## I. INTRODUCTION
Dry eye is a common condition diagnosed in ophthalmic clinics that can significantly affect a patient's quality of life. The clinical manifestations of patients with dry eye include tear secretion disorder, along with itchiness, photophobia, blurred vision, and foreign body sensation in the eye [1]. The prevalence of dry eye is expected to continue to rise as the elderly population grows [2]. Currently, mainstream treatments for dry eye include tear substitutes, anti-inflammatory drugs, immunosuppressive drugs, and hormones. However, in cases of severe dry eye, tear supplementation may not be enough therapeutically. Additionally, some anti-inflammatory drugs and hormone replacement therapies may cause side effects [3]. Chinese medicine (CM), originating in China, regards the human body as an organic whole, and its therapies involve the interaction of multiple viscera and the adjustment of qi, blood, fluid, and humor [4]. CM is an independent and complete system, which has shown unique therapeutic effects with fewer side effects in the treatment of dry eye [5][6]. The use of CM has broad prospects in the field of dry eye treatment. In CM theory, dry eye is considered a white xerotic syndrome, first proposed in Compendium of Ophthalmology (Shen Shi Yao Han). White xerotic syndrome is believed to be a result of latent heat in the qi aspect, and dampness-heat in the spleen-lung collateral [7]. Chrysanthemum is clinically considered to possess the effects of dispersing wind-heat and pacifying the liver to improve vision. Therefore, chrysanthemum is a common drug for dry eye treatment in CM. The effectiveness of chrysanthemum against dry eye is not only applied in the clinical practice of CM but also in daily life, such as in medicated tea and medicated diets.
As molecular biology, pharmacology, and bioinformatics continue to develop, the modernization of CM is necessary [8][9]. Our group has explored the anti-inflammatory mechanism of total flavonoids of chrysanthemum on dry eye. By inhibiting the expression of IL-1 $\beta$ and TNF- $\alpha$ and promoting the synthesis of TGF- $\beta 1$ mRNA and TGF- $\beta 1$, the flavonoid-class active ingredients can reduce inflammation in a castrated male rabbit dry eye model[10]. However, the potential pharmacological mechanism of chrysanthemum and its interaction with dry eye targets are still unclear and need further study.
Network pharmacology combined with molecular docking is a common method to study the effects of drugs on diseases. Network pharmacology is based on a network composed of chemicals, targets, and pathways, which combines computer science with medical science [11]. Its application in CM provides compelling evidence for the protein targets and potential mechanism of CM in the treatment of diseases. Additionally, molecular docking is a frequently used docking simulation method for predicting the optimal interaction of molecules. Our study aims to clarify the molecular targets and potential mechanism of chrysanthemum against dry eye by utilizing network pharmacology. Furthermore, we use molecular docking to simulate the binding mode of receptor proteins and ligands.
Chrysanthemum belongs to the Asteraceae family. In the Pharmacopeia of the National Health Commission of the People's Republic of China, chrysanthemum is listed as a dietary herbal medicine. Chrysanthemum contains a rich amount of flavonoids, which have superior free radical scavenging and antioxidant functions [12][13]. Additionally, it contains a number of other chemicals, such as phenylpropanoids, triterpenoids, and steroids [14]. What specific components of chrysanthemum act on dry eye and how do they work? We conducted this study to analyze the vital ingredients, important targets, and key pathways of chrysanthemum in dry eye treatment.
## II. MATERIALS AND METHODS
### a) Active components and targets retrieval of Chrysanthemum
The main active components and corresponding targets of chrysanthemum were collected by using the Traditional Chinese Medicine Systems Pharmacology Database and Analysis Platform (TCMSP, http://tcmspw.com/tcmsp.php) with screening conditions "oral bioavailability $(OB)\geq 30\%$ " and "drug-likelihood $(DL)\geq 0.18$ ". TCMSP is a commonly used, comprehensive botanical platform that can retrieve related compounds, related protein targets, and their pharmacokinetic properties [15]. Next, the UniProt database was utilized to obtain the protein sequence of humans. The protein names and annotations were standardized and matched with chrysanthemum component targets using Excel. After that, the targets of unmatched genes were searched again with TCMSP. All matching genes were considered to be the targets of chrysanthemum.
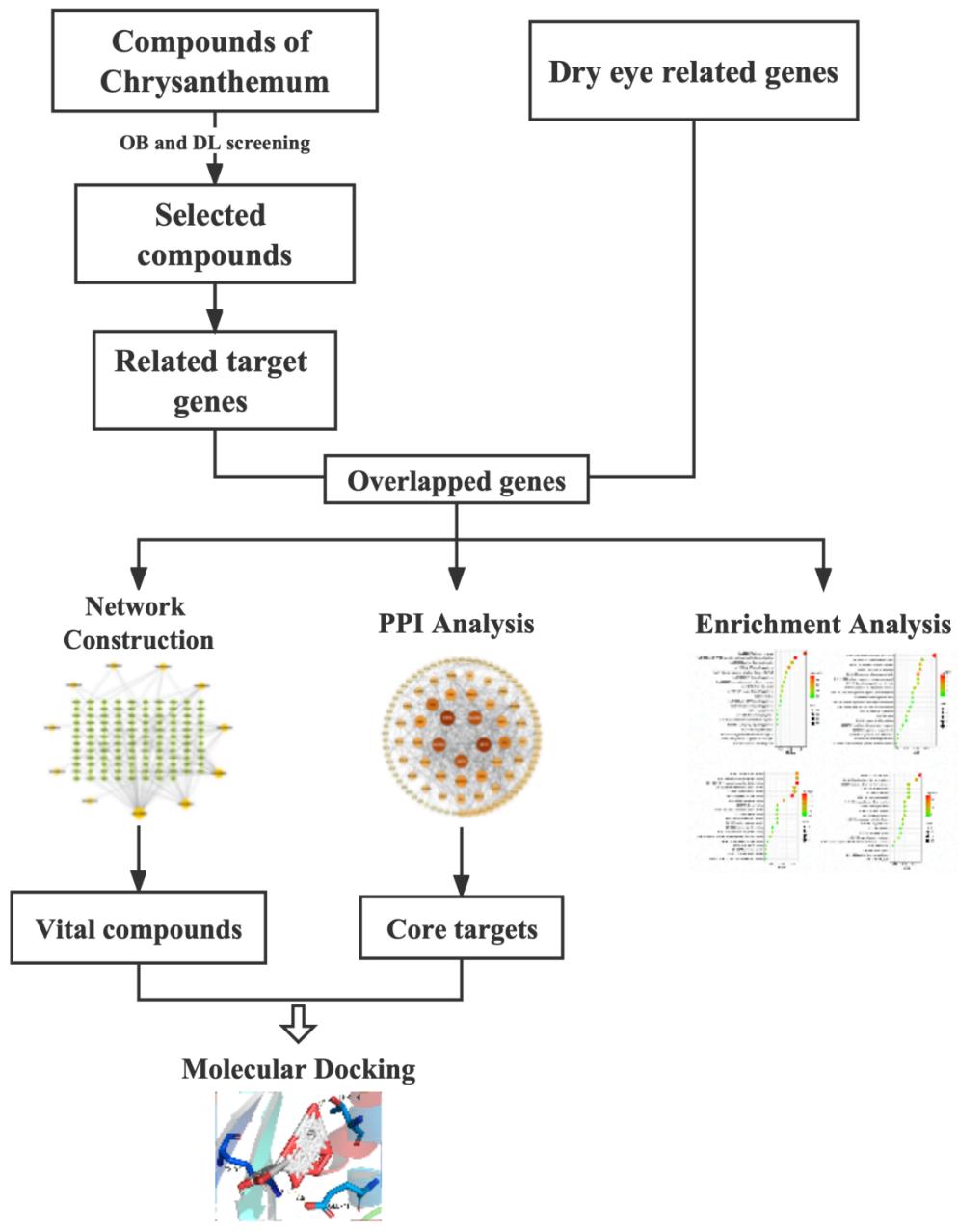 Fig.1: Flow chart of the experimental design
### b) Dry eye related targets retrieval and intersection targets visualization
The related targets of dry eye were searched for in the GeneCards database using the keywords "dry eye" and "xerophthalmia" [16]. The relevance scores of the search result were ranked and the median was calculated to set the screen-out threshold score at 3 $(\geq 3)$. The targets of dry eye identified, along with the chrysanthemum component targets, were inputted into VENNY 2.1.0 (https://bioinfogp.cnb.csic.es/tools/venny/index.html), and the intersection of drug targets and disease targets was collected as the potential targets of chrysanthemum for treating dry eye. The resulting chrysanthemum-dry eye target Venn diagram was drawn. The data for the chrysanthemum vital components and the intersection targets were then imported into Cytoscape 3.8.2 to construct the network of vital compound-disease interaction targets [17].
### c) PPI network constructing of chrysanthemum and dry eye
### d) GO and KEGG pathway enrichment analysis
The 118 intersection targets of chrysanthemum and dry eye were uploaded to Metascape database (https://metascape.org). Custom analysis was performed with Gene Ontology (GO) and KEGG pathway enrichment under the condition of P≤0.01. The enrichment results were imported into an online bioinformatics platform (http://www.bioinformatics.com.cn) to draw the relevant enrichment bubble plots.
### e) Molecular docking
We chose the top 5 proteins ranked by degree as protein receptors. The 3D structure of TP53, JUN, MAPK3, MAPK1 and AKT1 were downloaded from PDB database (https://www.rcsb.org). Then, we use PyMOL 2.5.2. software to remove the organics and solvent. 3D structures of the top 4 vital compounds were downloaded from PubChem database (https:// pubchem.ncbi.nlm.nih.gov). Additionally, molecular docking was performed using Autodock-Vina. TP53, JUN, MAPK3, MAPK1 and AKT1 were docked with quercetin (MOL000098), kaempferol (MOL000422), luteolin (MOL000006) and acacetin (MOL001689) respectively. The affinity score smaller than 0 indicates that the ligand and receptor can bind spontaneously. It is generally believed that the components with lower scores are the active ones interacting with the protein. Matrix heatmap was plotted by http://www.bioinformatics.com.cn, a free online platform for data analysis and visualization. At last, the docking results were analyzed by using PyMOL 2.5.2.
## III. RESULT
### a) Components and the corresponding targets of chrysanthemum
Twenty active components were obtained from chrysanthemum by using screening conditions of $OB \geq 30\%$ and $DL \geq 0.18$, including acacetin, linarin, chryseriol, isorhamnetin, kaempferol, and others. The corresponding targets were identified by searching for the chemical names and matching them with the protein sequence of human. After filtering out the remaining unmatched items, we obtained 220 targets of chrysanthemum components.
Table 1: The active compounds of chrysanthemum
<table><tr><td>MOLID</td><td>Molecule Name</td><td>OB</td><td>DL</td></tr><tr><td>MOL001689</td><td>acacetin</td><td>34.97%</td><td>0.24</td></tr><tr><td>MOL001790</td><td>Linarin</td><td>39.84%</td><td>0.71</td></tr><tr><td>MOL003044</td><td>Chryseriol</td><td>35.85%</td><td>0.27</td></tr><tr><td>MOL000354</td><td>isorhamnetin</td><td>49.60%</td><td>0.31</td></tr><tr><td>MOL000422</td><td>kaempferol</td><td>41.88%</td><td>0.24</td></tr><tr><td>MOL005100</td><td>5,7-dihydroxy-2-(3-hydroxy-4-methoxyphenyl)chroman-4-one</td><td>47.74%</td><td>0.27</td></tr><tr><td>MOL000006</td><td>luteolin</td><td>36.16%</td><td>0.25</td></tr><tr><td>MOL000098</td><td>quercetin</td><td>46.43%</td><td>0.28</td></tr><tr><td>MOL000358</td><td>beta-sitosterol</td><td>36.91%</td><td>0.75</td></tr><tr><td>MOL001506</td><td>Supraene</td><td>33.55%</td><td>0.42</td></tr><tr><td>MOL001733</td><td>EUPATORIN</td><td>30.23%</td><td>0.37</td></tr><tr><td>MOL001755</td><td>24-Ethylcholest-4-en-3-one</td><td>36.08%</td><td>0.76</td></tr><tr><td>MOL001771</td><td>poriferast-5-en-3beta-ol</td><td>36.91%</td><td>0.75</td></tr><tr><td>MOL001771</td><td>poriferast-5-en-3beta-ol</td><td>36.91%</td><td>0.75</td></tr><tr><td>MOL002881</td><td>Diosmetin</td><td>31.14%</td><td>0.27</td></tr><tr><td>MOL004328</td><td>naringenin</td><td>59.29%</td><td>0.21</td></tr><tr><td>MOL005229</td><td>Artemetin</td><td>49.55%</td><td>0.48</td></tr><tr><td>MOL007326</td><td>Cynarin(e)</td><td>31.76%</td><td>0.68</td></tr><tr><td>MOL011319</td><td>TruflexOBP</td><td>43.74%</td><td>0.24</td></tr><tr><td>MOL011802</td><td>(24r)-saringosterol</td><td>39.36%</td><td>0.79</td></tr><tr><td>MOL011816</td><td>[(1S,5S,7S)-7-acetoxy-5-isopropenyl-2,8-dimethylene-cyclodecyl]acetate</td><td>37.02%</td><td>0.19</td></tr></table>
### b) Candidate targets associated with Dry eye and chrysanthemum-dry eye intersection targets
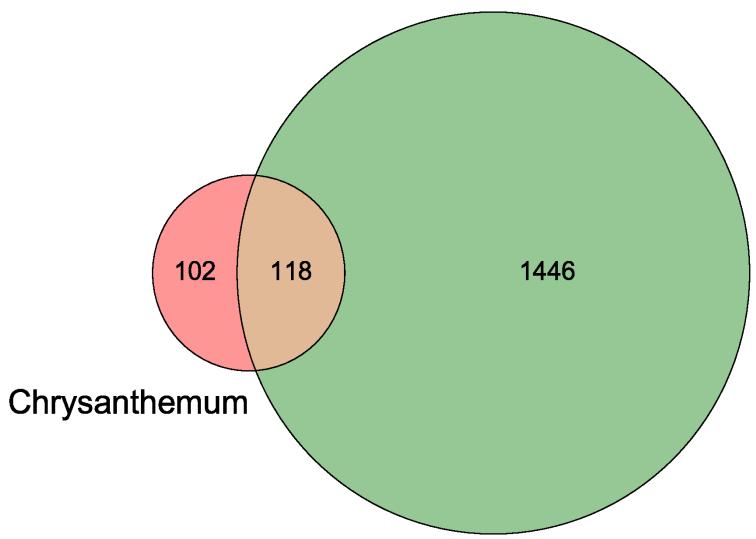
After filtering out the targets with a relevance score smaller than the median $(\geq 3)$, we obtained 1564 dry eye targets. Matching them with the chrysanthemum component targets resulted in a compound-target network of chrysanthemum on dry eye, which revealed
 Panel label: dry eye.
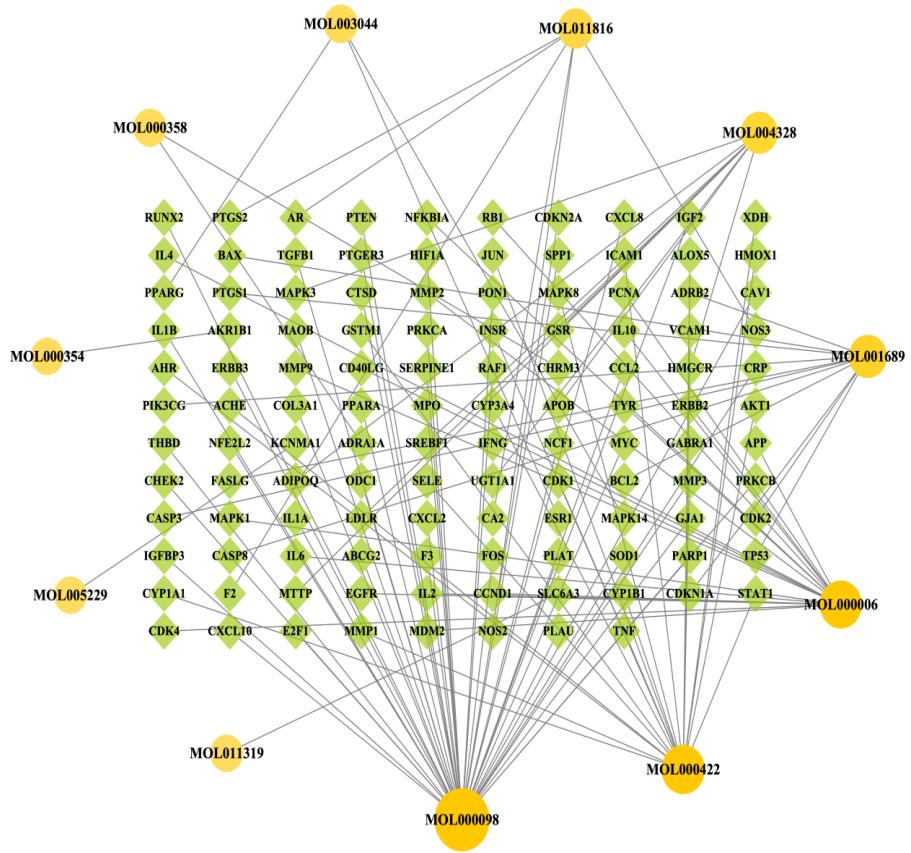 Fig. 2: Venn diagram of active compound targets of Chrysanthemum and related targets of dry eye.
### c) PPI network analysis
The PPI network (Fig. 4) displays 118 nodes and 467 edges, with an average node degree of 7.92. The PPI enrichment p-value is $< 1.0\mathrm{e} - 16$. The node degree indicates the number of edges of the target line. The higher the node degree, the more likely it is to be a key target of functional ingredients and play a significant role in the network. The nodes in the figure represent protein targets, and the edges represent protein-protein interactions. The more lines, the closer the relationship between targets. To illustrate this, the top 25 genes with the highest degree value were plotted in a histogram (Fig. 5), showing that TP53, JUN, MAPK3, MAPK1, and AKT1 are the most prominent proteins.
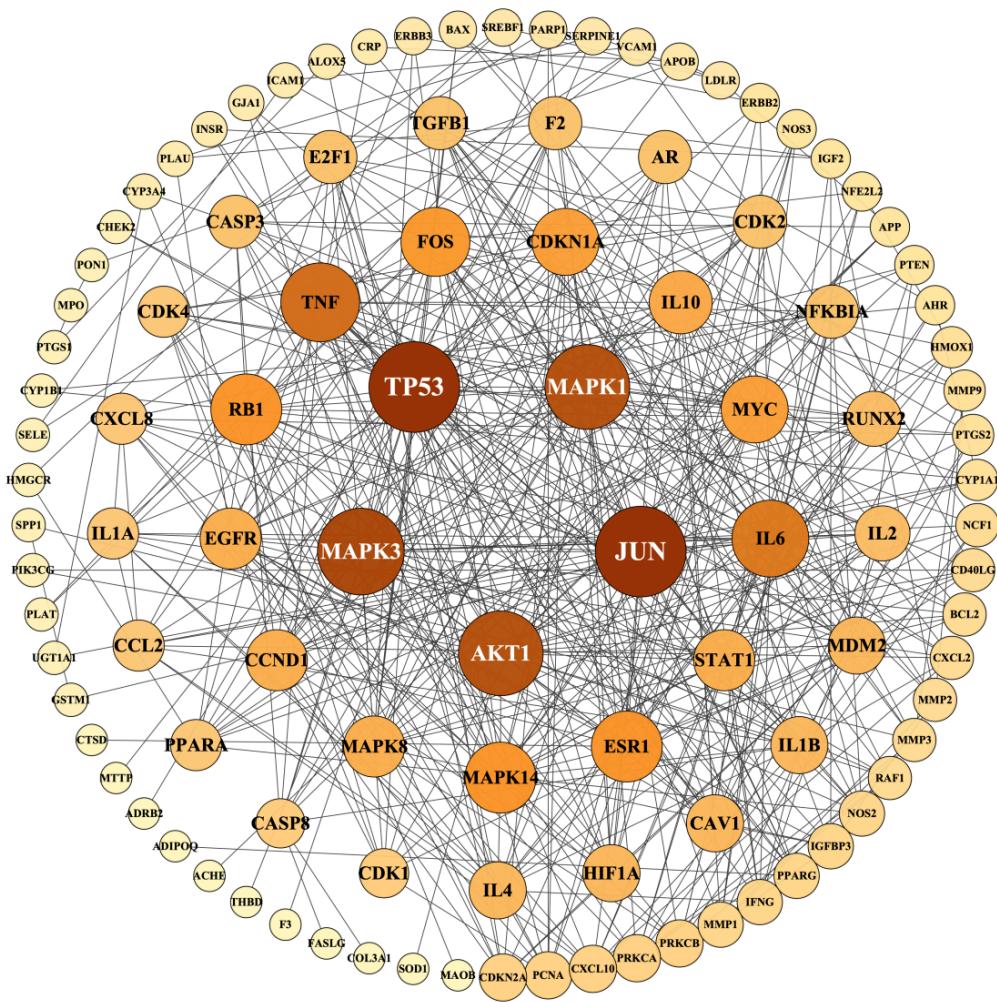 Fig. 3: The compound-target network of Chrysanthemum on dry eye. Yellow solid circles represent compounds and green diamonds represent targets.
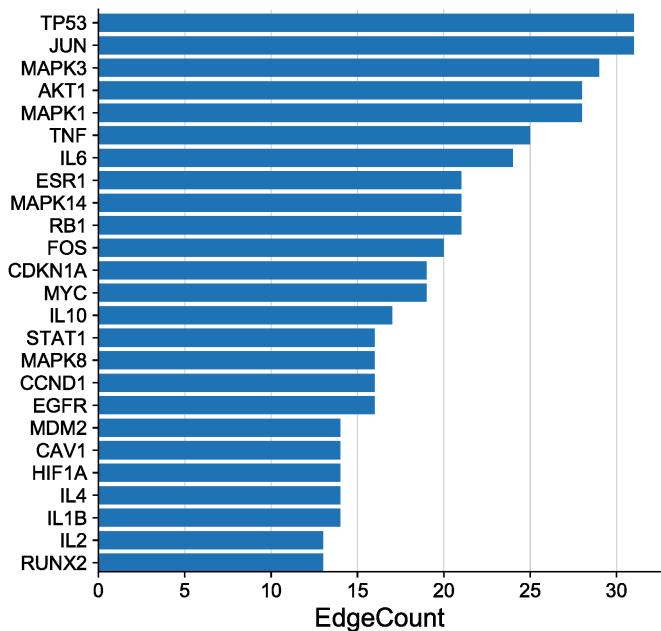 Fig. 4: Network diagram of protein-protein interaction (PPI)
### d) GO&KEGG pathway enrichment analysis
Enrichment result shows 431 cellular components (CC), 719 molecular functions (MF), 5365 biological processes (BP) and 455KEGG pathways. The top 20 significantly enriched entries in the results are arranged in an ascending order of p value (Fig. 6). The results showed important MF such as kinase binding, protease binding, cytokine receptor binding, DNA-binding transcription factor binding, kinase activity and cytokine activity; significant BP such as apoptotic signaling pathway, response to oxygen levels, regulation of inflammatory response, regulation of cellular response to stress and aging; important CC contains membrane, cytoplasm, endoplasmic reticulum, etc. The enrichment analysis results of KEGG pathways mainly manifested IL-17 signaling pathway (hsa04657), PI3K-Akt signaling pathway (hsa04151), HIF-1 signaling pathway (hsa04066), NF-kappa B signaling pathway (hsa04064), Jak-STAT signaling pathway (hsa04630), Calcium signaling pathway (hsa04020), Leukocyte transendothelial migration pathway (hsa04670), Regulation of lipolysis in adipocytes pathway (hsa04923) and Dopaminergic synapse pathway (hsa04728).
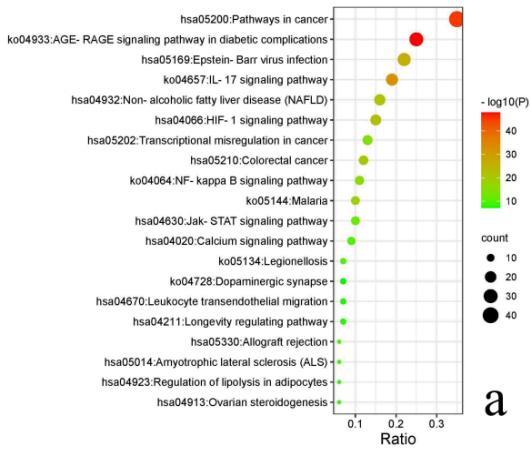 Fig. 5: Top25 targets by degree value
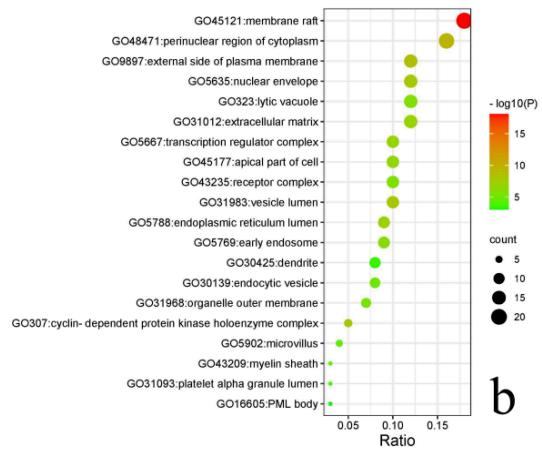
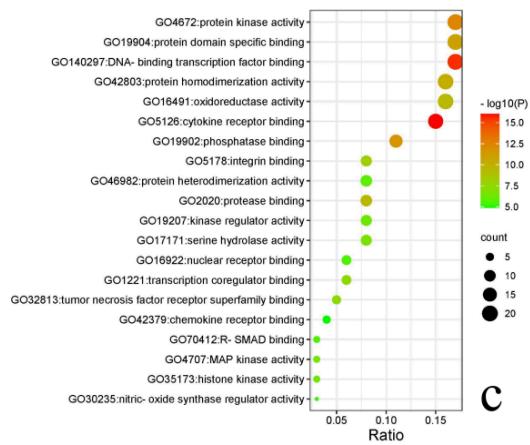
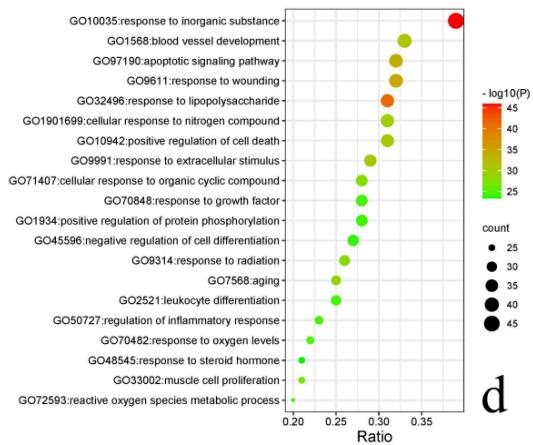 Fig. 6: Enrichment bubble plots of GO&KEGG pathway enrichment analysis
a Enrichment analysis results of KEGG pathways; b Enrichment analysis results of CC; c Enrichment analysis results of MF; d Enrichment analysis results of BP. Size and color of the dots represent the degree of GO enrichment analysis.
### e) Molecular docking results and analysis
The top 5 core targets were respectively docked onto the top 4 vital chemicals. All docking simulations show good combination states. The affinity scores of TP53, JUN, MAPK3 and MAPK1 are slightly more ideal than AKT1. The representative ones in the docking results are TP53 with luteolin (-9kcal/mol), JUN with quercetin (-9.4kcal/mol), MAPK3 with quercetin (-10kcal/mol) and MAPK1 with quercetin (-9.5kcal/mol). These four receptors and their best docked ligands were picked out to draw the molecular docking diagram subsequently. Notably, MAPK3 also shows remarkable docking state with other 3 ligands. Its docking score with kaempferol, luteolin and acacetin are -9kcal/mol, -9.4kcal/mol, -9.2kcal/mol, respectively. The affinity scores heatmap and docking simulation images are shown in figure 7 and figure 8.
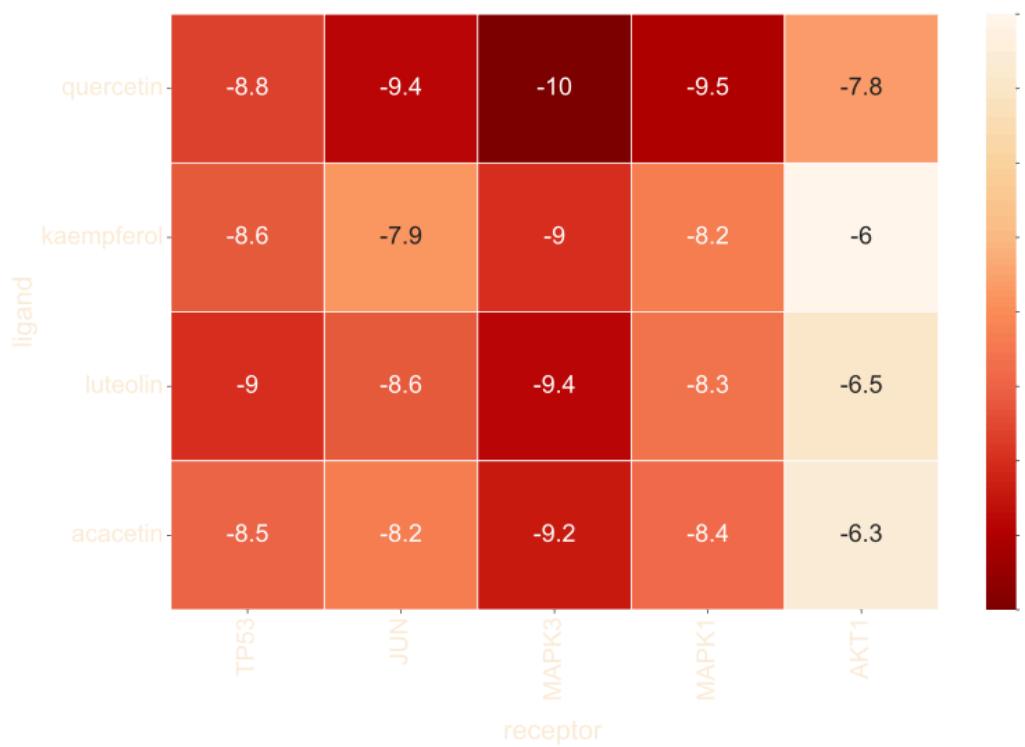 Fig. 7: The heat map of the affinity score
 Panel label: a.
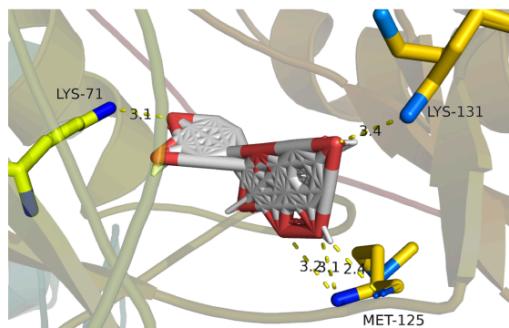 Panel label: b.
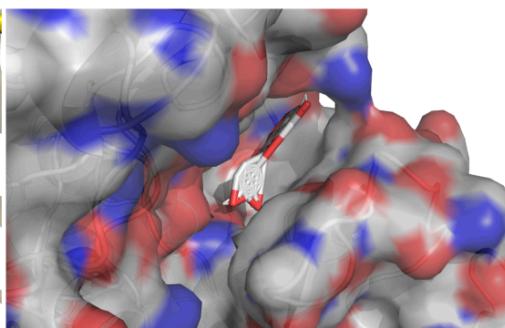
 Panel label: c.
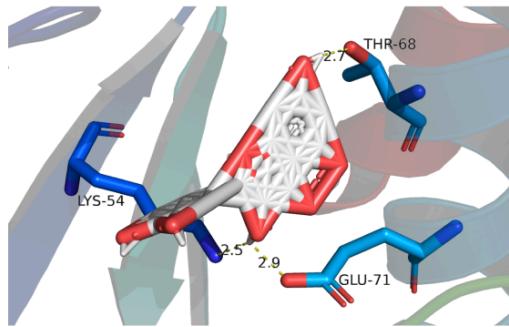
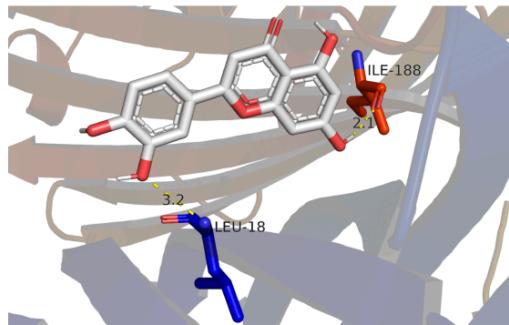 Panel label: d.
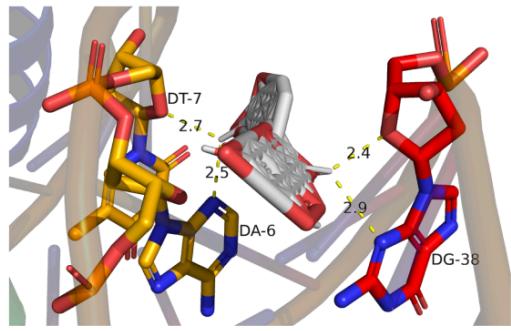
 Fig. 8: The notable results of molecular docking analysis. aAction mode of quercetin with target JUN(PDB ID:5T01); bAction mode of quercetin with target MAPK3(PDB ID:4QTB); cAction mode of quercetin and MAPK1(PDB ID:7NR8); dAction mode of luteolin and TP53(PDB ID:7BWN)
## IV. DISCUSSION
In CM theory, chrysanthemum is believed to be effective in treating dry eye by dispersing wind-heat and pacifying the liver, which in turn improves vision. However, due to the complex composition of herbal medicine, it is often difficult to understand its underlying mechanisms. To address this issue, we used network pharmacology to establish a drug-target-disease network, which helped us to explore the active ingredients and therapeutic targets of chrysanthemum. Our results suggest that chrysanthemum can treat dry eye through the action of multiple targets and components, which is consistent with the manifold therapeutic effects of CM. Although chrysanthemum and chrysanthemum-containing formulas have been the subject of some clinical experiments[10][19], there is still much to learn about the combination of its components and the enrichment of its targets. Therefore, we believe that exploring the vital components and targets before experimental validation may be more conducive to research in this field.
It is widely recognized that inflammation is a central mechanism of dry eye, as supported by numerous in vitro, in vivo, and human studies [20]. Inflammatory cytokines are the main culprits of dry eye-associated inflammation [21]. Moreover, research suggests that an abnormal immune response in the ocular system can also contribute to ocular surface injury, further exacerbating dry eye symptoms [22]. Additionally, oxidative stress is also a critical factor in the pathogenesis of dry eye. Oxidative stress mouse models have shown decreased tear secretion, leukocyte infiltration, and fibrosis [23]. Chrysanthemum's bioactive components have the effect of anti-inflammation, antibacterial, antifungal, anti-spirochete, anti-human immunodeficiency virus, and antioxidant [13]. This provides theoretical support for the anti-inflammatory effect of chrysanthemum treating dry eye.
According to the degree ranking of targets interaction, the corresponding most important components are quercetin, kaempferol, luteolin and acetin, which all belong to the ranks of natural flavonoids. Previous studies have already shown that flavonoid compound has strong anti-inflammatory and antioxidant effects [19][24], which is consistent with our earlier research[10]. Quercetin's topical application can not only reduce the irregularity of the ocular surface, but also increases the amount of tears and goblet cell density [25]. However, quercetin's oral bioavailability is still controversial, so we need more information about its pharmacokinetics in experiments [26]. Intragastric kaempferol feeding can inhibit nlrp1/NLRP3 inflammasomes and caspase8 through NF-κB and JNK pathway, so as to reduce the inflammatory damage of retinal ganglion cells in acute glaucoma mice model [27]. Kaempferol-contianing eye drops in rabbit dry eye model have also been proved to reduce corneal epithelial injury and increase tear secretion [28]. In the rat uveitis model, luteolin inhibits the inflammatory markers and activates NF-κB pathway, thereby reducing iris-ciliary body inflammation. The anti-inflammatory effect of 10 mg/kg luteolin being injected intraperitoneally was proved to be as strong as 1 mg/kg prednisolone [29]. Interestingly, Acacetin has shown therapeutic potential in inflammation, infections and other metabolic disorders [30][31][32]. However, there are few studies on its application in the field of Ophthalmology, which deserves our attention.
According to the our PPI network data as well as molecular docking results, we conclude that the core targets chrysanthemum regulating dry eye are TP53, JUN, MAPK3 and MAPK1, which all showed stable structure and high binding activity with the vital ingredients in molecular docking. TP53 cooperating with mitochondrial PPIF is involved in activating oxidative stress-induced necrosis, which damages the ocular surface and plays an important role in the mechanism of dry eye disease [33]. MAPK family is involved in many processes, such as cell proliferation, stress, inflammation, apoptosis and so on. Studies have proved the efficacy of p38 MAPK inhibitor on lacrimal gland secretion and neurotransmitter secretion in dry eye [34]. JUN protein is also related to cell aging induced by oxidative stress, however, its related studies in dry eye remain limited. Considering its good affinity scores with the ligands, it may serve as another latent therapeutic target of dry eye. Docking is broadly applied in the field of drug discovery and design as well as the identification of ligands binding to a target receptor [35]. We expect to see "pocket-like" docking simulation status. As can be seen in the docking image, the docking model reveals a good morphologic complementary between the receptors and ligands contact areas. Quercetin formed 4 hydrogen bonds with residue DT-6/7 and residue DG-38 of the JUN protein that shows a good binding affinity (-9.4kcal/mol). Whilst it could form 5 hydrogen bonds with residue LYS-71/131 and residue MET-125 of MPK3 which both show good binding affinity(-10kcal/mol). It also could form 3 hydrogen bonds with residue LYS-54, residue THR-68 and residue GLU-71 of MPK1 showing a good binding affinity (-9.5kcal/mol). Meanwhile, Luteolin could form 2 hydrogen bonds with residue ILE-188 and residue LEU-18 of TP53, displaying a good binding affinity (-9kcal/mol). These ligand-receptor combinations need to be further studied theoretically and experimentally.
The enrichment results have revealed several important KEGG pathways. The interleukin 17 (IL-17) family has been found to play a crucial role in both acute and chronic inflammatory responses. Topical application of IL-17 therapy has been shown to reduce ocular surface symptoms of tear evaporation and meibomian gland dysfunction [36]. The activation of the PI3K-AKT signaling pathway has also been found to have protective effects on injured RGCs [37]. Additionally, the NF- $\kappa$ B signaling pathway and JAK-STAT signaling pathway have been found to be critically involved in cellular stress and inflammation, and have been the focus of numerous studies in ophthalmology and Sjögren's syndrome [38][39][40]. It is worth noting that inflammation is also the primary mechanism of Sjögren's syndrome, which can trigger dry eye. Studies have shown that Dll4/Notch Signaling and HIF-1 $\alpha$ Stimulating lymphangiogenesis may protect lacrimal glands from dry eye-induced inflammation by helping to clear immune cells in the lacrimal glands[41]. The deficiency of HIF-1 $\alpha$ enhances the recruitment of inflammatory cells to lacrimal glands[42]. Our enrichment analysis has also identified some core functions and processes, including oxidoreductase activity and response to oxygen levels, which have drawn our attention. These results provide guidance for the direction of our subsequent experimental research and may provide new insights for the treatment of dry eye.
## V. CONCLUSION
The results of our enrichment analysis and molecular docking revealed that quercetin and luteolin are the important components of chrysanthemum in the treatment of dry eye, followed by kaempferol and acacetin. These components have shown promising anti-inflammatory and antioxidant properties. Interestingly, the anti-inflammatory mechanism of acacetin in ophthalmology has not been studied extensively, and further research is needed to explore its potential therapeutic effects. Our molecular docking studies showed that TP53, JUN, and MAPK3/1 are the core targets of chrysanthemum in the treatment of dry eye. Quercetin with JUN, quercetin with MAPK3/1, and luteolin with TP53 were identified as the most stable docking combinations, indicating their potential as effective therapeutic combinations for the treatment of dry eye. These findings provide valuable insights for further experimental studies on the potential therapeutic effects of chrysanthemum in the treatment of dry eye.
Declarations
Ethics approval and consent to participate
Not applicable
Availability of data and materials
Competing interests
The authors declare no conflict of interests.
Funding
This work is financially supported by National Natural Science Foundation of China (82174443); Natural Science Foundation of Hunan Province of China (2021JJ30527); Key Project of Educational Commission of Hunan Province of China (20A370); Key Program of Administration of Chinese Medicine of Hunan Province of China (2021023)
Authors' contributions
Yanxue Zhang wrote and revised the manuscript. Xiaolei Yao designed and adjusted structure of the manuscript. Yu Tang, Kai Wu collected and analyzed the data. All authors discussed the results and revised the manuscript.
Acknowledgements
Not applicable.
Generating HTML Viewer...
References
41 Cites in Article
P Rouen,M White (2018). Dry Eye Disease: Prevalence, Assessment, and Management.
Kimberly Farrand,Moshe Fridman,Ipek Stillman,Debra Schaumberg (2017). Prevalence of Diagnosed Dry Eye Disease in the United States Among Adults Aged 18 Years and Older.
Leisa Marshall,J Roach (2016). Treatment of Dry Eye Disease.
F Lozano (2014). Basic Theories of Traditional Chinese Medicine.
San-Hua Su,Tsung-Jung Ho,Cheng-Chan Yang (2021). Retrospective evaluation of the curative effect of traditional Chinese medicine on dry eye disease.
Huiyuan Tan,Guangyao Li,Qiumei Ruan,Chuanghui Yang,Xuelian Ma,Min Li (2021). Acupuncture combined with traditional Chinese medicine for dry eye: A systematic review and meta-analysis.
C Ou,Q Peng,X Chen (2019). A brief analysis of the treatment of dry eyes from the "Shen Shi Yao Han.
Liu Mengling,Zhang Xinyou,Ding Liang,Wu Shumao,Li Diyao,Xiuyun (2021). Data integration, data mining and visualization analysis of traditional Chinese medicine manufacturing process.
Junfei Zhang,Xinjun Wang,Yu Zhong,Shuming Gao,Lichao Zhang,Diwen Fan,Cong Zhou,Senlin Wang (2017). Optimization Design Method of Variable-Period TPMS Structures Based on Period Tensor.
Chen Shi Jian,Liu Lihao,Peng Qianhong,Tang Jun,Yao Yu,Liu Xiaolei,Guoguo (2020). Inflammatory Mechanism of Total Flavonoids of Chrysanthemum and Medicated Serum on Castrated Dry Eye Animal and Cell Models.
Won-Yung Lee,Choong-Yeol Lee,Youn-Sub Kim,Chang-Eop Kim (2019). The Methodological Trends of Traditional Herbal Medicine Employing Network Pharmacology.
Y Li,P Yang,Y Luo,B Gao,J Sun,W Lu,J Liu,P Chen,Y Zhang,L Yu (2019). Chemical compositions of chrysanthemum teas and their anti-inflammatory and antioxidant properties.
S Chen,J Liu,G Dong,X Zhang,Y Liu,W Sun,A Liu (2021). Flavonoids and caffeoylquinic acids in Chrysanthemum morifolium Ramat flowers: A potentially rich source of bioactive compounds.
Hanwen Yuan,Sai Jiang,Yingkai Liu,Muhammad Daniyal,Yuqing Jian,Caiyun Peng,Jianliang Shen,Shifeng Liu,Wei Wang (2020). The flower head of Chrysanthemum morifolium Ramat. (Juhua): A paradigm of flowers serving as Chinese dietary herbal medicine.
J Ru,P Li,J Wang,W Zhou,B Li,C Huang,P Li,Z Guo,W Tao,Y Yang (2014). TCMSP: a database of systems pharmacology for drug discovery from herbal medicines.
G Stelzer,I Dalah,T Stein,Y Satanower,N Rosen,N Nativ,D Oz-Levi,T Olender,F Belinky,I Bahir (2011). In-silico human genomics with GeneCards.
Michael Kohl,Sebastian Wiese,Bettina Warscheid (2011). Cytoscape: Software for Visualization and Analysis of Biological Networks.
Damian Szklarczyk,Annika Gable,David Lyon,Alexander Junge,Stefan Wyder,Jaime Huerta-Cepas,Milan Simonovic,Nadezhda Doncheva,John Morris,Peer Bork,Lars Jensen,Christian Mering (2019). STRING v11: protein–protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets.
Juntao Kan,Min Wang,Ying Liu,Hongyue Liu,Liang Chen,Xue Zhang,Chengrong Huang,Bryan Liu,Zhensheng Gu,Jun Du (2020). A novel botanical formula improves eye fatigue and dry eye: a randomized, double-blind, placebo-controlled study.
N Erdinest,A Solomon (2019). TOPICAL ANTI-INFLAMMATORY AGENTS FOR DRY EYE DISEASE.
Y Wei,P Asbell (2014). The core mechanism of dry eye disease is inflammation.
Laura Periman,Victor Perez,Daniel Saban,Meng Lin,Piergiorgio Neri (2020). The Immunological Basis of Dry Eye Disease and Current Topical Treatment Options.
Gabriele Carullo,Anna Cappello,Luca Frattaruolo,Mariateresa Badolato,Biagio Armentano,Francesca Aiello (2017). Quercetin and Derivatives: Useful Tools in Inflammation and Pain Management.
H Oh,C Kim,J Lee,J Yang (2015). Effects of Quercetin in a Mouse Model of Experimental Dry Eye.
Tina Mckay,Dimitrios Karamichos (2017). Quercetin and the ocular surface: What we know and where we are going.
Chaobin Lin,Fujin Wu,Tongmei Zheng,Xiuchun Wang,Yiwei Chen,Xiaomin Wu (2019). Kaempferol attenuates retinal ganglion cell death by suppressing NLRP1/NLRP3 inflammasomes and caspase-8 via JNK and NF-κB pathways in acute glaucoma.
H Chen,Z Chen,T Wang,V Drew,C Tseng,H Fang,F Lin (2017). Herbal Supplement in a Buffer for Dry Eye Syndrome Treatment.
K Kanai,T Hatta,S Nagata,Y Sugiura,K Sato,Y Yamashita,Y Kimura,N Itoh (2016). Luteolin attenuates endotoxin-induced uveitis in Lewis rats.
S Singh,P Gupta,A Meena,S Luqman (2020). Acacetin, a flavone with diverse therapeutic potential in cancer, inflammation, infections and other metabolic disorders.
Hao Wang,Zengxin Jiang,Zhiying Pang,Tianyao Zhou,Yutong Gu (2020). <p>Acacetin Alleviates Inflammation and Matrix Degradation in Nucleus Pulposus Cells and Ameliorates Intervertebral Disc Degeneration in vivo</p>.
M Dogru,T Kojima,C Simsek,K Tsubota (2018). Potential Role of Oxidative Stress in Ocular Surface Inflammation and Dry Eye Disease.
Xiaoyun Ma,Jun Zou,Linping He,Yun Zhang (2014). Dry eye management in a Sjögren’s syndrome mouse model by inhibition of p38-MAPK pathway.
Luca Pinzi,Giulio Rastelli (2019). Molecular Docking: Shifting Paradigms in Drug Discovery.
K Garbutcheon-Singh,N Carnt,U Pattamatta,C Samarawickrama,A White,V Calder (2019). A Review of the Cytokine IL-17 in Ocular Surface and Corneal Disease.
Xin-Gang Nie,Dong-Sheng Fan,Yan-Xia Huang,Ying-Ying He,Bo-Li Dong,Feng Gao (2018). Downregulation of microRNA-149 in retinal ganglion cells suppresses apoptosis through activation of the PI3K/Akt signaling pathway in mice with glaucoma.
Amandine Charras,Pinelopi Arvaniti,Christelle Le Dantec,Marina Arleevskaya,Kaliopi Zachou,George Dalekos,Anne Bordon,Yves Renaudineau (2020). JAK Inhibitors Suppress Innate Epigenetic Reprogramming: a Promise for Patients with Sjögren’s Syndrome.
Miaomiao Xin,Hongda Liang,Hongyue Wang,Dawei Wen,Liqin Wang,Lei Zhao,Mingshu Sun,Jibo Wang (2019). RETRACTED ARTICLE: Mirt2 functions in synergy with miR-377 to participate in inflammatory pathophysiology of Sjögren's syndrome.
D Zhang,Y Zhao,Y Yang,Y Zhao,D Wu,X Liu,Z Shi,J Hong,J Liu,X Ma (2020). A Mechanism Study of Electroacupuncture for Dry Eye Syndrome by Targeting Conjunctival Cytokine Expressions.
Ji Min,Chul Lee,Yong Ji,Areum Yeo,Hyemi Noh,Insil Song,Eung Kim,Hyung Lee (2016). Activation of Dll4/Notch Signaling and Hypoxia-Inducible Factor-1 Alpha Facilitates Lymphangiogenesis in Lacrimal Glands in Dry Eye.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Zhang Yanxue. 2026. \u201cIntegrating Network Pharmacology and Molecular Docking to Identify the Active Substances and Mechanisms of Chrysanthemum Against Dry Eye\u201d. Global Journal of Medical Research - B: Pharma, Drug Discovery, Toxicology & Medicine GJMR-B Volume 24 (GJMR Volume 24 Issue B1): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.