We propose to extend the existing theory of irreversible processes (TIP) to include reversible real processes associated with the performance of useful work. This is achieved by the fact that the main quantities used by this theory, thermodynamic forces and fluxes, are derived not from the principle of increasing entropy, but rather from the law of the conservation of energy. This way of constructing TIP prevents the occurrence of thermodynamic inequalities and allows one to substantiate all its provisions without invoking the postulates and considerations of a molecular-kinetic and statistical-mechanical nature. This opens up the possibility of further reducing the number of empirical coefficients and expanding the scope of TIP applicability to nonlinear systems and states that are far from equilibrium, as well as to energy conversion processes which are primarily of interest to power engineers, technologists, biophysicists and astro-physicists. At the same time, the unity of the laws of transformation of all forms of energy and the difference between their equations, reciprocity relations and efficiency criteria from the generally accepted ones are proved.
## I. INTRODUCTION
In the history of science there are frequent cases when a new theory brought in significant change in the natural science paradigm. The last part of the twentieth century was no exception; as this paradigm shift occurred in the fundamental theories of the thermodynamics of irreversible processes (TIP). Researchers from many countries contributed to its creation [1-11]. It enriched the theoretical thought of the twentieth century with the "principle of reciprocity" of heterogeneous phenomena, sometimes called the "fourth law of thermodynamics", and explained the many effects that arise at the junctions of fundamental disciplines due to the simultaneous occurrence of several non-static processes. However, later on, interest in this theory began to fade. To a large extent this is due, in our opinion, to the fact that the basic quantities underlying this theory, thermodynamic forces $X_{i}$ and fluxes $J_{i}$, are included on the basis of the principle of entropy increase, which exclude from consideration the reversible component of real processes. Yet it is precisely this component, connected to the execution of useful external work $W^{\mathrm{e}}$, which is of interest primarily in fields related to energy, technology, biophysics and astrophysics. This drawback can be eliminated by switching to finding these forces and fluxes on a basis of a more general law of conservation of energy. We shall consider the advantages that non equilibrium thermodynamics gains as a result.
## II. PREVENTING THE TRANSFORMATION OF EQUATIONS OF THERMODYNAMICS INTO INEQUALITIES
It is known that the equations of the 1st and 2nd laws of classical thermodynamics of open systems are combined in the form of the Gibbs relation[12]:
$$
d U = T d S - p c V + \Sigma_ {k} \mu_ {k} d N _ {k} \tag {1}
$$
This relation connects the internal energy $U$ of the object (or system) under study with its entropy $S$, volume $V$ and the number $N_{k}$ of moles of the $k^{\text{th}}$ substances, as well as with the generalized potentials $\psi_{j}$ conjugated with them (the chemical potentials of these substances is represented by $\mu_{k}$, absolute temperature by $T$ and pressure $p$ by.). This then becomes the inequalities:
$$
\delta Q \neq T d S; \delta W _ {\rho} \neq p d V; \delta W _ {k} \neq \mu_ {k} d N _ {k} \tag {2}
$$
This happens because in non-equilibrium systems the parameters $S, V, N_{k}$ change not only as a result of external energy exchange, but also as a result of internal relaxation processes (the number of moles $N_{k}$ is due to chemical reactions; the volume $V$ corresponds to the expanded form in a vacuum while not performing work; the entropy $S$ is due to friction and other irreversible processes.) As a result, the energy exchange of the system with the environment can no longer be found on the basis of changes in these parameters, and the mathematical apparatus of thermodynamics based on equation (1) turns out to be inapplicable. This disadvantage can be eliminated by going directly to the fluxes of these energy carriers across the boundaries of the system. For this we use the law of conservation of energy in the form proposed by N. Umov (1873) [13]:
$$
d U / d t = - \oint j _ {u} \cdot d \mathrm {f}, \tag {3}
$$
where $j_{u}$ ( $W \cdot m^{-2}$ ) is the internal energy flux density through the vector element $df$ of the closed surface $f$ of the system of constant volume $V$ in the direction of the external normal $n$ (Figure 1).
 Figure 1: Energy flux through the boundary of the system
Unlike the later equation of J. Poynting (1884), this form of the energy conservation law takes into account the kinetics of real processes, without making any assumptions about the mechanism of energy transfer in a solid media or about the internal structure of the system.
According to this equation about short-range effects, energy $U$ does not simply disappear at some points of space and arise in others, but rather carries the $i^{\text{th}}$ energy carrier $\Theta_{i}$ (with the number of moles $k$ of the substance $N_{k}$, their corresponding charges $Q_{k}$, their entropies $S_{k}$, the impulses $P_{k}$, etc.) through the fixed boundaries of the system. Let us now find the expanded form of this law, which is valid for any $i^{\text{th}}$ material carrier of energy. For this, we will take into account that the energy flux $j_{u}$ is the sum of the fluxes $j_{ui}$ carried by each of them. These fluxes, in turn, are expressed by the product of the flux density of the $i^{\text{th}}$ energy carrier $j_{i} = \rho_{i} v_{i}$ by its potential $\psi_{i} \equiv d U_{ii} / d \Theta_{i}$, where $\rho_{k} = d \Theta_{i} / d V$ and $v_{i} = d r_{i} / d t$ are the density of the $i^{\text{th}}$ energy carrier and the rate of its transfer across fixed boundaries systems, resp., i.e. $j_{ui} = \psi_{i} j_{i}$, so that
$$
j_{u} = \Sigma j_{ui} = \Sigma \psi_{i} j_{i},
$$
Using the Gauss-Ostrogradsky theorem, we transform the integral $\oint j_u\cdot df$ into a volume integral $\int \nabla \cdot j_{u}\mathrm{d}V$. Then, after decomposing $\nabla j_{\perp} = \nabla (\psi_j i)$ into independent components $\Sigma_{i}\Psi_{i}\nabla \cdot j_{i} + \Sigma_{i}j_{i}\cdot \nabla \psi_{ik}$, the energy conservation law (3) takes the form:
$$
d U / d t + \Sigma_ {i} \int \psi_ {i} \nabla \cdot j _ {i} d V + \Sigma_ {i} \int j _ {i} \cdot \nabla \psi_ {i} d V = 0 \tag {5}
$$
If we take the average value $\Psi_{i}$ of the potential $\psi_{i}$ and the average value $X_{i}$ of the potential gradient $\nabla \psi_{i}$ both from under the integral sign, then equation (5) can be expressed in terms of the parameters of the system as a whole, as is customary in classical thermodynamics:
$$
dU/dt+\Sigma_{i}\Psi_{i}J_{i}+\Sigma_{i}X_{i}\cdot J_{i}=0.
$$
Here $J_{i} = \int \nabla \cdot j_{i}dV = \oint j_{i}df$ is the scalar flux of the $i^{\mathrm{th}}$ energy carrier through the boundaries of the system; $J_{i} = \int \rho_{i}\nu_{i}dV = \Theta_{i}\overline{\pmb{v}}_{i}$ is its vector flux (impulse).
Unlike the Gibbs relation, Equation (6) contains 2 terms $(i = 1,2,\dots n)$ and describes not only the processes of introducing the $k^{\mathrm{th}}$ substance $N_{k}$ into the system as well as the series $Q_{k}$, the entropy $S_{k}$, the momentum $P_{k}$, etc. in the homogenous system being investigated, but also the processes of redistribution of the system volume of overcoming the forces of the $X_{i}$ and the performance of work "against equilibrium" in it. Therefore, it is applicable to a wide class of open $(N_{k} = var)$, non-closed $(X_{i} = var)$ and non-isolated systems $(U = var)$, which are the object of study in other fundamental disciplines. At the same time, it allows the irreversibility of the above processes. Indeed, considering (6) together with the integral equation of the energy carrier balance $\Theta_{i}$
$$
d\Theta_{i}/dt + \int\nabla\cdot j_{i}dV = \int\sigma_{i}dV.
$$
In this case, the densities of local and substantial fluxes $j_{u}$ coincide. We find that, besides the energy carrier $\Theta_{i}$ appearing in the Gibbs ratio, it takes into account the presence of these internal sources of density $\sigma_{i}$. It is easy to see that under the conditions of local equilibrium $(X_{i} = 0)$, Eq. (6) takes the form
$$
dU/dt = \Sigma_{i}\Psi_{i}d\Theta_{k}/dt-\Sigma_{i}\Psi_{i}\int\sigma_{i}dV,
$$
i.e., it transforms into a generalized Gibbs ratio for complex multivariable systems $dU = \Sigma_{i}\psi_{i}d\Theta_{i}$ only when the internal sources of entropy $d_{u}S / dt = \int \sigma_{s}dV$ and other energy carriers $d_{u}\Theta_{i} / dt = \int \sigma_{i}dV$ (including products of chemical reactions $d_{u}N_{k} / dt$ ) disappear. This testifies to the inconsistency of the hypothesis of local equilibrium, according to which the state of an element of the inhomogeneous continuum of the system is characterized by the same set of variables as in equilibrium. This follows from the fact that this assumption also means the absence of "production of entropy" ( $d_{u}S / dt > 0$ ). The latter makes it necessary to introduce the parameters of in homogeneity $X_{j}$ and the fluxes $J_{i}$ associated with them into the equations of nonequilibrium thermodynamics.
It is remarkable that Equation (6) does not become an inequality despite the obvious inclusion of the dynamic (irreversible) processes under consideration. This solves a major "problem of thermodynamic inequalities" which until now has prevented any application of the mathematical apparatus of nonequilibrium thermodynamics to real processes (i.e., those with fluxes at finite speeds).
It is also important that our derivation of an expanded form of the law of conservation of energy (6) contains definite forces $X_{i}$ and fluxes $J_{i}$. This bestows it with a definite sense corresponding to an energy field strength $\Theta_{ik}$ and averaged pulse $k^{\mathrm{th}}$ energy source $P_{i} = \Theta_{i}v_{i}$. Furthermore, it does not require a compilation of complex and cumbersome equations for the balance of matter, charge, momentum, energy and entropy. This dramatically simplifies the ability for thermodynamics to solve certain problems.
## III. THERMODYNAMIC DERIVATION OF THE ONSAGER RECIPROCAL RELATIONS
One of the most important provisions of the theory of irreversible processes is the "reciprocity relation" $L_{ij} = L_{ji}$ between the off-diagonal coefficients $L_{ij}$ and $L_i$ in the "phenomenological" laws postulated by L. Onsager:
$$
J_{i} = \sum_{j} L_{ij} X_{j}
$$
These ratios establish the relationship between dissimilar fluxes $J_{i}$ and forces $X_{j}$ and reduce the number of proportionality coefficients between them to be experimentally determined from $n^2$ to $n$ ( $n + 1$ /2). To prove these relations, the future Nobel laureate L. Onsager had to use the theory of fluctuations, the principle of microscopic reversibility and an additional postulate about the linear nature of the laws of decay of fluctuations [1]. All three of these assumptions are somewhat outside of classical thermodynamics; therefore, he rightly called his theory "quasithermodynamics".
Meanwhile, it can be shown that these relations gain support from the law of the conservation of energy (6). From that law, based on the independence of the mixed derivative from the order of differentiation with respect to the variables $X_{i}$ and $X_{j}$ ( $i,j = 1,2,\ldots n$ ), it follows:
$$
\partial^ {2} U / \partial X _ {i} \partial X _ {j} = \partial^ {2} U / \partial X _ {j} \partial X _ {i} \tag {10}
$$
This directly implies the relationship between unlike fluxes and forces, which we term differential reciprocal relations[13]:
$$
(\partial J_{j} / \partial X_{j}) = (\partial J_{j} / \partial X_{ ext{}}j).
$$
These relations are applicable to both linear and nonlinear transport laws and allow any dependence of the coefficients $L_{ij}$ on the parameters of the equilibrium state $\psi_i$ and $\Theta_j$. Application to the linear laws (9) directly leads to the symmetry of the matrix of phenomenological coefficients $L_{ij} = L_{ji}$:
$$
(\partial J_{i}/\partial X_{j}) = L_{ij} = (\partial J_{j}/\partial X_{ ext{ }j}) = L_{ji}
$$
Their derivation shows that these relationships are a consequence of more general reasons than the reversibility in time of micro processes. This explains why these relationships have often turned out to be valid far in domains far beyond the above conditions.
## IV. FINDING "SUPERPOSITION EFFECTS" WITHOUT USING ONSAGER RELATIONS
In isolated systems, the sum of internal forces $\Sigma_{i}F_{i}$ ( $i = 1,2\dots n$ ) is always zero. This means that, in accordance with Newton's $3^{\mathrm{rd}}$ law, any one of them can be expressed as the sum of $n = 1$ different forces of the $j^{\mathrm{th}}$ kind: $F_{i} = -\Sigma_{n-1}F_{j}$. The relationship of these forces to thermodynamic forces $X_{i}$ is easy to construct. From the expression for power $dW/dt = X_{i}J_{i} = F_{i}\overline{\theta}_{i}$ it follows that $X_{i} = F_{i}/\Theta_{i}$, i.e., it represents the precise meaning of force in its more general physical interpretation. Taking this into account, laws (17) can be represented in a form closer to (9):
$$
J_{i}=L_{i}\Sigma_{j}\Theta_{j}X_{j}
$$
Such a form of the laws of transfer and relaxation does not require the empirical coefficients $L_{i}$ to be constant; this expands the scope of these equations' applicability to nonlinear systems and states far from equilibrium. In addition, it allows to propose a new method for finding the "superposition effects" of irreversible process which are due to "partial" (incomplete) equilibria of the $i^{\text{th}}$ kind ( $J_{i} = 0$ ). The specificity of this method is easier to understand with the example of the diffusion of the $k^{\text{th}}$ substance in a continuous heterogeneous composition (with the concentration of components $c_{j}$, temperature $T$ and pressure $p$ ). According to laws (13), this process has the form:
$$
J _ {k} = - D _ {k} \nabla \mu_ {k}, \tag {14}
$$
where $D_{k}$ is the diffusion coefficient of the $k^{\mathrm{th}}$ substance; $\mu_{k}$ is its chemical potential.
If we represent $\nabla \mu_{k}$ through its derivatives with respect to the concentrations $c_{j}$ of its independent components, their temperature and pressure, then equation (14) can take the form:
$$
J _ {k} = - D _ {k} \left(\Sigma_ {j} \mu_ {k j} ^ {*} \nabla c _ {j} + s _ {k} ^ {*} \nabla T + v _ {k} ^ {*} \nabla p\right). \tag {15}
$$
where $\mu_{kj}^{*}\equiv (\partial \mu_{k} / \partial c_{j})$ $s_k^*\equiv (\partial \mu_k / \partial T)$ $\nu_{k}^{*}\equiv (\partial \mu_{k} / \partial p)$
Three components of the resulting force $F_{k}$ on the right side of this expression handle the usual (concentration) diffusion $F_{kc} = \Sigma_j\mu_{kj}^*\nabla c_j$, thermal diffusion $F_{kT} = s_k^*\nabla T$ and baro diffusion $F_{kp} = v_k^*\nabla p$. This allows one to separate the thermodynamic factors $\mu_{kj},s_k^*$, $v_{k}^{*}$ and the kinetic factors $D_{k}$ of multi component diffusion and establish a number of empirically established relationships between them[15]. Given the existing experimental means, it was mathematically unsound to obtain such results via the Onsager diffusion equation $J_{k} = -\Sigma_{i}D_{ki}\nabla \mu_{i}[1]$
As another example, consider an inhomogeneous system divided into two parts by a porous partition. If a temperature difference $(\Delta T \neq 0)$ is created in it, then a gas or liquid flux $J_{k} = D_{k}(s_{k}^{*}\nabla T - v_{k}^{*}\nabla p)$ occurs through the partition, leading, under conditions of incomplete equilibrium ( $J_{k} = 0$ ), to the occurrence of a pressure difference on both sides of the partition (Feddersen effect, 1873):
$$
(\Delta p / \Delta T)_{\mathrm{st}} = - q_{k}^{*} / T v_{k}^{*}
$$
where $q_{k}^{*} = T s_{k}^{*}$ represents the heat transfer of the $k^{\text{th}}$ substance.
This phenomenon is now called thermoosmosis. The opposite phenomenon has also been seen: the appearance of a temperature difference on both sides of the partition when air or other gas is forced through it. Both of these effects are of the same nature as the Knudsen effect (1910) - the appearance of a pressure difference in vessels connected by a capillary or a narrow slit and filled with gas of different temperatures. They are also of the same effect as the Allen and Jones "fountain effect" (1938), consisting of liquid helium II flowing out, at the slightest heating, from a vessel closed with a porous stopper. The opposite phenomenon - the occurrence of a temperature difference when a pressure difference is created on both sides of the partition - is called the mechanocaloric effect (Daunt-Mendelssohn).
In the case of systems that initially have the same pressure on both sides of the porous partition $(\Delta p = 0)$ and initially the same concentration of the $k^{\mathrm{th}}$ substance $(\Delta c_{k} = 0)$, when a temperature difference $\Delta T$ is created, a concentration difference occurs on both sides of it (the Soret effect, 1881):
$$
(\Delta c_{k} / \Delta T)_{\mathrm{cr}} = - q_{k}^{*} / T \mu_{k k} .
$$
The opposite phenomenon, the appearance of temperature gradients during diffusion mixing of components, was discovered by Dufour in 1872 and bears his name. In isothermal systems $(\Delta T = 0)$ for creating pressure differential across the membrane $\Delta p$ occurs via reverse osmosis, i.e., the separation of a binary solution with separation from the $k^{\text{th}}$ component (usually a solvent). This phenomenon is widely used in water treatment plants. This occurs when the concentration difference $k^{\text{th}}$ part is given by the expression:
$$
\left(\Delta c _ {k} / \Delta p\right) _ {\mathrm {s t}} = - v _ {k} / \mu_ {k k}. \tag {18}
$$
These results are consistent with those obtained in the framework of TIP [6,8]. However, for this it was not necessary to assume the linearity of phenomenological laws, postulate the constancy of the phenomenological coefficients $L_{i}$ or $D_{k}$ and resort to Onsager's reciprocity relations. At the same time, it becomes clear that these effects arise due to the onset of states of partial (incomplete) equilibrium; any multivariable system passes through such states on its way to full equilibrium. In this case, the "effects of superposition" are the result of superposition not of fluxes $J_{i}$, but rather of forces $F_{j}$ in full accordance with the principles of mechanics. The advantages of this method consist not only in the further number of phenomenological coefficients from $n(n + 1)/2$ in TIP to $n$ [14], but also in the possibility of finding superposition effects in nonlinear systems far from equilibrium. In this case, the TIP itself becomes free from any postulates expressing the coefficients $L_{i}$ as a function of the parameters of the system.
## V. ESTABLISHING THE FUNDAMENTAL
### DIFFERENCE BETWEEN THE LAWS OF
#### RELAXATION AND ENERGY CONVERSION
Consider an isolated system $(dU / dt = 0; J_{i} = 0)$ in which energy is converted from one form to another. For such a system, from (6) at once follows:
$$
\Sigma_{k} X_{k} \mathcal{J}_{k} = 0.
$$
For the process of converting some $j^{\mathrm{th}}$ form of energy into $j^{\mathrm{th}}$, this expression can be given the form:
$$
J_{i}/X_{j} = -J_{j}/X_{i}
$$
According to this expression, the direction of the internal flux $J_{i}$ of the $i^{\text{th}}$ energy carrier, induced by the "exterior" force $X_{j}$, is opposed to the direction of the "exterior" flux $J_{j}$, induced by the driving force $X_{i}$. If we denote the ratio $J_{i} / X_{j}$ by $L_{ij}$, and the ratio $J_{j} / X_{i}$ by $L_{i}$, then we come to the antisymmetric Onsager-Casimir reciprocity relations[6, 8]:
$$
L_{jj} = - L_{ji}.
$$
This provision keeps the opposing direction of diverse forces; fluxes in the processes of energy conversion and possesses a general physical status. In particular, Faraday's law of induction follows from it, if by $J_{i}$ we mean the flux of magnetic coupling (expressed by the number of lines of force), and by $X_{j}$, the resulting voltage.
However, these conditions of anti symmetric matrices with "phenomenological" coefficients indicate also that for the processes of intercom version of ordered forms of energy, in the law (9) of Onsager, the coefficients $L_{ij}$ and $L_{ji}$ must be modified by the opposite signs:
$$
J_{i}=L_{ij}X_{i}-L_{ji}X_{ ext{J}}
$$
$$
J_{j}=L_{j r}X_{r}-L_{j j}X_{j}.
$$
In particular, as is well known from the practice of working with a welding transformer, an increase in the voltage in the secondary circuit $X_{j}$ (approaching the "no-load" mode) causes a decrease in the current in the primary circuit $J_{j}$, and the "short circuit" mode ( $X_{j} = 0$ ) on the contrary, increases it. Thus, Equations (22, 23) are more consistent with the phenomenological status (based on experience) than is Equation (9).
It is no less important that the condition of interconnection of forces and fluxes (19) is a consequence of the law of conservation of energy (6). Under conditions of system relaxation, these conditions of counter-directional flux are absent, so that the fluxes $J_{i}$ and $J_{j}$ become independent. In this case, the reciprocity relations $L_{ij} = L_{ji}$ are fulfilled trivially (they vanish), and the equations of transfer and relaxation take the form of equations of heat conduction, electrical conductivity, diffusion, etc., in which the flux $J_{i}$ becomes a unique (eponymous) function of the thermodynamic force $X_{i}$. This independence was also assumed in the theory of L. Onsager, since he defined the scalar fluxes $J_{i}$ as time derivatives of the independent parameters of the system. Therefore, strictly speaking, he did not have sufficient grounds for postulating rules (9), in which each of the fluxes depends on all forces acting in the system.
## VI. DEVELOPMENT OF A UNIVERSAL CRITERION FOR THE EFFICIENCY OF ENERGY CONVERTERS
It is generally accepted that the energy conversion efficiency of any reversible non-thermal machine is equal to unity, while for a heat engine it is limited by the thermal efficiency of an ideal Carnot machine [15]:
$$
\eta_ {t} = 1 - T _ {2} / T _ {1} < 1, \tag {24}
$$
where $T_{1}, T_{2}$ are constant temperatures of supply and removal of heat in the heat engine cycle, equal to the absolute temperatures of the heat source and sink.
This "discrimination" of heat engines is based on the firm belief that "heat and work are, in principle, unequal" [15]. In fact, a closer look reveals that this reflects a misunderstanding of the concepts of absolute and relative efficiency. Thermal efficiency $\eta_{t}$, like its analog $\eta_{i}$ for ordered forms of energy, characterize the ratio of the work $W_{i}$ performed by the converter to the energy $U_{i}$ supplied from the source of the $i^{\text{th}}$ form of energy. Such efficiencies are usually called absolute.
According to the theorem of Carnot efficiency, an ideal cycle of the heat engine does not depend on the properties of its working body, nor on the design features of the machine or the mode of operation. Therefore, such "efficiency" more likely characterizes not its coefficient of performance, but rather the possibilities offered by nature thanks to its inherent spatial in homogeneity (difference of temperatures of the hot and cold heat sources). Strictly speaking, this figure should not have been called "machine efficiency" because this figure is characterized more by the "degree of instability" of the heat source.
The concept of efficiency of electric and other motors has a different meaning. Such efficiencies characterize the ratio of the work $W_{i}$ actually performed by the engineto the theoretically possible work $W_{i}^{t}$. They take into account the losses in the machine itself and are ideally equal to one. Such efficiencies are called relative internal $\eta_{oi}$. In thermodynamics such efficiency $\eta_{oi}$ is an evaluation of the performance of the processes of compression or expansion of a body upon which work is done. Naturally, the application of the same term "efficiency" to these two fundamentally different concepts causes non-specialists to misunderstand the inefficiency of heat engines.
In this respect, it is very useful to represent the efficiency through energy fluxes. Non equilibrium thermodynamics allows us to express the efficiency ratio in terms of the output power $N_{j}$ and the input transforming device $N_{i}[16]$:
$$
\eta_{N}=N_{j}/N_{i}=X_{j}\cdot J_{j}/X_{i}\cdot J_{\text{i}}\leq 1.
$$
This efficiency, which we term "power-based energy conversion efficiency", or henceforth "power efficiency" for short, is equally applicable to thermal and nonthermal, cyclic and acyclic, straight and reversed machines, including the "direct energy conversion" machines. It takes into account both the kinetics of the energy conversion process and all types of losses associated with both the delivery of energy to the energy converter and the energy conversion process itself. It also depends on the operating mode of the installation, twice turning to zero: in "idle" $(J_{j} = 0)$ and in "short circuit" modes $(X_{j} = 0)$. This also distinguishes it from the "exergy" efficiency, which is expressed by the ratio of free energies at the outlet and inlet of the installation. In a word, this efficiency most fully reflects the thermodynamic performance of the installation and the degree to which it realizes the possibilities that the source of ordered energy provides. Moreover, such an efficiency is the only possible indicator of the performance of an installation in those cases when the concept of absolute efficiency becomes inapplicable due to the impossibility of separating energy sources and receivers in a continuous medium. Examples include force fields, chemically reactive environments, polarized or magnetized bodies, or dissociated or ionized gases. All this makes it an irreplaceable tool for analyzing the efficiency of not only energy, but also technological installations, as well as energy converters created by nature itself. This is especially important due to the fact that many non-experts confuse efficiency with the heat transfer coefficient or the coefficient of performance, which may be distinguished by their units. Therefore, the use of power efficiency (24) not only reveals the unity of the laws of transformation of any forms of energy, but also allows us to propose a theory of the similarity of power plants of various types.
## VII. CONSTRUCTION OF A THEORY OF SIMILARITY OF POWER PLANTS
A proper generalization of TIP to the processes of useful energy conversion in various machines allows us to propose a theory of the similarity of power plants [16]. This would complement the classical theory of heat engines by analyzing the relationship of thermodynamic efficiency (energy conversion efficiency) with productivity (based on power $N$ ) and the operating mode of power and technological plants. As in the theory of similarity of heat transfer processes, the mathematical model of such systems includes, along with equations (22,23), the conditions for the uniqueness of the object of study. The latter contain boundary conditions determined in the case under consideration by the magnitude of the driving forces at the border with the energy source or object of work $X_{i}, X_{j}$, or by the magnitude of the fluxes $J_{i}, J_{j}$ at these boundaries, and by the initial conditions. These latter are set by the magnitude of these forces $X_{jo}$ or fluxes $J_{i}$ in the initial mode, for example, at the "idle" of the installation (at $J_{j} = 0$ ), or in the "short circuit" mode $J_{jk}$ (at $X_{j} = 0$ ), as well as the coefficients $L_{ij}$ ( $i, j = 1,2$ ) characterizing the transport properties of the system. These conditions make it possible to give the transport equations (22, 23) a dimensionless form
$$
X_{j}/X_{jo}+J_{j}/J_{jk}=1.
$$
and on its basis, propose a number of similarity criteria for power plants. One of them, which we called the load criterion, is composed of the boundary conditions set by the value of the forces $X_{j}$, $X_{j\circ}$ or fluxes $J_{j}$, $J_{jk}$
$$
\mathcal{B} = J_{j} / J_{j k} = 1 - X_{j} / X_{j o}.
$$
This criterion depends solely on the load of the installation and varies from zero in no-load mode $(J_{j} = 0)$ to one in the "short circuit" mode $(X_{j} = 0)$
Another criterion consists of the resistance coefficients $R_{ij}$, the reciprocal of the conductivity coefficients $L_{ij}$:
$$
\Phi = R_{ij} R_{ji} / R_{ii} R_{jj}
$$
This formula is similar in meaning to the ratio of reactive and active resistances, known in radio engineering as the "quality factor", or "Q-factor" for short, of the circuit, and therefore is called the "criterion of the Q-factor" of the installation. Its value fluctuates from zero to infinity $(0 < \Phi < \infty)$, increasing as the "active" resistances (from the side of scattering forces) $R_{ii}$ and $R_{jj}$ decrease and the "reactive" resistances $R_{ji}$ (from the side of "heterogeneous" forces) increase. Like thermal resistances in the theory of heat transfer, they depend on the transport properties of the system, i.e., ultimately, on the design performance of the installation.
Using these criteria in the expression for the power efficiency (24), it can be given the form of a critical equation for the energy conversion process:
$$
\eta_ {N} = (1 - \mathcal {B}) / (1 + 1 / B \Phi). \tag {29}
$$
Consequently, the efficiency of any energy converter under similar conditions $(\mathcal{B}, \Phi$ as above) is the same. It is expedient to call this provision the principle of similarity of power plants)[16].
This principle allows one to build a universal load characteristic of linear energy - converting systems (Figure 2) [16]. Solid lines in the diagram show the dependence of the power efficiency $\eta_{N}$ of the installation on the load criterion $B$ at different values of the quality factor $\Phi$, and the dash-dotted line shows the dependence on the load of its output power $N_{j}$.
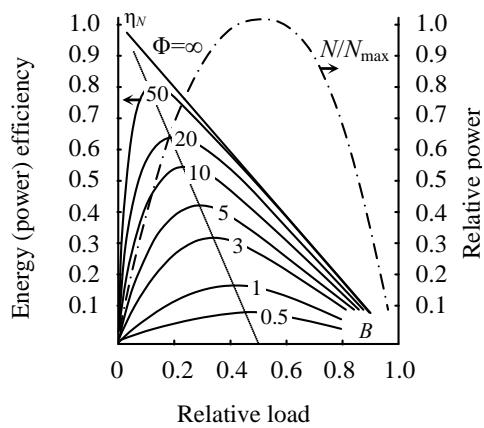 Fig. 2: Universal load characteristics of energy converters
As can be seen in Fig. 2, in the absence of energy losses $(\varPhi=\infty)$ and the steady state of the process of its conversion $(B\rightarrow 0)$, the efficiency of the installation reaches, as expected, unity. However, in all other cases the value of the power-based efficiency becomes zero twice, once in "idle" setting $(B=0, J_{j}=0)$ and once in the "short-circuit" setting $(B=1, X_{j}=0)$. This result is obtained by taking into account the energy consumption for the installation's own needs, as well as losses from irreversible energy exchange (including heat exchange) between the energy source and the working fluid of the installation, friction in pipelines and all kinds of energy "leaks" occurring at idle running of the installation.
The main feature of this characteristic is the presence of a pronounced maximum efficiency for a given load of the installation. Such modes, usually labeled nominal, move farther and farther from the maximum power mode, corresponding to the relative load $B = 0.5$, as the efficiency increases. As a result, the power-based efficiency of a real Carnot cycle (from quasi-statistical characteristics of the processes) at $O < \infty$ is not a maximum, but rather zero. Thus, taking into account the power and performance of the power plant brings the results of the thermodynamic analysis of its efficiency closer to reality.
Universal load characteristics are very useful not only for monitoring compliance with the most economical operating modes of basic, peak and transport power plants, but also when choosing the most promising of them with respect to future operating modes. In this way, non equilibrium thermodynamics of energy conversion processes acquire important practical applications.
## VIII. SUMMARY AND CONCLUSION
1. The main disadvantage in using their complete theory of thermodynamics of irreversible processes (TIP) as a general physical theory is its original limitations on processes of energy dissipation; this can be traced to its dependence on the principle of increasing entropy.
2. The approach to non-equilibrium thermodynamics from a more general position of an energy conservation law shows a failure of the hypothesis of local equilibrium. Thus, it highlights the necessity of introducing additional variables of a nonequilibrium state, where upon potential gradients and generalized process speeds would arise.
3. Determination of the basic quantities in which TIP is defined -- thermodynamic forces and energy fluxes - on a more general basis of a law governing the transfer of energy in path environments allows us to create a locally non-equilibrium thermodynamics. This version of thermodynamics does not exclude from consideration any (reversible or irreversible) component of real processes.
4. We propose an approach, which for brevity we term "energo dynamics", to prevent the occurrence of inequalities in the transition to non-static processes. This allows us to take into account the irreversibility of real processes of energy conversion not only in thermodynamics, but also in other fundamental disciplines.
5. Energodynamics allows us to give a strictly thermodynamic theory, free from the postulates and considerations of molecular-kinetic and statistical-
- mechanical theories, and one which validates all the provisions of a TIP, thus expanding it domain to nonlinear processes and states far from equilibrium.
6. The isolation of independent processes occurring in the system under study refutes Onsager 's postulate about the dependence of each of the fluxes on all forces acting in the system. It thus makes it possible to find, for each flux, a unique corresponding force whose disappearance results in the cessation of the process.
7. The proposed energy dynamic method for finding the superposition effects of heterogeneous processes allows further reduction in the number of empirical coefficients by $n(n + 1) / 2$ in TIP to $n$ and explains these superimposed effects not by fluxes, but rather by forces in full accordance with the principles of mechanics.
8. A suitable generalization of TIP to the processes of purposeful transformation of various forms of energy in natural and technical systems reveals their fundamental unity and difference from relaxation processes both in relation to their equations and their reciprocity relations.
9. The transition to the study of the kinetics of energy conversion processes allows us to propose a universal criterion for the efficiency of power and technological installations, taking into account, respectively, their power and performance, and combining the advantages of absolute, relative, exergy, etc. efficiency.
10. The unity of the laws of transformation of thermal and non-thermal forms of energy discovered within the framework of energo dynamics made it possible to propose a theory of the similarity of power and technological installations and to construct their universal load characteristics that facilitate the choice of nominal, peak, etc. modes of their operation.
Generating HTML Viewer...
References
18 Cites in Article
L Onsager (1931). Reciprocal relations in irreversible processes.
I Prigogine,J Wiame (1947). Biologie et thermodynamique des phénomènes irréversibles.
H Casimir (1945). On Onsager's Principle of Microscopic Reversibility.
K Denbig (1954). Thermodynamics of Irreversible Processes — Irreversible Thermodynamics.
I Meixner (1954). Thermodynamik der irreversiblen Prozesse.
S De Groot,P Mazur (1962). Nonequilibrium Thermodynamics.
I Gyarmati (1960). Introduction to Irreversible Thermodynamics.
Rolf Haase (1963). Thermodynamik der Irreversiblen Prozesse.
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.