations to Exercise in Humans: Historical Perspectives and Future Directions.
M Brand,A Orr,I Perevoshchikova,C Quinlan (2013). The Role of Mitochondrial Function and Cellular Bioenergetics in Ageing and Disease.
M Liesa,O Shirihai (2013). Mitochondrial Dynamics in the Regulation of Nutrient Utilization and Energy Expenditure.
I Kyriazis,E Vassi,M Alvanou,C Angelakis,Z Skaperda,F Tekos (2022). The Impact of Diet Upon Mitochondrial Physiology (Review).
P Niklowitz,B Brosche-Bockholt,I Dieffenbach,R Dieffenbach,W Andler,M Paulussen (2012). Coenzyme Q10 Concentration in Plasma and Blood Cells of Juvenile Patients Hospitalized for Anorexia Nervosa.
A Loucks,R Callister (1993). Induction and Prevention of Low-T3 Syndrome in Exercising Women.
J Slater,R Brown,R Mclay-Cooke,K Black (2017). Low Energy Availability in Exercising Women: Historical Perspectives and Future Directions.
A Loucks (2003). Energy Availability, Not Body Fatness, Regulates Reproductive Function in Women.
H Cabre,S Moore,A Smith-Ryan,A Hackney (2022). Relative Energy Deficiency in Sport (Red-S): Scientific, Clinical, and Practical Implications for the Female Athlete.
C Bennett,P Latorre-Muro,P Puigserver (2022). Mechanisms of Mitochondrial Respiratory Adaptation.
J Dempsey,P Hanson,K Henderson (1984). Exercise-Induced Arterial Hypoxaemia in Healthy Human Subjects at Sea Level.
C Harms,S Mcclaran,G Nickele,D Pegelow,W Nelson,J Dempsey (1998). Exercise-Induced Arterial Hypoxaemia in Healthy Young Women.
H Ameln,T Gustafsson,C Sundberg,K Okamoto,E Jansson,L Poellinger (2005). Physiological Activation of Hypoxia Inducible Factor-1 in Human Skeletal Muscle.
J Nakase,A Inaki,T Mochizuki,T Toratani,M Kosaka,Y Ohashi (2013). Whole Body Muscle Activity During the Fifa 11+ Program Evaluated by Positron Emission Tomography.
Rikuto Yoshimizu,Junsuke Nakase,Takafumi Mochizuki,Yasushi Takata,Kengo Shimozaki,Kazuki Asai,Kazu Toyooka,Mitsuhiro Kimura,Seigo Kinuya,Hiroyuki Tsuchiya (2022). Whole Body Muscle Activity During Weightlifting Exercise Evaluated By Positron Emission Tomography.
R Zou,C Wu,S Zhang,G Wang,Q Zhang,B Yu (2020). Euthyroid Sick Syndrome in Patients with Covid-19.
M Świstek,M Broncel,P Gorzelak-Pabiś,P Morawski,M Fabiś,E Woźniak (2022). Euthyroid Sick Syndrome as a Prognostic Indicator of Covid-19 Pulmonary Involvement, Associated with Poorer Disease Prognosis and Increased Mortality.
C Sparano,E Zago,A Morettini,C Nozzoli,D Yannas,V Adornato (2022). Euthyroid Sick Syndrome as an Early Surrogate Marker of Poor Outcome in Mild Sars-Cov-2 Disease.
Y Leyfman,T Erick,S Reddy,S Galwankar,Pwb Nanayakkara,Di Somma,S (2020). Potential Immunotherapeutic Targets for Hypoxia Due to Covi-Flu.
L Eades,M Drozd,R Cubbon (2022). Hypoxia Signalling in the Regulation of Innate Immune Training.
S Krishnan,H Nordqvist,A Ambikan,S Gupta,M Sperk,S Svensson-Akusjärvi (2021). Metabolic Perturbation Associated with Covid-19 Disease Severity and Sars-Cov-2 Replication.
P Kiatkittikul,C Promteangtrong,A Kunawudhi,D Siripongsatian,T Siripongboonsitti,P Ruckpanich (2022). Abnormality Pattern of F-18 Fdg Pet Whole Body with Functional Mri Brain in Post-Acute Covid-19.
Serbanescu-Kele Apor De Zalán,Cmc Banwarie,R Banwari,K Panka,B (2022). The Unfriendly Side of "Happy Hypoxaemia": Sudden Cardiac Death.
J Couzin-Frankel (2020). The Mystery of the Pandemic's 'Happy Hypoxia.
N Caputo,R Strayer,R Levitan (2020). Early Self-Proning in Awake, Non-Intubated Patients in the Emergency Department: A Single Ed's Experience During the Covid-19 Pandemic.
A De,M Bansal (2023). Association of Covid-19 with Shortand Long-Term Risk of Cardiovascular Disease and Mortality: A Prospective Cohort in Uk Biobank.
K Hansen,T Mogensen,J Agergaard,B Schiøttz-Christensen,L Østergaard,L Vibholm (2022). High-Dose Coenzyme Q10 Therapy Versus Placebo in Patients with Post Covid-19 Condition: A Randomized, Phase 2, Crossover Trial.
M Ranek,M Stachowski,J Kirk,M Willis (1738). The Role of Heat Shock Proteins and Co-Chaperones in Heart Failure.
M Iguchi,A Littmann,S Chang,L Wester,J Knipper,R Shields (2012). Heat Stress and Cardiovascular, Hormonal, and Heat Shock Proteins in Humans.
A Burk,S Timpmann,K Kreegipuu,M Tamm,E Unt,V Ööpik (2012). Effects of Heat Acclimation on Endurance Capacity and Prolactin Response to Exercise in the Heat.
G Stendig-Lindberg,D Moran,Y ; Shapiro,F Trautinger (1998). Heat Shock Proteins in the Photobiology of Human Skin.
K Richter,M Haslbeck,J Buchner (2010). The Heat Shock Response: Life on the Verge of Death.
E Waters (2014). The Mixed-up Evolutionary History of the Heat-Shock Proteins: Conservative Innovation.
E Laskowska,D Kuczyńska-Wiśnik,B Lipińska (2018). Proteomic Analysis of Protein Homeostasis and Aggregation.
A Bouchama (1995). Heatstroke: A New Look at an Ancient Disease.
A Bouchama,M Aziz,S Mahri,M Gabere,M Dlamy,S Mohammad (2017). A Model of Exposure to Extreme Environmental Heat Uncovers the Human Transcriptome to Heat Stress.
A Barrientos,M Barros,I Valnot,A Rotig,P Rustin,A Tzagoloff (2002). Cytochrome Oxidase in Health and Disease.
S Racinais,S Moussay,D Nichols,G Travers,T Belfekih,Y Schumacher (2018). Core Temperature up to 41.5 °C During the Uci Road Cycling World Championships in the Heat.
P Bi,S Williams,M Loughnan,G Lloyd,A Hansen,T Kjellstrom (2011). The Effects of Extreme Heat on Human Mortality and Morbidity in Australia: Implications for Public Health.
E Roselló-Lletí,J Alonso,R Cortés,L Almenar,L Martínez-Dolz,I Sánchez-Lázaro (2012). Cardiac Protein Changes in Ischaemic and Dilated Cardiomyopathy: A Proteomic Study of Human Left Ventricular Tissue.
D Lu,Y Xia,Z Chen,A Chen,Y Wu,J Jia (2019). Cardiac Proteome Profiling in Ischemic and Dilated Cardiomyopathy Mouse Models.
G Ilievska,S Dinevska-Kjovkarovska,B Miova (2017). Effect of Single and Repeated Heat Stress on Chemical Signals of Heat Shock Response Cascade in the Rat's Heart.
H Chen,X Zhang,Y Cheng,A Abdelnasir,S Tang,N Kemper (2015). Association of Heat Shock Protein 70 Expression with Rat Myocardial Cell Damage During Heat Stress in Vitro and in Vivo.
E Baljinnyam,N Hasebe,M Morihira,K Sumitomo,T Matsusaka,T Fujino (2006). Oral Pretreatment with Ebselen Enhances Heat Shock Protein 72 Expression and Reduces Myocardial Infarct Size.
A Liedtke,H Hughes (1976). Effects of Hyperthermic Stress on Myocardial Function During Experimental Coronary Ischemia.
X Huang,A Liedtke (1989). Alterations in Fatty Acid Oxidation in Ischemic and Reperfused Myocardium.
J Wang,W Chien,P Chu,C Chung,C Lin,S Tsai (2019). The Association between Heat Stroke and Subsequent Cardiovascular Diseases.
F Nzvere,E Tariq,K Nishanth,A Arshid,I Cancarevic (2020). Long-Term Cardiovascular Diseases of Heatstroke: A Delayed Pathophysiology Outcome.
A Bouchama,J Knochel (2002). Heat Stroke.
A Mujahid,K Sato,Y Akiba,M Toyomizu (2006). Acute Heat Stress Stimulates Mitochondrial Superoxide Production in Broiler Skeletal Muscle, Possibly Via Downregulation of Uncoupling Protein Content.
M Kikusato,K Nakamura,Y Mikami,A Mujahid,M Toyomizu (2015). The Suppressive Effect of Dietary Coenzyme Q10 on Mitochondrial Reactive Oxygen Species Production and Oxidative Stress in Chickens Exposed to Heat Stress.
A Emami,A Tofighi,S Asri-Rezaei,B Bazargani-Gilani (2018). The Effect of Short-Term Coenzyme Q10 Supplementation and Pre-Cooling Strategy on Cardiac Damage Markers in Elite Swimmers.
C Downs,S Heckathorn (1998). The Mitochondrial Small Heat-Shock Protein Protects Nadh: Ubiquinone Oxidoreductase of the Electron Transport Chain During Heat Stress in Plants.
A Jagodzińska,A Gondek,B Pietrzak,A Cudnoch-Jędrzejewska,A Mamcarz,M Wielgoś (2017). Peripartum Cardiomyopathy -from Pathogenesis to Treatment.
A Cénac,A Djibo,L Djangnikpo (1993). Peripartum Dilated Cardiomyopathy. A Model of Multifactor Disease?.
J Xu,B Yin,B Huang,S Tang,X Zhang,J Sun (2018). Co-Enzyme Q10 Protects Chicken Hearts from in Vivo Heat Stress Via Inducing Hsf1 Binding Activity and Hsp70 Expression.
D Zahger,A Moses,A Weiss (1989). Evidence of Prolonged Myocardial Dysfunction in Heat Stroke.
M Marchand,K Gin (2022). The Cardiovascular System in Heat Stroke.
S Liu,L Xing,J Wang,T Xin,H Mao,J Zhao (2022). The Relationship between 24-Hour Indicators and Mortality in Patients with Exertional Heat Stroke.
A Bouchama,B Abuyassin,C Lehe,O Laitano,O Jay,O' Connor,F (2022). Classic and Exertional Heatstroke.
H Mclennan,Degli Esposti,M (2000). The Contribution of Mitochondrial Respiratory Complexes to the Production of Reactive Oxygen Species.
K Duberley,A Abramov,A Chalasani,S Heales,S Rahman,I Hargreaves (2012). Human Neuronal Coenzyme Q10 Deficiency Results in Global Loss of Mitochondrial Respiratory Chain Activity, Increased Mitochondrial Oxidative Stress and Reversal of Atp Synthase Activity: Implications for Pathogenesis and Treatment.
A Galkin,A Abramov,N Frakich,M Duchen,S Moncada (2009). Lack of Oxygen Deactivates Mitochondrial Complex I: Implications for Ischemic Injury?.
H Tsukada,S Nishiyama,D Fukumoto,M Kanazawa,N Harada (2014). Novel Pet Probes 18f-Bcpp-Ef and 18f-Bcpp-Bf for Mitochondrial Complex I: A Pet Study in Comparison with 18f-Bms-747158-02 in Rat Brain.
K Yoshinaga,N Tamaki (2007). Imaging Myocardial Metabolism.
V Dilsizian,T Bateman,S Bergmann,Des Prez,R Magram,M Goodbody,A (2005). Metabolic Imaging with Β-Methyl-P-[(123)I]-Iodophenyl-Pentadecanoic Acid Identifies Ischemic Memory after Demand Ischemia.
M Morishima,T Kiriyama,Y Miyagi,T Otsuka,Y Fukushima,S Kumita (2021). Serial Change in Perfusion-Metabolism Mismatch after Coronary Artery Bypass Grafting.
K Yoshinaga,M Naya,T Shiga,E Suzuki,N Tamaki (2013). Ischaemic Memory Imaging Using Metabolic Radiopharmaceuticals: Overview of Clinical Settings and Ongoing Investigations.
K Yoshinaga,N Tamaki (2015). Current Status of Nuclear Cardiology in Japan: Ongoing Efforts to Improve Clinical Standards and to Establish Evidence.
M Yamagishi,N Tamaki,T Akasaka,T Ikeda,K Ueshima,S Uemura (2021). Jcs 2018 Guideline on Diagnosis of Chronic Coronary Heart Diseases.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Roy Moncayo. 2026. \u201cFrom the Thyroid to the Heart\u201d. Global Journal of Medical Research - K: Interdisciplinary GJMR-K Volume 23 (GJMR Volume 23 Issue K1): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
The pathogenesis of human disease is commonly considered to be unique to each organ or system. This has led to specialization in medical practice leaving little space for global pathogenesis concepts. In this review we have departed from a biochemical concept of coenzyme Q 10 (CoQ 10 ) deficiency and its relation to the initiation of hypoxia response in a systemic way. Several conditions of secondary CoQ 10 deficiency are known in the literature by which the organs involved could be affected by hypoxia and switch to glycolytic metabolism. The most salient biomarkers of this situation are the low T3 syndrome and the elevation of IL-6. These parameters together with CoQ 10 deficiency delineate a condition of acquired mitochondrial dysfunction. Additional related biochemical deficiency conditions affect magnesium, selenium, and iron levels. Visualization of glycolysis can be clearly achieved by diagnostic imaging methods based on the use of 18 F-fluoro-deoxyglucose ( 18 F-FDG). We present several examples of diagnostic imaging with 18 F-FDG to demonstrate our model of acquired mitochondrial dysfunction and disease.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.