## I. BACKGROUND
The Musk melon (Cucumis melo L.) is an annual herbaceous plant species native to intertropical Africa and belongs to the family Cucurbitaceae. The fruit, which is fleshy and edible, is gaining productive prominence in Cameroon, especially the North West, West and, Southwest Regions, because of its nutritional and ethnomedicinal values. Melons are naturally low in fat and sodium, have no cholesterol, and provide many essential nutrients such as potassium, in addition to being a rich source of beta-carotene and vitamin C [1, 2]. Chemical analysis has shown that C. melo is rich in moisture, carbohydrate, dietary fibers, minerals, carotenoids, folate and flavonoids such as B-carotene lending, xanthin cryptoxanthin, and phenolic compounds. Phenolic compounds are volatile. They are biosynthetically derived from fatty acids, carotenoids, amino acids and terpenes, while non-volatile constituents include constituents such as B-carotenes, flavonoids, carbohydrates, linoleic acid, acid a-linolenic, glycolipids, phospholipids, amino acids, phenolic compounds. glycosides [3, 4, 5, 6]. The total bioactive components of C. melo have important health values. Traditionally, C. melo is used to treat kidney stones, flatulence, leprosy, fever, jaundice, diabetes, obesity, cough, bronchitis, ascites, anemia, constipation and other abdominal disorders [7, 4, 8]. These total compounds have medicinal value, since the fruits and roots of C. melo are consumed for their therapeutic value [4]. In Cameroon, based on traditional knowledge that has accumulated over centuries, the leaves, the pulp of fruits, and the seeds of C. melo are eaten for their medicinal properties, while the roots are only used for medicinal purposes. The investigating the cytotoxicity and genotoxicity of the sources of C. melo will serve as a measure of safety for its continued use for medicinal purposes.
## II. METHODS
### a) Origin of Plant Material and Preparation of Extracts
In this study, the aqueous extract of the roots of Cucumis melo was used as the test substance, while the root of Allium cepa was used as the test system. The roots of C. melo were obtained from a farm in Baessing in the West Region of Cameroon. At the same time, the Violet Galmi variety of onion (A. cepa of pure line L78 to eliminate individual variations) was provided to us by the Institute of Agricultural Research for Development (IARD) of Maroua (Far North Region, Cameroon). The roots of C. melo were washed in clean water, chopped into small pieces, and then dried in an oven at $48^{\circ}\mathrm{C}$ for two days. Therefore, dry and brittle roots were ground and used to prepare the aqueous solution using the method of [9]. The Allium cepa tests were held at the Applied Biology and Ecology Research Unit (URBEA) Laboratory according to the general description of [10].
Thus, viable bulbs of A. cepa were placed in small transparent disposable cups with their basal ends dipping into various concentrations of the aqueous extract of C. melo roots (0 $\mu$ g/ml, five $\mu$ g/ml, ten $\mu$ g/ml, 20 $\mu$ g/ml, 30 $\mu$ g/ml and 40 $\mu$ g/ml and germinated at room temperature (25 - 30°C) for 96 hours. The control group of onion bulbs was dipped into distilled water (0 $\mu$ g/ml). For each treatment, five bulbs were used per concentration of the test samples.
### b) Macroscopic Examination
The onion bulbs were analysed for number of roots germinated and the length of the roots. Total number of roots grown per onion bulb was counted and recorded. To determine root length, ten cores were randomly taken from each treatment and measured with the help of a hardened stainless-steel digital caliper $150\mathrm{mm}$.
### c) Cytogenetic Analysis
Meristematic root tips were obtained from the roots used for the measurement of lengths. The squashes method was prepared as suggested by [11] using $2\%$ aceto-orcein. The chromosome smears thereof were used to determine the Mitotic Index (MI) and the presence of Chromosomal Aberrations (CA) induced by the aqueous extracts of the roots of C. melo. Five replicates were performed for each treatment, and scoring was done from the ten sources of each replicate. A minimum of 200 mitotic cells were counted from each slide. Mitotic indices (MI) were calculated for each treatment according to the formula: Number of dividing cells/Total number of cells observed $\times 100$. Chromosomal abnormalities were scored in the mitotic cells, and the results are shown in the tables and figures, while the most frequent chromosomal abnormalities are shown in photomicrographs.
### d) Statistical Analysis
The data expressed as means $\pm$ standard deviation and one-way ANOVA and Tukey's post-test (HSD) were applied to evaluate the significances $(\alpha < 0.05)$ of between-treatment differences in percentage germination, root length, mitotic index, and frequency of chromosome and nuclear abnormalities. Python software version 3.2, and Pandas packages, were used under the MacOSX operating system.
## III. RESULTS AND DISCUSSIONS
During the present study, the potential genotoxicity of the aqueous extract of the roots of C. melo was evaluated with the A. cepa essay. This cytotoxicity and genotoxicity were by analyzing two macroscopic parameters that included the number of roots sprouted and the lengths of the bases, as well as microscopic parameters such as the frequencies of MI and Cas in A. cepa cells plant growth, is an irreversible increase in size resulting from mitotic cell division and cell enlargement. The mitotic process also involves tight control and coordination of proliferative activity and growth in meristematic and differentiated tissues [12]. It is considered that, when the proliferative activity (mitosis) is disrupted, growth inhibition follows. In addition, disruption of mitosis could be the failure to respond appropriately to the stressor signal present in its environment [13]. During this study, the stressor signal was provided by the aqueous extract essential the roots of C. melo affected root growth by affecting mitosis in the meristem of the root tip. Therefore, follows that plant root growth inhibition is an indicator of macroscopic cytotoxicity [14, 15, 2].
The mean number of roots and mean lengths of the roots of onion bulbs grown in control (distilled water) and the different concentrations of aqueous extracts of C. melo are shown in Table 1. The results obtained for the further concentration of aqueous extracts $(5\mu \mathrm{g} / \mathrm{ml}$, ten $\mu \mathrm{g} / \mathrm{ml}$, $20\mu \mathrm{g} / \mathrm{ml}$, $30\mu \mathrm{g} / \mathrm{ml}$, and $40\mu \mathrm{g} / \mathrm{ml})$ indicated that the roots grew less when treated with various concentrations of aqueous extracts of C. melo $(102.10\pm 4.18\%)$, $94.44\pm 5.00\%$, $91.14\pm 2.02\%$, $59.02\pm 1.04\%$ and $35.11\pm 0.40\%$ respectively) than in the control group $(113.15\pm 6.10)$ (Table 1). Inhibition of root growth was, therefore, concentration-dependent and was statistically significant $(P < 0.05)$ at the test concentrations. For all concentrations of the aqueous extract of C. melo, restricted root germination implied toxicity. Mitotic activity expressed here as MI was the first parameter used to evaluate the cytotoxicity of the aqueous extract of C. melo. Table 3 carries the MI levels for the control and each treatment concentration. As per the data in Table 3, the cytotoxicity levels were observed to increase with a decreased rate of MI. It is evident from this data (Table 3) that the aqueous extract of the roots of C. melo reduced MI compared to the control group. However, the decrease in MI with the extract concentration was extract slight and essential there was stagnation in the mitotic division once the cells entered the Metaphase stage. The extracts reduce the number in cells moving from Metaphase to Anaphase. The data also showed that MI was positively correlated to root length, which decreased with increasing concentration of the aqueous root extracts of C. melo. The mean number of dividing cells was lowest in the highest concentration $(40\mu \mathrm{g} / \mathrm{ml})$ of the aqueous root extract of C. melo. Therefore, in this study, aqueous extract of the roots of C. melo decreased MI of A cepa root tip cells. This decrease was significant for all treatments concentrations $(5\mu \mathrm{g} / \mathrm{ml}$, ten $\mu \mathrm{g} / \mathrm{ml}$, $20\mu \mathrm{g} / \mathrm{ml}$, $30\mu \mathrm{g} / \mathrm{ml}$, and $40\mu \mathrm{g} / \mathrm{ml})$ when compared to the control group. These results showed that all treatment concentrations of the aqueous extract of the roots of C. melo were toxic on A. cepa root tip cells. It is suggested here that the bioactive compounds contained in the roots of C. melo have mitodepressant properties, that's to say slow down cell division in the meristematic cells of the root end of
A. cepa however, without killing them. The resumption of cell proliferation is observed when these mitodepressed roots are immersed again in distilled water. It has been variously shown that the mitodepressive effects of some plant extracts have the ability to block the synthesis of DNA and nucleus proteins [16, 17, 18] thereby reducing MI and hence plant growth. In the case of C. melo, bioactive substances may not have allowed the formation of spindle proteins hence reducing the movement of cells from metaphase to anaphase.
This study was conducted using chromosome aberrations to detect the clastogenic activity of the aqueous extract of the roots of C. melo on the meristematic cells in the root tips of A. cepa. A summary of information on aberrant chromosomes in dividing cells of the root tips of A. cepa treated with different concentrations of the aqueous extract of the roots of C. melo is shown in Table 3. In the meristematic cells of A. cepa treated with different concentrations of aqueous extract of the roots of C. melo, Anaphase bridges, laggards and disorientation, nuclear vacuoles, sticky metaphase and double nuclei (Fig. 2) were found and their frequencies were higher than in the control group. These chromosomal abnormalities were used to quantitatively and qualitatively evaluate the genotoxic potentials of the aqueous extract of the roots of C. melo. Chromosome abnormalities such as recorded in this study probably occurred due to lesions in DNA as well as chromosomal and spindle proteins that cause genetic damage [19]. The genetic damage brings about drastic changes in chromatin, spindle apparatus, and centromere thus preventing alignment at the metaphase plate, and abnormal spindle orientation. This has been shown to occur due to altered quality and quantity of kinetochore heterochromatin [20, 21]. Total chromosome aberration (CA) frequencies in the treated groups were found to be higher than in the control group, and all the differences were statistically significant $(p < 0.05)$. The total percentage of CAs significantly increased with the concentration of extract. Therefore, CAs were significantly dose-dependent. CA was not found in the control group. Although The frequencies of aberrant chromosomes increased after treatment, a dramatic increase was recorded with the highest concentration of the aqueous extracts of the root of C. melo (Table 3). The results of this study revealed that aqueous root extracts of C. melo had a clastogenic effect on the root tip cells of A. cepa, since all concentrations tested induced multiple chromosomal abnormalities.
 Figure 1: Examples of series of onions (A. cepa) cultivated for 96 h in different concentrations of the aqueous extracts of Cucumis melo
$A = 0\mu g / ml$ (control); $B = 5\mu g / ml$; $C = 10\mu g / ml$; $D = 20\mu g / ml$; $E = 30\mu g / ml$; $F = 40\mu g / ml$.
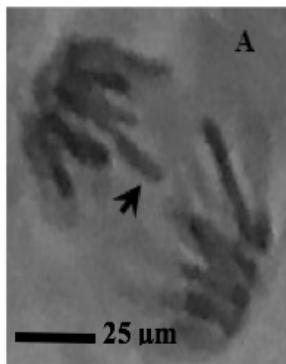
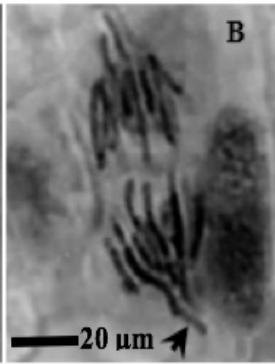
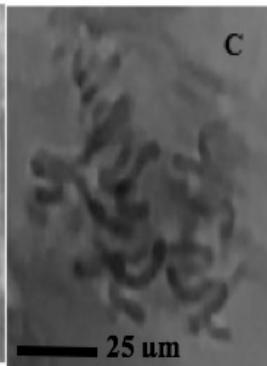
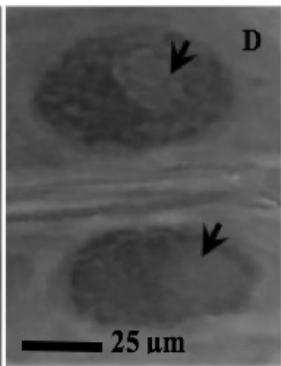
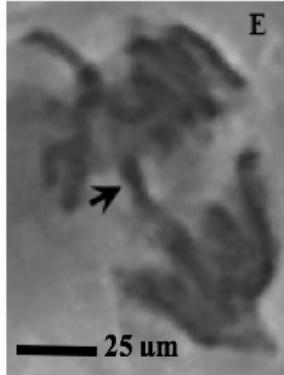

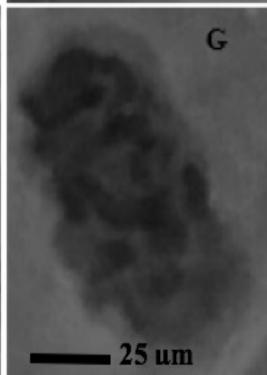
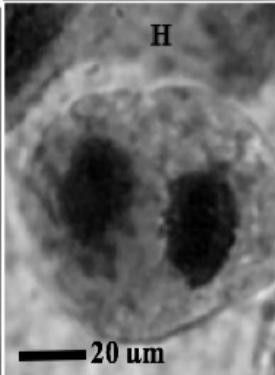 Figure 2: Mitotic and chromosomal aberrations (CA) (arrowed) induced in Allium cepa root tips by aqueous extracts of the roots of C. melo. A and B=Anaphase laggard; C= Disoriented Anaphase; D= Nuclear vacuoles; E=Anaphase Bridge; F=Sticky chromosomes; G=Mature cell showing puff; H=binucleated cell
Table 1: Effects of aqueous extract of Cucumis melo on the sprouting of Allium cepa roots
<table><tr><td>Concentration (μg/ml)</td><td>Mean root number ± SE</td><td>% Total root sprouted of NC</td><td>Percentage inhibition</td><td>95% confidence limit</td></tr><tr><td>0</td><td>113.15±6.10a</td><td>100</td><td>0</td><td>0.000</td></tr><tr><td>5</td><td>102.10±4.18*b</td><td>81</td><td>19</td><td>0.012</td></tr><tr><td>10</td><td>94.44±5.00**cd</td><td>59</td><td>41</td><td>0.010</td></tr><tr><td>20</td><td>91.14±2.02***ef</td><td>49</td><td>51</td><td>0.017</td></tr><tr><td>30</td><td>59.02±1.04***gh</td><td>15</td><td>85</td><td>0.011</td></tr><tr><td>40</td><td>35.11±0.40***i</td><td>3</td><td>97</td><td>0.010</td></tr></table>
Table 2: Effects of aqueous extract of Cucumis melo on the growth of Allium cepa roots
<table><tr><td>Concentration (μg/ml)</td><td>Mean root length ± SE</td><td>% Total root growth of NC</td><td>Percentage inhibition</td><td>95% confidence limit</td></tr><tr><td>0</td><td>5.77±0,5a</td><td>100</td><td>0</td><td>0.000</td></tr><tr><td>5</td><td>4.57±0,11*b</td><td>81</td><td>19</td><td>0.012</td></tr><tr><td>10</td><td>4.11±0,31*cd</td><td>59</td><td>41</td><td>0.010</td></tr><tr><td>20</td><td>4.06±0,13**e</td><td>49</td><td>51</td><td>0.017</td></tr><tr><td>30</td><td>3.88±0,26**e</td><td>15</td><td>85</td><td>0.011</td></tr><tr><td>40</td><td>2.89±0,24**f</td><td>3</td><td>97</td><td>0.010</td></tr></table>
Table 3: Cytogenetic analysis of A. cepa root tips exposed to different concentrations of aqueous extracts of the roots of C. melo
<table><tr><td rowspan="2">Conc. (μg/ml)</td><td rowspan="2">Total cells examined</td><td rowspan="2">% Prophase Index</td><td rowspan="2">% Metaphase Index</td><td rowspan="2">% Anaphase Index</td><td rowspan="2">Mitotic Index (MI)</td><td rowspan="2">Bridges</td><td colspan="3">Anaphase</td><td rowspan="2">Nuclear vacuoles</td><td rowspan="2">Sticky chromosomes</td><td rowspan="2">% Total abnormalities</td></tr><tr><td>Laggards</td><td>Disoriented</td><td>Nuclear vacuoles</td></tr><tr><td>0</td><td>454</td><td>15.80</td><td>14.20</td><td>14.40</td><td>45.40±1.18a</td><td>0</td><td>0</td><td>0</td><td>0</td><td>0</td><td>0</td><td>0a</td></tr><tr><td>5</td><td>439</td><td>15.50</td><td>15.00</td><td>13.40</td><td>43.90±1.53*b</td><td>5</td><td>3</td><td>6</td><td>2</td><td>4</td><td>3.83±1.47*b</td><td></td></tr><tr><td>10</td><td>424</td><td>10.40</td><td>19.80</td><td>12.20</td><td>42.40±1.81*c</td><td>6</td><td>5</td><td>8</td><td>2</td><td>6</td><td>5.16±2.04*cd</td><td></td></tr><tr><td>20</td><td>426</td><td>5.70</td><td>25.90</td><td>11.00</td><td>42.60±1.34***c</td><td>9</td><td>6</td><td>12</td><td>5</td><td>9</td><td>7.50±3.01*ef</td><td></td></tr><tr><td>30</td><td>452</td><td>4.60</td><td>30.10</td><td>10.50</td><td>45.20±1.06**ce</td><td>11</td><td>7</td><td>14</td><td>6</td><td>10</td><td>8.83±3.43**g</td><td></td></tr><tr><td>40</td><td>437</td><td>3.30</td><td>30.30</td><td>10.10</td><td>43.70±1.39***f</td><td>12</td><td>9</td><td>15</td><td>7</td><td>11</td><td>10.00±3.34***h</td><td></td></tr></table>
Abbreviations List
Not used
Declarations
Ethical Approval and Consent to Participate
All experimental studies on plants were complied with relevant institutional, national, and international guidelines and legislation.
Consent to Publication
We declare hereby that this work has not been published or accepted, in whole or in part, and that it is not selected for publication in another journal. All authors have approved the manuscript and agree with its submission.
Availability of Data and Material
Datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Competing Interests
The authors have no competing interests to declare that are relevant to the content of this article.
Funding
No funding was obtained for this study.
Author Contributions
N.A. and S.R.A. conceived and conducted research experiments, N.A. analysed data and conducted statistical analyses, N.A. wrote the original draft article, S.R.A reviewed the manuscript.
## ACKNOWLEDGEMENTS
Our sincere thanks go to Professor WATCHO Pierre, the Head, Reproduction and Sexual Disorders Sub-Unit, Physiology, University of Dschang for providing laboratory facilities and our heartful thanks go to Pr. MAYAKA Théodore Bileng, the Head, Applied Ecology Laboratory (LABEA), Faculty of Science, University of Dschang for providing laboratory facilities.
Generating HTML Viewer...
References
22 Cites in Article
Gene Lester,Melon (1997). Melon (Cucumis melo L.) Fruit Nutritional Quality and Health Functionality.
M Shivapriya,N Kotamballi,S Bihmanagouda (2021). Nutritional composition and health benefits.
Vassiliki Homatidou,Sophia Karvouni,Vassilis Dourtoglou,Constantine Poulos (1992). Determination of total volatile components of Cucumis melo L. variety cantaloupensis.
H Asif,N Akhtar,S Sultana,S Rehman,M Akram,J Rehman (2014). Medicinal properties of Cucumis melo L.
Sana Mallek-Ayadi,Neila Bahloul,Nabil Kechaou (2017). Characterization, phenolic compounds and functional properties of Cucumis melo L. peels.
Sana Mallek‐ayadi,Neila Bahloul,Semia Baklouti,Nabil Kechaou (2022). Bioactive compounds from <i>Cucumis melo</i> L. fruits as potential nutraceutical food ingredients and juice processing using membrane technology.
N Gill,J Bajwa,K Dhiman,P Sharma,S Sood,P Sharma,B Singh,M Bali (2011). Evaluation of Therapeutic Potential of Traditionally Consumed Cucumis melo Seeds.
Shweta Saboo,Priyanka Thorat,Ganesh Tapadiya,S Khadabadi (2013). DISTRIBUTION AND ANCIENT-RECENT MEDICINAL USES OF TRICHOSANTHES SPECIES.
R Seino,M Atonleu,A Ngnaniyyi,I Dongmo,R Tchoupou (2020). Cytogenotoxic assessment of the aqueous extract of Citrullus lanatus (Cucurbitaceae) leaves using the spermatogonial germ-linecells of Zonocerus variegatus L. (Orthoptera: Pyrgomoephidae).
Catalin Rosculete,Elena Bonciu,Elena Rosculete,Liviu Olaru (2019). Determination of the Environmental Pollution Potential of Some Herbicides by the Assessment of Cytotoxic and Genotoxic Effects on Allium cepa.
Al-Sabti K (1989). Allium test for air and water borne pollution control.
Séverine Planchais,Nathalie Glab,Dirk Inzé,Catherine Bergounioux (2000). Chemical inhibitors: a tool for plant cell cycle studies.
Feifei Qi,Fuxin Zhang (2002). Cell Cycle Regulation in the Plant Response to Stress.
T Çelik,O Aslantürk (2009). Investigation of cytotoxic and genotoxic effects of Ecballium elaterium.
Kai-Sung Wang,Chi-Yuan Lu,Shih-Hsien Chang (2011). Evaluation of acute toxicity and teratogenic effects of plant growth regulators by Daphnia magna embryo assay.
E Schulze,M Kirschner (2007). Microtubule dynamics in interphase cells..
A Akinboro,A Bakare (2007). Cytotoxic and genotoxic effects of aqueous extracts of five medicinal plants on Allium cepa Linn..
Robert Sablowski,Marcelo Carnier Dornelas (2013). Interplay between cell growth and cell cycle in plants.
E Zeiger (2010). Genetic Toxicology Testing.
A Jennifer,M Graves,P Zelesco (1988). Chromosome segregation from cell hybrids. V. Does segregation result from asynchronous centromere separation.
P Zelesco,A Jennifer,M Graves (1988). Chromosome segregation from cell hybrids IV. Movement and position of segregant set chromosomes in early phase interspecific cell hybrids.
Muhammad Aslam (2015). Specific Role of Proline Against Heavy Metals Toxicity in Plants.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Ngnaniyyi Abdoul. 2026. \u201cMusk Melon Root Extract (Cucumis Melo L.) Inhibits Mitosis and Growth of Meristematic Cells in the Allium Cepa Essay through Chromosomal Abnormalities\u201d. Global Journal of Science Frontier Research - C: Biological Science GJSFR-C Volume 23 (GJSFR Volume 23 Issue C1): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.