The present study sought to statistically relate the histological origin and anaplastic behavior of neoplasms diagnosed in the Pathology Unit of the Faculty of Veterinary Medicine and Zootechnics of the University of San Carlos of Guatemala in the years 2015 to 2019, as well as associate race, age, sex and affected body system. A total of 1,162 canine samples were diagnosed, and a subsample of 289 cases was determined by systematic stratified random sampling of proportional allocation of each year. Regarding histological origin, 44% were epithelial neoplasms, 29% connective and derivatives, 12% other types, 4% endothelial, and 1% nervous. Concerning the anaplastic behavior, 51% of neoplasms were benign and 49% malignant. Likewise, it was determined that the neoplasms affected mostly females, pure breeds, canines of 7-10 years, and affected the integumentary system and soft tissues.
## I. INTRODUCTION
In recent decades, special interest in oncology has been observed in veterinary medicine. The presentation of neoplasms in wild animals is not common, due to the shorter life expectancy, which is why knowledge of oncology in non-domestic animals is scarce. Regarding domestic animals, the dog is of particular interest, since this species is more frequently affected by tumors, due to the fact that neoplasms are among the most common causes of death in dogs $^{5,9}$.
Previous studies point out the importance of comparative medical research in this species that share the environment intimately with humans, since it often presents similar risk factors for the development of spontaneous neoplasms that have similar biological behavior and equivalent histological characteristics to the neoplasms seen in the human being. In many aspects, neoplasms in canines can be compared with the biological differences associated with neoplasms in humans, in addition to their close relationship with them concerning housing, environment and diet $^{1,3,18,20}$.
On the other hand, authors indicate that the presence of tumors in canines can be explained by improvements in their health care, better nutrition, immunizations, parasite control, use of antibiotics, etc., which make companion animals live longer, and therefore they are exposed to carcinogens for a longer time and exceed an age in which the cells reach the stage of tumor promotion and progression. For this reason, neoplasms are diagnosed with great efficiency and treated more frequently compared to previous years $^{9,18}$.
There is abundant clinical information on diagnosis and therapies for the distinct types of neoplasms, and some studies on risk factors. In an ideal situation, all neoplasms occurring in each population should be microscopically confirmed, either by histopathology or cytology, to identify and enumerate all individuals in the population at risk. Veterinary oncology is a young and growing specialty, and it does not have the resources for studies and treatments that human cancer has. In many cases, the patient is euthanized as soon as there is a presumptive diagnosis of cancer, although it is known that many patients with an early and correct diagnosis can be treated successfully. Likewise, there is limited information on the frequency, prevalence, and incidence of the distinct types of cancer in the canine population in many countries of the world, and Guatemala is no exception $^{4,6,19}$.
Different authors assert that the general benefit of these epidemiological studies is not only aimed at better understanding the epidemiology of canine tumors, but also at detecting and understanding future environmental hazards in the development of these diseases when comparing the situation of dogs with humans. Most epidemiological studies of neoplasms in canines in recent years have used canine populations derived from reference university veterinary hospitals[15,20,22].
In Guatemala, the last study conducted on neoplasms diagnosed through biopsies and necropsies was from 2012 to 2014. Despite the interest in the subject, no new epidemiological research has updated it, so the behavior of the disease has likely changed over the years due to the concern of the owners, and because of detection methods and greater diagnostic efficiency in veterinary practice[10]. The present study seeks to contribute to the knowledge of canine neoplasms in the country, through the analysis of data from canine samples sent in the years 2015 to 2019 to the Pathology Unit at the Faculty of Veterinary Medicine and Zootechnics of the University of San Carlos of Guatemala, a reference center for veterinary histopathological diagnosis in the country; and its objective is to determine and statistically relate the histological origin and anaplastic behavior of diagnosed neoplasms, as well as to associate with the patient's race, age, sex and affected body system.
## II. RESOURCES AND TECHNIQUES
A longitudinal study was conducted using canine biopsy and necropsy samples that turned out to be neoplasms sent to the veterinary histopathology and necropsy service provided by the Veterinary Pathology Unit at the Faculty of Veterinary Medicine and Zootechnics (FMVZ) of the University of San Carlos of Guatemala (USAC) in the period from 2015 to 2019. The diagnoses were made by experienced veterinary pathologists who performed a histopathological evaluation of samples stained with standard hematoxylin-eosin.
During that period, 1,162 patients were diagnosed with neoplasms. According to the total population, a probabilistic sample was calculated with the following equation when the size of the population is known:
$$
n = \frac {N}{1 + \frac {d ^ {2} (N - 1)}{z ^ {2} s ^ {2}}} n = \frac {1 1 6 2}{1 + \frac {(0 . 0 5) ^ {2} (1 1 6 1)}{(1 . 9 6) ^ {2} (0 . 2 5)}} n = 2 8 9
$$
n = size of the sample that you want to know
N = known population size d = deviation or error that is willing to tolerate, expressed in proportion. In this case 5% or 0.05.
$s^2 =$ population variance in the variable to be measured. In this case we have that $s^2 = pq = (0.50)(0.50) = 0.25$
The calculated size of the sample (289) was subjected to a stratified random sampling of proportional allocation. Subsequently, the subsample corresponding to each year was subjected to a systematic random sampling with which the specimens that made up each specific sample of each year were identified. The investigation focused on the neoplasm in each canine, and if multiple neoplasms were found in one animal, they were all addressed separately. However, biologically identical neoplasms located in the same animal at different anatomical sites were considered a single neoplasm.
For data collection purposes, a form or instrument was used for each canine, in which the data corresponding to the histological origin of the neoplasm (connective tissue and derivatives, endothelial, hematopoietic or lymphoreticular, epithelial, nervous, and others) and the anaplastic behavior were recorded (benign or malignant) according to the records of the pathology unit.
Additionally, general data on sex (female or male), race (pure or mongrel), age (<1 year, 1-6 years, 7-10 years, or >10 years), as well as the patient's affected body system was recorded (integumentary system and soft tissues, gastrointestinal, respiratory, urogenital, hemolymphatic, endocrine, nervous, cardiovascular, mammary gland, musculoskeletal or multicentric). Given the symmetric relationship between the variables to be observed and measured, and the nominal or frequency data they provide, the type of research is correlational, with which Cramér's V coefficient was used to indicate the association index between said study variables or categorical fields.
This work was approved by the Postgraduate Bioethics Committee of the University of San Carlos of Guatemala on October 19, 2021 (Ref. EPVirtual. 190.2021).
## III. RESULTS
A total of 1,162 samples of neoplasms from canine biopsies and autopsies were diagnosed during the years 2015-2019 in the FMVZ-USAC Pathology Unit, of which a subsample of 289 cases of neoplasm was determined through a stratified random sampling systematic proportional allocation of each year.
Concerning the histological origin, 128 (44%) were epithelial-type neoplasms, 84 (29%) connective and derivatives, 35 (12%) other types of neoplasms, being mostly mixed type, 12 (4%) endothelial, and 2 (1%) of the nervous type (Table 1). About anaplastic behavior, 146 (51%) neoplasms were benign, and 43 (49%) were malignant (Table 2).
Females presented a higher frequency of neoplasms, with a distribution of 172 (60%) females, compared to 117 (40%) males. Regarding the histological origin, both in males and females, there was a higher frequency of epithelial-type neoplasms (44 and 45% respectively), followed by connective tissue and derivatives (27 and 30%), others (11 and 13%), hematopoietic or lymphoreticular (12 and 8%), endothelial (5 and 3%) and nervous tissue (1%). In relation to anaplastic behavior,
Table 1: Number of canines according to histological origin of neoplasms
<table><tr><td>Histological origin</td><td>N°</td><td>%</td></tr><tr><td>Connective and derivatives</td><td>84</td><td>29</td></tr><tr><td>Endothelial</td><td>12</td><td>4</td></tr><tr><td>Hematopoietic or lymphoreticular</td><td>28</td><td>10</td></tr><tr><td>Epithelial</td><td>128</td><td>44</td></tr><tr><td>Nervous</td><td>2</td><td>1</td></tr><tr><td>Others</td><td>35</td><td>12</td></tr><tr><td>Total</td><td>289</td><td>100</td></tr></table>
Table 2: Number of canines according to anaplastic behavior of neoplasms.
<table><tr><td colspan="2">Benign</td><td colspan="2">Malignant</td><td colspan="2">Total</td></tr><tr><td>N°</td><td>%</td><td>N°</td><td>%</td><td>N°</td><td>%</td></tr><tr><td>146</td><td>51</td><td>143</td><td>49</td><td>289</td><td>100</td></tr></table>
53 (36%) male canines and 93 (64%) females presented benign neoplasms, while 62 (43%) males and 81 (57%) females presented malignant neoplasms.
Purebred canines presented a higher frequency of neoplasms, being 225 (78%) cases, compared to 64 (22%) of mongrel canines. Regarding histological origin, 105 (47%) purebred dogs presented epithelial-type neoplasms, 63 (28%) connective and derivative types, 27 (12%) other types of neoplasms, and 19 (8%) hematopoietic or lymphoreticular type. In contrast, 23 (36%) mongrel canines presented epithelial-type neoplasms, 21 (33%) connective and derivative types, 9 (14%) hematopoietic and lymphoreticular types, and 8 (12%) other types of neoplasms. Concerning anaplastic behavior, 110 (75%) purebred canines and 36 (25%) mongrel canines presented benign neoplasms, while 115 (80%) purebred canines and 28 (20%) mongrel canines had malignant neoplasms. Commonly found purebreds were Poodle (12.9%), Schnauzer (11.1%), Boxer (8.4%), English Cocker Spaniel (7.6%), Labrador Retriever (7.1%), Siberian Husky (5.8%), Golden Retriever (5.3%), Dachshund (3.6%), Basset Hound (3.1%) and Chihuahua (3.1%).
Concerning the age, canines between 7-10 years old presented the highest frequency, with 132 (46%) canines, followed by 78 (27%) dogs between 1-6 years, 74 (26%) canines over ten years, and 5 (1%) under one year old. Regarding the histological origin of neoplasms, canines under one year presented a higher frequency of neoplasms of epithelial origin (60%), followed by the endothelial type (20%) and other types (20%). Canines between 1-6 years old showed a higher frequency of neoplasms of connective origin and derivatives (40%), followed by epithelial neoplasms (31%), hematopoietic or lymphoreticular (11%), and others (10%). Canines between 7-10 years old showed a more significant number of cases of epithelial origin (42%), followed by neoplasms of connective origin and derivatives (29%), hematopoietic or lymphoreticular (13%), and others (11%). Very similar were the canines over ten years, where there was a higher percentage of neoplasms of epithelial type (61%), connective and derivatives (20%), other types of histological origin (15%), and hematopoietic or lymphoreticular (3%). Concerning the anaplastic behavior, benign neoplasms were shown in 70 (48%) canines between 7-10 years old, 42 (29%) canines between 1-6 years old, 30 (20%) canines over ten years old, and in 4 (3%) canines less than one year old while malignant neoplasms were present in 62 (43%) canines between 7-10 years, followed by 44 (31%) canines older than ten years, 36 (25%) canines between 1-6 years, and 1 (1%) canines less than one year old.
Regarding the affected body systems, there was a greater number of cases in the integumentary system and soft tissues (33%), followed by the mammary gland (18%), gastrointestinal system (13%), urogenital (12%), musculoskeletal (9%), multicentric (7%), respiratory (3%), hemolymphatic (3%), cardiovascular (1%), and nervous (1%). (Figure 1).
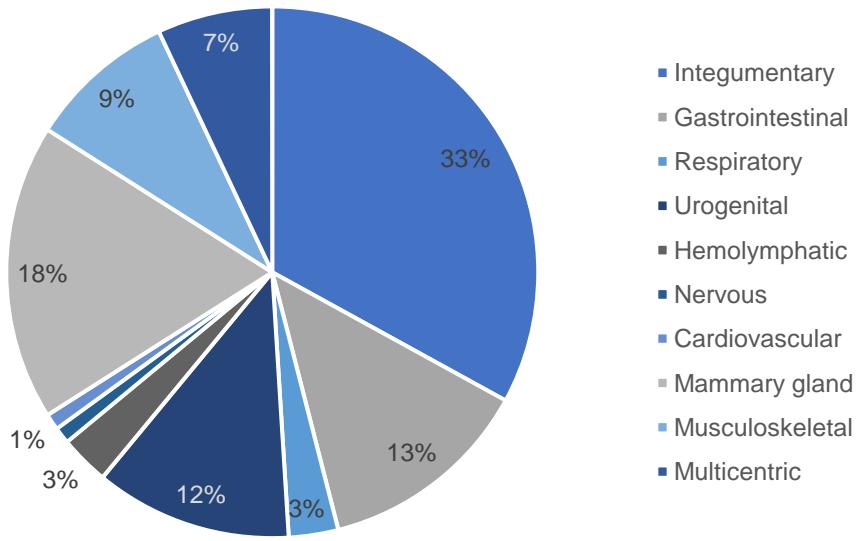 Figure 1: Distribution of canines according to the affected body system.
Concerning the anaplastic behavior according to the affected body systems, the benign neoplasms occurred in the integumentary system and soft tissues, mammary gland, urogenital, musculoskeletal, and gastrointestinal systems; while malignant neoplasms were shown in a higher percentage in the integumentary system and soft tissues, mammary gland, gastrointestinal system, multicentric and urogenital system.
Finally, Cramér's V coefficient was used to indicate the association index between the study variables, where the value was $V = 0.525$, indicating a moderate level of association between the histological origin and the anaplastic behavior of the neoplasms in the observed canine sample (Table 3).
Table 3: Number of canines according to the histological origin and anaplastic behavior of neoplasms
<table><tr><td rowspan="2">Histological origin</td><td colspan="2">Benign</td><td colspan="2">Malignant</td><td colspan="2">Total</td></tr><tr><td>N°</td><td>%</td><td>N°</td><td>%</td><td>N°</td><td>%</td></tr><tr><td>Connective and derivatives</td><td>68</td><td>47</td><td>16</td><td>11</td><td>84</td><td>29</td></tr><tr><td>Endothelial</td><td>10</td><td>7</td><td>2</td><td>1</td><td>12</td><td>4</td></tr><tr><td>Hematopoyetic or lymphoreticular</td><td>2</td><td>1</td><td>26</td><td>18</td><td>28</td><td>10</td></tr><tr><td>Epithelial</td><td>41</td><td>28</td><td>87</td><td>61</td><td>128</td><td>44</td></tr><tr><td>Nervous</td><td>2</td><td>1</td><td>0</td><td>0</td><td>2</td><td>1</td></tr><tr><td>Others</td><td>23</td><td>16</td><td>12</td><td>9</td><td>35</td><td>12</td></tr><tr><td>Total</td><td>146</td><td>100</td><td>143</td><td>100</td><td>289</td><td>100</td></tr></table>
## IV. DISCUSSION
The characterization of a population at risk is essential to understand the epidemiology of a disease. In recent decades there has been a growing interest in comparative oncology, due to the progressive understanding of environmental biological monitoring. In domestic animals such as dogs, there is a high manifestation of tumors, probably because they are allowed to reach old age, as well as their close relationship with humans regarding the environment and food. The analysis of neoplasms in a population based on their histological origin and anaplastic behavior, in addition to their association with characteristics of the patient's data, can contribute to accurate diagnoses, and early and more appropriate treatments in daily practice $^{9,17,19}$.
The results of this study reveal interesting aspects of the current epidemiology of neoplasms in canines in Guatemala. Compared to studies previously conducted from 2012 to 2014, where 183 biopsies and 113 autopsies with neoplasm cases were analyzed, the number of cases increased considerably in the years examined in this study, with a total of 1,162 cases as a whole[10].
According to the histological origin, the most common type of tumor was epithelial, followed by connective and derivatives, similar to what was found by Aco (2020) in Peru and Cevallos (2013) in Ecuador; and different from De la Cruz et al. (2017) in Mexico, Mayorga and Ruiz (2016) in Colombia, and Tordova (2016) in Bulgaria, who found a higher frequency of mesenchymal neoplasms $(51.5 - 55\%)$, followed by those of epithelial origin $(41.3 - 45\%)$. Epithelial tumors are usually the most common type in companion animals, primarily affecting the integument and soft tissues, followed by mammary neoplasms. Various authors with equivalent results regarding the affected body system consider that it is because the skin is the tissue most exposed to a wide variety of physical and chemical aggressions, and to various environmental factors that can trigger the development of neoplasms. Likewise, they are easier to recognize through clinical examination as it is the most visible organ, easily explored and accessible to obtain samples, unlike those that affect internal organs that require specific imaging studies, such as X-rays, ultrasound, computed tomography (CT) and magnetic resonance imaging $(\mathsf{MRI})^{8,14,16,22}$. In second place are mammary gland tumors, which are influenced by sex, being less prevalent in males, and their development in females is due to hormonal factors and increases the probability of suffering from it if they are sterilized, starting from $0.05\%$ if spayed before the first heat, up to $26\%$ if spayed after the second heat, and if spayed later in life, the risk of developing mammary tumors is the same as for an intact female[8,21]. Other authors (Elgue et al, 2012; Tordova, 2006) differ with this, since they indicate that mammary tumors are the most frequent in canines, and skin neoplasms are in second place.
Regarding anaplastic behavior, there was a similar result for benign and malignant neoplasms, with benign neoplasms being slightly more frequent, mainly those of the connective type and derivatives, like Mayorga and Ruiz (2016) who demonstrated that benign neoplasms and of mesenchymal type were the most frequent. In most studies, benign neoplasms are usually twice as frequent as malignant ones. The close numerical proximity between benign and malignant neoplasms in this study could be due to the fact that benign neoplasms are generally slow-growing, do not represent a danger to the patient, are often missed or detected incidentally during clinical examination, and that owners are reluctant to allow surgical resection and/or histopathological examination, causing the condition to migrate towards malignancy. A very different result was evidenced by Aco (2020) in Peru, and De la Cruz et al. (2017) in Mexico, where most malignant neoplasms were presented $^{1,4,11,13}$.
Concerning sex, females presented a higher frequency of neoplasms than males, like results seen by many authors, who consider that gender may be a factor that influences the development of some neoplasms due to the presence of hormones that can stimulate the abnormal cell division and turn into tumors. For this reason, it is considered that hormonal overstimulation of some organs increases the probability of the appearance of neoplasms, as well as working together with other carcinogens that could induce a mutation and neoplastic transformation of tissues $^{4,8,14,23}$.
Regarding the breed, it was shown that the pure breeds presented a higher frequency of neoplasms, of a malignant nature. Authors who agree with these results have determined that there is a racial predisposition toward the malignant growth of neoplasms, and the reason for this natural predisposition is unknown; it is believed that in specific individuals there is inactivation of tumor suppressor genes or activation of inherited oncogenes, or mutant versions of normal genes, which provide morphological characteristics of the breed together with its other genetic traits $^{1,8,11,23}$. When comparing dog breeds, neoplasms were found more frequently in poodles, schnauzers and boxers, similar to the results found by Juarez (2017), with the difference that they showed a major presence of neoplasms in canines of the schnauzer breed and in second place the poodles. The results could be influenced by the fact that they are the most popular breeds in Guatemala. However, it is essential to consider that owners tend to exaggerate or idealize the breed of their pets, so the quality of the breed data that depends primarily on owner reports should be questioned $^{19}$. Different results were found by Elgue et al (2012) in Uruguay, and De la Cruz et al. (2017) in Mexico, which determined that mongrel canines had a higher presentation of neoplasms than purebreds.
Concerning the age of presentation, it was more frequent in canines between 7-10 years, like Fajardo et al. (2013), Torres et al. (2015), and Aco (2020). This is as a consequence that the tissues have been exposed for extended periods to risk factors, as well as decreased cell repair as the animals age and a reduced immune response that leads to greater vulnerability to activity of carcinogens. The fact that malignant tumors mainly occurred in the older age groups (7-10 years and $>10$ years), suggests that age could be a significant risk factor, since exposure to endogenous and exogenous risk factors, together with a weakened immune system, can trigger the development of malignant tumors[8,23].
According to the Cramér index value, it is concluded that there is a moderate level of association between the histological origin of a neoplasm and anaplastic behavior. De la Cruz et al. (2017) in Mexico carried out a similar study where they evaluated the existence of a significant statistical association between the five most frequent tumors in the country, and the sex, race, and age of the patients in the development of neoplasms, to which they determined that there was no difference significant after evaluating the behavior of the neoplasm with race and sex, but with the age of the patients. Furthermore, Elgue et al. (2012) concluded through their study in Uruguay that sex, age, and race were factors associated with cancer. When analyzing this information, it is inferred that not only age is an important factor in the development of neoplasms and that senior age increases the risk of developing malignant neoplasms, as well as race and sex, but also the different histological origins of neoplasms are more or less likely to develop malignant tumor behavior, for example, according to the present study, neoplasms of epithelial and hematopoietic/ lymphoreticular origin are more likely to be malignant, while those of connective origin and derivatives are more likely to be benign $^{4,7}$.
## V. CONCLUSION
Studies of neoplasms from the records of a reference veterinary diagnostic center allow a deep understanding of the epidemiology of the disease and point out the differences that may occur from one country to another, which can support a better diagnosis and treatment in daily practice. This study focused on determining and statistically relating the histological origin and anaplastic behavior of neoplasms diagnosed between 2015 and 2019, as well as with the sex, race, age, and affected body system of the patient, revealing interesting aspects about the current epidemiology of neoplasms in canines in Guatemala. With the results obtained, it was found that epithelial-type neoplasms, and of a benign nature, were more frequently presented. In addition, an important level of association between the histological origin and the anaplastic behavior of these neoplasms was evidenced, which represents an important epidemiological factor. Likewise, it was determined that neoplasms in the country mainly affect females in terms of gender, purebred canines, the age group between 7 and 10 years old, and that the disease affects to a greater extent the integumentary system and soft tissues. From all this, it is concluded that it is essential to expand this type of research to understand the behavior of the disease in domestic animals in the country, and to make a continuous effort to evaluate the risk factors and compare them with the risk factors in humans.
### ACKNOWLEDGEMENTS
The authors thank the Pathology Unit of the Faculty of Veterinary Medicine and Zootechnics of the
University of San Carlos of Guatemala for the assistance with accessing data from the medical records, and to José R. Monroy and Gabriela Vásquez for the assistance on the manuscript drafting.
No third-party funding or support was received in connection with this study, or the writing or publication of the manuscript. The authors declare that there were no conflicts of interests.
REFERENCES RÉFERENCES REFERENCI A
1. Aco Alburqueque, P. R. (2020). Characterizacion de las neoplasias caninas diagnosticadas por histopatologia en el Laboratorio de Histologia y Patologia Veterinaria de la Universidad Peruana Cayetano Heredia: Periodo 2003-2015. Revista de Investigaciones Veterinarias del Perú, 31(2). http://dx.doi.org/10.15381/rivep.v3i2.16155
2. Cevallos Varas, V. M. (2013). Hallazgos Anatomo-Histopatologicos en tumores cutaneos extraídos en caninos de las clínicas Veterinarias de la Ciudad de Quito. [Tesis de prerado, Universidad Central de Ecuador]. Facultad de Medicina Veterinaria y Zootecnia, Universidad Central del Ecuador.
3. Cohen, D., Reif, J. S., Brodey, R. S., & Keiser, H. (1974). Epidemiological analysis of the most prevalent sites and types of canine neoplasia observed in a veterinary hospital. Cancer research, 34(11), 2859-2868.
4. De la Cruz Hernández, N. I., Monreal García, A. E., Carvajal de la Fuente, V., Barrón Vargas, C. A., Martínez Burnes, J., Zarate Terán, A.,... & Rangel Lucio, J. A. (2017). Frecuencia y caracterización de las principales neoplasias presentes en el perro domístico en Tamaulipas (México). Revista de Medicina Veterinaria, 1(35), 53-71. https://doi.org/10.19052/mv.4389
5. Dobson, J. (2019). Significant advances in veterinary oncology - 60 years on. The Journal of small animal practice, 60(12), 711-712. https://doi.org/10.1111/jsap.13076
6. Dobson, J. M., Samuel, S., Milstein, H., Rogers, K., & Wood, J. L. N. (2002). Canine neoplasia in the UK: estimates of incidence rates from a population of insured dogs. Journal of small animal practice, 43(6), 240-246. https://doi.org/10.1111/j.1748-5827.2002.tb00066.x
7. Elgue, V., Piaggio, J., Amaral, C., & Pessina, P. (2012). Factores asociados a la presentación del tipo de cancer en caninos atendidos en el Hospital de la Facultad de Veterinaria de Uruguay. Veterinaria (Montevideo), 48(187), 25-30.
8. Fajardo, R., Alpizar, A., Pérez, L. S., Martínez, J. S., & Córdova, E. (2013). Prevalence of tumors in dogs from the municipality of Toluca, México, from 2002 to 2008. Archivos de Medicina Veterinaria, 45(3),
- 305-309. http://dx.doi.org/10.4067/S0301-732X2013000300011
9. Gamlem, H., Nordstoga, K., &Glattre, E. (2008). Canine neoplasia-introductory paper. Apmis, 116(s125), 5-18. https://doi.org/10.1111/j.1600-0463.2008.125m2.x
10. Juarez López, M. Y. (2017). Caracterización de las neoplasias de caninos diagnósticas en la Unidad de Patología de la Facultad de Medicina Veterinaria y Zootecnia, durante el periodo de 2012 al 2014. [Tesis de prerado, Universidad de San Carlos de Guatemala]. Facultad de Medicina Veterinaria y Zootecnia, Universidad de San Carlos de Guatemala.
11. Kaldrymidou, H., Leontides, L., Koutinas, A. F., Saridomichelakis, M. N., & Karayannopoulou, M. (2002). Prevalence, distribution and factors associated with the presence and the potential for malignancy of cutaneous neoplasms in 174 dogs admitted to a clinic in northern Greece. Journal of Veterinary Medicine Series A, 49(2), 87-91. https://doi.org/10.1046/j.1439-0442.2002.jv408.x
12. MacVean, D. W., Monlux, A. W., Anderson, P. S., Silberg Jr, S. L., & Roszel, J. F. (1978). Frequency of canine and feline tumors in a defined population. Veterinary Pathology, 15(6), 700-715.
13. Mayorga J.S. y Ruiz, J. (2016) Frecuencia de neoplasias depiel en caninos emitidos a un laboratorio de patologia veterinaria de Bogota: Estudio retrospectivo en el periodo 2015-2016. [Tesis de prerado, Universidad de Ciencias Aplicadas y Ambientales]. Facultad de Medicina Veterinaria, Universidad de Ciencias Aplicadas y Ambientales.
14. Merlo, D. F., Rossi, L., Pellegrino, C., Ceppi, M., Cardellino, U., Capurro, C., Ratto, A., Sambucco, P.L., Sestito, V., Tanara, G. & Bocchini, V. (2008). Cancer incidence in pet dogs: findings of the Animal Tumor Registry of Genoa, Italy. Journal of veterinary internal medicine, 22(4), 976-984. https://doi.org/10.1111/j.1939-1676.2008.0133.x
15. O'Brien, D. J., Kaneene, J. B., Getis, A., Lloyd, J. W., Swanson, G. M., & Leader, R. W. (2000). Spatial and temporal comparison of selected cancers in dogs and humans, Michigan, USA, 1964-1994. Preventive Veterinary Medicine, 47(3), 187-204. https://doi.org/10.1016/S0167-5877(00)00168-9
16. Pakhrin, B., Kang, M. S., Bae, I. H., Park, M. S., Jee, H., You, M. H., Kim, J.H., Yoon, B.I., Choi, Y.K. & Kim, D. Y. (2007). Retrospective study of canine cutaneous tumors in Korea. Journal of Veterinary Science, 8(3), 229-236. https://doi.org/10.4142/jvs.2007.8.3.229
17. Pérez-Enriquez, J. M., Romero-Romero, L., Alonso-Morales, R. A., & Fuentes-Panana, E. M. (2020). Tumor prevalence in cats: experience from a reference diagnostic center in Mexico City (2006-
- 2018). Veterinaria México OA, 7(4). http://dx.doi.org/10.22201/fmvz.24486760e.2020.4.837
18. Prier, J. E., & Brodey, R. S. (1963). Canine Neoplasia: a prototype for human cancer study. Bulletin of the World Health Organization, 29(3), 331.
19. Reid-Smith, R., Bonnett, B., Sw, M., Sa, K., Abrams-Ogg, A., &Mj, H. (2000). The incidence of neoplasia in the canine and feline patient populations of private veterinary practices in Southern Ontario. [Tesis de doctorado, The University of Guelph] Faculty of Graduated Studies, University of Guelph.
20. Richards, H. G., McNeil, P. E., Thompson, H., & Reid, S. W. J. (2001). An epidemiological analysis of a canine-biopsies database compiled by a diagnostic histopathology service. Preventive Veterinary Medicine, 51(1-2), 125-136. https://doi.org/10.1016/S0167-5877(01)00211-2
21. Salas, Y., Márquez, A., Diaz, D., & Romero, L. (2015). Epidemiological study of mammary tumors in female dogs diagnosed during the period 2002-2012: a growing animal health problem. PloS one, 10(5), e0127381. https://doi.org/10.1371/journal.pone.0127381
22. Sostarić-Zuckermann, I. C., Severin, K., Hohsteter, M., Artuković, B., Beck, A., Kurilj, A. G., Sabočanec, R., Džaja, P. & Grabarevic, Z. (2013). Incidence and types of canine tumours in Croatia. Veterinarski Arhiv, 83, 31-45.
23. Todorova, I. (2006). Prevalence and etiology of the most common malignant tumours in dogs and cats. *Bulgarian Journal of Veterinary Medicine*, 9(2), 85-98.
24. Torres González-Chávez, M., Peraza González, B., Fabré Rodríguez, Y., Rodríguez Aurrecochea, J. C., Calña Seoane, L., Márquez Álvarez, M., Zamora Montalvo, Y., Rubio García, J.L., Martín Romero, J.A., & Camacho Socarrás, C. (2015). Frecuencia de presentación de neoplasias en caninos del municipio San Miguel del Padrón, La Habana, Cuba. Revista de Salud Animal, 37(1), 39-46.
25. Vascellari, M., Baioni, E., Ru, G., Carminato, A., & Mutinelli, F. (2009). Animal tumour registry of two provinces in northern Italy: incidence of spontaneous tumours in dogs and cats. BMC veterinary research, 5(1), 1-9.
26. Withrow, S. J. (2007). Withrow and MacEwen's small animal clinical oncology. Elsevier Health Sciences.
### SUPPLEMENTARY MATERIAL
Table 1: Sample size of each year
<table><tr><td>Year</td><td>PT</td><td>%</td><td>N</td></tr><tr><td>2015</td><td>106</td><td>9</td><td>26</td></tr><tr><td>2016</td><td>215</td><td>19</td><td>55</td></tr><tr><td>2017</td><td>269</td><td>23</td><td>67</td></tr><tr><td>2018</td><td>290</td><td>25</td><td>72</td></tr><tr><td>2019</td><td>282</td><td>24</td><td>69</td></tr><tr><td>Total</td><td>1,162</td><td>100</td><td>289</td></tr></table>
Table 2: Number of canines according to sex and histological origin of neoplasms
<table><tr><td rowspan="2">Histological origin</td><td colspan="2">Male</td><td colspan="2">Female</td><td colspan="2">Total</td></tr><tr><td>N°</td><td>%</td><td>N°</td><td>%</td><td>N°</td><td>%</td></tr><tr><td>Connective and derivatives</td><td>32</td><td>27</td><td>52</td><td>30</td><td>84</td><td>29</td></tr><tr><td>Endothelial</td><td>6</td><td>5</td><td>6</td><td>3</td><td>12</td><td>4</td></tr><tr><td>Hematopoietic or lymphoreticular</td><td>14</td><td>12</td><td>14</td><td>8</td><td>28</td><td>10</td></tr><tr><td>Epithelial</td><td>51</td><td>44</td><td>77</td><td>45</td><td>128</td><td>44</td></tr><tr><td>Nervous</td><td>1</td><td>1</td><td>1</td><td>1</td><td>2</td><td>1</td></tr><tr><td>Others</td><td>13</td><td>11</td><td>22</td><td>13</td><td>35</td><td>12</td></tr><tr><td>Total</td><td>117</td><td>100</td><td>172</td><td>100</td><td>289</td><td>100</td></tr></table>
Table 3: Number of canines according to sex and anaplastic behavior of neoplasms
<table><tr><td rowspan="2">Sex</td><td colspan="2">Benign</td><td colspan="2">Malignant</td><td colspan="2">Total</td></tr><tr><td>N°</td><td>%</td><td>N°</td><td>%</td><td>N°</td><td>%</td></tr><tr><td>Male</td><td>53</td><td>36</td><td>62</td><td>43</td><td>115</td><td>40</td></tr><tr><td>Female</td><td>93</td><td>64</td><td>81</td><td>57</td><td>174</td><td>60</td></tr><tr><td>Total</td><td>146</td><td>100</td><td>143</td><td>100</td><td>289</td><td>100</td></tr></table>
Table 4: Number of canines according to breed and histological origin of neoplasms
<table><tr><td rowspan="2">Histological origin</td><td colspan="2">Purebred</td><td colspan="2">Mongrel</td><td colspan="2">Total</td></tr><tr><td>N°</td><td>%</td><td>N°</td><td>%</td><td>N°</td><td>%</td></tr><tr><td>Connective and derivatives</td><td>63</td><td>28</td><td>21</td><td>33</td><td>84</td><td>29</td></tr><tr><td>Endothelial</td><td>9</td><td>4</td><td>3</td><td>5</td><td>12</td><td>4</td></tr><tr><td>Hematopoyetic or lymphoreticular</td><td>19</td><td>8</td><td>9</td><td>14</td><td>28</td><td>10</td></tr><tr><td>Epithelial</td><td>105</td><td>47</td><td>23</td><td>36</td><td>128</td><td>44</td></tr><tr><td>Nervous</td><td>2</td><td>1</td><td>0</td><td>0</td><td>2</td><td>1</td></tr><tr><td>Others</td><td>27</td><td>12</td><td>8</td><td>12</td><td>35</td><td>12</td></tr><tr><td>Total</td><td>225</td><td>100</td><td>64</td><td>100</td><td>289</td><td>100</td></tr></table>
Table 5: Number of canines according to breed and anaplastic behavior of neoplasms
<table><tr><td rowspan="2">Breed</td><td colspan="2">Benign</td><td colspan="2">Malignant</td><td colspan="2">Total</td></tr><tr><td>N°</td><td>%</td><td>N°</td><td>%</td><td>N°</td><td>%</td></tr><tr><td>Purebred</td><td>110</td><td>75</td><td>115</td><td>80</td><td>225</td><td>78</td></tr><tr><td>Mongrel</td><td>36</td><td>25</td><td>28</td><td>20</td><td>64</td><td>22</td></tr><tr><td>Total</td><td>146</td><td>100</td><td>143</td><td>100</td><td>289</td><td>100</td></tr></table>
Table 6: Number of canines according to age and histological origin of neoplasms
<table><tr><td rowspan="2">Histological origin</td><td colspan="2"><1 year</td><td colspan="2">1-6 years</td><td colspan="2">7-10 years</td><td colspan="2">>10 years</td><td colspan="2">Total</td></tr><tr><td>N°</td><td>%</td><td>N°</td><td>%</td><td>N°</td><td>%</td><td>N°</td><td>%</td><td>N°</td><td>%</td></tr><tr><td>Connective and derivatives</td><td>0</td><td>0</td><td>31</td><td>40</td><td>38</td><td>29</td><td>15</td><td>20</td><td>84</td><td>29</td></tr><tr><td>Endothelial</td><td>1</td><td>20</td><td>6</td><td>8</td><td>5</td><td>4</td><td>0</td><td>0</td><td>12</td><td>4</td></tr><tr><td>Hematopoyetic or lymphoreticular</td><td>0</td><td>0</td><td>9</td><td>11</td><td>17</td><td>13</td><td>2</td><td>3</td><td>28</td><td>10</td></tr><tr><td>Epithelial</td><td>3</td><td>60</td><td>24</td><td>31</td><td>56</td><td>42</td><td>45</td><td>61</td><td>128</td><td>44</td></tr><tr><td>Nervous</td><td>0</td><td>0</td><td>0</td><td>0</td><td>1</td><td>1</td><td>1</td><td>1</td><td>2</td><td>1</td></tr><tr><td>Others</td><td>1</td><td>20</td><td>8</td><td>10</td><td>15</td><td>11</td><td>11</td><td>15</td><td>35</td><td>12</td></tr><tr><td>Total</td><td>5</td><td>100</td><td>78</td><td>100</td><td>132</td><td>100</td><td>74</td><td>100</td><td>289</td><td>100</td></tr></table>
Table 7: Number of canines according to age and anaplastic behavior of neoplasms
<table><tr><td rowspan="2">Age</td><td colspan="2">Benign</td><td colspan="2">Malignant</td><td colspan="2">Total</td></tr><tr><td>N°</td><td>%</td><td>N°</td><td>%</td><td>N°</td><td>%</td></tr><tr><td><1 year</td><td>4</td><td>3</td><td>1</td><td>1</td><td>5</td><td>2</td></tr><tr><td>1-6 years</td><td>42</td><td>29</td><td>36</td><td>25</td><td>78</td><td>27</td></tr><tr><td>7-10 years</td><td>70</td><td>48</td><td>62</td><td>43</td><td>132</td><td>46</td></tr><tr><td>>10 years</td><td>30</td><td>20</td><td>44</td><td>31</td><td>74</td><td>25</td></tr><tr><td>Total</td><td>146</td><td>100</td><td>143</td><td>100</td><td>289</td><td>100</td></tr></table>
Table 8: Number of canines according to the affected body system and anaplastic behavior of neoplasms
<table><tr><td rowspan="2">Affected body system</td><td colspan="2">Benign</td><td colspan="2">Malignant</td><td colspan="2">Total</td></tr><tr><td>N°</td><td>%</td><td>N°</td><td>%</td><td>N°</td><td>%</td></tr><tr><td>Integumentary and soft tissues</td><td>56</td><td>38</td><td>40</td><td>28</td><td>96</td><td>33</td></tr><tr><td>Gastrointestinal</td><td>16</td><td>11</td><td>21</td><td>15</td><td>37</td><td>13</td></tr><tr><td>Respiratory</td><td>4</td><td>3</td><td>4</td><td>3</td><td>8</td><td>3</td></tr><tr><td>Urogenital</td><td>23</td><td>16</td><td>12</td><td>8</td><td>35</td><td>12</td></tr><tr><td>Hemolymphatic</td><td>0</td><td>0</td><td>8</td><td>5</td><td>8</td><td>3</td></tr><tr><td>Endocrine</td><td>0</td><td>0</td><td>0</td><td>0</td><td>0</td><td>0</td></tr><tr><td>Nervous</td><td>2</td><td>1</td><td>0</td><td>0</td><td>2</td><td>1</td></tr><tr><td>Cardiovascular</td><td>2</td><td>1</td><td>1</td><td>1</td><td>3</td><td>1</td></tr><tr><td>Mammary gland</td><td>26</td><td>18</td><td>27</td><td>19</td><td>53</td><td>18</td></tr><tr><td>Musculoskeletal</td><td>17</td><td>12</td><td>9</td><td>6</td><td>26</td><td>9</td></tr><tr><td>Multicentric</td><td>0</td><td>0</td><td>21</td><td>15</td><td>21</td><td>7</td></tr><tr><td>Total</td><td>146</td><td>100</td><td>143</td><td>100</td><td>289</td><td>100</td></tr></table>
Table 9: Contingency table for statistical analysis of Cramér's V association index
<table><tr><td rowspan="2">Histological origin</td><td colspan="2">Anaplastic behavior</td><td rowspan="2">Total</td></tr><tr><td>Benign</td><td>Malignant</td></tr><tr><td>Connective and derivatives</td><td>68 (42.43)</td><td>16 (41.56)</td><td>84</td></tr><tr><td>Endothelial</td><td>10 (6.06)</td><td>2 (5.93)</td><td>12</td></tr><tr><td>Hematopoietic or lymphoreticular</td><td>2 (14.14)</td><td>26 (13.85)</td><td>28</td></tr><tr><td>Epithelial</td><td>41 (64.66)</td><td>87 (63.33)</td><td>128</td></tr><tr><td>Nervous</td><td>2 (1.01)</td><td>0 (0.98)</td><td>2</td></tr><tr><td>Others</td><td>23 (17.68)</td><td>12 (17.31)</td><td>35</td></tr><tr><td>Total</td><td>146</td><td>143</td><td>289</td></tr></table>
Table 10: Distribution of cases of neoplasms according to purebred canines
<table><tr><td>Breed</td><td>No.</td><td>%</td></tr><tr><td>Poodle</td><td>29</td><td>12.9</td></tr><tr><td>Schnauzer</td><td>25</td><td>11.1</td></tr><tr><td>Boxer</td><td>19</td><td>8.4</td></tr><tr><td>English Cocker Spaniel</td><td>17</td><td>7.6</td></tr><tr><td>Labrador Retriever</td><td>16</td><td>7.1</td></tr><tr><td>Husky</td><td>13</td><td>5.8</td></tr><tr><td>Golden Retriever</td><td>12</td><td>5.3</td></tr><tr><td>Dachshund</td><td>8</td><td>3.6</td></tr><tr><td>Chihuahua</td><td>7</td><td>3.1</td></tr><tr><td>Basset Hound</td><td>7</td><td>3.1</td></tr><tr><td>Pitbull</td><td>6</td><td>2.7</td></tr><tr><td>Shih Tzu</td><td>6</td><td>2.7</td></tr><tr><td>German shepherd</td><td>6</td><td>2.7</td></tr><tr><td>Beagle</td><td>5</td><td>2.2</td></tr><tr><td>Guatemala dogo</td><td>4</td><td>1.8</td></tr><tr><td>Rottweiler</td><td>3</td><td>1.3</td></tr><tr><td>French bulldog</td><td>3</td><td>1.3</td></tr><tr><td>Sharpei</td><td>3</td><td>1.3</td></tr><tr><td>Bulldog</td><td>2</td><td>0.9</td></tr><tr><td>Fila brasiliero</td><td>2</td><td>0.9</td></tr><tr><td>Doberman</td><td>2</td><td>0.9</td></tr><tr><td>American Bully</td><td>2</td><td>0.9</td></tr><tr><td>Chowchow</td><td>2</td><td>0.9</td></tr><tr><td>Dalmatian</td><td>2</td><td>0.9</td></tr><tr><td>Great dane</td><td>2</td><td>0.9</td></tr><tr><td>Others</td><td>22</td><td>9.7</td></tr><tr><td>Total</td><td>225</td><td>100</td></tr></table>
HISTOLOGICAL ORIGIN AND ANAPLASTIC BEHAVIOR OF NEOPLASMS DIAGNOSED IN CANINES IN GUATEMALA (2015-2019)
## I. General Data
1. Patient's ID:
2. Year
2015
1
2016
#
2017
#
2018
2019
### 3. Breed
Mongrel
#
Purebred
#### 4. Age
<1 year
1-6 years
1
7-10 years
1
>10 years
#### 5. Sex
Male
Female
#
#### 6. Affected body system
- Integumentary and soft tissues
- Gastrointestinal
- Respiratory
- Urogenital
- Hemolymphatic
- Endocrine
- Nervous
- Cardiovascular
- Mammary gland
- Musculoskeletal
- Multicentric
## I. Histological origin
- Connective and derivatives
- Endothelial
- Hematopoietic or lymphoreticular
- Epithelial
- Nervous
- Others
## II. Anaplastic behavior
Benign Malignant
Figure 1: Instrument used in data collection
Generating HTML Viewer...
References
26 Cites in Article
P Aco Alburqueque (2020). Caracterización de las neoplasias caninas diagnosticadas por histopatología en el Laboratorio de Histología y Patología Veterinaria de la Universidad Peruana Cayetano Heredia: Periodo 2003-2015.
Daniel Degado-Piedra,Elizabeth Padilla Paltas,David Flores Cuvi,Mayra Paltas Miranda (2013). Guía quirúrgica en 3D para la resección de exostosis vestibular y torus mandibular: reporte de un caso.
D Cohen,J Reif,R Brodey,H Keiser (1974). Epidemiological analysis of the most prevalent sites and types of canine neoplasia observed in a veterinary hospital.
Cruz De La,N Hernández,A García,V Carvajal De La Fuente,Barrón,C Vargas,J Martínez Burnes,A Zarate Terán,. Rangel Lucio,J (2017). Frecuencia y caracterización de las principales neoplasias presentes en el perro doméstico en Tamaulipas (México).
J Dobson (2019). Significant advances in veterinary oncology -60 years on.
J Dobson,S Samuel,H Milstein,K Rogers,J Wood (2002). Canine neoplasia in the UK: estimates of incidence rates from a population of insured dogs.
V Elgue,J Piaggio,C Amaral,P Pessina (2012). Factores asociados a la presentación del tipo de cáncer en caninos atendidos en el Hospital de la Facultad de Veterinaria de Uruguay.
R Fajardo,A Alpízar,L Pérez,J Martínez,E Córdova (2013). Prevalence of tumors in dogs from the municipality of Toluca, México, from 2002 to 2008.
Hans Gamlem,Knut Nordstoga,Eystein Glattre (2008). Canine neoplasia – Introductory paper.
M Juárez López (2017). Caracterización de las neoplasias de caninos diagnosticadas en la Unidad de Patología de la Facultad de Medicina Veterinaria y Zootecnia, durante el periodo de 2012 al 2014.
H Kaldrymidou,L Leontides,A Koutinas,M Saridomichelakis,M Karayannopoulou (2002). Prevalence, Distribution and Factors Associated with the Presence and the Potential for Malignancy of Cutaneous Neoplasms in 174 Dogs Admitted to a Clinic in Northern Greece.
D Macvean,A Monlux,P Anderson,S Silberg,J Roszel (1978). Frequency of Canine and Feline Tumors in a Defined Population.
J Mayorga,J Ruiz (2016). Frecuencia de neoplasias de piel en caninos emitidos a un laboratorio de patología veterinaria de Bogotá: Estudio retrospectivo en el periodo 2015-2016.
D Merlo,L Rossi,C Pellegrino,M Ceppi,U Cardellino,C Capurro,A Ratto,P Sambucco,V Sestito,G Tanara,V Bocchini (2008). Cancer Incidence in Pet Dogs: Findings of the Animal Tumor Registry of Genoa, Italy.
Daniel O’brien,John Kaneene,Arthur Getis,James Lloyd,G Swanson,Robert Leader (2000). Spatial and temporal comparison of selected cancers in dogs and humans, Michigan, USA, 1964–1994.
Bidur Pakhrin,Min-Soo Kang,Il-Hong Bae,Mi-Sun Park,Hyang Jee,Mi-Hyeon You,Jae-Hoon Kim,Byung-Il Yoon,Yang-Kyu Choi,Dae-Yong Kim (2007). Retrospective study of canine cutaneous tumors in Korea.
Juan Pérez-Enriquez,Laura Romero-Romero,Rogelio Alonso-Morales,Ezequiel Fuentes-Pananá (2020). Tumor prevalence in cats: experience from a reference diagnostic center in Mexico City (2006-2018).
J Prier,R Brodey (1963). Canine Neoplasia: a prototype for human cancer study.
Adronie Verbrugghe,Alexandra Rankovic (2000). Dietary choline in feline nutrition and its role in obesity prevention and liver health.
H Richards,P Mcneil,H Thompson,S Reid (2001). An epidemiological analysis of a canine-biopsies database compiled by a diagnostic histopathology service.
Yaritza Salas,Adelys Márquez,Daniel Diaz,Laura Romero (2015). Epidemiological Study of Mammary Tumors in Female Dogs Diagnosed during the Period 2002-2012: A Growing Animal Health Problem.
I Šoštarić-Zuckermann,K Severin,M Hohšteter,B Artuković,A Beck,A Kurilj,R Saboc Ǎnec,P Dzǎja,Ž Grabarevi Ć (2013). Incidence and types of canine tumours in Croatia.
I Todorova (2006). Prevalence and etiology of the most common malignant tumours in dogs and cats.
Ana Testa González,Marislay Torres Antela,Marelys Estupiñán Álvarez (2015). Methodological alternative fordevelopinglistening comprehension in english among medical students.
Marta Vascellari,Elisa Baioni,Giuseppe Ru,Antonio Carminato,Franco Mutinelli (2009). Animal tumour registry of two provinces in northern Italy: incidence of spontaneous tumours in dogs and cats.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Andrea Daniela Lemus-Centes. 2026. \u201cHistological Origin and Anaplastic Behavior of Neoplasms Diagnosed in Canines in Guatemala (2015-2019)\u201d. Global Journal of Medical Research - G: Veterinary Science & Medicine GJMR-G Volume 22 (GJMR Volume 22 Issue G1).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.