This study aims to explore the green technology strategies for the preparation of fluoxetine, through the research on the greenization of catalysts and membrane treatment of wastewater. The study show that green technology strategies can effectively reduce the environmental pollution and resource consumption during the preparation of fluoxetine. At the same time, the integration and prospects of green technology strategies were analyzed, and its potential value and broad application prospects were provided in the preparation of fluoxetine. This research is of great significance for promoting the development of fluoxetine preparation processes towards a green, environmentally friendly, high efficiency and sustainable development.
## I. INTRODUCTION
Fluoxetine, N-methyl-3-phenyl-3-[4-(trifluoromethyl) phenoxy] propylamine hydrochloride, Prozac®, is a new generation of non-tricyclic antidepressants developed by Eli Lilly Company, the structure is shown in Figure 1. Fluoxetine contains a chiral center with a pair of enantiomers, namely (R)-and (s)-fluoxetine. Most of fluoxetine hydrochloride sold on the market is racemate. The main effects of (R)-fluoxetine is the treatment of depression, but (s)-fluoxetine is the prevention of migraine. [1,2] a clinical study of chiral (R)- or (s)-fluoxetine by SEPRACOR Inc. found that (R)-fluoxetine had a shorter half-life and action time than commercially available racemic fluoxetine, it can greatly reduce the adverse side effects of racemic fluoxetine, such as headache, anxiety and suicidal impulse. Because of its high selectivity, safety and bioavailability, it is widely used in clinical practice and is one of the essential drugs listed by the World Health Organization[3-5].
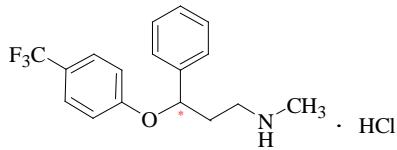 Fig. 1: Molecule Structure of Fluoxetine Panel label: Author.
$\alpha$ $\sigma$: Henan Medical College, Henan Xinzheng, 451191, China.
## II. GREEN TECHNOLOGY STRATEGY FOR PREPARATION OF FLUOXETINE
# a) The Selection and Utilization of Raw Materials
The selection and utilization of raw materials are crucial to green technology strategy during the preparation process in fluoxetine. First, we need to consider the choice of renewable, biodegradable raw materials, in order to reduce environmental pollution and consumption of resources. Second, the use of green synthetic pathways to avoid the use of toxic or refractory compounds to reduce environmental and human risks. In addition, the utilization rate of raw materials can be increased and the generation of wastes can be reduced by improving the synthesis process. Through the greening of raw materials, the environmental load in the preparation process of fluoxetine can be effectively reduced to achieve the goal of green and sustainable development. The fluoxetine steps used in the past are shown in Figure 2. $\mathrm{KBH}_4$ is used in this process and has a corrosive effect on the skin and mucous membranes and is usually slightly harmful to water. DMSO, also known as Dimethyl sulfoxide, interacts with the hydrophobic groups of proteins and causes Denaturation, which is toxic to human skin and irritating to the eyes. According to the guiding principles of chemical drug residue solvent and 12 principles of green chemistry, so should minimize or avoid the side-use of such solvents.
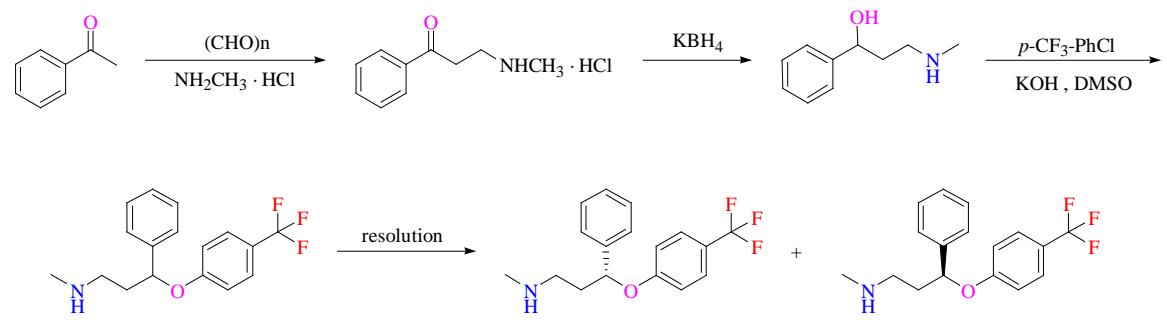 Fig. 2: Preparation Steps of Fluoxetine Hydrochloride
### b) The Green Solvent
The solvent plays a crucial role in the preparation of fluoxetine, and the choice of the solvent is directly related to the environmental friendliness and energy consumption of the preparation process. Therefore, in the green technology strategy, we need to consider the choice of low-toxic, non-volatile organic solvents or water solvents to reduce the harm to the environment and human body. In addition, the emission and consumption of solvents can be reduced by optimizing the amount of solvents used and recycling. In order to make the preparation of fluoxetine green, we should consider the factors of toxicity, volatility, price and reproducibility, and choose the most suitable solvent. In order to further improve the green degree of fluoxetine preparation process, we can consider the implementation of solvent recycling strategy. The recovery and reuse of waste solvents by physical or chemical methods can reduce the consumption of solvents and the discharge of wastes, and reduce the demand of resources in the preparation process. In addition, the idea of circular economy can be introduced to reuse the waste solvent as a resource and realize the closed-loop utilization of the solvent to further improve the sustainability and environmental protection of the preparation process of fluoxetine. The environmental pollution and resource consumption in the preparation of fluoxetine can be reduced by recycling the solvent, and the green technology strategy can be realized.
### c) The Green Catalysts of Fluoxetine
There are many methods for the synthesis and resolution of the green catalysts of fluoxetine. Most of the (R)-fluoxetine currently used in the market are obtained by chiral resolution, a large number of chiral reagents are required and the whole process is time-consuming and laborious. Asymmetric catalytic synthesis has great Atom economy advantages over chiral resolution. Asymmetric catalytic synthesis has become a hot research field in the field of organic chemistry. In particular, many chemical companies are developing asymmetric catalytic reactions into chiral and Enantioselective synthesis processes, such as L-Dopa synthesis from L-Dopa catalyzed by chiral rhodium in American Monsanto, and (-)-menthol synthesis from asymmetric rearrangement catalyzed by rhodium in Japanese Enantioselective synthesis company, and developed into a promising industrial chiral technology.
In a pioneering study [2], Duan Yu and colleagues embarked on a chemical journey, initiating their work with cinnamaldehyde, ultimately shaping it into chiral fluoxetine via an intricate sequence of reactions. They navigated through asymmetric epoxidation of chiral secondary amines, ring-opening to ester, Ester Exchange, reduction, and the transformative etherification of ester amines.
Chengyu Liu, on another scientific expedition [3], relied on the prowess of an enantioselective catalyst to forge an (R)-fluoxetine intermediate. Wang Jiahao, with deft precision, synthesized $\gamma$ -amino alcohol, a beacon of optical purity, by way of enantioselective synthesis of the prochiral $\beta$ -amino ketone [4]. This pivotal compound then paved the way for the synthesis of fluoxetine.
Li Zhen-zhong, employing the [RUCL2((s)-BINAP)((s, S)-dpen)] complex as a catalyst [5], skillfully constructed a photoactive fluoxetine intermediate, (R)-N, n-dimethyl-2-hydroxyamphetamine. This masterpiece was then honed into the radiant (R)-fluoxetine, culminating in an impressive overall yield of $49.1\%$.
Kinetic resolution, a technique that discriminates enantiomers via differential rates of reaction with chiral reagents or catalysts, was employed to synthesize fluoxetine. Researchers, through the hydrolysis kinetic resolution of racemic epoxide catalyzed by Salen Co (III) chiral catalyst, sculpted the elusive chiral r-phenyl ethylene oxide [6]. A series of transformations then led to the birth of the coveted fluoxetine drug. The allure of kinetic resolution lies in its simplicity and efficiency, with the capacity to enhance excess enantiomers by adjusting conversion rates. However, it demands an extra step to resolve nontargeted stereoisomers.
Xiang Peng [7] ventured into uncharted territories, revealing a novel path for (S)-fluoxetine synthesis, harnessing chiral secondary alcohols, bearing $\alpha$ -position substituents, as pivotal intermediates. Cheng Qingfang and collaborators took an innovative approach [8,9], deploying their homemade chiral oxazaborolane as a catalyst to achieve enantioselective hydrogenation of $\beta$ -chlorophenylacetone, yielding (S)- or (R)-chiral alcohols, subsequently steering the synthesis of (S)- and (R)-fluoxetine. This breakthrough marked another milestone in the evolving landscape of fluoxetine synthesis.
Table 1: Methods and Conditions for Preparation of Fluoxetine
<table><tr><td>[1]</td><td>Starting Material</td><td>Methods</td><td>Synthetic Products</td><td>The End Product</td></tr><tr><td>[2]</td><td>Cinnamaldehyde</td><td>Chiral Enantioselective synthesis</td><td>Chiral fluoxetine</td><td></td></tr><tr><td>[3]</td><td></td><td>Iridium catalyst Enantioselective synthesis</td><td>(R)-Fluoxetine Intermediate</td><td></td></tr><tr><td>[4]</td><td>Latent Chiral β-aminophenone</td><td>Enantioselective synthesis</td><td>The intermediate γ-amino alcohol</td><td>fluoxetine</td></tr><tr><td>[5]</td><td>N, n-dimethyl-1-phenylacetone</td><td>[RuCl2 (S) -BINAP](S,S)-DPEN), The complex is the catalyst</td><td>The fluoxetine intermediate (R) -- N, N-dimethyl-2-hydroxyamphetamine</td><td>fluoxetine</td></tr><tr><td>[6]</td><td>Racemic Epoxide</td><td>Hydrolysis kinetic resolution of Salen Co (III) chiral catalyst</td><td>Chiral intermediate r-phenyl ethylene oxide</td><td>Chiral fluoxetine</td></tr><tr><td>[7]</td><td>Chiral secondary alcohols containing substituent groups at α-position</td><td>Kinetic resolution of Stereoselectivity synthesis catalyzed by organometallic catalysts and biological enzymes</td><td></td><td>(S)-fluoxetine</td></tr><tr><td>[8]</td><td>B-chlorophenone</td><td>Asymmetric catalytic hydrogenation reduction</td><td>(s)-or (R)-chiral alcohols</td><td>Enantioselective synthesis (s)-and (R)-fluoxetine</td></tr><tr><td>[9]</td><td>Latent chiral ketone</td><td>Asymmetric reduction of ketone reductase</td><td>Intermediate chiral alcohols</td><td>Fluoxetine drugs</td></tr></table>
The strategy of using natural chiral sources to synthesize some complex chiral compounds is not only helpful for configuration retention, but also suitable for the synthesis of new chiral compounds by configuration transformation and other organic reactions.
## i. The Selection and Design of Catalysts
The pivotal role of catalyst selection and design emerges as a cornerstone within the green methodology for crafting fluoxetine. An astute choice of catalysts can significantly boost reaction proficiency, decrease energy utilization, and mitigate environmental implications. Hence, a comprehensive exploration of catalyst selection and design is imperatively advocated.
Primarily, the catalytic prowess and specificity of the catalyst should be weighed, assuring its proficient performance in fluoxetine synthesis. Subsequently, the catalyst's architecture should be ingeniously devised for enhanced durability and recycling efficiency. Adapting to distinct reaction scenarios and catalytic mechanisms, surface modification and functional treatments can also be employed to fine-tune their attributes.
Through meticulous experimental trials and theoretical scrutiny, a bevy of proficient and eco-friendly catalysts have been ingeniously crafted and sifted, thereby fostering a robust foundation for the sustainable synthesis of fluoxetine. Of particular interest is the selective production of the two enantiomers of fluoxetine, an area drawing substantial attention.
Corey's innovation was instrumental in enhancing the catalyst, enabling the facile acquisition of both enantiomers and augmenting reaction efficacy. Sharpless, on the other hand, unfolded a novel path for synthesizing both enantiomers, with the pivotal step being Sharpless epoxidation. This synthetic pathway, depicted in Figure 3, exhibits practicality and boasts a total yield of $49\%$. [7] In this discourse, the intricate dance between catalyst choice, design, and the quest for greener fluoxetine synthesis is portrayed, with key milestones and advancements underpinning the journey.
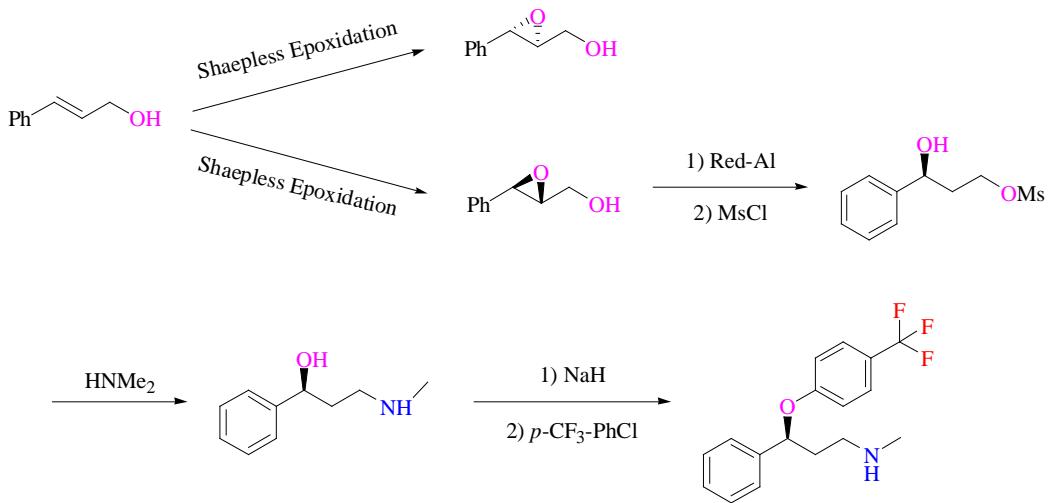 Fig. 3: Sharpless Synthetic Route of Fluoxetine
## ii. The Revitalization of Catalysts: A Pillar of Eco-Technology
- The resurrection of catalysts occupies a pivotal role in the eco-friendly technology roadmap. In conventional fluoxetine synthesis, catalysts are customarily disposed after a solitary application, resulting in both resource squandering and environmental devastation. Consequently, a profound exploration of catalyst recycling emerges as indispensable[8,9]. Harnessing surface engineering and rejuvenation methodologies, catalysts can undergo multiple reincarnations, significantly curbing waste management volumes and minimizing environmental footprints. Moreover, extensive studies on catalyst deactivation during recycling have exposed a plethora of proficient regeneration techniques, thereby enhancing the catalyst's longevity and minimizing the energy expenditure and production costs of fluoxetine. iii. The Sustainable Management of Catalyst Waste: An Integral Green Strategy
Catalyst waste disposal stands as a crucial facet in the holistic green technology approach. Throughout the catalyst's operational cycle, waste production is an inescapable byproduct, posing severe environmental hazards if not dealt with cautiously. Thus, extensive investigations into waste catalyst management are paramount. Techniques such as physical adsorption, chemical reduction, and pyrolysis, known for their energy efficiency, are employed to convert waste catalysts into reusable resources and render them environmentally benign. Simultaneously, a comprehensive experimental and simulation-based assessment of various waste treatment methodologies enables a comparative evaluation of their environmental impact. This generates scientifically grounded and feasible strategies for waste catalyst management, thereby offering substantial technical backing for the environmentally sustainable synthesis of fluoxetine.
### d) The Innovative Approach: Membrane-Driven Wastewater Management
## i. Embracing the Potential of Membrane Separation Techniques
As an avant-garde and ecologically conscientious technique, membrane separation has carved a niche for itself across diverse sectors. [10-15] In the intricate process of manufacturing fluoxetine, the wastewater generated is replete with an array of organic compounds and impurities, necessitating the judicious deployment of membrane separation. This advanced technology leverages selective membrane materials and tailored operational parameters to proficiently sieve out organic contaminants, ions, and microorganisms from the aqueous milieu.
A gamut of membrane separation methodologies exists, including microfiltration, ultrafiltration, nanofiltration, and reverse osmosis. Each method possesses unique attributes, allowing for tailored solutions based on the distinctive traits of the wastewater at hand, thereby optimizing the purification process and facilitating water reuse.
The integration of membrane separation not only ensures the efficient eradication of impurities and organic compounds but also facilitates meticulous water quality control, thereby fulfilling the stringent purity criteria in fluoxetine synthesis. Moreover, this technology curbs chemical reliance, decreases energy consumption, and lightens the financial burden of wastewater management, thereby delivering substantial economic and environmental dividends.
In essence, the strategic utilization of membrane separation technology in wastewater management significantly enhances the environmental footprint of fluoxetine production. It propels the manufacturing process towards a greener, more sustainable trajectory, fostering a harmonious coexistence between industry and the environment. Thus, the marriage of membrane separation with wastewater treatment stands as a testament to innovation and responsible stewardship of our planet's resources.
## ii. Enhancing the efficacy of membrane-based wastewater treatment methodology
The pivotal step in accomplishing proficient wastewater management and boosting resource recovery lies in refining the wastewater membrane treatment process. While formulating fluoxetine, the industrial effluent exhibits a substantial concentration of organic compounds, posing significant hurdles for the treatment process. Consequently, it is imperative to tailor the membrane treatment strategy, considering the unique characteristics of the wastewater, to enhance purification efficiency and elongate the membrane's operational lifespan.
The initial stage of improvement revolves around optimizing the preliminary wastewater treatment, encompassing pH balancing, suspended solids and sediment removal. These measures mitigate membrane fouling and damage, thereby fortifying membrane stability and service duration.
Subsequently, the membrane material and operational parameters are meticulously fine-tuned. A membrane material particularly suited for wastewater treatment is selected, and operational conditions are adeptly regulated, thereby enhancing the membrane's capacity to intercept fouling and resist contamination.
Ultimately, the integration of sophisticated automation technologies brings about online monitoring and intelligent control of the membrane treatment workflow. This ensures the seamless functioning of the wastewater management system and sustains a high level of treatment efficacy.
Through the meticulous optimization of the wastewater membrane treatment process, the efficacious extraction of organic substances and impurities from the wastewater is realized, contributing to an extended membrane lifespan, lowered treatment costs, and heightened resource recovery efficiency. This, in turn, furnishes vital technical backing for the sustainable advancement of the fluoxetine synthesis process, fostering green development.
## iii. The Recycled Symphony of Membrane-Treated Effluent
The intricate dance of elements, both organic and ionic, within the aqueous remnants of membrane filtration, has been skillfully harnessed to comply with stringent water quality benchmarks. This transformative process not only ensures the elimination of impurities but also reshapes waste into a precious commodity, fostering a virtuous cycle of zero discharge and wastewater reuse.
In the intricate ballet of fluoxetine synthesis, a residue of fluoxetine and organic solvent lingers in the water trail. However, through the alchemy of membrane treatment, this detritus is transmuted into a treasure trove of high-purity fluoxetine and solvent, ripe for reincarnation within the production cycle, enhancing efficiency and sustainability.
Beyond industrial rejuvenation, the reclaimed water, having undergone its metamorphosis, can grace the fields as a life-giving elixir for agriculture, or cool the industrious engines as efficient coolant. Thus, maximizing resource utilization and maintaining a watertight seal on wastewater discharge.
This innovative utilization of wastewater not only shields natural water sources from excessive -extraction and contamination but also alleviates the ecological toll of waste management, contributing to energy conservation and diminished emissions. The marriage of membrane treatment and wastewater reuse embodies a union of environmental stewardship and economic prudence, serving as a pivotal cornerstone in the management of fluoxetine waste streams.
As the tide of membrane technology rises, the embrace of wastewater recycling is poised to evolve into an indispensable pillar of the fluoxetine manufacturing process. It extends a sturdy arm of support to the industry's pursuit of sustainable development, fostering a future where progress and preservation intertwine in a harmonious symphony.
## III. INTEGRATION OF GREEN TECHNOLOGY STRATEGIES AND PROSPECTS
### a) Integration of Green Technology Strategies
In the preparation of fluoxetine, the integration of green technology strategies can effectively reduce the environmental pollution and resource consumption during the preparation of fluoxetine. The key to the integration of green technology strategy lies in the organic combination of each link to achieve the overall optimization of green preparation.
In green technology strategy integration, we need to take into account the synergy between different links to ensure that the effective convergence and complementarity of various technology strategies. For example, the goal of waste reduction and resource recycling in the preparation of fluoxetine can be achieved by optimizing the selection and preparation of catalysts, combined with membrane technology for wastewater treatment.
Green technology strategy integration needs to consider not only the feasibility and benefits of the technology itself, but also its practical application in industrial production to ensure the Operability and economy of the technology strategy. Through the integration of green technology strategy, we can optimize the preparation process of fluoxetine and improve the environmental performance, providing strong support for the green development of the preparation process of fluoxetine.
### b) Green Evaluation of Fluoxetine Preparation Process
The green preparation process of fluoxetine need to conduct a comprehensive evaluation and analysis. First of all, the selection of raw materials, production processes, waste emissions and energy consumption can be evaluated to fully understand the current process of environmental and resource problems. Secondly, suggestions can be made for the existing problems, such as using green catalysts, optimizing reaction conditions, improving wastewater treatment technology. The green evaluation needs to fully consider the Operability and economics of fluoxetine preparation process in actual production, and ensure the implementation of green technology strategy can meet the requirements of industrial production. In addition, a comprehensive assessment of the green process is needed to ensure that significant improvements can be made in environmental protection, resource utilization and economic benefits. The green evaluation of the preparation process of fluoxetine can provide scientific basis for the implementation of green technology strategy and provide important support for the sustainable development of the preparation process of fluoxetine.
## IV. PROSPECT AND CONCLUSION
Green technology has broad development prospect in pharmaceutical industry. With the society's attention to environmental protection and sustainable development, the application of green technology in pharmaceutical industry will be further promoted and deepened. Especially in the process of drug preparation, green technology can effectively reduce waste emissions and resource consumption, and provide important support for the sustainable development of pharmaceutical industry.
In the future, the application of green technology in pharmaceutical industry will be more extensive. For example, green catalyst, waste water membrane treatment technology, green solvent and so on will become the important technological means in the pharmaceutical process, providing more environmental protection and efficient solution for the pharmaceutical preparation process. At the same time, the continuous innovation and development of green technology will inject new vitality into the pharmaceutical industry, driving the entire industry towards green, environmental protection, sustainable direction.
Considering the development prospect of green technology in pharmaceutical industry, we can foresee that green technology will become an important direction of pharmaceutical industry in the future and inject new impetus into the sustainable development of the industry.
Conflicts of interest
There are no conflicts to declare.
Generating HTML Viewer...
References
15 Cites in Article
Zhao Mingrui,Peng Peng (2023). Thermodynamic Parameters of Fluoxetine Estimated by Group Contribution Method.
Chengyu Liu,Lei Zhang,Liming Cao,Yan Xiong,Yueyue Ma,Ruihua Cheng,Jinxing Ye (2022). Iridium-catalyzed enantioselective synthesis of chiral γ-amino alcohols and intermediates of (S)-duloxetine, (R)-fluoxetine, and (R)-atomoxetine.
Wong Jiahao (2013). Studies on the asymmetric catalytic synthesis of antidepressant psychoactive drug fluoxetine and its analogues.
Li Zhenzhong,Xu Baihong,He Shanzhen (2009). Antidepressant (R)-Enantioselective synthesis of fluoxetine [J].
Yang Runsheng (2007). Chiral Hypervalent Iodine(III) Catalyst Promotes Highly Enantioselective Sulfonyl- and Phosphoryl-oxylactonizations.
Xiang Peng (2012). Chiral Thiourea Catalysts Containing Fluxional Groups: Synthesis of Fluorinated Chiral Building Blocks.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Zhao Mingrui. 2026. \u201cGreen Technology Strategy For Fluoxetine Preparation\u201d. Global Journal of Medical Research - B: Pharma, Drug Discovery, Toxicology & Medicine GJMR-B Volume 24 (GJMR Volume 24 Issue B2): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
This study aims to explore the green technology strategies for the preparation of fluoxetine, through the research on the greenization of catalysts and membrane treatment of wastewater. The study show that green technology strategies can effectively reduce the environmental pollution and resource consumption during the preparation of fluoxetine. At the same time, the integration and prospects of green technology strategies were analyzed, and its potential value and broad application prospects were provided in the preparation of fluoxetine. This research is of great significance for promoting the development of fluoxetine preparation processes towards a green, environmentally friendly, high efficiency and sustainable development.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.