Since most of medicinal mushrooms are rare in nature production of fungal fruiting bodies using artificial cultivation in a form of farming has been intensively established during the last 40 years. Solid state cultivation of various medicinal mushroom mycelia in various types of bioreactors, suitable for veterinary use, appears slightly in last few decades. Developing submerged technologies, using stirred tank and air lift bioreactors, are the most promising technologies for fast and large cultivation of medicinal pharmaceutically active products for human need. This potential initiates the development of new drugs and some of the most attractive over the counter human and veterinary remedies. This article is an overview of the engineering achievements in comprehensive medicinal mushroom mycelia cultivation.
## I. INTRODUCTION
A total of more than 200 medicinal functions are thought to be produced by medicinal mushrooms (MM) and fungi including antitumor, immunomodulating, antioxidant, radical scavenging, cardiovascular, cholesterol-lowering, antiviral, antibacterial, anti-parasitic, antifungal, detoxification, hepatoprotective, anti-diabetic, anti-obesity, neuroprotective, neuroregenerative, and other effects. Also, substances derived from MM can be used as painkillers or analgetics [4, 5, 6]. The best implementation of MM drugs and dietary supplements has been in preventing immune disorders and maintaining a good quality of life, especially in immunodeficient and immuno-depressed patients, patients under chemotherapy or radiotherapy, patients with different types of cancers, chronic blood-borne viral infections of Hepatitis B, C, and D, different types of anemia, the human immunodeficiency virus/acquired immunodeficiency syndrome (HIV/AIDS), herpes simplex virus (HSV), Epstein Bar virus, Influenza viruses A and B, H5N1 [7], COVID-19 [8-10], West Nile virus, chronic fatigue syndrome, patients with chronic gastritis and gastric ulcers caused by Helicobacter pylori, and people suffering from dementia (especially Alzheimer's disease) [1, 2, 3, 5].
To combat these threats, humankind is focusing more and more attention on mushrooms and mushroom products. Mushrooms themselves are consumed regularly as part of the human diet and are treated as healthy or functional foods. On the other hand, the term mushroom nutraceuticals or dietary supplements has been applied to products derived from medicinal mushrooms that are taken to enhance general health and fitness but are not a regular food but a dietary food supplements [1].
## II. MAIN PHARMACEUTICALLY ACTIVE COMPOUNDS
The main components of these supplements are polysaccharides, triterpenes and immunomodulatory proteins. Polysaccharide components, in particular, have been widely investigated as a source of anti-tumor and immunostimulating agents. They are widely distributed in mushrooms, with over 660 species from 183 genera reported to contain pharmacologically active polysaccharides. About $77\%$ of all medicinal mushroom products are derived from the fruiting bodies, which have been either commercially farmed or collected from the wild, $21\%$ from culture mycelium and $2\%$ from culture broths. Precisely how these products work is still a matter of conjecture, but numerous laboratory animal tests as well as human clinical trials have shown them to be effective. In some cases, attention has focused on a single bioactive mushroom component and its effectiveness in treating specific disease conditions, much like a pharmaceutical. In the case of nutraceuticals/dietary supplements, emphasis has been placed on a combination of components that collectively impact on an individual's overall health and quality of life [2].
Many such products are currently available, and their market value worldwide increased from 1.2 billion in 1991 to 3.6 billion USD in 1994. The combined market value of medicinal mushrooms, mushroom extracts and derived products in 1999 was estimated to be 6.0 billion USD. That year, the United States nutraceutical market alone was valued at 35 million USD. Since then, demand has increased between $20\%$ and $40\%$ annually depending on the species, with Ganoderma-based dietary supplements alone valued at 1.6 billion USD. The MM industry has grown from small-scale (cottage-based) operations aimed at supplementing household incomes, to medium and mega-sized industrial
ventures. This review examines the past, present and future of MM development, and includes a pyramid model addressing key issues [3].
They are of different chemical composition, such as polysaccharides, glycopeptide-protein complexes, proteoglycans, proteins and triterpenoids, with most scientific attention focussed to the group of non-cellulosic $\beta$ -glucans with $\beta$ -(1-3) linkages in the main chain of the glucan, and additional $\beta$ -(1-6) branch points, that are characteristic for the antitumor and immuno-stimulating action. Mushroom polysaccharides do not attack cancer cells directly, but produce their antitumor effects by activating different immune responses in the host. Their mechanisms of action involve them being recognized by several immune cells receptors as non-self molecules, so the immune system is stimulated by their presence. Structurally different $\beta$ -glucans have different affinities toward receptors and thus generate different host responses [12].
Immunomodulating and antitumor activities of these metabolites are related to immune cells such as hematopoietic stem cells, lymphocytes, macrophages, T cells, dendritic cells, and natural killer cells, involved in the innate and adaptive immunity, resulting in the
production of biologic response modifiers [13]. Clinical evidence for antitumor and other medicinal activities come primarily from some commercialised purified polysaccharides, such as lentinan from Shiitake - Lentinula edodes, krestin from Coriolus versicolor, grifolan from Grifola frondosa, and schizophyllan from Schizophyllum commune [12,14], but polysaccharide preparations of some other medicinal mushrooms also show promising results.
## III. WOOD DEGRADING BASIDIOMYCETES WITH PHARMACOLOGICAL EFFECTS
Almost unknown in Western Scientific Research only three decades ago, some of the wood degrading Basidiomycetes became intensely and systematically studied due to their promising pharmacological effects (Figures 1 A-E). Among them, Ganoderma lucidum - Ling zhi or Reishi, Grifola frondosa - Maitake, Hericium erinaceus - Lion mane and Cordyceps militaris have been known from the traditional Asian medicine, and Trametes versicolor Turkey tale, (previous Coriolus versicolor) are the subject of this review.






Figure 1: Ganoderma lucidum (A), Trametes versicolor (B) (previous Coriolus versicolor), Grifola frondosa (C), Hericium erinaceus (D) and Cordyceps militaris (E)
Technological possibilities for commercial cultivation gave rise to the number of patents, which are protecting inventions related to new methods and technologies for cultivation of fruit bodies and/or mycelium biomass, methods of active compounds isolation, and development of new commercial formulations and products. Ganoderma lucidum has been identified as a medicinal mushroom with the largest number of patented inventions [11].
### a) Ganoderma Lucidum
Ganoderma (W. Curt.: Fr.) Lloyd is a white rot wood Basidiomycete that degrades lignin and possess hard fruiting body. G. lucidum is from Ganoderma sp. the most often reported as a source of various medicinal compounds. In Asian traditional medicine, the fruiting body of G. lucidum, named Ling-zhi in Chinese and Reishi in Japanese language, has been used for treatment of several diseases for thousands of years, as
reported in Shen Nong's Materia Medica (Leung et.al., 2002; Kim and Kim, 2002; Lin, 2009).
Modern uses of Ganoderma include treatment of coronary heart diseases, arteriosclerosis, hepatitis, arthritis, nephritis, bronchitis, asthma, hypertension, cancer and gastric ulcer [15,16] Publications also report on Ganoderma antiallergenic constituents [17], immunomodulatory action [18-20], antitumor activity [21-23] cardiovascular effects [24], liver protection and detoxification, and effects on nervous system [22,25]. New reports emphasize its potential in treatment of viral, especially HIV infections [26, 27].
Pharmaceutically active compounds from Ganoderma lucidum include triterpenoids, polysaccharides (1,6-β-D-glucans and 1,3-β-D-glucans), proteins, proteoglycans, steroids, alkaloids, nucleotides, lactones and fatty acids, amino acids, nucleotides, alkaloids, steroids, lactones and enzymes. Over 100 triterpenoids were found in Ganoderma spp., such as ganoderic (highly oxygenated $C_{30}$ lanostane-type triterpenoids), lucidenic, ganodermic, ganoderenic and ganolucidic acids, lucidones, ganoderals and ganoderols [11].
A large and diverse spectrum of chemical compounds with a pharmacological activity has been isolated from the mycelium, fruiting bodies and sclerotia of Ganoderma mushrooms: triterpenoids, polysaccharides, proteins, amino acids, nucleotides, alkaloids, steroids, lactones, fatty acids and enzymes [28, 29]. There is abundant evidence that polysaccharides isolated from G. lucidum are immunomodulatory effective [4, 30-34]. Studies have shown that the most active immunomodulatory polysaccharides are $(1\rightarrow 3)$ - $\beta$ -D and $(1\rightarrow 6)$ - $\beta$ -D glucans, that can be precipitated by ethanol. Their prevailing structure is $\beta$ -1,3 D-glucopyronan with 1-15 units of $\beta$ -1,6 monoglucosyl side chains. Their 1,3-linked backbone, relatively small side chains, and an organized helical structure are beneficial for the immunostimulation. Although they are chemically heterogeneous, these polysaccharides are usually termed as $\beta$ -glucans [30-32, 36, 37].
Bioactive polysaccharides have been isolated from different sources of G. lucidum: basidiocarps, spores and from the mycelial biomass cultivated in liquid culture. Few have been isolated from the culture medium [37]. Though different models of the fungal cell wall differ somewhat, scientists agree that $\beta$ -glucan is not located on the surface of the wall but is more or less immersed in the wall material. Generally, the cell wall of most fungi contains five main components: $(1 \rightarrow 3)$ - $\beta$ -D glucan, $(1 \rightarrow 6)$ - $\beta$ -D glucan, chitin, and glycoproteins. $\beta$ -glucan forms $9 - 46 \%$ of cell wall mass [36, 37]. A large number of studies have shown that polysaccharides of G. lucidum, especially $\beta$ -D glucan, can modulate the functions of many components of the immune system
such as the antigen-presenting cells, T and B lymphocytes, NK cells, neutrophil granulocytes, dendritic cells and, on cytokine production [30, 32, 38, 39].
### b) Grifola Frondosa
Grifola frondosa also known as Maitake is white rot lignin degrading Basidiomycete with excellent nutritional and medicinal properties. Grifola frondosa active compounds primarily belong to the group of polysaccharides (especially 1,6-β-D-glucans and 1,3-β-D-glucans), glycoproteins, and proteins. This products have been used for treatment of a series of diseases, including hepatitis, arthritis, nephritis, bronchitis, coronary heart diseases, asthma, arteriosclerosis, hypertension, cancer and gastric ulcer. Newer investigations report on Grifola frondosa antiallergenic constituents, immunomodulatory action and treatment of HIV infections, antitumor and cardiovascular effects, liver protection and detoxification and effects on nervous system [40].
G. frondosa has gained in popularity among consumers, not only because as a gastronomic delight of its taste and flavor, but also because of its reported medicinal value. Its active compounds primarily belong to the group of polysaccharides (especially 1,6-β-D-glucans and 1,3-β-D-glucans), glycoproteins, and proteins. Medicinal effects of G. frondosa are numerous, including anti-cancer activity [41, 42], immune system stimulation [43,44], effects on angiogenesis [45], reduction of benign prostatic hyperplasia [46], antibacterial [47], and antiviral effects [48], effects on lipid metabolism and hypertension [41], antidiabetic activity [49], vitality and performance enhancement [50], antioxidant effects, and beneficial cosmetic effects on skin [51]. According to Shen (2001), more than 20 anti-tumor polysaccharides have been isolated and purified from G. frondosa [52].
### c) Hericium Erinaceus
In $H.$ erinaceus various pharmaceutically active substances were found. Phytosterols (β-sitosterol and ergosterol), lower the content of low density lipoproteins (LDL) and triglycerides that operate anticarcinogenic as well they reduce the metabolism of fats [53]. In $H.$ erinaceus fruitbody numerous constituents such as are polysaccharides, proteins, lectins, phenols, hericenones, erinacines and terpenoids were identified. They strengthen the immune system, relieve gastritis and gastrointestinal infections, reflux and upset stomach due to stress [54].
H. erinaceus water-soluble polysaccharides increased activity of macrophages and other immune cells in the fight against cancer cells, but also demonstrated the reduction of formation of metastases. The most outstanding activity of the extract of H. erinaceus is that it strengthens the immune system and
activate the synthesis of nerve growth factor [55]. Due to the increased proliferation of T and B-lymphocytes it strengthens the immune system and strengthens the body's natural defences Thus, H. erinaceus expresses very positive effects on prolongation of quality of life of the cancer patients [53].
Among the compounds isolated from fruiting bodies and cultured mycelia of $H.$ erinaceum, most interesting are the low-molecular-weight compounds belonging to a group of cyathin diterpenoids (erinacines A-K, P, and Q). Several of them, i.e. erinacines A-H, are known to have a potent stimulating effect on nerve growth factor (NGF) synthesis in vitro [56-61].
H. erinaceus polysaccharides (HEP) derived from fruiting bodies and mycelium severed as effective therapeutic agents in liver damage-associated diseases. A study demonstrated that the serum levels of aspartate aminotransferase and glutamic pyruvic transaminase activities in carbon tetrachloride-induced hepatic injury were decreased by administration with extracellular and intracellular HEP (200, 400, and $800\mathrm{mg/kg/day}$ ) from mycelium, but the blood lipid levels in the serum of mice were enhanced [62]. Moreover, Kim et al. found that 10 mg/kg/day HEP markedly alleviated Salmonella typhimurium-induced liver damage and reduced infected mice mortality [63]. Zhang et al. revealed that endo-HEP potent hepatoprotective effect in vivo, which may be due to its powerful antioxidant capacity. Taken together, the HEP could be exploited as a supplement in the prevention of hepatic diseases [64].
### d) Trametes Versicolor
Trametes versicolor, previous Coriolus versicolor, also known as Turkey tail mushroom is one of the most attractive medical fungi. It is known for its use and success as a remedy in Asian traditional medicine [65-67]. T. versicolor pharmaceutical activities include immunomodulation, antibody production, activation of apoptosis etc.
The two most prominent products of $T.$ versicolor are polysaccharide Krestin (PSK) and polysaccharide peptide (PSP) both potentially highly active pharmaceutical substances in complementary cancer treatments. PSP has a variety of physiological effects, such as immunological enhancement, antitumor, liver protecting, oxidation resistance, and reducing blood fat. PSP has been clinically used in treating cancers, hepatitis, hyperlipidemia, chronic bronchitis, and other diseases. The clinical data also demonstrate that PSP has diverse functions such as improving the quality of patients' life, enhancing learning and memory, and antiulcer effects [70-73].
Current studies support PSP as an immunotherapeutic. PSP activates and enhances the function and recognition ability of immune cells, strengthens the phagocytosis of macrophages, increases the expressions of cytokines
and chemokines such as tumor necrosis factor- $\alpha$ (TNF- $\alpha$ ), interleukins (IL-1 $\beta$ and IL-6), histamine, and prostaglandin E, stimulates the filtration of both dendritic cells and T-cells into tumors, and ameliorates the adverse events associated with chemotherapy. In recent years, immunotherapy has been widely used in cancer treatment. However, to use PSP as an immunotherapeutic at world stage, further chemical, biochemical and pharmacological studies of PSP are needed [66].
In vitro and in vivo studies have shown that the mixture of PSP and PSK has a synergistic action that highly affects immune cell proliferation and highly expresses antitumor activities [65, 67, 68, 74-76].
### e) Cordyceps Militaris
The usage of natural/herbal medicines over the synthetic ones has seen an upward trend in the recent past. Cordyceps being an ancient medicinal mushroom used as a crude drug for the welfare of mankind in old civilization is now a matter of great concern because of its unexplored potentials obtained by various culture techniques and being an excellent source of bioactive metabolites with more than 21 clinically approved benefits on human health [77-78].
The studies by many researchers in the past on Cordyceps have demonstrated that it has antibacterial, anti-fungal, larvicidal, anti-inflammatory, anti-diabetic, anti-oxidant, anti-tumor, pro-sexual, apoptotic, immunomodulatory, anti-HIV, remarkable clinical health effects including action on hepatic, renal, cardiovascular, respiratory, nervous, besides having anti-cancer, anti-oxidant, anti-inflammatory and antimicrobial activities [79-82].
Cordycepin has received much attention due to its broad-spectrum biological activity. It is known to interfere with various biochemical and molecular processes including purine biosynthesis [83, 84], DNA/RNA synthesis [85] and mTOR (mammalian target of rapamycin) signaling transduction [86]. Cordyceps has been included as one of the growing numbers of fungal traditional Chinese medicine (FTCM) used as cures for modern diseases with many products available commercially.
Great potential of pharmaceutical active compounds and Cordyceps militaris extract contains many biological bioactive materials, such as the terpenoids cordycepin and cordycepic acid, polysaccharides, sterols and other compounds [87]. Cordyceps militaris main active component is terpenoid cordycepin that inhibits the development of cancer cells including antitumor, anti-metastatic, insecticidal, anti-proliferative, anti-bacterial properties, anti-fungal, larvicidal, anti-inflammatory, anti-diabetic, anti-oxidant, pro-sexual, apoptotic, immunomodulatory, anti-HIV, remarkable clinical health effects including action on hepatic, renal, cardiovascular, respiratory, nervous,
besides having anti-cancer, anti-oxidant, anti-inflammatory and anti-microbial activities, anti-leukemia and antimalarial activities [77-82].
The second main active components are polysaccharides, which research have shown to be effective in regulating blood sugar and also have antimetastatic and antitumor properties [87-89]. The most outstanding active substance of C. militaris is Cordycepin. The structure of Cordycepin is very much similar with cellular nucleoside, adenosine. Cordycepin, i.e., $3^{\prime}$ -deoxyadenosine, is the main active constituent which is most widely studied for its medicinal value having a broad spectrum biological activity and acts like a nucleoside analogue [90].
Cordycepin alone has been widely explored for its anti-cancer/anti-oxidant activities, thus, holding a strong pharmacological and therapeutic potential to cure many dreadful diseases in future. Further investigations need to be focused on to study the mechanistic insight into the mysterious potential of this medicinal mushroom on human health and promoting
its cultivation strategies for commercialization and ethno-pharmacological use of this wonderful herb [91, 92].
## IV. CULTIVATION TECHNOLOGIES
Since medicinal mushrooms are scarce in nature, cultivation of fruit bodies on artificial media has been introduced. Traditional cultivation of fruit bodies on wood logs has been known for centuries. With time, cultivation methods have diversified, modified and developed (Figure 2). [11].
Besides on wood logs, fruit bodies are being produced on sawdust substrates in trays or beds, and in sterilised plastic bags or in bottles. In addition, production of fungal mycelia has been developed in bioreactors, utilizing submerged cultivation in liquid media, or solid state cultivation on various secondary wastes substrates from wood and agricultural industry [93].
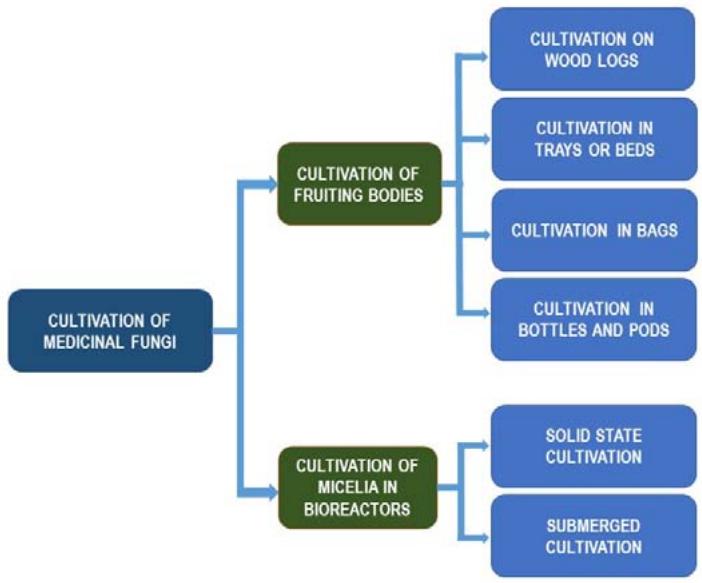
Figure 2: Technological possibilities of cultivating medicinal fungi fruit bodies or fungal biomass on a commercial scale adapted from [11]
### a) Farming Fruit Bodies Cultivation
In the wild, wood degrading mushrooms grow primarily on hardwood of trees. However, under artificial cultivation conditions, they thrive on various other substrates containing lignin and cellulose, and therefore have a high potential for recycling different types of organic waste materials from wood and agriculture industry. As lignocellulose containing wastes are produced worldwide in large quantities, and in many instances they pose a threat to the environment. Cultivation of edible and medicinal wood degrading fungi on lignocellulosic substrates offers almost unlimited possibilities and economically viable potentials
for large scale commercial cultivation on a World scale [94].
In farming fruit bodies cultivation substrate cultivation methods are divided mostly into bottle cultivation and bag cultivation, sometimes also called synthetic logs. Bag cultivation has more advantages, such as the use of more substrate, a strong body and convenient manipulation, so it is more widely used. Both production processes include the following main steps: raw material preparation, mixing, bagging (bottling) and sterilization, inoculation, spawning, embedding in soil (or transfer to mushroom house), fruiting body development, management and harvesting [94].

Figure 3: Farming production of Ganoderma lucidum fruit boddies (Photo A.Gregori )
For a production of G. lucidum fruit bodies supplemented sawdust is performed in heat-resistant polypropylene bottles or bags. Sawdust can be supplemented with rice bran (10%) and $\mathrm{CaCO}_3$ (3%), moistened with water and filled (700 g) into plastic bags. A plastic collar is then fitted onto each bag and stopped with a cotton plug. After 5h of heat treatment (95-100°C) and cooling, substrate is inoculated with grain or sawdust spawn, and incubated for 3 to 4 weeks (or until the spawn fully colonizes the substrate) [95].
### b) Solid State Cultivation
Solid state cultivation (SSC) is a three-phase heterogeneous process taking place in various bioreactors, comprising solid, liquid and gaseous phases, which offers potential benefits for the microbial cultivation for bioprocesses and products development.
Microbial growth on solid state substrate particles is very close to the growth of fungi in the natural environment. Main source of water, carbohydrates, phosphorous, nitrogen and sulphur are intrapartically bounded, therefore the microbial culture applied have to possess the abilities to access the water and essential element sources out the solid matrix Concerning that on the tips of young growing hipphae fungal polysaccharides are produced. Polysaccharide gel has actually two functions. Primary it serve as a gel media where from lignocellulitic enzymes are secreted in the solid wooden matrix and secondary as a sticky material used for anchoring of hyphae and for moving on the solid surface. Produced fungal polysaccharides have also a whole palette of their pharmaceutical activities that are used in traditional Eastern medicine already for centuries [96].



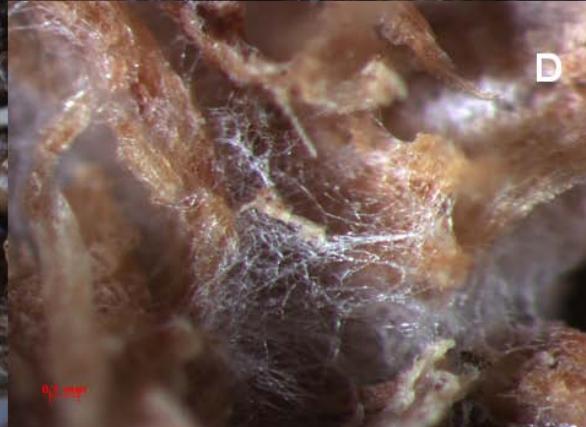
Figure 4: Growth of Hericium erinaceus on solid substrate 124 h (D), 145 h (A), 234h (B) and 280 h (C), (Photo M. Vittori)
SSC in bioreactors involves the growth and metabolism of microorganisms in fully control environment. Mycobial growth in takes place in aerated beds of moist solid materials in which the interparticle spaces contain a continuous gas phase and little or no free water. The upper limit of moisture content for solid state cultivations is determined by the absorbancy of the solid, which varies between substrates, although for most substrates a free water becomes apparent before $80\%$ moisture level is reached. Although fungal mycelia growth in SSC is very close to the growth in nature habitats fungal fruit bodies are not produced in this technology [93]. An example of SSC mycelia growth in presented in Figure 4.
Over the last two decades, SSC has gained significant attention for the development of industrial
bioprocesses, particularly due to lower energy requirement associated with higher product yields and less wastewater production with lesser risk of bacterial contamination.
An important advantage of solid state cultivation over other techniques is that a concentrated product can be obtained from a cheap substrate, such as wood and agricultural secondary residue with little pretreatment or enrichment. For this reason, solid state cultivation seems to be most appropriate for the production of pharmaceutically active animal feed supplements, for which the whole fermented substrate can be used as the product [97]. Results of solid state medicinal mushroom cultivation on various substrates are presented in Table 1.
Table 1: Solid state cultivation of various medicinal mushroom in 15 L horizontal stirred tank bioreactor (HSTR). Own results
<table><tr><td>Fungus</td><td>Substrate</td><td>Biomass (mg/g)</td><td>Intracellular IPS (mg/g)</td></tr><tr><td>Ganoderma lucidum*</td><td>Beech saw-dust</td><td>68</td><td>7.45</td></tr><tr><td>Grifola frondosa</td><td>Beech saw-dust</td><td>54</td><td>4.70</td></tr><tr><td>Trametes versicolor</td><td>Corn straw</td><td>83.5</td><td>5.95</td></tr><tr><td>Hericium erinaceus</td><td>Husked and paddy millet</td><td>350</td><td>3.07</td></tr><tr><td>Cordyceps militaris</td><td>Husked paddy millet+ rice</td><td>236</td><td>10.42</td></tr></table>
In recent years, substantial credibility in employing solid state cultivation (SSC) technique has been witnessed owing to its numerous advantages over
submerged bioprocessing (SC). In spite of enormous advantages, true potential of SSC technology has not yet been fully realized at industrial scale [96].
### c) Submeged Cultivation
Submerged cultivation of mushrooms represent the best and the fastest technology for a large scale production of medicinal mushroom mycelia and their products for a human use. In recent years, its submerged cultivation has received great interest in Asian countries as a promising alternative for efficient production of medicinal mushroom mycelia and its valuable metabolites [99].
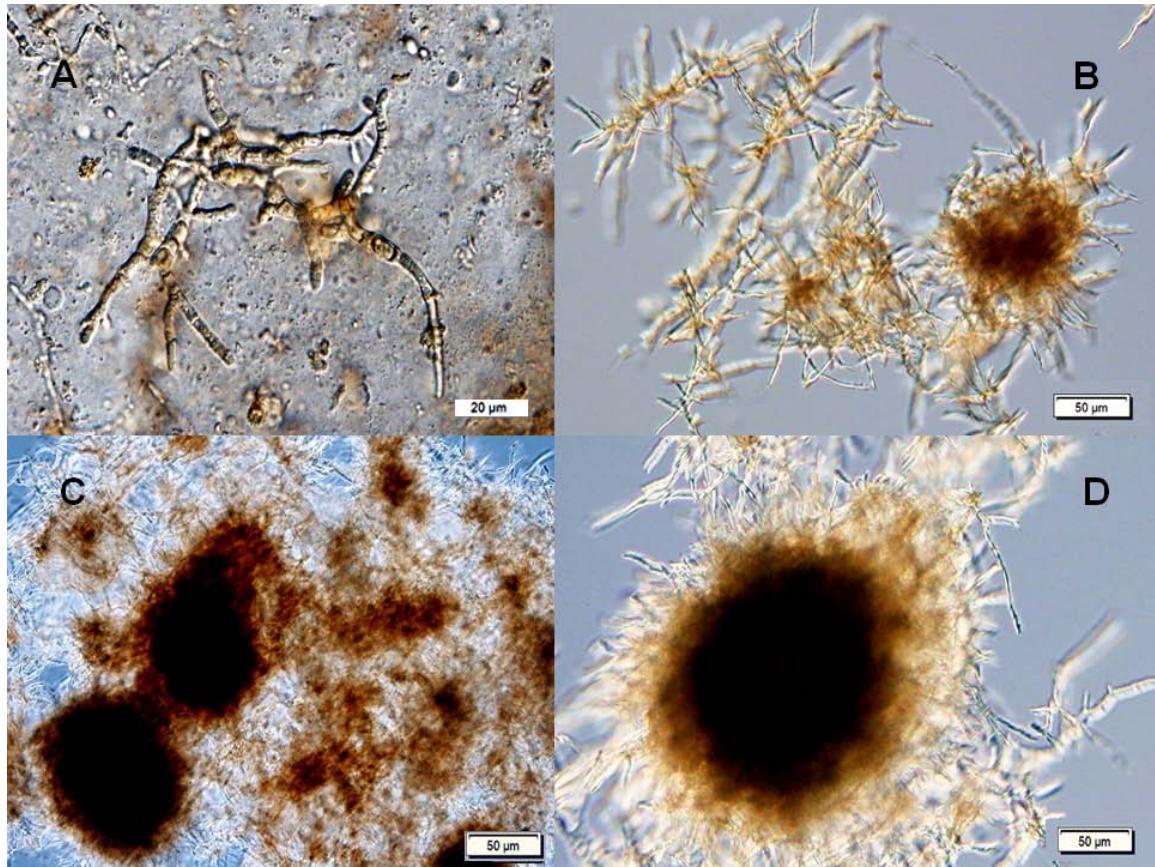
Figure 5: Growth of Hericium erinaceus in submerged cultivation. (A) fungal mycelia (14 h)(magnification 1000x); (B) mycelia clumps (218 h) (400x), (C) mycelia pellets (262 h) (100x), (D) mycelia pellets (346 h) (100x) (Photo M.Vittori)
Mycelial growth and the results of submerged medicinal mushroom cultivation of five species on various substrates are presented in Figure 5 and Table 2.
The problems in submerged cultivation of fungal biomass increase with increase the broth viscosity during cultivation because of changes in the morphology, fungal biomass and extracellular polysaccharide (EPS) production. Therefore one of the most important factors of large-scale submerged cultures in bioreactors is related to the heath and mass transfer liquid phase oxygen supply. It is necessary to characterize the variations that occur during the
submerged cultivation in bioreactors and their effects on growth and product formation [100,101].
Table 2: Results of submerged cultivation of five species of medicinal mushrooms in bioreactors
<table><tr><td>Species</td><td>Substrate</td><td>Bioreactor</td><td>Products</td><td></td></tr><tr><td>Grifola frondosa</td><td>Optimized medium for mycelial biomass: 45.2 gL-1glucose, 2.97 gL-1KH2PO4, 6.58 gL-1peptone Optimized medium for extracellular polysaccharides 58.6 gL-1glucose, 4.06 gL-1KH2PO4 3.79 gL-1peptone</td><td>15 L STR bioreactor innoculum 10% (v/v), T 25 °C, initial pH 5.5, aeration rate 8.0 vvm, agitation speed 80 rpm</td><td>Biomass 22.50 gL-1Extracellular polysaccharides 1.32 gL-1</td><td>[103]</td></tr><tr><td>Ganoderma lucidum</td><td>Potato dextrose 101.2 gL-1glucose 2% olive oil</td><td>10 L STR bioreactor innoculum 10% (v/v) T 30 °C, initial pH 5.8, aeration rate 1.0 vvm, agitation speed 300 rpm</td><td>Biomass 15.9 gL-1Extracellular polysaccharides 9.6 gL-1Intracellular polysaccharides 6.3 gL-1</td><td>[104]</td></tr><tr><td>Hericium erinaceus</td><td>30 gL-1corn flour 10 gL-1glucose 3.0 gL-1yeast extract 1.0 gL-1KH2PO4, 0.5 gL-1CaCO315 mL of corn steep liquor</td><td>15 L STR bioreactor innoculum 10% (v/v), T 28 °C initial pH 5.7 aeration rate 0.8 vvm, agitation speed 80 rpm</td><td>Biomass 20.5 gL-1Extracellular polysaccharides 4.25 gL-1</td><td>[105]</td></tr><tr><td>Trametes versicolor</td><td>35 gL-1glucose 0.5 gL-1yeast extract, 5.0 gL-1pepton 1.0 gL-1KH2PO4 0.5 gL-1MgSO4 x 7 H2O 0,05 gL-1tiamin</td><td>10 L STR inoculum 10% (v/v), T 28 °C initial pH 5.7 aeration rate 1.0 vvm, agitation speed 400 rpm</td><td>Biomass 18,5 gL-1Extracellular polysaccharide 3.8 gL-1</td><td>[106]</td></tr><tr><td>Cordyceps militaris</td><td>80 gL-1glucose, 10 gL-1yeast extract, 0.5 gL-1MgSO4·7H2O 0.5 gL-1KH2PO4</td><td>5 L STR bioreactor T 24 °C, pH 5.8, agitation 200 rpm, aeration 1.5 vvm,</td><td>Biomass 40.60 gL-1Extracellular polysaccharide 6.74 gL-1</td><td>[107]</td></tr></table>
### d) Submerged Cultivation of the other Species in Bioreactors
Besides the cultures present in this chapter, there are also some other medicinal mushrooms that were submerge cultivated in mostly in lab scale bioreactors.
Inonotus levis and Agaricus nevoi cultivation was proceed in a $10 \mathrm{~L}$ stirred-tank bioreactor using substrate based on glucose and corn steep liquor at pH 5.5. Agitation speed gradually increased from 50 to 300 rpm and T $28^{\circ} \mathrm{C}$. I.levis developed very rapidly and after 5 days of cultivation the culture reached the stationary phase of growth with a high level of mycelia biomass of $16 \mathrm{~g} / \mathrm{L}$ at level of EPS concentration $4.2 \mathrm{~g} / \mathrm{L}$. A. nevoi was distinguished by a much lower growth rate and entered the stationary growth phase on day 10 with mushroom biomass $12 \mathrm{~g} / \mathrm{L}$ and EPS of $3.9 \mathrm{~g} / \mathrm{L}$ [102].
Submerged cultivation of Agaricus brasiliensis was studied in 1 L stirred tank reactor. Sucrose was found to be most effective for EPS production. Yeast extract was the best for EPS among the inorganic and organic nitrogen sources tested. The factorial experiment demonstrated that a temperature of $30^{\circ}\mathrm{C}$ and a pH of 6.1 were the best for the EPS production.
Glucose $10 \, \mathrm{g/L}$, yeast extract $3 \, \mathrm{g/L}$, $\mathrm{K}_2\mathrm{HPO}_4$ $0.6 \, \mathrm{g/L}$ and $\mathrm{MgSO}_4$ $0.3 \, \mathrm{g/L}$ [108].
Armillaria mellea was cultivated on glucose 40 g/L and yeast extract based substrate in 5 L stirred tank reactor at $22^{\circ}\mathrm{C}$; the two-stage aeration rate strategy $(1.2\rightarrow 0.6\mathrm{~v~m})$; 150 rpm, controlled pH 4.0, 6.65 g/L fungal biomass and 233.2 m g/L of extracellular polysaccharides with antioxidant properties were obtained [109].
Pleurotus pulmonarius was studied in submerged cultivation in a 2 L stirred-tank reactor. Substrate was composed by 20 g/L of brown sugar, 4 g/L rice bran, 4 g/L malt extract, and 4 g/L of yeast extract with initial pH of 5.5 Incubated at $28^{\circ}\mathrm{C}$ with agitation speed of 250 rpm and oxygen partial pressure of $30 - 40\%$. Maximum P. pulmonarius dry biomass production of 11.8 g/L was achieved after 3 days of cultivation [110].
Pleurotus saca was submerged cultivated on substrate consist of beer worth substrate (batch mode in 10-L stirred tank reactor. Agitation speed was 500 rpm and aeration $5 \mathrm{~L} \mathrm{~min}^{-1}$ and pH 6.2, up to $48.5 \mathrm{~g} / \mathrm{L}$ of dry biomass was obtained [111].
Pleurotus ostreatus was cultivated in a 20 L stirred tank bioreactor in a submerged process with enhanced glucan and dietary fibres content, using 57 g/L xylose and 37 g/L corn steep liquor. High yields 39.2 g/L of dry biomass was obtained [112,113].
Ganoderma lingzhi were studied in $5 \mathrm{~L}$ stirred bioreactor. The optimum conditions were an initial pH of 5.9, $20.0\%$ DO and $T 29^{\circ} \mathrm{C}$. These conditions resulted in a triterpene acids (TA) of $0.31 \mathrm{~g} / \mathrm{L}$. Furthermore this optimized conditions were then successfully scaled up
to a production scale of 200 L, and maximum TA production and productivity of 0.29 g/L and 0.05 g/L day $^{-1}$ were achieved. [114].
### e) Differences between Solid State and Submerged Cultivation
Main difference and benefits of solid state and submerged medicinal mushroom cultivation are presented in Table 3.
Table 3: Comparison of solid-state and submerged cultivation [96]
<table><tr><td>Solid state cultivation</td><td>Submerged cultivation</td></tr><tr><td>Some products can only be produced well under low moisture conditions. For other products, if the producing organisms require free water, solid state cultivation cannot be used.</td><td>A wide range of products can be produced, from a wide range of microorganisms and fungi. Many products are produced best under submerged cultivation.</td></tr><tr><td>The medium is relatively simple (eg. grain) and unrefined. It may contain all nutrients necessary for growth, or only require wetting with a mineral solution. Pretreatment can be as simple as cooking or grinding. However, the substrate composition and characteristics can be variable.</td><td>The medium often contains more highly processed ingredients and is therefore more expensive. Unprocessed ingredients may need processing to extract and solubilize the nutrients. With defined media good reproducibility is possible</td></tr><tr><td>The low water availability helps to select and protect against growth of contaminants.</td><td>The water activity is usually very high and many contaminants can grow well.</td></tr><tr><td>Media are concentrated and smaller bioreactors can be used, leading to higher volumetric productivities, even when growth rates and yields are lower.</td><td>Media are dilute and therefore occupy larger volumes, leading to lower volumetric productivities.</td></tr><tr><td>High substrate concentrations can enable high product concentrations.</td><td>High substrate concentrations can cause rheological problems. Substrate feeding systems may be required.</td></tr><tr><td>Aeration requires less power since pressures are lower. Gas transfer is easier since the particles have a large surface area.</td><td>High air pressures can be required. Gas transfer from the gas to liquid phase is slow and can be limiting.</td></tr><tr><td>Mixing within particles is not possible, and growth can be limited by the diffusion of nutrients.</td><td>Vigorous mixing can be used, and diffusion of nutrients is usually not limiting.</td></tr><tr><td>Ability to remove metabolic heat is restricted, leading to overheating problems.</td><td>High water content and more dilute nature makes temperature control easier.</td></tr><tr><td>Process control can be difficult due to difficulties in making on-line measurements, and in measuring biomass. The addition of substances during the process is difficult.</td><td>Many on-line sensors are available and more are being developed. Additions of substances can be made to control the process.</td></tr><tr><td>Downstream processing may be simpler since products are more concentrated. However, extracts can be contaminated with substrate components.</td><td>Downstream processing requires removal of large volumes of water, and is more expensive. However, with defined media, product purification may be easier.</td></tr><tr><td>Liquid waste is not produced.</td><td>Usually large volumes of liquid wastes are produced.</td></tr><tr><td>Growth kinetics and transport phenomena have received little attention and are poorly characterized.</td><td>Much kinetic and transport information is available in literature, which can guide reactor design and operation.</td></tr><tr><td>Research results and information from the solid-state cultivation can be scaled-up, or even transferred and applied in liquid-state cultivation.</td><td>In scaling up fungal submerged cultivation processes, various technical problems need to be solved, such as an increased broth viscosity and oxygen supply.</td></tr><tr><td>Solid-state cultivation of fungal mycelia is less labor intensive.</td><td>Submerged cultivation is more demanding and labor intensive.</td></tr></table>
## V. DOWN STREAM PROCESSING
Disruption of medicinal mushrooms by mechanical, chemical or enzymatic methods is greatly required for the efficient extraction of active compounds from them. In addition, ultrafine powder of medicinal mushroom by mechanical method can be used for functional food or dietary supplement. Other products used for pharmaceuticals are produced at the stages of extraction, fractionation and purification by varying techniques including microwave assisted extraction, membrane separation, adsorptive separation and chromatography. RecrySTALLIZATION, lyophilisation, drying and formulation are used as final product treatments [115].
Current disruption methods can be classified into mechanical, chemical and enzymatic methods in terms of their principles and characteristics. Mechanical methods are often preferred due to short residence time and lower operating costs [116]. The most common mechanical means for disruption are bead mill and homogenizer [117]. Another most frequently used sample disruption method is air jet milling which uses high velocity jets of gas to impart energy to particles for size reduction. The main features of air jet mills include [118].
Extraction is the first step to separate the desired products from the raw materials. Nearly $80 - 85\%$ of all medicinal mushroom products are extracted from their fruiting bodies while only $15\%$ are derived from mycelium culture [119].
Solvent is one of the most important parameters for a successful extraction. Selectivity, solubility, cost and safety should be considered in selection of solvents. Alcohols (ethanol and methanol) are universal solvents for the extraction of natural products although their low selectivity.
In recent years, advanced and greener extraction methods such as supercritical fluid extraction, pressurized liquid extraction, ultrasound assisted extraction, microwave assisted extraction, pulsed electric field extraction and enzyme assisted extraction have also been applied for extraction of natural products, and they offer some advantages such as lower organic solvent consumption, shorter extraction time and higher selectivity. In particular, supercritical fluid extraction gains increasing attention due to its higher efficiency and greener characteristics.
A brief summary of the various extraction methods used for medicinal mushroom products is shown in Table 4 [120].
Table 4: A brief summary of various extraction method used for medicinal mushroom product adapted from [120]
<table><tr><td>Applied Method</td><td>Solvent</td><td>Volume of consumed solvent</td><td>Temperature</td><td>Time</td><td>Pressure</td><td>Polarity of extracted products</td></tr><tr><td>Maceration</td><td>Various solvent</td><td>Large</td><td>Room temperature</td><td>Atmospheric</td><td>Long</td><td>Dependent on the solvent</td></tr><tr><td>Percolation</td><td>Various solvent</td><td>Large</td><td>Room temperature, sometimes under heat</td><td>Atmospheric</td><td>Long</td><td>Dependent on the solvent</td></tr><tr><td>Decoration</td><td>Water</td><td>None</td><td>Under heat</td><td>Atmospheric</td><td>Moderate</td><td>Polar compounds</td></tr><tr><td>Reflux extraction</td><td>Various solvent</td><td>Moderate</td><td>Under heat</td><td>Atmospheric</td><td>Moderate</td><td>Dependent on the solvent</td></tr><tr><td>Soxhlet extraction</td><td>Organic solvent</td><td>Moderate</td><td>Under heat</td><td>Atmospheric</td><td>Long</td><td>Dependent on the solvent</td></tr><tr><td>Pressurized liquid extraction</td><td>Organic solvent</td><td>Small</td><td>Under heat</td><td>High</td><td>Short</td><td>Dependent on the solvent</td></tr><tr><td>Supercritical fluid extraction</td><td>Supercritical fluid (usually CO2)</td><td>None or small</td><td>Near room temperature</td><td>High</td><td>Short</td><td>None polar or moderate polar</td></tr><tr><td>Ultrasound assisted extraction</td><td>Various solvent</td><td>Moderate</td><td>Room temperature or under heat</td><td>Atmospheric</td><td>Short</td><td>Compounds dependent on the solvent</td></tr><tr><td>Microwave assisted extraction</td><td>Various solvent</td><td>Moderate</td><td>Room temperature or under heat</td><td>Atmospheric</td><td>Short</td><td>Dependent on the solvent</td></tr><tr><td>Pulsed electric field extraction</td><td>Various solvent</td><td>Moderate</td><td>Room temperature or under heat</td><td>Atmospheric</td><td>Short</td><td>Dependent on the solvent</td></tr><tr><td>Enzyme assisted extraction</td><td>Various solvent</td><td>Moderate</td><td>Room temperature or under heat</td><td>Atmospheric</td><td>Moderate</td><td>Dependent on the solvent</td></tr><tr><td>Distillation</td><td>Water</td><td>Moderate</td><td>Under heat</td><td>Atmospheric</td><td>Long</td><td>Essential oil</td></tr></table>
The obtained fraction containing the desired products via fractionation steps described above may consist of several compounds with highly similar chemical and physical properties. Sometimes these compounds are analogues or even isomers as already stated, presenting a huge challenge for the purification. Column chromatography packed with separation media with small particle size is the most prevailing technique to implement the task, because it can offer very high number of theoretical plates and thus high resolution. However, in industrial scale, the particle size of the packed material is much larger because no pump can generate the pressure in manufacturing scale as that in a HPLC system [120].
## VI. CONCLUSION
Various pharmaceutically active substances from medicinal mushrooms represent effective potential in human life. Great demand for medicinal fungi biomass production could be fulfilled using various cultivation techniques. Medicinal fungi biomass in a present time is mostly covered by farming. Farming cultivation represents cheap but long time consuming technology. Using cultivation on a wooden locks a few year coughing time is need. Cultivation on sawdust substrates in trays or beds and in sterilised plastic bags or in bottles represents an advance and much faster production of fungal fruit bodies than conventional farming.
Solid state a few week time cultivation is a comprehensive, well controlled technology that enables much faster medium scale technology for medicinal mushroom mycelia production. In this technology various secondary waste from wood and agriculture industry are successfully used. No fungal fruit bodies are produced. Final product delignolized, wooden material is overgrown by medicinal fungi biomass in two to four weeks, enreach with proteins and various pharmaceutically products need to be dryed and pulverized and in such form it could be directly used in a veterinary need. Solid state cultivation of medicinal mushroom biomass is cheap in non time consuming technology perfectly suitable in veterinary use.
Submerged cultivation of medicinal fungi biomass represents fast and comprehensive technology method. Fungal biomass is in its final state from 10 to 28 days. The main benefit of cultivation of medicinal mushroom biomass in bioreactors is in using of higher sterility, comprehensive technology and bioprocess control, for large scale production of various pharmaceutically active compounds as are fungal
polysaccharides, terpenoides or proteinoglucans in much shorter cultivation time. Reports on pharmacological activity of extracts, partly purified preparations and isolated compounds from biomass of G.lucidum, G.frondosa, T.versicolor, H.erinaceus and C.militaris and the other reported species in laboratory and pilot scale, are very convincing. Production of medicinal fungi polysaccharides enhanced by fed-batch or two-stage cultivation strategy was found very useful for improving the production. Some of present results of lab scale research of various medicinal fungi are already transferred to pilot and lower industrial scale and they represents suitable starting platform for development of medicinal fungi biomass in large scale pharmaceutical industrial production. Comparing the economy of the same product production solid state processing is $30\%$ less than those of the one produced in submerged cultivation.
Isolation of medicinal fungi pharmaceutically compounds in all three technologies is is based on precipitation with hot water and ethanol. Crude extracts often show equal or stronger pharmacological activity as purified compounds, which suggests potential synergistic effects of several naturally occurring compounds. In the future, more precise capture (or enrichment) and separation techniques such as affinity separation, and more integrated bioprocesses for medicinal mushroom products should be developed which would enable a higher product yield and better process performance.
As pointed out submerged cultivation of medicinal mushrooms has significant large scale industrial potential, but its success on a commercial scale depends on existing field-cultivation technology as well as pharmaceutical market economy. Production of various medicinal mushrooms products, terpenoids, polysaccharides and proteinoglucans represents great business in Asiatic space where it is traditional. Great demand of this active ingredients definitively need to include new and fast large scale industrial production technologies as are solid state and submerged cultivations. In opposite, Western Pharmaceutical Industry has no tradition in natural isolates from herbal and fungal sources. Unfortunately, it is too convenient and much baseded on classical pharmaco-chemical biosynthesis including its all side effects.
However, from the viewpoint of Western Science, pharmaceutical legislation and regulations might be one of the main obstacles hindering the introduction of medicinal mushrooms products as registered pharmaceuticals. In any case, further


research is needed to fully understand all mechanisms of pharmaceutical effects of medicinal mushrooms products and to identify their potential side effects.
### ACKNOWLEDGEMENTS
Special gratitude to Univ.dipl.ing. Branko Škrinjar, Head of FKKT Library, University of Ljubljana, for his great assistance to the Literature support and doc. dr. Miloš Vettori, Biotechnical Faculty, University of Ljubljana, dr.Andrej Gregori, MycoMedica d.o.o. for their excellent photos.
Generating HTML Viewer...
References
116 Cites in Article
S Wasser (2010). Medicinal mushroom science: history, current status, future trends, and unsolved problems.
Shu-Ting Chang,Solomon Wasser (2012). The Role of Culinary-Medicinal Mushrooms on Human Welfare with a Pyramid Model for Human Health.
Shu Chang,Solomon Wasser (2018). Current and Future Research Trends in Agricultural and Biomedical Applications of Medicinal Mushrooms and Mushroom Products (Review).
Bojana Boh (2013). Ganoderma lucidum: A Potential for Biotechnological Production of Anti-Cancer and Immunomodulatory Drugs.
I Zmitrovich,N Belova,M Balandaykin,M Bondartseva,S Wasser (2019). Cancer without pharmacological illusions and a niche for mycotherapy (Review).
S Griffiths (2016). Unknown Title.
S Chang,J Buswell,Ganoderma Lucidum (curt,) Fr,Karst (1999). Aphyllophoromycetideae) -A mushrooming medicinal mushroom.
S Baldauf,A Roger,I Wenk-Siefert,W Doolittle (2000). A kingdom-level phylogeny of eukaryotes based on combined protein data.
Laura Katz,Jessica Grant,Laura Parfrey,J Burleigh (2012). Turning the Crown Upside Down: Gene Tree Parsimony Roots the Eukaryotic Tree of Life.
S Chang,J Buswell (2023). Medicinal Mushrooms -Past, Present and Future, Advances in Biochemial Engineering and Biotechnology.
B Boh Podgornik,M Berovic (2011). Ganoderma lucidum Production of Pharmaceuticals.
Jiezhong Chen,Robert Seviour (2007). Medicinal importance of fungal β-(1→3), (1→6)-glucans.
M Moradali,H Mostafavi,S Ghods,G Hedjaroude (2007). Immunomodulating and anticancer agents in the realm of macromycetes fungi (macrofungi).
S Wasser (2002). Medicinal mushrooms as a source of antitumor and immunomodulating polysaccharides.
S Jong,J Birmingham (1992). Medicinal Benefits of the Mushroom Ganoderma.
K Perumal (2009). Indigenous technology on organic cultivation of Reishi (Ganoderma lucidum) in India.
K Tasaka,M Akagi,K Miyoshi,M Mio,T Makino (1988). Anti-allergic constituents in the culture medium ofGanoderma lucidum. (I) Inhibitory effect of oleic acid on histamine release.
K Kino,T Sone,J Watanabe,A Yamashita,H Tsuboi,H Miyajima,H Tsunoo (1991). Immunomodulator, LZ-8, prevents antibodyproduction in mice.
Z Ji,Q Tang,J Zhang,Y Yang,Y Liu,C Tang,Y Pan (2009). Immunomodulation of bone marrow macrophages by GLIS, a proteoglycan fraction from Ganoderma lucidum.
Libin Ye,Xiaolin Zheng,Jingsong Zhang,Qingqiu Tang,Yan Yang,Xiangyang Wang,Jianrong Li,Yanfang Liu,Yingjie Pan (2011). Biochemical characterization of a proteoglycan complex from an edible mushroom Ganoderma lucidum fruiting bodies and its immunoregulatory activity.
J Yuen,M Gohel,C Ng (2011). The differential immunological activities of Ganoderma lucidum on human pre-cancerous uroepithelial cells.
J Zhang,Q Tang,C Zhou,W Jia,L Da Silva,D Nguyen,W Reutter,H Fan (2009). Mice and inhibition Medicinal Mushroom Biotechnology Global Journal of Science Frontier Research ( B ) XXIV Issue I Version I Year 2024 of tumour growth by GLIS, a bioactive proteoglycan from Ganoderma lucidum.
Zijing Jiang,Tiantian Jin,Feng Gao,Jianwen Liu,Jianjiang Zhong,Heng Zhao (2011). Effects of Ganoderic acid Me on inhibiting multidrug resistance and inducing apoptosis in multidrug resistant colon cancer cells.
I Jakopovich (2009). Medicinal mushroom preparations against lung cancer.
Seung Lee,Hee Rhee (1990). Cardiovascular effects of mycelium extract of Ganoderma lucidum: Inhibition of sympathetic outflow as a mechanism of its hypotensive action..
S El-Mekkawy,M Meselhy,N Nakamura,Y Tezuka,M Hattori,N Kakiuchi,K Shimotohno,T Kawahata,T Otake (1998). Anti-HIV-1 and anti-HIV-1 protease substances from Ganoderma lucidum.
H Pan,Y Wang,Y Xie,X Li,S Li,Z Zhi,L Chong,T Peng (2009). The 10th International Medicinal Mushrooms Conference (September 19–22, 2019, Nantong, China).
S Wasser,A Weiss,Medicinal Mushrooms -G,Lucidum (1997). Reishi mushroom.
Z Lin,B Yang,Ganoderma (2019). Unknown Title.
Z Lin (2005). Cellular and molecular mechanisms of immuno-modulation by Ganoderma lucidum.
R Russell,M Paterson (2006). Ganoderma lucidum -A therapeutic fungal biofactory.
Bojana Boh,Marin Berovic,Jingsong Zhang,Lin Zhi-Bin (2007). Ganoderma lucidum and its pharmaceutically active compounds.
C Lai,J Hung,H Lin,A Yu,S Chen,Y Tsai (2010). Immunomodulatory and adjuvant activities of a polysaccharide extract of Ganoderma lucidum in vivo and in vitro.
Jingsong Zhang,Qingjiu Tang,Changyan Zhou,Wei Jia,Luis Da Silva,Long Nguyen,Werner Reutter,Hua Fan (2010). GLIS, a bioactive proteoglycan fraction from Ganoderma lucidum, displays anti-tumour activity by increasing both humoral and cellular immune response.
M Novak,V Vetvicka (2007). <i>β</i>-Glucans, History, and the Present: Immunomodulatory Aspects and Mechanisms of Action.
G Chan,W Chan,D Sze (2009). The effect of βglucan on human immune and cancer cells.
J Zhe,Q Tang,J Zhang,Y Yang,W Jia,Y Pan (2007). Immunomodulation of RAW264.7 macrophages by GLIS, a proteopolysaccharide from Ganoderma lucidum.
Hung-Sen Chen,Yow-Fu Tsai,Steven Lin,Chia-Ching Lin,Kay-Hooi Khoo,Chun-Hung Lin,Chi-Huey Wong (2004). Studies on the immuno-modulating and anti-tumor activities of Ganoderma lucidum (Reishi) polysaccharides.
M Berovic,J Habijanic,I Zore,B Wraber,D Hodzar,B Boh (2003). Submerged cultivation of Ganoderma lucidum biomass and immunostimulatory effects of fungal polysaccharides.
Julie Frank,Rijin Xiao,Shanggong Yu,Matthew Ferguson,Leah Hennings,Pippa Simpson,Martin Ronis,Nianbai Fang,Thomas Badger,Frank Simmen (2006). Effect of Shiitake mushroom dose on colon tumorigenesis in azoxymethane-treated male Sprague-Dawley rats.
H Nanba,K Kubo (1997). Effect of Maitake D-fraction on cancer prevention.
N Harada,N Kodama,H Nanba (2003). Relationship between dendritic cells and the D-fraction-induced Th-1 dominant response in BALB/c tumor-bearing mice.
Noriko Kodama,Yukihito Murata,Akihiro Asakawa,Akio Inui,Masahiko Hayashi,Norio Sakai,Hiroaki Nanba (2005). Maitake D-Fraction enhances antitumor effects and reduces immunosuppression by mitomycin-C in tumor-bearing mice.
Hong Lin,Yu-Hong She,Barrie Cassileth,Frank Sirotnak,Susanna Cunningham Rundles (2004). Maitake beta-glucan MD-fraction enhances bone marrow colony formation and reduces doxorubicin toxicity in vitro.
R Barry,G Govindan,A Miller (2003). Annex 6.A2. Agricultural patent applications filed under the Patent Co-operation Treaty (PCT) in agriculture, 2006-11.
N Kodama,M Yamada,H Nanba (2001). Addition of Maitake D-fraction reduces the effective dosage of vancomycin for the treatment of Listeria-infected mice.
A Mlinarič,M Kač,F Pohleven (2005). Screening of selected wood-damaging fungi for the HIV-1 reverse Medicinal Mushroom Biotechnology Global Journal of Science Frontier Research ( B ) XXIV Issue I Version I Year 2024 transcriptase inhibitors.
V Manohar,N Talpur,B Echard,S Lieberman,H Preuss (2002). Effects of a water‐soluble extract of maitake mushroom on circulating glucose/insulin concentrations in KK mice.
Kyungah Jung,In-Ho Kim,Daeseok Han (2004). Effect of medicinal plant extracts on forced swimming capacity in mice.
J Bae,G Sim,D Lee,B Lee,H Pyo,T Choe,J Yun (2005). Production of exopolysaccharide from mycelial culture of Grifola frondosa and its inhibitory effect on matrix metalloproteinase -1 expression in UV-irradiated human dermal fibroblasts.
Q Shen (2001). Molecular phylogenetic analysis of Grifola frondosa (Maitake) and related species and the influence of selected nutrient supplements on mushroom yield.
T Mizuno (1995). Bioactive biomolecules of mushrooms: food function and medicinal effect of mushroom fungi.
Takashi Mizuno (1999). Bioactive Substances in Hericium erinaceus (Bull.: Fr.) Pers. (Yamabushitake), and Its Medicinal Utilization.
E Kolotushkina,M Moldavan,K Voronin,G Skibo (2003). The influence of Hericium erinaseus extract on myelination process in vitro.
Hirokazu Kawagishi,Atsushi Shimada,Ryoko Shirai,Kenji Okamoto,Fumihiro Ojima,Hideki Sakamoto,Yukio Ishiguro,Shoei Furukawa (1994). Erinacines A, B and C, strong stimulators of nerve growth factor (NGF)-synthesis, from the mycelia of Hericium erinaceum.
Hirokazu Kawagishi,,Atsushi Simada,,Kazue Shizuki,,Fumihiro Ojima,,Hironobu Mori,,Kenji Okamoto,,Hideki Sakamoto,,Shoei Furukawa, (1996). ERINACINE D, A STIMULATOR OF NGF-SYNTHESIS, FROM THE MYCELIA OF HERICIUM ERINACEUM.
Hirokazu Kawagishi,Atsushi Shimada,Satoshi Hosokawa,Hironobu Mori,Hideki Sakamoto,Yukio Ishiguro,Shiichi Sakemi,Jon Bordner,Nakao Kojima,Shoei Furukawa (1996). Erinacines E, F, and G, stimulators of nerve growth factor (NGF)-synthesis, from the mycelia of Hericium erinaceum.
H Kawagishi,A Masui,S Tukuyama,T Nakamura,Erinacines (2006). K from the mycelia of Hericium erinaceum.
H Kenmoku,T Sassa,N Kato (2000). Isolation of Erinacine P, a new parental metabolite of cyathanexylosides, from Hericium erinaceum and its biomimetic conversion into erinacines A and B.
Hiromichi Kenmoku,Takashi Shimai,Tomonobu Toyomasu,Nobuo Kato,Takeshi Sassa (2002). Erinacine Q, a New Erinacine from<i>Hericium erinaceum</i>, and its Biosynthetic Route to Erinacine C in the Basidiomycete.
Fangyuan Cui,Xia Gao,Jianjun Zhang,Min Liu,Chen Zhang,Nuo Xu,Huajie Zhao,Lin Lin,Meng Zhou,Le Jia (2016). Protective Effects of Extracellular and Intracellular Polysaccharides on Hepatotoxicity by Hericium erinaceus SG-02.
Sung Kim,Eunpyo Moon,Seok Nam,Mendel Friedman (2012). Hericium erinaceus Mushroom Extracts Protect Infected Mice against Salmonella Typhimurium-Induced Liver Damage and Mortality by Stimulation of Innate Immune Cells.
Zuofa Zhang,Guoying Lv,Huijuan Pan,Ashok Pandey,Weiqiang He,Leifa Fan (2012). Antioxidant and hepatoprotective potential of endo-polysaccharides from Hericium erinaceus grown on tofu whey.
K Chu,S Ho,A Chow (2002). Coriolus versicolor: A Medicinal Mushroom with Promising Immunotherapeutic Values.
Huaiqian Dou,Yajing Chang,Lijuan Zhang (2019). Coriolus versicolor polysaccharopeptide as an immunotherapeutic in China.
K Cheng,P Leung (2008). General review of polysaccharopeptides (PSP) from C. versicolor: Pharmacological and clinical studies.
T Ng (1998). A review of research on the protein-bound polysaccharide (polysaccharopeptide, PSP) from the mushroom Coriolus versicolor (basidiomycetes: Polyporaceae).
Takako Hattori,Nobukazu Komatsu,Shigeki Shichijo,Kyogo Itoh (2004). Protein-bound polysaccharide K induced apoptosis of the human Burkitt lymphoma cell line, Namalwa.
C Ho,C Lau,C Kim (2004). Differnetial effect of Trametes versicolor (Yunzhi) extract on cytokine production by murine lymphocytes in vitro.
C Ho,C Kim,K Leung (2006). Trametes versicolor (Yunzhi) extract attenuates growth of human leukemia xenografts and induces apoptosis through the mitochondrial pathway.
L Harhaji,S Mijatović,D Maksimović-Ivanić,I Stojanović,M Momčilović,V Maksimović,S Tufegdžić,Ž Marjanović,M Mostarica-Stojković,Ž Vučinić,S Stošić-Grujičić (2008). Anti-tumor effect of Coriolus versicolor methanol extract against mouse B16 melanoma cells: In vitro and in vivo study.
B Sevastere,I Prodan,R Manalachioae (2010). Antitumor Effect of Trametes versicolor Queil Medicinal Mushroom Biotechnology Global Journal of Science Frontier Research ( B ) XXIV Issue I Version I Year 2024 Alcoholic Extract.
P Kidd (2000). The use of mushroom glucans and proteoglycans in cancer treatment.
Jian Cui,Yusuf Chisti (2003). Polysaccharopeptides of Coriolus versicolor: physiological activity, uses, and production.
Lisa Price,Cynthia Wenner,Daniel Sloper,Joel Slaton,Jeffrey Novack (2010). Role for toll-like receptor 4 in TNF-alpha secretion by murine macrophages in response to polysaccharide Krestin, a Trametes versicolor mushroom extract.
Z Wang,X Peng,K Lee,J Tang,P Cheung,J Wu (2011). Structural characterisation and immunomodulatory property of an acidic polysaccharide from mycelial culture of Cordyceps sinensis fungus Cs-HK1.
Xiaoxia Zhou,Liping Luo,Waike Dressel,Gulibahaer Shadier,Doreen Krumbiegel,Peter Schmidtke,Fred Zepp,Claudius Meyer (2008). Cordycepin is an Immunoregulatory Active Ingredient of <i>Cordyceps sinensis</i>.
B Lee,J Park,J Park,H Shin,S Kwon,M Yeom,B Sur,S Kim,M Kim,H Lee,S Yoon,D Hahm (2011). Cordyceps militaris improves neurite outgrowth in neuro2A cells and reverses memory impairment in rats.
Ju-Hyon Lee,Soon-Min Hong,Jun-Yong Yun,Hoon Myoung,Myung-Jin Kim (2011). Anti-Cancer Effects of Cordycepin on Oral Squamous Cell Carcinoma Proliferation and Apoptosis in Vitro.
S Patel,A Goyal (2012). Recent developments in mushrooms as anti-cancer therapeutics: a review.
Kai Yue,Meng Ye,Zuji Zhou,Wen Sun,Xiao Lin (2013). The genus <i>Cordyceps</i>: a chemical and pharmacological review.
K Overgaard (1964). The inhibition of 5-phosphoribosyl-1-pyrophosphate formation by Cordycepin triphosphate in extracts of Ehrlich ascites tumor cells.
Fritz Rottman,Armand Guarino (1964). The inhibition of phosphoribosyl-pyrophosphate amidotransferase activity by cordycepin monophosphate.
S Holbein,A Wengi,L Decourty,F Freimoser,A Jacquier,I Dichtlrna (2009). Cordycepin interferes with 3′ end formation in yeast independently of its potential to terminate RNA chain elongation.
Y Wong,A Moon,R Duffin,A Barthet-Barateig,H Meijer,M Clemens,C De Moor (2010). Cordycepin inhibits protein synthesis and cell adhesion through effects on signal transduction.
X Chen,G Wu,Z Huang (2013). Structural analysis and antioxidant activities of polysaccharides from cultured Cordyceps militaris.
Jia Guan,Jing Zhao,Kun Feng,De-Jun Hu,Shao-Ping Li (2011). Comparison and characterization of polysaccharides from natural and cultured Cordyceps using saccharide mapping.
R Russell,M Paterson (2008). Cordyceps -A traditional Chinese medicine and another fungal therapeutic biofactory?.
K Cunningham,William Manson,F Spring,S Hutchinson (1950). Cordycepin, a Metabolic Product isolated from Cultures of Cordyceps militaris (Linn.) Link..
Shonkor Das,Mina Masuda,Masanori Hatashita,Akihiko Sakurai,Mikio Sakakibara (2010). Optimization of culture medium for cordycepin production using Cordyceps militaris mutant obtained by ion beam irradiation.
S Das,M Masuda,A Sakurai,M Sakakibara (2010). Medicinal uses of the mushroom Cordyceps militaris: current state and prospects.
B Boh Podgornik,M Berovic (2012). Cultivation of Ganoderma lucidum and Grifola frondosa and production of their pharmaceutical active compounds.
M Nikšić,B Boh Podgornik,M Berovic,Farming,Mushrooms (2023). Advances in Biochemial Engineering and Biotechnology.
Daniel Royse (1996). Speciality Mushrooms and Their Cultivation.
D Mitchell,M Berovic (1998). Solid state cultivation.
A Gregori,N Kretschmer,S Wagner,H Boechzelt,D Klinar,R Bauer,F Pohleven (2012). Influence of Olive Oil Press Cakes on Shiitake Culinary-Medicinal Mushroom, Lentinus edodes (Berk.) Singer (Higher Basidiomycetes) Fruiting Bodies Production and Effect of their Crude Polysaccharides on CCRF-CEM Cell Proliferation.
Sidharth Arora,Richa Rani,Sanjoy Ghosh (2018). Bioreactors in solid state fermentation technology: Design, applications and engineering aspects.
Hiroshi Hikino,Chohachi Konno,Yoshiaki Mirin,Teruaki Hayashi (1985). Isolation and Hypoglycemic Activity of Ganoderans A and B, Glycans of<i>Ganoderma lucidum</i>Fruit Bodies.
M Berovic,M Popovi Ć (2018). Submerged cultivation of Ganoderma lucidum biomass in stirred tank reactor.
V Elisashvili (2012). Submerged Cultivation of Medicinal Mushrooms: Bioprocesses and Products (Review).
F Cui,Y Li,Z Xu,H Xu,K Sun,W Tao (2006). Optimization of the medium composition for production of mycelial biomass and exo-polymer by Grifola frondosa GF9801 using response surface methodology.
I Zore,M Berovic,B Boh Podgornik,D Hodzar,F Pohleven (1998). Procedure for preparation of inoculum for growing of fungus Ganoderma lucidum by submersion cultivation.
D Huang,F Cui,Y Li,Z Zhang,J Zhao,X Han,X Xiao,J Qian,Q Wu,G Guan (2007). Nutritional Requirements for the Mycelial Biomass and Exopolymer Production by Hericium erinaceus CZ-2.
J Smole (2009). Optimization of culture conditions and medium components for the production of mycelial biomass and exo-polysaccharides with Cordyceps militaris in liquid culture biotechnology.
L Fana,A Soccol,A Pandey,R Soccol (2007). Effect of nutritional and environmental conditions on the production of exo-polysaccharide of Agaricus brasiliensis by submerged cultivation and its antitumor activity.
M Lung,C Hsieh (2011). Production of liquid spawn of an edible grey oyster mushroom, Pleurotus pulmonarius (Fr.) Quél by submerged cultivation and sporophore yield on rubber wood sawdust.
M Berovič,M Knežević (1995). Influence of tetracycline on the submerge Pleurotus saca cultivation.
Lefki-Maria Papaspyridi,Petros Katapodis,Zacharoula Gonou-Zagou,Evangelia Kapsanaki-Gotsi,Paul Christakopoulos (2010). Optimization of biomass production with enhanced glucan and dietary fibres content by Pleurotus ostreatus ATHUM 4438 under submerged culture.
L Papaspyridi,N Aligiannis,P Christakopoulos,A Skaltsounis,N Fokialakis (2011). Production of bioactive metabolites with pharmaceutical and nutraceutical interest by submerged cultivation of Pleurotus ostreatus in a batch stirred tank bioreactor.
Xiao-Ling Wang,Zhong-Yang Ding,Yan Zhao,Gao-Qiang Liu,Guo-Ying Zhou (2017). Efficient Accumulation and In Vitro Antitumor Activities of Triterpene Acids from Submerged Batch-Cultured Lingzhi or Reishi Medicinal Mushroom, Ganoderma lucidum (Agaricomycetes).
Hui Shi,Min Zhang,Sakamon Devahastin (2020). New Development of Efficient Processing Techniques on Typical Medicinal Fungi: A Review.
R Hatti-Kaul,B Mattiasson (1995). J. Process-scale disruption of microorganisms.
Y Xiao,A Qiang,W Lian,F Yiang,J Sun,S Guo (2009). Application of superfine pulverization technology in biomaterial industry.
Ulrike Lindequist,Timo Niedermeyer,Wolf-Dieter Jülich (2005). The Pharmacological Potential of Mushrooms.
H Luo,Y Li (2023). Down Stream Processing of Medicinal Mushrooms Products, Advances in Biochemial Engineering and Biotechnology.
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Since most of medicinal mushrooms are rare in nature production of fungal fruiting bodies using artificial cultivation in a form of farming has been intensively established during the last 40 years. Solid state cultivation of various medicinal mushroom mycelia in various types of bioreactors, suitable for veterinary use, appears slightly in last few decades. Developing submerged technologies, using stirred tank and air lift bioreactors, are the most promising technologies for fast and large cultivation of medicinal pharmaceutically active products for human need. This potential initiates the development of new drugs and some of the most attractive over the counter human and veterinary remedies. This article is an overview of the engineering achievements in comprehensive medicinal mushroom mycelia cultivation.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.