## I. INTRODUCTION
One of the most important modern phenomena that demands careful thought is the astounding rise in the world's population. Between 6 and 7 billion people are thought to live on the planet today. The exponential growth of their populations is a significant problem for developing countries like India.
An imbalance in socioeconomic infrastructure is likely to result from this population boom's negative effects on social and economic policies. Since human fertility is limited, controlling it becomes a vital and pressing biosocial and medical concern. Many medications, including hormonal and other compounds, have been developed in response to the need for fertility control. To mitigate the potential adverse effects associated with chemically synthesized drugs, there is a preference for indigenous plants, which are not only cost-effective and readily available but also considered safe (1).
Often known as oral contraceptives, antifertility agents are medications that control fertility (2). These drugs affect women's ovulation and menstrual cycles. Estrogen and progesterone are commonly found in birth control pills. The active ingredients in these antifertility medications work on females by blocking ovulation and implantation, preventing fertilization, and either killing the zygote or causing abortion. These substances function in males by influencing gonadotropins and sperm viability, lowering testosterone levels, or suppressing spermatogenesis (3). Population growth presents serious problems for natural, social, and economic resources (4). The pressing need for efficient contraceptive methods is highlighted by the growing population in developing countries (5).
It has long been known that medicinal plants are useful tools for treating a variety of human health conditions. These plants have been used for centuries to treat physical and mental illnesses; in developing nations, about $80\%$ of medical treatments are used.
6. This field is aided by phytoestrogens, which are novel compounds present in a variety of plants. Furthermore, a number of medications, such as testosterone, gossypol, tamoxifen, and triptolide, are being investigated as antifertility agents (7). Oral contraceptives, also referred to as antifertility agents, are medications that control fertility (2). These drugs affect women's ovulation and menstrual cycles. Estrogen and progesterone are commonly found in birth control pills. The active ingredients in these antifertility medications work on females by blocking ovulation and implantation, preventing fertilization, and either killing the zygote or causing abortion. These substances affect gonadotropins and sperm viability, lower testosterone levels, or inhibit spermatogenesis in males. Many developing nations are currently controlling their populations (3). By interfering with a number of normal reproductive processes in both males and females, antifertility medications reduce fertility. $100\%$ effectiveness, reversibility of effects, lack of side effects, and ease of use are the best qualities in a contraceptive agent (8). Due to a lack of written records, a lack of scientific validation, and comparatively low economic resources within these traditions, these traditional knowledge systems have started to deteriorate over time.
It has been determined that a wide variety of plant species can influence fertility (9). Traditional medicine has long used plant-based remedies to manage fertility in many places, such as Ethiopia and India. Numerous medicinal plants have been used to treat a range of conditions, including infertility, in addition to being used as dietary supplements, frequently without a thorough understanding of their mechanisms (10). A considerable number of herbal plants also show varied degrees of toxicity, even though many of them have a variety of antifertility qualities, including oestrogenic, spermicidal, ebolic, abortifacient, and anti-implantation effects (10). Numerous products made from plants have the ability to reduce fertility in both men and women, which raises the possibility that they could be developed as forms of birth control. Only a small number of native plants have had their antifertility effects thoroughly studied, despite evidence that many of them can prevent conception. To find new oral active non-steroidal contraceptive agents, the World Health Organization (WHO) formed a task force on plant research. Numerous medicinal plant extracts have been tested for their ability to prevent infertility in both sexes (11).
It is not a new idea to create safe and efficient oral fertility-regulating substances for human use that are derived from higher plants. Almost all indigenous cultures have used a variety of plants to try to manage population growth for centuries. Many plants have the ability to regulate fertility. There are currently initiatives underway to turn these plants into antifertility products. Economically disadvantaged populations could greatly benefit from plant-based contraceptive methods, such as crude extracts or scientifically validated composite preparations, as these options would be more affordable. The possible abortifacient and antifertility effects of many plants, including those traditionally used in folk contraceptives, are the subject of extensive research worldwide (12). In light of the negative consequences of traditional approaches, fertility control, including contraception and infertility treatment, is an important component of reproductive health for both men and women (13). Numerous efficient techniques for causing infertility have been investigated over time, such as hormonal, chemical, and immunological approaches (14). However, women are less likely to accept chemical methods because they frequently result in a number of side effects, including obesity, gallstones, gastrointestinal problems, and an increased risk of breast and cervical cancers, asthma, and thromboembolism. Hormonal contraceptives are also linked to an increased risk of cancer. Because of their negligible or nonexistent adverse effects, scientists are therefore becoming more interested in plant-derived products as a major source of naturally occurring fertility-regulating agents (15). Health, population growth, and women's empowerment are all directly correlated with the rising use of contraceptive methods
(16). In both males and females, a variety of herbal plants have antifertility effects (17).
In recent years, population control has become more and more important. There are many different synthetic contraceptive methods available, but the side effects that come with them frequently limit their long-term use. Both male and female populations have been the focus of efforts to prevent conception. The goal of research in the field of male contraception is to find spermicidal agents that work. On the other hand, female contraception consists of several steps that can be controlled with medication, such as ovulation, fertilization, implantation of the fertilized ovum, and the final maturation of the fetus. As a result, methods to interfere with fertilization have mostly focused on these phases using different substances that are said to be abortifacient, antiovulatory, or anti-implantation. Although there are currently alternatives like steroidal pills, injections, IUDs, barrier methods, and sterilization techniques, the changing lifestyle and growing population challenge suggest that the perfect contraceptive solution has not yet been found (18). The exponential growth of the human population, which can negatively impact economic policies and destabilize financial structures, is one of the major issues facing developing countries. Thus, it is crucial to keep an eye on population growth (19). The demand for herbal remedies made from medicinal plants has increased due to the high cost of new medications, their inaccessibility in remote areas, and the many negative effects of current synthetic fertility control methods, including weight gain, hypertension, hormonal imbalances, and an increased risk of cancer. According to research, women use contraceptives at a higher rate than men worldwide, especially in rural and developing areas where access to contemporary contraceptives is restricted. For women, especially those living in rural areas of developing nations with high population densities like Bangladesh, China, India, and Africa, herbal contraceptives provide an affordable and easily accessible alternative. These substitutes are distinguished by their lower adverse effects and possible efficacy. However, because herbal medicines may pose minor risks, extensive testing is necessary to determine their safety and efficacy (20).
Pharmaceuticals that control fertility are known as oral contraceptives, or antifertility drugs (2). These medications affect the menstrual cycle and female ovulation. A combination of progesterone and estrogen is commonly found in birth control pills. When a contraceptive stops women from ovulating, implantation, fertilization, zygote destruction, or abortion, it is considered effective. It also has an effect on gonadotrophin levels or sperm viability, suppresses testosterone, and stops male spermatogenesis. At the moment, many developing nations are taking action to curb population growth (21). By preventing the production of prostaglandins, drugs like oxyphenbutazone, indomethacin, and acetylsalicylic acid have shown antifertility effects in studies involving albino male and female rabbits. In particular, indomethacin and oxyphenbutazone affect reproductive processes in male rabbits. In many developing countries, the trend of population control is common. Additionally, it has been demonstrated that the aforementioned compounds in albino rabbits decrease prostaglandin synthesis and have antifertility effects. Oxygen butazone and indomethacin have a significant impact on male rabbit reproductive processes (22). Because they are less toxic and have been used for a long time in traditional medical practices like Ayurveda, people are increasingly choosing plant-derived medications over synthetic ones. To encourage family planning, a variety of contraceptive methods have been promoted. However, there is now more interest in indigenous herbs for their possible contraceptive qualities due to the serious side effects linked to synthetic steroidal contraceptives. Consequently, it is essential to explore suitable native plant products that could serve as alternatives to conventional tablets (23).
In many parts of the world, such as Morocco, Saudi Arabia, Taiwan, and Trinidad and Tobago, ethnobotanical research on medicinal plants used by local populations has been carried out. Several plant species have been found to have antifertility properties. The use of plant-based remedies has long been a part of traditional medicine practices for fertility control in many parts of Ethiopia, India, and the rest of the world. Without a thorough understanding of their mechanisms, a variety of medicinal plants have been used as dietary supplements and to treat a wide range of illnesses, including infertility. A sizable fraction of these medicinal plants also show varied degrees of toxicity, even though many herbal plants have a variety of antifertility qualities, including anti-implantation, abortifacient, estrogenic, and spermicidal effects (24). Since the dawn of civilization, traditional plants have been essential to human society, helping to fight off a variety of illnesses. Historically, natural products—including plants, animals, and minerals—have been the cornerstone of disease treatment. Nearly $80\%$ of developing countries, according to the World Health Organization, struggle to obtain synthetic drugs and must instead rely on traditional medicines, which are mostly made from plants, to meet their basic medical needs (25).
Although estrogen and progesterone-containing contraceptives are currently widely used and effective for family planning, many countries have banned the use of hormonal contraceptives due to the serious side effects of synthetic steroidal contraceptives, including gonadal toxicity, temporary or permanent infertility, testicular germ cell cancer, breast and prostate cancer, brain developmental issues, endometriosis, obesity, cholelithiasis, gastrointestinal disturbances, asthma, venous thromboembolism, and early puberty. The dangers associated with these drugs have led to research into novel compounds made from medicinal plants that could replace conventional antifertility drugs.
The objective of the current study is to review the antifertility properties of various medicinal plants.
## II. MATERIALS AND METHODS
The information presented in this review is the outcome of a comprehensive bibliographic investigation, which involved the analysis of classical textbooks, scientific journals, and consultation of globally recognized databases. Peer-reviewed articles were collected from various sources, including SCOPUS, PUBMED, GOOGL SCHOLAR, and INFLIBNET.
### a) Reproductive Systems
The conceptive framework is a sex organ inside a life form that works with the end goal of sexual propagation. Numerous non-living substances, for example, liquids, hormones, and pheromones, are the most significant types of gear for regenerative frameworks (26).
## i. Male Reproductive System
The different sex organs that play a major role in human generation are part of the male conceptual framework. These organs are located inside the pelvis and outside the body. An ovum in the female's body is fertilized by the sperm and semen produced by the penis and gonads, the main male sex organs. The fertilized ovum grows into a fetus, which is subsequently born as an infant (26).
## ii. Female Reproductive System
The inner and outer sex organs make up the female conceptual framework. It is attempting to increase the number of new generations. When the female human reproductive system matures after being immature at birth. One can produce gametes and carry a fetus to term through puberty. The ovaries, fallopian tubes, and uterus are the internal sex organs. Undeveloped organisms that develop into fetuses are called uterus or belly obliges. Additionally, the uterus produces uterine and vaginal discharges that facilitate sperm transit to the Fallopian tubes. The egg cells are made in the ovaries. Genitals and vaginal openings are other names for the external sex organs. The cervix is where the vagina and uterus are joined (26).
### b) Antifertility
Antifertility agents are substances that can inhibit ovulation or fertilization, ultimately leading to the termination of a pregnancy (27). Medications designed to prevent fertilization are referred to as having antifertility effects, which are also known as contraceptive effects. Contraception encompasses methods that disrupt the natural processes of ovulation, fertilization, and the implantation of the ovum, thereby preventing pregnancy (28).
A concise overview of plants exhibiting antifertility properties, along with their active components, is presented in Table 1. The investigation of various antifertility medicinal plants led to the conclusion that the efficacy of different plant parts is ranked as follows: Leaf > Seed > Whole Plant > Root > Aerial Part = Bark > Stem > Fruit = Flower > Tuber > Stem Bark > Rhizome. The leaves demonstrate the highest potential for antifertility activity, while the rhizome shows the least potential (see Figures 1 and 2).
### c) Medicinal plants used as antifertility agents
## i. Antiovulation Activity
Polygonum hydropiper Linn (Marsh Pepper) belongs to the family Polygonaceae, which is in part valued for its roots and leaves and adds such active ingredients as formic acid, acetic acid, beldianic acid, tannin, essential oil, and oxymethyl-anthraquinones. It is used in situations involving diarrhea, skin problems, hemorrhoids, and dyspepsia. It is used in folk medicine as an anti-cancer and anti-rheumatic agent. Biologically, these constituents can have antioxidant, antimicrobial, anti-inflammatory, and antifertility effects in humans. In one study, Kapoor et al. (1974) (30) have reported on the anti-ovulatory activity in this plant. Their study using three varieties of extracts (petroleum, aqueous, and alcohol) was conducted to examine the antifertility activity of this particular plant. Antifertility activity was noticed in rabbits with copper-induced ovulation. Petroleum ether extract of the roots of Polygonum hydropiper was detected adequately in inhibiting ovulation in $60\%$ of the animals. All the other extracts prohibited ovulation in $40\%$ or less of the animals (30).
## ii. Anti-Implantation Activity
Ailanthus excelsa Roxb is a deciduous tree from the Simaroubaceae family and is widely distributed in Asia and northern Australia. Its native origin is China and is known as the "tree of heaven" (6). In Maharashtra, the above plants were used traditionally for anti-implantation and abortion activity (Table 2). Ailanthus excelsa Roxb is a deciduous tree from the Simaroubaceae family and is widely distributed in Asia and northern Australia. Its native origin is China and is known as the "tree of heaven" (6). In Maharashtra, the above plants were used traditionally for anti-implantation and abortion activity (Table 2). Ailanthus excelsa Roxb is a deciduous tree from the Simaroubaceae family and is widely distributed in India and northern Australia. Its native origin is China and is known as the "tree of heaven" (6). In Maharashtra, the above plants were used traditionally for anti-implantation and abortifacient activity (Table 2). Ailanthus excelsa Roxb is an abscission tree from the Simaroubaceae family and is extensively distributed in Asia and northern Australia. Its ancient origin is China and is known as the "tree of heaven" (32). In Maharashtra, the above plants were used habitually for anti-implantation and abortifacient activity.
The anti-implantation activity was purposive according to the method of Olagbende-Dada Stella O et al., 2009 (33). Eighteen mature, female, colony-bred Wistar albino rats were divided into three groups (6 female rats per group). One group was used as a control, and the other two groups were used as a test group. Female rats in the proestrous phase were kept with males with confirmed fertility in a ratio of 2:1. The female rats were examined in the following morning for verification of copulation; the vaginal smear was examined for thick clumps of spermatozoa. The day on which the spermatozoa were found in the smear was observed the first day of pregnancy (Day 1). A 150 mg/kg of body weight and 300 mg per kg of body weight of the extract was administrated intragastrically for 10 days from day 1 to day 10 of pregnancy for the test group and equal volume of vehicle for the control group. On day 11, all groups of rats were laparotomized under light ether anesthesia to determine the number of implantation sites in the horns of the uteri. The presence of a difference in the mean number of propagation sites between the extract and the control was taken as a positive response.
## iii. Antispermatogenic Activity
Plumbago zeylanica belongs to the family Plumbaginaceae, and its antifertility ingredients include roots and leaves. Its active rules are plumbagin, isoshinanolone, transcinnamic acid, vanillic acid, betasitosterol, 4-hydroxybenzaldehyde, and plumbagic acid, and it is used to cure piles, leukoderma, and other skin diseases. It developed to foster diverse biological activities, including anti-Helicobacter pylori, antidiabetic, antioxidant, and antifertility. An earlier rat study was initiated using the plant's ethanol extract. When the applied extract dosage was $159\mathrm{mg/kg}$, seminiferous tubule diameters became smaller, and spermatocyte and spermatid production was reduced. Furthermore, a decline in immature and mature Leydig cells occurred, and degenerating cells were significantly increased. Lastly, the testicular cell population was decreased. Overall, this study showed palpable plant-based antifertility activity (34).
## iv. Abortifacient Activity
Plumeria rubra L. are secreting latex trees and shrubs that belong to the Apocynaceae family. The commixture of bark & roots of Plumeria rubra is traditionally used to treat asthma, ease constipation, stimulate menstruation, and reduce fever, and the latex is used to soothe irritation (35). In India, however, its fruit is used as an abortifacient (36).
The plant extracts were checked in female albino rats for abortifacient activity as per Khanna et al. (1969) (37). The female rats in the pro-estrous stage were caged with males of proven fertility in the ratio of 2:1 in the evening and examined the successive day for the evidence of copulation. Rats exhibiting a thick clump of spermatozoa in their vaginal smear were partitioned, and that day was designated as day 1 of pregnancy. These rats were irregularly distributed into 13 groups, one control group and 12 experimental groups of 6 animals each. On the day of pregnancy, animals were laparotomized below light ether anesthesia using sterile conditions. The two horns of uteri were inspected to determine the implantation sites. Thereafter the abdominal wound was sutured in layers (38).
### d) Hormonal Control of Fertility
The birth control pill, the most effective form of birth control, is based on the oral administration of steroids. Either progestins and estrogens are used together, or progestins are used alone, as with the minipill. Furthermore, different combinations of steroids can be given intrauterine or as long-acting injectable preparations. Estradiol and progesterone are not suitable for use in oral pills because they are metabolized in the liver and gastrointestinal tract. Therefore, different synthetic progestins like norethindrone, norethindrone acetate, norgestrel, ethinodiol diacetate, or norethynodrel are used in conjunction with synthetic estrogens like mestranol or ethinyl estradiol. The hormones are administered in a cyclical manner for 21 days, starting on the fifth day of the menstrual cycle and ending with either no pills or a placebo for 7 days. Through negative feedback effects on the hypothalamus, the high levels of progestin and estrogen prevent ovulation and the midcycle LH surge. While FSH levels are typically suppressed, irregular LH peaks can occasionally be seen. Estrogens are still secreted, but ovarian progesterone production is reduced. Depending on the type and dosage of the contraceptive, the effects on the endometrium can vary. Within a few days of beginning daily intake, there is a rapid progression from proliferation to early secretory changes, which are followed by regressive changes (39).
### e) Mechanism of Action of Antifertility Plants
It has been reported that medicinal plants have antifertility effects through a variety of mechanisms. Their impact on sex hormones, specifically for reducing fertility, regulating the menstrual cycle, alleviating dysmenorrhea, treating enlarged prostate, menopausal symptoms, breast pain, etc., is one of their main functions (40). Furthermore, by peripherally modulating follicle-stimulating hormone (FSH) and luteinizing hormone (LH), plants with estrogenic qualities can directly affect pituitary action, reducing their secretions and preventing ovulation (41). On the other hand, plants that have anti-estrogenic properties have abortifacient effects and interfere with the development of the ovum and endometrium (42). In females, the hypothalamus, anterior pituitary, ovary, oviduct, uterus, and vagina are the sites of action of antifertility medications. Antifertility effects primarily occur in the mammalian uterus (40). In immature rats, typical estrogenic compounds can cause cornification and vaginal opening, as well as increase the uterine wet weight, all of which have antiimplantation effects (43). When given to male rats, plant extracts have also demonstrated encouraging antifertility effects. Plants have a variety of effects on the male reproductive system that can cause antifertility, such as antispermatogenic, post-testicular, spermicidal, sperm immobilizing, and antiandrogenic effects.
### f) Medicinal plants with significant antifertility activity
Although some herbal contraceptives have been developed, their potential for human use is limited. People are now searching for herbal remedies to combat a variety of illnesses and regulate fertility as a result of these issues (44). There are a number of preventive and corrective contraceptive methods available thanks to modern medicine, but none of them are particularly safe or free of major side effects. Drugs that are synthetic or chemically based have the potential to disrupt the endocrine system and have effects on the body's metabolism, development, neurological function, and reproduction. Natural hormone synthesis, secretion, transport, and activity may all be adversely affected by these substances. By preventing the synthesis and metabolism of hormones or by obstructing their action, they disrupt the normal level of hormones. Among them are Alkylphenols, bisphenol A, dioxins, heavy metals, fungicides, and insecticides prevent the synthesis of estrogen and progesterone, which impacts female sexual development by causing toxicity to the gonads, testicular germ cell cancer, breast/prostate cancer, and endometriosis. Pesticides, phthalates, and plasticizers also prevent the production of androgens, which impacts male sexual development. Other negative effects of these chemicals on the reproductive system have been demonstrated, including temporary or permanent infertility (45). These factors make it essential to create a highly effective, entirely herbal medication that doesn't negatively impact the reproductive system. Worldwide, over 35,000 plant species are utilized for medicinal purposes in a variety of human cultures. For primary healthcare, almost $80\%$ of people worldwide rely on traditional medicines, the majority of which use plant extracts (46). People have been using plants to treat illnesses and ease physical pain since ancient times. Many traditional medicines are now recognized for their effectiveness, reduced side effects, and improved cultural acceptability and compatibility with the human body. The need for the development of safe and effective herbal contraceptives Even the savages of ancient societies used herbal contraceptives to manage their fertility and avoid getting pregnant. Although some significant anti-fertility drugs (contraceptives) for women have been discovered by conventional medicine, their use and popularity among women are limited because of certain undesirable and problematic side effects. Obesity, cholelithiasis, stomach issues, breast and cervical cancer, asthma, and venous thromboembolism are among the frequent adverse effects (47).
Medical professionals are therefore looking for herbal contraceptives that are both safe and effective. Numerous plants have anti-fertility properties that have been scientifically proven. Both men and women may find these plants to be a useful source of herbal contraceptives. Due to their minimal or nonexistent adverse effects, plant products have caught the interest of numerous scientists as a major source of naturally occurring fertility-regulating agents. There have been reports of several plant extracts acting as antifertility agents (48). Given India's long-standing concerns about population growth, medicinal plants have been examined for their potential as contraceptives and antifertility effects. There are fewer options for effective, reversible, non-irritating, and highly expectable contraceptives available to men who are willing to share family planning responsibilities, and female contraceptive methods have always been given priority. Additionally, some herbs have been shown to disrupt the regular movement or production of sperm. Since every herb has a unique use, it's critical to have a basic understanding of how they are or might be used. Let's clarify the potential courses of action in more detail. Traditional herbal medicine-based sterilization techniques, such as abortion during the first few weeks, preventing conception, or rendering either partner sterile, are employed to regulate population growth rates. A review of the literature showed that, with the exception of gynecological disorders, herbal remedies that induce abortion, and plants that induce abortion, sufficient research has been done on the various medicinal uses of plants in this region (49). Numerous plant products have the potential to be developed into contraceptives by inhibiting both male and female fertility. Only a small number of native plants have been studied for their anti-fertility properties thus far, despite the fact that many of them have been demonstrated to prevent conception. The anti-fertility effects of a variety of medicinal plant extracts have been investigated in both males and females. Hormone levels were changed and spermicidal in some of these plants (50). Currently, there is a global effort to investigate the effectiveness of herbal products as a form of birth control (51). Synthetic drugs are losing ground to plant-based products. Their low toxicity and extensive exposure to these medications in traditional medical systems such as Ayurveda are the main reasons for this in recent years. Therefore, it is necessary to look for appropriate products made from local medicinal plants that can be used in place of pills. In an effort to reduce adverse effects and increase efficacy, the types and quantities of these ingredients have evolved over time (52). There are various ways that medicinal plants can cause infertility in females. In addition to interfering with implantation and sperm penetration, they may have an impact on the ovary, uterus, hormone production, and inhibition of hormonal action. Some of them create a protective layer around an egg to stop fertilization. Since antifertility plants are medications that prevent gametes from forming and disrupt the fertilization process, the plants can be categorized based on these actions. Ovulation is suppressed by antioestrogenic plants. These medications are administered by injection or by mouth. Anti-implantation plants stop fertilized ovum from attaching or penetrating the uterus. Abortionfacients The fetus is expelled early by plants (53). In females, the hypothalamus, anterior pituitary, ovary, oviduct, uterus, and vagina are the sites of action of antifertility medications. By releasing follicle-stimulating hormone (FSH) and luteinizing hormone (LH), the hypothalamus regulates the uterus's activity. Therefore, antifertility drugs may work at this level by interfering with the pituitary and/or hypothalamus's hormonal function or by blocking the neural pathway to the hypothalamus that regulates the release of hormones that release gonadotropin.
Male contraceptive options and progress are still limited and slow, despite significant advancements in the development of highly effective, acceptable, and reversible methods for females (13). New methods of male contraception must be developed in light of recent advancements in our understanding of male reproductive physiology. Numerous possible methods for causing infertility have been studied for a long time, including immunological, chemical, and hormonal methods. A variety of chemical groups, including steroidal and non-steroidal ones, have an impact on testicular function. These include melatonin, $\alpha$ -chlorohydrin, serotonin, levonorgestrel, depot medroxyprogesterone acetate (DMPA), cyproterone acetate (CPA), Danazol, and metapiron. However, their use has failed due to a number of risks, as they have been shown to be toxic or idiosyncratic in both short- and long-term use in the reproductive organs (54). Even though there are many different forms of contraception, finding newer, more effective ones is one of the most difficult tasks in the field of pharmaceutical and medical sciences. Exploration of the hidden wealth of medicinal plants for use as contraceptives has recently begun. A large portion of the global population still has access to herbal medicine as a common form of therapy for both illness treatment and health maintenance. Information about the screening of plants with antifertility efficacy has been steadily accumulating (55). The antifertility program can benefit from the knowledge found in folklore and ancient literature about plants and herbs. Many plants have been identified recently, and researchers have evaluated extracts and active ingredients from various plant parts, such as seeds, roots, leaves, flowers, stems, or stem barks (56).
## III. RESULTS
To investigate the traditional and folkloric uses of plants with antifertility properties, a thorough analysis of a large number of scientific peer-reviewed publications was carried out. Several plants that have been asserted and proven to have antifertility properties were included in the study. A list of plants that have been shown to have antifertility properties is provided below, along with information on the precise parts used and how they work.
## IV. DISCUSSION
Medicinal plants have been utilized for their therapeutic properties throughout history across various regions of the globe. In India and other countries, numerous medicinal plants are documented to exhibit antifertility effects (57).
This review aims to provide a comprehensive analysis of ethnopharmacological data concerning plant species utilized for the regulation of fertilization and conception by various tribes worldwide over recent decades. Table 3 includes the names of these plants, along with their respective families, the parts used, the animal models employed, and their mechanisms of action. As indicated in Table 3, the plants are categorized based on their effects as antifertility agents, with some exhibiting multiple properties that vary according to dosage. Furthermore, this review presents a compilation of plants that play a significant role in fertility control for both males and females. The literature survey revealed that among the different parts of plants, leaves are predominantly used for the purpose of controlling fertilization, while other parts such as fruits, stems, bark, roots, seeds, and flowers are utilized in lesser amounts (58).
## V. CONCLUSION
To sum up, this review has brought together data that has been verified by science about the phytochemical components and antifertility properties of medicinal plants that have been used for centuries. The results show that these medicinal plants' extracts have strong antifertility effects. Additionally, the findings show that the previously mentioned plants have dose-dependent antifertility effects.
## ACKNOWLEDGEMENTS
The authors wish to express their sincere gratitude to the Department of Zoology, Sri Venkateswara University, Tirupati, and Andhra Pradesh,
India, for providing necessary facilities to carry out this research work.
Conflict of Interest: The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Funding: The authors declared that this study has received no financial support.
Author Contributions: PVR, Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Resources, Software, Writing-original draft. MSR, Supervision, Validation, Visualization, Writing-review &editing. All authors have read and agree to the published version of the manuscript.
Ethical Approval: It is not applicable.
Institutional Review Board Statement: It is not applicable. Informed Consent Statement: It is not applicable.
Data Availability Statement: Data will be made available on request.
- albino rats. Indian Journal of Medical Research. 1969: 57: 237-244.
38. Abdulazeez MA, Mansurah DA, Ameh AD, Ahmadu D, Ibrahim S, Sani A, Ayo JO, Joseph O, Ambali SF & Suleiman F. Effect of fermented seed extract of Carica papaya on litters of female wistar rats. African Journal of Biotechnology., 2009; 8: 854.
39. Dusmanta Kumar Pradhan, Manas Ranjan Mishra, Ashutosh Mishra, Ashok Kumar Panda, Rajani Kanta Behera, Shivesh Jha and Sanjaya Choudhury. A comprehensive review of plants used as contraceptives. International Journal of Pharmaceutical Sciences and Research. 2012; 4(1): 148-155.
40. Williamson EM, Okpako DT, Evans FJ. Pharmacological methods in phytotherapy research: Selection preparation and pharmacological evaluation of plant material. John Wiley and Sons Ltd., London. 1996; 1:191-212.
41. Brinker F. Inhibition of endocrine function by botanical agents, antigonadotropic activity. British journal of phytotherapy. 1997; 4:123-145.
42. Prakash AO, Saxena V, Shukla S, Mathur R. Contraceptive potency of Pueraria tuberosa D.C. and its hormonal status. Acta Europaea fertilitatis. 1985; 16(1):59-65.
43. Turner DC. General endocrinology 4th ed. Tokyo, WB Saunders Company, Topan Company Ltd.
1971.
44. Patil SJ & Patil SB. Antiovulatory activity of petroleum ether extract of chromatographic fractions of Citrus medica seeds in albino rats. International Journal of Medical Sciences. 2013; 13(6): 410-417.
45. Schug TT, Janesick A, Blumber B & Heindela JJ. Endocrine disrupting chemicals and disease susceptibility. Journal of Steroid Biochemistry & Molecular Biology. 2011; 127: 204-15.
46. Kaur R, Sharma A, Kumar R & Kharb R. Rising Trends towards Herbal Contraceptives. Journal of Natural Product and Plant Resources. 2011; 1(4): 5-12.
47. Kumud Bala, Mahima Arya & Deepshikha Pandey
Katare. Herbal Contraceptive: An Overview. World Journal of Pharmacy and Pharmaceutical Sciences. 2014; 3(8): 1305-26.
48. Shah GM, Khan MA, Ahmad M, Zafar M & Khan AA. Observations on antifertility and abortifacient herbal drugs. African Journal of Biotechnology. 2009; 8(9): 1959-64.
49. Dehghan MH, Martin T & Dehghanan R. Antifertility effect of Iranian neem seed alcoholic extract on epididymal sperm of mice. Iranian Journal of Reproductive Medicine. 2005; 3(2): 83-89.
50. Mishra N, Joshi S, Tondon VL & Munjal A. Evaluation of Antifertility potential of aqueous extract of Bougainvillea spectabilis leaves in swiss albino
- mice. International Journal of Pharmaceutical Sciences and Drug Research. 2009; 1(1): 19-23.
51. Ahmad S, Jamal Y & Mannan A. Review of Some Medicinal Plants with Anti-fertility Activities. Unani Research. 2011; 1(2): 24-28.
52. Qureshi AA, Sanghai DB & Padgilwar SS. Herbal options for contraception: A review. Pharmacognosy Magazine. 2006; 2(8): 204-15.
53. Kabra M, Bhandari S, Gupta R & Sharma A. A review on herbal contraception. World Journal of Pharmacy and Pharmaceutical Sciences. 2013; 2(5): 2569-77.
54. Prasad MRN. Control of fertility in the male, In: Pharmacology and the future of man, Proceedings 5th International Congress of Pharmacology, San Francisco, Karger S, Basel, 1973; 1: 208-20.
55. Farnsworth NR, Bingel AS, Cordell GA, Crane FA & Fong HHS. Potential value of plants as source of new antifertility agents I. Journal of Pharmaceutical Sciences. 1975; 64: 535-49.
56. Zhu D. Plant constituents with biological activity reported in recent years. Zhongcaoyao. 1982; 13: 377-82.
57. Hiremanth SP, Rudresh K, Badami S, Patil SB, Patil SR. Post-coital antifertility activity of Acalypha indica L. Journal of Ethnopharmacology. 1999; 67(3):253-258.
58. Afsar Shaik, Prasanna Raju Yalavarthi and Chandrasekhar Kothapalli Bannoth. Role of Antifertility Medicinal Plants on Male & Female Reproduction. Journal of Complementary and Alternative Medical Research. 3(2): 1-22, 2017.
Table 1: List of Antifertility Plants with Chemical Constituents (29)
<table><tr><td>S.No.</td><td>Plant name</td><td>Common name</td><td>Type of extract</td><td>Plant part used</td><td>Activities</td><td>Chemical Constituents</td></tr><tr><td>1.</td><td>Abrus precatorius</td><td>Chirmi</td><td>Methanolic</td><td>Seed</td><td>Antifertility</td><td>Precatorine, trigonelline, choline, abrine abricin, abridin</td></tr><tr><td>2.</td><td>Acacia catechu</td><td>Katha</td><td>-</td><td>Exudate</td><td>Anti-implantation</td><td>-</td></tr><tr><td>3.</td><td>A. leucophloea</td><td>Reonja</td><td>Alcoholic</td><td>Root</td><td>Antifertility</td><td>Tannins, flavonoids, terpenes, alkaloids</td></tr><tr><td>4.</td><td>Acacia nilotica</td><td>Babul</td><td>Aqueous</td><td>Pod</td><td>Antispermatogenic</td><td>Phytosterols, phenolic compounds, saponins</td></tr><tr><td>5.</td><td>Azadirachta indica</td><td>Khokli</td><td>Petroleum ether and ethanolic</td><td>Whole plant</td><td>Post-coital activity</td><td>β-Sitosterol, acalyphine, triacetonamine, kaempferol, tannin, stigmasterol</td></tr><tr><td>6.</td><td>Achyranthus aspera</td><td>Chirchira</td><td>Ethanolic</td><td>Root</td><td>Anti-implantation</td><td>Ecdysterone, oleanolic acid, n-hexacos-14-enoic acid</td></tr><tr><td>7.</td><td>Adathoda vasica</td><td>Arusa</td><td>-</td><td>Leaves</td><td>Anti-implantation</td><td>Vasicine</td></tr><tr><td>8.</td><td>Aegle marmelos</td><td>Bael</td><td>Ethanolic</td><td>Leaves</td><td>Antifertility</td><td>Alkaloids, caumarins, steroids</td></tr><tr><td>9.</td><td>Aerva lanata</td><td>Bui</td><td>Ethanolic</td><td>Root</td><td>Anti-implantation</td><td>Alkaloids, kaempferol, quercetin, β-sitosteryl acetate, tannic acid</td></tr><tr><td>10.</td><td>Albizia lebbek</td><td>Siris</td><td>Methanolic</td><td>Pod</td><td>Spermicidal activity</td><td>Lebbekanin-E</td></tr><tr><td>11.</td><td>Ammania baccifera</td><td>Aginbuti</td><td>Ethanolic</td><td>Whole plant</td><td>Antisteroidogenic</td><td>Steroids, triterpenoids, Flavonoids, and tannins</td></tr><tr><td>12.</td><td>Amaranthus spinosus</td><td>Kanta chaulai</td><td>Acetone</td><td>Root</td><td>Anti-spermatogenic and anti-androgenic</td><td>Alkaloids, flavonoids, saponins, β-sitosterol, stigmasterol, Kaempferol, glycosides</td></tr><tr><td>13.</td><td>Amaramthu viridis</td><td>Jangli cholai</td><td>Aqueous</td><td>Root</td><td>Abortifacient</td><td>Alkaloids, anthraquinon, saponins</td></tr><tr><td>14.</td><td>Anagallis arvensis</td><td>Dhartidhak</td><td>-</td><td>Whole plant</td><td>Spermicidal activity</td><td>Oleanolic acid</td></tr><tr><td>15.</td><td>Andrographis paniculate</td><td>Kiryat</td><td>Dry leaf powder</td><td>Leaves</td><td>Antispermatogenic</td><td>Flavonoids, andrographilode, diterpenoids, phenylpropanoids, oleanolic acid, and β-sitosterol</td></tr><tr><td>16.</td><td>Aristolochia indica</td><td>Indian Birthwort</td><td>Ethanolic</td><td>Root</td><td>Antispermatogenic/anti-androgenic</td><td>Aristololic acid, p-coumric acid, methyl aristolate</td></tr><tr><td>17.</td><td>Argemone maxicana</td><td>Satyanashi</td><td>-</td><td>Seed</td><td>Anti-spermatogenic</td><td>Isoquinoline alkaloids, dihydro palmatine hydroxide, berberine, protopine</td></tr><tr><td>18.</td><td>Azardiracha indica</td><td>Neem</td><td>Alcoholic</td><td>Flower</td><td>Antifertility</td><td>Steroids, triterpenoids, alkaloids, phenolic compound, flavonoids</td></tr><tr><td>19.</td><td>Balanites aegyptiaca</td><td>Desert date</td><td>Methanolic</td><td>Bark</td><td>Antiimplantation</td><td>β-sitosterol, bergaptem, marmesin, β-sitosterol glucoside</td></tr><tr><td>20.</td><td>Balanites roxburghii</td><td>Desert date</td><td>Ethanolic</td><td>Fruit</td><td>Abortifacient</td><td>Alkaloids, saponins, tannins, flavonoids, phenolic compound</td></tr><tr><td>21.</td><td>Bbiophytum sensitivum</td><td>Lakshmana</td><td>Ethanolic</td><td>Whole plant</td><td>Antifertility activity</td><td>Phenolic and polyphenolic compound, saponins</td></tr><tr><td>22.</td><td>Boerhavia diffusa</td><td>Khapra-ara</td><td>Methanolic</td><td>Root</td><td>Antiimplantation, antiestrogenic</td><td>β-sitosterol, alkaloids, ursolic acid</td></tr><tr><td>23.</td><td>Butea monosperma</td><td>Dhak</td><td>Petroleum ether and Chloroform</td><td>Root</td><td>Anti-steroidogenic</td><td>Glycine, glycoside, aromatic hydroxyl compound</td></tr><tr><td>24.</td><td>Cajanus cajan (L)</td><td>Arhar</td><td>Methanolic</td><td>Seed</td><td>Antifertility</td><td>Sitosterol</td></tr><tr><td>25.</td><td>Calotropis gigantea</td><td>Madar</td><td>Ethanolic</td><td>Root</td><td>Anti-implantation</td><td>Akundarin, calotropin</td></tr><tr><td>26.</td><td>Calotropis</td><td>Aak</td><td>Ethanolic</td><td>Root</td><td>Anti-implantation</td><td>Alkaloids, flavonoids, tannins,</td></tr><tr><td></td><td>procera</td><td></td><td></td><td></td><td></td><td>saponins, and cardiac glycosides</td></tr><tr><td>27.</td><td>Capparis decidua(aphylla)</td><td>Kair</td><td>Ethanolic</td><td>Whole plant</td><td>Antispermatogenic</td><td>Capparin, capparilin, capparinin, sitosterol, n-triacontanol</td></tr><tr><td>28.</td><td>Cassia fistula</td><td>Amaltash</td><td>Aqueous</td><td>Seed</td><td>Antiestrogenic</td><td>Anthraquinone, glycosides, flavonoids, phenolic compound</td></tr><tr><td>29.</td><td>Cassia occidentalis</td><td>Kajondi</td><td>Ethanolic</td><td>Root</td><td>Anti-implantation and abortifacient</td><td>β-sitosterol, campesterol, emodin, 1,8-dihydroxyanthraquinone, quercetin</td></tr><tr><td>30.</td><td>Celsia cromandeliana</td><td>Kokhima</td><td>Methanolic</td><td>Aerial part</td><td>Antiovulatory</td><td>-</td></tr><tr><td>31.</td><td>Convolvulus arvensis</td><td>Field bindweed</td><td>Alcoholic</td><td>Aerial part</td><td>Antispermatogenic</td><td>α-amyrin, campesterol, stigmasterol, β-sitosterol, quercetin, kaemferol, p-caumaric acid</td></tr><tr><td>32.</td><td>Corchorus olitorius</td><td>Nalta jute</td><td>Methanolic</td><td>Seed</td><td>Antisteroidogenic</td><td>Hydrocyanin, cardiac glycosides, tannins, flavonoids, anthraquinones, saponins, Corchoroside A, helveticoside, corloside, digitoxigenin, peripogenin</td></tr><tr><td>33.</td><td>Cordia dichotoma</td><td>Lasora</td><td>Methanolic</td><td>Bark</td><td>Antiimplantation</td><td>α-amyrins, lupeol-3-rhamnoside, β-sitosterol, β-sitosterol-3-glucoside, toxifolin-3,5-dirhmnoside</td></tr><tr><td>34.</td><td>Crotalaria juncea</td><td>Sunnhemp</td><td>Petroleum ether, Benzene and ethanol</td><td>Seed</td><td>Antispermatogenic</td><td>Flavonoids, alkaloids, saponins, volatile oil</td></tr><tr><td>35.</td><td>Cuscuta reflexa</td><td>Amarbel</td><td>Methanolic</td><td>Stem</td><td>Anti steroidogeic</td><td>Kaemferol-3-o-glucoside quercetin, quercetin-3-o-glucoside</td></tr><tr><td>36.</td><td>dactylon</td><td>Durva</td><td>Aqueous</td><td>Whole plant</td><td>Anti-implantation</td><td>Flavonoids, tannins, phenolic compound</td></tr><tr><td>37.</td><td>Cyperus rotundus</td><td>Nut grass</td><td></td><td>Tuber</td><td>Antifertility</td><td>Tannins, flavonoids, coumarins, sterols</td></tr><tr><td>38.</td><td>Dactyloctenium aegypticum</td><td>Crowfoot grass</td><td>Ethanolic</td><td>Whole plant</td><td>Antifertility activity</td><td>Saponins, flavonoids, tannins, terpenoids, alkaloids</td></tr><tr><td>39.</td><td>Dalbergia sisso</td><td>Seesam</td><td>Ethanolic</td><td>Stem bark</td><td>Anti-spermatogenic</td><td>Isoflavones, flavone, β-amyrin, β-sitosterol, stigmasterol</td></tr><tr><td>40.</td><td>Datura metal</td><td>Datura</td><td>Acetone</td><td>Seed</td><td>antifertility</td><td>Saponins, flavonoids, tannins, glycosides, alkaloids, terpenoids</td></tr><tr><td>41.</td><td>Dendrophthoe falcata</td><td>Banda</td><td>Methanolic</td><td>Stem</td><td>Depression of spermatogenesis</td><td>β-amyrin-6-acetate, oleonic acid, β-sitosterol, stigmasterol</td></tr><tr><td>42.</td><td>Dolichos billorus</td><td>Kulattha</td><td>Acetone</td><td>Seed</td><td>Anti spermatogenic antiandrogenic</td><td>Isoflavone diglycoside, aglycone</td></tr><tr><td>43.</td><td>Emblica officinalis</td><td>Amala</td><td></td><td>Fruit</td><td>Abortifacient</td><td></td></tr><tr><td>44.</td><td>Feronia limonia</td><td>Wood apple</td><td>Ethanolic</td><td>Fruit pulp</td><td>Antispermatogenic</td><td>Polyphenols, phytosterols, saponins, tannin, coumarins, Triterpenoids</td></tr><tr><td>45.</td><td>Ficus benghalensis</td><td>Bargad</td><td>Ethanolic</td><td>Leaves</td><td>Suppression of the spermatogenesis</td><td>Tannins, flavonoids, steroids</td></tr><tr><td>46.</td><td>Ficus religiosa</td><td>Peepal</td><td>-</td><td>Fruit</td><td>Anti-implantation</td><td>n-hexadecanoic acid, 9,12-oct-adecadienoic acid, 9,12,15-octadecatrienoic acid, butyl 9,12,15-oct-adecatrienoate</td></tr><tr><td>47.</td><td>Gnaphalium indicum</td><td>Cudweed</td><td>Ethanolic</td><td>Whole plant</td><td>Anti-implantation</td><td>Luteolin, quercetin, quercetin-3-methyl ether</td></tr><tr><td>48.</td><td>Grangea maderaspatana</td><td>Mukhatari</td><td>Flavonoid extract</td><td>Whole plant</td><td>Anti-implantation</td><td>Sesquiterpenoids, γ-gurjunene, terpinyl acetate, hinesol</td></tr><tr><td>49.</td><td>Ipomoea fistulosa</td><td>Pink morning glory</td><td>Alcoholic</td><td>Plant without root</td><td>Postcoital antifertility</td><td>Alkaloids, glycosides, phenolics, tannins, phytosterols, flavonoids, saponins</td></tr><tr><td>50.</td><td>Mangifera indica</td><td>Mango</td><td>Methanolic</td><td>Leaves</td><td>Antispermatogenic</td><td>Saponin, anthraquinone, steroids, tannin, flavonoids</td></tr><tr><td>51.</td><td>Maytenus emarginate</td><td>Kankero</td><td>Methanolic</td><td>Leaves</td><td>Inhibition of spermatogenesis</td><td>Tannins, flavonoids, alkaloids, steroids</td></tr><tr><td>52.</td><td>Melia azedarach</td><td>Chinaberry</td><td>-</td><td>Seed</td><td>Abortifacient</td><td>Alkaloids, tannins, saponins, phenols, glycosides, steroids, terpenoids, flavonoids</td></tr><tr><td>53.</td><td>Mimosa pudica</td><td>Touch me not</td><td>-</td><td>Root</td><td>Contraception and abortion</td><td>Alkaloids, glycosides, steroids, flavonoids, phenols</td></tr><tr><td>54.</td><td>Nelumbo nucifera</td><td>Lotus</td><td>Ethanolic</td><td>Seed</td><td>Antiestrogenic</td><td>Alkaloids, flavonoids, ursane triterpenoid ester</td></tr><tr><td>55.</td><td>Nyctanthes arbortistis</td><td>Har singar</td><td>Methanolic</td><td>Stem bark</td><td>Antispermatogenic</td><td>Alkaloid, phytosterols, phenolics, tannins, flavonoids, saponins</td></tr><tr><td>56.</td><td>Ocimum</td><td>Shyam Tulsi</td><td>Hydroalcoholic</td><td>Leaves</td><td>Antifertility</td><td>Saponins, glycosides, alkaloids,</td></tr></table>
<table><tr><td></td><td>basilicum</td><td></td><td></td><td></td><td></td><td>tannins, and phenolic compounds</td></tr><tr><td>57.</td><td>Opuntia dillenii</td><td>Naagfani</td><td>Methanolic</td><td>Phylloclade</td><td>Antispermatogenic</td><td>Vitexin, isorhamnetin</td></tr><tr><td>58.</td><td>Purgularia deamia</td><td>Sagovani</td><td>Ethanolic</td><td>Stem, leaves</td><td>Antifertility</td><td>Flavonoids, terpenoids, steroids, alkaloids</td></tr><tr><td>59.</td><td>Polygonum glabrum</td><td>Neli</td><td>-</td><td>Root</td><td>Contraceptive</td><td>Sterol</td></tr><tr><td>60.</td><td>Portulaca oleracea</td><td>Purslane</td><td>Petroleum ether, chloroform, and ethanol crude</td><td>Aerial part</td><td>Abortifacient</td><td>Alkaloids, tannins, flavonoids, saponins, and triterpenoids</td></tr><tr><td>61.</td><td>Rivea hypocrateriform</td><td>Night glory</td><td>Ethanolic</td><td>Aerial part</td><td>Antiovulatory</td><td>Alkaloids, glycosides, saponins, tannins, phenolic compound</td></tr><tr><td>62.</td><td>Salvadora persic</td><td>Meswak</td><td>Aqueous</td><td>Leaf and stem</td><td>Antifertility</td><td>Octacosanol, 1-triacantanol, β-sitosterol, β-sitosterol-3-o-β-D-glycopyranoside</td></tr><tr><td>63.</td><td>Sida acuta</td><td>common wireweed</td><td>Ethanolic</td><td>Leaf</td><td>Antiimplantation</td><td>Alkaloids, steroids, glycosides, saponins, flavones, phenolic compound</td></tr><tr><td>64.</td><td>Syzygium cumini</td><td>Jamun</td><td>Alcoholic</td><td>Seed</td><td>Antispermatogenic</td><td>β-pinene, terpinolene, eugenol, rutin, quercetin,β-sitosterol</td></tr><tr><td>65.</td><td>Terminalia bellirica</td><td>Harad</td><td>Ethanolic</td><td>Bark</td><td>Anti-implantation</td><td>Phytosterols, flavonoids, phenolic comp., tannins</td></tr><tr><td>66.</td><td>Terminalia chebula</td><td>Harad</td><td>Acetone, Methanol, Ethanol, Aqueous</td><td>Bark</td><td>Antispermatogenic</td><td>Tannins, flavonoids, sterolstriterpenoids</td></tr><tr><td>67.</td><td>Tactona grandis</td><td>Teak</td><td>Petroleum ether</td><td>Stem</td><td>Antifertility</td><td>Lapachol</td></tr><tr><td>68.</td><td>Tamarindus indica</td><td>Imli</td><td>-</td><td>Fruit</td><td>abortifacient</td><td>-</td></tr><tr><td>69.</td><td>Tephrosia purpurea</td><td>Unhali</td><td>-</td><td>Seed</td><td>Purpurin, rutin</td><td>-</td></tr><tr><td>70.</td><td>Terminalia arjuna</td><td>Arjun tree</td><td>-</td><td>Bark</td><td>Antiimplantation, Abortifacient</td><td>Lupeol, oleanolic acid, arjuncic acid, arjunetin, arjunolitin</td></tr><tr><td>71.</td><td>Tinospora cordifolia</td><td>Giloya</td><td>Methanolic</td><td>Stem</td><td>Antifertility</td><td>Alkaloids, sesquiterpenoid, β-sitosterol, cordifolia, columbin</td></tr><tr><td>72.</td><td>Tribulus terrestris</td><td>Gokhru</td><td>-</td><td>Seed</td><td>Abortifacient</td><td>Alkaloids, flavonoids, saponins, tannins</td></tr><tr><td>73.</td><td>Vicoa indica</td><td>Banjhauri</td><td>-</td><td>Plant</td><td>Antiimplantation</td><td>Vicolid B, Vicolid D</td></tr><tr><td>74.</td><td>Wrightia tinctorial</td><td>Duhi</td><td>Ethanolic</td><td>Stem bark</td><td>Post-coital interceptive activity</td><td>Lupeol, stigmasterol, campesterol</td></tr><tr><td>75.</td><td>Zizyphus mauritiana</td><td>Ber</td><td>Aqueous, methanolic</td><td>Bark</td><td>Spermicidal</td><td>Mauritine A, B, oleonic acid, betulonic acid</td></tr></table>
Table 2: List of Antifertility Medicinal Plants (31)
<table><tr><td>Cichorium intybus</td><td>50% ethanolic extract</td><td>50</td><td>Anti-implantation</td></tr><tr><td>Cuscuta reflexa</td><td>Ethanolic extract</td><td>800</td><td>Anti-implantation</td></tr><tr><td>Rubia cordifolia</td><td>Ethanolic extract</td><td>250</td><td>Anti-implantation</td></tr><tr><td>Urtica dioica</td><td>Ethanolic extract</td><td>250</td><td>Anti-implantation</td></tr><tr><td>Abroma augusta</td><td>Petroleum ether</td><td>50</td><td>Anti-implantation</td></tr><tr><td>Curcuma longa</td><td>Petroleum ether</td><td>200</td><td>Anti-implantation</td></tr><tr><td>Plumbago rosea</td><td>Acetone extract</td><td>200</td><td>Anti-implantation</td></tr><tr><td>Aloe barbadensis</td><td>Aqueous extract</td><td>100</td><td>Anti-implantation</td></tr><tr><td>Abutilon indicum</td><td>50% aqueous methanolic extract</td><td>500</td><td>Anti-implantation</td></tr><tr><td>Artemisia vulgaris</td><td>Methanolic extract</td><td>300 and 600</td><td>Anti-implantation</td></tr></table>
Table 3: List of Medicinal Plants Reported to Possess Antifertility Effects (58)
<table><tr><td>S. no.</td><td>Name of the plant</td><td>Family</td><td>Part used</td><td>Animal model</td><td>Mechanism of action</td></tr><tr><td>1.</td><td>Abroma angusta Linn.</td><td>Sterculiaceae</td><td>Roots</td><td>Rat</td><td>Antiimplantation & Abortifacient</td></tr><tr><td>2.</td><td>Abrus precatorius Linn.</td><td>Fabaceae</td><td>Seeds</td><td>Rat</td><td>Reduced sperm motility, Post-testicular antifertility effect</td></tr><tr><td>3.</td><td>Acacia auriculaeformis A. Cunn.</td><td>Fabaceae</td><td>-</td><td>-</td><td>Sperm immobilizing effect</td></tr><tr><td>4.</td><td>Acacia caesia Wight & Arn</td><td>Leguminosae</td><td>Fruit</td><td>-</td><td>Immobilization of spermatozoa</td></tr><tr><td>5.</td><td>Acacia concinna DC</td><td>Fabaceae</td><td>Stem bark</td><td>Rat</td><td>Spermicidal and semen coagulating activities</td></tr><tr><td>6.</td><td>Acalypha indica Linn.</td><td>Euphorbiaceae</td><td>Whole plant</td><td>-</td><td>Anti-estrogenic activity</td></tr><tr><td>7.</td><td>Achillea millefolium Linn.</td><td>Asteraceae</td><td>Flowers</td><td>Mice</td><td>Antispermatogenic effect</td></tr><tr><td>8.</td><td>Achryanthus aspera Linn.</td><td>Amranthaceae</td><td>Root</td><td>Rat</td><td>Spermicidal action</td></tr><tr><td>9.</td><td>Actiniopteris dichotoma Kuhn</td><td>Pteridaceae</td><td>Whole plant</td><td>Rat</td><td>Antifertility effect</td></tr><tr><td>10.</td><td>Adhatoda vasica Nees Syn. Justice adhatoda L.</td><td>Acanthaceae</td><td>Leaves</td><td>Rat</td><td>Antiimplantation & Abortifacient</td></tr><tr><td>11.</td><td>Aegle marmelos Corr. Ex Roxb.</td><td>Rutaceae</td><td>Leaf</td><td>Rat</td><td>Resist process of spermatogenesis and decrease sperm motility</td></tr><tr><td>12.</td><td>Aerva lanata (L.) Juss. Ex. Shult</td><td>Amaranthaceae</td><td>Aerial parts</td><td>Rat</td><td>Antiimplantation effect</td></tr><tr><td>13.</td><td>Afromosia laxiflora (Baker) Harms</td><td>Fabaceae</td><td>Stem bark</td><td>Rat</td><td>Antigonadotropic activity and blocks oestrous cycle</td></tr><tr><td>14.</td><td>Ailanthus excelsa Roxb.</td><td>Simaroubaceae</td><td>Leaf, Stem, Bark</td><td>Rat</td><td>Antiimplantation effect and Early Abortifacient</td></tr><tr><td>15.</td><td>Alangium Salvifolium (L.f.)</td><td>Alangiaceae</td><td>Stem, Bark</td><td>Rat</td><td>Antiimplantation & Abortifacient</td></tr><tr><td>16.</td><td>Albizia procera (Roxb.) Benth.</td><td>Leguminosae</td><td>Seed and Root</td><td>Rat</td><td>Spermicidal and semen coagulating activities</td></tr><tr><td>17.</td><td>Albizia lebbek (Linn.) Benth.</td><td>Mimosacaeae</td><td>Pod, Bark</td><td>Rat</td><td>Antifertility activity</td></tr><tr><td>18.</td><td>Allium cepa Linn.</td><td>Liliaceae</td><td>Bulb</td><td>Rat</td><td>Antiimplantation activity</td></tr><tr><td>19.</td><td>Allium sativum Linn.</td><td>Amaryllidaceae</td><td>Pod</td><td>Rat</td><td>Antispermatogenic activity</td></tr><tr><td>20.</td><td>Aloe barbadensis Mill. Syn. Acalypha indica, A. littoralis, A. vera</td><td>Liliaceae</td><td>Leaves</td><td>Dog</td><td>Antiandrogenic activity</td></tr><tr><td>21.</td><td>Alstonia scholars R.Br.</td><td>Apocynaceae</td><td>Stem bark</td><td>Rat</td><td>Antifertility activity</td></tr><tr><td>22.</td><td>Amaranthus spinous Linn.</td><td>Amaranthaceae</td><td>Root</td><td>Rat</td><td>Inhibit fusion of Sperm and Ovum</td></tr><tr><td>23.</td><td>Amaranthus viridis L.</td><td>Amaranthaceae</td><td>Root</td><td>Rat</td><td>Contraception Activity</td></tr><tr><td>24.</td><td>Anacardium occidentale Linn.</td><td>Anacardiaceae</td><td>Nut Shell</td><td>Rat</td><td>Spermicidal</td></tr><tr><td>25.</td><td>Anagalis arvensis Linn.</td><td>Primulaceae</td><td>Whole Plant</td><td>Rat</td><td>Spermicidal and semen coagulating activities</td></tr><tr><td>26.</td><td>Ananas comosus Merr.</td><td>Bromeliaceae</td><td>Unripe fruit</td><td>Rat</td><td>Antispermatogenic activity</td></tr><tr><td>27.</td><td>Andrographis paniculata Wall. Ex Nees</td><td>Acanthaceae</td><td>Leaves</td><td>Rat</td><td>Antispermatogenic and antiandrogenic</td></tr><tr><td>28.</td><td>Arctium lappa Linn.</td><td>Asteraceae</td><td>Leaves and roots</td><td>Rat</td><td>Abortifacient</td></tr><tr><td>29.</td><td>Ardisia solanacea Roxb.</td><td>Myrsinacea</td><td>Plants excluding roots</td><td>Rat</td><td>Spermicidal Activity</td></tr><tr><td>30.</td><td>Aristolochia indica Linn.</td><td>Aristolochiaceae</td><td>Root</td><td>Presbytes langur</td><td>Antispermatogenic and antiandrogenic</td></tr><tr><td>31.</td><td>Artemisia atra Jacq. Ex Wild.</td><td>Asteraceae</td><td>Leaf</td><td>Rats</td><td>Abortion</td></tr><tr><td>32.</td><td>Artemisia vulgaris Linn.</td><td>Asteraceae</td><td>Leaves</td><td>Rats</td><td>Antiimplantation and Estrogenic activity</td></tr><tr><td>33.</td><td>Aspilia Africana (pers.) C.D. Adams</td><td>Asteraceae</td><td>Leaves</td><td>Rats</td><td>Antiovulatory Activity</td></tr><tr><td>34.</td><td>Austroplenckia populnea (Reiss.) Lundell.</td><td>Celastraceae</td><td>Pods</td><td>Rats</td><td>Affects the sexual behavior and epididymal sperm concentration</td></tr><tr><td>35.</td><td>Azardirachta indica A. Juss.</td><td>Maliaceae</td><td>Seed Oil</td><td>Rats</td><td>Antispermatogenic and antiandrogenic</td></tr><tr><td>36.</td><td>Bacopa monnieri (L.) Pennell</td><td>Scrophulariaceae</td><td>Whole plant</td><td>Rats</td><td>Contraception Activity</td></tr><tr><td>37.</td><td>Balanites roxburghii Linn.</td><td>Zygophyllaceae</td><td>Fruits</td><td>Dog</td><td>Antispermatogenic activity and testicular necrosis and atrophy</td></tr><tr><td>38.</td><td>Ballota undulate (Sieber ex. Fresen.) Benth.</td><td>Labiatae</td><td>Leaves, Flowers</td><td>Rats</td><td>Antimplantation activity</td></tr><tr><td>39.</td><td>Bambusa arundinacea Willd.</td><td>Graminae</td><td>Shoots, Stem</td><td>Rats</td><td>Impaired the structural and functional activity of epididymis, Reduced sperm motility</td></tr><tr><td>40.</td><td>Barleria prionitis Linn.</td><td>Acanthaceae</td><td>Roots</td><td>Rat</td><td>Antifertility effect</td></tr><tr><td>41.</td><td>Berberis chitria Buch.-Ham.ex Lindl.</td><td>Berberidaceae</td><td>Root</td><td>Dog</td><td>Antispermatogenic activity</td></tr><tr><td>42.</td><td>Biophytum sensitum (L.) DC.</td><td>Oxalidaceae</td><td>Leaves</td><td>Rats</td><td>Antimplantation Activity</td></tr><tr><td>43.</td><td>Bougainvillea Comm. Ex Juss.</td><td>Nyctaginaceae</td><td>Leaves</td><td>Rats</td><td>Antifertility effect</td></tr><tr><td>44.</td><td>Butea monosperma (Lam.) Kuntze</td><td>Fabaceae</td><td>Seed</td><td>Rat, Dog</td><td>Effects on testicular function</td></tr><tr><td>45.</td><td>Calotropis procera (Ait.) R. Br.</td><td>Asclepiadaceae</td><td>Roots</td><td>Rabbit, Mice</td><td>Antispermatogenic effect and leydig cell atrophy Functional alteration in the genital organs and inhibition of fertility</td></tr><tr><td>46.</td><td>Cananga odorata (Lam.) Hook. F. & Thomson</td><td>Annonaceae</td><td>Root, Bark</td><td>Rat</td><td>Spermicidal Activity</td></tr><tr><td>47.</td><td>Cannabis sativa Linn.</td><td>Cannabaceae</td><td>Leaves</td><td>Presbytis Monkey</td><td>Testicular lesions and atrophy of Leydig cells</td></tr><tr><td>48.</td><td>Cardiospermum Helicacabum L</td><td>Spindaceae</td><td>Whole plant</td><td>Rat</td><td>Antimplantation activity</td></tr><tr><td>49.</td><td>Carica papaya Linn.</td><td>Caricaceae</td><td>Fruit</td><td>Rat</td><td>Antispermatogenic activity</td></tr><tr><td>50.</td><td>Carum carvi Linn.</td><td>Apiaceae</td><td>Rhizome</td><td>Rat</td><td>Antioestrogenic activity</td></tr><tr><td>51.</td><td>Cassis fistula Linn.</td><td>Caesalpiniaceae</td><td>Pods, Seeds</td><td>Rat</td><td>Antioestrogenic activity</td></tr><tr><td>52.</td><td>Catharanthus roseus G. Don syn. Vinca rosea Linn.</td><td>Apocynaceae</td><td>Leaves</td><td>Mice</td><td>Antioestrogenic activity</td></tr><tr><td>53.</td><td>Celastrus paniculatus Willd.</td><td>Celastraceae</td><td>Seeds</td><td>Rat</td><td>Antispermatogenic action</td></tr><tr><td>54.</td><td>Cicer arietinum Linn.</td><td>Fabaceae</td><td>Seeds</td><td>Rat</td><td>Abortifacient and estrogenic activity</td></tr><tr><td>55.</td><td>Cichorium intybus Linn.</td><td>Asteraceae</td><td>Whole plant</td><td>Rat</td><td>Antispermatogenic activity</td></tr><tr><td>56.</td><td>Cinnamomum</td><td>Lauraceae</td><td>Seed</td><td>Sparrow</td><td>Arrest and inhibition of spermatogenesis</td></tr><tr><td>57.</td><td>Camphora Nees & Eberm.</td><td></td><td></td><td></td><td></td></tr><tr><td>58.</td><td>Cissampelos pareira Linn.</td><td>Menispermaceae</td><td>Leaves</td><td>Mice</td><td>Antioestrogenic activity</td></tr><tr><td>59.</td><td>Citrullus colocynthis Schrad.</td><td>Cucurbitaceae</td><td>Fruit, Root</td><td>Rat</td><td>Induced reversible antifertility effects and Antispermatogenic effect</td></tr><tr><td>60.</td><td>Clerodendrum serratum L.</td><td>Lamiaceae/Verb enaceae</td><td>Whole plant (Excluding Roots)</td><td>Rats</td><td>Spermicidal activity</td></tr><tr><td>61.</td><td>Cnidoscolous aconitifolius (Mill.)J.M.Johnst.</td><td>Euphorbiaceae</td><td>Leaves</td><td>Rats</td><td>Contraception</td></tr><tr><td>62.</td><td>Cola nitida Schott & Endl.</td><td>Sterculiaceae</td><td>Stem Bark</td><td>Rats</td><td>Antigonadotropic activity and</td></tr><tr><td>63.</td><td>Colebrookia oppositifolia Sm.</td><td>Lamiaceae</td><td>Leaf</td><td>Rats</td><td>Antifertility Effect</td></tr><tr><td>64.</td><td>Combretodendron macrocarpum (P.Beauv.) Keay</td><td>Barringtoniaceae</td><td>Stem bark</td><td>Rats</td><td>Antigonadotropic activity and</td></tr><tr><td>65.</td><td>Convolvulus microphyllus Sieb. ex Spreng</td><td>Convolvulaceae</td><td>Whole Plant</td><td>Rat</td><td>Antispermatogenic effect</td></tr><tr><td>66.</td><td>Crataeva nurvala Buch.Ham.</td><td>Capparidaceae</td><td>Stem Bark</td><td>Rat</td><td>Antiimplantation and Antioestrogenic activity</td></tr><tr><td>67.</td><td>Crotalaria juncea Linn.</td><td>Papilionaceae</td><td>Seeds</td><td>Mice</td><td>Antifertility Activity, Arrest of spermatogenesis and antiandrogenic Effect</td></tr><tr><td>68.</td><td>Croton roxburghii Balak.</td><td>Euphorbiaceae</td><td>Bark</td><td>Mouse</td><td>Anti-steroidogenic activity</td></tr><tr><td>69.</td><td>Cumifiga racemosa L.</td><td>Apocyanaceae</td><td>Root</td><td>Rats</td><td>Spermatogenesis</td></tr><tr><td>70.</td><td>Cuminum cyminum Linn.</td><td>Apiaceae</td><td>Seed</td><td>Rat</td><td>Antispermatogenic effect</td></tr><tr><td>71.</td><td>Curcuma aromatica Salisb.</td><td>Zingiberaceae</td><td>Rhizome</td><td>Rats</td><td>Antifertility Activity</td></tr><tr><td>72.</td><td>Curcuma longa Linn.</td><td>Zingiberaceae</td><td>Root</td><td>Rats</td><td>Interference with Spermatogenesis</td></tr><tr><td>73.</td><td>Cyclamen persicum Mill.</td><td>Primulaceae</td><td>Whole Plant</td><td>-</td><td>Spermicidal activity</td></tr><tr><td>74.</td><td>Cyclea burmanni Miers</td><td>Menispermaceae</td><td>Roots</td><td>Rat</td><td>Decrease Sperm Count</td></tr><tr><td>75.</td><td>Cynomorum coccineum Linn.</td><td>Cynomoraceae</td><td>Inner pulp of stem and root</td><td>Rats</td><td>Effect on epididymal sperm pattern</td></tr><tr><td>76.</td><td>Daucus Carota Linn.</td><td>Apiaceae</td><td>Seeds</td><td>Rat</td><td>Blastocystotoxic and Antiplantation effects; Postcoital contraceptive effects</td></tr><tr><td>77.</td><td>Dendrophthoe falcate (Linn. f.)</td><td>Loranthaceae</td><td>Aerial parts</td><td>Rats</td><td>Antifertility effect</td></tr><tr><td>78.</td><td>Derris brevipes Baker.</td><td>Fabaceae</td><td>Root Powder</td><td>Rats</td><td>Abortifacient</td></tr><tr><td>79.</td><td>Desmodium gangeticum DC.</td><td>Fabaceae</td><td>Whole plant</td><td>Rat</td><td>Antifertility effect</td></tr><tr><td>80.</td><td>Dioscorea bulbifera L.</td><td>Dioscoreaceae</td><td>Tuber</td><td>-</td><td>Contraceptive</td></tr><tr><td>81.</td><td>Diploclasia echinatus Linn.</td><td>Asteraceae</td><td>Stem</td><td>-</td><td>Spermicidal</td></tr><tr><td>82.</td><td>Dipsacus mitis D.Don</td><td>Spindaceae</td><td>Root</td><td>Hamster</td><td>Contraceptive</td></tr><tr><td>83.</td><td>Ecballium elaterium A. Rich.</td><td>Cucurbitaceae</td><td>-</td><td>Rabbit</td><td>Decreases sperm motility</td></tr><tr><td>84.</td><td>Echeveria gibbiflora DC</td><td>Crassulaceae</td><td>Whole plant</td><td>Guinea Pig</td><td>Decreased sperm motility</td></tr><tr><td>85.</td><td>Echinops echinatus Roxb.</td><td>Asteraceae</td><td>Root</td><td>Rat</td><td>Sperm antimotility</td></tr><tr><td>86.</td><td>Embelia Ribes Burm.f.</td><td>Myrsinaceae</td><td>Berry</td><td>Rat</td><td>Antifertility activity</td></tr><tr><td>87.</td><td>Epilobium angustifolium Linn.</td><td>Onagraniiaceae</td><td>-</td><td>Rat</td><td>Reduction in weight of accessory sex organs</td></tr><tr><td>88.</td><td>Eupatorium odoratum Linn.</td><td>Asteraceae</td><td>-</td><td>-</td><td>Spermicidal activity</td></tr><tr><td>89.</td><td>Euphorbia neriifolia Linn.</td><td>Euphorbiaceae</td><td>Root</td><td>Rat</td><td>Antispermatogenic effects</td></tr><tr><td>90.</td><td>Eugenia jambolana L.</td><td>Myrtaceae</td><td>Flowers</td><td>Rat</td><td>Antifertility effect</td></tr><tr><td>91.</td><td>Ehretia cymosa Thonn.</td><td>Boraginaceae</td><td>Leaf, Bark</td><td>-</td><td>Contraceptive</td></tr><tr><td>92.</td><td>Eleutherine bulbosa Urb.</td><td>Iridaceae</td><td>Bulb</td><td>Rat</td><td>Abortifacient</td></tr><tr><td>93.</td><td>Fevillea passiflora Vell.</td><td>Cucurbitaceae</td><td>Seed</td><td>-</td><td>Abortifacient</td></tr><tr><td>94.</td><td>Ferula assa-foetida Linn.</td><td>Apiaceae</td><td>Resin</td><td>-</td><td>Emmenagogue</td></tr><tr><td>95.</td><td>Ficus religiosa Linn.</td><td>Moraceae</td><td>Fruit</td><td>Goat</td><td>Anti-implantation</td></tr><tr><td>96.</td><td>Ficus wassa Roxb.</td><td>Moraceae</td><td>Root</td><td>-</td><td>Contraceptive</td></tr><tr><td>97.</td><td>Flagellaria indica Linn.</td><td>Flagellariaceae</td><td>Leaf</td><td>-</td><td>Contraceptive</td></tr><tr><td>98.</td><td>Flemingia strobulifera (L.) J. St. Hill syn. Moghania strobulifera (L.) J. St.-Hill.</td><td>Fabaceae</td><td>Seed</td><td>-</td><td>Contraceptive</td></tr><tr><td>99.</td><td>Fleura aestuans Linn.</td><td>Utricaceae</td><td>Root</td><td>-</td><td>Abortifacient</td></tr><tr><td>100.</td><td>Foeniculum vulgare Mill.</td><td>Apiaceae</td><td>Seed</td><td>Rat</td><td>Sperm toxic</td></tr><tr><td>101.</td><td>Fragaria vesca Linn.</td><td>Rosaceae</td><td>Leaf</td><td>-</td><td>-</td></tr><tr><td>102.</td><td>Fransenia artemisiodes Willd.</td><td>Asteraceae</td><td>Whole plant</td><td>-</td><td>Contraceptive</td></tr><tr><td>103.</td><td>Galium mexicanum Var.</td><td>Rubiaceae</td><td>Leaves</td><td>Cat</td><td>Abortifacient</td></tr><tr><td>104.</td><td>Garcinia tambogia Desr.</td><td>Clusiaceae</td><td>Fruit</td><td>Rat</td><td>Testicular atrophy</td></tr><tr><td>105.</td><td>Gardenia jasminoides Ellis.</td><td>Rubiaceae</td><td>Fruits</td><td>-</td><td>Abortifacient</td></tr><tr><td>106.</td><td>Gloriosa superb Linn.</td><td>Liliaceae</td><td>Roots</td><td>Rat, mice</td><td>Oxytocic activity, Abortifacient</td></tr><tr><td>107.</td><td>Glossocardia bosvallia DC.</td><td>Asteraceae</td><td>Whole plant</td><td>-</td><td>Emmenagogue</td></tr><tr><td>108.</td><td>Glycyrrhiza glabra Linn.</td><td>Fabaceae</td><td>Root</td><td>-</td><td>Emmenagogue</td></tr><tr><td>109.</td><td>Gossypium barbadense Linn.</td><td>Malvaceae</td><td>Cotton Seed</td><td>rat</td><td>Testicular</td></tr><tr><td>110.</td><td>Grewia columnaris Sm.</td><td>Trillaceae</td><td>Root</td><td>-</td><td>Sterilizer</td></tr><tr><td>111.</td><td>Hagenia abyssinica.syn. Brayera anthalmintica</td><td>Rosaceae</td><td>-</td><td>-</td><td>Abortifacient</td></tr><tr><td>112.</td><td>Haematoxylon campechianum L.</td><td>Fabaceae</td><td>Whole plant</td><td>-</td><td>Abortifacient</td></tr><tr><td>113.</td><td>Hamelia erecta Jacq</td><td>Rubiaceae</td><td>Leaf</td><td>-</td><td>Abortifacient</td></tr><tr><td>114.</td><td>Hedeoma pulegoides Linn.</td><td>Labiateae</td><td>Plant without root</td><td>-</td><td>Contraceptive and Abortifacient</td></tr><tr><td>115.</td><td>Hedera helix Linn.</td><td>Araliaceae</td><td>Fruit</td><td>-</td><td>Contraceptive</td></tr><tr><td>116.</td><td>Hibiscus rosa-sinensis Linn.</td><td>Malvaceae</td><td>Root</td><td>Rats & Mice</td><td>Anti-implantation & Uterotropic activity</td></tr><tr><td>117.</td><td>Hyptis suaveolens Poit.</td><td>Labiateae</td><td>Whole plant</td><td>Mice</td><td>Antifertility</td></tr><tr><td>118.</td><td>Hypochoeris brasiliensis (Less.) Benth</td><td>Asteraceae</td><td>Leaf & Root</td><td>-</td><td>Contraceptive</td></tr><tr><td>119.</td><td>Hypericum chinensis Linn.</td><td>Clusiaceae</td><td>Leaf</td><td>-</td><td>Emmenagogue</td></tr><tr><td>120.</td><td>Hymenaea stigonocarpa Mart. Ex Hayne</td><td>Fabaceae</td><td>Bark</td><td>-</td><td>Contraceptive</td></tr><tr><td>121.</td><td>Indigofera linnaei Ali</td><td>Fabaceae</td><td>Herb</td><td>rats</td><td>Anti-fertility activity</td></tr><tr><td>122.</td><td>Jacaranda copaia (Aublet.) D. Don</td><td>Bignoniaceae</td><td>Tuber</td><td>-</td><td>Contraceptive</td></tr><tr><td>123.</td><td>Jasminum multiflorum (Burm.f.) Andrews</td><td>Oleaceae</td><td>-</td><td>-</td><td>Emmenagogue</td></tr><tr><td>124.</td><td>Jodinia rhombifolia (Hook. & Arn.) Reissek.</td><td>Santalaceae</td><td>Leaf</td><td>-</td><td>Abortifacient</td></tr><tr><td>125.</td><td>Juglans regia Linn.</td><td>Juglandaceae</td><td>Leaf</td><td>-</td><td>Contraceptive</td></tr><tr><td>126.</td><td>Juniperus communis Linn.</td><td>Cupressaceae</td><td>Stem & Fruit</td><td>-</td><td>Anti-implantation activity</td></tr><tr><td>127.</td><td>Juniperus oxycedrus Linn.</td><td>Cupressaceae</td><td>Berry</td><td>-</td><td>Abortifacient</td></tr><tr><td>128.</td><td>Justicia simplex D. Don</td><td>Acanthaceae</td><td>Root</td><td>-</td><td>Contraceptive</td></tr><tr><td>129.</td><td>Kopsia SP</td><td>Apocynaceae</td><td>Leaf</td><td>-</td><td>Contraceptive</td></tr><tr><td>130.</td><td>Laurus nobilis Linn.</td><td>Lauraceae</td><td>Leaf</td><td>Rats</td><td>Testicular dysfunction</td></tr><tr><td>131.</td><td>Lawsonia inermis Linn. syn. L. alba</td><td>Lythraceae</td><td>Leaves</td><td>rats</td><td>Abortifacient</td></tr><tr><td>132.</td><td>Leonotis nepetaefolia R.Br.</td><td>Labiatae</td><td>Leaf</td><td>Rats</td><td>Anti-implantation</td></tr><tr><td>133.</td><td>Lepidium meyenii Walp.</td><td>Brassicaceae</td><td>Root</td><td>Rats</td><td>invigorates spermatogenesis in male rats</td></tr><tr><td>134.</td><td>Lepidium sativum Linn.</td><td>Brassicaceae</td><td>Herb</td><td>-</td><td>Abortifacient & Anti-Ovulatory</td></tr><tr><td>135.</td><td>Licuala SP.</td><td>Arecaceae</td><td>Root bark</td><td>-</td><td>Contraceptive</td></tr><tr><td>136.</td><td>Ligusticum porter Coult. And Rose</td><td>Apiaceae</td><td>Root</td><td>-</td><td>Emmenagogue</td></tr><tr><td>137.</td><td>Lithospermum officinale Linn.</td><td>Broaginaceae</td><td>Leaves</td><td>Rat</td><td>Inhibition of hypophyseal hormone secretion</td></tr><tr><td>138.</td><td>Lobelia nicotianifolia Heyne</td><td>Campanulaceae</td><td>Whole plant</td><td>-</td><td>Contraceptive</td></tr><tr><td>139.</td><td>Lonicera ciliosa</td><td>Caprifoliaceae</td><td>Leaf</td><td>-</td><td>Contraceptive</td></tr><tr><td>140.</td><td>Malvaviscus conzattii Greenm</td><td>Malvaceae</td><td>Flower</td><td>Albino Mice</td><td>Antifertility activity</td></tr><tr><td>141.</td><td>Martynia annua Linn.</td><td>Martyniacceae</td><td>Root</td><td>Rats</td><td>Antifertility Effect</td></tr><tr><td>142.</td><td>Melodirus fusiformis Champ. Ex Benth.</td><td>Apocynaceae</td><td>-</td><td>-</td><td>Spermicidal Effect</td></tr><tr><td>143.</td><td>Mentha arvensis Linn.</td><td>Labiatae</td><td>Leaves</td><td>Rabbits</td><td>Anti-Ovulatory</td></tr><tr><td>144.</td><td>Millettia auriculata Baker. ex, Brand.</td><td>Fabaceae</td><td>Leaves</td><td>Rat</td><td>Anti-Implantation effect</td></tr><tr><td>145.</td><td>Momordica charantia Linn.</td><td>Cucurbitaceae</td><td>Seeds</td><td>Rats</td><td>Antispermatogenic</td></tr><tr><td>146.</td><td>Mondia whiteii Skeels</td><td>Apocynaceae</td><td>Root bark</td><td>Rat</td><td>Antispermatogenic & Anti fertility activities</td></tr><tr><td>147.</td><td>Mucuna urens Medik.</td><td>Fabaceae</td><td>Seed</td><td>Rat</td><td>Antispermatogenic</td></tr><tr><td>148.</td><td>Myristica fragrans Houtt</td><td>Myristiacaceae</td><td>Seed</td><td>-</td><td>Abortifacient</td></tr><tr><td>149.</td><td>Mesua ferrea Linn.</td><td>Clusiaceae</td><td>Flowers</td><td>Rat</td><td>Anti-implantation</td></tr><tr><td>150.</td><td>Nardostachys jatamansi DC.</td><td>Valerianaceae</td><td>Root</td><td>-</td><td>Emmenagogue</td></tr><tr><td>151.</td><td>Nasturtium officinalis R.Br.</td><td>Brassicaceae</td><td>Whole Plant</td><td>-</td><td>Abortifacient</td></tr><tr><td>152.</td><td>Nerium indicum Mill.</td><td>Aocynaceae</td><td>Whole Plant</td><td>-</td><td>Emmenagogue</td></tr><tr><td>153.</td><td>Nicotiana tabacum Linn.</td><td>Solanaceae</td><td>Leaves</td><td>Rat</td><td>Antiandrogenic effects</td></tr><tr><td>154.</td><td>Nigella sativa Linn.</td><td>Ranunculaceae</td><td>Seeds</td><td>Rat</td><td>Post-Coital Antifertility effect</td></tr><tr><td>155.</td><td>Nothocnide repanda (Bl.) Bl.</td><td>Ultricaceae</td><td>Leaf</td><td>-</td><td>Abortifacient</td></tr><tr><td>156.</td><td>Ochna jabotapita Linn.</td><td>Ochnaceae</td><td>Plant (Without</td><td>-</td><td>Semen coagulating activity</td></tr><tr><td>157.</td><td>Ocimum sanctum Linn.</td><td>Labiatae</td><td>Leaves</td><td>Rats</td><td>Antiandrogenic Property</td></tr><tr><td>158.</td><td>Olea europea Linn.</td><td>Oleaceae</td><td>Fruit</td><td>Rats</td><td>Contraceptive</td></tr><tr><td>159.</td><td>Ophiopogon intermedius (D.Don) Maxim</td><td>Asparagaceae</td><td>Rhizomes</td><td>-</td><td>Spermicidal</td></tr><tr><td>160.</td><td>Opuntia dilleni Haw.</td><td>Cactaceae</td><td>Phylloclade</td><td>Rats</td><td>Spermatotoxic</td></tr><tr><td>161.</td><td>Origanum vulgare Linn.</td><td>Labiatae</td><td>-</td><td>-</td><td>Abortifacient</td></tr><tr><td>162.</td><td>Oxalis physocalyx Zucc.ex Progel</td><td>Oxalidaceae</td><td>Whole Plant</td><td>-</td><td>Abortifacient</td></tr><tr><td>163.</td><td>Oxytenanthera abyssinica Munero</td><td>Poaceae</td><td>Leaf</td><td>-</td><td>Abortifacient</td></tr><tr><td>164.</td><td>Papaver somniferum Linn.</td><td>Papaveraceae</td><td>Fruit</td><td>-</td><td>Induces Abortion</td></tr><tr><td>165.</td><td>Peganum harmala Linn.</td><td>Zygophyllaceae</td><td>Epigeal Plants</td><td>Rats</td><td>Abortifacient</td></tr><tr><td>166.</td><td>Petrocarpus santalinus Linn.f.</td><td>Fabaceae</td><td>Stem Bark</td><td>Rats</td><td>Anti-implantation activity</td></tr><tr><td>167.</td><td>Piper longum Linn.</td><td>Piperaceae</td><td>Fruit</td><td>Rats</td><td>Antifertility Activity</td></tr><tr><td>168.</td><td>Pittosporum neelgherrense Wight & Arn.</td><td>Pittosporaceae</td><td>Plant (Without Root)</td><td>Rats</td><td>Spermicidal and Semen Coagulation</td></tr><tr><td>169.</td><td>Plumbago zeylanica Linn.</td><td>Plumbaginaceae</td><td>Leaves & Root</td><td>Rats</td><td>oestrogenic activity</td></tr><tr><td>170.</td><td>Plumeria rubra Linn.</td><td>Apocynaceae</td><td>Pod Extract</td><td>Rats</td><td>Anti-implantation activity</td></tr><tr><td>171.</td><td>Polemonium caeruleum Linn.</td><td>Polemoniacceae</td><td>-</td><td>-</td><td>Antispermatogenic effect</td></tr><tr><td>172.</td><td>Primula vulgaris Huds.</td><td>Primulaceae</td><td>-</td><td>-</td><td>Spermicidal effect</td></tr><tr><td>173.</td><td>Pueraria tuberose DC.</td><td>Fabaceae</td><td>Tubers</td><td>Rats</td><td>Antifertility activity</td></tr><tr><td>174.</td><td>Portulaca oleracea Linn.</td><td>Portulacaceae</td><td>Seed</td><td>Mice</td><td>Impairement of Spermatogenesis</td></tr><tr><td>175.</td><td>Pyrus cuspidata Bertol</td><td>Rosaceae</td><td>Whole Plant</td><td>-</td><td>Spermicidal effect</td></tr><tr><td>176.</td><td>Quassia amara Linn.</td><td>Simaroubaceae</td><td>Stem wood</td><td>Rats</td><td>Antifertility activity</td></tr><tr><td>177.</td><td>Randia dumetorum Lamk.</td><td>Rubiaceae</td><td>-</td><td>-</td><td>Anti-implantation effect</td></tr><tr><td>178.</td><td>Randia spinosa (Thumb.) Bl.</td><td>Rubiaceae</td><td>Fruit</td><td>-</td><td>Antifertility activity</td></tr><tr><td>179.</td><td>Ranunculus sceleratus Linn.</td><td>Ranunculaceae</td><td>Whole Plant</td><td>-</td><td>Antifertility activity</td></tr><tr><td>180.</td><td>Rauwolfia serpentine Benth.</td><td>Apocynaceae</td><td>Root</td><td>-</td><td>Antifertility activity</td></tr><tr><td>181.</td><td>Rhamnus catharticus Linn.</td><td>Rhamnaceae</td><td>-</td><td>-</td><td>Emmenagogue</td></tr><tr><td>182.</td><td>Ricinus communis Linn.</td><td>Euphorbiaceae</td><td>Seed</td><td>Guinea Pigs</td><td>Anti-implantation and Abortifacient</td></tr><tr><td>183.</td><td>Rubia cordifolia Linn.</td><td>Rubiaceae</td><td>Root</td><td>-</td><td>Antifertility activity</td></tr><tr><td>184.</td><td>Rubus ellipticus Sm.</td><td>Rosaceae</td><td>Leaves</td><td>Rats</td><td>Anti-implantation Effect</td></tr><tr><td>185.</td><td>Ruta angustifolia Linn.</td><td>Rutaceae</td><td>Leaf</td><td>-</td><td>Antifertility activity</td></tr><tr><td>186.</td><td>Ruta graveolens Linn.</td><td>Rutaceae</td><td>Aerial parts and Roots</td><td>Rats and hamsters</td><td>Anticonceptive activity</td></tr><tr><td>187.</td><td>Salvia fruticosa Mill.</td><td>Labiatae</td><td>Leaves</td><td>Rats</td><td>Anti-implantation Effect</td></tr><tr><td>188.</td><td>Samida rosea Sims.</td><td>Flacourtiaceae</td><td>Leaf</td><td>Rats</td><td>Abortifacient and Emmenagogue</td></tr><tr><td>189.</td><td>Santalum album Linn.</td><td>Santalaceae</td><td>Whole Plant</td><td>-</td><td>Abortifacient</td></tr><tr><td>190.</td><td>Sapindus mukorossi Gactn</td><td>Sapindaceae</td><td>Fruit Pericarp</td><td>Rats</td><td>Alteration in Sperm membrane physiology</td></tr><tr><td>191.</td><td>Sarcostemma acidum (Roxb) Voigt</td><td>Apocynaceae</td><td>Stem</td><td>Rats</td><td>Arrests Spermatogenesis</td></tr><tr><td>192.</td><td>Scilla indica (Baker)</td><td>Liliaceae</td><td>Bulb</td><td>-</td><td>Emmenagogue</td></tr><tr><td>193.</td><td>Semecarpus anacardium Linn.</td><td>Anacardiaceae</td><td>Fruits</td><td>Rats</td><td>Spermatogenic arrest</td></tr><tr><td>194.</td><td>Solanum surattense Burm.f.</td><td>Solanaceae</td><td>Seed</td><td>Rats</td><td>Deplete the oxidative stress of cauda epididymal spermatozoa</td></tr><tr><td>195.</td><td>Stephania hemandifolia Willd.</td><td>Menispermaceae</td><td>Leaf</td><td>Rats</td><td>Inhibition of spermatogenesis</td></tr><tr><td>196.</td><td>Stevia rebaudiana Bertoni</td><td>Asteraceae</td><td>Whole plant</td><td>Rats</td><td>Decrease in Testosterone Level</td></tr><tr><td>197.</td><td>Striga orobanchoides Benth</td><td>Scrophulariaceae</td><td>Whole Plant</td><td>Rats</td><td>Antispermatogenic effect</td></tr><tr><td>198.</td><td>Syzygium cuminii Linn. Syn. Eugenia jambolana Lam.</td><td>Myrtaceae</td><td>Oleanolic acid isolated from the flowers of Eugenia jambolana</td><td>Rats</td><td>Arrest of spermatogenesis</td></tr><tr><td>199.</td><td>Tagetes erecta L.</td><td>Asteraceae</td><td>leaves</td><td>-</td><td>Emmenagogue</td></tr><tr><td>200.</td><td>Tanacetum parthenium L.Sch.</td><td>Asteraceae</td><td>Plant without Root</td><td>-</td><td>Abortifacient</td></tr><tr><td>201.</td><td>Taxus baccata Linn.</td><td>Taxaceae</td><td>Leaves</td><td>Rats</td><td>Antifertility</td></tr><tr><td>202.</td><td>Terminalia arjuna Wight & Am.</td><td>Combretaceae</td><td>Bark</td><td>-</td><td>Antispermatogenic effect</td></tr><tr><td>203.</td><td>Tinospora cordifolia (Willd.) Miers ex Hook.f. Thoms</td><td>Menispermaceae</td><td>Stem</td><td>Rats</td><td>Reduction in testosterone levels</td></tr><tr><td>204.</td><td>Trichosanthes cucumerina Linn.</td><td>Curcubitaceae</td><td>Whole plant</td><td>Rats</td><td>Antiovulatory activity</td></tr><tr><td>205.</td><td>Trigonella foenumgraecum Linn.</td><td>Fabaceae</td><td>Seeds</td><td>Rabbits</td><td>Antifertility activity</td></tr><tr><td>206.</td><td>Tripterygium hypoglaucum (Level) Hutch</td><td>Celastraceae</td><td>Root Xylem</td><td>Humans</td><td>Reduced Sperm concentration and motility</td></tr><tr><td>207.</td><td>Tripterygium wilfordii Hook f.</td><td>Celastraceae</td><td>Root and Isolated plant fractions</td><td>Rats and Humans</td><td>Reversible infertility</td></tr><tr><td>208.</td><td>Tylophora asthmatica Wight & Am</td><td>Apocynaceae</td><td>Leaf and Stem</td><td>Rat</td><td>Antispermatogenic effect</td></tr><tr><td>209.</td><td>Uraria lagopodioides Desv.</td><td>Fabaceae</td><td>Whole plant</td><td>-</td><td>Abortifacient effect</td></tr><tr><td>210.</td><td>Urena lobata Linn.</td><td>Malvaceae</td><td>Root</td><td>Rat</td><td>Inhibition of Spermatogenesis and</td></tr><tr><td>211.</td><td>Urginea indica Kunth.</td><td>Liliaceae</td><td>Bulb</td><td>-</td><td>Abortifacient effect</td></tr><tr><td>212.</td><td>Uritica dioica Linn.</td><td>Urticaceae</td><td>-</td><td>-</td><td>Abortifacient effect</td></tr><tr><td>213.</td><td>Uropatha antisylleptica R.E. Schult.</td><td>Araceae</td><td>-</td><td>-</td><td>Contraceptive</td></tr><tr><td>214.</td><td>Valeriana Montana Linn.</td><td>Valerianaceae</td><td>Root</td><td>-</td><td>Sterilizer</td></tr><tr><td>215.</td><td>Ventilago neo-caledonica Schlecht.</td><td>Rhamnaceae</td><td>Leaf</td><td>-</td><td>Contraceptive</td></tr><tr><td>216.</td><td>Vernonia amygdalina Delile</td><td>Asteraceae</td><td>Root</td><td>-</td><td>Antifertility effect</td></tr><tr><td>217.</td><td>Viburnum foetidum wall</td><td>Caprifoliaceae</td><td>Leaf</td><td>-</td><td>Emmenagogue</td></tr><tr><td>218.</td><td>Vigna unguiculata (Linn.) Walp (Cowpeas)</td><td>Fabaceae</td><td>-</td><td>Rat</td><td>Antifertility effect</td></tr><tr><td>219.</td><td>Vitex negundo L.</td><td>Lamiaceae</td><td>Seeds</td><td>Dog</td><td>Anti-Androgenic Effect</td></tr><tr><td>220.</td><td>Waltheria Americana Linn</td><td>Sterculiaceae</td><td>-</td><td>-</td><td>Abortifacient Effect</td></tr><tr><td>221.</td><td>Wedelia gracilis Rich</td><td>Asteraceae</td><td>Whole plant</td><td>-</td><td>Abortifacient Effect</td></tr><tr><td>222.</td><td>Wedelia trilobata (L.) Hitch.</td><td>Asteraceae</td><td>-</td><td>-</td><td>Antifertility effect</td></tr><tr><td>223.</td><td>Withania coagulans (Stocks.) Dunal</td><td>Solanaceae</td><td>Fruit</td><td>-</td><td>Emmenagogue</td></tr><tr><td>224.</td><td>Withania somnifera Dunal</td><td>Solanaceae</td><td>Fruit</td><td>Rats</td><td>Decreased Sperm motility</td></tr><tr><td>225.</td><td>Xanthium spinosum Linn.</td><td>Asteraceae</td><td>Leaf</td><td>-</td><td>Contraceptive</td></tr><tr><td>226.</td><td>Xylopia aethiopica (Dunal) A.Rich</td><td>Annonaceae</td><td>Fruit</td><td>Rats</td><td>Antifertility effect</td></tr><tr><td>227.</td><td>Zaluzania triloba (Ort.) Pers.</td><td>Asteraceae</td><td>Plant without root</td><td>-</td><td>Abortifacient</td></tr><tr><td>228.</td><td>Zingiber roseum (Roxb.) Roscoe</td><td>Zinziberaceae</td><td>Stem</td><td>-</td><td>Antifertility</td></tr><tr><td>229.</td><td>Zinziber officinale Rosc.</td><td>Zinziberaceae</td><td>Rhizome</td><td>Rats</td><td>Abortifacient</td></tr><tr><td>230.</td><td>Ziziphora tenuior Linn</td><td>Labiatae</td><td>Seed</td><td>-</td><td>Emmenagogue</td></tr><tr><td>231.</td><td>Ziziphus nummularia (Burm.f.)</td><td>Rhamnaceae</td><td>Root bark</td><td>-</td><td>Abortifacient</td></tr><tr><td>232.</td><td>Zizyphus jujuba Mill.</td><td>Rhamnaceae</td><td>Bark</td><td>-</td><td>Antifertility</td></tr><tr><td>233.</td><td>Zizyphus xylopyrus (Retz.) Willd.</td><td>Rhamnaceae</td><td>Fruit</td><td>-</td><td>Induces Sterility</td></tr></table>
 Panel label: Lupeol.
Alpha - spinasterol
 Panel label: Hexacosanal.
 Panel label: Vasicine.
 Panel label: Isoadiantone.
 Panel label: Lebbekanin -E.
 Panel label: Oleanolic acid.
 Panel label: Leupelol acetate.
 Panel label: Stigmastane.
Beta - Sitosterol
 Panel label: Ergosterol peroxide.
 Panel label: Andrographolide.
 Panel label: Aristolic acid.
 Panel label: p-coumaric acid.
 Panel label: Methyl aristolate.
 Panel label: Luteolin.
 Panel label: Apigenin.
 Panel label: Butin.
 Panel label: Calotropin.
 Panel label: Pectin.
 Panel label: Leucoanthocyanidin.
 Panel label: Sennoside A.
 Panel label: Sennoside B.
 Panel label: Fraxinellone.
 Panel label: Ecdysterone.
 Panel label: Rohitukene.
 Panel label: Embelin.
 Panel label: Anethole.
 Panel label: Gardenic acid.
Gardenolic acid B
 Panel label: Colchicine.
Gossypol acetic acid
 Panel label: Leonitin.
7 -Oxo -beta -sitosterol
 Panel label: Tinctoramine.
 Panel label: Tinctoralactone.
 Panel label: kaurenoic acid.
 Panel label: Yuechukene H.
 Panel label: Nicotine.
 Panel label: Piperine.
 Panel label: Plumbagin.
 Panel label: Lupeol.
 Panel label: Chalepensin.
2-(4-acetoxyphenyl)-2-chloro-N-methyl-ethyl ammonium chloride
 Panel label: Senecionine.
Senecionine N -oxide
 Panel label: Acacetin.
 Panel label: Luteolin.
 Panel label: Apigenin.
 Panel label: Purpurin.
 Panel label: Rutin.
 Panel label: Berberine.
 Panel label: Toddaline.
 Panel label: Triptolide.
 Panel label: Tripdiolide.
 Panel label: Triphchlorolide.
 Panel label: Vincristine.
5,7,3'-trihydroxy -6,8,4' -trimethoxy (Acerosin)
 Panel label: Diosgenin.
 Panel label: Yamogenin.
 Panel label: Tigogenin.
 Panel label: Gossypol.
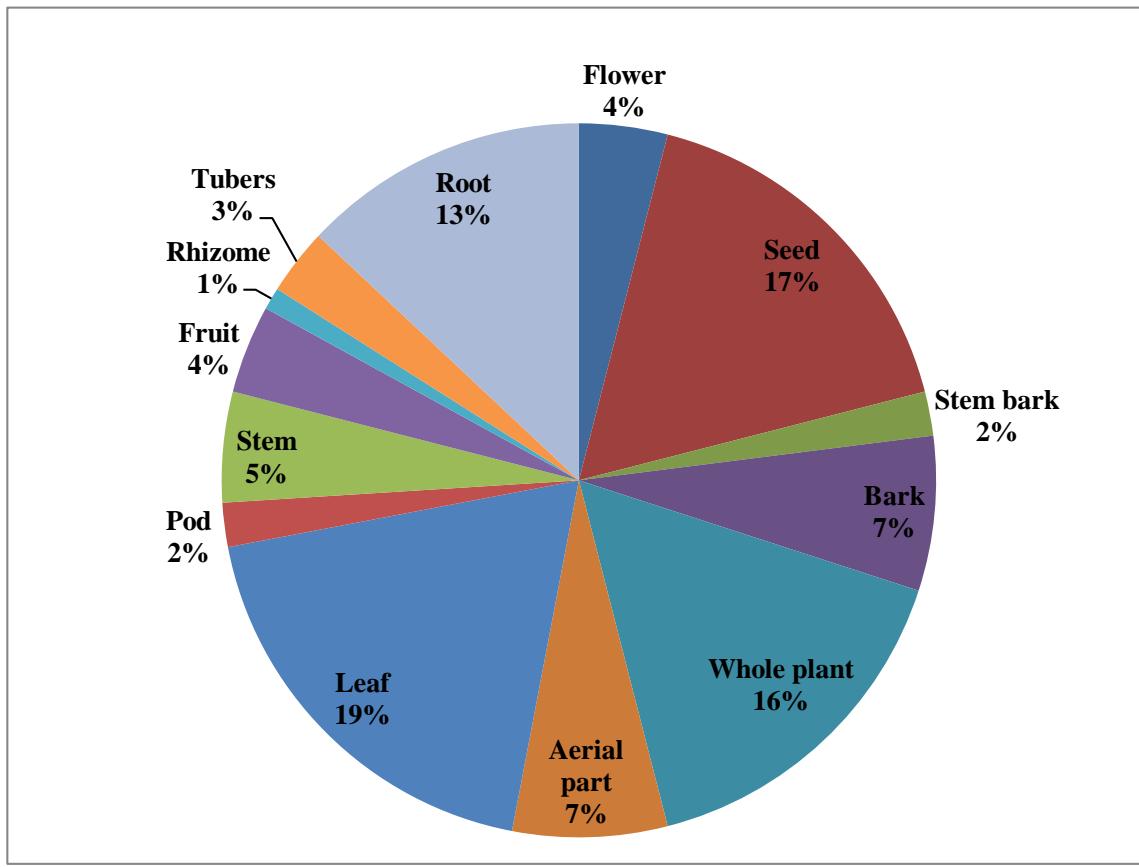
Figure 1: Structure of a Few Chemical Components that Were Isolated from the Plants on the List Figure 2: Percentage of Different Plant Parts Responsible for Antifertility Activity
Generating HTML Viewer...
References
59 Cites in Article
D Dabhadkar,V Thakare,V Zade,A Charjan,M Dhore,S Deosthale (2015). Review on some ethnomedicinal plants having antifertility activity in female albino rats.
D Kumar,A Kumar,O Prakash (2012). Potential antifertility agents from plants: a comprehensive review.
X Pei,W Nai (1991). Can ethnopharmacology contribute to the development of antifertility drugs?.
W Shibeshi,E Makonne,L Zerihum,A Debella (2006). Effect of Achyranthes aspera L. on fetal abortion, uterine and pituitary weights, serum lipids and hormones.
H Khushalani,P Tatke,K Singh (2006). Antifertility activity of dried flowers of Woodfordia fruticosa kurz.
Stephen Bent,Richard Ko (2004). Commonly used herbal medicines in the United States: a review.
Yong Zha,Ying-Li Cun,Yun-Chao Huang (2008). Matrine promotes cell apoptosis in human hepatocellular carcinoma cell line SMMC-7721.
J John (1981). The continuing need for contraceptive research.
C Lin (1992). Crude drugs used for the treatment of diabetes mellitus in Taiwan.
Leon Muntean (2019). HOP AND MEDICINAL PLANTS CROP RESEARCH CENTER, YEAR XXV.
R Marles,N Farnsworth (1994). Antidiabetic plants and their active constituents.
C Stalin,K Vivekanandan,E Bhavya (2013). Medicinal Plants with Antifertility Activity-An Overview.
V Murugan,G Shareef,R Sarma,M Ramanathan,B Suresh (2000). Antifertility activity of Oroxylum indicum Vent. stem bark on female Wistar rats.
I Allag,K Rangari (2002). Extragenomic action of steroids on spermatozoa: Prospects for regulation of fertility.
R Gupta,R Sharma (2006). A review on medicinal plants exhibiting antifertility activity in males.
D Bhowmik,M Umadevi,Pks Kumar,S Duraivel (2013). Medicinal Plants with Potential Antifertility Activity.
Shyam Agrawal,Ashok Kumar,Sumeet Gullaiya,Vishal Dubey,Ashish Nagar,Poonam Tiwari,Parul Dhar,Varun Singh (2012). Antifertility activity of methanolic bark extract of Aegle marmelos (l.) in male wistar rats.
K Saravanan,G Priya,C Renuka (2012). Medicinal plants with potential antifertility activity-A review of sixteen years of herbal medicine research (1994-2010).
G Priya,K Saravanan,C Renuka (2012). Medicinal plants with potential antifertility activity-A review of sixteen years of herbal medicine research.
Priyanka Goswami,Moksood Laskar,Mrinmoy Basak (2020). A Review on Medicinal Plants of North Eastern Region with Potential Antifertility Activity.
K Bala,M Arya,D Katare (2014). Herbal contraceptive: an overview.
X Peri,W Nai (1991). Can ethnopharmacology contribute to the development of antifertility drugs?.
Radha Yegnanarayan,G Joglekar (1978). Anti-Fertility Effect of Non-Steroidal Anti-Inflammatory Drugs.
(2019). World population prospects 2019.
M Umadevi,Sampath Kumar,P,Debjit Bhowmik,S Duraivel (2013). Medicinal Plants with Antifertility Activity.
A Choudhury,B Marbaniang,I Sutnga,G Hazarika,P Goswami,B Dey (2020). Pharmacognostic and preliminary phytochemical screening of Trachyspermum khasianum H. Wolff.
Harold Mahadevan,Ellis,Vishy (2013). Clinical anatomy applied anatomy for students and junior doctors.
Afsar Shaik,Rupesh Kanhere,Rajaram Cuddapah,Kumar Nelson,Prasanth Vara,Saisaran Sibyala (2014). Antifertility activity of Artemisia vulgaris leaves on female Wistar rats.
T Lemke,D Williams,V Roche,S Zito (2008). Foye's principles of medicinal chemistry.
Sandeep Dewal,Rachna Sharma,R Gupta (2018). A REVIEW ON ANTIFERTILITY ACTIVITY OF PLANTS OF CHAMBAL RIVERINE.
M Kapoor,S Garg,V Mathur (1974). Anthiovulatory activity of five indigenous plants in rabbits.
S Tejashri,Dattaprasad Ravindra,S (2021). Androgenic and Antifertility Properties of Medicinal Plants: A Review.
Said Ataa,Hawas Usama,W,El-Shenawy Siham,Nofal Salwa,M,Rashed Khaled (2010). Flavonoids and some biological activities of Ailanthus excelsa leaves.
Olagbende-Dada Stella,O,Ukpo Grace,E Herbert,A Coker,Samuel (2009). Adesina Oxytocic and anti-implantation activities of the leaf extracts of Graptophyllum pictum (Linn.) Griff. (Acanthaceae).
A Parohit,K Surendra,B Keshav (2008). Contraceptive efficacy of Plumbago zeylanica root extract (50% ETOH) in male albino rats with special emphasis on testicular cell population dynamics.
Ahmad Koraini,Zainey Konjing,Marahizal Malihan (2002). Tertiary palynomorph assemblage from eastern Chenor, Pahang.
Z Zaheer,A Konale,K Patel,S Khan,R Ahmed (2010). Comparative phytochemical screening of flowers of Plumeria alba and Plumeria rubra.
U Khanna,S Garg,S Vohra,H Walia,R Choudhary (1969). Antifertility screening of plants. II. Effect of six indigenous plants on early pregnancy in Impact of Medicinal Plants on Antifertility Activities: A Review Global Journal of Medical Research ( B ) XXV Issue I Version I Year 2025 albino rats.
M Abdulazeez,D Mansurah,A Ameh,D Ahmadu,S Ibrahim,A Sani,J Ayo,O Joseph,S Ambali,F Suleiman (2009). Effect of Methanol Extract of Carica papaya Seed on Some Hormone Function in Male Albino Rats.
Dusmanta Kumar Pradhan,Manas Ranjan Mishra,Ashutosh Mishra,Ashok Kumar Panda,Rajani Kanta Behera,Shivesh Jha,Sanjaya Choudhury (2012). A comprehensive review of plants used as contraceptives.
E Williamson,D Okpako,F Evans (1996). Pharmacological methods in phytotherapy research: Selection preparation and pharmacological evaluation of plant material.
F Brinker (1997). Inhibition of endocrine function by botanical agents, antigonadotropic activity.
A Prakash,V Saxena,S Shukla,R Mathur (1985). Pueraria tuberosa DC: contraceptive efficacy and toxicological profile.
D Turner (1971). Turner, C. Donnell. General Endocrinology. Philadelphia: W. B. Saunders Company, 1955. 553 p..
S Patil,S Patil (2013). Antiovulatory activity of petroleum ether extract of chromatographic fractions of Citrus medica seeds in albino rats.
T Schug,A Janesick,B Blumber,J Heindela (2011). Endocrine disrupting chemicals and disease susceptibility.
R Kaur,A Sharma,R Kumar,R Kharb (2011). Rising Trends towards Herbal Contraceptives.
Kumud Bala,Mahima Arya,Deepshikha Pandey Katare (2014). Herbal Contraceptive: An Overview.
G Shah,M Khan,M Ahmad,M Zafar,A Khan (2009). Observations on antifertility and abortifacient herbal drugs.
Mohammad Dehghan,T Martin,R Dehghanan (2005). Acute toxicity study on neem (Iranian Azadirachta indica) seed alcoholic extract in mice.
N Mishra,S Joshi,L Tandon,A Munjal (2009). EVALUATION OF ANTI-FERTILITY POTENTIAL OF AQUEOUS EXTRACT OF BOUGAINVILLEA SPECTABILIS LEAVES IN SWISS ALBINO MICE.
Shoaib Ahmad,Yusuf Jamal,Abdul Mannan (2011). Review of Some Medicinal Plants with Anti-fertility Activities.
A Qureshi,D Sanghai,S Padgilwar (2006). Herbal options for contraception: A review.
M Kabra,S Bhandari,R Gupta,A Sharma (2013). A review on herbal contraception.
Mrn Prasad (1973). Toxicological Problems.
Norman Farnsworth,Audrey Bingel,Geoffrey Cordell,Frank Crane,Harry Fong (1975). Potential Value of Plants As Sources of New Antifertility Agents I *.
D Zhu (1982). Plant constituents with biological activity reported in recent years.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Srinivasulu Reddy Motireddy. 2026. \u201cImpact of Medicinal Plants on Antifertility Activities: A Review\u201d. Global Journal of Medical Research - B: Pharma, Drug Discovery, Toxicology & Medicine GJMR-B Volume 25 (GJMR Volume 25 Issue B1): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.