## I. INTRODUCTION
Intracerebral hemorrhage (ICH) accounts for approximately $20\%$ of all strokes, and outcomes are often catastrophic, resulting in 30-day mortality up to $40\%^{1}$ and 6-month functional independence of only $20\%$. In addition to direct tissue damage at the time of the initial hemorrhagic event, there is profound secondary injury as the body responds to the presence of the hematoma, including cytotoxic and excitotoxic factors, oxidative stress, ferroptosis, and inflammatory pathways. While there has been some preclinical success in identifying therapeutic targets for secondary injury, no clinical studies have demonstrated success with any trialed therapies to date; in part due to lack of preclinical models that reliably recapitulate the comorbidities seen in patients with ICH.
Multiple preclinical ICH models have been developed over time, including cortical vessel avulsion, $^{8}$ microballoon, $^{9}$ bacterial collagenase, $^{10}$ and several variations of an autologous blood injection, $^{11-14}$ each with distinct advantages and shortcomings. A comparative study of the cortical vessel avulsion model, the bacterial collagenase injection model, and an autologous blood injection model showed that while the models demonstrate similar temporal patterns of injury, differences in cell death, inflammatory cell infiltration, and microglial reaction showed autologous blood injection to most closely mimic spontaneous human ICH. $^{15}$ Cortical vessel avulsion demonstrated a mixed pathology of ischemia and hemorrhage, whereas, bacterial collagenase elicited a more intense and prolonged inflammatory reaction. $^{15}$
Various modifications of a single-injection technique include blood injection via a permanently implanted needle into the basal ganglia, $^{11}$ bolus injection through a stereotaxically-placed intracerebral needle, $^{16}$ direct circuit from an intra-arterial femoral catheter to simulate hemorrhage induced at arterial pressure, $^{12}$ and utilization of a microinfusion pump to deliver a consistent volume of blood at a specified rate. $^{13}$ Each model aimed to improve reliability and to better simulate human pathology. However, techniques based on a single continuous injection often face issues with retrograde egress of blood along the needle tract, inconsistent and insufficient hematoma volume, subarachnoid and subdural extension of blood, and overall meager reproducibility. $^{17,18}$
To address the shortcomings of the single-injection technique, a double-injection modification was devised to allow for the formation of a "clot barrier" with time between injections, thus facilitating a more reproducible and stable hematoma during the second injection.[14] This model as initially described was performed with a stereotaxically directed catheter permanently placed into the basal ganglia with fresh autologous blood from a femoral artery cutdown and cannulation.[14] Femoral artery sampling does allow for fresh blood to be delivered, but requires an extended duration of anesthesia, which may confound ICH outcomes[20] and impairs post-surgical behavioral evaluation[21] critical to ICH studies.[22]
Our lab focuses on translational study of ICH with emphasis on biologic variables such as sex, age, and common ICH comorbidities like hypertension, insulin resistance and obesity. To accomplish these goals, we tested the modified double-injection autologous blood method against an optimized single-injection technique[24] in order to produce a reliable and cost-effective model in rats that best recapitulates human pathology in ICH. We present here our troubleshooting efforts and a granular, stepwise methodology that is not present in the current literature. This knowledge avoids a steep learning curve requiring significant time, expense, and animal expenditure.
## II. MATERIAL AND METHODS
All procedures were conducted in compliance with the NIH Guide for the Care and Use of Laboratory Animals and approved by the Institutional Animal Care and Use Committee at Wake Forest School of Medicine (protocol number A19-089). Matched Hanover Sprague-Dawley (SD) normotensive rats and hypertensive (mREN2)27 rats with cardiometabolic syndrome to recapitulate human disease $^{23}$ ages 12-55 weeks, both male and female, were used for the development of this model. Rats were randomly selected into the double-injection model ( $n = 10$ per group (male SD, female SD, male (mREN2)27, female (mREN2)27): $n = 40$ total) or the optimized single-injection model ( $n = 13$ per group (male SD, female SD, male (mREN2)27, female (mREN2)27): n=52 total). Ages were matched to account for the biologic variable of age.
Same sex litter mates were housed together in pairs in ventilated cages and maintained on a regular diurnal light cycle (12:12 light:dark) with ad libitum access to food and water. All animals were housed in the Animal Resources Program facilities at Wake Forest Baptist Health, accredited by AAALAC (The Association for Assessment and Accreditation of Laboratory Animal Care International). Animals underwent either the double injection method[14] or the optimized single-injection model described below. Following the procedure, animals underwent behavioral testing (Garcia score and Corner Turn) at 1 day and 3 days and were sacrificed at 72 hours. Brains were perfused with PBS, and serial cutting or hemoglobin assay was performed to evaluate hematoma location and volume.
### a) Stepwise Optimized Single-Injection Methodology Pre-Surgical Phase
- 1.1 Weigh the rat to calculate medication doses.
- 1.2 Transfer the rat to the anesthesia induction chamber and initiate the induction procedure with an oxygen flow rate equivalent to the volume of the chamber, approximately 1-2 L/min, and up to $3\%$ MAC vaporized isoflurane until the rat is completely immobilized and has lost its righting reflex.
- 1.3 Remove rat from the induction chamber and transfer to the stereotaxic apparatus. Position the rat prone on a heating pad at the base of the stereotaxic frame and place the rat's nose into a gas adaptor stereotaxic nose cone for maintenance anesthesia.
- 1.4 Reduce the anesthetic to $1.5\%$ MAC and ensure that the circuit is open to the stereotaxic nose cone adaptor. Rats weighing over 400g may require up to $2.5\%$ MAC maintenance anesthesia.
- 1.5 Apply ophthalmic lubricating ointment for corneal protection.
- 1.6 Position the rat with all midline structures in alignment; forepaws directed anteriorly, and hindpaws directed posteriorly. The stereotaxic nose cone should be adjusted to ensure the nose, head, and spine are in line and parallel to the floor without angulation (Figure 1). The stereotaxic machine may be used to ensure that the bregma and lambda are in the same plane. Gauze may be used to support the head as needed. Ensure the nose fits snugly into the chamber. This is critical as movement throughout the procedure due to inadequate maintenance anesthesia may displace the stereotaxic apparatus and lead to poorly formed hematomas.
- 1.7 Place a protective earpiece to prevent otic damage with the headband wire directed posteriorly.
- 1.8 Ensure the blink reflex is absent and affix the head in the stereotaxic apparatus such that it is centered
in the sagittal, coronal, and axial planes. Confirm that the head is secure before proceeding.
- 1.9 Apply heat to the tail to dilate the tail veins. This may be achieved by placing the tail under a nitrile or latex glove filled with water and heated in a water bath at $40^{\circ} \mathrm{C}$, not to exceed $43^{\circ} \mathrm{C}$.
- 1.10 Shave the surgical site with clippers and use tape to clear the site of hair. Disinfect the surgical site with povidone-iodine solution or equivalent and use appropriate drapes for aseptic technique. At this point we aseptically inject weight appropriate SQ buprenorphine $0.01 \, \text{mg/kg}$ for post-operative pain control.
- 1.11 Program the automated syringe pump (we use Harvard Apparatus Model 11 Elite Syringe Pump; Holliston, MA) with an injection rate of $10~\mu \mathrm{L} / \mathrm{min}$, injection volume of $100~\mu \mathrm{L}$, and syringe volume of $1 \mathrm{~mL}$.
- 1.12 Attach the shortest possible segment of PE20 polyethylene catheter tubing (PE20; I.D. 0.38 mm, O.D. 1.09 mm; Intramedic Clay Adams; Sparks, MD) to a 1 mL syringe with a blunt 26-gauge needle. Flush with non-heparinized saline and ensure an uninterrupted stream with minimal resistance to light finger pressure. If there is resistance, ensure that the needle is not occluded by pieces of plastic tubing, which can be avoided by pulling the needle back in the tubing. Using another 26-gauge needle, pre-dilate the distal end of the tubing to facilitate attachment to the Hamilton needle.
- 1.13 Prepare a second set of flushed and pre-dilated tubing to use as a back-up, in case clotting or occlusion of the tubing occurs during the injection.
#### Surgical Phase
- 1.14 Make a 2 cm midline scalp incision starting at the posterior aspect of the eyes and extending caudally to the lambda. Incise the periosteum laterally and locate the bregma, which often appears V-shaped (Figure 2).
- 1.15 Mobilize the periosteum laterally, until it cannot be mobilized any further. The burr hole will be at the junction pf the posterior-most aspect of the bregma intersecting where the periosteum remains attached at is lateral aspect, approximately 4.5mm in females and 5mm male rats. (Figure 2). Use a circular motion with a 1 mm burr drill (75,000 rpm) to make the smallest burr hole that will accommodate the needle. The depth of the skull is approximately 1-1.5 mm deep at this location. We then place sterile bone wax to seal the hole and prevent blood egress.
- 1.16 Affix a Hamilton needle (26 gauge with hub; 50.8 mm length; #7784-08; Reno, NV) to the stereotaxic frame and insert the stylet to maintain patency of the needle during stereotaxic placement. Set the needle
at a 20-degree angle along the coronal plane to the right of midline. Do not attach the needle too low on the stereotaxic frame as the needle will collide with the animal when positioning. With the stylet in place, carefully advance the needle through the center of the burr hole $6\mathrm{mm}$ in males and $5.5\mathrm{mm}$ in females deep to the surface of the skull. Note: The stereotaxic placement of the Hamilton needle into the striatum is completed prior to the collection of venous blood.
- 1.17 Using aseptic technique, disinfect the distal tail of the rat with alcohol and use sharp scissors to snip the distal-most $0.5 - 1 \mathrm{~mm}$ of the tail. Collect $0.2 \mathrm{~mL}$ of venous blood directly into a needleless $1 \mathrm{~mL}$ syringe. Note: after tail snip, it may be necessary for one lab technician to gently massage the tail from proximal to distal while another collects blood into the upright distal tip of the syringe. Avoid contacting the syringe with the epithelium of the tail. Remove any air bubbles that form in the syringe during blood collection, as air will induce clotting. A tail vein blood draw is another viable option (22-gauge needle or smaller), but we found this to prolong overall surgical time.
- 1.18 Connect the syringe containing fresh autologous blood to the flushed tubing with fluid-to-fluid interface, avoiding any air bubbles. Advance blood to the end of the tubing. Affix the syringe with freshly collected blood to the automated pump and P20 polyethylene catheter tubing connected to the 26 gauge Hamilton needle flushed with saline. Note: air bubbles promote hematogenous clotting and must be avoided.
- 1.19 Withdraw the Hamilton needle $0.5 \mathrm{~mm}$ to reach a final depth of $5.5 \mathrm{~mm}$ in males and $5 \mathrm{~mm}$ in females, creating a potential space for hematoma formation.
- 1.20 Remove the stylet from the stereotaxically placed Hamilton needle and connect the pre-dilated P20 tubing. A drop of saline may be needed to lubricate the needle and facilitate attaching the blood-filled tubing.
- 1.21 Perform a small bolus injection to get the blood to the end of the needle (one tap of the advance arrow button).
- 1.22 Begin the injection of $100~\mu \mathrm{L}$ at $10~\mu \mathrm{L} / \mathrm{min}$. \*\*\*Note: To ensure successful injection, note the starting volume of blood in the syringe and monitor for progression at a rate of $10~\mu \mathrm{L} / \mathrm{min}$. Inspect the site of the burr hole periodically to monitor for egress of blood. If impeded flow through the tubing is suspected, manually advance the microinfusion pump to dislodge any early clot formation within the tubing. If this maneuver is unsuccessful, discard the occluded tubing and connect the back-up tubing to the apparatus. Adjust the microinfusion pump settings to account for the lost blood volume and resume the injection. Note the new starting volume upon infusion resumption and continue to monitor for signs of injection failure. As this protocol carries a very low risk of intraventricular hematoma extrusion, we advise repeating portions of the injection ad libitum in the event of suspected injection failure.
- 1.23 After the injection has completed, allow 5 minutes for consolidation of the hematoma. Then, reinsert the stylet, pull back the needle $2\mathrm{mm}$ and wait 2 minutes, then we remove the Hamilton needle slowly over the course of 1 minute to prevent reflux of injected blood along the needle tract.
- 1.24 Reapproximate the scalp edges and suture the incision. When suturing is complete, turn off the anesthetic, remove the rat from the stereotaxic apparatus, and transfer the rat from the stereotaxic apparatus to a heated recovery chamber for observation.
- 1.25 Flush the Hamilton syringe needle with saline between each animal and clean with $70\%$ ethanol. It is recommended to use new tubing on every animal to prevent clotting. Do not reuse the syringe following injection, this creates a high risk of clotting.
Total procedural duration including total time under anesthesia is approximately 30-40 minutes.
## III. RESULTS
Of the 40 animals subjected to the double-injection method, successful hematoma formation was observed in only 19 animals, yielding a $47.5\%$ success rate. Of the 21 unsuccessful procedure attempts, 13 failed due to lack of hematoma/insufficient hematoma volume, 1 due to intraventricular extension, and 7 had visible blood egress along the needle tract. Due to the unacceptably high failure rate of the double-injection protocol, the modified single-injection method was adopted and implemented in a total of 52 animals. Successful hematoma formation was observed in 48 animals, yielding a $92.3\%$ success rate, which was significantly greater than that observed with the double-injection method $(x2 = 22.94, p < 0.00001)$. Of the 4 unsuccessful procedure attempts, 3 were deemed unsuccessful due to lack of hematoma/insufficient hematoma volume, 0 due to intraventricular extension, and 1 due to blood egress along the needle tract. Brain slices were imaged and characterized (Figure 3). Brain homogenization was performed on 14 animals with 7mRENs and 7 SDs. Hemispheric hemoglobin concentrations collected at 72 hours after ICH for SDs were 0.138 with a standard deviation of 0.018 and for mRENs were 0.168 with a standard deviation 0.022 (Figure 4). Behavioral testing demonstrated both Garcia scores at 1-day $(p <.001)$ and 3-day $(p=.023)$ and Corner Turn1-day $(p=.017)$ and 3-day $(p=.009)$ post-ICH were significantly different from baseline.
## IV. DISCUSSION
We developed a protocol to address shortcomings in existing ICH models: an autologous, single-injection rat model that reliably creates consistent basal ganglia ICH and allows for assessment of behavioral, physiologic, and histologic outcomes in a transgenic rat model to mimic human disease. This 100uL venous injection can be performed in 30 minutes per animal by new and emerging laboratories with relatively low costs. As with previous studies, we initially encountered complications necessitating method modification to achieve reproducibility. Through a discussion of these complications and our means of overcoming them, we hope to remove barriers encountered by new researchers in order to further the field of ICH research.
For the collection of autologous blood, we found that the tail snip was the most efficient method, minimizing the duration of anesthesia, reducing the amount of handling between collection and injection, and reliably performed by all lab members. Further, it can be repeated as needed during the procedure without additional tail cutting and does not impair post-ICH functional evaluation. Early application of heat through a glove filled with warmed water improves the ease of collection.
The presence of more than one lab member to allow tasks to be completed simultaneously, including collecting the blood, flushing and connecting the tubing, and programming the pump was an additional means to minimize the opportunity for clotting. Most importantly, we found that transitioning to a continuous injection model helped circumvent the above complication, as the injection is completed in a single continuous infusion and provided more reliable results.
To overcome the concern for reflux along the needle tract described in previous studies, we found that the burr hole must be no larger than $1.5\mathrm{mm}$, initial stereotaxic placement of the Hamilton needle to a depth of $6\mathrm{mm}$ (male) and subsequent retraction by $0.5\mathrm{mm}$ to reach a final depth of $5.5\mathrm{mm}$ created a potential space for hematoma formation, minimizing reflux along the needle.
One complication encountered, as noted above, was clot formation within the blood collection vial and the injection tubing. Minimizing the amount of handling and the amount of time elapsed between blood acquisition and blood injection is imperative to prevent clot formation, regardless of the method being used. Ensuring that there is no air in the syringe or tubing is essential to prevent clotting. It is also critical to visually monitor the blood delivery throughout the injection period. By noting the starting volume of the syringe and ensuring that it is advancing appropriately through the infusion period, we prevented episodes where the appropriate volume of blood had inadvertently not been injected. A simple bump forward with the pump if blood infused is slower than pump measurements usually dislodges any forming clot and infusion can continue uninterrupted. Having a spare flushed tubing prepared to replace any clotted tubing, as well as having extra saline for flushing the tubing, is critical. Again, as this model carries a low chance of hematoma rupture into the ventricle, and we have found that if in doubt that the full $100~\mu \mathrm{L}$ was infused into the brain parenchyma, it is better to re-inject that volume. Failure to do this was the most common reason for failed ICH creation at the beginning of our model development.
## V. CONCLUSIONS
This is the first paper that clearly details an autologous single-injection ICH model in rats with $>90\%$ rate of reproducibility. This basal ganglion ICH model results in accompanying neurobehavioral deficits on both the Garcia and corner turn testing. Importantly, we outline the pitfalls and proposed means of overcoming such complications in using this model. We hope for this report to facilitate other labs in reproducing the model effectively and efficiently and ultimately lower the expense of developing a translational animal model of ICH. This model is a form of pragmatic science that aims to recapitulate human disease, allowing for identifying possible biomarkers and potential targets for therapeutic interventions in a disease that causes significant morbidity and mortality.
### ACKNOWLEDGEMENTS
This work was supported through a pilot grant from the Wake Forest Cardiovascular Sciences Center, funded in part by the National Center for Advancing Translational Sciences (NCATS), National Institutes of Health, through Grant Award Number UL1TR001420. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH. Additional funds were provided by the Farley Hudson Foundation and the Hypertension & Vascular Research Center.
The authors report no conflicts of interest that could interfere with the integrity of this science. No part of this work has been previously published.
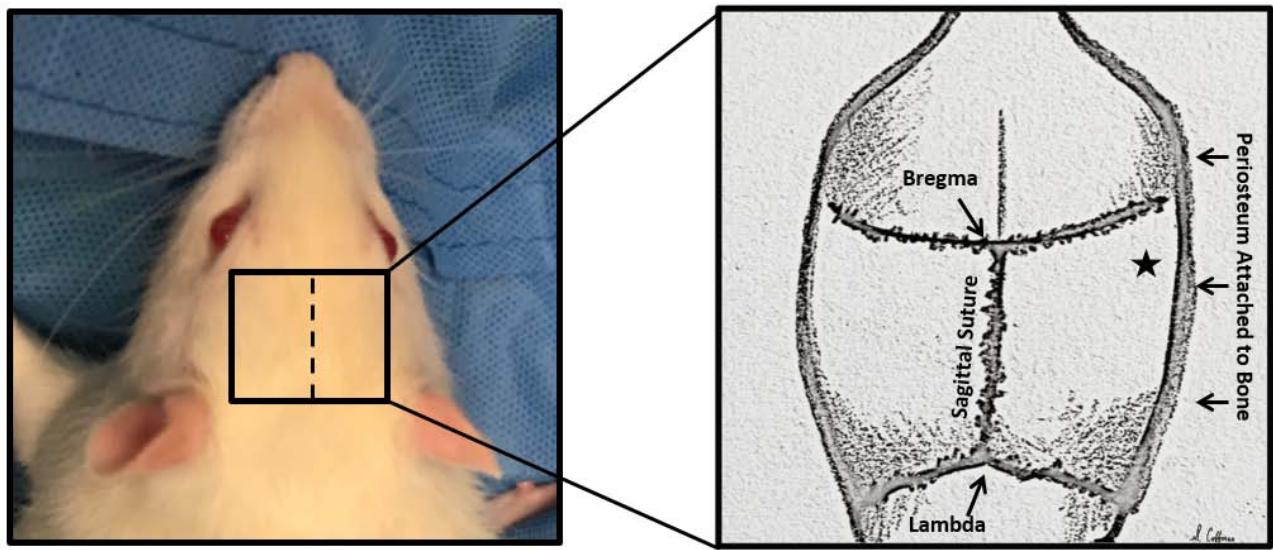
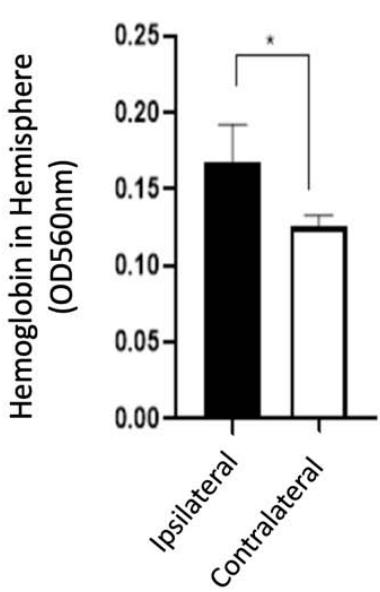 Figure 2: The incision (dashed line, left image) is approximately $2\mathrm{cm}$ long, midline on the scalp, from the back of the eyes to the lambda. After the incision is made, the periosteum is dissected and the bregma located, with the burr hole (star, right image) placed at the posterior-most point of the bregma and $5\mathrm{mm}$ to the right of the midline, where the periosteum remains attached to the skull. Original artwork by author Stephanie A. Coffman.
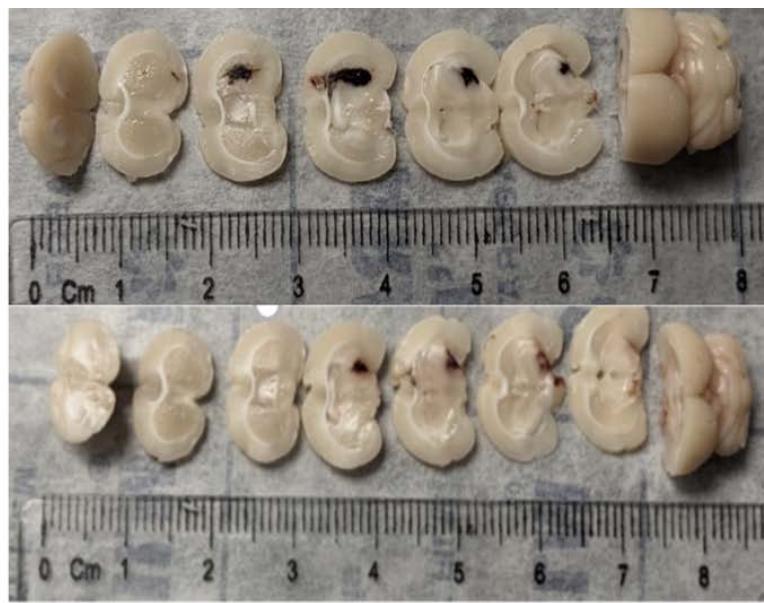 Figure 3: Graph (left) demonstrates mean total hemoglobin concentration in the ipsilateral hemisphere after induced autologous-ICH, demonstrating consistent hematoma volume and images (right) display serial brain sections demonstrating consistent basal ganglia hematoma.
Table 1: Comparative results of success rate and reasons for procedural failure in our experience of single-injection and double-injection methods for ICH creation.
<table><tr><td></td><td>Single-Injection Method*</td><td>Double-Injection Method</td><td>X2</td><td>p-value</td></tr><tr><td># successful attempts</td><td>48</td><td>19</td><td rowspan="2">22.94</td><td rowspan="2"><0.00001</td></tr><tr><td># unsuccessful attempts</td><td>4</td><td>21</td></tr><tr><td>Lack of hematoma or insufficient volume</td><td>3</td><td>13</td><td></td><td></td></tr><tr><td>Intraventricular extension</td><td>0</td><td>1</td><td></td><td></td></tr><tr><td>Blood egress along needle tract</td><td>1</td><td>7</td><td></td><td></td></tr></table>
Generating HTML Viewer...
References
25 Cites in Article
Charlotte Van Asch,Merel Luitse,Gabriël Rinkel,Ingeborg Van Der Tweel,Ale Algra,Catharina Klijn (2010). Incidence, case fatality, and functional outcome of intracerebral haemorrhage over time, according to age, sex, and ethnic origin: a systematic review and meta-analysis.
R Fogelholm,K Murros,A Rissanen,S Avikainen (2005). Long term survival after primary intracerebral haemorrhage: a retrospective population based study.
Huimin Zhu,Zhiqiang Wang,Jixu Yu,Xiuli Yang,Feng He,Zhenchuan Liu,Fengyuan Che,Xuemei Chen,Honglei Ren,Michael Hong,Jian Wang (2019). Role and mechanisms of cytokines in the secondary brain injury after intracerebral hemorrhage.
Praveen Belur,Jason Chang,Shuhan He,Benjamin Emanuel,William Mack (2013). Emerging experimental therapies for intracerebral hemorrhage: targeting mechanisms of secondary brain injury.
W Funnell,Dušica Maysinger,A Cuello (1990). Three-dimensional reconstruction and quantitative evaluation of devascularizing cortical lesions in the rat.
E Sinar,A Mendelow,D Graham,G Teasdale (1987). Experimental intracerebral hemorrhage: effects of a temporary mass lesion.
G Rosenberg,S Mun-Bryce,M Wesley,M Kornfeld (1990). Collagenase-induced intracerebral hemorrhage in rats..
Allan Ropper,Nicholas Zervas (1982). Cerebral blood flow after experimental basal ganglia hemorrhage.
R Bullock,A Mendelow,G Teasdale,D Graham (1984). Intracranial haemorrhage induced at arterial pressure in the rat: Part 1: Description of technique, ICP changes and neuropathological findings.
Guo-Yuan Yang,A Betz,Thomas Chenevert,James Brunberg,Julian Hoff (1994). Experimental intracerebral hemorrhage: relationship between brain edema, blood flow, and blood-brain barrier permeability in rats.
Wolfgang Deinsberger,Johannes Vogel,Wolgang Kuschinsky,Ludwig Michael Auer,Dieter-Karsten Böker (1996). Experimental intracerebral hemorrhage: Description of a double injection model in rats.
Mengzhou Xue,Marc Del Bigio (2003). Comparison of brain cell death and inflammatory reaction in three models of intracerebral hemorrhage in adult rats.
Anatol Manaenko,Hank Chen,John Zhang,Jiping Tang (2011). Comparison of Different Preclinical Models of Intracerebral Hemorrhage.
Qingyi Ma,Nikan Khatibi,Hank Chen,Jiping Tang,John Zhang (2011). History of Preclinical Models of Intracerebral Hemorrhage.
Dave Brodbelt (1997). Perioperative mortality in small animal anaesthesia.
Cassandra Wilkinson,Anna Kalisvaart,Tiffany Kung,D Maisey,Ana Klahr,Clayton Dickson,Frederick Colbourne (2020). The collagenase model of intracerebral hemorrhage in awake, freely moving animals: The effects of isoflurane.
Sharon Ohayon,Shaun Gruenbaum,Alan Artru,Matthew Boyko,Benjamin Gruenbaum,Michael Dubilet,Akiva Leibowitz,Yoram Shapira,Vivian Teichberg,Alexander Zlotnik (2012). Anatomical location of arterial and venous lines significantly affects motor performance in rats.
Ya Hua,Timothy Schallert,Richard Keep,Jimin Wu,Julian Hoff,Guohua Xi (2002). Behavioral Tests After Intracerebral Hemorrhage in the Rat.
M Langheinrich,M Lee,M Böhm,Y Pinto,D Ganten,M Paul (1996). The hypertensive Ren-2 transgenic rat TGR (mREN2) 27 in hypertension research Characteristics and functional aspects.
T O'lynnger,Y Hua,G Xi (2009). Blood Injection Intracerebral Hemorrhage Rat Model.
David Dorman,Brian Wong,Melanie Struve,R James,Krista Laperle,Marianne Marshall,Brad Bolon (1996). Development of a Mouse Whole-Body Exposure System from a Directed-Flow, Rat Nose-Only System.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Brock R. Yager, BS. 2026. \u201cModifications and Optimization of an Autologous Intracerebral Hemorrhage Rat Model\u201d. Global Journal of Medical Research - A: Neurology & Nervous System GJMR-A Volume 24 (GJMR Volume 24 Issue A1).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.