Multiple Sclerosis (MS) is a progressive neurodegenerative disease affecting the Central Nervous System (CNS), leading to demyelination and neurological impairment. Early diagnosis and continuous monitoring of disease progression are crucial for effective treatment. Magnetic Resonance Imaging (MRI) remains the primary tool for detecting MS lesions; however, traditional segmentation methods rely heavily on visual analysis and struggle to detect earlystage lesions. This study reviews the application of Convolutional Neural Networks (CNNs) for automated lesion segmentation in MS.
## I. INTRODUCTION
Neurodegenerative diseases are progressive disorders that affect the Central Nervous System (CNS), leading to the gradual loss of neurons[1].
Their development involves multiple risk factors, including protein dysfunctions, genetic alterations, oxidative stress, and exposure to toxic substances[2,3]. Landmark studies have demonstrated that oxidative stress exacerbates neuronal death in Alzheimer's and Parkinson's diseases[4], while genetic mutations such as SNCA in Parkinson's and APP in Alzheimer's directly contribute to pathological protein aggregation[5,6]. Additionally, environmental toxins like pesticides have been implicated in increasing the risk of neurodegeneration[7].
Diseases such as Parkinson's, Alzheimer's, Huntington's, amyotrophic lateral sclerosis, and frontotemporal dementias are considered neuropathies and neurodegenerative disorders[3]. Although they have distinct clinical manifestations, they share common pathological mechanisms, particularly the accumulation of misfolded proteins and chronic neuroinflammation. Misfolded proteins, such as amyloid-beta in Alzheimer's and alpha-synuclein in Parkinson's, aggregate into toxic structures that disrupt cellular homeostasis and lead to neuronal loss[8]. These aggregates impair essential cellular functions by sequestering normal proteins and damaging mitochondria. Furthermore, chronic activation of innate immune cells, such as microglia and astrocytes, sustains an inflammatory environment, contributing to progressive neurodegeneration[9].
Multiple Sclerosis (MS) represents a unique intersection between neurodegeneration and autoimmunity. While traditionally categorized separately, MS shares features with other neurodegenerative diseases, including chronic neuroinflammation, oxidative stress, and axonal damage. MS is a chronic, progressive inflammatory disease that affects the CNS[10], Characterized by demyelination, where degeneration of the myelin sheath, the protective covering of nerve fibers, disrupts the conduction of electrical impulses. Although some lesions may initially repair without permanent loss of function, sustained inflammation leads to scarring and irreversible damage[11,12].
MS is an autoimmune disease where activated T cells cross the blood-brain barrier, triggering inflammatory responses that result in demyelination and neuronal injury. The precise mechanisms underlying disease onset remain unclear, but it is widely accepted that MS results from the interplay between genetic susceptibility and environmental triggers[13]. Genetic variants in the HLA-DRB1 locus have been strongly associated with increased MS risk[14], while environmental factors such as Epstein-Barr virus infection and vitamin D deficiency are believed to influence disease activation[15]. Oligodendrocyte death, driven by immune-mediated damage and oxidative stress, impairs myelin repair and exacerbates neurodegeneration[16].
MS typically follows one of several clinical courses: Relapsing-Remitting Multiple Sclerosis (RRMS), Primary Progressive Multiple Sclerosis (PPMS), Secondary Progressive Multiple Sclerosis (SPMS), and Progressive-Relapsing Multiple Sclerosis (PRMS). Early-stage symptoms often overlap with other neurological conditions, complicating diagnosis. Initial manifestations may include transient sensory disturbances, optic neuritis, or mild motor weakness, which can easily be misattributed to less serious illnesses[17]. As the disease progresses, more disabling symptoms such as ataxia, dysarthria, paresis, diplopia, and tremors emerge, leading to cumulative neurological disability[18].
Although there is no cure for MS, advances in immunosuppressive and immunomodulatory therapies have significantly improved disease management. Treatments such as interferon-beta, glatiramer acetate, and monoclonal antibodies aim to modulate the immune response, reduce the frequency of relapses, and slow the progression of disability[19,20]. Corticosteroids are used during acute exacerbations to dampen inflammation[21].
In Brazil, MS has a prevalence of approximately 15 cases per 100,000 inhabitants, with a higher incidence among white women. Symptoms commonly begin around the age of 30, but delays in diagnosis mean many individuals are not formally diagnosed until their mid-40s[22]. As research continues to elucidate the immunopathological mechanisms underlying MS, new therapeutic avenues emerge, offering hope for improved outcomes and quality of life for patients[23].
## II. THEORETICAL FRAMEWORK
Medical image segmentation plays a crucial role in the diagnosis and monitoring of various neurological diseases, including Multiple Sclerosis (MS).
Traditional approaches, relying on visual observation by specialists, although widely used, have significant limitations, such as the time required for identification and the difficulty in detecting small lesions, which are typical in the early stages of the disease. While traditional methods have proven effective in certain contexts, the limitations of manual interpretation, particularly in identifying small or subtle lesions, have necessitated the exploration of AI-based alternatives.
MRI remains the gold standard in diagnosing MS due to its ability to provide high-resolution images of brain structures and lesions. MRI uses different sequences to highlight contrasts between brain tissues, with the most common being FLAIR, T1, T2, and PD. In T1-weighted images, white matter appears lighter than gray matter, while cerebrospinal fluid (CSF) appears dark. In T2-weighted images, white matter is darker and CSF appears bright. The FLAIR sequence, like T2, suppresses the CSF signal, making it easier to visualize lesions typical of Multiple Sclerosis. These lesions appear hypointense in T1 and hyperintense in T2, PD, and FLAIR sequences, with FLAIR providing the greatest contrast with surrounding tissues.37 With the advancement of Artificial Intelligence (AI) and deep neural networks, these barriers are progressively being overcome. Recent studies have enhanced MRI's predictive capability through AI, with a particular focus on machine learning. New automated methods for lesion monitoring in MRI scans are being developed, offering greater precision and efficiency in diagnosing MS. According to Yousef et al.[17](2024), deep learning techniques, particularly Convolutional Neural Networks (CNNs), have demonstrated superior performance in segmenting brain structures and lesions compared to conventional methods. CNNs are particularly effective because they can automatically learn hierarchical features from raw image data, capturing complex spatial patterns and subtle differences that are difficult for traditional algorithms to detect.
Lesion segmentation in MS is particularly challenging due to the heterogeneity of lesions, variations in tissue contrast, and differences in image acquisition protocols. Research such as that by SadeghiBakh et al. $^{18}$ (2022) indicates that CNN-based models can recognize complex patterns in MRI images, improving accuracy in identification and reducing reliance on manual intervention. Furthermore, the use of AI for segmentation enhances reproducibility and sensitivity in lesion detection. Studies like those by Salem et al. $^{19}$ (2022) suggest that automated segmentation methods can be integrated into clinical workflows to assist professionals in decision-making, contributing to faster and more accurate diagnoses. Thus, the application of deep neural networks in lesion segmentation for MRI represents a significant advancement in neurology, enabling better assessment of MS progression and offering personalized therapeutic strategies for patients.
## III. METHODOLOGY
This study is an integrative literature review with a qualitative and exploratory approach, focused on studies addressing Multiple Sclerosis (MS), particularly the application of neural networks for lesion segmentation in Magnetic Resonance Imaging (MRI) aimed at improving early diagnosis. The central research question guiding the review was: "How can artificial intelligence, specifically neural networks, improve lesion segmentation in Magnetic Resonance Imaging (MRI) for the early diagnosis of Multiple Sclerosis?"
Steps were defined to ensure methodological rigor, including the formulation of the research question, comprehensive database searches, selection, reading, thematic analysis of the selected studies, and the use of the PRISMA checklist (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) to structure the review process. The articles were analyzed using thematic analysis, allowing for the identification of recurring themes related to AI applications in MS lesion segmentation.
The PRISMA checklist guided the establishment of inclusion and exclusion criteria, the systematic selection of studies, and the transparent reporting of findings, enhancing the reproducibility and reliability of the review process. The literature search included studies published in Portuguese, English, and Spanish, accessed remotely through databases such as PubMed (National Library of Medicine), Virtual Health Library (BVS), Nature, Google Scholar, and Arxiv. Search Strategy and Keywords:
Specific keywords and Boolean operators were used to ensure the comprehensiveness of the search. For instance.
1. The search in the PUBMED database was conducted using relevant terms to ensure the retrieval of pertinent studies. The search terms were combined using Boolean operators as follows:
(((("Hyperintense lesion"[Title/Abstract]) OR ("Hyperintensity"[Title/Abstract]) OR ("Hypointense lesion"[Title/Abstract]) OR ("Hypointensity"[Title/Abstract])) AND (Artificial Intelligence[Title/Abstract] OR AI[Title/Abstract])) AND (Multiple Sclerosis[Title/Abstract])) AND (Black Holes[Title/Abstract])) AND (Magnetic Resonance Imaging[Title/Abstract] OR MRI[Title/Abstract] OR MR[Title/Abstract])) AND (Neural Network[Title/Abstract] OR Tiramisu Network[Title/Abstract]) AND (Segmentation[Title/Abstract]).
2. Second Search: The search conducted in the Virtual Health Library (BVS) applied the keywords: "multiple sclerosis," "magnetic resonance imaging," "artificial intelligence," or "AI."
3. Third Search: In the Nature database, the terms "magnetic resonance imaging," "artificial intelligence," "multiple sclerosis," and "black holes" were used.
4. Fourth Search: The terms used in the Arxiv database were the same as in BVS: "multiple sclerosis," "magnetic resonance imaging," and "artificial intelligence."
5. Fifth Search: In Google Scholar, the following keywords were used: "artificial intelligence," "multiple sclerosis," "black holes," "magnetic resonance imaging," "Neural Network," "Hypointense lesion," "Hypointensity," "Hyperintensity," and "Hyperintense lesion."
### Reproducibility and Explainability
The methodology was carefully structured to ensure the reproducibility of the research. All search criteria, selection procedures, and thematic analysis methods were transparently documented. The qualitative analysis focused on identifying thematic patterns, segmentation methods, and AI advancements, ensuring objectivity and reliability in the interpretation of the studies.
#### Limitations and Ethical Considerations
Despite the methodological rigor, limitations such as potential publication bias and heterogeneity in MRI acquisition protocols across studies were acknowledged. Ethical considerations were strictly followed, with only publicly available data used and no involvement of patient data or experimental interventions. Studies were excluded if they lacked summaries, were inaccessible, or did not focus specifically on the use of artificial intelligence in Multiple Sclerosis. Included works comprised articles, research papers, theses, dissertations, final projects, and book chapters, published from 2022 onward, to ensure the relevance and currency of the findings.
## IV. RESULTS
The search in the PUBMED database resulted in 472 articles, and BVS yielded 31 articles. The third search conducted in Nature resulted in 3 works, the terms used in ARXIV produced 11 results, and Google Scholar returned 27 articles. The Search across databases resulted in 472 articles, of which 9 were eliminated due to duplication. Of the 463 works, 19 predated the established publication deadline, and after reading the abstracts, 419 articles were eliminated. Therefore, 25 studies were potentially eligible for this review, which, after full-text reading, were used to prepare this report.
Table 1: Representing the Articles used and Subitems were Depicted
<table><tr><td>Title</td><td>Key Points</td><td>Study Type</td></tr><tr><td>Boosting Multiple Sclerosis Lesion Segmentation through Attention Mechanism</td><td>- Comparison of unsupervised, supervised, and deep learning techniques
- Use of residual U-Net with channel attention
- Intensity thresholds in FLAIR</td><td>Original Research</td></tr><tr><td>Multiple Sclerosis Lesions Segmentation Using Attention-Based CNNs in FLAIR Images</td><td>- ResNet-based architecture with spatial and channel attention
- Superior performance in Dice Score
- Clinical validation</td><td>Original Research</td></tr><tr><td>AI-based Detection of Contrast-Enhancing MRI Lesions in MS</td><td>- Expert-labeled lesions
- 3D probability masks for lesion segmentation
- Comparable performance to human readers</td><td>Original Research</td></tr><tr><td>Assessment of AI Automatic MS Lesion Delineation Tool</td><td>- Comparison with existing segmentation tools
- Transfer learning for local data adaptation
- High accuracy and clinical acceptance</td><td>Original Research</td></tr><tr><td>Framework to Segment MS Lesions Using VGG-UNet</td><td>- Comparison between UNet, SegNet, VGG-Unet, and VGG19
- Python implementation with 80% training/20% validation</td><td>Experimental Study</td></tr><tr><td>3D CNN Framework for Nuclear Medicine</td><td>- Use of CNNs with convolutional and pooling layers
- U-Net effective for biomedical</td><td>Original Research</td></tr><tr><td></td><td>segmentation</td><td></td></tr><tr><td>Deep 3D Neural Network for Brain Structures Segmentation</td><td>- CNN-Transformer hybrid for improved segmentation
- Efficient segmentation, with future optimization plans</td><td>Original Research</td></tr><tr><td>Deep Whole Brain Segmentation with 7T MRI</td><td>- 7T MRI provides better spatial resolution
- Higher accuracy in segmenting brain structures</td><td>Original Research</td></tr><tr><td>DBSegment for Deep Brain Structures</td><td>- Use of MNI and CIT168-based registration
- Improved accuracy through registration for 3T-to-7T adaptation</td><td>Original Research</td></tr><tr><td>Deep Learning-Based PET/MR Radiomics for MS</td><td>- 2D U-Net segmentation
- Multimodal PET/MR models improved ARR prediction</td><td>Original Research</td></tr><tr><td>Single-Timepoint MS Lesion Classification Using CNN</td><td>- Acute vs chronic lesion detection using radiomics
- Effective classifier with CNNs</td><td>Original Research</td></tr><tr><td>New MS Lesion Segmentation with Residual Attention U-Net</td><td>- Use of U-Net with attention gates for improved lesion detection</td><td>Original Research</td></tr><tr><td>Improving MS Lesion Detection with Cascaded 3D CNN</td><td>- Use of deformation fields for sequential scans
- Better sensitivity and fewer false positives</td><td>Original Research</td></tr><tr><td>Thalamus Segmentation Using 3D CNN</td><td>- 3D CNN outperforms FastSurfer and FSL in detecting thalamic atrophy</td><td>Original Research</td></tr><tr><td>Automated Cervical Spinal Cord Segmentation</td><td>- U-Net 2D with residual attention for spinal cord segmentation
- High segmentation accuracy</td><td>Original Research</td></tr><tr><td>Active/Inactive Plaque Segmentation Using DeepLabV3+</td><td>- Efficient and accurate segmentation of plaques in FLAIR images</td><td>Original Research</td></tr><tr><td>Predicting MS Disease Progression with MRI Biomarkers</td><td>- MRI biomarkers crucial for personalized MS prognosis
- Machine learning models offer promising potential</td><td>Literature Review</td></tr><tr><td>Prognostic Relevance of MRI in Early Relapsing MS</td><td>- MRI identifies lesions that guide treatment decisions
- Early treatment reduces risk</td><td>Literature Review</td></tr><tr><td>Repurposing MRI Archives for MS Research</td><td>- AI-based model for segmenting brain structures and lesions
- High correlation with EDSS and treatment effects</td><td>Original Research</td></tr><tr><td>MLV2-Net for Meningeal Lymphatic Vessel Segmentation</td><td>- First automatic method for meningeal vessel segmentation using CNN</td><td>Original Research</td></tr><tr><td>Cortical Lesion Segmentation</td><td>- Quantifies uncertainty in cortical lesion</td><td>Original</td></tr><tr><td>Uncertainty</td><td>segmentation
- Improved detection reliability</td><td>Research</td></tr><tr><td>Instance-Level Quantitative Saliency for MS Lesion Segmentation</td><td>- Use of SmoothGrad and Grad-CAM++ for segmentation explanations</td><td>Original Research</td></tr><tr><td>Ensemble CNN and Uncertainty Modeling for MS Lesion Detection</td><td>- Improved segmentation with multiple evaluators and optimized U-Net 2D</td><td>Original Research</td></tr><tr><td>Multi-Modal CNN for MS Lesion Detection</td><td>- 2D U-Net CNN for segmenting MS lesions
- Improved performance with fewer computational costs</td><td>Original Research</td></tr><tr><td>Capuchin Search Algorithm for MS Diagnosis</td><td>- Use of CNNs for MRI image processing
- Improved precision and recall with ensemble models</td><td>Original Research</td></tr></table>
## v. DISCUSSION
Rigorous lesion segmentation in MRI images is essential for obtaining an accurate diagnosis and monitoring of Multiple Sclerosis (MS). Traditional segmentation approaches based on manual or semi-automatic techniques have significant drawbacks, such as dependence on expert domain knowledge and inter- and intra-observer variability. In clinical practice, traditional methods are often time-consuming, require specialized expertise, and struggle to detect subtle lesions, particularly in the early stages of MS, potentially delaying diagnosis and treatment. To overcome these limitations, AI-based approaches, particularly deep neural networks, have gained increasing attention for automating and improving lesion segmentation.
Figure 1 illustrates the number of publications that have used different AI models in the lesion segmentation process in MS, from image acquisition to result analysis and validation. The pipeline begins with image acquisition, followed by preprocessing, based on different MRI sequences (T1, T2, and FLAIR), essential for identifying lesions. Preprocessing steps, such as skull stripping to remove non-brain tissues, normalization of image intensity to reduce noise, and artifact removal, are critical for enhancing input data quality and improving segmentation accuracy. Feature extraction then focuses on highlighting areas of interest using contrast enhancement techniques and improving lesion detection.

Figure 1: Use of AI in Publications Described as Model and Variation of Segmentation Methodologies (OHLC) For segmentation, classic methods such as thresholding and probabilistic models are employed, alongside modern methods like deep neural networks, including CNN, U-Net, and Attention U-Net. Post-processing is applied to refine results by reducing false positives and applying smoothing techniques, improving model precision. The final phase includes analysis and validation, comparing automated segmentation with manual segmentation by specialists using metrics such as Dice Score and sensitivity.
Recent investigations have demonstrated that deep learning-based models can outperform conventional techniques, offering higher accuracy and efficiency in identifying lesions, as described by Du et al.[20](2023). Deep neural networks (DNNs), particularly Convolutional Neural Networks (CNNs), are particularly effective for medical image segmentation because they can automatically learn hierarchical patterns from raw MRI data, capturing complex spatial features that are difficult for traditional algorithms to detect. CNN-based models such as U-Net[21] and nnU-Net[22] have shown excellent performance in segmenting hyperintense and hypointense lesions in brain white matter.
Furthermore, optimization algorithms such as the Capuchin Search Algorithm (CapSA) and fuzzy c-means (FCM), when combined with CNNs, have enhanced lesion detection capabilities in MS. Despite these advancements, challenges persist, particularly the need for large, well-labeled datasets to train deep learning models effectively $^{21}$. Dataset heterogeneity and variation in MRI acquisition protocols between institutions can impact model performance, raising concerns about generalizability $^{22,25}$.
Various strategies have been proposed to improve model accuracy and robustness, including attention mechanisms, residual learning, and hybrid architectures. Networks like U-Net, VGG-UNet $^{26}$, ResNet $^{18}$, and EfficientNet $^{27}$ have demonstrated significant improvements in lesion segmentation by optimizing feature detection and minimizing error rates. Improving sensitivity - the ability to correctly identify true positives - remains a major focus, with models like U-Net $^{20,21,25,28,29}$, DeepLabV3 $^{+27}$, and attention-based networks being widely adopted to enhance this parameter.
AI-based models have shown effectiveness particularly in segmenting hyperintense lesions on FLAIR images $^{18,21,24,25,27,30}$, enabling better longitudinal assessment of MS progression. For example, the VicoroCascade model $^{19}$ demonstrated high sensitivity and reduced false positives, supporting its use for disease monitoring. Combining information from multiple sequences, such as T2 and FLAIR, has further improved lesion detection, especially in early and subtle cases.
Uncertainty modeling techniques, like Grad-CAM++ and SmoothGrad $^{30}$, have been employed to quantify prediction confidence, improving interpretability and reducing the risk of misclassification. Additionally, hybrid networks combining CNNs with spatial and channel attention modules $^{18,24,31,32}$ have increased sensitivity without compromising specificity.
Comparative studies show that some deep learning models now approach the performance of experienced radiologists, achieving segmentation results with less inter-observer variability $^{20,25,33}$. Other innovations include techniques for differentiating active from inactive lesions without the need for contrast agents like gadolinium, reducing patient risk. For example, DeepLabV3+ combined with EfficientNetBO can distinguish lesion types in FLAIR images with high accuracy $^{27}$.
Table 2: Table about the Main Models Discussed
<table><tr><th>Model</th><th>Architecture</th><th>Segmentation Strategy</th><th>Input Modality</th><th>Dice</th><th>Differentials</th></tr><tr><td>U-Net</td><td>Multi-branch Fully Convolutional Neural Network (FCNN)</td><td>Automatic extraction of radiomic features for ARR prediction</td><td>PET/MR</td><td>0.81</td><td>Combination of PET and MRI for better prediction of relapse rate (ARR)</td></tr><tr><td>MindGlide</td><td>Variant of U-Net with automatic hyperparameter tuning</td><td>Volumetric extraction of lesions and brain regions for longitudinal analysis</td><td>Qualquer RM</td><td>0.61</td><td>Works with incomplete modalities and can be applied to heterogeneous clinical data</td></tr><tr><td>DeepLab V3+</td><td>Encoder-decoder with ASPP (spatial pyramid) and SE-Block (Squeeze and Excitation)</td><td>Pre-processing with bone removal, normalization, and use of SE and ASPP blocks to improve segmentation</td><td>FLAIR</td><td>0.7624</td><td>Use of EfficientNetBO as backbone for performance and efficiency optimization</td></tr><tr><td>ResNet</td><td>Based on residual blocks with spatial and channel attention (SCA-VoxRes)</td><td>Patches of 80x80x80 applied to 4D images (CLAHE + borders)</td><td>FLAIR</td><td>0.6430</td><td>Use of spatial and channel attention to improve lesion detection</td></tr><tr><td>Vicoro Cascade</td><td>Cascaded architecture with 3D convolutional networks and 3D multimodal fusion Fully Convolutional Network (FCNN)</td><td>Two-stage network: 1st detects candidates for new lesions, 2nd reduces false positives</td><td>T2-FLAIR (Baseline + Follow-up)</td><td>0.42</td><td>Focus on detecting new lesions over time, improving prognostics</td></tr></table>
Five recent CNN architectures were compared in this study (Table 2), highlighting different strengths depending on the clinical objective: diagnosis, prognosis, or disease monitoring. Models like DeepLabV3+ SE EfficientNetB0 used Squeeze and Excitation (SE) and Atrous Spatial Pyramid Block (ASPP) techniques for enhanced segmentation, while nnU-Net demonstrated adaptability across heterogeneous clinical data. Vicorob, with its cascaded FCNN pipeline, stood out in detecting new lesions over time by minimizing false positives.
Figure 2 shows the prevalence of model use, with U-Net being the most reported in the literature (15 studies), followed by growing interest in more automated, adaptable frameworks like nnU-Net. These trends indicate the shift toward scalable, flexible solutions in MS lesion segmentation.
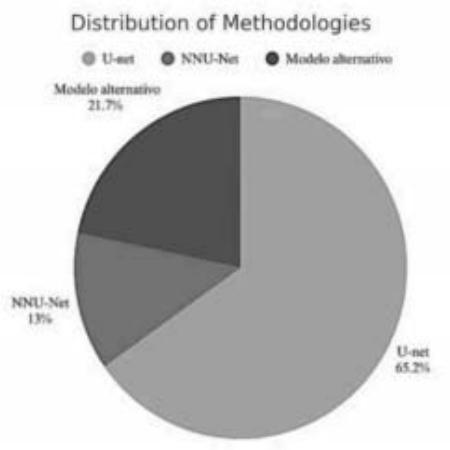
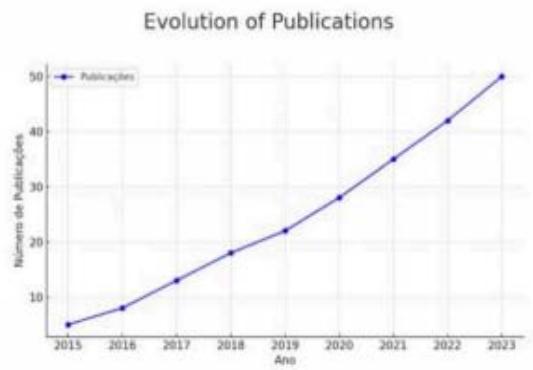
Figure 2: Pie Chart Showing the Distribution of the most commonly used Models
Source: Prepared by the Authors Themselves
The development of these advanced segmentation methods is essential to support early MS diagnosis and progression monitoring. However, as noted by Sadeghibakhi et al.[18](2022) and Schlaeger et al.[33](2023), rigorous clinical validation, population diversity testing[26], and interpretability improvements are necessary for safe adoption into clinical workflows[34]. Reducing algorithmic bias, improving model explainability, and ensuring compatibility with different imaging protocols remain key goals for future research.
Thus, the combination of MRI with deep neural networks represents a transformative advancement for diagnosing and monitoring MS. As emphasized by Manimegalai et al.[28](2022), continuous development is vital to optimize these tools for clinical use, enabling faster, safer, and more accurate care for patients with MS.
## VI. CONCLUSION
The application of neural networks in lesion segmentation for magnetic resonance imaging (MRI) has proven to be a promising approach for the diagnosis and monitoring of Multiple Sclerosis (MS). The use of artificial intelligence in this context enables faster and more accurate image analysis, reduces the subjectivity inherent in manual segmentation, and assists specialists in identifying and tracking disease progression over time. Although advancements in the field are notable, important obstacles remain. There is a critical need for the standardization of image acquisition protocols across centers and the development of models that are robust and generalizable to diverse patient populations and different MRI equipment. Collaborations between research institutions, hospitals, and imaging manufacturers could help establish standardized imaging protocols, while multi-center clinical trials would be crucial to validate AI models across heterogeneous populations.
Furthermore, the interpretation of algorithm-generated results still requires extensive clinical validation to ensure their applicability in real-world settings. In addition to clinical validation, AI models must undergo rigorous regulatory evaluation and approval, such as by agencies like the FDA or through CE marking, to guarantee that they meet the necessary standards for widespread clinical implementation. Future research should focus on creating more resilient, interpretable, and transferable models, trained on diverse datasets, to improve the generalizability and robustness of segmentation tools across different clinical environments. Emphasis on model explainability and minimizing bias will also be critical for building trust among clinicians and patients.
Thus, the integration of artificial intelligence with healthcare professionals' expertise represents a promising path for enhancing MS diagnosis and treatment. The continuous evolution of these technologies holds the potential to improve decision-making accuracy, optimize healthcare resources, and ultimately provide a better quality of life for patients affected by MS.
### ACKNOWLEDGMENTS
We thank FAEP/UMC for their support and instrumentation of this research, which may contribute to improving lives through early diagnosis and better disease management.
Declarations Consent to Participate
This study used public datasets and did not involve direct human subjects; therefore, informed consent was not required.
Conflicts of Interest
The authors declare no conflicts of interest.
Generating HTML Viewer...
Funding
No external funding was declared for this work.
Conflict of Interest
The authors declare no conflict of interest.
Ethical Approval
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
Almir Rodrigues Tavares. 2026. \u201cLeveraging Neural Networks for Longitudinal Analysis of Multiple Sclerosis and Other Neurodegenerative Diseases\u201d. Global Journal of Computer Science and Technology - D: Neural & AI GJCST-D Volume 25 (GJCST Volume 25 Issue D1): .
Subject: Global Journal of Computer Science and Technology - D: Neural & AI
Authors:
Almir Rodrigues Tavares, Vitória Lorrani dos Santos, Bruna Soares Mucoucah, Manuel Pereira Coelho Filho, Cleber Silva de Oliveira, Felipe Cabral, Thiago de Souza Franco, Gabriely Gomes de Sa,· Maria Fernanda Mendes, Antonio Jose da Rocha, Marcia Aparecida (PhD/Dr. count: 0)
Multiple Sclerosis (MS) is a progressive neurodegenerative disease affecting the Central Nervous System (CNS), leading to demyelination and neurological impairment. Early diagnosis and continuous monitoring of disease progression are crucial for effective treatment. Magnetic Resonance Imaging (MRI) remains the primary tool for detecting MS lesions; however, traditional segmentation methods rely heavily on visual analysis and struggle to detect earlystage lesions. This study reviews the application of Convolutional Neural Networks (CNNs) for automated lesion segmentation in MS.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
×
This Page is Under Development
We are currently updating this article page for a better experience.
Thank you for connecting with us. We will respond to you shortly.